Virus Diseases of Cereal and Oilseed Crops in Australia: Current Position and Future Challenges
Abstract
1. Introduction
2. Background Information
2.1. Australian Cereals and Oilseeds Industries
2.2. Epidemiology
2.3. General Management Principles
2.4. Viruses Infecting Cereal and Oilseed Crops
2.5. Quantification of Yield Losses
3. Cereal Viruses
3.1. Yellow Dwarf Viruses
3.2. Wheat Streak Mosaic Virus
3.3. Johnsongrass Mosaic Virus
3.4. Other Cereal Viruses
3.5. Possible Occurrence of Soil-Borne Cereal Viruses
3.6. Recommendations for Further Research
4. Oilseed Viruses
4.1. Turnip Yellows Virus
4.2. Turnip Mosaic Virus
4.3. Tobacco Streak Virus
4.4. Cotton Bunchy Top Virus
4.5. Other Oilseed Viruses
4.6. Recommendations for Further Research
5. Future Threats to Virus Management
5.1. Biosecurity Threats
5.2. Threats from Vector Insecticide Resistance
5.3. Threats from Resistance Breaking Virus Strains
5.4. Climate Change Threats
5.5. Insufficient Industry Awareness
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bos, L. Crop losses caused by viruses. Crop Prot. 1982, 1, 263–282. [Google Scholar] [CrossRef]
- Bos, L. New plant virus problems in developing countries: A corollary of agricultural modernization. In Advances in Virus Research; Maramorosch, K., Murphy, F.A., Shatkin, A.J., Eds.; Academic Press: Cambridge, MA, USA, 1992; Volume 41, pp. 349–407. [Google Scholar]
- Thresh, J.M. Crop Viruses and Virus Diseases: A Global Perspective. In Virus Diseases and Crop Biosecurity; NATO Security through Science Series; Cooper, I., Kühne, T., Polishchuk, V.P., Eds.; Springer: Dordecht, The Netherlands, 2006; pp. 9–32. [Google Scholar]
- Thresh, J.M. Control of Tropical Plant Virus Diseases. Adv. Virus Res. 2006, 67, 245–295. [Google Scholar]
- Jones, R.A.C. Plant virus emergence and evolution: Origins, new encounter scenarios, factors driving emergence, effects of changing world conditions, and prospects for control. Virus Res. 2009, 141, 113–130. [Google Scholar] [CrossRef]
- Jones, R.A.C. Future scenarios for plant virus pathogens as climate change progresses. Adv. Virus Res. 2016, 95, 87–147. [Google Scholar]
- Jones, R.A.C. Disease pandemics and major epidemics arising from new encounters between indigenous viruses and introduced crops. Viruses 2020, 12, 1388. [Google Scholar] [CrossRef]
- Jones, R.A.C. Global plant virus disease pandemics and epidemics. Plants 2021, 10, 233. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.A.C.; Barbetti, M.J. Influence of climate change on plant disease infections and epidemics caused by viruses and bacteria. CAB Rev. 2012, 7, 1–33. [Google Scholar] [CrossRef]
- Hull, R. Plant Virology, 5th ed.; Academic Press: San Diego, CA, USA, 2014. [Google Scholar]
- Trębicki, P. Climate change and plant virus epidemiology. Virus Res. 2020, 286, 198059. [Google Scholar] [CrossRef] [PubMed]
- Sastry, K.S.; Zitter, T.A. Management of virus and viroid diseases of crops in tropics. In Plant Virus and Viroid Diseases in the Tropics; Epidemiology and Management; Springer: Heidelberg, Germany, 2014; Volume 2, pp. 149–480. [Google Scholar]
- Buchen-Osmond, C.; Crabtree, K.; Gibbs, A.J.; McLean, G.D. Viruses of Plants in Australia: Descriptions and Lists from the VIDE Database (No. 632.8 V821v); Australian National University: Canberra, ACT, Australia, 1988. [Google Scholar]
- Jones, R.A.C.; Coutts, B.A. Spread of introduced viruses to new plants in natural ecosystems and the threat this poses to plant biodiversity. Mol. Plant Pathol. 2015, 16, 541–545. [Google Scholar] [CrossRef]
- Rodoni, B.C. The role of plant biosecurity in preventing and controlling emerging plant virus disease epidemics. Virus Res. 2009, 141, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Gerritsen, R. Evidence for indigenous Australian agriculture. Australas Sci. 2010, 31, 35–37. [Google Scholar]
- Mulvaney, J.; Kamminga, J. Prehistory of Australia; Routledge Publishing: Abingdon, UK, 2020. [Google Scholar]
- Henzell, T. Australian Agriculture: Its History and Challenges; CSIRO Publishing: Melbourne, VIC, Australia, 2007. [Google Scholar]
- Brown, A.; De Costa, C.; Guo, F. Our Food Future: Trends and Opportunities, Canberra. 2020. [CrossRef]
- Johnstone, G.R.; Barbetti, M.J. Impact of fungal and virus diseases on pasture. In Temperate Pastures: Their Production, Use and Management; Wheeler, J.L., Pearson, C.J., Roberts, G.E., Eds.; Australian Wool Corporation/CSIRO: Melbourne, VIC, Australia, 1987; pp. 235–248. [Google Scholar]
- Panetta, F.D.; Ridsdell-Smith, T.J.; Barbetti, M.J.; Jones, R.A.C. The ecology of weeds, invertebrate pests and diseases of australian sheep pastures. In Pests of Pastures: Weed, Invertebrate and Disease Pests of Australian Sheep Pastures; Delfosse, E.S., Ed.; Australian Wool Corporation/CSIRO: Melbourne, VIC, Australia, 1993; pp. 87–114. [Google Scholar]
- Barbetti, M.J.; Jones, R.A.C.; Riley, I.T. Problems and progress in assessing direct and indirect yield losses caused by pathogens in pasture species. In Pasture and Forage Crop Pathology; Chakraborty, S., Leath, K.T.L., Skipp, R.A., Pederson, G.A., Bray, R.A., Latch, G.C.M., Nutter, F.W., Eds.; American Society of Agronomy, Inc.: Madison, WI, USA, 1996. [Google Scholar]
- Barbetti, M.J.; Pei You, M.P.; Jones, R.A.C. Medicago truncatula and other annual Medicago Spp.–Interactions with root and foliar fungal, oomycete, and viral pathogens. In The Model Legume Medicago truncatula; de Brujin, F., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2020; pp. 293–306. [Google Scholar]
- Nichols, P.G.H.; Jones, R.A.C.; Ridsdill-Smith, T.J.; Barbetti, M.J. Genetic improvement of subterranean clover (Trifolium subterraneum L.). 2. Breeding for disease and pest resistance. Crop Pasture Sci. 2014, 65, 1207–1229. [Google Scholar] [CrossRef]
- Salam, M.U.; Davidson, J.A.; Thomas, G.J.; Ford, R.; Jones, R.A.C.; Lindbeck, K.D.; Macleod, W.J.; Kimber, R.B.; Galloway, J.; Mantri, N.; et al. Advances in winter pulse pathology research in Australia. Austral. Plant. Pathol. 2011, 40, 549–567. [Google Scholar] [CrossRef]
- Inturrisi, F.C.; Barbetti, M.J.; Tirnaz, S.; Patel, D.A.; Edwards, D.; Batley, J. Molecular characterization of disease resistance in Brassica juncea—The current status and the way forward. Plant. Pathol. 2020, 70, 13–34. [Google Scholar] [CrossRef]
- Jones, R.A.C. Virus diseases of annual pasture legumes: Incidences, losses, epidemiology and management. Crop Pasture Sci. 2012, 63, 399–418. [Google Scholar] [CrossRef]
- Jones, R.A.C. Virus diseases of perennial pasture legumes: Incidences, losses, epidemiology and management. Crop Pasture Sci. 2013, 64, 199–215. [Google Scholar] [CrossRef]
- Johnstone, G.R.; McLean, G.D. Virus diseases of subterranean clover. Ann. Appl. Biol. 1987, 110, 421–440. [Google Scholar] [CrossRef]
- Jones, R.A.C. Virus Diseases of Australian Pastures. In Pasture and Forage Crop Pathology; Chackraborty, S.L.K., Skipp, R.A., Pederson, G.A., Bray, R., Latch, G.C.M., Nutter, F.W., Eds.; American Society of Agronomy: Madison, WI, USA, 1996; pp. 288–307. [Google Scholar]
- Jones, R.A.C. Virus diseases of pasture grasses in Australia: Incidences, losses, epidemiology, and management. Crop Pasture Sci. 2013, 64, 199. [Google Scholar] [CrossRef]
- Jones, R.A.C. Plant virus ecology and epidemiology: Historical perspectives, recent progress and future prospects. Ann. Appl. Biol. 2014, 164, 320–347. [Google Scholar] [CrossRef]
- Jones, R.A.C. Developing integrated disease management strategies against non-persistently aphid-borne viruses: A model programme. Integr. Pest Manag. Rev. 2001, 6, 15–46. [Google Scholar] [CrossRef]
- Jones, R.A.C.; McLean, G.D. Virus diseases of lupins. Ann. Appl. Biol. 1989, 114, 609–637. [Google Scholar] [CrossRef]
- Parry, H.R.; Macfadyen, S.; Kriticos, D.J. The geographical distribution of yellow dwarf viruses and their aphid vectors in Australian grasslands and wheat. Austral. Plant. Pathol. 2012, 41, 375–387. [Google Scholar] [CrossRef]
- Zekulich, M. The Grain Journey: The History of the Grain Pool of Western Australia; The Grain Pool of Western Australia: Perth, WA, Australia, 1997. Available online: https://catalogue.nla.gov.au/Record/227832 (accessed on 12 September 2021).
- Pratley, J.; Kirkegaard, J. (Eds.) Australian Agriculture in 2020: From Conservation to Automation; Agronomy Australia and Charles Sturt University: Wagga Wagga, NSW, Australia, 2020. Available online: http://www.agronomyaustraliaproceedings.org/images/sampledata/specialpublications/Australian%20Agriculture%20in%202020.pdf (accessed on 12 September 2021).
- Shaw, J.H. Collins Australian Encyclopedia; William Collins Pty Ltd.: Sydney, Australia, 1984. [Google Scholar]
- Freund, M.; Henley, B.J.; Karoly, D.J.; Allen, K.J.; Baker, P.J. Multi-century cool- and warm-season rainfall reconstructions for Australia’s major climatic regions. Clim. Past 2017, 13, 1751–1770. [Google Scholar] [CrossRef]
- Bates, B.C.; Kundzewicz, Z.W.; Shaohong, W.; Palutikof, J.P. Climate Change and Water; Technical Paper of the Intergovernmental Panel on Climate Change; IPCC Secretariat: Geneva, Switzerland, 2008; p. 210. [Google Scholar]
- Stephens, D.J.; Lyons, T.J. Rainfall-yield relationships across the Australian wheatbelt. Aust. J. Agric. Res. 1998, 49, 211–224. [Google Scholar] [CrossRef]
- Turner, N.C.; Asseng, S. Productivity, sustainability, and rainfall-use efficiency in Australian rainfed Mediterranean agricultural systems. Aust. J. Agric. Res. 2005, 56, 1123–1136. [Google Scholar] [CrossRef]
- Turner, N.C.; Molyneux, N.; Yang, S.; Xiong, Y.-C.; Siddique, K.H.M. Climate change in south-west Australia and north-west China: Challenges and opportunities for crop production. Crop Pasture Sci. 2011, 62, 445–456. [Google Scholar] [CrossRef]
- Australian Export Grains Innovation Centre. What grows where? In Australian Grain Production—A Snapshot. Australian Grain Note; AEGIC: Sydney/Perth, Australia, 2020; Available online: https://www.aegic.org.au/australian-grain-production-a-snapshot/ (accessed on 12 September 2021).
