Mosquito Vector Competence for Japanese Encephalitis Virus
Abstract
:1. Introduction
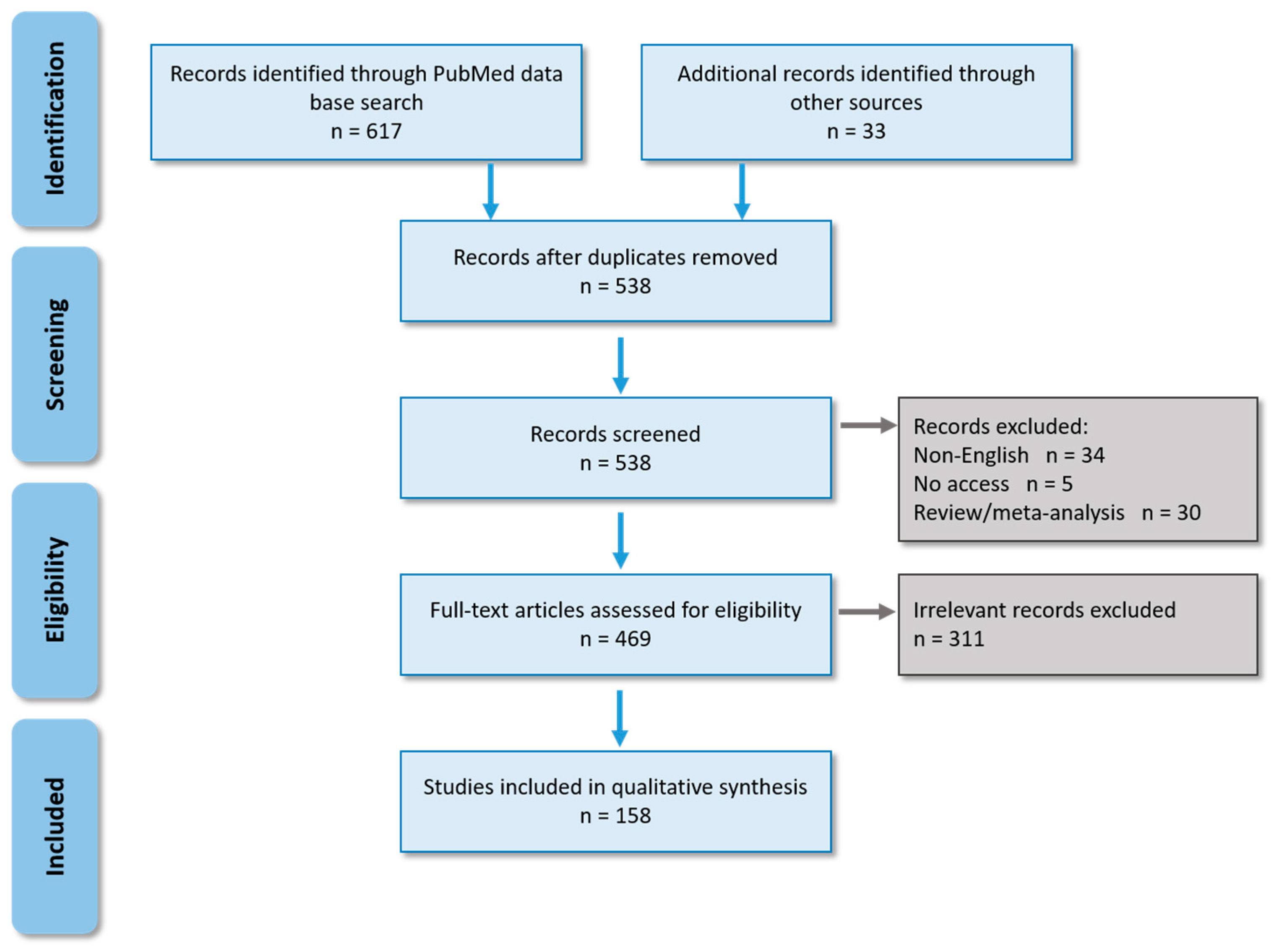
2. Materials and Methods
2.1. Literature Search
2.2. Mosquito Classification and Taxonomy
2.3. Classification in Confirmed and Potential Vector Species
3. Results
3.1. Confirmed Vectors
3.2. Potential Vectors
3.3. Mosquito Species with JEV Isolation in the Field
| Mosquito Species | Virus Detection in Field-Caught Mosquitoes | Virus Isolation from Field-Caught Mosquitoes |
|---|---|---|
| Aedes butleri | 1992 in Malaysia [29]; 1992–1993 in Malaysia [59] | |
| Aedes curtipes | 1968 in Malaysia [74] | |
| Aedes lineatopennis | 1992–1993 in Malaysia [59] | |
| Aedes vexans nocturnus | 2000–2004 in Taiwan [116] | |
| Anopheles ssp | 1969 in Malaysia [74] | |
| Anopheles annularis | 1978–1980 in Indonesia [57]; 1979 in Indonesia [133] | |
| Anopheles barbirostris | 1973 in India [158]; 2011–2013 in India [39] | |
| Anopheles hyrcanus | 1973 in India [158]; 1974–1975 in India [179] | |
| Anopheles pallidus | 2011–2013 in India [39] | |
| Anopheles peditaeniatus | 1985–1987 in India [58] | |
| Anopheles subpictus | 1996 in India [121]; 1997–1999 in India [180]; 2011–2013 in India [39] | 1977–1979 in India [102] |
| Anopheles sinensis | 2007 in China [40]; 2007–2009 in China [142]; 2009–2010 in China [41] | |
| Anopheles vagus | 1978–1980 in Indonesia [57]; 1979 in Indonesia [133] | |
| Coquillettidia ochracea | 2015 in China [89] | |
| Culex annulus | 1967 in Taiwan [177]; 1969 in Taiwan [131,178]; 1974–1976 in Taiwan [56]; 1995–1996 in Taiwan [28] | |
| Culex epidesmus | 1974–1975 in India [179] | |
| Culex fuscanus | 1995–1996 in Taiwan [28] | |
| Culex infula | 2011–2013 in India [39] | |
| Culex orientalis | 2012 in South Korea [84] | |
| Culex rubithoracis | 2002–2004 in Taiwan [116] | |
| Culex whitmorei | 1962–1966 in India [129]; 1987–1988 in Sri Lanka [52] | |
| Mansonia ssp | 1969 in Malaysia | |
| Mansonia bonneae/dives | 1969 in Malaysia [74] | |
| Mansonia annulifera | 1999–2000 in India [65]; 2011–2013 in India [39] | |
| Mansonia indiana | 1996 in India [121]; 1999–2000 in India [65] | |
| Mansonia uniformis | 1987–1988 in Sri Lanka [52]; 1996 in India [121]; 1999–2000 in India [65]; 2011–2013 in India [39] | 1969 in Malaysia [74] |
4. Discussion
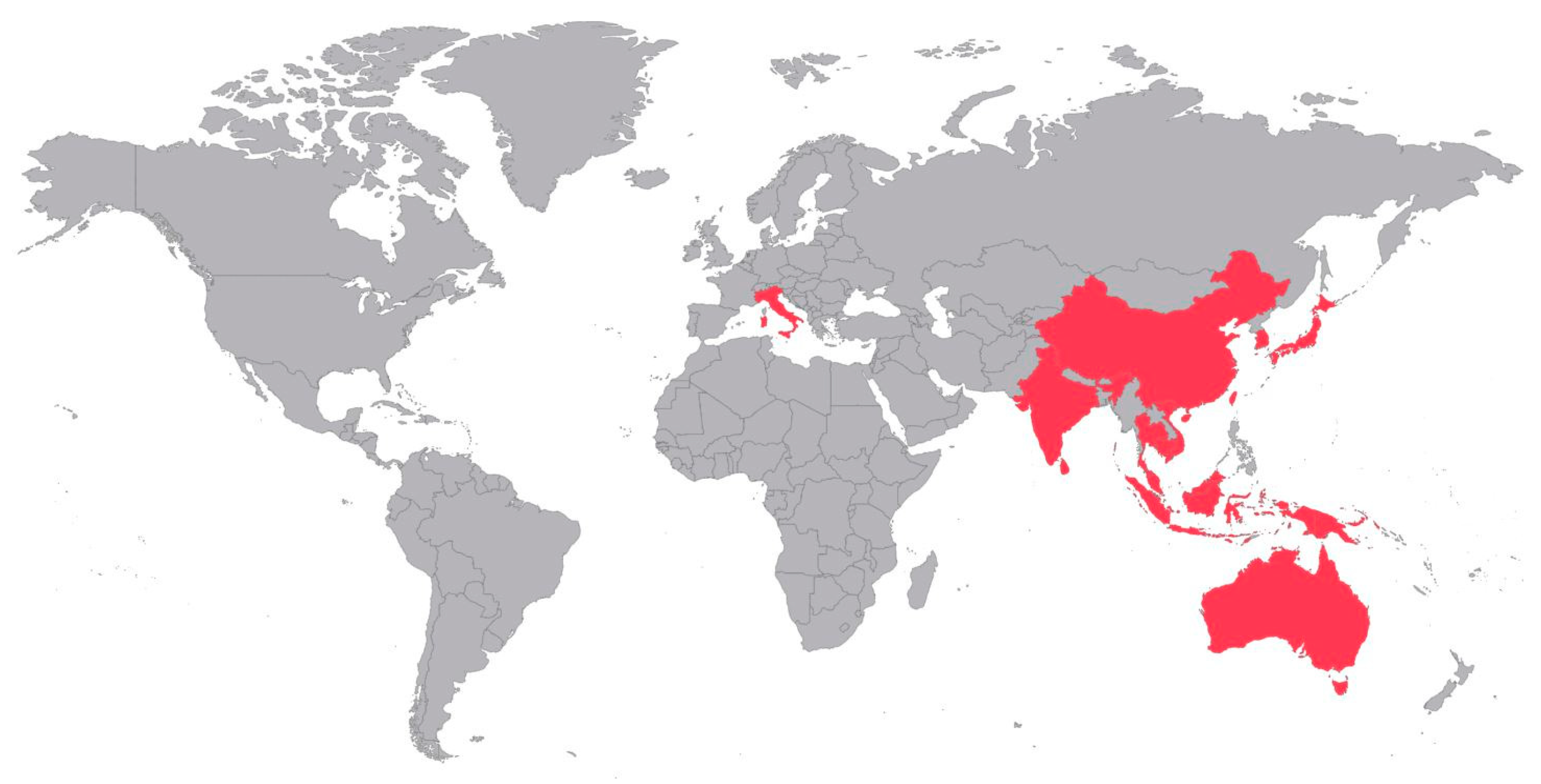
4.1. Diversity of Mosquito Vector Species and Consequence in Terms of Public Health
4.2. Diversity of Vector Competence Experiments: Problems and Solutions
4.2.1. Influence of Mosquito Origin and Rearing on Vector Competence
4.2.2. Influence of Virus Strain on Vector Competence
4.2.3. Influence of Applied Techniques on Vector Competence
4.2.4. Other Factors Influencing Vector Competence
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Lindahl, J.F. Japanese Encephalitis Virus in Pigs and Vectors in the Mekong Delta. With Special Reference to Urban Farming. Ph.D. Thesis, Swedish University of Agricultural Sciences, Upsalla, Sweden, 2012. [Google Scholar]
- Mackenzie, J.S. Emerging Zoonotic Encephalitis Viruses: Lessons from Southeast Asia and Oceania. J. Neurovirol. 2005, 11, 434–440. [Google Scholar] [CrossRef]
- Campbell, G.L.; Hills, S.L.; Fischer, M.; Jacobson, J.A.; Hoke, C.H.; Hombach, J.M.; Marfin, A.A.; Solomon, T.; Tsai, T.F.; Tsu, V.D.; et al. Estimated Global Incidence of Japanese Encephalitis: A Systematic Review. Bull. World Health Organ. 2011, 89, 766–774, 774A–774E. [Google Scholar] [CrossRef] [PubMed]
- Mathers, C.D.; Ezzati, M.; Lopez, A.D. Measuring the Burden of Neglected Tropical Diseases: The Global Burden of Disease Framework. PLoS Negl. Trop. Dis. 2007, 1, e114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Solomon, T.; Ni, H.; Beasley, D.W.C.; Ekkelenkamp, M.; Cardosa, M.J.; Barrett, A.D.T. Origin and Evolution of Japanese Encephalitis Virus in Southeast Asia. J. Virol. 2003, 77, 3091–3098. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ladreyt, H.; Durand, B.; Dussart, P.; Chevalier, V. How Central Is the Domestic Pig in the Epidemiological Cycle of Japanese Encephalitis Virus? A Review of Scientific Evidence and Implications for Disease Control. Viruses 2019, 11, 949. [Google Scholar] [CrossRef] [Green Version]
- Ricklin, M.E.; García-Nicolás, O.; Brechbühl, D.; Python, S.; Zumkehr, B.; Nougairede, A.; Charrel, R.N.; Posthaus, H.; Oevermann, A.; Summerfield, A. Vector-Free Transmission and Persistence of Japanese Encephalitis Virus in Pigs. Nat. Commun. 2016, 7, 10832. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diallo, A.O.I.; Chevalier, V.; Cappelle, J.; Duong, V.; Fontenille, D.; Duboz, R. How Much Does Direct Transmission between Pigs Contribute to Japanese Encephalitis Virus Circulation? A Modelling Approach in Cambodia. PLoS ONE 2018, 13, e0201209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliveira, A.R.S.; Strathe, E.; Etcheverry, L.; Cohnstaedt, L.W.; McVey, D.S.; Piaggio, J.; Cernicchiaro, N. Assessment of Data on Vector and Host Competence for Japanese Encephalitis Virus: A Systematic Review of the Literature. Prev. Vet. Med. 2018, 154, 71–89. [Google Scholar] [CrossRef]
- Oliveira, A.R.S.; Cohnstaedt, L.W.; Cernicchiaro, N. Japanese Encephalitis Virus: Placing Disease Vectors in the Epidemiologic Triad. Ann. Entomol. Soc. Am. 2018, 111, 295–303. [Google Scholar] [CrossRef]
- Pearce, J.C.; Learoyd, T.P.; Langendorf, B.J.; Logan, J.G. Japanese Encephalitis: The Vectors, Ecology and Potential for Expansion. J. Travel Med. 2018, 25, S16–S26. [Google Scholar] [CrossRef] [Green Version]
- Sudeep, A.B. Culex Gelidus: An Emerging Mosquito Vector with Potential to Transmit Multiple Virus Infections. J. Vector Borne Dis. 2014, 51, 251–258. [Google Scholar] [PubMed]
- Boussès, P.; Dehecq, J.S.; Brengues, C.; Fontenille, D. Inventaire actualisé des moustiques (Diptera: Culicidae) de l’île de La Réunion, océan Indien. Bull. Soc. Pathol. Exot. 2013, 106, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Alves, J.; de Pina, A.; Diallo, M.; Dia, I. First Report of Culex (Culex) Tritaeniorhynchus Giles, 1901 (Diptera: Culicidae) in the Cape Verde Islands. Zool. Caboverdiana 2014, 5, 14–19. [Google Scholar]
- Fall, A.; Diaïté, A.; Seck, M.; Bouyer, J.; Lefrançois, T.; Vachiéry, N.; Aprelon, R.; Faye, O.; Konaté, L.; Lancelot, R. West Nile Virus Transmission in Sentinel Chickens and Potential Mosquito Vectors, Senegal River Delta, 2008–2009. Int. J. Environ. Res. Public Health 2013, 10, 4718–4727. [Google Scholar] [CrossRef] [PubMed]
- Lytra, I.; Emmanouel, N. Study of Culex Tritaeniorhynchus and Species Composition of Mosquitoes in a Rice Field in Greece. Acta Trop. 2014, 134, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Sirivanakarn, S. Medical Entomology Studies—III. A Revision of the Subgenus Culex in the Oriental Region (Diptera: Culicidae). Cont. Am. Entomol. Inst. 1976, 12, 1–271. [Google Scholar]
- Barnett, H.C.; Gould, D.J. Colonization of Culex Gelidus Theobald and Some Resultant Effects on Its Biology. Trans. R Soc. Trop. Med. Hyg. 1962, 56, 423–428. [Google Scholar] [CrossRef]
- Gould, D.J.; Barnett, H.C.; Suyemoto, W. Transmission of Japanese Encephalitis Virus by Culex Gelidus Theobald. Trans. R Soc. Trop. Med. Hyg. 1962, 56, 429–435. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, A.R.S.; Cohnstaedt, L.W.; Strathe, E.; Hernández, L.E.; McVey, D.S.; Piaggio, J.; Cernicchiaro, N. Meta-Analyses of the Proportion of Japanese Encephalitis Virus Infection in Vectors and Vertebrate Hosts. Parasites Vectors 2017, 10, 418. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, A.R.S.; Cohnstaedt, L.W.; Strathe, E.; Etcheverry, L.; McVey, D.S.; Piaggio, J.; Cernicchiaro, N. Meta-Analyses of Japanese Encephalitis Virus Infection, Dissemination, and Transmission Rates in Vectors. Am. J. Trop. Med. Hyg. 2018, 98, 883–890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rückert, C.; Ebel, G.D. How Do Virus–Mosquito Interactions Lead to Viral Emergence? Trends Parasitol. 2018, 34, 310–321. [Google Scholar] [CrossRef]
- Takashima, I.; Rosen, L. Horizontal and Vertical Transmission of Japanese Encephalitis Virus by Aedes Japonicus (Diptera: Culicidae). J. Med. Entomol. 1989, 26, 454–458. [Google Scholar] [CrossRef] [PubMed]
