2A and 2A-like Sequences: Distribution in Different Virus Species and Applications in Biotechnology
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. 2A/2A-Like Distribution on Viruses
3.2. pssRNA Viruses
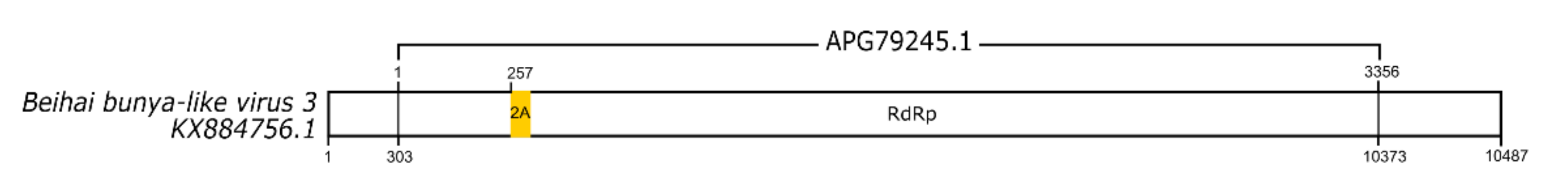
3.3. dsRNA Viruses
3.4. nssRNA Virus
3.5. 2A/2A-Likes Sequences and Viral Evolution
3.6. Biotechnology Applications
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Donnelly, M.L.L.; Hughes, L.E.; Luke, G.; Mendoza, H.; Ten Dam, E.; Gani, D.; Ryan, M.D. The “cleavage” activities of foot-and-mouth disease virus 2A site-directed mutants and naturally occurring “2A-like” sequences. J. Gen. Virol. 2001, 82, 1027–1041. [Google Scholar] [CrossRef]
- Luke, G.A.; de Felipe, P.; Lukashev, A.; Kallioinen, S.E.; Bruno, E.A.; Ryan, M.D. Occurrence, function and evolutionary origins of ‘2A-like’ sequences in virus genomes. J. Gen. Virol. 2008, 89, 1036–1042. [Google Scholar] [CrossRef]
- Ryan, M.D.; Donnelly, M.; Lewis, A.; Mehrotra, A.P.; Wilkie, J.; Gani, D. A model for nonstoichiometric, cotranslational protein scission in eukaryotic ribosomes. Bioorg. Chem. 1999, 27, 55–79. [Google Scholar] [CrossRef] [Green Version]
- Donnelly, M.L.L.; Luke, G.; Mehrotra, A.; Li, X.; Hughes, L.E.; Gani, D.; Ryan, M.D. Analysis of the aphthovirus 2A/2B polyprotein “cleavage” mechanism indicates not a proteolytic reaction, but a novel translational effect: A putative ribosomal “skip”. J. Gen. Virol. 2001, 82, 1013–1025. [Google Scholar] [CrossRef]
- Atkins, J.F.; Wills, N.M.; Loughran, G.; Wu, C.Y.; Parsawar, K.; Ryan, M.D.; Wang, C.H.; Nelson, C.C. A case for “StopGo”: Reprogramming translation to augment codon meaning of GGN by promoting unconventional termination (Stop) after addition of glycine and then allowing continued translation (Go). RNA 2007, 13, 803–810. [Google Scholar] [CrossRef] [Green Version]
- Brown, J.D.; Ryan, M.D. Ribosome “Skipping”: “Stop-Carry On” or “StopGo” Translation. In Recoding: Expansion of Decoding Rules Enriches Gene Expression; Atkins, J.F., Gesteland, R.F., Eds.; Springer: New York, NY, USA, 2010; pp. 101–121. [Google Scholar]
- de Felipe, P. Skipping the co-expression problem: The new 2A “CHYSEL” technology. Genet. Vaccines Ther. 2004, 2, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ryan, M.D.; King, A.M.Q.; Thomas, G.P. Cleavage of foot-and-mouth disease virus polyprotein is mediated by residues located within a 19 amino acid sequence. J. Gen. Virol. 1991, 72, 2727–2732. [Google Scholar] [CrossRef]
- Ryan, M.D.; Drew, J. Foot-and-mouth disease virus 2A oligopeptide mediated cleavage of an artificial polyprotein. EMBO J. 1994, 13, 928–933. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, M.L.L.; Gani, D.; Flint, M.; Monaghan, S.; Ryan, M.D. The cleavage activities of aphthovirus and cardiovirus 2A proteins. J. Gen. Virol. 1997, 78, 13–21. [Google Scholar] [CrossRef]
- Nibert, M.L. “2A-like” and “shifty heptamer” motifs in penaeid shrimp infectious myonecrosis virus, a monosegmented double-stranded RNA virus. J. Gen. Virol. 2007, 88, 1315–1318. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.E.; Brameld, J.M.; Hill, P.; Barrett, P.; Ebling, F.J.P.; Jethwa, P.H. The use of a viral 2A sequence for the simultaneous over-expression of both the vgf gene and enhanced green fluorescent protein (eGFP) in vitro and in vivo. J. Neurosci. Methods 2015, 256, 22–29. [Google Scholar] [CrossRef]
- Doronina, V.A.; de Felipe, P.; Wu, C.; Sharma, P.; Sachs, M.S.; Ryan, M.D.; Brown, J.D. Dissection of a co-translational nascent chain separation event. Biochem. Soc. Trans. 2008, 36, 712–716. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Cheng, A.; Wang, M.; Jia, R.; Sun, K.; Pan, K.; Yang, Q.; Wu, Y.; Zhu, D.; Chen, S.; et al. Structures and corresponding functions of five types of picornaviral 2A proteins. Front. Microbiol. 2017, 8, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Luke, G.A.; Escuin, H.; De Felipe, P.; Ryan, M. 2A to the Fore–Research, Technology and Applications 2A to the Fore–Research, Technology and Applications. Biotechnol. Genet. Eng. Rev. 2009, 26, 223–260. [Google Scholar] [CrossRef] [PubMed]
- Luke, G.A.; Pathania, U.S.; Roulston, C.; De Felipe, P.; Ryan, M.D. DxExNPGP-Motives for the motif. In Recent Research Developments in Virology; Research Signpost: Kerala, India, 2014; Volume 9, pp. 25–42. [Google Scholar]
- Luke, G.A.; Ryan, M.D. The protein coexpression problem in biotechnology and biomedicine: Virus 2A and 2A-like sequences provide a solution. Future Virol. 2013, 8, 983–996. [Google Scholar] [CrossRef] [Green Version]
- Gorbalenya, A.E.; Pringle, F.M.; Zeddam, J.L.; Luke, B.T.; Cameron, C.E.; Kalmakoff, J.; Hanzlik, T.N.; Gordon, K.H.J.; Ward, V.K. The palm subdomain-based active site is internally permuted in viral RNA-dependent RNA polymerases of an ancient lineage. J. Mol. Biol. 2002, 324, 47–62. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, J.; Lu, J.; Yi, F.; Liu, C.; Hu, Y. Sequence analysis and genomic organization of a new insect picorna-like virus, Ectropis obliqua picorna-like virus, isolated from Ectropis obliqua. J. Gen. Virol. 2004, 85, 1145–1151. [Google Scholar] [CrossRef]