- Blakeney, A.B.; Lewin, L.; Batten, G.; Welsh, L. Rice Cultivation and Quality in Australia; Ricegrowers’ Cooperative Limited: Sydney, NSW, Australia, 1996. [Google Scholar]
- Kemmler, G. Some notes on rice cultivation in Australia. Jpn. J. Trop. Agric. 1965, 8, 135–139. [Google Scholar]
- Matsui, T.; Kobayasi, K.; Yoshimoto, M.; Hasegawa, T. Stability of rice pollination in the field under hot and dry conditions in the Riverina region of New South Wales, Australia. Plant. Prod. Sci. 2007, 10, 57–63. [Google Scholar] [CrossRef]
- Kimberley Development Commission. Primary Industries: The Ord River Irrigation Area. 2020. Available online: https://kdc.wa.gov.au/economic-profile/primaryindustries/#:~:text=The%20Ord%20Valley%20produces%20a,stages%20over%20the%20last%20years (accessed on 8 September 2021).
- Cooperative Research Centre for Northern Australia. Northern Australian Broadacre Cropping Situational Analysis. 2020. Available online: https://www.crcna.com.au/resources/publications/northern-australian-broadacre-cropping-situational-analysis (accessed on 8 September 2021).
- Northern Territory Government. Agriculture, Forestry and Fishing. Northern Territory Economy. 2017. Available online: https://nteconomy.nt.gov.au/industry-analysis/agriculture,-foresty-and-fishing (accessed on 6 September 2021).
- Grains Research and Development Corporation. Australian Grains Focus 2010–2011. 2010. Available online: https://grdc.com.au/__data/assets/pdf_file/0025/208735/grdc-australian-grains-focus-20102011.pdf (accessed on 9 September 2021).
- Jeger, M.J.; Madden, L.V.; van den Bosch, F. Plant Virus Epidemiology: Applications and prospects for mathematical modeling and analysis to improve understanding and disease control. Plant Dis. 2017, 102, 837–854. [Google Scholar] [CrossRef]
- Jones, R.A.C.; Naidu, R. Global dimensions of plant virus diseases: Current status and future perspectives. Ann. Rev. Virol. 2019, 6, 387–409. [Google Scholar] [CrossRef] [PubMed]
- Thresh, J.M. The origins and epidemiology of some important plant virus diseases. Appl. Biol. 1980, 5, 1–65. [Google Scholar]
- Jones, R.A.C.; Salam, M.U.; Maling, T.J.; Diggle, A.J.; Thackray, D.J. Principles of predicting plant virus disease epidemics. Ann. Rev. Phytopathol. 2010, 48, 179–203. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.A.C. Control of plant virus diseases. Adv. Virus Res. 2006, 67, 205–244. [Google Scholar]
- Makkouk, K.M.; Kumari, S.G.; van Leur, J.A.G.; Jones, R.A.C. Control of plant virus diseases in cool-season legume crops. Adv. Virus Res. 2014, 90, 207–253. [Google Scholar]
- Thresh, J.M. Cropping practices and virus spread. Ann. Rev. Phytopathol. 1982, 20, 193–218. [Google Scholar] [CrossRef]
- Jones, R.A.C. Using epidemiological information to develop effective integrated virus disease management strategies. Virus Res. 2004, 100, 5–30. [Google Scholar] [CrossRef]
- Peng, Y.; Dallas, M.; Ascencio-Ibáñez, J.T.; Hoyer, S.; Legg, J.; Hanley-Bowdoin, L.; Grieve, B.; Yin, H. Early detection of plant virus infection using multispectral imaging and spatial-spectral machine learning. Sensors 2021, 21, 742. [Google Scholar]
- Jones, R.A.C. Trends in plant virus epidemiology: Opportunities from new or improved technologies. Virus Res. 2014, 186, 3–19. [Google Scholar] [CrossRef]
- Watson, A.; Ghosh, S.; Williams, M.J.; Cuddy, W.S.; Simmonds, J.; Rey, M.D.; Hatta, M.A.M.; Hinchliffe, A.; Steed, A.; Reynolds, D.; et al. Speed breeding is a powerful tool to accelerate crop research and breeding. Nat. Plants 2018, 4, 23–29. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, X.; Zhou, G.; Zhang, T. Engineering plant virus resistance: From RNA silencing to genome editing strategies. Plant Biotechnol. J. 2020, 18, 328–336. [Google Scholar] [CrossRef]
- Taliansky, M.; Samarskaya, V.; Zavriev, S.K.; Fesenko, I.; Kalinina, N.O.; Love, A.J. RNA-based technologies for engineering plant virus resistance. Plants 2021, 10, 82. [Google Scholar] [CrossRef] [PubMed]
- Farooq, T.; Adeel, M.; He, Z.; Umar, M.; Shakoor, N.; da Silva, W.; Elmer, W.; White, J.C.; Rui, Y. Nanotechnology and plant viruses: An emerging disease management approach for resistant pathogens. ACS Nano 2021, 15, 6030–6037. [Google Scholar] [CrossRef]
- Blyuss, K.B.; Al Basir, F.; Tsygankova, V.A.; Biliavska, L.O.; Iutynska, G.O.; Kyrychko, S.N.; Dziuba, S.V.; Tsyliuryk, O.I.; Izhboldin, O.O. Control of mosaic disease using microbial biostimulants: Insights from mathematical modelling. Ric. Mat. 2020, 69, 437–455. [Google Scholar] [CrossRef]
- Kraberger, S.; Geering, A.D.W.; Walters, M.; Martin, D.P.; Varsani, A. Novel mastreviruses identified in Australian wild rice. Virus Res. 2017, 238, 193–197. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jones, R.A.C.; Boonham, N.; Adams, I.P.; Fox, A. Historical virus isolate collections: An invaluable resource connecting plant virology’s pre-sequencing and post-sequencing eras. Plant Pathol. 2021, 70, 235–248. [Google Scholar] [CrossRef]
- Banks, P.M.; Davidson, J.L.; Bariana, H.; Larkin, P.J. Effects of barley yellow dwarf virus on the yield of winter wheat. Aust. J. Agric. Res. 1995, 46, 935–946. [Google Scholar] [CrossRef]
- McKirdy, S.J.; Jones, R.A.C. Use of imidacloprid and newer generation synthetic pyrethroids to control the spread of barley yellow dwarf luteovirus in cereals. Plant Dis. 1996, 80, 895–901. [Google Scholar] [CrossRef]
- McKirdy, S.J.; Jones, R.A.C.; Nutter, F.W. Quantification of yield losses caused by barley yellow dwarf virus in wheat and oats. Plant Dis. 2002, 86, 769–773. [Google Scholar] [CrossRef]
- Thackray, D.J.; Ward, L.T.; Thomas-Carroll, M.L.; Jones, R.A.C. Role of winter-active aphids spreading barley yellow dwarf virus in decreasing wheat yields in a Mediterranean-type environment. Aust. J. Agric. Res. 2005, 56, 1089–1099. [Google Scholar] [CrossRef]
- Nancarrow, N.; Aftab, M.; Hollaway, G.; Rodoni, B.C.; Trębicki, P. Yield losses caused by barley yellow dwarf virus-PAV infection in wheat and barley: A three-year field study in south-eastern Australia. Microorganisms 2021, 9, 645. [Google Scholar] [CrossRef]
- Fahim, M.; Larkin, P.J.; Haber, S.; Shorter, S.; Lonergan, P.F.; Rosewarne, G.M. Effectiveness of three potential sources of resistance in wheat against wheat streak mosaic virus under field conditions. Austral. Plant Pathol. 2012, 41, 301–309. [Google Scholar] [CrossRef]
- Persley, D.M.; Greber, R.S.; Henzell, R.G.; Fletcher, D.S. Effect of sugarcane mosaic virus on the yield of grain sorghum and maize in Queensland. Aust. Plant Pathol. Soc. Newsl. 1976, 5, 73. [Google Scholar]
- Henzell, R.G.; Persley, D.M.; Fletcher, D.S.; Greber, R.S.; Slobbe, L. The effect of sugarcane mosaic virus on the yield of eleven grain sorghum (Sorghum bicolor) cultivars. Aust. J. Exp. Agric. 1979, 19, 225–232. [Google Scholar] [CrossRef]
- Jones, R.A.C.; Coutts, B.A.; Hawkes, J.R. Yield-limiting potential of beet western yellows virus in Brassica napus. Aust. J. Agric. Res. 2007, 58, 788–801. [Google Scholar] [CrossRef]
- McLean, G.D.; Khan, T.N. Assessing the incidence and severity of barley yellow dwarf virus in the field. Austral. Plant Pathol. 1983, 12, 50–51. [Google Scholar] [CrossRef]
- Guy, P.L.; Johnstone, G.R.; Morris, D. Barley yellow dwarf viruses in, and aphids on, grasses (including cereals) in Tasmania. Aust. J. Agric. Res. 1987, 38, 139–152. [Google Scholar] [CrossRef]
- Sward, R.J.; Lister, R.M. The incidence of barley yellow dwarf viruses in wheat in Victoria. Aust. J. Agric. Res. 1987, 38, 821–828. [Google Scholar] [CrossRef]
- Sward, R.J.; Lister, R.M. The identity of barley yellow dwarf virus isolates in cereals and grasses from mainland Australia. Aust. J. Agric. Res. 1988, 39, 375–384. [Google Scholar] [CrossRef]
- Greber, R.S. Leafhopper and aphid-borne viruses affecting subtropical cereal and grass crops. In Current Topics in Vector Research; Harris, K.F., Ed.; Praeger Publishers: New York, NY, USA, 1984; pp. 141–183. [Google Scholar]
- Greber, R.S. Ecology of barley yellow dwarf virus in south-east Queensland. Austral. Plant Pathol. 1988, 17, 101–104. [Google Scholar] [CrossRef]
- Jones, R.A.C.; McKirdy, S.J.; Shivas, R.G. Occurrence of barley yellow dwarf viruses in over-summering grasses and cereal crops in Western Australia. Austral. Plant Pathol. 1990, 19, 90–96. [Google Scholar] [CrossRef]
- Guy, P.L. Barley yellow dwarf virus infection of the Gramineae in Tasmania. Acta Phytopathol. Entomol. Hung. 1991, 26, 21–26. [Google Scholar]
- Henry, M.; Francki, R.I.B.; Wallwork, H. Occurrence of barley yellow dwarf virus in cereals and grasses of the low-rainfall wheatbelt of South Australia. Plant Pathol. 1992, 41, 713–721. [Google Scholar] [CrossRef]
- Coutts, B.A.; Strickland, G.R.; Kehoe, M.A.; Severtson, D.L.; Jones, R.A.C. The epidemiology of wheat streak mosaic virus in Australia: Case histories, gradients, mite vectors, and alternative hosts. Aust. J. Agric. Res. 2008, 59, 844–853. [Google Scholar] [CrossRef]
- Coutts, B.A.; Banovic, M.; Kehoe, M.A.; Severtson, D.L.; Jones, R.A.C. Epidemiology of wheat streak mosaic virus in wheat in a Mediterranean-type environment. Eur. J. Plant Pathol. 2014, 140, 797–813. [Google Scholar] [CrossRef]
- Nancarrow, N.; Aftab, M.; Freeman, A.J.; Rodoni, B.C.; Hollaway, G.; Trębicki, P. Prevalence and incidence of yellow dwarf viruses across a climatic gradient: A four-year field study in southeastern Australia. Plant Dis. 2018, 102, 2465–2472. [Google Scholar] [CrossRef]
- Smith, P.R.; Sward, R.J. Crop loss assessment studies on the effects of barley yellow dwarf virus in wheat in Victoria. Aust. J. Agric. Res. 1982, 33, 179–185. [Google Scholar] [CrossRef]
- Smith, H.C. Cereal yellow dwarf virus seen in Australia. Commonw. Phytopathol. News 1957, 3, 10–11. [Google Scholar]
- Smith, H.C. A survey of barley yellow dwarf virus in Australia 1963. N. Z. J. Agric. Res. 1964, 7, 239–247. [Google Scholar] [CrossRef][Green Version]
- Teakle, D.S.; Moore, R.S.; George, D.L.; Byth, D.E. Inheritance of the necrotic and mosaic reactions in sorghum infected with a ‘Johnson grass’ strain of sugarcane mosaic virus. Aust. J. Agric. Res. 1970, 21, 549–556. [Google Scholar] [CrossRef]
- Persley, D.M.; Greber, R.S.; Moore, R.S. A new source of mosaic resistance in sorghum. Austral. Plant Pathol. 1972, 1, 11–12. [Google Scholar] [CrossRef]
- Persley, D.M.; Martin, I.F.; Greber, R.S. The resistance of maize inbred lines to sugarcane mosaic virus in Australia. Aust. J. Agric. Res. 1981, 32, 741–748. [Google Scholar] [CrossRef]
- Karan, M.; Noone, D.F.; Teakle, D.S.; Hacker, J.B. Susceptibility of pearl millet accessions and cultivars to johnsongrass mosaic and sugarcane mosaic viruses in Queensland. Austral. Plant Pathol. 1992, 21, 128–130. [Google Scholar] [CrossRef]
- McLean, G.D.; Khan, T.N.; McLean, R.J.; Portman, P.A. Effect of barley yellow dwarf virus on two near isogenic lines of barley. SABRAO J. 1984, 16, 143–184. [Google Scholar]
- McKirdy, S.J.; Jones, R.A.C. Effect of sowing time on barley yellow dwarf virus infection in wheat: Virus incidence and grain yield losses. Aust. J. Agric. Res. 1997, 48, 199–206. [Google Scholar] [CrossRef]
- Choudhury, S.; Larkin, P.J.; Meinke, H.; Hasanuzzaman, M.D.; Johnson, P.; Zhou, M. Barley yellow dwarf virus infection affects physiology, morphology, grain yield and flour pasting properties of wheat. Crop Pasture Sci. 2019, 70, 16–25. [Google Scholar] [CrossRef]
- Coutts, B.A.; Hammond, N.E.B.; Kehoe, M.A.; Jones, R.A.C. Finding wheat streak mosaic virus in south-west Australia. Aust. J. Agric. Res. 2008, 59, 836–843. [Google Scholar] [CrossRef]
- Jones, R.A.C. Report on Fact Finding Mission to Assess the Wheat Streak Mosaic Virus (WSMV) Epidemic in New South Wales and the Potential Threat Posed to the Western Australian Wheat Industry; Department of Agriculture for Western Australia: Perth, WA, Australia, 17–22 November 2005. [Google Scholar]
- Murray, G.M.; Wratten, K. Wheat Streak Mosaic Virus. In Plant Disease Notes; New South Wales Department of Primary Industries: Wagga Wagga, NSW, Australia, 2005. [Google Scholar]
- CABI Invasive Species Compendium. 2021. Available online: https://www.cabi.org/isc/ (accessed on 6 September 2021).