- Rosen, L.; Tesh, R.; Lien, J.; Cross, J. Transovarial Transmission of Japanese Encephalitis Virus by Mosquitoes. Science 1978, 199, 909–911. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, T.P. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [Green Version]
- Harbach, R. Mosquito Taxonomic Inventory—Updated 22 January 2021. Available online: http://mosquito-taxonomic-inventory.info/sites/mosquito-taxonomic-inventory.info/files/Valid%20Species%20List_92.pdf (accessed on 28 January 2021).
- Wilkerson, R.C.; Linton, Y.-M.; Fonseca, D.M.; Schultz, T.R.; Price, D.C.; Strickman, D.A. Making Mosquito Taxonomy Useful: A Stable Classification of Tribe Aedini That Balances Utility with Current Knowledge of Evolutionary Relationships. PLoS ONE 2015, 10, e0133602. [Google Scholar] [CrossRef] [PubMed]
- Weng, M.H.; Lien, J.C.; Wang, Y.M.; Lin, C.C.; Lin, H.C.; Chin, C. Isolation of Japanese Encephalitis Virus from Mosquitoes Collected in Northern Taiwan between 1995 and 1996. J. Microbiol. Immunol. Infect. 1999, 32, 9–13. [Google Scholar]
- Vythilingam, I.; Oda, K.; Chew, T.K.; Mahadevan, S.; Vijayamalar, B.; Morita, K.; Tsuchie, H.; Igarashi, A. Isolation of Japanese Encephalitis Virus from Mosquitoes Collected in Sabak Bernam, Selangor, Malaysia in 1992. J. Am. Mosq. Control Assoc. 1995, 11, 94–98. [Google Scholar]
- Su, C.-L.; Yang, C.-F.; Teng, H.-J.; Lu, L.-C.; Lin, C.; Tsai, K.-H.; Chen, Y.-Y.; Chen, L.-Y.; Chang, S.-F.; Shu, P.-Y. Molecular Epidemiology of Japanese Encephalitis Virus in Mosquitoes in Taiwan during 2005–2012. PLoS Negl. Trop. Dis. 2014, 8, e3122. [Google Scholar] [CrossRef] [Green Version]
- Weng, M.H.; Lien, J.C.; Wang, Y.M.; Wu, H.L.; Chin, C. Susceptibility of Three Laboratory Strains of Aedes Albopictus (Diptera: Culicidae) to Japanese Encephalitis Virus from Taiwan. J. Med. Entomol. 1997, 34, 745–747. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.J.; Dong, C.F.; Chiou, L.Y.; Chuang, W.L. Potential Role of Armigeres Subalbatus (Diptera: Culicidae) in the Transmission of Japanese Encephalitis Virus in the Absence of Rice Culture on Liu-Chiu Islet, Taiwan. J. Med. Entomol. 2000, 37, 108–113. [Google Scholar] [CrossRef]
- Nicholson, J.; Ritchie, S.A.; van den Hurk, A.F. Aedes Albopictus (Diptera: Culicidae) as a Potential Vector of Endemic and Exotic Arboviruses in Australia. J. Med. Entomol. 2014, 51, 661–669. [Google Scholar] [CrossRef]
- de Wispelaere, M.; Desprès, P.; Choumet, V. European Aedes Albopictus and Culex Pipiens Are Competent Vectors for Japanese Encephalitis Virus. PLoS Negl. Trop. Dis. 2017, 11, e0005294. [Google Scholar] [CrossRef] [PubMed]
- Hodes, H.L. Experimental Transmission of Japanese B. Encephalitis by Mosquitoes and Mosquito Larvae. Bull. Johns Hopkins Hosp. 1946, 79, 358. [Google Scholar]
- Reeves, W.C.; Hammon, W.M. Laboratory Transmission of Japanese B Encephalitis Virus by Seven Species (Three Genera) of North American Mosquitoes. J. Exp. Med. 1946, 83, 185–194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanna, J.N.; Ritchie, S.A.; Phillips, D.A.; Lee, J.M.; Hills, S.L.; van den Hurk, A.F.; Pyke, A.T.; Johansen, C.A.; Mackenzie, J.S. Japanese Encephalitis in North Queensland, Australia, 1998. Med. J. Aust. 1999, 170, 533–536. [Google Scholar] [CrossRef]
- van den Hurk, A.F.; Nisbet, D.J.; Hall, R.A.; Kay, B.H.; Mackenzie, J.S.; Ritchie, S.A. Vector Competence of Australian Mosquitoes (Diptera: Culicidae) for Japanese Encephalitis Virus. J. Med. Entomol. 2003, 40, 82–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thenmozhi, V.; Balaji, T.; Selvam, A.; Venkatasubramani, K.; Dhananjeyan, K.J. A Longitudinal Study on Abundance and Infection Frequency of Japanese Encephalitis Vectors in Tirunelveli District, Tamil Nadu, India. Int. J. Mol. Res. 2015, 2, 166–169. [Google Scholar]
- Feng, Y.; Fu, S.; Zhang, H.; Li, M.; Zhou, T.; Wang, J.; Zhang, Y.; Wang, H.; Tang, Q.; Liang, G. Distribution of Mosquitoes and Mosquito-Borne Viruses along the China-Myanmar Border in Yunnan Province. Jpn. J. Infect. Dis. 2012, 65, 215–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, H.; Lu, H.-J.; Liu, Z.-J.; Jing, J.; Ren, J.-Q.; Liu, Y.-Y.; Lu, F.; Jin, N.-Y. Japanese Encephalitis Virus in Mosquitoes and Swine in Yunnan Province, China 2009-2010. Vector-Borne Zoonotic Dis. 2013, 13, 41–49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsai, K.-H.; Huang, C.-G.; Wu, W.-J.; Chuang, C.-K.; Lin, C.-C.; Chen, W.-J. Parallel Infection of Japanese Encephalitis Virus and Wolbachia within Cells of Mosquito Salivary Glands. J. Med. Entomol. 2006, 43, 752–756. [Google Scholar] [CrossRef]
- Ritchie, S.A.; Phillips, D.; Broom, A.; Mackenzie, J.; Poidinger, M.; van den Hurk, A. Isolation of Japanese Encephalitis Virus from Culex Annulirostris in Australia. Am. J. Trop. Med. Hyg. 1997, 56, 80–84. [Google Scholar] [CrossRef]
- van den Hurk, A.F.; Smith, C.S.; Field, H.E.; Smith, I.L.; Northill, J.A.; Taylor, C.T.; Jansen, C.C.; Smith, G.A.; Mackenzie, J.S. Transmission of Japanese Encephalitis Virus from the Black Flying Fox, Pteropus Alecto, to Culex Annulirostris Mosquitoes, despite the Absence of Detectable Viremia. Am. J. Trop. Med. Hyg. 2009, 81, 457–462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takhampunya, R.; Kim, H.-C.; Tippayachai, B.; Kengluecha, A.; Klein, T.A.; Lee, W.-J.; Grieco, J.; Evans, B.P. Emergence of Japanese Encephalitis Virus Genotype V in the Republic of Korea. Virol. J. 2011, 8, 449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.C.; Takhampunya, R.; Tippayachai, B.; Chong, S.-T.; Park, J.-Y.; Kim, M.-S.; Seo, H.-J.; Yeh, J.-Y.; Lee, W.-J.; Lee, D.-K.; et al. Japanese Encephalitis Virus in Culicine Mosquitoes (Diptera: Culicidae) of the Republic of Korea, 2008–2010. Mil. Med. 2015, 180, 158–167. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.C.; Klein, T.A.; Takhampunya, R.; Evans, B.P.; Mingmongkolchai, S.; Kengluecha, A.; Grieco, J.; Masuoka, P.; Kim, M.-S.; Chong, S.-T.; et al. Japanese Encephalitis Virus in Culicine Mosquitoes (Diptera: Culicidae) Collected at Daeseongdong, a Village in the Demilitarized Zone of the Republic of Korea. J. Med. Entomol. 2011, 48, 1250–1256. [Google Scholar] [CrossRef] [PubMed]
- Seo, H.-J.; Kim, H.C.; Klein, T.A.; Ramey, A.M.; Lee, J.-H.; Kyung, S.-G.; Park, J.-Y.; Cho, Y.S.; Cho, I.-S.; Yeh, J.-Y. Molecular Detection and Genotyping of Japanese Encephalitis Virus in Mosquitoes during a 2010 Outbreak in the Republic of Korea. PLoS ONE 2013, 8, e55165. [Google Scholar] [CrossRef]
- Dhanda, V.; Banerjee, K.; Deshmukh, P.K.; Ilkal, M.A. Experimental Viraemia and Transmission of Japanese Encephalitis Virus by Mosquitoes in Domestic Ducks. Indian J. Med. Res. 1977, 66, 881–888. [Google Scholar]
- Banerjee, K.; Deshmukh, P.K.; Ilkal, M.A.; Dhanda, V. Transmission of Japanese Encephalitis Virus by Culex Bitaeniorhynchus Giles. Indian J. Med. Res. 1978, 67, 889–893. [Google Scholar] [PubMed]
- Banerjee, K.; Deshmukh, P.K.; Ilkal, M.A.; Dhanda, V. Comparative Susceptibility of Three Species of Mosquitoes to Infection with Japanese Encephalitis Virus. Indian J. Med. Res. 1983, 78, 603–606. [Google Scholar] [PubMed]
- Peiris, J.S.M.; Amerasinghe, F.P.; Amerasinghe, P.H.; Ratnayake, C.B.; Karunaratne, S.H.P.P.; Tsai, T.F. Japanese Encephalitis in Sri Lanka—The Study of an Epidemic: Vector Incrimination, Porcine Infection and Human Disease. Trans. R Soc. Trop. Med. Hyg. 1992, 86, 307–313. [Google Scholar] [CrossRef]
- Gajanana, A.; Rajendran, R.; Samuel, P.P.; Thenmozhi, V.; Tsai, T.F.; Kimura-Kuroda, J.; Reuben, R. Japanese Encephalitis in South Arcot District, Tamil Nadu, India: A Three-Year Longitudinal Study of Vector Abundance and Infection Frequency. J. Med. Entomol. 1997, 34, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-C.; Wang, C.-Y.; Teng, H.-J.; Chen, C.-F.; Chang, M.-C.; Lu, L.-C.; Lin, C.; Jian, S.-W.; Wu, H.-S. Comparison of the Efficacy of CO2-Baited and Unbaited Light Traps, Gravid Traps, Backpack Aspirators, and Sweep Net Collections for Sampling Mosquitoes Infected with Japanese Encephalitis Virus. J. Vector Ecol. 2011, 36, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Gould, D.J.; Edelman, R.; Grossman, R.A.; Nisalak, A.; Sullivan, M.F. Study of Japanese Encephalitis Virus in Chiangmai Valley, Thailand. IV. Vector Studies. Am. J. Epidemiol. 1974, 100, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.H.; Huang, W.C.; Cross, J.H. The Isolation of Japanese Encephalitis Virus from Taiwan Mosquitoes by Mosquito Cell Cultures and Mouse Inoculation. J. Med. Entomol. 1978, 14, 698–701. [Google Scholar] [CrossRef] [PubMed]