- Wu, C.Y.; Lo, C.F.; Huang, C.J.; Yu, H.T.; Wang, C.H. The complete genome sequence of Perina nuda picorna-like virus, an insect-infecting RNA virus with a genome organization similar to that of the mammalian picornaviruses. Virology 2002, 294, 312–323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, F.; Browning, G.F.; Studdert, M.J.; Crabb, B.S. Equine rhinovirus 1 is more closely related to foot-and-mouth disease virus than to other picornaviruses. Proc. Natl. Acad. Sci. USA 1996, 93, 990–995. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wutz, G.; Auer, H.; Nowotny, N.; Grosse, B.; Skern, T.; Kuechler, E. Equine rhinovirus serotypes 1 and 2: Relationship to each other and to aphthoviruses and cardioviruses. J. Gen. Virol. 1996, 77, 1719–1730. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, A.M.; Johansson, S. Phylogenetic analysis of Ljungan virus and A-2 plaque virus, new members of the Picornaviridae. Virus Res. 2002, 85, 61–70. [Google Scholar] [CrossRef]
- Doherty, M.; Todd, D.; McFerran, N.; Hoey, E.M. Sequence analysis of a porcine enterovirus serotype 1 isolate: Relationships with other picornaviruses. J. Gen. Virol. 1999, 80, 1929–1941. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.S.; Lukashov, V.V.; Ganac, R.D.; Schnurr, D.P. Discovery of a novel human picornavirus in a stool sample from a pediatric patient presenting with fever of unknown origin. J. Clin. Microbiol. 2007, 45, 2144–2150. [Google Scholar] [CrossRef] [Green Version]
- Hagiwara, K.; Kobayashi, J.; Tomita, M.; Yoshimura, T. Nucleotide sequence of genome segment 5 from Bombyx mori cypovirus 1. Arch. Virol. 2001, 146, 181–187. [Google Scholar] [CrossRef]
- Yang, H.; Makeyev, E.V.; Kang, Z.; Ji, S.; Bamford, D.H.; Van Dijk, A.A. Cloning and sequence analysis of dsRNA segments 5, 6 and 7 of a novel non-group A, B, C adult rotavirus that caused an outbreak of gastroenteritis in China. Virus Res. 2004, 106, 15–26. [Google Scholar] [CrossRef]
- Zell, R.; Delwart, E.; Gorbalenya, A.E.; Hovi, T.; King, A.M.Q.; Knowles, N.J.; Lindberg, A.M.; Pallansch, M.A.; Palmenberg, A.C.; Reuter, G.; et al. ICTV virus taxonomy profile: Picornaviridae. J. Gen. Virol. 2017, 98, 2421–2422. [Google Scholar] [CrossRef] [PubMed]
- Hughes, P.J.; Stanway, G. The 2A proteins of three diverse picornaviruses are related to each other and to the H-rev107 family of proteins involved in the control of cell proliferation. J. Gen. Virol. 2000, 81, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Walter, C.T.; Pringle, F.M.; Nakayinga, R.; De Felipe, P.; Ryan, M.D.; Ball, L.A.; Dorrington, R.A. Genome organization and translation products of Providence virus: Insight into a unique tetravirus. J. Gen. Virol. 2010, 91, 2826–2835. [Google Scholar] [CrossRef] [PubMed]
- de Lima, J.G.S.; Teixeira, D.G.; Freitas, T.T.; Lima, J.P.M.S.; Lanza, D.C.F. Evolutionary origin of 2A-like sequences in Totiviridae genomes. Virus Res. 2019, 259, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Poulos, B.T.; Tang, K.F.J.; Pantoja, C.R.; Bonami, J.R.; Lightner, D.V. Purification and characterization of infectious myonecrosis virus of penaeid shrimp. J. Gen. Virol. 2006, 87, 987–996. [Google Scholar] [CrossRef]
- Zhai, Y.; Attoui, H.; Mohd Jaafar, F.; Wang, H.-Q.; Cao, Y.-X.; Fan, S.-P.; Sun, Y.-X.; Liu, L.-D.; Mertens, P.P.C.; Meng, W.-S.; et al. Isolation and full-length sequence analysis of Armigeres subalbatus totivirus, the first totivirus isolate from mosquitoes representing a proposed novel genus (Artivirus) of the family Totiviridae. J. Gen. Virol. 2010, 91, 2836–2845. [Google Scholar] [CrossRef]
- Isawa, H.; Kuwata, R.; Hoshino, K.; Tsuda, Y.; Sakai, K.; Watanabe, S.; Nishimura, M.; Satho, T.; Kataoka, M.; Nagata, N.; et al. Identification and molecular characterization of a new nonsegmented double-stranded RNA virus isolated from Culex mosquitoes in Japan. Virus Res. 2011, 155, 147–155. [Google Scholar] [CrossRef]
- Wu, Q.; Luo, Y.; Lu, R.; Lau, N.; Lai, E.C.; Li, W.X.; Ding, S.W. Virus discovery by deep sequencing and assembly of virus-derived small silencing RNAs. Proc. Natl. Acad. Sci. USA 2010, 107, 1606–1611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Virol, A.; Mor, S.K.; Benjamin, N.; Phelps, D. Molecular detection of a novel totivirus from golden shiner (Notemigonus crysoleucas) baitfish in the USA. Arch. Virol. 2016. [Google Scholar] [CrossRef]
- Danielle, M.; Dantas, A.; Henrique, G.; Cavalcante, O.; Oliveira, R.A.C.C.; Lanza, D.C.F.F.; Dantas, M.D.A.; Cavalcante, G.H.O.; Oliveira, R.A.C.C.; Lanza, D.C.F.F. New insights about ORF1 coding regions support the proposition of a new genus comprising arthropod viruses in the family Totiviridae. Virus Res. 2016, 211, 159–164. [Google Scholar] [CrossRef] [Green Version]
- Modrow, S.; Falke, D.; Truyen, U.; Schätzl, H. How Do Mutations Lead to the Emergence of Novel Viruses? Springer: Berlin/Heidelberg, Germany, 2013; Volume 9783642207, ISBN 978-3-642-20717-4. [Google Scholar]