- McKirdy, S.J.; Jones, R.A.C. Barley yellow dwarf virus in cereals. J. Agric. West. Aust. Fourth Ser. 1993, 34, 3–8. [Google Scholar]
- Johnstone, G.R. Epidemiology of barley yellow dwarf in Australia. In Barley Yellow Dwarf—40 Years of Progress; D’Arcy, C.J., Burnett, P.A., Eds.; APS Press: St. Paul, MN, USA, 1995; pp. 129–143. [Google Scholar]
- Smith, P.R.; Plumb, R.T. Barley yellow dwarf virus infectivity of cereal aphids trapped at two sites in Victoria. Aust. J. Agric. Res. 1981, 32, 249–255. [Google Scholar] [CrossRef]
- Clarke, R.; Kehoe, M.A.; Broughton, S.; Jones, R.A.C. Host plant affiliations of aphid vector species found in a remote tropical environment. Virus Res. 2020, 281, 197934. [Google Scholar] [CrossRef]
- Waterhouse, P.M.; Helms, K. Metopolophium dirhodum (Walker): A newly arrived vector of barley yellow dwarf virus in Australia. Austral. Plant Pathol. 1985, 14, 64–66. [Google Scholar] [CrossRef]
- Hales, D.F.; Chapman, R.L.; Lardner, R.M.; Cowen, R.; Turak, E. Aphids of the genus Sitobion occurring on grasses in Southern Australia. Aust. J. Entomol. 1990, 29, 19–25. [Google Scholar] [CrossRef]
- McKirdy, S.J.; Jones, R.A.C. Occurrence of barley yellow dwarf virus serotypes MAV and RMV in over-summering grasses. Aust. J. Agric. Res. 1993, 44, 1195–1209. [Google Scholar] [CrossRef]
- De Barro, P.J. A survey of Rhopalosiphum padi (L.) (Hemiptera: Aphididae) and other wheat-infesting cereal aphids flying over South Australia in 1989. Aust. J. Entomol. 1992, 31, 345–349. [Google Scholar] [CrossRef]
- Guy, P.L.; Johnstone, G.R.; Duffus, J.E. Occurrence and identity of barley yellow dwarf virus in Tasmanian pasture grasses. Aust. J. Agric. Res. 1986, 37, 43–53. [Google Scholar] [CrossRef]
- International Committee on Taxonomy of Viruses. ICTV Master Species List 2019.v1 Checklist Data Set. 2020. Available online: https://talk.ictvonline.org/files/master-species-lists/m/msl/9601 (accessed on 3 March 2021).
- Aradottir, G.I.; Crespo-Herrera, L. Host plant resistance in wheat to barley yellow dwarf viruses and their aphid vectors: A review. Curr. Opin. Insect Sci. 2021, 45, 59–68. [Google Scholar] [CrossRef]
- Guy, P.L. Pasture ecology of barley yellow dwarf viruses at Sandford, Tasmania. Plant Pathol. 1988, 37, 546–550. [Google Scholar] [CrossRef]
- Hawkes, J.R.; Jones, R.A.C. Incidence and distribution of barley yellow dwarf virus and cereal yellow dwarf virus in over-summering grasses in a Mediterranean-type environment. Aust. J. Agric. Res. 2005, 56, 257–270. [Google Scholar] [CrossRef]
- Rochow, W.F. Biological properties of four isolates of barley yellow dwarf virus. Phytopathology 1969, 59, 1580–1589. [Google Scholar]
- De Barro, P.J. Ecology of the Bird Cherry-Oat Aphid, Rhopalosiphum padi (L.) (Hemiptera: Aphididae) in the Low Rainfall Wheat Belt of South Australia. Ph.D. Thesis, University of Adelaide—Waite Institute, Urrbrae, SA, Australia, 1991. [Google Scholar]
- Milgate, A.; Adorada, D.; Chambers, G.; Terras, M.A. Occurrence of winter cereal viruses in New South Wales, Australia, 2006 to 2014. Plant Dis. 2016, 100, 313–317. [Google Scholar] [CrossRef]
- Michael, P. Cereal aphids and direct feeding damage to cereals. In Agribusiness Cereals Update 2002; Jettner, R., Ed.; Department of Agriculture: Perth, WA, Australia, 2002; pp. 55–56. [Google Scholar]
- Coutts, B.A.; Jones, R.A.C. Temporal dynamics of spread of four viruses within mixed species perennial pastures. Ann. Appl. Biol. 2002, 140, 37–52. [Google Scholar] [CrossRef]
- Eagling, D.R.; Cunningham, P.J.; Sward, R.J.; Halloran, G.M. The incidence of barley yellow dwarf virus isolates in perennial ryegrass (Lolium perenne) in south-western Victoria. Plant Pathol. 1989, 38, 408–413. [Google Scholar] [CrossRef]
- Thackray, D.J.; Jones, R.A.C. Roles of nearby pastures in development of epidemics of aphid-borne virus diseases in crops in regions with Mediterranean-type climates. In Proceedings of the 7th Australasian Conference on Grassland Invertebrate Ecology, Perth, WA, Australia, 4–6 October 1999; Matthiessen, J.N., Ed.; CSIRO: Perth, WA, Australia; p. 298. [Google Scholar]
- Thackray, D.J.; Diggle, A.J.; Jones, R.A.C. BYDV Predictor: A simulation model to predict aphid arrival, epidemics of barley yellow dwarf virus and yield losses in wheat crops in a Mediterranean-type environment. Plant Pathol. 2009, 58, 186–202. [Google Scholar] [CrossRef]
- Wang, K.; Zhang, M.; Huang, Y.; Yang, Z.; Su, S.; Chen, M. Characterization of imidacloprid resistance in the bird cherry-oat aphid, Rhopalosiphum padi, a serious pest on wheat crops. Pest Manag. Sci. 2018, 74, 1457–1465. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Han, Z.; Qiao, X.; Qu, M. Resistance mechanisms and associated mutations in acetylcholinesterase genes in Sitobion avenae (Fabricius). Pestic. Biochem. Physiol. 2007, 87, 189–195. [Google Scholar] [CrossRef]
- Vertigan, W.A. Shannon: The first Tasmanian-bred barley. Tasman. J. Agric. 1980, 51, 33–35. [Google Scholar]
- Larkin, P.; Banks, P.M.; Lagudah, E.S.; Appels, R.; Xiao, C.; Zhiyong, X.; Ohm, H.W.; McIntosh, R.A. Disomic Thinopyrum intermedium addition lines in wheat with barley yellow dwarf virus resistance and with rust resistances. Genome 1995, 38, 385–394. [Google Scholar] [CrossRef]
- Collins, N.C.; Paltridge, N.G.; Ford, C.M.; Symons, R.H. The Yd2 gene for barley yellow dwarf virus resistance maps close to the centromere on the long arm of barley chromosome 3. Theor. Appl. Genet. 1996, 92, 858–864. [Google Scholar] [CrossRef]
- Francki, M.G.; Ohm, H.W.; Anderson, J.M. Novel germplasm providing resistance to barley yellow dwarf virus in wheat. Aust. J. Agric. Res. 2001, 52, 1375–1382. [Google Scholar] [CrossRef]
- Zhang, W.; Carter, M.; Matsay, S.; Stoutjesdijk, P.; Potter, R.; Jones, M.G.K.; Kleven, S.; Wilson, R.E.; Larkin, P.J.; Turner, M.; et al. Implementation of probes for tracing chromosome segments conferring barley yellow dwarf virus resistance. Crop Pasture Sci. 2001, 52, 1389–1392. [Google Scholar] [CrossRef]
- Ayala-Navarrete, L.I.; Tourton, E.; Mechanicos, A.A.; Larkin, P.J. Comparison of Thinopyrum intermedium derivatives carrying barley yellow dwarf virus resistance in wheat. Genome 2009, 52, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Ayala-Navarrete, L.I.; Larkin, P.J. Wheat virus diseases: Breeding for resistance and tolerance. In World Wheat Book: A History of Wheat Breeding; Bonjean, A., Angus, W., van Ginkel, M., Eds.; Lavoisier: Paris, France, 2011; Volume 2, pp. 1073–1107. [Google Scholar]
- Choudhury, S.; Al-Shammari, D.; Hu, H.; Meinke, H.; Birchall, C.; Larkin, P.J.; Zhou, M. A screening method to detect BYDV-PAV resistance in cereals under glasshouse conditions. Plant Pathol. 2018, 67, 1987–1996. [Google Scholar] [CrossRef]
- Choudhury, S.; Hu, H.; Larkin, P.J.; Meinke, H.; Shabala, S.; Ahmed, I.; Zhou, M. Agronomical, biochemical and histological response of resistant and susceptible wheat and barley under BYDV stress. Peer J. 2018, 6, e4833. [Google Scholar] [CrossRef]
- Choudhury, S.; Hu, H.; Meinke, H.; Shabala, S.; Larkin, P.J.; Zhou, M. Barley yellow dwarf viruses: Infection mechanisms and breeding strategies. Euphytica 2017, 213, 168. [Google Scholar] [CrossRef]
- Choudhury, S.; Larkin, P.J.; Xu, R.; Hayden, M.; Forrest, K.; Meinke, H.; Hu, H.; Zhou, M.; Fan, Y. Genome wide association study reveals novel QTL for barley yellow dwarf virus resistance in wheat. BMC Genom. 2019, 20, 891. [Google Scholar] [CrossRef]
- Hu, H.; Choudhury, S.; Shabala, S.; Gupta, S.; Zhou, M. Genomic regions on chromosome 5H containing a novel QTL conferring barley yellow dwarf virus-PAV (BYDV-PAV) tolerance in barley. Sci. Rep. 2019, 9, 11298. [Google Scholar] [CrossRef]
- Suneson, C.A. Breeding for resistance to yellow dwarf virus in barley. Agron. J. 1955, 47, 283. [Google Scholar] [CrossRef]
- Rasmusson, D.C.; Schaller, C. The inheritance of resistance in barley to the yellow-dwarf virus. Agron. J. 1959, 51, 661–664. [Google Scholar] [CrossRef]
- Niks, R.E.; Habekuß, A.; Bekele, B.; Ordon, F. A novel major gene on chromosome 6H for resistance of barley against the barley yellow dwarf virus. Theor. Appl. Genet. 2004, 109, 1536–1543. [Google Scholar] [CrossRef]
- Scholz, M.; Ruge-Wehling, B.; Habekuß, A.; Schrader, O.; Pendinen, G.; Fischer, K.; Wehling, P. Ryd4Hb: A novel resistance gene introgressed from Hordeum bulbosum into barley and conferring complete and dominant resistance to the barley yellow dwarf virus. Theor. Appl. Genet. 2009, 119, 837–849. [Google Scholar] [CrossRef]
- Del Blanco, I.A.; Hegarty, J.; Gallagher, L.; Falk, B.W.; Brown-Guedira, G.; Pellerin, E.; Dubcovsky, J. Mapping of QTL for tolerance to cereal yellow dwarf virus in two-rowed spring barley. Crop Sci. 2014, 54, 1468–1475. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Larkin, P.J.; Schwinghamer, M.W.; Coutts, B.A.; Birchall, C.; Johnson, P.; Westmore, G.; Davey, B. Studies on barley yellow dwarf virus (BYDV) in wheat. In Proceedings of the 17th Australian Society of Agronomy Conference: Building Productive, Diverse and Sustainable Landscapes, Hobart, TAS, Australia, 20–24 September 2015; Available online: http://www.agronomyaustraliaproceedings.org/images/sampledata/2015_Conference/pdf/agronomy2015final00295.pdf (accessed on 12 September 2021).