- Olson, J.G.; Ksiazek, T.G.; Tan, R.; Atmosoedjono, S.; Lee, V.H.; Converse, J.D. Correlation of Population Indices of Female Culex Tritaeniorhynchus with Japanese Encephalitis Viral Activity in Kapuk, Indonesia. Southeast Asian J. Trop. Med. Public Health 1985, 16, 337–342. [Google Scholar]
- Mourya, D.T.; Ilkal, M.A.; Mishra, A.C.; Jacob, P.G.; Pant, U.; Ramanujam, S.; Mavale, M.S.; Bhat, H.R.; Dhanda, V. Isolation of Japanese Encephalitis Virus from Mosquitoes Collected in Karnataka State, India from 1985 to 1987. Trans. R Soc. Trop. Med. Hyg. 1989, 83, 550–552. [Google Scholar] [CrossRef]
- Vythilingam, I.; Oda, K.; Mahadevan, S.; Abdullah, G.; Thim, C.S.; Hong, C.C.; Vijayamalar, B.; Sinniah, M.; Igarashi, A. Abundance, Parity, and Japanese Encephalitis Virus Infection of Mosquitoes (Diptera:Culicidae) in Sepang District, Malaysia. J. Med. Entomol. 1997, 34, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Muangman, D.; Edelman, R.; Sullivan, M.J.; Gould, D.J. Experimental Transmission of Japanese Encephalitis Virus by Culex Fuscocephala. Am. J. Trop. Med. Hyg. 1972, 21, 482–486. [Google Scholar] [CrossRef] [PubMed]
- Okuno, T.; Mitchell, C.J.; Chen, P.S.; Hsu, S.; Ryu, E. Experimental Transmission of Japanese Encephalitis Virus by Culex Tritaeniorhynchus and C. Fuscocephalus. Ann. Trop. Med. Parasitol. 1975, 69, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Tewari, S.C.; Thenmozhi, V.; Rajendran, R.; Appavoo, N.C.; Gajanana, A. Detection of Japanese Encephalitis Virus Antigen in Desiccated Mosquitoes: An Improved Surveillance System. Trans. R Soc. Trop. Med. Hyg. 1999, 93, 525–526. [Google Scholar] [CrossRef]
- Tewari, S.C.; Thenmozhi, V.; Arunachalam, N.; Philip Samuel, P.; Tyagi, B.K. Desiccated Vector Mosquitoes Used for the Surveillance of Japanese Encephalitis Virus Activity in Endemic Southern India. Trop. Med. Int. Health 2008, 13, 286–290. [Google Scholar] [CrossRef]
- Rajendran, R.; Thenmozhi, V.; Tewari, S.C.; Balasubramanian, A.; Ayanar, K.; Manavalan, R.; Gajanana, A.; Kabilan, L.; Thakare, J.P.; Satyanarayana, K. Longitudinal Studies in South Indian Villages on Japanese Encephalitis Virus Infection in Mosquitoes and Seroconversion in Goats. Trop. Med. Int. Health 2003, 8, 174–181. [Google Scholar] [CrossRef]
- Arunachalam, N.; Philip Samuel, P.; Hiriyan, J.; Thenmozhi, V.; Gajanana, A. Japanese Encephalitis in Kerala, South India: Can Mansonia (Diptera: Culicidae) Play a Supplemental Role in Transmission? J. Med. Entomol. 2004, 41, 456–461. [Google Scholar] [CrossRef]
- Upadhyayula, S.M.; Rao Mutheneni, S.; Nayanoori, H.K.; Natarajan, A.; Goswami, P. Impact of Weather Variables on Mosquitoes Infected with Japanese Encephalitis Virus in Kurnool District, Andhra Pradesh. Asian Pac. J. Trop. Med. 2012, 5, 337–341. [Google Scholar] [CrossRef] [Green Version]
- Ritchie, S.A.; van den Hurk, A.F.; Zborowski, P.; Kerlin, T.J.; Banks, D.; Walker, J.A.; Lee, J.M.; Montgomery, B.L.; Smith, G.A.; Pyke, A.T.; et al. Operational Trials of Remote Mosquito Trap Systems for Japanese Encephalitis Virus Surveillance in the Torres Strait, Australia. Vector-Borne Zoonotic Dis. 2007, 7, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, D.; Muniaraj, M.; Samuel, P.P.; Thenmozhi, V.; Venkatesh, A.; Nagaraj, J.; Tyagi, B.K. Seasonal Abundance & Role of Predominant Japanese Encephalitis Vectors Culex Tritaeniorhynchus & Cx. Gelidus Theobald in Cuddalore District, Tamil Nadu. Indian J. Med. Res. 2015, 142, S23–S29. [Google Scholar] [CrossRef] [PubMed]
- Thenmozhi, V.; Paramasivan, R.; Philip Samuel, P.; Kamaraj, T.; Balaji, T.; Dhananjeyan, K.J.; Venkatasubramani, K.; Leo, S.V.J.; Babu, R.S.; Govindarajan, R.; et al. Japanese Encephalitis Virus Isolation from Mosquitoes during an Outbreak in 2011 in Alappuzha District, Kerala. J. Vector Borne Dis. 2013, 50, 229–231. [Google Scholar]
- Pantawane, P.B.; Dhanze, H.; Ravi Kumar, G.V.P.P.S.; Grace, M.R.; Dudhe, N.C.; Bhilegaonkar, K.N. TaqMan Real-Time RT-PCR Assay for Detecting Japanese Encephalitis Virus in Swine Blood Samples and Mosquitoes. Anim. Biotechnol. 2019, 30, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Simasathien, P.; Rohitayodhin, S.; Nisalak, A.; Singharaj, P.; Halstead, S.B.; Russell, P.K. Recovery of Japanese Encephalitis Virus from Wild Caught Mosquitoes in Thailand. Southeast Asian J. Trop Med. Public Health 1972, 3, 52–54. [Google Scholar] [PubMed]
- Simpson, D.I.; Bowen, E.T.; Platt, G.S.; Way, H.; Smith, C.E.; Peto, S.; Kamath, S.; Lim, B.L.; Lim, T.W. Japanese Encephalitis in Sarawak: Virus Isolation and Serology in a Land Dyak Village. Trans. R Soc. Trop. Med. Hyg. 1970, 64, 503–510. [Google Scholar] [CrossRef]
- Heathcote, O.H.U. Japanese Encephalitis in Sarawak: Studies on Juvenile Mosquito Populations. Trans. R Soc. Trop. Med. Hyg. 1970, 64, 483–488. [Google Scholar] [CrossRef]
- Simpson, D.I.; Bowen, E.T.; Way, H.J.; Platt, G.S.; Hill, M.N.; Kamath, S.; Lim, T.W.; Bendell, P.J.; Heathcote, O.H. Arbovirus Infections in Sarawak, October 1968--February 1970: Japanese Encephalitis Virus Isolations from Mosquitoes. Ann. Trop. Med. Parasitol. 1974, 68, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Thoa, N.T.K.; Vien, N.T.; Mai, T.T.; Xuan, N.T.N. Japanese Encephalitis Vectors: Isolation of Virus from Culicine Mosquitoes in the Saigon Area. Southeast Asian J. Trop. Med. Public Health 1974, 5, 408–412. [Google Scholar]
- Van Peenen, P.F.D.; Joseph, P.L.; Atmosoedjono, S.; Irsiana, R.; Saroso, J.S. Japanese Encephalitis Virus from Pigs and Mosquitoes in Jakarta, Indonesia. Trans. R Soc. Trop. Med. Hyg. 1975, 69, 477–479. [Google Scholar] [CrossRef]
- Gingrich, J.B.; Nisalak, A.; Latendresse, J.R.; Sattabongkot, J.; Hoke, C.H.; Pomsdhit, J.; Chantalakana, C.; Satayaphanta, C.; Uechiewcharnkit, K.; Innis, B.L. Japanese Encephalitis Virus in Bangkok: Factors Influencing Vector Infections in Three Suburban Communities. J. Med. Entomol. 1992, 29, 436–444. [Google Scholar] [CrossRef]
- Simpson, D.I.; Smith, C.E.G.; Marshall, T.F.; Platt, G.S.; Way, H.J.; Bowen, E.T.W.; Bright, W.F.; Day, J.; McMahon, D.A.; Hill, M.N.; et al. Arbovirus Infections in Sarawak: The Role of the Domestic Pig. Trans. R Soc. Trop. Med. Hyg. 1976, 70, 66–72. [Google Scholar] [CrossRef]
- Pyke, A.T.; Williams, D.T.; Nisbet, D.J.; van den Hurk, A.F.; Taylor, C.T.; Johansen, C.A.; Macdonald, J.; Hall, R.A.; Simmons, R.J.; Mason, R.J.; et al. The Appearance of a Second Genotype of Japanese Encephalitis Virus in the Australasian Region. Am. J. Trop. Med. Hyg. 2001, 65, 747–753. [Google Scholar] [CrossRef] [PubMed]
- van den Hurk, A.F.; Nisbet, D.J.; Johansen, C.A.; Foley, P.N.; Ritchie, S.A.; Mackenzie, J.S. Japanese Encephalitis on Badu Island, Australia: The First Isojation of Japanese Encephalitis Virus from Culex Gelidus in the Australasian Region and the Role of Mosquito Host-Feeding Patterns in Virus Transmission Cycles. Trans. R Soc. Trop. Med. Hyg. 2001, 95, 595–600. [Google Scholar] [CrossRef]
- Arunachalam, N.; Murty, U.S.N.; Narahari, D.; Balasubramanian, A.; Philip Samuel, P.; Thenmozhi, V.; Paramasivan, R.; Rajendran, R.; Tyagi, B.K. Longitudinal Studies of Japanese Encephalitis Virus Infection in Vector Mosquitoes in Kurnool District, Andhra Pradesh, South India. J. Med. Entomol. 2009, 46, 633–639. [Google Scholar] [CrossRef]
- Sudeep, A.B.; Ghodke, Y.S.; George, R.P.; Ingale, V.S.; Dhaigude, S.D.; Gokhale, M.D. Vectorial Capacity of Culex Gelidus (Theobald) Mosquitoes to Certain Viruses of Public Health Importance in India. J. Vector Borne Dis. 2015, 52, 153–158. [Google Scholar] [PubMed]
- Ravanini, P.; Huhtamo, E.; Ilaria, V.; Crobu, M.G.; Nicosia, A.M.; Servino, L.; Rivasi, F.; Allegrini, S.; Miglio, U.; Magri, A.; et al. Japanese Encephalitis Virus RNA Detected in Culex Pipiens Mosquitoes in Italy. Eurosurveillance 2012, 17. [Google Scholar] [CrossRef]
- Kim, H.; Cha, G.-W.; Jeong, Y.E.; Lee, W.-G.; Chang, K.S.; Roh, J.Y.; Yang, S.C.; Park, M.Y.; Park, C.; Shin, E.-H. Detection of Japanese Encephalitis Virus Genotype V in Culex Orientalis and Culex Pipiens (Diptera: Culicidae) in Korea. PLoS ONE 2015, 10, e0116547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fang, Y.; Zhang, Y.; Zhou, Z.-B.; Xia, S.; Shi, W.-Q.; Xue, J.-B.; Li, Y.-Y.; Wu, J.-T. New Strains of Japanese Encephalitis Virus Circulating in Shanghai, China after a Ten-Year Hiatus in Local Mosquito Surveillance. Parasites Vectors 2019, 12, 22. [Google Scholar] [CrossRef] [PubMed]