- Orton, R.J.; Wright, C.F.; King, D.P.; Haydon, D.T. Estimating viral bottleneck sizes for FMDV transmission within and between hosts and implications for the rate of viral evolution. Interface Focus 2019, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khodamoradi, S.; Shenagari, M.; Kheiri, M.T.; Sabahi, F.; Jamali, A.; Heidari, A.; Ashrafkhani, B. IRES-based co-expression of influenza virus conserved genes can promote synergistic antiviral effects both in vitro and in vivo. Arch. Virol. 2018, 163, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Akiyoshi, S.; Ishii, T.; Bai, Z.; Mombaerts, P. Subpopulations of vomeronasal sensory neurons with coordinated coexpression of type 2 vomeronasal receptor genes are differentially dependent on Vmn2r1. Eur. J. Neurosci. 2018, 47, 887–900. [Google Scholar] [CrossRef] [Green Version]
- Zhang, K.; Su, L.; Duan, X.; Liu, L.; Wu, J. High-level extracellular protein production in Bacillus subtilis using an optimized dual-promoter expression system. Microb. Cell Fact. 2017, 16, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bayat, H.; Hossienzadeh, S.; Pourmaleki, E.; Ahani, R.; Rahimpour, A. Evaluation of different vector design strategies for the expression of recombinant monoclonal antibody in CHO cells. Prep. Biochem. Biotechnol. 2018, 48, 160–164. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, W.; Ehrlich, I.; Wolff, S.B.E.; Michalski, A.M.; Wölfl, S.; Hasan, M.T.; Lüthi, A.; Sprengel, R. Faithful expression of multiple proteins via 2A-peptide self-processing: A versatile and reliable method for manipulating brain circuits. J. Neurosci. 2009, 29, 8621–8629. [Google Scholar] [CrossRef] [PubMed]
- Radcliffe, P.A.; Mitrophanous, K.A. Multiple gene products from a single vector: “Self-cleaving” 2A peptides. Gene Ther. 2004, 11, 1673–1674. [Google Scholar] [CrossRef]
- Arbab, A.S.; Yocum, G.T.; Wilson, L.B.; Parwana, A.; Jordan, E.K.; Kalish, H.; Frank, J.A. Comparison of transfection agents in forming complexes with ferumoxides, cell labeling efficiency, and cellular viability. Mol. Imaging 2004, 3, 24–32. [Google Scholar] [CrossRef] [PubMed]
- de Felipe, P.; Luke, G.A.; Hughes, L.E.; Gani, D.; Halpin, C.; Ryan, M.D. E unum pluribus: Multiple proteins from a self-processing polyprotein. Trends Biotechnol. 2006, 24, 68–75. [Google Scholar] [CrossRef]
- Hellen, C.U.T.; Sarnow, P. Internal ribosome entry sites in eukaryotic mRNA molecules. Genes Dev. 2001, 15, 1593–1612. [Google Scholar] [CrossRef] [Green Version]
- Bouabe, H.; Fässler, R.; Heesemann, J. Improvement of reporter activity by IRES-mediated polycistronic reporter system. Nucleic Acids Res. 2008, 36, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Momose, F.; Morikawa, Y. Polycistronic expression of the influenza A virus RNA-dependent RNA polymerase by using the Thosea asigna virus 2A-like self-processing sequence. Front. Microbiol. 2016, 7, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Sun, Y.F.; Lin, Y.; Zhang, J.H.; Zheng, S.P.; Ye, Y.R.; Liang, X.X.; Han, S.Y. Double Candida antarctica lipase B co-display on Pichia pastoris cell surface based on a self-processing foot-and-mouth disease virus 2A peptide. Appl. Microbiol. Biotechnol. 2012, 96, 1539–1550. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Yao, Q.; Tao, J.; Qiao, Y.; Zhang, Z. Co-ordinate expression of glycine betaine synthesis genes linked by the FMDV 2A region in a single open reading frame in Pichia pastoris. Appl. Microbiol. Biotechnol. 2007, 77, 891–899. [Google Scholar] [CrossRef]
- Subramanian, V.; Schuster, L.A.; Moore, K.T.; Ii, L.E.T.; Baker, J.O.; Wall, T.A.; Vander Linger, J.G.; Himmel, M.E.; Decker, S.R. Biotechnology for Biofuels A versatile 2A peptide - based bicistronic protein expressing platform for the industrial cellulase producing fungus, Trichoderma reesei. Biotechnol. Biofuels 2017, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Schuetze, T.; Meyer, V. Polycistronic gene expression in Aspergillus niger. Microb. Cell Fact. 2017, 16, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, F.; Liu, Q.; Li, X.; Zhang, C.; Li, J.; Sun, W.; Liu, D.; Xiao, D.; Tian, C. Construction of a new thermophilic fungus Myceliophthora thermophila platform for enzyme production using a versatile 2A peptide strategy combined with efficient CRISPR-Cas9 system. Biotechnol. Lett. 2020, 42, 1181–1191. [Google Scholar] [CrossRef]
- Osborn, M.J.; Panoskaltsis-Mortari, A.; McElmurry, R.T.; Bell, S.K.; Vignali, D.A.A.; Ryan, M.D.; Wilber, A.C.; McIvor, R.S.; Tolar, J.; Blazar, B.R. A picornaviral 2A-like sequence-based tricistronic vector allowing for high-level therapeutic gene expression coupled to a dual-reporter system. Mol. Ther. 2005, 12, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Schwirz, J.; Yan, Y.; Franta, Z.; Schetelig, M.F. Bicistronic expression and differential localization of proteins in insect cells and Drosophila suzukii using picornaviral 2A peptides. Insect Biochem. Mol. Biol. 2020, 119, 103324. [Google Scholar] [CrossRef]
- Zhang, B.; Rapolu, M.; Kumar, S.; Gupta, M.; Liang, Z.; Han, Z.; Williams, P.; Su, W.W. Coordinated protein co-expression in plants by harnessing the synergy between an intein and a viral 2A peptide. Plant Biotechnol. J. 2017, 15, 718–728. [Google Scholar] [CrossRef] [Green Version]
- de Felipe, P.; Hughes, L.E.; Ryan, M.D.; Brown, J.D. Co-translational, intraribosomal cleavage of polypeptides by the foot-and-mouth disease virus 2A peptide. J. Biol. Chem. 2003, 278, 11441–11448. [Google Scholar] [CrossRef] [Green Version]
