Orthohantavirus Survey in Indigenous Lands in a Savannah-Like Biome, Brazil
Abstract
1. Introduction
2. Materials and Methods
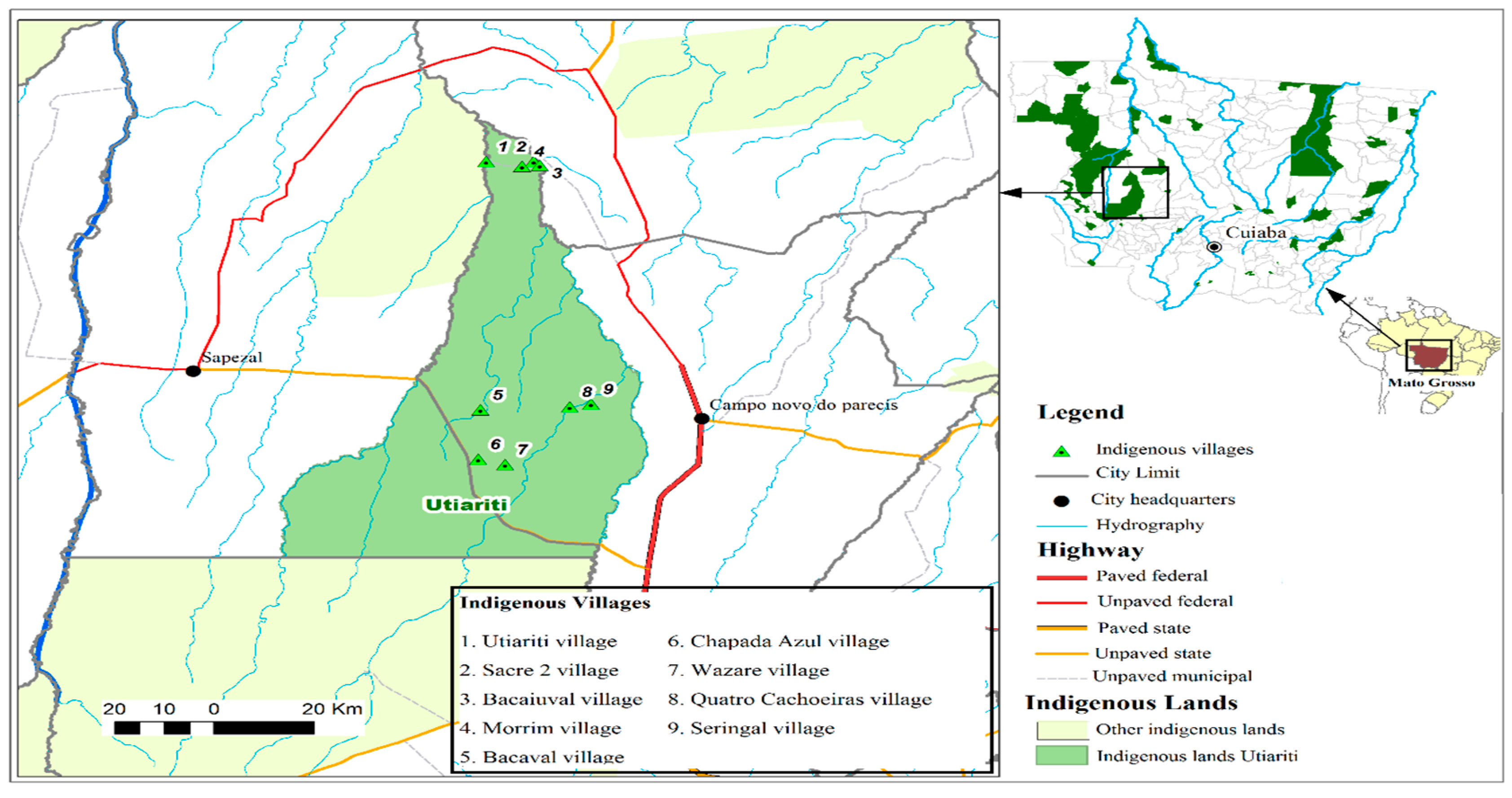
2.1. Study Area
2.2. Sample Collection
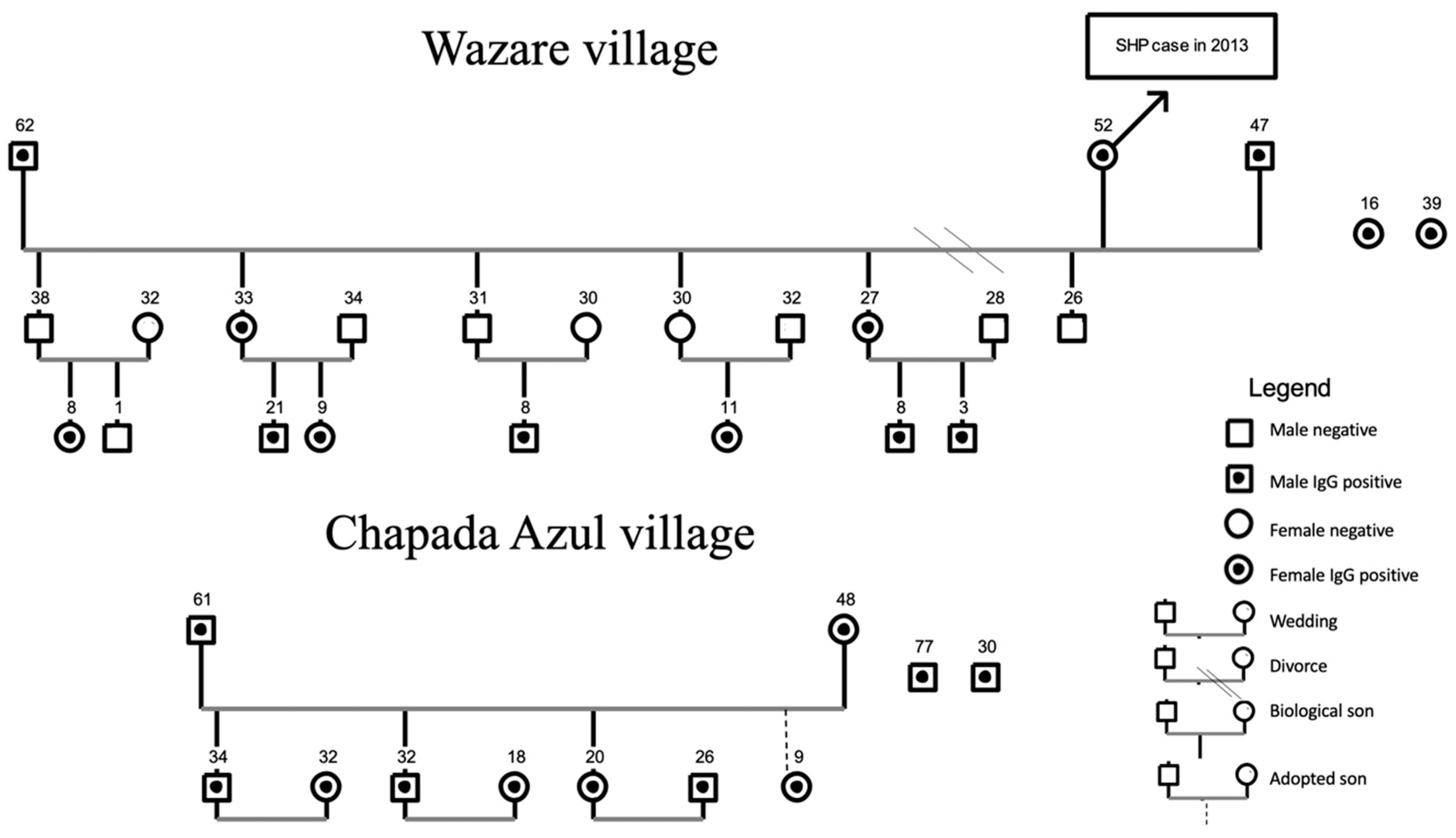
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Lee, H.W.; Lee, P.W.; Johnson, K.M. Isolation of the etiologic agent of Korean Hemorrhagic fever. J. Infect. Dis. 1978, 137, 298–308. [Google Scholar] [CrossRef]
- Nichol, S.T.; Spiropoulou, C.F.; Morzunov, S.; Rollin, P.E.; Ksiazek, T.G.; Feldmann, H.; Sanchez, A.; Childs, J.; Zaki, S.; Peters, C.J. Genetic Identification of a Hantavirus Associated with an Outbreak of Acute Respiratory Illness. Science 1993, 262, 914–917. [Google Scholar] [CrossRef]