- Buescher, E.L.; Scherer, W.F.; Rosenberg, M.Z.; Gresser, I.; Hardy, J.L.; Bullock, H.R. Ecologic Studies of Japanese Encephalitis Virus in Japan. II. Mosquito Infection. Am. J. Trop. Med. Hyg. 1959, 8, 651664. [Google Scholar] [CrossRef]
- Lee, H.W.; Min, B.W.; Lee, Y.W. Japanese Encephalitis Virus Isolation from Mosquitoes of Korea. J. Korean Med. Assoc. 1969, 12, 429–440. [Google Scholar]
- Huang, C.H. Studies of Japanese Encephalitis in China. In Advances in Virus Research; Elsevier: Amsterdam, The Netherlands, 1982; Volume 27, pp. 71–101. ISBN 978-0-12-039827-0. [Google Scholar]
- Shi, Q.; Song, X.; Lv, Y.; Huang, X.; Kou, J.; Wang, H.W.; Zhang, H.; Cheng, P.; Gong, M. Potential Risks Associated with Japanese Encephalitis Prevalence in Shandong Province, China. Vector-Borne Zoonotic Dis. 2019, 19, 640–645. [Google Scholar] [CrossRef] [PubMed]
- Hammon, W.M.; Rees, D.M.; Casals, J.; Meiklejohn, G. Experimental Transmission of Japanese B Encephalitis Virus by Culex Tritaeniorhynchus and Culex Pipiens Var. Pallens, Suspected Natural Vectors. Am. J. Hyg. 1949, 50, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Doi, R.; Oya, A.; Shirasaka, A.; Yabe, S.; Sasa, M. Studies on Japanese Encephalitis Virus Infection of Reptiles. II. Role of Lizards on Hibernation of Japanese Encephalitis Virus. Jpn. J. Exp. Med. 1983, 53, 125–134. [Google Scholar]
- Weng, M.H.; Lien, J.C.; Lin, C.C.; Yao, C.W. Vector Competence of Culex Pipiens Molestus (Diptera: Culicidae) from Taiwan for a Sympatric Strain of Japanese Encephalitis Virus. J. Med. Entomol. 2000, 37, 780–783. [Google Scholar] [CrossRef]
- Turell, M.J.; Mores, C.N.; Dohm, D.J.; Komilov, N.; Paragas, J.; Lee, J.S.; Shermuhemedova, D.; Endy, T.P.; Kodirov, A.; Khodjaev, S. Laboratory Transmission of Japanese Encephalitis and West Nile Viruses by Molestus Form of Culex Pipiens (Diptera: Culicidae) Collected in Uzbekistan in 2004. J. Med. Entomol. 2006, 43, 296–300. [Google Scholar] [CrossRef]
- Hameed, M.; Liu, K.; Anwar, M.N.; Wahaab, A.; Safdar, A.; Di, D.; Boruah, P.; Xu, J.; Wang, X.; Li, B.; et al. The Emerged Genotype I of Japanese Encephalitis Virus Shows an Infectivity Similar to Genotype III in Culex Pipiens Mosquitoes from China. PLoS Negl. Trop. Dis. 2019, 13, e0007716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chapman, G.E.; Sherlock, K.; Hesson, J.C.; Blagrove, M.S.C.; Lycett, G.J.; Archer, D.; Solomon, T.; Baylis, M. Laboratory Transmission Potential of British Mosquitoes for Equine Arboviruses. Parasites Vectors 2020, 13, 413. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Xiao, C.; Xi, S.; Hameed, M.; Wahaab, A.; Shao, D.; Li, Z.; Li, B.; Wei, J.; Qiu, Y.; et al. Mosquito Defensins Enhance Japanese Encephalitis Virus Infection by Facilitating Virus Adsorption and Entry within the Mosquito. J. Virol. 2020, 94. [Google Scholar] [CrossRef] [PubMed]
- Di, D.; Li, C.; Zhang, J.; Hameed, M.; Wang, X.; Xia, Q.; Li, H.; Xi, S.; Li, Z.; Liu, K.; et al. Experimental Infection of Newly Hatched Domestic Ducklings via Japanese Encephalitis Virus-Infected Mosquitoes. Pathogens 2020, 9, 371. [Google Scholar] [CrossRef]
- Folly, A.J.; Dorey-Robinson, D.; Hernández-Triana, L.M.; Ackroyd, S.; Vidana, B.; Lean, F.Z.X.; Hicks, D.; Nuñez, A.; Johnson, N. Temperate Conditions Restrict Japanese Encephalitis Virus Infection to the Mid-Gut and Prevents Systemic Dissemination in Culex Pipiens Mosquitoes. Sci. Rep. 2021, 11, 6133. [Google Scholar] [CrossRef]
- Dhanda, V.; Mourya, D.T.; Mishra, A.C.; Ilkal, M.A.; Pant, U.; Jacob, P.G.; Bhat, H.R. Japanese Encephalitis Virus Infection in Mosquitoes Reared from Field-Collected Immatures and in Wild-Caught Males. Am. J. Trop. Med. Hyg. 1989, 41, 732–736. [Google Scholar] [CrossRef]
- Borah, J.; Dutta, P.; Khan, S.A.; Mahanta, J. Epidemiological Concordance of Japanese Encephalitis Virus Infection among Mosquito Vectors, Amplifying Hosts and Humans in India. Epidemiol. Infect. 2013, 141, 74–80. [Google Scholar] [CrossRef]
- Nyari, N.; Singh, D.; Kakkar, K.; Sharma, S.; Pandey, S.N.; Dhole, T.N. Entomological and Serological Investigation of Japanese Encephalitis in Endemic Area of Eastern Uttar Pradesh, India. J. Vector Borne Dis. 2015, 52, 321–328. [Google Scholar]
- George, S.; Jacob, P.G.; Rao, J.A. Isolation of Japanese Encephalitis & West Nile Viruses from Mosquitoes Collected in Kolar District of Karnataka State during 1977-79. Indian J. Med. Res. 1987, 85, 235–238. [Google Scholar]
- Naik, P.S.; Ilkal, M.A.; Pant, U.; Kulkarni, S.M.; Dhanda, V. Isolation of Japanese Encephalitis Virus from Culex Pseudovishnui Colless, 1957 (Diptera: Culicidae) in Goa. Indian J. Med. Res. 1990, 91, 331–333. [Google Scholar]
- Mourya, D.T.; Mishra, A.C.; Soman, R.S. Transmission of Japanese Encephalitis Virus in Culex Pseudovishnui & C. Tritaeniorhynchus Mosquitoes. Indian J. Med. Res. 1991, 93, 250–252. [Google Scholar] [PubMed]
- Mourya, D.T.; Mishra, A.C. Antigen Distribution Pattern of Japanese Encephalitis Virus in Culex Tritaeniorhynchus, C. Vishnui & C. Pseudovishnui. Indian J. Med. Res. 2000, 111, 157–161. [Google Scholar] [PubMed]
- Lindahl, J.F.; Ståhl, K.; Chirico, J.; Boqvist, S.; Thu, H.T.V.; Magnusson, U. Circulation of Japanese Encephalitis Virus in Pigs and Mosquito Vectors within Can Tho City, Vietnam. PLoS Negl. Trop. Dis. 2013, 7, e2153. [Google Scholar] [CrossRef] [PubMed]
- Nitatpattana, N.; Apiwathnasorn, C.; Barbazan, P.; Leemingsawat, S.; Yoksan, S.; Gonzalez, J.-P. First Isolation of Japanese Encephalitis from Culex Quinquefasciatus in Thailand. Southeast Asian J. Trop. Med. Public Health 2005, 36, 875–878. [Google Scholar]
- Hurlbut, H.S.; Thomas, J.I. Potential Vectors of Japanese Encephalitis in the Caroline Islands. Am. J. Trop. Med. Hyg. 1949, s1-29, 215–217. [Google Scholar] [CrossRef] [PubMed]
- Hurlbut, H.S.; Thomas, J.I. Observations on the Experimental Transmission of Japanese Encephalitis by Mosquitoes. Am. J. Trop. Med. Hyg. 1950, s1-30, 683–688. [Google Scholar] [CrossRef]
- Banerjee, K.; Deshmukh, P.K.; Ilkal, M.A.; Dhanda, V. Experimental Transmission of Japanese Encephalitis Virus through Anopheles Tessellatus and Culex Fatigans Mosquitoes. Indian J. Med. Res. 1977, 65, 746–752. [Google Scholar]
- Mackenzie-Impoinvil, L.; Impoinvil, D.E.; Galbraith, S.E.; Dillon, R.J.; Ranson, H.; Johnson, N.; Fooks, A.R.; Solomon, T.; Baylis, M. Evaluation of a Temperate Climate Mosquito, Ochlerotatus Detritus (=Aedes Detritus), as a Potential Vector of Japanese Encephalitis Virus. Med. Vet. Entomol. 2015, 29, 1–9. [Google Scholar] [CrossRef]
- Huang, Y.-J.S.; Harbin, J.N.; Hettenbach, S.M.; Maki, E.; Cohnstaedt, L.W.; Barrett, A.D.T.; Higgs, S.; Vanlandingham, D.L. Susceptibility of a North American Culex Quinquefasciatus to Japanese Encephalitis Virus. Vector-Borne Zoonotic Dis. 2015, 15, 709–711. [Google Scholar] [CrossRef]
- Huang, Y.-J.S.; Hettenbach, S.M.; Park, S.L.; Higgs, S.; Barrett, A.D.T.; Hsu, W.-W.; Harbin, J.N.; Cohnstaedt, L.W.; Vanlandingham, D.L. Differential Infectivities among Different Japanese Encephalitis Virus Genotypes in Culex Quinquefasciatus Mosquitoes. PLoS Negl. Trop. Dis. 2016, 10, e0005038. [Google Scholar] [CrossRef] [Green Version]
- Karna, A.K.; Bowen, R.A. Experimental Evaluation of the Role of Ecologically-Relevant Hosts and Vectors in Japanese Encephalitis Virus Genotype Displacement. Viruses 2019, 11, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vythilingam, I.; Oda, K.; Tsuchie, H.; Mahadevan, S.; Vijayamalar, B. Isolation of Japanese Encephalitis Virus from Culex Sitiens Mosquitoes in Selangor, Malaysia. J. Am. Mosq. Control Assoc. 1994, 10, 228–229. [Google Scholar]
- Weng, M.H.; Lien, J.C.; Ji, D.D. Monitoring of Japanese Encephalitis Virus Infection in Mosquitoes (Diptera: Culicidae) at Guandu Nature Park, Taipei, 2002–2004. J. Med. Entomol. 2005, 42, 1085–1088. [Google Scholar] [CrossRef]