- de Felipe, P.; Luke, G.A.; Brown, J.D.; Ryan, M.D. Inhibition of 2A-mediated “cleavage” of certain artificial polyproteins bearing N-terminal signal sequences. Biotechnol. J. 2010, 5, 213–223. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Wang, M.; Wang, T.; Wei, Y.; Guo, X.; Mi, C.; Zhao, C.; Cao, X.; Dou, Y. Effects of different 2A peptides on transgene expression mediated by tricistronic vectors in transfected CHO cells. Mol. Biol. Rep. 2020, 47, 469–475. [Google Scholar] [CrossRef]
- Shin, S.; Kim, S.H.; Shin, S.W.; Grav, L.M.; Pedersen, L.E.; Lee, J.S.; Lee, G.M. Comprehensive Analysis of Genomic Safe Harbors as Target Sites for Stable Expression of the Heterologous Gene in HEK293 Cells. ACS Synth. Biol. 2020, 9, 1263–1269. [Google Scholar] [CrossRef]
- Rasala, B.A.; Lee, P.A.; Shen, Z.; Briggs, S.P.; Mendez, M.; Mayfield, S.P. Robust expression and secretion of xylanase1 in Chlamydomonas reinhardtii by fusion to a selection gene and processing with the FMDV 2A peptide. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeong, I.; Kim, E.; Seong, J.Y.; Park, H.C. Overexpression of Spexin 1 in the Dorsal Habenula Reduces Anxiety in Zebrafish. Front. Neural Circuits 2019, 13, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Pontes-Quero, S.; Fernández-Chacón, M.; Luo, W.; Lunella, F.F.; Casquero-Garcia, V.; Garcia-Gonzalez, I.; Hermoso, A.; Rocha, S.F.; Bansal, M.; Benedito, R. High mitogenic stimulation arrests angiogenesis. Nat. Commun. 2019, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Wang, F.; Xu, S.; Wang, R.; Chen, W.; Hou, K.; Tian, C.; Wang, F.; Zhao, P.; Xia, Q. Optimization of a 2A self-cleaving peptide-based multigene expression system for efficient expression of upstream and downstream genes in silkworm. Mol. Genet. Genomics 2019, 294, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Beekwilder, J.; Van Rossum, H.M.; Koopman, F.; Sonntag, F.; Buchhaupt, M.; Schrader, J.; Hall, R.D.; Bosch, D.; Pronk, J.T.; Van Maris, A.J.A.; et al. Polycistronic expression of a β-carotene biosynthetic pathway in Saccharomyces cerevisiae coupled to β-ionone production. J. Biotechnol. 2014, 192, 383–392. [Google Scholar] [CrossRef]
- Park, M.; Kang, K.; Park, S.; Kim, Y.S.; Ha, S.H.; Lee, S.W.; Ahn, M.J.; Bae, J.M.; Back, K. Expression of serotonin derivative synthetic genes on a single self-processing polypeptide and the production of serotonin derivatives in microbes. Appl. Microbiol. Biotechnol. 2008, 81, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Geier, M.; Fauland, P.; Vogl, T.; Glieder, A. Compact multi-enzyme pathways in P. pastoris. Chem. Commun. 2015, 51, 1643–1646. [Google Scholar] [CrossRef] [Green Version]
- Park, S.; Kang, K.; Kim, Y.S.; Back, K. Endosperm-specific expression of tyramine N-hydroxycinnamoyltransferase and tyrosine decarboxylase from a single self-processing polypeptide produces high levels of tyramine derivatives in rice seeds. Biotechnol. Lett. 2009, 31, 911–915. [Google Scholar] [CrossRef]
- Quilis, J.; López-García, B.; Meynard, D.; Guiderdoni, E.; San Segundo, B. Inducible expression of a fusion gene encoding two proteinase inhibitors leads to insect and pathogen resistance in transgenic rice. Plant Biotechnol. J. 2014, 12, 367–377. [Google Scholar] [CrossRef]
- Yeo, E.T.; Kwon, H.B.; Han, S.E.; Lee, J.T.; Ryu, J.C.; Byun, M.O. Genetic engineering of drought resistant potato plants by introduction of the trehalose-6-phosphate synthase (TPS1) gene from Saccharomyces cerevisiae. Mol. Cells 2000, 10, 263–268. [Google Scholar]
- Ralley, L.; Enfissi, E.M.A.; Misawa, N.; Schuch, W.; Bramley, P.M.; Fraser, P.D. Metabolic engineering of ketocarotenoid formation in higher plants. Plant J. 2004, 39, 477–486. [Google Scholar] [CrossRef]
- Chu, V.T.; Weber, T.; Wefers, B.; Wurst, W.; Sander, S.; Rajewsky, K.; Kühn, R. Increasing the efficiency of homology-directed repair for CRISPR-Cas9-induced precise gene editing in mammalian cells. Nat. Biotechnol. 2015, 33, 543–548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Browne, E.P. An Interleukin-1 Beta-Encoding Retrovirus Exhibits Enhanced Replication In Vivo. J. Virol. 2015, 89, 155–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yi, G.; Choi, J.G.; Bharaj, P.; Abraham, S.; Dang, Y.; Kafri, T.; Alozie, O.; Manjunath, M.N.; Shankar, P. CCR5 gene editing of resting CD4+ T cells by transient ZFN expression from HIV envelope pseudotyped nonintegrating lentivirus confers HIV-1 resistance in humanized mice. Mol. Ther.-Nucleic Acids 2014, 3, 1–10. [Google Scholar] [CrossRef]
- Peng, Y.; Yang, T.; Tang, X.; Chen, F.; Wang, S. Construction of an Inducible CRISPR/Cas9 System for CXCR4 Gene and Demonstration of its Effects on MKN-45 Cells. Cell Biochem. Biophys. 2020, 78, 23–30. [Google Scholar] [CrossRef]
- Fang, Y.; Stroukov, W.; Cathomen, T.; Mussolino, C. Chimerization enables gene synthesis and lentiviral delivery of customizable tale-based effectors. Int. J. Mol. Sci. 2020, 21, 795. [Google Scholar] [CrossRef] [Green Version]
- Mizote, Y.; Masumi-Koizumi, K.; Katsuda, T.; Yamaji, H. Production of an antibody Fab fragment using 2A peptide in insect cells. J. Biosci. Bioeng. 2020, 130, 205–211. [Google Scholar] [CrossRef]