- Wang, M.B.; Abbott, D.C.; Waterhouse, P.M. A single copy of a virus-derived transgene encoding hairpin RNA gives immunity to barley yellow dwarf virus. Mol. Plant Pathol. 2000, 1, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.A.C. Progress in Understanding Wheat Disease Epidemiology and Management in Western Australia; Department of Primary Industries and Regional Development Internal Report; Department of Primary Industries and Regional Development: South Perth, WA, Australia, 2017. [Google Scholar]
- Ellis, M.H.; Silva, T.F.; Stiller, W.N.; Wilson, L.J.; Vaslin, M.F.S.; Sharman, M.; Llewellyn, D.J. Identification of a new polerovirus (family Luteoviridae) associated with cotton bunchy top disease in Australia. Austral. Plant Pathol. 2013, 42, 261–269. [Google Scholar] [CrossRef]
- Jones, R.A.C.; Coutts, B.A.; Mackie, A.E.; Dwyer, G.I. Seed transmission of wheat streak mosaic virus shown unequivocally in wheat. Plant Dis. 2005, 89, 1048–1050. [Google Scholar] [CrossRef]
- Lanoiselet, V.M.; Hind-Lanoiselet, T.L.; Murray, G.M. Studies on the seed transmission of wheat streak mosaic virus. Austral. Plant Pathol. 2008, 37, 584–588. [Google Scholar] [CrossRef]
- Dwyer, G.I.; Gibbs, M.J.; Gibbs, A.J.; Jones, R.A.C. Wheat streak mosaic virus in Australia: Relationship to isolates from the Pacific northwest of the USA and its dispersion via seed transmission. Plant Dis. 2007, 91, 164–170. [Google Scholar] [CrossRef]
- Schiffer, M.; Umina, P.A.; Carew, M.; Hoffmann, A.A.; Rodoni, B.C.; Miller, A.D. The distribution of wheat curl mite (Aceria tosichella Keifer) lineages in Australia and their potential to transmit wheat streak mosaic virus. Ann. Appl. Biol. 2009, 155, 371–379. [Google Scholar] [CrossRef]
- Miller, A.D.; Umina, P.A.; Weeks, A.R.; Hoffmann, A.A. Population genetics of the wheat curl mite (Aceria tosichella Keifer) in Australia: Implications for the management of wheat pathogens. Bull. Entomol. Res. 2012, 102, 199–212. [Google Scholar] [CrossRef]
- Oliveira-Hofman, C.; Wegulo, S.N.; Tatineni, S.; Hein, G.L. Impact of wheat streak mosaic virus and triticum mosaic virus coinfection of wheat on transmission rates by wheat curl mites. Plant Dis. 2015, 99, 1170–1174. [Google Scholar] [CrossRef]
- Singh, K.; Wegulo, S.N.; Skoracka, A.; Kundu, J.K. Wheat streak mosaic virus: A century old virus with rising importance worldwide. Mol. Plant Pathol. 2018, 19, 2193–2206. [Google Scholar] [CrossRef]
- Fahim, M.; Ayala-Navarrete, L.I.; Millar, A.A.; Larkin, P.J. Hairpin RNA derived from viral NIa gene confers immunity to Wheat streak mosaic virus infection in transgenic wheat plants. Plant Biotechnol. J. 2010, 8, 821–834. [Google Scholar] [CrossRef] [PubMed]
- Fahim, M.; Mechanicos, A.A.; Ayala-Navarrete, L.I.; Haber, S.; Larkin, P.J. Resistance to wheat streak mosaic virus– a survey of resources and development of molecular markers. Plant Pathol. 2012, 61, 425–440. [Google Scholar] [CrossRef]
- Fahim, M.; Millar, A.A.; Wood, C.C.; Larkin, P.J. Resistance to wheat streak mosaic virus generated by expression of an artificial polycistronic microRNA in wheat. Plant Biotechnol. J. 2012, 10, 150–163. [Google Scholar] [CrossRef]
- Fahim, M.; Larkin, P.J. Designing Effective amiRNA and Multimeric amiRNA against Plant Viruses. In siRNA Design: Methods and Protocols; Taxman, D.J., Ed.; Humana Press: Totowa, NJ, USA, 2013; pp. 357–377. [Google Scholar]
- Richardson, K.; Miller, A.D.; Hoffmann, A.A.; Larkin, P.J. Potential new sources of wheat curl mite resistance in wheat to prevent the spread of yield-reducing pathogens. Exp. Appl. Acarol. 2014, 64, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, G.I.; Welsh, B.A.; Kehoe, M.A.; Jones, R.A.C. Development of a Seed Test for Wheat Streak Mosaic Virus in Bulk Samples of Wheat. Agribusiness Crop Updates 2007; Burswood Entertainment Complex: Perth, WA, Australia, 2007; pp. 96–98. [Google Scholar]
- Cox, B.A.; Luo, H.; Jones, R.A.C. Development of a quantitative, bulk seedling test for seed transmission of Wheat streak mosaic virus in wheat seed samples using real-time PCR. In Proceedings of the 10th Australasian Plant Virology Workshop, Hammer Springs, New Zealand, 19–22 November 2012; p. 24. [Google Scholar]
- Congdon, B.S.; Coutts, B.A.; Renton, M.; Jones, R.A.C. Pea seed-borne mosaic virus: Stability and wind-mediated contact transmission in field pea. Plant Dis. 2016, 100, 953–958. [Google Scholar] [CrossRef] [PubMed]
- Taylor, R.H.; Pares, R.D. The relationship between sugarcane mosaic virus and mosaic viruses of maize and Johnson grass in Australia. Aust. J. Agric. Res. 1968, 19, 767–773. [Google Scholar] [CrossRef]
- Teakle, D.S.; Grylls, N.E. Four strains of sugarcane mosaic virus infecting cereals and other grasses in Australia. Aust. J. Agric. Res. 1973, 24, 465–477. [Google Scholar] [CrossRef]
- Shukla, D.D.; Teakle, D.S. Johnsongrass mosaic virus. In Descriptions of Plant Viruses; Association of Applied Biologists: Wellesbourne, UK, 1989; Volume 340. [Google Scholar]
- Shukla, D.D.; Gough, K.H.; Ward, C.W. Coat protein of potyviruses. Arch. Virol. 1987, 96, 59–74. [Google Scholar] [CrossRef]
- Shepherd, R.J.; Holdeman, Q.L. Seed transmission of the Johnson grass strain of the sugarcane mosaic virus in corn. Plant Dis. Rep. 1965, 49, 468–469. [Google Scholar]
- Persley, D.M. Maize dwarf mosaic disease. Qld Agric. J. 1976, 102, 531–532. [Google Scholar]
- Penrose, L.J. Distribution of a Johnson grass strain of sugarcane mosaic virus in New South Wales and studies of the host range of the Johnson grass and sugarcane strains. Aust. J. Agric. Res. 1974, 25, 99–104. [Google Scholar] [CrossRef]
- Gough, K.H.; Shukla, D.D. Nucleotide sequence of johnsongrass mosaic potyvirus genomic RNA. Intervirology 1993, 36, 181–192. [Google Scholar] [CrossRef]
- Conde, B.D.; Moore, R.F.; Fletcher, D.S.; Teakle, D.S. Inheritance of the resistance of Krish sorghum to sugarcane mosaic virus. Aust. J. Agric. Res. 1976, 27, 45–52. [Google Scholar] [CrossRef]
- Persley, D.M.; Greber, R.S.; Henzell, R.G. Isolates of sugarcane mosaic virus—Johnson grass strain infecting Krish resistant grain sorghum genotypes in Australia. Sorghum Newsl. 1987, 30, 72–73. [Google Scholar]
- Teakle, D.S.; Pritchard, A.J. Resistance of Krish sorghum to four strains of sugarcane mosaic virus in Queensland. Plant Dis. Rep. 1971, 55, 596–598. [Google Scholar]
- Persley, D.M.; Henzell, R.G.; Greber, R.S.; Teakle, D.S.; Toler, R.W. Use of set of differential sorghum inbred lines to compare isolates of sugarcane mosaic virus from sorghum and maize in nine countries. Plant Dis. 1985, 69, 1046–1049. [Google Scholar] [CrossRef]
- Monaghan, N. Problems caused by Sorghum halepense in Australia. Int. J. Pest Manag. 1978, 24, 172–176. [Google Scholar]