- Van Den Hurk, A.F.; Montgomery, B.L.; Northill, J.A.; Smith, I.L.; Zborowski, P.; Ritchie, S.A.; Mackenzie, J.S.; Smith, G.A. Short Report: The First Isolation of Japanese Encephalitis Virus from Mosquitoes Collected from Mainland Australia. Am. J. Trop. Med. Hyg. 2006, 75, 21–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johansen, C.A.; van den Hurk, A.F.; Ritchie, S.A.; Zborowski, P.; Nisbet, D.J.; Paru, R.; Bockarie, M.J.; Macdonald, J.; Drew, A.C.; Khromykh, T.I.; et al. Isolation of Japanese Encephalitis Virus from Mosquitoes (Diptera: Culicidae) Collected in the Western Province of Papua New Guinea, 1997–1998. Am. J. Trop. Med. Hyg. 2000, 62, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Johansen, C.A.; van den Hurk, A.F.; Pyke, A.T.; Zborowski, P.; Phillips, D.A.; Mackenzie, J.S.; Ritchie, S.A. Entomological Investigations of an Outbreak of Japanese Encephalitis Virus in the Torres Strait, Australia, in 1998. J. Med. Entomol. 2001, 38, 581–588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johansen, C.A.; Nisbet, D.J.; Foley, P.N.; Van Den Hurk, A.F.; Hall, R.A.; Mackenzie, J.S.; Ritchie, S.A. Flavivirus Isolations from Mosquitoes Collected from Saibai Island in the Torres Strait, Australia, during an Incursion of Japanese Encephalitis Virus. Med. Vet. Entomol. 2004, 18, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Dhanda, V.; Thenmozhi, V.; Kumar, N.P.; Hiriyan, J.; Arunachalam, N.; Balasubramanian, A.; Ilango, A.; Gajanana, A. Virus Isolation from Wild-Caught Mosquitoes during a Japanese Encephalitis Outbreak in Kerala in 1996. Indian J. Med. Res. 1997, 106, 4–6. [Google Scholar]
- Das, B.P.; Sharma, S.N.; Kabilan, L.; Lal, S.; Saxena, V.K. First Time Detection of Japanese Encephalitis Virus Antigen in Dry and Unpreserved Mosquito Culex Tritaeniorhynchus Giles, 1901, from Karnal District of Haryana State of India. J. Commun. Dis. 2005, 37, 131–133. [Google Scholar] [PubMed]
- Jeong, Y.E.; Jeon, M.J.; Cho, J.E.; Han, M.G.; Choi, H.J.; Shin, M.Y.; Park, H.J.; Kim, W.; Moon, B.C.; Park, J.-S.; et al. Development and Field Evaluation of a Nested RT-PCR Kit for Detecting Japanese Encephalitis Virus in Mosquitoes. J. Virol. Methods 2011, 171, 248–252. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-Y.; Fan, Y.-C.; Tu, W.-C.; Chang, R.-Y.; Shih, C.-C.; Lu, I.-H.; Chien, M.-S.; Lee, W.-C.; Chen, T.-H.; Chang, G.-J.; et al. Japanese Encephalitis Virus Genotype Replacement, Taiwan, 2009–2010. Emerg. Infect. Dis. 2011, 17, 2354–2356. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Hori, K.; Kitagawa, Y.; Oikawa, Y.; Kamimura, K.; Takegami, T. An Ecological Survey of Mosquitoes and the Distribution of Japanese Encephalitis Virus in Ishikawa Prefecture, Japan, between 2010 and 2014. Jpn. J. Infect. Dis. 2017, 70, 362–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Philip Samuel, P.; Thenmozhi, V.; Muniaraj, M.; Ramesh, D.; Victor Jerald Leo, S.; Balaji, T.; Venkatasubramani, K.; Nagaraj, J.; Paramasivan, R. Changing Paradigm in the Epidemiology of Japanese Encephalitis in a Non-Endemic Region. J. Vector Borne Dis. 2018, 55, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Tao, Z.; Liu, G.; Wang, M.; Wang, H.; Lin, X.; Song, L.; Wang, S.; Wang, H.; Liu, X.; Cui, N.; et al. Molecular Epidemiology of Japanese Encephalitis Virus in Mosquitoes during an Outbreak in China, 2013. Sci. Rep. 2014, 4, 4908. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fang, Y.; Zhang, W.; Xue, J.-B.; Zhang, Y. Monitoring Mosquito-Borne Arbovirus in Various Insect Regions in China in 2018. Front. Cell Infect. Microbiol. 2021, 11, 640993. [Google Scholar] [CrossRef] [PubMed]
- Carey, D.E.; Reuben, R.; Myers, R.M. Japanese Encephalitis Studies in Vellore, South India. I. Virus Isolation from Mosquitoes. Indian J. Med. Res. 1968, 56, 1309–1318. [Google Scholar]
- Yamamoto, H.; Ishida, N.; Akiyama, K.; Shiratori, T.; Konno, J. Epidemiological Analyses of Japanese Encephalitis Virus Spread from Mosquitoes to Pigs through 5 Years. Jpn. J. Med. Sci. Biol. 1970, 23, 75–85. [Google Scholar] [CrossRef] [Green Version]
- Okuno, T.; Tseng, P.T.; Liu, S.Y.; Hsu, S.T.; Huang, C.T. Rates of Infection with Japanese Encephalitis Virus of Two Culicine Species of Mosquito in Taiwan. Bull. World Health Organ. 1971, 44, 599–604. [Google Scholar] [PubMed]
- Van Peenen, P.F.D.; Irsiana, R.; Saroso, J.S.; Joseph, S.W.; Shope, R.E.; Joseph, P.L. First Isolation of Japanese Encephalitis Virus from Java. Mil. Med. 1974, 139, 821–823. [Google Scholar] [CrossRef]
- Olson, J.G.; Ksiazek, T.G.; Lee, V.H.; Tan, R.; Shope, R.E. Isolation of Japanese Encephalitis Virus from Anopheles Annularis and Anopheles Vagus in Lombok, Indonesia. Trans. R. Soc. Trop. Med. Hyg. 1985, 79, 845–847. [Google Scholar] [CrossRef] [Green Version]
- Rosen, L.; Lien, J.C.; Lu, L.C. A Longitudinal Study of the Prevalence of Japanese Encephalitis Virus in Adult and Larval Culex Tritaeniorhynchus Mosquitoes in Northern Taiwan. Am. J. Trop. Med. Hyg. 1989, 40, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Leake, C.J.; Ussery, M.A.; Nisalak, A.; Hoke, C.H.; Andre, R.G.; Burke, D.S. Virus Isolations from Mosquitoes Collected during the 1982 Japanese Encephalitis Epidemic in Northern Thailand. Trans. R. Soc. Trop. Med. Hyg. 1986, 80, 831–837. [Google Scholar] [CrossRef]
- Tadano, M.; Kanemura, K.; Hasegawa, H.; Makino, Y.; Fukunaga, T. Epidemiological and Ecological Studies of Japanese Encephalitis in Okinawa, Subtropical Area in Japan. I. Investigations on Antibody Levels to Japanese Encephalitis Virus in Swine Sera and Vector Mosquito in Okinawa, Miyako and Ishigaki Islands. Microbiol. Immunol. 1994, 38, 117–122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pant, U.; Ilkal, M.A.; Soman, R.S.; Shetty, P.S.; Kanojia, P.C.; Kaul, H.N. First Isolation of Japanese Encephalitis Virus from the Mosquito, Culex Tritaeniorhynchus Giles, 1901 (Diptera: Culicidae) in Gorakhpur District, Uttar Pradesh. Indian J. Med. Res. 1994, 99, 149–151. [Google Scholar] [PubMed]
- Liu, H.; Liu, Z.-J.; Jing, J.; Ren, J.-Q.; Liu, Y.-Y.; Guo, H.-H.; Fan, M.; Lu, H.-J.; Jin, N.-Y. Reverse Transcription Loop-Mediated Isothermal Amplification for Rapid Detection of Japanese Encephalitis Virus in Swine and Mosquitoes. Vector-Borne Zoonotic Dis. 2012, 12, 1042–1052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turell, M.J.; O’Guinn, M.L.; Wasieloski, L.P.; Dohm, D.J.; Lee, W.-J.; Cho, H.-W.; Kim, H.-C.; Burkett, D.A.; Mores, C.N.; Coleman, R.E.; et al. Isolation of Japanese Encephalitis and Getah Viruses from Mosquitoes (Diptera: Culicidae) Collected near Camp Greaves, Gyonggi Province, Republic of Korea, 2000. J. Med. Entomol. 2003, 40, 580–584. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Fu, S.; Gong, Z.; Ge, J.; Meng, W.; Feng, Y.; Wang, J.; Zhai, Y.; Wang, H.; Nasci, R.; et al. Distribution of Arboviruses and Mosquitoes in Northwestern Yunnan Province, China. Vector-Borne Zoonotic Dis. 2009, 9, 623–630. [Google Scholar] [CrossRef]
- Kuwata, R.; Nga, P.T.; Yen, N.T.; Hoshino, K.; Isawa, H.; Higa, Y.; Hoang, N.V.; Trang, B.M.; Loan, D.P.; Phong, T.V.; et al. Surveillance of Japanese Encephalitis Virus Infection in Mosquitoes in Vietnam from 2006 to 2008. Am. J. Trop. Med. Hyg. 2013, 88, 681–688. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Guo, X.; Zhao, Q.; Tong, Y.; Fan, H.; Sun, Q.; Xing, S.; Zhou, H.; Zhang, J. Investigation on Mosquito-Borne Viruses at Lancang River and Nu River Watersheds in Southwestern China. Vector-Borne Zoonotic Dis. 2017. [Google Scholar] [CrossRef]
- Kumari, R.; Kumar, K.; Rawat, A.; Singh, G.; Yadav, N.K.; Chauhan, L.S. First Indigenous Transmission of Japanese Encephalitis in Urban Areas of National Capital Territory of Delhi, India. Trop. Med. Int. Health 2013, 18, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Yap, G.; Mailepessov, D.; Lim, X.F.; Chan, S.; How, C.B.; Humaidi, M.; Yeo, G.; Chong, C.S.; Lam-Phua, S.G.; Lee, R.; et al. Detection of Japanese Encephalitis Virus in Culex Mosquitoes in Singapore. Am. J. Trop. Med. Hyg. 2020, 103, 1234–1240. [Google Scholar] [CrossRef] [PubMed]
- Kuwata, R.; Sugiyama, H.; Yonemitsu, K.; Van Dung, N.; Terada, Y.; Taniguchi, M.; Shimoda, H.; Takano, A.; Maeda, K. Isolation of Japanese Encephalitis Virus and a Novel Insect-Specific Flavivirus from Mosquitoes Collected in a Cowshed in Japan. Arch. Virol. 2015, 160, 2151–2159. [Google Scholar] [CrossRef]