- Arevalo-Villalobos, J.I.; Govea-Alonso, D.O.; Bañuelos-Hernández, B.; González-Ortega, O.; Zarazúa, S.; Rosales-Mendoza, S. Inducible expression of antigens in plants: A study focused on peptides related to multiple sclerosis immunotherapy. J. Biotechnol. 2020, 318, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.H.; Hu, C.C.; Liao, J.T.; Lee, Y.L.; Huang, Y.W.; Lin, N.S.; Lin, Y.L.; Hsu, Y.H. Production of Japanese encephalitis virus antigens in plants using bamboo mosaic virus-based vector. Front. Microbiol. 2017, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Hung, C.Y.; Bhattacharya, C.; Nichols, S.; Rahimuddin, H.; Kittur, F.S.; Leung, T.C.; Xie, J. An effective way of producing fully assembled antibody in transgenic tobacco plants by linking heavy and light chains via a self-cleaving 2a peptide. Front. Plant Sci. 2018, 9, 1–11. [Google Scholar] [CrossRef]
- Chen, L.; Yang, X.; Luo, D.; Yu, W. Efficient Production of a Bioactive Bevacizumab monoclonal antibody using the 2A self-cleavage peptide in transgenic rice callus. Front. Plant Sci. 2016, 7, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Monreal-Escalante, E.; Bañuelos-Hernández, B.; Hernández, M.; Fragoso, G.; Garate, T.; Sciutto, E.; Rosales-Mendoza, S. Expression of Multiple Taenia Solium Immunogens in Plant Cells Through a Ribosomal Skip Mechanism. Mol. Biotechnol. 2015, 57, 635–643. [Google Scholar] [CrossRef]
- Jin, S.; Yang, C.; Huang, J.; Liu, L.; Zhang, Y.; Li, S.; Zhang, L.; Sun, Q.; Yang, P. Conditioned medium derived from FGF-2-modified GMSCs enhances migration and angiogenesis of human umbilical vein endothelial cells. Stem Cell Res. Ther. 2020, 11, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knoll, A.; Kankowski, S.; Schöllkopf, S.; Meier, J.C.; Seitz, O. Chemo-biological mRNA imaging with single nucleotide specificity. Chem. Commun. 2019, 55, 14817–14820. [Google Scholar] [CrossRef] [PubMed]
- Quenneville, S.; Labouèbe, G.; Basco, D.; Metref, S.; Viollet, B.; Foretz, M.; Thorens, B. Hypoglycemia Sensing Neurons of the Ventromedial Hypothalamus Require AMPK-Induced Txn2 Expression But Are Dispensable For Physiological Counterregulation. Diabetes 2020, db200577. [Google Scholar] [CrossRef]
- Shibuta, M.K.; Matsuoka, M.; Matsunaga, S. 2a Peptides Contribute To the Co-Expression of Proteins for Imaging and Genome Editing. Cytologia 2019, 84, 107–111. [Google Scholar] [CrossRef] [Green Version]
- Sinha, D.; Steyer, B.; Shahi, P.K.; Mueller, K.P.; Valiauga, R.; Edwards, K.L.; Bacig, C.; Steltzer, S.S.; Srinivasan, S.; Abdeen, A.; et al. Human iPSC Modeling Reveals Mutation-Specific Responses to Gene Therapy in a Genotypically Diverse Dominant Maculopathy. Am. J. Hum. Genet. 2020, 107, 278–292. [Google Scholar] [CrossRef] [PubMed]
- Kawano, F.; Okazaki, R.; Yazawa, M.; Sato, M. A photoactivatable Cre-loxP recombination system for optogenetic genome engineering. Nat. Chem. Biol. 2016, 12, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, V.V.; Khamo, J.S.; Mei, W.; Turgeon, A.J.; Ashraf, H.M.; Mondal, P.; Patel, D.B.; Risner, N.; Cho, E.E.; Yang, J.; et al. Reversible optogenetic control of kinase activity during differentiation and embryonic development. Development 2016, 143, 4085–4094. [Google Scholar] [CrossRef] [Green Version]
- Taslimi, A.; Zoltowski, B.; Miranda, J.G.; Pathak, G.P.; Hughes, R.M.; Tucker, C.L. Optimized second-generation CRY2-CIB dimerizers and photoactivatable Cre recombinase. Nat. Chem. Biol. 2016, 12, 425–430. [Google Scholar] [CrossRef] [Green Version]
- Luke, G.A.; Ryan, M.D. Therapeutic applications of the ‘NPGP’ family of viral 2As. Rev. Med. Virol. 2018, 28, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
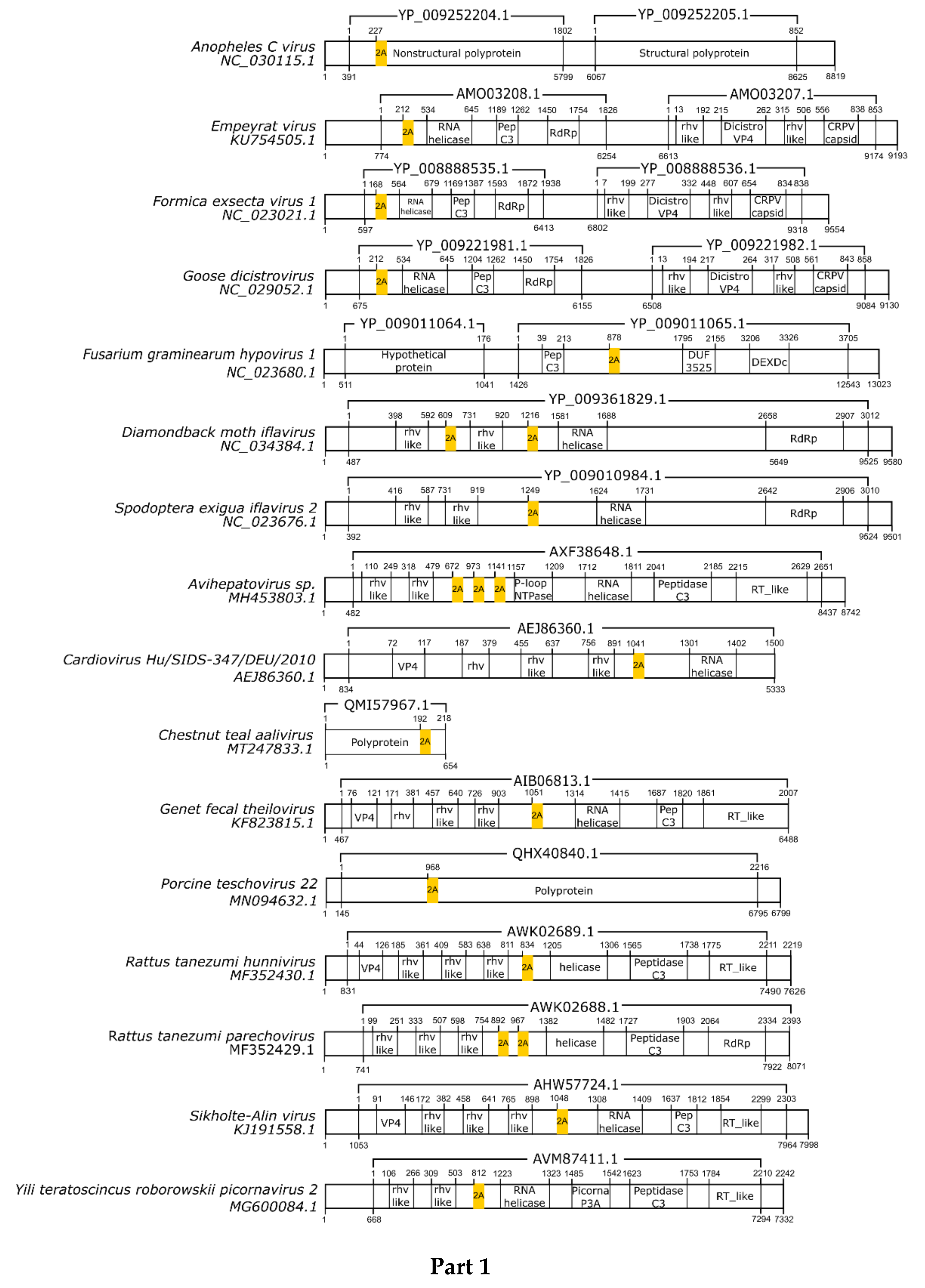
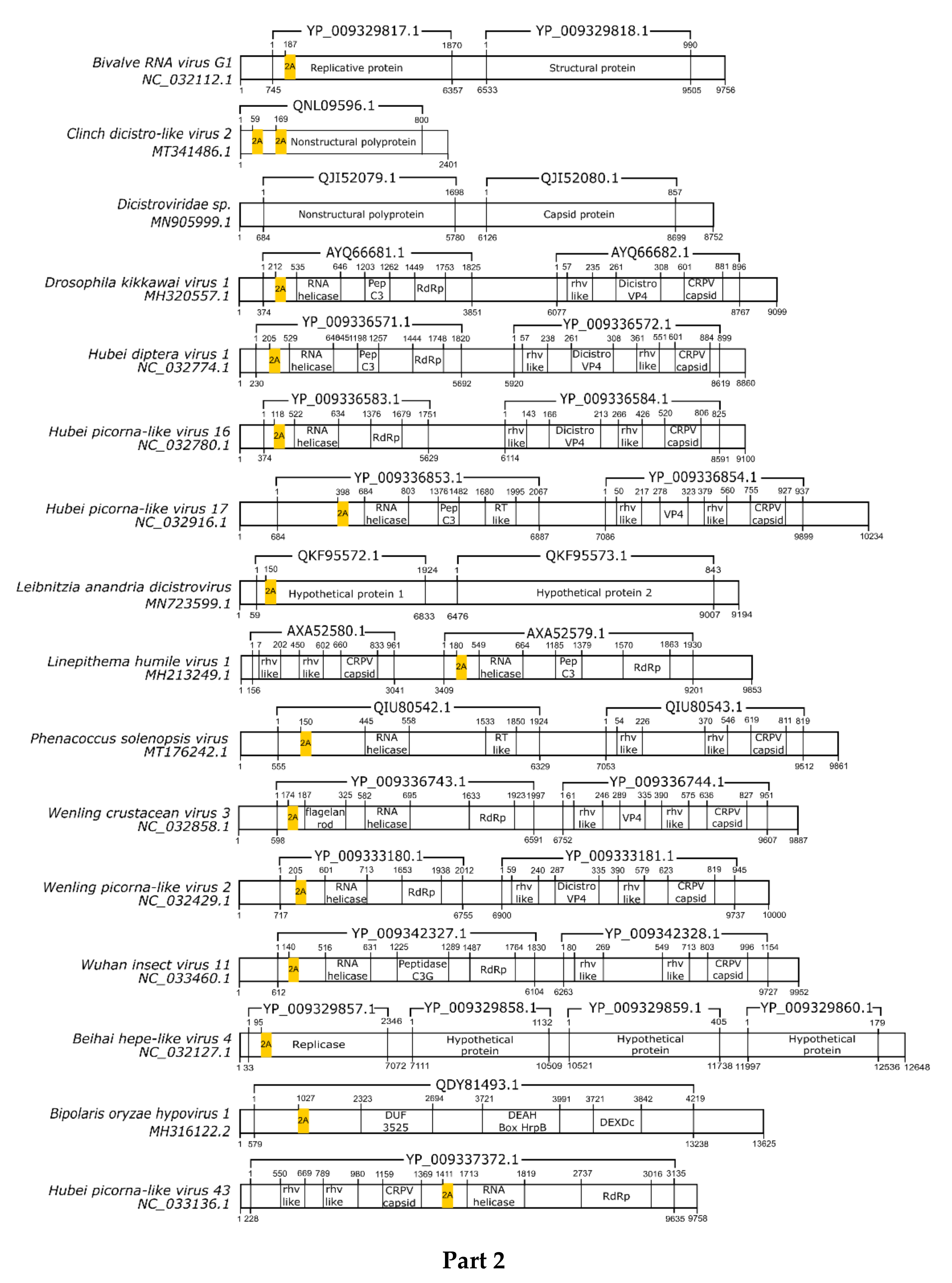
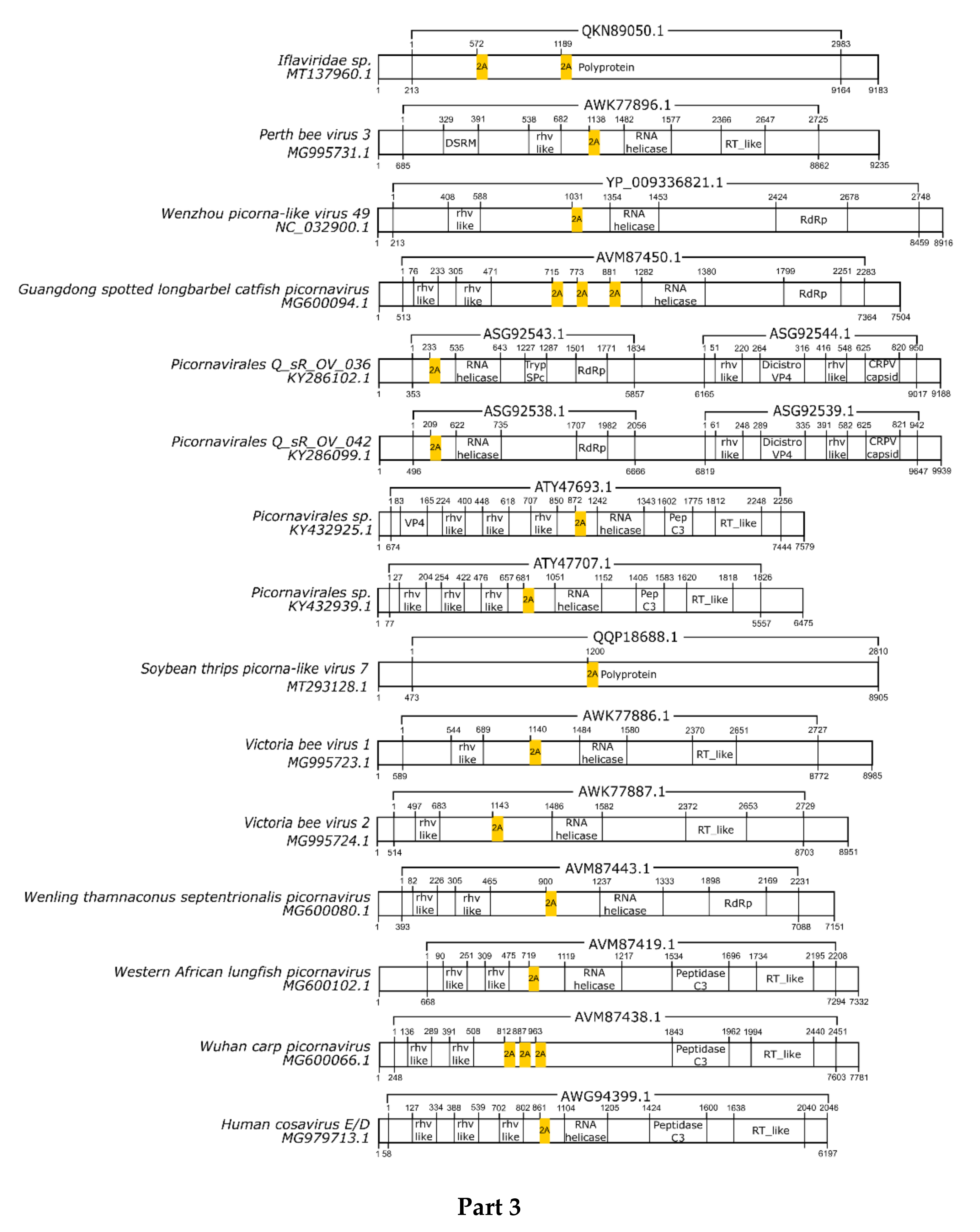

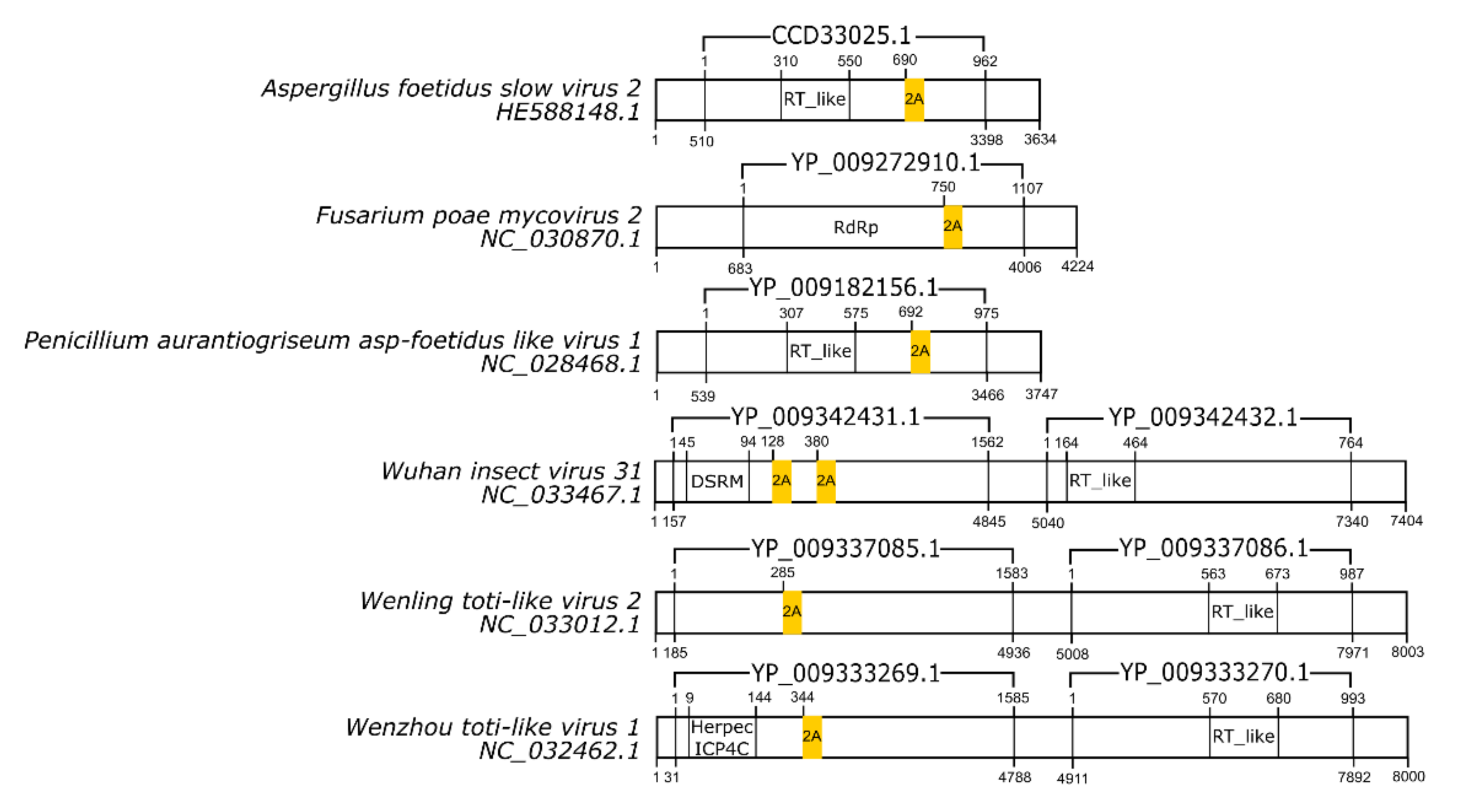
| Virus | Family | Motif | Cleavage Efficiency | References |
|---|---|---|---|---|
| Euprosterna elaeasa virus (EeV) | Alphatetraviridae | GDVEENPGP | ~99% | [2,18] |
| Providence virus (PrV) | Alphatetraviridae | GDVESNPGP | ~99% | [2] |
| Providence virus (PrV) | Alphatetraviridae | GDIEKNPGP | ~94% | [2] |
| Providence virus (PrV) | Alphatetraviridae | GDVEKNPGP | ~99% | [2] |
| Thosea asigna virus (TaV) | Alphatetraviridae | GDVEENPGP | ~99% | [1] |
| Acute bee paralysis virus (ABPV) | Dicistroviridae | GDVETNPGP | ~94% | [1,2] |
| Cricket paralysis virus (CrPV) | Dicistroviridae | GDVESNPGP | ~90% | [1,2] |
| Drosophila C virus (DCV) | Dicistroviridae | GDVETNPGP | ~95% | [1] |
| Ectropis oblique picorna-like virus (EoPV) | Iflaviridae | GDVESNPGP | ~99% | [2,19] |
| Ectropis oblique picorna-like virus (EoPV) | Iflaviridae | GDIESNPGP | ~99% | [2,19] |
| Infectious flacherie virus (IFV) | Iflaviridae | AGIESNPGP | ~99% | [1,2] |
| Perina nuda picorna-like virus (PnPV) | Iflaviridae | GDVESNPGP | ~99% | [2,20] |
| Perina nuda picorna-like virus (PnPV) | Iflaviridae | GDIESNPGP | ~99% | [2,20] |
| Encephalomyocarditis virus (EMCV) | Picornaviridae | HDIETNPGP | ~91% | [1,8] |
| Equine rhinitis A virus (ERAV) | Picornaviridae | GDVESNPGP | ~99% | [1,21] |
| Equine rhinitis B virus (ERBV-1) | Picornaviridae | GDVELNPGP | ~99% | [2,22] |
| Foot-and-mouth disease virus (FMDV) | Picornaviridae | GDVESNPGP | ~99% | [8,10] |
| Ljungan virus (LV) | Picornaviridae | GDVETNPGP | ~99% | [2,23] |
| Porcine teschovirus 1 (PTV-1) | Picornaviridae | GDVEENPGP | ~94% | [1,24] |
| Saffold virus (SAF-V) | Picornaviridae | HDVETNPGP | ~99% | [2,25] |