- Ryley, M.J.; Persley, D.M.; Jordan, D.R.; Henzell, R.G. Status of sorghum and pearl millet diseases in Australia. In Sorghum and Millet Diseases; Leslie, J.F., Ed.; Iowa State Press: Iowa City, IA, USA, 2003. [Google Scholar]
- Persley, D.M.; Moore, R.F.; Fletcher, D.S. The inheritance of the red leaf reaction of grain sorghum to sugarcane mosaic virus infection. Aust. J. Agric. Res. 1977, 28, 853–858. [Google Scholar] [CrossRef]
- Grogan, P.W.; Teakle, D.S. Resistance of some Australian inbred maize lines to maize dwarf mosaic disease. Aust. J. Exp. Agric. 1969, 9, 541–544. [Google Scholar] [CrossRef]
- Laidlaw, H.K.; Persley, D.M.; Pallaghy, C.K.; Godwin, I.D. Development of durable johnsongrass mosaic virus (JGMV) resistance in sorghum via a transgenic approach. In Sorghum and Millet Diseases; Iowa State Press: Iowa City, IA, USA, 2003; pp. 473–474. [Google Scholar]
- Henzell, R.G.; Persley, D.M.; Greber, R.S.; Fletcher, D.S.; Slobbe, L.V. Development of grain sorghum lines with resistance to sugarcane mosaic and other sorghum diseases. Plant Dis. 1982, 66, 900–901. [Google Scholar] [CrossRef]
- Laidlaw, H.K.; Persley, D.M.; Pallaghy, C.K.; Godwin, I.D. Sequence diversity in the coat protein coding region of the genome RNA of Johnsongrass mosaic virus in Australia. Arch. Virol. 2004, 149, 1633–1641. [Google Scholar] [CrossRef]
- Kerns, M.R.; Pataky, J.K. Reactions of sweet corn hybrids with resistance to maize dwarf mosaic. Plant Dis. 1997, 81, 460–464. [Google Scholar] [CrossRef] [PubMed]
- Signoret, P.A. Cereal Viruses: Maize/Corn. In Encyclopedia of Virology, 3rd ed.; Mahy, B.W., van Regenmortel, M.H., Eds.; Academic Press: Cambridge, MA, USA, 2008; pp. 475–482. [Google Scholar]
- Coutts, B.A.; Cox, B.A.; Thomas, G.J.; Jones, R.A.C. First report of wheat mosaic virus infecting wheat in Western Australia. Plant Dis. 2014, 98, 285. [Google Scholar] [CrossRef] [PubMed]
- Forster, R.L.; Seifers, D.L.; Strausbaugh, C.A.; Jensen, S.G.; Ball, E.M.; Harvey, T.L. Seed transmission of the High Plains virus in sweet corn. Plant Dis. 2001, 85, 696–699. [Google Scholar] [CrossRef]
- Tatineni, S.; Hein, G.L. High Plains wheat mosaic virus: An enigmatic disease of wheat and corn causing the High Plains disease. Mol. Plant Pathol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Nancarrow, N.; Aftab, M.; Zheng, L.; Maina, S.; Freeman, A.J.; Rodoni, B.C.; Spackman, M.E.; Trębicki, P. First report of barley virus G in Australia. Plant Dis. 2019, 103, 1799. [Google Scholar] [CrossRef]
- Greber, R.S. Barley stripe mosaic virus on Cape barley in Queensland. QLD J. Agric. Anim. Sci. 1971, 28, 121–129. [Google Scholar]
- Shivas, R.G.; Williamson, P.M.; Jones, R.A.C. Barley stripe mosaic virus in the Western Australian barley germplasm collection. Aust. Plant Pathol. 1989, 18, 29–31. [Google Scholar] [CrossRef]
- Greber, R.S. Maize sterile stunt—A delphacid transmitted rhabdovirus disease affecting some maize genotypes in Australia. Aust. J. Agric. Res. 1982, 33, 13–23. [Google Scholar] [CrossRef]
- Grylls, N.E. A striate mosaic virus disease of grasses and cereals in Australia, transmitted by the cicadellid Nesoclutha obscura. Aust. J. Agric. Res. 1963, 14, 143–153. [Google Scholar] [CrossRef]
- Greber, R.S. Maize stripe disease in Australia. Aust. J. Agric. Res. 1981, 32, 27–36. [Google Scholar] [CrossRef]
- Thompson, J.P.; Clewett, T.G.; Jennings, R.E.; Sheedy, J.G.; Owen, K.J.; Persley, D.M. Detection of Polymyxa graminis in a barley crop in Australia. Aust. Plant Pathol. 2010, 40, 66–75. [Google Scholar] [CrossRef]
- Cox, B.A.; Luo, H.; Jones, R.A.C. Polymyxa graminis isolates from Australia: Identification in wheat roots and soil, molecular characterization, and wide genetic diversity. Plant Dis. 2014, 98, 1567–1575. [Google Scholar] [CrossRef]
- Lebas, B.S.M.; Ochoa Corona, F.M.; Elliott, D.R.; Tang, J.; Blouin, A.G.; Timudo, O.E.; Ganev, S.; Alexander, B.J.R. Investigation of an outbreak of soil-borne wheat mosaic virus in New Zealand. Aust. Plant Pathol. 2009, 38, 85–90. [Google Scholar] [CrossRef]
- Australian Oilseeds Federation. Australian Oilseeds Industry. Industry Facts and Figures. 2020. Available online: http://www.australianoilseeds.com/oilseeds_industry/industry_facts_and_figures (accessed on 9 September 2021).
- Encyclopaedia Britannica, 8th ed. 2009. Available online: https://www.britannica.com (accessed on 9 September 2021).
- Johnstone, G.R.; Duffus, J.E. Some luteovirus diseases in Tasmania caused by beet western yellows and subterranean clover red leaf viruses. Aust. J. Agric. Res. 1984, 35, 821–830. [Google Scholar] [CrossRef]
- Coutts, B.A.; Jones, R.A.C. Viruses infecting canola (Brassica napus) in south-west Australia: Incidence, distribution, spread and infection reservoir in wild radish (Raphanus raphanistrum). Aust. J. Agric. Res. 2000, 51, 925–936. [Google Scholar] [CrossRef]
- Hertel, K.; Schwinghamer, M.W.; Bambach, R.W. Virus Diseases in Canola and Mustard. Agnote DPI 495, 1st ed. NSW Department of Primary Industries. 2004. Available online: http://www.australianoilseeds.com/__data/assets/pdf_file/0017/4436/Virus_in_canola.pdf (accessed on 12 September 2021).
- Coutts, B.A.; Hawkes, J.R.; Jones, R.A.C. Occurrence of beet western yellows virus and its aphid vectors in over-summering broad-leafed weeds and volunteer crop plants in the grainbelt region of south-western Australia. Aust. J. Agric. Res. 2006, 57, 975–982. [Google Scholar] [CrossRef]
- Coutts, B.A.; Walsh, J.A.; Jones, R.A.C. Evaluation of resistance to turnip mosaic virus in Australian Brassica napus genotypes. Aust. J. Agric. Res. 2007, 58, 67–74. [Google Scholar] [CrossRef]
- Coutts, B.A.; Webster, C.G.; Jones, R.A.C. Control of beet western yellows virus in Brassica napus crops: Infection resistance in Australian genotypes and effectiveness of imidacloprid seed dressing. Crop Pasture Sci. 2010, 61, 321–330. [Google Scholar] [CrossRef]
- Kehoe, M.A.; Coutts, B.A.; Jones, R.A.C. Resistance phenotypes in diverse accessions, breeding lines, and cultivars of three mustard species inoculated with turnip mosaic virus. Plant Dis. 2010, 94, 1290–1298. [Google Scholar] [CrossRef]
- Schwinghamer, M.W.; Schilg, M.A.; Walsh, J.A.; Bambach, R.W.; Cossu, R.M.; Bambridge, J.M.; Hind-Lanoiselet, T.L.; McCorkell, B.E.; Cross, P. Turnip mosaic virus: Potential for crop losses in the grain belt of New South Wales, Australia. Aust. Plant Pathol. 2014, 43, 663–678. [Google Scholar] [CrossRef]
- Van Leur, J.A.G.; Aftab, M.; Sharman, M.; Lindbeck, K. Viral Diseases in Canola and Winter Pulses. GRDC Update Papers. 2014. Available online: http://www.grdc.com.au/Research-and-Development/GRDC-Update-Papers/2014/02/Viral-diseases-in-canola-and-winter-pulses (accessed on 12 September 2021).