- Duong, V.; Choeung, R.; Gorman, C.; Laurent, D.; Crabol, Y.; Mey, C.; Peng, B.; Di Francesco, J.; Hul, V.; Sothy, H.; et al. Isolation and Full-Genome Sequences of Japanese Encephalitis Virus Genotype I Strains from Cambodian Human Patients, Mosquitoes and Pigs. J. Gen. Virol. 2017, 98, 2287–2296. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Rehman, M.U.; Li, K.; Luo, H.; Lan, Y.; Nabi, F.; Zhang, L.; Iqbal, M.K.; Zhu, S.; Javed, M.T.; et al. Epidemiologic Survey of Japanese Encephalitis Virus Infection, Tibet, China, 2015. Emerg. Infect. Dis. 2017, 23, 1023–1024. [Google Scholar] [CrossRef] [PubMed]
- Mitamura, T.; Kitaoka, M.; Watanabe, M.; Okuba, K.; Tnejin, S.; Yamada, S.; Mori, K.; Asada, J. Study on Japanese Encephalitis Virus. Animal Experiments and Mosquito Transmission Experiments. Kansai Iji 1936, 1, 260–261. [Google Scholar]
- Hale, J.H.; Colless, D.H.; Lim, K.A. Investigation of the Malaysian Form of Culex Tritaeniorhynchus as a Potential Vector of Japanese B Encephalitis Virus on Singapore Island. Ann. Trop. Med. Parasitol. 1957, 51, 17–25. [Google Scholar] [CrossRef]
- Gresser, I.; Hardy, J.L.; Hu, S.M.K.; Scherer, W.F. Factors Influencing Transmission of Japanese B Encephalitis Virus by a Colonized Strain of Culex Tritaeniorhynchus Giles, from Infected Pigs and Chicks to Susceptible Pigs and Birds. Am. J. Trop. Med. Hyg. 1958, 7, 365–373. [Google Scholar] [CrossRef]
- Gould, D.J.; Byrne, R.J.; Hayes, D.E. Experimental Infection of Horses with Japanese Encephalitis Virus by Mosquito Bite. Am. J. Trop. Med. Hyg. 1964, 13, 742–746. [Google Scholar] [CrossRef] [PubMed]
- Carey, D.E.; Reuben, R.; Myers, R.M. Japanese Encephalitis Studies in Vellore, South India. V. Experimental Infection and Transmission. Indian J. Med. Res. 1969, 57, 282–289. [Google Scholar] [PubMed]
- Takahashi, M. The Effects of Environmental and Physiological Conditions of Culex Tritaeniorhynchus on the Pattern of Transmission of Japanese Encephalitis Virus. J. Med. Entomol. 1976, 13, 275–284. [Google Scholar] [CrossRef]
- Soman, R.S.; Rodrigues, F.M.; Guttikar, S.N.; Guru, P.Y. Experimental Viraemia and Transmission of Japanese Encephalitis Virus by Mosquitoes in Ardeid Birds. Indian J. Med. Res. 1977, 66, 709–718. [Google Scholar]
- Chen, B.Q.; Beaty, B.J. Japanese Encephalitis Vaccine (2-8 Strain) and Parent (SA 14 Strain) Viruses in Culex Tritaeniorhynchus Mosquitoes. Am. J. Trop. Med. Hyg. 1982, 31, 403–407. [Google Scholar] [CrossRef]
- Turell, M.J.; Mores, C.N.; Dohm, D.J.; Lee, W.-J.; Kim, H.-C.; Klein, T.A. Laboratory Transmission of Japanese Encephalitis, West Nile, and Getah Viruses by Mosquitoes (Diptera: Culicidae) Collected near Camp Greaves, Gyeonggi Province, Republic of Korea 2003. J. Med. Entomol. 2006, 43, 1076–1081. [Google Scholar] [CrossRef]
- Faizah, A.N.; Kobayashi, D.; Amoa-Bosompem, M.; Higa, Y.; Tsuda, Y.; Itokawa, K.; Miura, K.; Hirayama, K.; Sawabe, K.; Isawa, H. Evaluating the Competence of the Primary Vector, Culex Tritaeniorhynchus, and the Invasive Mosquito Species, Aedes Japonicus Japonicus, in Transmitting Three Japanese Encephalitis Virus Genotypes. PLoS Negl. Trop. Dis. 2020, 14, e0008986. [Google Scholar] [CrossRef] [PubMed]
- Chakravarty, S.K.; Sarkar, J.K.; Chakravarty, M.S.; Mukherjee, M.K.; Mukherjee, K.K.; Das, B.C.; Hati, A.K. The First Epidemic of Japanese Encephalitis Studied in India—Virological Studies. Indian J. Med. Res. 1975, 63, 77–82. [Google Scholar]
- Sahu, S.S.; Dash, S.; Sonia, T.; Muthukumaravel, S.; Sankari, T.; Gunasekaran, K.; Jambulingam, P. Entomological Investigation of Japanese Encephalitis Outbreak in Malkangiri District of Odisha State, India. Mem. Inst. Oswaldo Cruz 2018, 113, e170499. [Google Scholar] [CrossRef] [Green Version]
- Sarkar, A.; Taraphdar, D.; Mukhopadhyay, S.K.; Chakrabarti, S.; Chatterjee, S. Molecular Evidence for the Occurrence of Japanese Encephalitis Virus Genotype I and III Infection Associated with Acute Encephalitis in Patients of West Bengal, India, 2010. Virol. J. 2012, 9, 271. [Google Scholar] [CrossRef] [Green Version]
- Hurlbut, H.S. The Pig-Mosquito Cycle of Japanese Encephalitis Virus in Taiwan. J. Med. Entomol. 1964, 1, 301–307. [Google Scholar] [CrossRef]
- Bhatt, T.R.; Crabtree, M.B.; Guirakhoo, F.; Monath, T.P.; Miller, B.R. Growth Characteristics of the Chimeric Japanese Encephalitis Virus Vaccine Candidate, ChimeriVax-JE (YF/JE SA14--14--2), in Culex Tritaeniorhynchus, Aedes Albopictus, and Aedes Aegypti Mosquitoes. Am. J. Trop. Med. Hyg. 2000, 62, 480–484. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.; Zhang, Q.; Zhou, J.; Yu, S.; Zheng, X.; Chen, Q. Susceptibility of Aedes albopictus and Culex pipiens quinquefasciatus to infection with bat Japanese encephalitis virus isolates. Nan Fang Yi Ke Da Xue Xue Bao 2012, 32, 515–518. [Google Scholar]
- Banerjee, K.; Deshmukh, P.K. Transmission of Japanese Encephalitis Virus to Chicks by Individual Culex Bitaeniorhynchus Mosquitoes. Indian J. Med. Res. 1987, 86, 726–727. [Google Scholar]
- Van Peenen, P.F.D.; Joseph, P.L.; Atmosoedjono, S.; Irsiana, R.; Saroso, J.S. Isolation of Japanese Encephalitis Virus from Mosquitoes near Bogor, West Java, Indonesia. J. Med. Entomol. 1975, 12, 573–574. [Google Scholar] [CrossRef]
- Kramer, L.D.; Chin, P.; Cane, R.P.; Kauffman, E.B.; Mackereth, G. Vector Competence of New Zealand Mosquitoes for Selected Arboviruses. Am. J. Trop. Med. Hyg. 2011, 85, 182–189. [Google Scholar] [CrossRef]
- Liu, K.; Hou, F.; Wahaab, A.; Kang, L.; Xie, F.; Ma, X.; Xia, Q.; Xiao, C.; Shao, D.; Li, B.; et al. Mosquito Defensin Facilitates Japanese Encephalitis Virus Infection by Downregulating the C6/36 Cell-Surface Antiviral Protein HSC70B. Vet. Microbiol. 2021, 253, 108971. [Google Scholar] [CrossRef]
- Liu, K.; Qian, Y.; Jung, Y.-S.; Zhou, B.; Cao, R.; Shen, T.; Shao, D.; Wei, J.; Ma, Z.; Chen, P.; et al. MosGCTL-7, a C-Type Lectin Protein, Mediates Japanese Encephalitis Virus Infection in Mosquitoes. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doi, R.; Shirasaka, A.; Sasa, M.; Oya, A. Studies on the Susceptibility of Three Species of Mosquitoes to Japanese Encephalitis Virus. J. Med. Entomol. 1977, 13, 591–594. [Google Scholar] [CrossRef]
- Hurlbut, H.S. The Transmission of Japanese B Encephalitis by Mosquitoes after Experimental Hibernation. Am. J. Hyg. 1950, 51, 265–268. [Google Scholar] [CrossRef] [PubMed]
- Mourya, D.T.; Gokhale, M.D.; Pidiyar, V.; Barde, P.V.; Patole, M.; Mishra, A.C.; Shouche, Y. Study of the Effect of the Midgut Bacterial Flora of Culex Quinquefasciatus on the Susceptibility of Mosquitoes to Japanese Encephalitis Virus. Acta Virol. 2002, 46, 257–260. [Google Scholar] [PubMed]
- Sudeep, A.B.; Bondre, V.P.; George, R.; Ghodke, Y.S.; Aher, R.V.; Gokhale, M.D. Bagaza Virus Inhibits Japanese Encephalitis & West Nile Virus Replication in Culex Tritaeniorhynchus & Cx. Quinquefasciatus Mosquitoes. Indian J. Med. Res. 2015, 142 (Suppl. S1), S44–S51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitamura, T.; Kitaoka, M.; Mori, K.; Okuba, K. Isolation of the Virus of Japanese Epidemic Encephalitis from Mosquitoes Caught in Nature. Tokyi Iji Shinshi 1938, 62, 820–831. [Google Scholar]
- Takahashi, M. Variation in Susceptibility among Colony Strains of Culex Tritaeniorhynchus to Japanese Encephalitis Virus Infection. Jpn. J. Med. Sci. Biol. 1980, 33, 321–329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huber, K.; Jansen, S.; Leggewie, M.; Badusche, M.; Schmidt-Chanasit, J.; Becker, N.; Tannich, E.; Becker, S.C. Aedes Japonicus Japonicus (Diptera: Culicidae) from Germany Have Vector Competence for Japan Encephalitis Virus but Are Refractory to Infection with West Nile Virus. Parasitol. Res. 2014, 113, 3195–3199. [Google Scholar] [CrossRef]
- Dhanda, V.; Kaul, H.N. Mosquito Vectors of Japanese Encephalitis Virus and Their Bionomics in India. Proc. Indian Natl. Sci. Acad. 1980, 6, 759–768. [Google Scholar]
- Cates, M.D.; Detels, R. Japanese Encephalitis Virus in Taiwan: Preliminary Evidence for Culex Annulus Theob. as a Vector. J. Med. Entomol. 1969, 6, 327–328. [Google Scholar] [CrossRef] [PubMed]
- Cross, J.H.; Lien, J.C.; Huang, W.C.; Lien, S.C.; Chiu, S.F. Japanese Encephalitis Virus Surveillance in Taiwan. II. Isolations from Mosquitoes and Bats in Taipei Area 1969-1970. Taiwan Yi Xue Hui Za Zhi 1971, 70, 681–686. [Google Scholar] [PubMed]