- Lemos, E.R.S.; Silva, M.V. Hantavírus. In Dinâmica das Doenças Infecciosas e Parasitárias; Coura, J.R., Ed.; Guanabara Koogan: Rio de Janeiro, Brazil, 2013; pp. 1885–1897. [Google Scholar]
- Jonsson, C.B.; Figueiredo, L.T.; Vapalahti, O. A global perspective on hantavirus ecology, epidemiology, and disease. Clin. Microbiol. Rev. 2010, 23, 412–444. [Google Scholar] [CrossRef]
- Oliveira, R.C.; Sant’ana, M.M.; Guterres, A.; Fernandes, J.; Hillesheim, N.L.; Lucini, C.; Gomes, R.; Lamas, C.; Bochner, R.; Zeccer, S.; et al. Hantavirus pulmonary syndrome in a highly endemic area of Brazil. Epidemiol. Infect. 2016, 144, 1096–1106. [Google Scholar] [CrossRef]
- Lee, H.W.; Lee, P.W.; Baek, L.J.; Song, C.K.; Seong, I.W. Intraspecific transmission of Hantaan virus, etiologic agent of Korean hemorrhagic fever, in the rodent Apodemus agrarius. Am. J. Trop. Med. Hyg. 1981, 30, 1106–1112. [Google Scholar] [CrossRef] [PubMed]
- Childs, J.C.; Peter, C.J. Ecology and epidemiology of arenaviruses and their hosts. In The Arenaviridae; Salvato, M.S., Ed.; Plenun Press: New York, NY, USA, 1993; pp. 331–373. [Google Scholar]
- Hutchinson, K.L.; Rollin, P.E.; Peters, C.J. Pathogenesis of a North American hantavirus, Black Creek Canal virus, in experimentally infected Sigmodon hispidus. Am. J. Trop. Med. Hyg. 1998, 59, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde; Ministério da Saúde: Brasília, Brazil, 2014.