- Aftab, M.; van Leur, J.A.G.; Davidson, J.; Freeman, A.J.; Rodoni, B.C.; Trebicki, P. Beet western yellows virus in canola crops: High infection in south-eastern Australia. In Proceedings of the 20th Australasian Plant Pathology Conference, Fremantle, WA, Australia, 14–16 September 2015; p. 115. [Google Scholar]
- Nyalugwe, E.P.; Barbetti, M.J.; Jones, R.A.C. Studies on resistance phenotypes to turnip mosaic virus in five species of Brassicaceae, and identification of a virus resistance gene in Brassica juncea. Eur. J. Plant Pathol. 2015, 141, 647–666. [Google Scholar] [CrossRef]
- Nyalugwe, E.P.; Jones, R.A.C.; Barbetti, M.J.; Kehoe, M.A. Biological and molecular variation amongst Australian turnip mosaic virus isolates. Plant Pathol. 2015, 64, 1215–1223. [Google Scholar] [CrossRef]
- Guerret, M.G.L.; Nyalugwe, E.P.; Maina, S.; Barbetti, M.J.; van Leur, J.A.G.; Jones, R.A.C. Biological and molecular properties of a turnip mosaic virus (TuMV) strain that breaks TuMV resistances in Brassica napus. Plant Dis. 2017, 101, 674–683. [Google Scholar] [CrossRef]
- Congdon, B.S.; Matson, P.; Begum, F.; Kehoe, M.A.; Coutts, B.A. Application of loop-mediated isothermal amplification in an early warning system for epidemics of an externally sourced plant virus. Plants 2019, 8, 139. [Google Scholar] [CrossRef] [PubMed]
- Congdon, B.S.; Baulch, J.R.; Coutts, B.A. Impact of turnip yellows virus infection on seed yield of an open-pollinated and hybrid canola cultivar when inoculated at different growth stages. Virus Res. 2020, 277, 197847. [Google Scholar] [CrossRef]
- Congdon, B.S.; Baulch, J.R.; Coutts, B.A. Novel sources of turnip yellows virus resistance in Brassica and impacts of temperature on their durability. Plant Dis. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kehoe, M.A.; Coutts, B.A. Turnip yellows virus and soybean dwarf virus in Western Australia. Aust. Plant Pathol. 2019, 48, 323–329. [Google Scholar] [CrossRef]
- Nyalugwe, E.P.; Barbetti, M.J.; Clode, P.L.; Jones, R.A.C. Systemic hypersensitive resistance to turnip mosaic virus in Brassica juncea is associated with multiple defense responses, especially phloem necrosis and xylem occlusion. Plant Dis. 2016, 100, 1261–1270. [Google Scholar] [CrossRef]
- Sharman, M. Epidemiology and Genetic Diversity of Tobacco Streak Virus and Related Subgroup 1 Ilarviruses. Ph.D. Thesis, University of Queensland, Brisbane, QLD, Australia, 2015. [Google Scholar]
- Sharman, M.; Persley, D.M.; Thomas, J.E. Distribution in Australia and seed transmission of tobacco streak virus in Parthenium hysterophorus. Plant Dis. 2009, 93, 708–712. [Google Scholar] [CrossRef] [PubMed]
- Sharman, M.; Thomas, J.E.; Persley, D.M. First report of tobacco streak virus in sunflower (Helianthus annuus), cotton (Gossypium hirsutum), chickpea (Cicer arietinum) and mung bean (Vigna radiata) in Australia. Aust. Plant Dis. Notes 2008, 3, 27–29. [Google Scholar] [CrossRef]
- Sharman, M.; Thomas, J.E.; Persley, D.M. Natural host range, thrips and seed transmission of distinct tobacco streak virus strains in Queensland, Australia. Ann. Appl. Biol. 2015, 167, 197–207. [Google Scholar] [CrossRef]
- Sharman, M.; Pagendam, D.E.; Persley, D.M.; Drenth, A.; Thomas, J.E. Field evaluation of tolerance to tobacco streak virus in sunflower germplasm, and observations of seasonal disease spread. Ann. Appl. Biol. 2016, 168, 390–399. [Google Scholar] [CrossRef]
- Anonymous. Cotton bunchy top update. In Cotton Information Sheet; Australian Cotton Co-operative Research Centre: Narrabri, NSW, Australia, 1999; pp. 1–4. [Google Scholar]
- Reddall, A.; Ali, A.; Able, J.A.; Stonor, J.; Tesoriero, L.; Wright, P.R.; Rezaian, M.A.; Wilson, L.J. Cotton bunchy top: An aphid and graft transmitted cotton disease. Aust. Plant Pathol. 2004, 33, 197–202. [Google Scholar] [CrossRef]
- Ali, A.; Reddall, A.; Roberts, J.; Wilson, L.J.; Rezaian, M.A. Cytopathology, mode of aphid transmission and search for the causal agent of cotton bunchy top disease. J. Phytopathol. 2007, 155, 220–227. [Google Scholar] [CrossRef]
- Sharman, M.; Wilson, L.J.; Smith, T.; Grundy, P.; Webb, M. Cotton bunchy top disease and related biosecurity threats. In The Australian Cotton Grower; Greenmount Press: Toowoomba, QLD, Australia, 2014; pp. 30–31. [Google Scholar]
- Duffus, J.E.; Johnstone, G.R. The probable long association of beet western yellows virus with the potato leaf roll syndrome in Tasmania. Aust. J. Exp. Agric. 1982, 22, 353–356. [Google Scholar] [CrossRef]
- Johnstone, G.R.; Ashby, J.W.; Gibbs, A.J.; Duffus, J.E.; Thottappilly, G.; Fletcher, J.D. The host ranges, classification and identification of eight persistent aphid-transmitted viruses causing diseases in legumes. Neth. J. Plant Pathol. 1984, 90, 225–245. [Google Scholar] [CrossRef]
- Johnstone, G.R.; Duffus, J.E.; Guy, P.L. New records on the occurrence of beet western yellows virus in Australia, New Zealand and Mexico. Aust. J. Agric. Res. 1989, 40, 353–358. [Google Scholar] [CrossRef]
- Guy, P.L. Beet western yellow virus infection of Cardamine spp. and Cheesemania radicata. Aust. Plant Pathol. 1987, 16, 43–44. [Google Scholar] [CrossRef]
- Skotnicki, M.L.; Skotnicki, A.H.; Gibbs, A.J.; Mackenzie, A.M.; Torronen, M. Detection of beet western yellows virus by nucleic acid hybridization. Microbios 1987, 49, 17–24. [Google Scholar]
- Latham, L.J.; Smith, L.J.; Jones, R.A.C. Incidence of three viruses in vegetable brassica plantings and wild radish weeds in south-west Australia. Aust. Plant Pathol. 2003, 32, 387–391. [Google Scholar] [CrossRef]
- Sharman, M.; van Leur, J.A.G.; Aftab, M.; Thomas, J.T. Virus diseases of chickpeas and pulse crops in Australia. In Proceedings of the 5th International Working Group on Legume and Vegetable Viruses, Haarlem, The Netherlands, 30 August–3 September 2015; Available online: https://agris.fao.org/agris-search/search.do?recordID=AU2019A03607 (accessed on 12 September 2021).
- Nancarrow, N.; Trebicki, P.; Aftab, M. Turnip Yellows Virus in Western Victoria. Agriculture Victoria: Horsham, VIC Australia, 2020. Available online: https://extensionaus.com.au/FieldCropDiseasesVic/tuyv-western-vic. (accessed on 20 August 2021).
- Coutts, B.A.; Jones, R.A.C. Incidence and distribution of viruses infecting cucurbit crops in the Northern Territory and Western Australia. Aust. J. Agric. Res. 2005, 56, 847–858. [Google Scholar] [CrossRef]
- Coutts, B.A.; Jones, R.A.C.; Umina, P.A.; Davidson, J.; Baker, G.; Aftab, M. Beet western yellows virus (synonym: Turnip yellows virus) and green peach aphid in canola. In Proceedings of the 2015 Grains Research and Development Updates, Perth, WA, Australia, 10–11 February 2015; Available online: https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2015/02/beet-western-yellows-virus-synonym-turnip-yellows-virus-and-green-peach-aphid-in-canola (accessed on 12 September 2021).
- Maling, T.; Diggle, A.J.; Thackray, D.J.; Siddique, K.H.M.; Jones, R.A.C. An epidemiological model for externally acquired vector-borne viruses applied to beet western yellows virus in Brassica napus crops in a Mediterranean-type environment. Crop Pasture Sci. 2010, 61, 132–144. [Google Scholar] [CrossRef]
- Freeman, A.J.; Aftab, M. Effective management of viruses in pulse crops in south eastern Australia should include management of weeds. Aust. Plant Pathol. 2011, 40, 430–441. [Google Scholar] [CrossRef]
- Wilson, C.R.; Lambert, S.J.; Dann, A.; Cross, P.; Hay, F.S. Occurrence of viruses within Tasmanian vegetable crops and identification of a novel polerovirus infecting pea. Aust. Plant Pathol. 2012, 41, 311–319. [Google Scholar] [CrossRef]
- Congdon, B.S.; Matson, P.; Begum, F.; Dore, A.; Kehoe, M.A.; Coutts, B.A. Turnip Yellows Virus Epidemic in 2018—Time to Get One Step Ahead of the Green Peach Aphid; 2019 GRDC Research Updates; GRDC: Perth, WA, Australia, 2019. [Google Scholar]
- Filardo, F.F.; Nancarrow, N.; Kehoe, M.A.; McTaggart, A.R.; Congdon, B.S.; Kumari, S.G.; Aftab, M.; Trebicki, P.; Rodoni, B.C.; Thomas, J.E.; et al. Genetic diversity and recombination between turnip yellows virus strains in Australia. Arch. Virol. 2021, 166, 813–829. [Google Scholar] [CrossRef]
- Filardo, F.F.; Sharman, M. Siratro latent polerovirus (SLPV): A new polerovirus from Australia with a non-functional open reading frame 0. Aust. Plant Pathol. 2019, 48, 491–501. [Google Scholar] [CrossRef]
- Mayo, M.A. ICTV at the Paris ICV: Results of the plenary session and the binomial ballot. Arch. Virol. 2002, 147, 2254–2260. [Google Scholar] [CrossRef]
- Schliephake, E.; Graichen, K.; Rabenstein, F. Investigations on the vector transmission of the beet mild yellowing virus (BMYV) and the turnip yellows virus (TuYV). J. Plant Dis. Prot. 2000, 107, 81–87. [Google Scholar]
- Kyriakou, A.; Close, R.C.; Ashby, J.W. A strain of beet western yellows virus in Canterbury, New Zealand. N. Z. J. Agric. Res. 1983, 26, 271–277. [Google Scholar] [CrossRef]
- Henry, F.; Aftab, M.; Trebicki, P.; Nuttall, J.; Wallace, A. Viruses in Spring-Sown Dual-Purpose Canola in the High Rainfall Zone of Southern Victoria. 2018 GRDC Research Updates, Bendigo, Australia. 2018. Available online: http://agronomyaustraliaproceedings.org/images/sampledata/2017/74_ASA2017_Henry_Frank_FinalL.pdf (accessed on 12 September 2021).
- Congdon, B.S.; Kehoe, M.A.; Filardo, F.F.; Coutts, B.A. In-field capable loop-mediated isothermal amplification detection of turnip yellows virus in plants and its principal aphid vector Myzus persicae. J. Virol. Methods 2019, 265, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Thackray, D.J.; Hawkes, J.R.; Jones, R.A.C. Forecasting aphid and virus risk in canola. In Crop Updates: 2000 Oilseeds Updates; Cox, A., Ed.; Agriculture Western Australia: Perth, WA, Australia, 2000; pp. 36–37. Available online: https://researchlibrary.agric.wa.gov.au/cgi/viewcontent.cgi?article=1005&context=crop_up (accessed on 12 September 2021).
- Thackray, D.J.; Hawkes, J.R.; Jones, R.A.C. Further developments in forecasting aphid and virus risk in canola. In Crop Updates: 2001 Oilseeds Updates; Zaicou-Kunesch, C., Ed.; Agriculture Western Australia: Perth, WA, Australia, 2001; pp. 55–57. [Google Scholar]
- Thackray, D.J.; Hawkes, J.R.; Jones, R.A.C. Influence of climate on aphid outbreaks and virus epidemics in canola. In Crop Updates: 2002 Oilseeds Updates; Ecksteen, D., Ed.; Agriculture Western Australia: Perth, WA, Australia, 2002; pp. 44–46. [Google Scholar]
- Kirkegaard, J.A.; Lilley, J.M.; Morrison, M.J. Drivers of trends in Australian canola productivity and future prospects. Crop Pasture Sci. 2016, 67, 1–9. [Google Scholar] [CrossRef]
- Scanlon, T.T.; Doncon, G. Rain, rain, gone away: Decreased growing-season rainfall for the dryland cropping region of the south-west of Western Australia. Crop Pasture Sci. 2020, 71, 128–133. [Google Scholar] [CrossRef]
- Coutts, B.A.; Jones, R.A.C. Controlling aphids and beet western yellows virus in canola using imidacloprid seed dressing. In Crop Updates: 2005 Oilseeds Updates; Hamilton, D., Ed.; Department of Agriculture: Perth, WA, Australia, 2005; pp. 19–20. [Google Scholar]
- Umina, P.A.; Edwards, O.; Carson, P.; Van Rooyen, A.; Anderson, A. High levels of resistance to carbamate and pyrethroid chemicals widespread in Australian Myzus persicae (Hemiptera: Aphididae) populations. J. Econ. Entomol. 2014, 107, 1626–1638. [Google Scholar] [CrossRef]
- De Little, S.C.; Edwards, O.R.; van Rooyen, A.R.; Weeks, A.; Umina, P.A. Discovery of metabolic resistance to neonicotinoids in green peach aphids (Myzus persicae) in Australia. Pest Manag. Sci. 2017, 73, 1611–1617. [Google Scholar] [CrossRef]
- Umina, P.A. Green Peach Aphid Shows Signs of Low-Level Resistance to Insecticide. GRDC Ground Cover. 2019. Available online: https://groundcover.grdc.com.au/story/6215223/aphids-resistance-signs-starting-to-show-in-australia/ (accessed on 9 September 2021).
- Department of Primary Industries and Regional Development. Managing turnip yellows virus in canola. 2021. Available online: https://www.agric.wa.gov.au/canola/managing-turnip-yellows-virus-canola (accessed on 6 September 2021).