- Banerjee, K.; Mahadev, P.V.; Ilkal, M.A.; Mishra, A.C.; Dhanda, V.; Modi, G.B.; Geevarghese, G.; Kaul, H.N.; Shetty, P.S.; George, P.J. Isolation of Japanese Encephalitis Virus from Mosquitoes Collected in Bankura District (West Bengal) during October 1974 to December 1975. Indian J. Med. Res. 1979, 69, 201–205. [Google Scholar] [PubMed]
- Thenmozhi, V.; Rajendran, R.; Ayanar, K.; Manavalan, R.; Tyagi, B.K. Long-Term Study of Japanese Encephalitis Virus Infection in Anopheles Subpictus in Cuddalore District, Tamil Nadu, South India. Trop. Med. Int. Health 2006, 11, 288–293. [Google Scholar] [CrossRef]
- Hemmerter, S.; Šlapeta, J.; van den Hurk, A.F.; Cooper, R.D.; Whelan, P.I.; Russell, R.C.; Johansen, C.A.; Beebe, N.W. A Curious Coincidence: Mosquito Biodiversity and the Limits of the Japanese Encephalitis Virus in Australasia. BMC Evol. Biol. 2007, 7, 100. [Google Scholar] [CrossRef] [Green Version]
- Delatte, H.; Desvars, A.; Bouétard, A.; Bord, S.; Gimonneau, G.; Vourc’h, G.; Fontenille, D. Blood-Feeding Behavior of Aedes Albopictus, a Vector of Chikungunya on La Réunion. Vector-Borne Zoonotic Dis. 2010, 10, 249–258. [Google Scholar] [CrossRef] [Green Version]
- Srinivas, S.D.; Pandian, R.S.; Dwarakanath, S.K. Biting Behaviour of Armigeres Subalbatus (Coquillett) with Reference to Host Selection and Landing. Indian J. Exp. Biol. 1994, 32, 348–350. [Google Scholar] [PubMed]
- Lien, J.C.; Huang, W.C.; Cross, J.H. Japanese Encephalitis Virus Surveillance in the Taipei Area, Taiwan in 1978. Southeast Asian J. Trop. Med. Public Health 1980, 11, 177–183. [Google Scholar] [PubMed]
- Prummongkol, S.; Panasoponkul, C.; Apiwathnasorn, C.; Lek-Uthai, U. Biology of Culex Sitiens, a Predominant Mosquito in Phang Nga, Thailand after a Tsunami. J. Insect Sci. 2012, 12, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Hall-Mendelin, S.; Jansen, C.C.; Cheah, W.Y.; Montgomery, B.L.; Hall, R.A.; Ritchie, S.A.; van den Hurk, A.F. Culex Annulirostris (Diptera: Culicidae) Host Feeding Patterns and Japanese Encephalitis Virus Ecology in Northern Australia. J. Med. Entomol. 2012, 49, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Murty, U.S.; Rao, M.S.; Arunachalam, N. The Effects of Climatic Factors on the Distribution and Abundance of Japanese Encephalitis Vectors in Kurnool District of Andhra Pradesh, India. J. Vector Borne Dis. 2010, 47, 26–32. [Google Scholar] [PubMed]
- Reuben, R.; Thenmozhi, V.; Samuel, P.P.; Gajanana, A.; Mani, T.R. Mosquito Blood Feeding Patterns as a Factor in the Epidemiology of Japanese Encephalitis in Southern India. Am. J. Trop. Med. Hyg. 1992, 46, 654–663. [Google Scholar] [CrossRef] [PubMed]
- Zinser, M.; Ramberg, F.; Willott, E. Scientific Note Culex Quinquefasciatus (Diptera: Culicidae) as a Potential West Nile Virus Vector in Tucson, Arizona: Blood Meal Analysis Indicates Feeding on Both Humans and Birds. J. Insect Sci. 2004, 4, 20. [Google Scholar] [CrossRef] [Green Version]
- Hasegawa, M.; Tuno, N.; Yen, N.T.; Nam, V.S.; Takagi, M. Influence of the Distribution of Host Species on Adult Abundance of Japanese Encephalitis Vectors Culex Vishnui Subgroup and Culex Gelidus in a Rice-Cultivating Village in Northern Vietnam. Am. J. Trop. Med. Hyg. 2008, 78, 159–168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boyer, S.; Durand, B.; Yean, S.; Brengues, C.; Maquart, P.-O.; Fontenille, D.; Chevalier, V. Host-Feeding Preference and Diel Activity of Mosquito Vectors of the Japanese Encephalitis Virus in Rural Cambodia. Pathogens 2021, 10, 376. [Google Scholar] [CrossRef] [PubMed]
- Farajollahi, A.; Fonseca, D.M.; Kramer, L.D.; Marm Kilpatrick, A. “Bird Biting” Mosquitoes and Human Disease: A Review of the Role of Culex Pipiens Complex Mosquitoes in Epidemiology. Infect. Genet. Evol. 2011, 11, 1577–1585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molaei, G.; Andreadis, T.G.; Armstrong, P.M.; Bueno, R.; Dennett, J.A.; Real, S.V.; Sargent, C.; Bala, A.; Randle, Y.; Guzman, H.; et al. Host Feeding Pattern of Culex Quinquefasciatus (Diptera: Culicidae) and Its Role in Transmission of West Nile Virus in Harris County, Texas. Am. J. Trop. Med. Hyg. 2007, 77, 73–81. [Google Scholar] [CrossRef] [Green Version]
- Mottram, P.; Kettle, D.S. Development and Survival of Immature Culex Annulirostris Mosquitoes in Southeast Queensland. Med. Vet. Entomol. 1997, 11, 181–186. [Google Scholar] [CrossRef]
- Medlock, J.M.; Avenell, D.; Barrass, I.; Leach, S. Analysis of the Potential for Survival and Seasonal Activity of Aedes Albopictus (Diptera: Culicidae) in the United Kingdom. J. Vector Ecol. 2006, 31, 292–304. [Google Scholar] [CrossRef]
- Rochlin, I.; Ninivaggi, D.V.; Hutchinson, M.L.; Farajollahi, A. Climate Change and Range Expansion of the Asian Tiger Mosquito (Aedes Albopictus) in Northeastern USA: Implications for Public Health Practitioners. PLoS ONE 2013, 8, e60874. [Google Scholar] [CrossRef] [PubMed]
- Giordano, B.V.; Gasparotto, A.; Liang, P.; Nelder, M.P.; Russell, C.; Hunter, F.F. Discovery of an Aedes (Stegomyia) Albopictus Population and First Records of Aedes (Stegomyia) Aegypti in Canada. Med. Vet. Entomol. 2020, 34, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Ree, H.I.; Hong, H.K.; Lee, J.S.; Wada, Y.; Lolivet, P. Dispersal Experiment on Culex Tritaeniorhynchus in Korea. Korean J. Zool. 1978, 21, 59–66. [Google Scholar]
- Masuoka, P.; Klein, T.A.; Kim, H.-C.; Claborn, D.M.; Achee, N.; Andre, R.; Chamberlin, J.; Taylor, K.; Small, J.; Anyamba, A.; et al. Modeling and Analysis of Mosquito and Environmental Data to Predict the Risk of Japanese Encephalitis. In Proceedings of the ASPRS 2009 Annual Conference, Baltimore, MD, USA, 9–13 March 2009. [Google Scholar]
- Longbottom, J.; Browne, A.J.; Pigott, D.M.; Sinka, M.E.; Golding, N.; Hay, S.I.; Moyes, C.L.; Shearer, F.M. Mapping the Spatial Distribution of the Japanese Encephalitis Vector, Culex Tritaeniorhynchus Giles, 1901 (Diptera: Culicidae) within Areas of Japanese Encephalitis Risk. Parasites Vectors 2017, 10, 148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dickson, L.B.; Jiolle, D.; Minard, G.; Moltini-Conclois, I.; Volant, S.; Ghozlane, A.; Bouchier, C.; Ayala, D.; Paupy, C.; Moro, C.V.; et al. Carryover Effects of Larval Exposure to Different Environmental Bacteria Drive Adult Trait Variation in a Mosquito Vector. Sci. Adv. 2017, 3, e1700585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- White, L.A. Susceptibility of Aedes Albopictus C6/36 Cells to Viral Infection. J. Clin. Microbiol. 1987, 25, 1221–1224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takahashi, M.; Yabe, S.; Okada, T. Effects of Various Passages on Some Properties of an Attenuated Strain of Japanese Encephalitis Virus with Special Regard to Mosquito Infectivity. Jpn. J. Med. Sci. Biol. 1969, 22, 163–174. [Google Scholar] [CrossRef]
- Park, S.L.; Huang, Y.-J.S.; Lyons, A.C.; Ayers, V.B.; Hettenbach, S.M.; McVey, D.S.; Burton, K.R.; Higgs, S.; Vanlandingham, D.L. North American Domestic Pigs Are Susceptible to Experimental Infection with Japanese Encephalitis Virus. Sci. Rep. 2018, 8, 7951. [Google Scholar] [CrossRef] [PubMed]
- Scherer, W.F.; Moyer, J.T.; Izumi, T. Immunologic Studies of Japanese Encephalitis Virus in Japan. V. Maternal Antibodies, Antibody Responses and Viremia Following Infection of Swine. J. Immunol. 1959, 83, 620–626. [Google Scholar] [PubMed]
- Cleton, N.B.; Bosco-Lauth, A.; Page, M.J.; Bowen, R.A. Age-Related Susceptibility to Japanese Encephalitis Virus in Domestic Ducklings and Chicks. Am. J. Trop. Med. Hyg. 2014, 90, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Carrington, L.B.; Seifert, S.N.; Armijos, M.V.; Lambrechts, L.; Scott, T.W. Reduction of Aedes Aegypti Vector Competence for Dengue Virus under Large Temperature Fluctuations. Am. J. Trop. Med. Hyg. 2013, 88, 689–697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lambrechts, L.; Chevillon, C.; Albright, R.G.; Thaisomboonsuk, B.; Richardson, J.H.; Jarman, R.G.; Scott, T.W. Genetic Specificity and Potential for Local Adaptation between Dengue Viruses and Mosquito Vectors. BMC Evol. Biol. 2009, 9, 160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zouache, K.; Fontaine, A.; Vega-Rua, A.; Mousson, L.; Thiberge, J.-M.; Lourenco-De-Oliveira, R.; Caro, V.; Lambrechts, L.; Failloux, A.-B. Three-Way Interactions between Mosquito Population, Viral Strain and Temperature Underlying Chikungunya Virus Transmission Potential. Proc. Biol. Sci. 2014, 281, 20141078. [Google Scholar] [CrossRef] [PubMed]