| Theiler’s murine encephalomyelitis virus (TMEV) | Picornaviridae | HDVEMNPGP | ~99% | [10] |
| Bombyx mori reoviridae 1 (BmCPV-1) | Reoviridae | GDIESNPGP | ~99% | [2,26] |
| Human reoviridae C (HurV-C) | Reoviridae | GDIELNPGP | ~82% | [2] |
| New adult diarrhea virus (ADRV-N) | Reoviridae | ECIESNPGP | ~97% | [2,27] |
| Operophtera brumata reoviridae 18 (OpbuCPV-18) | Reoviridae | GDVESNPGP | ~99% | [2] |
| Porcine reoviridae A (Porv-C) | Reoviridae | GDVELNPGP | ~89% | [1,2] |
| Infectious myonecrosis virus (IMNV) | Unassigned Totiviridae | GDVESNPGP | ~99% | [2,11] |
| Infectious myonecrosis virus (IMNV) | Unassigned Totiviridae | GDVEENPGP | ~99% | [2,11] |
| Accession Number | Virus | 2A Motif | Taxon |
|---|---|---|---|
| YP_003620399.1 | Providence virus—2A1 | GDVEKNPGP | Carmotetraviridae |
| Providence virus—2A2 | GDVESNPGP | ||
| Providence virus—2A3 | GDIEKNPGP | ||
| NP_066241.1 | Acute bee paralysis virus | GDVETNPGP | Dicistroviridae |
| YP_009252204.1 | Anopheles C virus | GDVELNPGP | Dicistroviridae |
| NP_647481.1 | Cricket paralysis virus | GDVESNPGP | Dicistroviridae |
| NP_044945.1 | Drosophila C virus | GDVETNPGP | Dicistroviridae |
| AMO03208.1 | Empeyrat virus | GDVELNPGP | Dicistroviridae |
| YP_008888535.1 | Formica exsecta virus 1 | GDIESNPGP | Dicistroviridae |
| YP_009221981.1 | Goose dicistrovirus | GDVELNPGP | Dicistroviridae |
| ASS83246.1 | Israeli acute paralysis virus | GDVEENPGP | Dicistroviridae |
| NP_851403.1 | Kashmir bee virus | GDIELNPGP | Dicistroviridae |
| YP_009011065.1 | Fusarium graminearum hypovirus 1 | HDVEKNPGP | Hypoviridae |
| YP_009361829.1 | Diamond back moth iflavirus—2A1 | GDVESNPGP | Iflaviridae |
| Diamond back moth iflavirus—2A2 | GDVESNPGP | ||
| NP_919029.1 | Ectropis obliqua picorna-like virus—2A1 | GDVESNPGP | Iflaviridae |
| Ectropis obliqua picorna-like virus—2A2 | GDIESNPGP | ||
| NP_277061.1 | Perina nuda virus—2A1 | GDVESNPGP | Iflaviridae |
| Perina nuda virus—2A2 | GDIESNPGP | ||
| YP_009010984.1 | Spodoptera exigua iflavirus 2 | GDVESNPGP | Iflaviridae |
| NP_573542.1 | Euprosterna elaeasa virus | GDVEENPGP | Permutotetraviridae |
| AAC97195.1 | Thosea asigna virus | GDVEENPGP | Permutotetraviridae |
| AXF38648.1 | Avihepatovirus sp.—2A1 | GDVESNPGP | Picornaviridae |
| Avihepatovirus sp.—2A2 | GDVESNPGP | ||
| Avihepatovirus sp.—2A3 | GDVEPNPGP | ||
| Avihepatovirus sp.—2A4 | GDVESNPGP | ||
| AUX16868.1 | Avisivirus AVE052/AsV | GDIEENPGP | Picornaviridae |
| YP_009345900.1 | Bat crohivirus | GDIESNPGP | Picornaviridae |
| YP_006607894.1 | Bluegill picornavirus—2A1 | GDVESNPGP | Picornaviridae |
| Bluegill picornavirus—2A2 | GDVEQNPGP | ||
| YP_006792625.1 | Bovine hungarovirus 1 | GDVELNPGP | Picornaviridae |
| YP_009116874.1 | Bovine picornavirus | GDIESNPGP | Picornaviridae |
| AQX17368.1 | Bovine rhinitis B virus | GDIESNPGP | Picornaviridae |
| ANN02879.1 | Bovine rhinitis B virus | GDIETNPGP | Picornaviridae |
| YP_009352243.1 | Bovine rhinovirus 1 | GDVETNPGP | Picornaviridae |
| QEQ92497.1 | Burpengary virus | GDVEQNPGP | Picornaviridae |
| ACG61138.2 | Cardiovirus D | HDIETNPGP | Picornaviridae |
| AEJ86360.1 | Cardiovirus Hu/SIDS-347/DEU/2010 | HDIETNPGP | Picornaviridae |
| YP_008992026.1 | Carp picornavirus 1—2A1 | GDVEQNPGP | Picornaviridae |
| Carp picornavirus 1—2A2 | GDVESNPGP | ||
| QMI57967.1 | Chestnut teal aalivirus | GDVEENPGP | Picornaviridae |
| YP_002956074.1 | Cosavirus A | GDIESNPGP | Picornaviridae |
| YP_002956076.1 | Cosavirus D | GDIETNPGP | Picornaviridae |
| YP_009361830.1 | Cosavirus F | GDVEENPGP | Picornaviridae |
| YP_009104360.1 | Crohivirus | GDIESNPGP | Picornaviridae |
| YP_009345900.1 | Crohivirus B | GDIESNPGP | Picornaviridae |
| YP_009026377.1 | Duck picornavirus GL/12—2A1 | GDVESNPGP | Picornaviridae |
| Duck picornavirus GL/12—2A2 | GDVEENPGP | ||
| Duck picornavirus GL/12—2A3 | GDVEMNPGP | ||
| Duck picornavirus GL/12—2A4 | GDIEQNPGP | ||
| AAA43035.1 | Encephalomyocarditis virus | HDIETNPGP | Picornaviridae |
| AKE44318.1 | Encephalomyocarditis virus | HDVETNPGP | Picornaviridae |
| AGU38152.1 | Encephalomyocarditis virus | HDVELNPGP | Picornaviridae |
| AFO66759.1 | Encephalomyocarditis virus type 2 | HDVETNPGP | Picornaviridae |
| NP_653077.1 | Equine rhinitis B virus 1 | GDVELNPGP | Picornaviridae |
| ANJ20934.1 | Equine rhinitis B virus 2 | GDVESNPGP | Picornaviridae |
| ANJ20932.1 | Erbovirus A | GDVESNPGP | Picornaviridae |
| ANJ20933.1 | Erbovirus A | GDVELNPGP | Picornaviridae |
| YP_009423853.1 | Falcon picornavirus—2A1 | GDVEENPGP | Picornaviridae |
| Falcon picornavirus—2A2 | GDVELNPGP | ||
| AHL26986.1 | Fathead minnow picornavirus—2A1 | GDVEQNPGP | Picornaviridae |
| Fathead minnow picornavirus—2A2 | GDVESNPGP | ||
| AYJ71467.2 | Feline hunnivirus | GDVELNPGP | Picornaviridae |
| AAT01719.1 | Foot-and-mouth disease virus—type A | GDVESNPGP | Picornaviridae |
| AFM56034.1 | Foot-and-mouth disease virus—type O | GDVESNPGP | Picornaviridae |
| AAT01787.1 | Foot-and-mouth disease virus—type SAT 1 | GDVESNPGP | Picornaviridae |
| AFE84748.1 | Foot-and-mouth disease virus—type SAT 2 | GDVESNPGP | Picornaviridae |
| AAT01795.1 | Foot-and-mouth disease virus—type SAT 3 | GDVESNPGP | Picornaviridae |
| AIB06813.1 | Genet fecal theilovirus | HDVEMNPGP | Picornaviridae |