- van Leur, J.A.G.; Lindbeck, K.; Aftab, M.; Freeman, A.J.; McCaffery, D. Virus Development in Canola Crops during 2014 in New South Wales and Implications for the Oilseed and Pulse Industry. In GRDC Research Updates. 2015. Available online: https://grdc.com.au/resources-644and-publications/grdc-update-papers/tab-content/grdc-update-papers/2015/02/virus-645development-in-canola-crops-during-2014 (accessed on 12 September 2021).
- Bus, A.; Korber, N.; Snowdon, R.J.; Stich, B. Patterns of molecular variation in a species-wide germplasm set of Brassica napus. Appl. Genet. 2011, 123, 1413–1423. [Google Scholar] [CrossRef]
- Juergens, M.; Paetsch, C.; Kramer, I.; Zahn, M.; Rabenstein, F.; Schondelmaier, J.; Schliephake, E.; Snowdon, R.; Friedt, W.; Ordon, F. Genetic analyses of the host-pathogen system turnip yellows virus (TuYV)-rapeseed (Brassica napus L.) and development of molecular markers for TuYV-resistance. Theor. Appl. Genet. 2010, 120, 735–744. [Google Scholar] [CrossRef]
- Conroy, R.Y. Black ringspot disease of crucifers. J. Aust. Inst. Agric. Sci. 1959, 25, 64–67. [Google Scholar]
- Letham, D.B.; Daines, D.; Hennissey, J. Turnip mosaic—A serious problem in hybrid cabbage crops. Agric. Gaz. New South Wales 1975, 86, 52–53. [Google Scholar]
- Plant Health Australia. Australian Plant Pest Database. 2001. Available online: www.planthealthaustralia.com.au/appd (accessed on 9 September 2021).
- Walsh, J.A.; Tomlinson, J.A. Viruses infecting winter oilseed rape (Brassica napus ssp. oleifera). Ann. Appl. Biol. 1985, 107, 485–495. [Google Scholar] [CrossRef]
- Edwardson, J.R.; Christie, R.G. CRC Handbook of Viruses Infecting Legumes; CRC Press: Boca Raton, FL, USA, 1991. [Google Scholar]
- Nyalugwe, E.P.; Barbetti, M.J.; Jones, R.A.C. Strain specificity of turnip mosaic virus resistance gene TuRBJU 01 in Brassica juncea. Eur. J. Plant Pathol. 2016, 145, 209–213. [Google Scholar] [CrossRef]
- Nyalugwe, E.P.; Barbetti, M.J.; Jones, R.A.C. Preliminary studies on resistance phenotypes to turnip mosaic virus in Brassica napus and B. carinata from different continents and effects of temperature on their expression. Eur. J. Plant Pathol. 2014, 139, 687–706. [Google Scholar] [CrossRef]
- Jenner, C.E.; Walsh, J.A. Pathotypic variation in turnip mosaic virus with special reference to European isolates. Plant Pathol. 1996, 45, 848–856. [Google Scholar] [CrossRef]
- Yasaka, R.; Ohba, K.; Schwinghamer, M.W.; Fletcher, J.; Ochoa Corona, F.M.; Thomas, J.E.; Ho, S.Y.W.; Gibbs, A.J.; Ohshima, K. Phylodynamic evidence of the migration of turnip mosaic potyvirus from Europe to Australia and New Zealand. J. Gen. Virol. 2014, 96, 701–713. [Google Scholar] [CrossRef]
- Walsh, J.A.; Jenner, C.E. Turnip mosaic virus and the quest for durable resistance. Mol. Plant Pathol. 2002, 3, 289–300. [Google Scholar] [CrossRef]
- Walsh, J.A.; Jenner, C.E. Resistance to Turnip Mosaic Virus in the Brassicaceae. In Natural Resistance Mechanisms of Plants to Viruses; Springer: Dordrecht, The Netherlands, 2006; pp. 415–430. [Google Scholar]
- Hughes, S.L.; Green, S.K.; Lydiate, D.J.; Walsh, J.A. Resistance to turnip mosaic virus in Brassica rapa and B. napus and the analysis of genetic inheritance in selected lines. Plant Pathol. 2002, 51, 567–573. [Google Scholar] [CrossRef]
- Nyalugwe, E.P.; Barbetti, M.J.; Clode, P.L.; Jones, R.A.C. Programmed cell death pathways induced by early plant-virus infection are determined by isolate virulence and stage of infection. Plant Pathol. 2016, 65, 1518–1528. [Google Scholar] [CrossRef]
- Kim, C.K.; Seol, Y.J.; Perumal, S.; Lee, J.; Waminal, N.E.; Jayakodi, M.; Lee, S.C.; Jin, S.; Choi, B.S.; Yu, Y.; et al. Re-exploration of U’s triangle brassica species based on chloroplast genomes and 45S nrDNA sequences. Sci. Rep. 2018, 8, 7353. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.A.; Rusholme Pilcher, R.L.; Hughes, S.L.; Jenner, C.E.; Bambridge, J.M.; Lydiate, D.J.; Green, S.K. Different classes of resistance to turnip mosaic virus in Brassica rapa. Eur. J. Plant Pathol. 2002, 108, 15–20. [Google Scholar] [CrossRef]
- Rusholme, R.L.; Higgins, E.E.; Walsh, J.A.; Lydiate, D.J. Genetic control of broad-spectrum resistance to turnip mosaic virus in Brassica rapa (Chinese cabbage). J. Gen. Virol. 2007, 88, 3177–3186. [Google Scholar] [CrossRef] [PubMed]
- Greber, R.S. Some characteristics of tobacco streak virus isolates from Queensland. Qld. J. Agric. Anim. Sci. 1971, 28, 105–114. [Google Scholar]
- Finlay, J.R. Tobacco streak virus in tobacco. Australas. Plant Pathol. Soc. Newsl. 1974, 3, 71. [Google Scholar] [CrossRef]
- Greber, R.S. Virus diseases of Queensland strawberries and the epidemiological effects of the strawberry runner approval scheme. QLD J. Agric. Anim. Sci. 1979, 36, 93–103. [Google Scholar]
- Sdoodee, R.; Teakle, D.S. Transmission of tobacco streak virus by Thrips tabaci: A new method of plant virus transmission. Plant Pathol. 1987, 36, 377–380. [Google Scholar] [CrossRef]
- Sdoodee, R.; Teakle, D.S. Seed and pollen transmission of tobacco streak virus in tomato (Lycopersicon esculentum cv. Grosse Lisse). Aust. J. Agric. Res. 1988, 39, 469–474. [Google Scholar] [CrossRef]
- Greber, R.S.; Klose, M.J.; Teakle, D.S.; Milne, J.R. High incidence of tobacco streak virus in tobacco and its transmission by Microcephalothrips abdominalis and pollen from Ageratum houstonianum. Plant Dis. 1991, 75, 450. [Google Scholar] [CrossRef]
- Klose, M.J.; Webb, R.; Teakle, D.S. Studies on the association of tobacco streak virus and pollen using an environmental scanning electron microscope and molecular distillation drying techniques. J. Comput. Assist. Microsc. 1992, 4, 213–220. [Google Scholar]
- Sdoodee, R.; Teakle, D.S. Studies on the mechanism of transmission of pollen–associated tobacco streak ilarvirus by Thrips tabaci. Plant Pathol. 1993, 42, 88–92. [Google Scholar] [CrossRef]
- Klose, M.J.; Sdoodee, R.; Teakle, D.S.; Milne, J.R.; Greber, R.S.; Walter, G.H. Transmission of three strains of tobacco streak ilarvirus by different thrips species using virus-infected pollen. J. Phytopathol. 1996, 144, 281–284. [Google Scholar] [CrossRef]
- Sdoodee, R. Biological and Biophysical Properties of Tobacco Streak Virus. Ph.D. Thesis, University of Queensland, Brisbane, Australia, 1989. [Google Scholar]
- Shukla, D.D.; Gough, K.H. Tobacco streak, broad bean wilt, cucumber mosaic, and alfalfa mosaic viruses associated with ring spot of Ajuga reptans in Australia. Plant Dis. 1983, 67, 221–224. [Google Scholar] [CrossRef]
- Sharman, M.; Thomas, J.E. Genetic diversity of subgroup 1 ilarviruses from eastern Australia. Arch. Virol. 2013, 158, 1637–1647. [Google Scholar] [CrossRef]
- Shepherd, A.; Kong, G. Central Queensland Sunflower Disorder—Tobacco Streak Virus. Crop Note. 2006. Available online: https://www.daf.qld.gov.au/__data/assets/pdf_file/0018/50463/CQSFS-Sunflower-TobaccoStreakVirus.pdf (accessed on 7 September 2021).
- Horticulture Innovation Australia Limited. Sunflower. 2010. Available online: https://www.agrifutures.com.au/wp-content/uploads/publications/10-140.pdf (accessed on 8 September 2021).
- Tennakoon, S.B.; Milroy, S.P. Crop and water use efficiency on irrigated farms in Australia. Agric. Water Manag. 2003, 61, 179–194. [Google Scholar] [CrossRef]
- Smith, T.; Wilson, L.J.; Heimoana, S.; Herron, G.A.; Franzmann, B. Overwinter host plants of cotton aphid (Aphis gossypii) and implications for managing abundance and resistance. Proceedings of 13th Australian Cotton Growers Conference: Product, Production, Profit—Progressing our Natural Advantage, Gold Coast, QST, Australia, 8–10 August 2006; pp. 8–10. Available online: http://www.insidecotton.com/xmlui/handle/1/2545 (accessed on 15 September 2021).
- Herron, G.A.; Wilson, L.J. Neonicotinoid resistance in Aphis gossypii Glover (Aphididae: Hemiptera) from Australian cotton. Aust. J. Entomol. 2011, 50, 93–98. [Google Scholar] [CrossRef]
- Sharman, M. Surveillance and monitoring for endemic and exotic virus diseases of cotton. Final Report for project DAQ1201, Cotton Research and Development Corporation. Narrabri New South Wales, Australia. 2015. Available online: http://www.insidecotton.com/xmlui/handle/1/4309. (accessed on 15 September 2021).
- Herron, G.A.; Wilson, L.J. Can resistance management strategies recover insecticide susceptibility in pests? A case study with cotton aphid Aphis gossypii (Aphididae: Hemiptera) in Australian cotton. Aust. Entomol. 2017, 56, 1–13. [Google Scholar] [CrossRef]
- Ellis, M.H.; Stiller, W.N.; Phongkham, T.; Tate, W.A.; Gillespie, V.J.; Gapare, W.J.; Zhu, Q.-H.; Llewellyn, D.J.; Wilson, I.W. Molecular mapping of bunchy top disease resistance in Gossypium hirsutum L. Euphytica 2016, 210, 135–142. [Google Scholar] [CrossRef]
- Fang, D.D.; Xiao, J.; Canci, P.C.; Cantrell, R.G. A new SNP haplotype associated with blue disease resistance gene in cotton (Gossypium hirsutum L.). Theor. Appl. Genet. 2010, 120, 943–953. [Google Scholar] [CrossRef] [PubMed]
- Randles, J.W.; Crowley, N.C. Epidemiology of cauliflower mosaic virus in South Australia. Aust. J. Agric. Res. 1967, 18, 289–298. [Google Scholar] [CrossRef]
- Garrett, R.G.; Martindale, W.A. The occurrence of broccoli necrotic yellows virus in Victoria. Aust. Plant Pathol. 1973, 2, 17–18. [Google Scholar] [CrossRef]
- Schwinghamer, M.W.; Thomas, J.E.; Schilg, M.A.; Parry, J.N.; Dann, E.K.; Moore, K.J.; Kumari, S.G. Mastreviruses in chickpea (Cicer arietinum) and other dicotyledonous crops and weeds in Queensland and northern New South Wales, Australia. Aust. Plant Pathol. 2010, 39, 551–561. [Google Scholar] [CrossRef]
- Thomas, J.E.; Persley, D.M.; McGrath, D.J.; Hibberd, A.M. Virus diseases of tomato and pepper in Queensland and some aspects of their control. Proceedings of International Symposium on Integrated Management Practices, Tainan, Taiwan, 21–28 March 1989; Asian Vegetable Research and Development Center: Tainan, Taiwan. Available online: https://worldveg.tind.io/record/12188/ (accessed on 12 September 2021).