- Martinez, V.P.; Bellomo, C.M.; Cacace, M.L.; Suarez, P.; Bogni, L.; Padula, P.J. Hantavirus pulmonary syndrome in Argentina, 1995–2008. Emerg. Infect. Dis. 2010, 16, 1853–1860. [Google Scholar] [CrossRef] [PubMed]
- MacNeil, A.; Ksiazek, T.G.; Rollin, P.E. Hantavirus pulmonary syndrome, United States, 1993–2009. Emerg. Infect. Dis. 2011, 17, 1195–1201. [Google Scholar] [CrossRef]
- López, F.; Muñoz, M.; Galeano, R.; Rojas Silva, A.; Pereira, Y.; Gómez, P.; Paredes, H.; Samudio, M.; Cabello, M.A.; Ojeda, A.; et al. Características epidemiológicas del síndrome pulmonar por hantavirus y los factores asociados al óbito en el Chaco Central—Paraguay. Año 2011. Rev. Parag. Epidemiol. 2012, 2, 27–28. [Google Scholar]
- Montgomery, J.M.; Blair, P.J.; Carroll, D.S.; Mills, J.N.; Gianella, A. Hantavirus Pulmonary Syndrome in Santa Cruz, Bolivia: Outbreak Investigation and Antibody Prevalence Study. PLoS Negl. Trop. Dis. 2012, 6, 1840. [Google Scholar] [CrossRef] [PubMed]
- Riquelme, R.; Rioseco, M.L.; Bastidas, L.; Trincado, D.; Riquelme, M.; Loyola, H.; Valdivieso, F. Hantavirus pulmonary syndrome, Southern Chile, 1995–2012. Emerg. Infect. Dis. 2015, 21, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Drebot, M.A.; Jones, S.; Grolla, A.; Safronetz, D.; Strong, J.E.; Kobinger, G.; Lindsay, R.L. Hantavirus pulmonary syndrome in Canada: An overview of clinical features, diagnostics, epidemiology and prevention. Can. Commun. Dis. Rep. 2015, 41, 65–69. [Google Scholar] [CrossRef]
- Brasil. Casos Confirmados de Hantavirose 1993–2018; Ministério da Saúde: Brasília, Brazil, 2019. Available online: https://antigo.saude.gov.br/images/pdf/2019/junho/07/CONF-HANTA-93-19-ATUAL-24-05-2019.pdf (accessed on 20 August 2020).
- Medeiros, D.B.A.; Travassos da Rosa, E.S.; Marques, A.A.R.; Simith, D.B.; Carneiro, A.R.; Chiang, J.O. Circulation of hantaviruses in the influence area of the Cuiabá-Santarém Highway. Mem. Inst. Oswaldo Cruz. 2010, 105, 665–671. [Google Scholar] [CrossRef] [PubMed]
- Travassos da Rosa, E.S.; Medeiros, D.B.A.; Nunes, M.R.T.; Simith, D.B.; Pereira, A.S.; Elkhoury, M.R.; Terças, A.C.P. Pygmy Rice Rat as Potential Host of Castelo dos Sonhos Hantavirus. Emerg. Infect. Dis. 2011, 17, 1527–1530. [Google Scholar] [CrossRef]
- Rosa, E.S.T.; Medeiros, D.B.; Nunes, M.R.; Simith, D.B.; Pereira, A.S.; Elkhoury, M.R. Molecular epidemiology of Laguna Negra Virus, Mato Grosso State, Brazil. Emerg. Infect. Dis. 2012, 18, 982–985. [Google Scholar] [CrossRef]
- Ferrer, J.F.; Jonsson, C.B.; Esteban, E.; Galligan, D.; Basombrio, M.A.; Peralta Ramos, M. High prevalence of hantavirus infection in Indian communities of the Paraguayan and Argentinean Gran Chaco. Am. J. Trop. Med. Hyg. 1998, 59, 438–444. [Google Scholar] [CrossRef]
- Chu, Y.K.; Owen, R.D.; Gonzalez, L.M.; Jonsson, C.B. The complex ecology of hantavirus in Paraguay. Am. J. Trop. Med. Hyg. 2003, 69, 263–268. [Google Scholar] [CrossRef]
- Pini, N.; Levis, S.; Calderon, G.; Ramirez, J.; Bravo, D.; Lozano, E. Hantavirus infection in humans and rodents, northwestern Argentina. Emerg. Infect. Dis. 2003, 9, 1070–1076. [Google Scholar] [CrossRef]
- Lopes, L.; Guterres, A.; Rozental, T.; Carvalho de Oliveira, R.; Mares-Guia, M.A.; Fernandes, J.; Figueredo, J.F.; Anschau, I.; de Jesus, S.; Almeida, A.B.M.V.; et al. Rickettsia bellii, Rickettsia amblyommii, and Laguna Negra hantavirus in an Indian reserve in the Brazilian Amazon. Parasit Vectors 2014, 7, 191. [Google Scholar] [CrossRef] [PubMed]
- Barrera, S.; Martínez, S.; Tique-Salleg, V.; Miranda, J.; Guzmán, C.; Mattar, S. Seroprevalencia de Hantavirus, Rickettsia y Chikungunya en población indígena del municipio de Tuchín, Córdoba. Infectio 2015, 19, 75–82. [Google Scholar] [CrossRef]
- DATASUS. Informações em saúde: Epidemiológicas e Morbidade; Doenças e Agravos de Notificação, Hantavirose: Brasília, Brazil, 2021; Available online: http://www2.datasus.gov.br/DATASUS/index.php?area=0203&id=29892133&VObj=http://tabnet.datasus.gov.br/cgi/deftohtm.exe?sinannet/cnv/hanta and http://www2.datasus.gov.br/DATASUS/index.php?area=0203&id=30010028&VObj=http://tabnet.datasus.gov.br/cgi/deftohtm.exe?sinanwin/cnv/hanta; (accessed on 20 August 2020).