| YP_009026376.1 | Human cosavirus | GDIETNPGP | Picornaviridae |
| AFJ04537.1 | Human cosavirus A20 | GDIESNPGP | Picornaviridae |
| YP_002956075.1 | Human cosavirus B | HDIETNPGP | Picornaviridae |
| ADF28539.1 | Human TMEV-like cardiovirus | HDIETNPGP | Picornaviridae |
| AMT85188.1 | Hunnivirus | GDVEENPGP | Picornaviridae |
| YP_009118270.1 | Lesavirus 2 | GDIEPNPGP | Picornaviridae |
| ACJ48052.1 | Ljungan virus | GDVEENPGP | Picornaviridae |
| AVX29482.1 | Marmot mosavirus—2A1 | GDVETNPGP | Picornaviridae |
| Marmot mosavirus—2A2 | GDVETNPGP | ||
| ANX14418.1 | Mengo virus | HDVETNPGP | Picornaviridae |
| YP_009361319.1 | Miniopterus schreibersii picornavirus 1 | GDVEENPGP | Picornaviridae |
| AWC68493.1 | Mischivirus B | GDIEENPGP | Picornaviridae |
| YP_009026384.1 | Mosavirus A2 | GDVESNPGP | Picornaviridae |
| YP_009109563.1 | Norway rat hunnivirus | GDVELNPGP | Picornaviridae |
| ADO85550.2 | Ovine hungarovirus | GDVELNPGP | Picornaviridae |
| AIU94297.1 | Pasivirus A | GDVEQNPGP | Picornaviridae |
| SNQ28005.1 | Pasivirus A | GDIEQNPGP | Picornaviridae |
| APA29021.1 | Picornaviridae sp. rodent | GDVELNPGP | Picornaviridae |
| ADN52625.1 | Porcine encephalomyocarditis virus | HDIETNPGP | Picornaviridae |
| AAK12398.1 | Porcine teschovirus 1 | GDVEENPGP | Picornaviridae |
| AAK12413.1 | Porcine teschovirus 10 | GDVEENPGP | Picornaviridae |
| AAK12390.1 | Porcine teschovirus 11 | GDVEENPGP | Picornaviridae |
| AAK12381.1 | Porcine teschovirus 2 | GDVEENPGP | Picornaviridae |
| AAK12382.1 | Porcine teschovirus 3 | GDVEENPGP | Picornaviridae |
| AGB67759.1 | Porcine teschovirus 4 | GDVEENPGP | Picornaviridae |
| ACT66681.1 | Porcine teschovirus 5 | GDVEENPGP | Picornaviridae |
| AAK12409.1 | Porcine teschovirus 6 | GDVEENPGP | Picornaviridae |
| AAK12386.1 | Porcine teschovirus 7 | GDVEENPGP | Picornaviridae |
| AAK12388.1 | Porcine teschovirus 9 | GDVEENPGP | Picornaviridae |
| QHX40840.1 | Porcine teschovirus 22 | GDIEENPGP | Picornaviridae |
| ACD67870.1 | Rat theilovirus 1 | HDVETNPGP | Picornaviridae |
| AWK02689.1 | Rattus tanezumi hunnivirus | GDVEENPGP | Picornaviridae |
| AWK02688.1 | Rattus tanezumi parechovirus—2A1 | GDVEENPGP | Picornaviridae |
| Rattus tanezumi parechovirus—2A2 | GDVEENPGP | ||
| ACO92353.1 | Saffold virus | HDIETNPGP | Picornaviridae |
| YP_001210296.2 | Saffold virus | HDVETNPGP | Picornaviridae |
| APZ85840.1 | Senecavirus A | GDIETNPGP | Picornaviridae |
| AHW57724.1 | Sikhote-Alin virus | HDVEMNPGP | Picornaviridae |
| AUK47911.1 | Swine pasivirus SPaV1/US/17-50816IA60467-1/2001 | GDVEQNPGP | Picornaviridae |
| BAU71153.1 | Swine picornavirus | GDVEENPGP | Picornaviridae |
| NP_653143.1 | Teschovirus A | GDVEENPGP | Picornaviridae |
| ACG55799.1 | Theiler’s encephalomyelitis virus | HDVETNPGP | Picornaviridae |
| BAC58035.1 | Theiler’s-like virus of rats | HDVETNPGP | Picornaviridae |
| AIY68187.1 | Tortoise picornavirus | GDVEVNPGP | Picornaviridae |
| AIY68186.1 | Tortoise picornavirus | GDVEQNPGP | Picornaviridae |
| ACG55801.1 | Vilyuisk human encephalomyelitis virus | HDVEMNPGP | Picornaviridae |
| AVM87411.1 | Yili teratoscincus roborowskii picornavirus 2 | GDVEQNPGP | Picornaviridae |
| YP_009329817.1 | Bivalve RNA virus G1 | GDVETNPGP | Unassigned Dicistroviridae |
| QNL09596.1 | Clinch dicistro-like virus 2—2A1 | GDVEMNPGP | Unassigned Dicistroviridae |
| Clinch dicistro-like virus 2—2A2 | GDVETNPGP | ||
| QJI52079.1 | Dicistroviridae sp. | GDVEMNPGP | Unassigned Dicistroviridae |
| AYQ66681.1 | Drosophila kikkawai virus 1 | GDVELNPGP | Unassigned Dicistroviridae |
| YP_009336571.1 | Hubei diptera virus 1 | GDVELNPGP | Unassigned Dicistroviridae |
| YP_009336583.1 | Hubei picorna-like virus 16 | GDVELNPGP | Unassigned Dicistroviridae |
| YP_009336853.1 | Hubei picorna-like virus 17 | GDVELNPGP | Unassigned Dicistroviridae |
| QKF95572.1 | Leibnitzia anandria dicistrovirus | GDIEENPGP | Unassigned Dicistroviridae |
| AXA52579.1 | Linepithema humile virus 1 | GDIELNPGP | Unassigned Dicistroviridae |
| QIU80542.1 | Phenacoccus solenopsis virus | GDIEENPGP | Unassigned Dicistroviridae |
| YP_009336743.1 | Wenling crustacean virus 3 | GDVEENPGP | Unassigned Dicistroviridae |
| YP_009333180.1 | Wenling picorna-like virus 2 | GDIELNPGP | Unassigned Dicistroviridae |
| YP_009342327.1 | Wuhan insect virus 11 | GDIEANPGP | Unassigned Dicistroviridae |
| YP_009329857.1 | Beihai hepe-like virus 4 | GDIESNPGP | Unassigned Hepeviridae |
| QDY81493.1 | Bipolaris oryzae hypovirus 1 | GDVEANPGP | Unassigned Hypoviridae |
| YP_009337372.1 | Hubei picorna-like virus 43 | GDIESNPGP | Unassigned Iflaviridae |
| QKN89050.1 | Iflaviridae sp.—2A1 | GDVESNPGP | Unassigned Iflaviridae |
| Iflaviridae sp.—2A2 | GDIESNPGP | ||
| AWK77896.1 | Perth bee virus 3 | GDVETNPGP | Unassigned Iflaviridae |
| YP_009336821.1 | Wenzhou picorna-like virus 49 | HDVELNPGP | Unassigned Iflaviridae |
| AVM87450.1 | Guangdong spotted longbarbel catfish picornavirus—2A1 | GDVEENPGP | Unassigned Picornavirales |
| Guangdong spotted longbarbel catfish picornavirus—2A2 | GDIESNPGP | ||
| Guangdong spotted longbarbel catfish picornavirus—2A3 | GDVERNPGP | ||
| ASG92543.1 | Picornavirales Q_sR_OV_036 | GDVEANPGP | Unassigned Picornavirales |
| ASG92538.1 | Picornavirales Q_sR_OV_042 | GDIEENPGP | Unassigned Picornavirales |