- Miot, E.F.; Calvez, E.; Aubry, F.; Dabo, S.; Grandadam, M.; Marcombe, S.; Oke, C.; Logan, J.G.; Brey, P.T.; Lambrechts, L. Risk of Arbovirus Emergence via Bridge Vectors: Case Study of the Sylvatic Mosquito Aedes Malayensis in the Nakai District, Laos. Sci. Rep. 2020, 10, 7750. [Google Scholar] [CrossRef] [PubMed]
- Pongsiri, A.; Ponlawat, A.; Thaisomboonsuk, B.; Jarman, R.G.; Scott, T.W.; Lambrechts, L. Differential Susceptibility of Two Field Aedes Aegypti Populations to a Low Infectious Dose of Dengue Virus. PLoS ONE 2014, 9, e92971. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fontaine, A.; Lequime, S.; Moltini-Conclois, I.; Jiolle, D.; Leparc-Goffart, I.; Reiner, R.C.; Lambrechts, L. Epidemiological Significance of Dengue Virus Genetic Variation in Mosquito Infection Dynamics. PLoS Pathog. 2018, 14, e1007187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dickson, L.B.; Ghozlane, A.; Volant, S.; Bouchier, C.; Ma, L.; Vega-Rúa, A.; Dusfour, I.; Jiolle, D.; Paupy, C.; Mayanja, M.N.; et al. Diverse Laboratory Colonies of Aedes Aegypti Harbor the Same Adult Midgut Bacterial Microbiome. Parasites Vectors 2018, 11, 207. [Google Scholar] [CrossRef] [Green Version]
- Baidaliuk, A.; Miot, E.F.; Lequime, S.; Moltini-Conclois, I.; Delaigue, F.; Dabo, S.; Dickson, L.B.; Aubry, F.; Merkling, S.H.; Cao-Lormeau, V.-M.; et al. Cell-Fusing Agent Virus Reduces Arbovirus Dissemination in Aedes Aegypti Mosquitoes In Vivo. J. Virol. 2019, 93. [Google Scholar] [CrossRef] [Green Version]
- Huang, C.H.; Liang, H.C.; Jia, F.L. Beneficial Role of a Nonpathogenic Orbi-like Virus: Studies on the Interfering Effect of M14 Virus in Mice and Mosquitoes Infected with Japanese Encephalitis Virus. Intervirology 1985, 24, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Carvajal, T.M.; Capistrano, J.D.R.; Hashimoto, K.; Go, K.J.D.; Cruz, M.A.I.J.; Martinez, M.J.L.B.; Tiopianco, V.S.P.; Amalin, D.M.; Watanabe, K. Detection and Distribution of Wolbachia Endobacteria in Culex Quinquefasciatus Populations (Diptera: Culicidae) from Metropolitan Manila, Philippines. J. Vector Borne Dis. 2018, 55, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Nugapola, N.W.N.P.; De Silva, W.A.P.P.; Karunaratne, S.H.P.P. Distribution and Phylogeny of Wolbachia Strains in Wild Mosquito Populations in Sri Lanka. Parasites Vectors 2017, 10, 230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeffries, C.L.; Walker, T. The Potential Use of Wolbachia-Based Mosquito Biocontrol Strategies for Japanese Encephalitis. PLoS Negl. Trop. Dis. 2015, 9, e0003576. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Mosquito Species | Virus Detection in Field-Caught Mosquitoes | Virus Isolation from Field-Caught Mosquitoes | Proven Vector Competence * |
|---|---|---|---|
| Aedes albopictus | 1995–1996 in Taiwan [28]; 1992 in Malaysia [29]; 2005–2012 in Taiwan [30] | [31,32]; [33,34] | |
| Aedes vexans | 1995–1996 in Taiwan [28] | [35,36] | |
| Aedes vigilax | 1997–1998 in Australia [37] | [38] | |
| Armigeres subalbatus | 2011–2013 in India [39] | 1995–1996 in Taiwan [28]; 2007 in China [40]; 2009–2010 in China [41] | [32,42] |
| Culex annulirostris | 1995 in Australia [43]; 1997–1998 in Australia [37] | [35]; [38,44] | |
| Culexbitaeniorhynchus | 1992 in Malaysia [29]; 2008–2010 in South Korea [45]; 2008–2010 in South Korea [46]; 2010 in South Korea [47,48]; 2011–2013 in India [39] | 1992 in Malaysia [29] | [49,50,51] |
| Culexfuscocephala | 1987–1988 in Sri Lanka [52]; 1991–1994 in India [53]; 2006–2008 in Taiwan [54]; 2011–2013 in India [39] | 1970 in Thailand [55]; 1974–1976 in Taiwan [56]; 1978–1980 in Indonesia [57]; 1985–1987 in India [58]; 1991–1994 in India [53]; 1992 in Malaysia [59] | [60,61] |
| Culexgelidus | 1987–1988 in Sri Lanka [52]; 1991–1994 in India [53]; 1995–1997 in India [62]; 1996–2004 in India [63]; 1998–2000 in India [64]; 1999–2000 in India [65]; 2002–2005 in India [66]; 2003–2004 in Australia [67]; 2010–2013 in India [68]; 2011 in India [69]; 2016 in India [70] | 1954–1956 in Malaysia [19]; 1962–1968 in Thailand [71]; 1968–1969 in Malaysia [72]; 1969 in Malaysia [73,74]; 1972–1973 in Vietnam [75]; 1972–1974 in Indonesia [76]; 1978–1980 in Indonesia [57]; 1985–1987 in India [58]; 1986–1987 in Thailand [77]; 1987–1988 in Sri Lanka [52]; 1991–1994 in India [53]; 1992 in Malaysia [29]; 1992–1994 in Malaysia [78]; 2000 in Australia [79,80]; 2002–2006 in India [81] | [19,38,82] |
| Culexpipiens (including subspecies molestus, pallens, pipiens) | 2008–2010 in South Korea [46]; 2010 in South Korea [48]; 2010–2011 in Italy [83]; 2012 in South Korea [84]; 2016 in China [85] | 1952–1957 in Japan [86]; 1960s in South Korea [87]; 1970s in China [88]; 2015 in China [89] | [34,36,90,91,92,93,94,95,96,97,98] |
| Culexpseudovishnui | 1985 + 1987 in India [99]; 2009–2010 in India [100]; 2010–2013 in India [101]; | 1977–1979 in India [102]; 1988 in India [103] | [104,105] |
| Culexquinquefasciatus | 2009 in Vietnam [106]; 2011–2013 in India [39] | 1972–1973 in Vietnam [75]; 1985–1987 in India [58]; 1995–1996 in Taiwan [28]; 2003 in Thailand [107] | [32,35,36,38,51,91,108,109,110,111,112,113,114] |
| Culexsitiens | 1992 in Malaysia [29,115]; 2002–2004 in Taiwan [116]; 2003–2005 in Australia [67]; 2004 in Australia [117] | 1992 in Malaysia [29]; 1995–1996 in Taiwan [28]; 1997–1998 in Papua New Guinea [118]; 1998 in Australia [119]; 2000 in Australia [120] | [38] |
| Culextritaeniorhynchus | 1985–1987 in India [99]; 1987–1988 in Sri Lanka [52]; 1991-1994 in India [53]; 1992 in Malaysia [29]; 1995–1997 in India [62]; 1996 in India [121]; 1996–2004 in India [63]; 1998–2000 in India [64]; 1999–2000 in India [65]; 2001–2003 in India [122]; 2002–2004 in Taiwan [116]; 2002–2005 in India [66]; 2006–2008 in Taiwan [54]; 2008–2009 in South Korea [123]; 2008–2010 in South Korea [46]; 2008–2010 in South Korea [45]; 2009 in Vietnam [106]; 2009–2010 in India [100]; 2009–2010 in Taiwan [124]; 2010 in South Korea [47,48]; 2010–2013 in India [68,101]; 2010–2014 in Japan [125]; 2011 in India [69]; 2011–2013 in India [39]; 2011–2014 in India [126]; 2013 in China [127]; 2016 in China [85]; 2016 in India [70] 2018 in China [128] | 1962–1966 in India [129]; 1962–1968 in Thailand [71]; 1964–1968 in Japan [130]; 1968–1969 in Malaysia; 1968–1970 in Malaysia [74] 1969 in Taiwan [131] 1972 in Indonesia [132]; 1972–1973 in Vietnam [75]; 1974–1976 in Taiwan [56]; 1977–1979 in India [102]; 1978–1980 in Indonesia [57]; 1979 in Indonesia [133]; 1980s in Taiwan [134]; 1982 in Thailand [135]; 1985–1987 in India [58]; 1986–1991 in Japan [136]; 1990s in India [137]; 1991–1994 in India [53]; 1992–1993 in Malaysia [59]; 1995–1996 in Taiwan [28]; 2000s in China [138]; 2000 in South Korea [139]; 2005–2006 in China [140]; 2006–2008 in Vietnam [141]; 2007 in China [40]; 2007–2009 in China [142]; 2009–2010 in China [41]; 2011 in India [143]; 2011–2013 in Singapore [144]; 2013 in Japan [145]; 2014 in Cambodia [146]; 2015 in China [89]; 2015 in China [147] | [23,49,51,60,61,90,92,105,110,148,149,150,151,152,153,154,155,156,157] |
| Culexvishnui | 1970s in India [158]; 1991–1994 in India [53]; 1992 in Malaysia [29]; 1995–1997 in India [62]; 1996–2004 in India [63]; 2006–2008 in Vietnam [141]; 2009–2010 in India [100]; 2010–2013 in India [101]; 2011 in India [143]; 2016 in India [159] | 1978–1980 in Indonesia [57]; 1982 in Thailand [135]; 1991–1994 in India [53]; 2010 in India [160] | [105] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Auerswald, H.; Maquart, P.-O.; Chevalier, V.; Boyer, S. Mosquito Vector Competence for Japanese Encephalitis Virus. Viruses 2021, 13, 1154. https://doi.org/10.3390/v13061154
Auerswald H, Maquart P-O, Chevalier V, Boyer S. Mosquito Vector Competence for Japanese Encephalitis Virus. Viruses. 2021; 13(6):1154. https://doi.org/10.3390/v13061154
Chicago/Turabian StyleAuerswald, Heidi, Pierre-Olivier Maquart, Véronique Chevalier, and Sebastien Boyer. 2021. "Mosquito Vector Competence for Japanese Encephalitis Virus" Viruses 13, no. 6: 1154. https://doi.org/10.3390/v13061154
APA StyleAuerswald, H., Maquart, P.-O., Chevalier, V., & Boyer, S. (2021). Mosquito Vector Competence for Japanese Encephalitis Virus. Viruses, 13(6), 1154. https://doi.org/10.3390/v13061154