- Terças, A.C.P.; Atanaka-Santos, M.; Pignatti, M.G.; Espinosa, M.M.; de Melo Via, A.V.G.; Menegatti, J.A. Hantavirus pulmonary syndrome outbreak, Brazil, December 2009–January 2010. Emerg. Infect. Dis. 2013, 19, 1824–1827. [Google Scholar] [CrossRef] [PubMed]
- Terças, A.C.P.; Nascimento, V.F.; Hattori, T.Y.; Zenazokenae, L.E.; Atanaka, M.; Lemos, E.R.S. Clinical research in indigenous lands: The experience with Haliti-Paresi. Rev. Enferm. UFPE 2016, 10, 680–685. [Google Scholar]
- Terças, A.C.P.; Espinosa, M.M.; Santos, M.A. Fatores associados ao óbito por sindrome cardiopulmonar por hantavírus em Mato Grosso, Brasil. In Saúde Coletiva: Múltiplos Olhares em Pesquisa, 1st ed.; Guimarães, L.V., Pignatti, M.G., Eds.; EdUFMT: Cuiabá, Brazil, 2012; pp. 309–325. [Google Scholar]
- Oliveira, S.V.; Fonseca, L.X.; Vilges, K.M.A.; Maniglia, F.V.P.; Pereira, S.V.C.; Caldas, E.P.; Tauil, P.L.; Gurgel-Goncalves, R. Vulnerability of Brazilian municipalities to hantavirus infections based on multi-criteria decision analysis. Emerg. Themes Epidemiol. 2015, 12, 15. [Google Scholar] [CrossRef] [PubMed]
- Terças, A.C.P.; Nascimento, V.F.; Hattori, T.Y.; Zenazokenae, L.E.; Atanaka, M.; Lemos, E.R.S. Os Haliti-Paresí: Uma reflexão sobre saúde e demografia da população residente nas terras indígenas Paresí. Revista Espaço Ameríndio 2016, 10, 226–253. [Google Scholar] [CrossRef][Green Version]
- Figueiredo, L.T.M.; Moreli, M.L.; Borges, A.A.; de Figueiredo, G.G.; Badra, S.J.; Bisordi, I.; Suzuki, A.; Capria, S.; Padula, P. Evaluation of an enzyme-linked immunosorbent assay based on Araraquara virus recombinant nucleocapsid protein. Am. J. Trop. Med. Hyg. 2009, 81, 273–276. [Google Scholar] [CrossRef]
- Brasil. Instituto Brasileiro de Geografia e Estatística (IBGE). Os Indígenas no Censo Demográfico 2010: Primeiras Considerações Com Base no Quesito cor ou Raça; Ministério do Planejamento, Orçamento e Gestão, Instituto Brasileiro de Geografia e Estatística, Diretoria de Pesquisas: Rio de Janeiro, Brazil, 2012.