| ATY47693.1 | Picornavirales sp. | GDVEENPGP | Unassigned Picornavirales |
| ATY47707.1 | Picornavirales sp. | GDVELNPGP | Unassigned Picornavirales |
| AWK02666.1 | Rhinolophus sinicus picornavirus | GDIEENPGP | Unassigned Picornavirales |
| QQP18688.1 | Soybean thrips picorna-like virus 7 | GDVETNPGP | Unassigned Picornavirales |
| AWK02669.1 | Suncus murinus picornavirus | GDVETNPGP | Unassigned Picornavirales |
| AWK77886.1 | Victoria bee virus 1 | GDVETNPGP | Unassigned Picornavirales |
| AWK77887.1 | Victoria bee virus 2 | GDIETNPGP | Unassigned Picornavirales |
| AVM87443.1 | Wenling thamnaconus septentrionalis picornavirus | GDIESNPGP | Unassigned Picornavirales |
| AVM87419.1 | Western African lungfish picornavirus | GDVEENPGP | Unassigned Picornavirales |
| AVM87438.1 | Wuhan carp picornavirus—2A1 | GDVESNPGP | Unassigned Picornavirales |
| Wuhan carp picornavirus—2A2 | GDVESNPGP | ||
| Wuhan carp picornavirus—2A3 | GDVESNPGP | ||
| ANN02882.1 | Bovine rhinitis B virus 5 | GDVETNPGP | Unassigned Picornaviridae |
| AQM40272.1 | Human cosavirus (Cosavirus-zj-1) | GDVEENPGP | Unassigned Picornaviridae |
| AWG94399.1 | Human cosavirus E/D | GDVEENPGP | Unassigned Picornaviridae |
| AVX29481.1 | Marmot cardiovirus | HDVETNPGP | Unassigned Picornaviridae |
| AWK02672.1 | Niviventer confucianus hunnivirus | GDVELNPGP | Unassigned Picornaviridae |
| AFV31450.1 | Parechovirus-like virus | GDVEQNPGP | Unassigned Picornaviridae |
| QBH68005.1 | Parechovirus sp. QAPp32 | GDVEENPGP | Unassigned Picornaviridae |
| QKE55061.1 | Picornaviridae sp. | GDIEENPGP | Unassigned Picornaviridae |
| QKE55028.1 | Picornaviridae sp.—2A1 | GDVESNPGP | Unassigned Picornaviridae |
| Picornaviridae sp.—2A2 | GDVEQNPGP | ||
| Picornaviridae sp.—2A3 | GDVESNPGP | ||
| QIM74091.1 | Picornaviridae sp. | HDVETNPGP | Unassigned Picornaviridae |
| YP_009336671.1 | Wenzhou picorna-like virus 48—2A1 | GDIEENPGP | Unassigned Picornaviridae |
| Wenzhou picorna-like virus 48—2A2 | GDIESNPGP | ||
| Wenzhou picorna-like virus 48—2A3 | GDIEENPGP | ||
| AZT88626.1 | Aspergillus homomorphus yadokarivirus 1 | GDIEENPGP | Unassigned pssRNA |
| APG77930.1 | Beihai picorna-like virus 76 | GDVETNPGP | Unassigned pssRNA |
| YP_009333551.1 | Beihai picorna-like virus 85 | GDVETNPGP | Unassigned pssRNA |
| AYN75548.1 | Halhan virus 1 | GDVEQNPGP | Unassigned pssRNA |
| AZT88627.1 | Penicillium digitatum yadokarivirus 1 | GDVETNPGP | Unassigned pssRNA |
| QOI17269.1 | Picoa juniperi yado-kari virus 1 | GDIESNPGP | Unassigned pssRNA |
| QHD64758.1 | Plasmopara viticola lesion associated yadokari virus 1 | GDIEENPGP | Unassigned pssRNA |
| QIJ25855.1 | Warroolaba Creek virus 2 | GDVETNPGP | Unassigned pssRNA |
| AVD68673.2 | Yado-kari virus 2 | GDVEENPGP | Unassigned pssRNA |
| Accession Number | Virus | 2A Motif | Taxon |
|---|---|---|---|
| AAU88188.1 | Adult diarrhea virus | ECIESNPGP | Reoviridae |
| BAB20437.1 | Bombyx mori cypovirus 1 | GDIESNPGP | Reoviridae |
| BAO73973.1 | Bovine rotavirus C | GDVELNPGP | Reoviridae |
| AAO32344.1 | Dendrolimus punctatus cypovirus 1 | GDVESNPGP | Reoviridae |
| BAU80889.1 | Human rotavirus C | GDIELNPGP | Reoviridae |
| AAK73524.1 | Lymantria dispar cypovirus 1 | GDVESNPGP | Reoviridae |
| ABB17215.1 | Operophtera brumata cypovirus 18 | GDVESNPGP | Reoviridae |
| BAV31546.1 | Porcine rotavirus C | GDVELNPGP | Reoviridae |
| QBJ02264.1 | Porcine rotavirus H | GDVELNPGP | Reoviridae |
| AQX34666.1 | Rotavirus I | GDIESNPGP | Reoviridae |
| CCD33025.1 | Aspergillus foetidus slow virus 2 | GDIEENPGP | Unassigned dsRNA |
| YP_009272910.1 | Fusarium poae mycovirus 2 | GDIEENPGP | Unassigned dsRNA |
| YP_009182156.1 | Penicillium aurantiogriseum asp-foetidus like virus 1 | GDIEENPGP | Unassigned dsRNA |
| YP_009342431.1 | Wuhan insect virus 31—2A1 | GDVELNPGP | Unassigned dsRNA |
| Wuhan insect virus 31—2A2 | GDVERNPGP | ||
| YP_003934933.1 | Armigeres subalbatus | GDVESNPGP | Unassigned Totiviridae |
| YP_009256208.1 | Golden shiner totivirus | GDIESNPGP | Unassigned Totiviridae |
| AIC34742.2 | Penaeid shrimp infectious myonecrosis virus—2A1 | GDVESNPGP | Unassigned Totiviridae |
| Penaeid shrimp infectious myonecrosis virus—2A2 | GDVEENPGP | ||
| YP_009337085.1 | Wenling toti-like virus 2 | GDIETNPGP | Unassigned Totiviridae |
| YP_009333269.1 | Wenzhou toti-like virus 1 | GDVEMNPGP | Unassigned Totiviridae |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Lima, J.G.S.; Lanza, D.C.F. 2A and 2A-like Sequences: Distribution in Different Virus Species and Applications in Biotechnology. Viruses 2021, 13, 2160. https://doi.org/10.3390/v13112160
de Lima JGS, Lanza DCF. 2A and 2A-like Sequences: Distribution in Different Virus Species and Applications in Biotechnology. Viruses. 2021; 13(11):2160. https://doi.org/10.3390/v13112160
Chicago/Turabian Stylede Lima, Juliana G. S., and Daniel C. F. Lanza. 2021. "2A and 2A-like Sequences: Distribution in Different Virus Species and Applications in Biotechnology" Viruses 13, no. 11: 2160. https://doi.org/10.3390/v13112160
APA Stylede Lima, J. G. S., & Lanza, D. C. F. (2021). 2A and 2A-like Sequences: Distribution in Different Virus Species and Applications in Biotechnology. Viruses, 13(11), 2160. https://doi.org/10.3390/v13112160