- Mascarenhas-Batista, A.V.; da Rosa, E.S.; Ksiazek, T.G.; da Rosa, A.P.; Leduc, J.W.; Pinheiro, F.; Tavares-Neto, J. Anti-hantavirus antibodies in school children in Salvador, Bahia. Rev. Soc. Bras. Med. Trop. 1998, 31, 433–440. [Google Scholar] [CrossRef]
- Figueiredo, L.T.; Moreli, M.L.; de-Sousa, R.L.; Borges, A.A.; de-Figueiredo, G.G.; Machado, A.M.; Bisordi, I.; Nagasse-Sugahara, T.K.; Suzuki, A.; Pereira, L.E.; et al. Hantavirus pulmonary syndrome, central plateau, southeastern, and southern Brazil. Emerg. Infect. Dis. 2009, 15, 561–567. [Google Scholar] [CrossRef]
- Mendes, W.S.; da Silva, A.A.; Neiva, R.F.; Costa, N.M.; de Assis, M.S.; Vidigal, P.M.; Leite, M.; da Rosa, E.S.; Medeiros, D.B.; Simith, D.; et al. Serologic survey of hantavirus infection, Brazilian Amazon. Emerg. Infect. Dis. 2010, 16, 889–891. [Google Scholar] [CrossRef]
- Souza, W.M.D.; Machado, A.M.; Figueiredo, L.T.M.; Boff, E. Serosurvey of hantavirus infection in humans in the border region between Brazil and Argentina. Rev. Soc. Bras. Med. Trop. 2011, 44, 131–135. [Google Scholar] [CrossRef][Green Version]
- Pereira, G.W.; Teixeira, A.M.; Souza, M.S.D.; Braga, A.D.; Junior, S.; Figueiredo, G.G.D.; Figueiredo, L.T.M.; Borges, A.A. Prevalence of serum antibodies to hantavirus in a rural population from the southern state of Santa Catarina, Brazil. Rev. Soc. Bras. Med. Trop. 2012, 45, 117–119. [Google Scholar] [CrossRef]
- Gimaque, J.B.L.; Bastos, M.D.S.; Braga, W.S.M.; Oliveira, C.M.C.D.; Castilho, M.D.C.; Figueiredo, R.M.P.D.; Mourão, M.P.G. Serological evidence of hantavirus infection in rural and urban regions in the state of Amazonas, Brazil. Mem. Inst. Oswal. Cruz. 2012, 107, 135–137. [Google Scholar] [CrossRef]
- Santos, I.O.; Figueiredo, G.G.D.; Figueiredo, L.T.M.; Azevedo, M.R.A.D.; Novo, N.F.; Vaz, C.A.C. Serologic survey of hantavirus in a rural population from the northern State of Mato Grosso, Brazil. Rev. Soc. Bras. Med. Trop. 2013, 46, 30–33. [Google Scholar] [CrossRef]
- Muñoz-Zanzi, C.; Saavedra, F.; Otth, C.; Domancich, L.; Hott, M.; Padula, P. Serological evidence of hantavirus infection in apparently healthy people from rural and slum communities in southern Chile. Viruses 2015, 7, 2006–2013. [Google Scholar] [CrossRef]
- Terças-Trettel, A.C.P.; Oliveira, E.C.; Fontes, C.J.F.; Melo, A.V.G.; Oliveira, R.C.; Guterres, A.; Fernandes, J.; Silva, R.G.; Atanaka, M.; Espinosa, M.M.; et al. Malaria and Hantavirus Pulmonary Syndrome in Gold Mining in the Amazon Region, Brazil. Int. J. Environ. Res. Public Health 2019, 16, 1852. [Google Scholar] [CrossRef]
- Campos, G.M.; Moro de Sousa, R.L.; Badra, S.J.; Pane, C.; Gomes, U.A.; Figueiredo, L.T. Serological survey of hantavirus in Jardinópolis County, Brazil. J. Med. Virol. 2003, 71, 417–422. [Google Scholar] [CrossRef]
- Zenazokenae, L.E.; Terças-Trettel, A.C.P.; Nascimento, V.; Hattori, T.Y.; Atanaka, M.; Lemos, E.; Bóia, M. Prevalence of enteroparasitosis in the indigenous community of Mato Grosso, Brazil: A look into the sanitation and ethno-development. Saúde e Pesquisa 2019, 12, 253–264. [Google Scholar] [CrossRef]
- Schatzmayr, H.G. Viroses emergentes e reemergentes. Cad Saúde Pública 2001, 17, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.A.C. A questão ambiental na promoção da saúde: Uma oportunidade de ação multiprofissional sobre doenças emergentes. Physis 2007, 17, 373–392. [Google Scholar] [CrossRef]
- Oliveira, R.C.; Guterres, A.; Fernandes, J.; D’Andrea, P.S.; Bonvicino, C.R.; Lemos, E.R.S. Hantavirus reservoirs: Current status with an emphasis on data from Brazil. Viruses 2014, 6, 1929–1973. [Google Scholar] [CrossRef] [PubMed]
- Rocha, C.R.; Ribeiro, R.; Marinho-Filho, J. Influence of temporal variation and seasonality on population dynamics of three sympatric rodents. Mamm. Biol. 2017, 84, 20–29. [Google Scholar] [CrossRef]
- Pignatti, M.G. Saúde e Ambiente: As doenças emergentes no Brasil. Rev. Ambient. Soc. 2004, VII, 133–148. [Google Scholar] [CrossRef]
- O’Shea, T.J.; Neubaum, D.J.; Neubaum, M.A.; Cryan, P.M.; Ellison, L.E.; Stanley, T.R.; Rupprecht, C.E.; Pape, W.J.; Bowen, R.A. Bat ecology and public health surveillance for rabies in an urbanizing region of Colorado. Urban Ecosyst. 2011, 14, 665–697. [Google Scholar] [CrossRef]
| Variables | Total Population | Anti-Hantavirus IgG+ Indigenous People | |||
|---|---|---|---|---|---|
| N | % | N | N | ||
| Gender | Male | 155 | 51.5 | 16 | 45.7 |
| Female | 146 | 48.5 | 19 | 54.3 | |
| Education | No education | 22 | 7.3 | 1 | 2.9 |
| Not of school age | 18 | 5.9 | 2 | 5.7 | |
| Kindergarten | 5 | 1.7 | - | - | |
| Elementary school | 165 | 54.8 | 20 | 57.1 | |
| High school | 77 | 25.6 | 10 | 28.6 | |
| Higher education | 14 | 4.7 | 2 | 5.7 | |
| Ethnicity | Paresí | 269 | 89.4 | 31 | 88.6 |
| Non-Indigenous people | 11 | 3.7 | 2 | 5.7 | |
| Other Indigenous ethnicity | 21 | 6.9 | 2 | 5.7 | |
| Village of residence | Bacaval | 63 | 20.9 | 5 | 14.3 |
| Wazare | 56 | 18.6 | 13 | 37.2 | |
| Bacaiuval | 48 | 15.9 | 3 | 8.6 | |
| Utiariti | 39 | 13 | - | - | |
| Seringal/Cabeceira do Seringal | 29 | 9.6 | 1 | 2.9 | |
| Chapada Azul | 27 | 9 | 11 | 31.4 | |
| Quatro Cachoeiras | 22 | 7.3 | - | - | |
| Sacre 2 | 16 | 2.3 | 2 | 5.7 | |
| Morrim | 1 | 0.3 | - | - | |
| Type of habitation | Wood | 119 | 39.5 | 11 | 31.4 |
| Traditional Indigenous housing | 97 | 39.5 | 14 | 40 | |
| Brickwork | 85 | 28.2 | 10 | 28.6 | |
| Variables | Total Population | Anti-Hantavirus IgG+ Indigenous People | |||
|---|---|---|---|---|---|
| N | % | N | % | ||
| Contact with wild rodents | No | 163 | 54.2 | 22 | 62.9 |
| Yes | 138 | 45.8 | 13 | 37.1 | |
| - Inside the house | 42 | 30.4 | 7 | 53.9 | |
| - In the village | 24 | 17.4 | 5 | 38.5 | |
| - On the farm | 15 | 10.9 | 1 | 7.7 | |
| - On the soy/corn plantation | 7 | 5.1 | 2 | 15.4 | |
| - Other sites | 37 | 26.8 | 1 | 7.7 | |
| Contact with other wild animals | No | 155 | 51.5 | 8 | 22.9 |
| Yes | 146 | 48.5 | 27 | 77.1 | |
| Signs and symptoms reported 60 days before serum collection | Headache | 69 | 22.9 | 5 | 14.3 |
| Fever | 53 | 17.6 | 5 | 14.3 | |
| Nausea | 29 | 9.6 | 1 | 2.9 | |
| Diarrhea | 24 | 8 | 3 | 6.6 | |
| Myalgia | 23 | 7.6 | 7 | 20 | |
| Abdominal pain | 22 | 7.3 | 2 | 5.7 | |
| Dizziness | 15 | 5 | 2 | 5.7 | |
| Low back pain | 14 | 4.7 | 4 | 11.4 | |
| Dyspnea | 13 | 4.3 | 2 | 5.7 | |
| Asthenia | 11 | 3.7 | - | - | |
| Cough | 10 | 3.3 | - | - | |
| Hypotension | 6 | 2 | - | - | |
| Chest pain | 5 | 1.7 | 1 | 2.9 | |
| Other diseases in the past 60 days | Influenza | 3 | 0.9 | - | - |
| Dengue | 1 | 0.3 | - | - | |
| Cutaneous Leishmaniosis | 1 | 0.3 | - | - | |
| Zika virus | 1 | 0.3 | - | - | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terças-Trettel, A.C.P.; de Melo, A.V.G.; de Oliveira, R.C.; Guterres, A.; Fernandes, J.; Pereira, L.S.; Atanaka, M.; Espinosa, M.M.; Teixeira, B.R.; Bonvicino, C.R.; et al. Orthohantavirus Survey in Indigenous Lands in a Savannah-Like Biome, Brazil. Viruses 2021, 13, 1122. https://doi.org/10.3390/v13061122
Terças-Trettel ACP, de Melo AVG, de Oliveira RC, Guterres A, Fernandes J, Pereira LS, Atanaka M, Espinosa MM, Teixeira BR, Bonvicino CR, et al. Orthohantavirus Survey in Indigenous Lands in a Savannah-Like Biome, Brazil. Viruses. 2021; 13(6):1122. https://doi.org/10.3390/v13061122
Chicago/Turabian StyleTerças-Trettel, Ana Cláudia Pereira, Alba Valéria Gomes de Melo, Renata Carvalho de Oliveira, Alexandro Guterres, Jorlan Fernandes, Liana Stretch Pereira, Marina Atanaka, Mariano Martinez Espinosa, Bernardo Rodrigues Teixeira, Cibele Rodrigues Bonvicino, and et al. 2021. "Orthohantavirus Survey in Indigenous Lands in a Savannah-Like Biome, Brazil" Viruses 13, no. 6: 1122. https://doi.org/10.3390/v13061122
APA StyleTerças-Trettel, A. C. P., de Melo, A. V. G., de Oliveira, R. C., Guterres, A., Fernandes, J., Pereira, L. S., Atanaka, M., Espinosa, M. M., Teixeira, B. R., Bonvicino, C. R., D’Andrea, P. S., & de Lemos, E. R. S. (2021). Orthohantavirus Survey in Indigenous Lands in a Savannah-Like Biome, Brazil. Viruses, 13(6), 1122. https://doi.org/10.3390/v13061122