- Harveson, R.M.; Markell, S.G.; Block, C.C.; Gulya, T.J. (Eds.) Compendium of Sunflower Diseases and Pests; APS Press: St Paul, MN, USA, 2016. [Google Scholar]
- Lapidot, M.D.; Guenoune-Gelbart, D.; Leibman, D.; Holdengreber, V.; Davidovitz, M.; Machbash, Z.; Klieman-Shoval, S.; Cohen, S.; Gal-On, A. Pelargonium zonate spot virus is transmitted vertically via seed and pollen in tomato. Phytopathology 2010, 100, 798–804. [Google Scholar] [CrossRef]
- Kamitani, M.; Nagano, A.J.; Honjo, M.N.; Kudoh, H. A survey on plant viruses in natural Brassicaceae communities using RNA-Seq. Microb. Ecol. 2019, 78, 113–121. [Google Scholar] [CrossRef]
- Li, H.; Zhang, C.; Luo, H.; Jones, M.G.K.; Sivasithamparam, K.; Koh, S.; Ong, J.W.L.; Wylie, S.J. Yellow tailflower mild mottle virus and pelargonium zonate spot virus co-infect a wild plant of red-striped tailflower in Australia. Plant Pathol. 2016, 65, 503–509. [Google Scholar] [CrossRef]
- Luo, H.; Wylie, S.J.; Jones, M.G.K. Identification of plant viruses using one-dimensional gel electrophoresis and peptide mass fingerprints. J. Virol. Methods 2010, 165, 297–301. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Persley, D.M.; Steele, V.; Sharman, M.; Campbell, P.; Geering, A.D.W.; Gambley, C. First report of a carlavirus infecting plants in the Fabaceae in Australia. New Dis. Rep. 2020, 41, 26. [Google Scholar] [CrossRef]
- Sharman, M.; Filardo, F.F.; Brier, H.; Gambley, C.; Steele, V.; Persley, D.M. Phytoplasma disease outbreaks in multiple crops in eastern Australia. In Proceedings of the Science Protecting Plant Health, Brisbane, QLD, Australia, 26–28 September 2017; Available online: https://agris.fao.org/agris-search/search.do?recordID=AU2019A05207 (accessed on 9 September 2021).
- Sharman, M.; Brier, H.; Filardo, F.F.; Thompson, S.; Holding, M.; Persley, D.M. Phytoplasma and virus problems in soybean and mungbean crops. 2016. Available online: http://thebeatsheet.com.au/crops/pulses/soybeans/phytoplasma-and-virus-problems-in-soybean-and-mungbean-crops/ (accessed on 9 September 2021).
- Behncken, G.M. The occurrence of peanut mottle virus in Queensland. Aust. J. Agric. Res. 1970, 21, 465–472. [Google Scholar] [CrossRef]
- Persley, D.M.; Thomas, J.E.; Sharman, M. Tospoviruses—An Australian perspective. Aust. Plant Pathol. 2006, 35, 161–180. [Google Scholar] [CrossRef]
- Sharman, M.; Thomas, J.E.; Tree, D.; Persley, D.M. Natural host range and thrips transmission of capsicum chlorosis virus in Australia. Aust. Plant Pathol. 2020, 49, 45–51. [Google Scholar] [CrossRef]
- Srinivasan, R.; Abney, M.R.; Culbreath, A.K.; Kemerait, R.C.; Tubbs, R.S.; Monfort, W.S.; Pappu, H.R. Three decades of managing tomato spotted wilt virus in peanut in southeastern United States. Virus Res. 2017, 241, 203–212. [Google Scholar] [CrossRef]
- Jeggo, M. The Australian perspective, the biosecurity continuum from preborder, to border and postborder. In Improving Food Safety through a One Health Approach: Workshop Summary; National Academies Press: Washington, DC, USA, 2012; p. 198. Available online: https://books.google.com.au/books?hl=en&lr=&id=iv-9DwAAQBAJ&oi=fnd&pg=PA198&dq=The+Australian+perspective,+the+biosecurity+continuum+from+preborder,+to+border+and+postborder (accessed on 12 September 2021).
- Rodoni, B.C.; Zheng, L. Enhanced diagnostic platforms for post entry quarantine (PEQ) and market access; Final Report. CRC, 40050. National Plant Biosecurity Cooperative Research Centre: Geelong, VIC, Australia, 2010. Available online: http://legacy.pbcrc.com.au/sites/all/files/40050%20Final%20Report.pdf (accessed on 19 September 2021).
- Zheng, L.; Tang, J.; Clover, G.R.; Spackman, M.E.; Freeman, A.J.; Rodoni, B.C. Novel genus-specific broad range primers for the detection of furoviruses, hordeiviruses and rymoviruses and their application in field surveys in South-East Australia. J. Virol. Methods 2015, 214, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Maina, S.; Coutts, B.A.; Edwards, O.R.; Almeida, L.; Kehoe, M.A.; Ximenes, A.; Jones, R.A.C. Zucchini yellow mosaic virus populations from East Timorese and northern Australian cucurbit crops: Molecular properties, genetic connectivity, and biosecurity implications. Plant Dis. 2017, 101, 1236–1245. [Google Scholar] [CrossRef]
- Maina, S.; Barbetti, M.J.; Edwards, O.R.; Minemba, D.; Areke, M.W.; Jones, R.A.C. Genetic connectivity between papaya ringspot virus genomes from Papua New Guinea and northern Australia, and new recombination insights. Plant Dis. 2018, 103, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, A.J.; Mackenzie, A.M.; Wei, K.J.; Gibbs, M.J. The potyviruses of Australia. Arch. Virol. 2008, 153, 1411–1420. [Google Scholar] [CrossRef] [PubMed]
- Department of Agriculture, Water and Environment. Northern Australia Quarantine Strategy (NAQS). 2021. Available online: https://www.agriculture.gov.au/biosecurity/australia/naqs (accessed on 6 September 2021).
- Yazdani, M.; Baker, G.; DeGraaf, H.; Henry, K.; Hill, K.; Kimber, B.; Malipatil, M.; Perry, K.; Valenzuela, I.; Nash, M.A. First detection of Russian wheat aphid Diuraphis noxia Kurdjumov (Hemiptera: Aphididae) in Australia: A major threat to cereal production. Aust. Entomol. 2018, 57, 410–417. [Google Scholar] [CrossRef]
- Damsteegt, V.D.; Gildow, F.E.; Hewings, A.D.; Carroll, T.W. A clone of the Russian wheat aphid (Diuraphis noxia) as a vector of barley yellow dwarf, barley stripe mosaic, and brome mosaic viruses. Plant Dis. 1992, 76, 1155–1160. [Google Scholar] [CrossRef]
- Redinbaugh, M.G.; Stewart, L.R. Maize Lethal Necrosis: An emerging, synergistic viral disease. Ann. Rev. Virol. 2018, 5, 301–322. [Google Scholar] [CrossRef]
- Nagaraja, A.; Chethana, B.S.; Jain, A.K. Chapter 7—Biotic stresses and their management. In Millets and Pseudo Cereals; Singh, M., Sood, S., Eds.; Woodhead Publishing: Sawston, UK, 2021; pp. 119–142. [Google Scholar]
- Byamukama, E.; Wegulo, S.N.; Tatineni, S.; Hein, G.L.; Graybosch, R.A.; Baenziger, P.S.; French, R. Quantification of yield loss caused by triticum mosaic virus and wheat streak mosaic virus in winter wheat under field conditions. Plant Dis. 2013, 98, 127–133. [Google Scholar] [CrossRef]
- Jones, R.A.C. Barley Mild Mosaic Virus (BaMMV); In Pest Risk Review—Plant Health Australia Canberra, Australia. 2005. Available online: https://www.planthealthaustralia.com.au/biosecurity/risk-mitigation/risk-assessment/ (accessed on 12 September 2021).
- Kanyuka, K.; Ward, E.; Adams, M.J. Polymyxa graminis and the cereal viruses it transmits: A research challenge. Mol. Plant Pathol. 2003, 4, 393–406. [Google Scholar] [CrossRef]
- Jones, R.A.C. A National diagnostic protocol for soil-borne viruses of wheat. In National Grains Industry Biosecurity Plan; Plant Health Australia Ltd.: Canberra, ACT, Australia, 2004. [Google Scholar]
- Thouvenel, J.-C.; Fauquet, C.; Fargette, D.; Fishpool, L.D.C. Peanut Clump Virus in West Africa. In Viruses with Fungal Vectors; Cooper, J.I., Asher, M.J.C., Eds.; The Association of Applied Biologists: Wellesbourne, UK, 1988; pp. 247–254. [Google Scholar]
- Delfosse, P.; Reddy, A.S.; Legrève, A.; Devi, P.S.; Devi, T.K.T.; Maraite, H.; Reddy, D.V.R. Indian peanut clump virus (IPCV) infection on wheat and barley: Symptoms, yield loss and transmission through seed. Plant Pathol. 1999, 48, 273–282. [Google Scholar] [CrossRef]
- Kühne, T. Soil-borne viruses affecting cereals—Known for long but still a threat. Virus Res. 2009, 141, 174–183. [Google Scholar] [CrossRef]
- Briddon, R.W.; Markham, P.G. Cotton leaf curl virus disease. Virus Res. 2000, 71, 151–159. [Google Scholar] [CrossRef]
- Sequeira, R.V.; Naranjo, S.E. Sampling and management of Bemisia tabaci (Genn.) biotype B in Australian cotton. Crop Prot. 2008, 27, 1262–1268. [Google Scholar] [CrossRef]
- Ray, J.D.; Sharman, M.; Quintao, V.; Rossel, B.; Westaway, J.; Gambley, C. Cotton leafroll dwarf virus detected in Timor-Leste. Aust. Plant Dis. Notes 2016, 11, 29. [Google Scholar] [CrossRef]
- Sharman, M.; Lapbanjob, S.; Sebunruang, P.; Belot, J.L.; Galbieri, R.; Giband, M.; Suassuna, N. First report of cotton leafroll dwarf virus in Thailand using a species-specific PCR validated with isolates from Brazil. Aust. Plant Dis. Notes 2015, 10, 24. [Google Scholar] [CrossRef]
- Reddy, S.V.; Kumar, P.L. Transmission and properties of a new luteovirus associated with chickpea stunt disease in India. Curr. Sci. 2004, 86, 1157–1161. [Google Scholar]
- Pappu, H.R.; Jones, R.A.C.; Jain, R.K. Global status of tospovirus epidemics in diverse cropping systems: Successes achieved and challenges ahead. Virus Res. 2009, 141, 219–236. [Google Scholar] [CrossRef]
- Hill, J.H.; Whitham, S.A. Chapter Seven—Control of Virus Diseases in Soybeans. In Advances in Virus Research; Loebenstein, G., Katis, N., Eds.; Academic Press: Cambridge, MA, USA, 2014; Volume 90, pp. 355–390. [Google Scholar]
- Rimmer, S.R.; Shattuck, V.I.; Buchwaldt, L. Compendium of Brassica Diseases; APS Press: St. Paul, MN, USA, 2007. [Google Scholar]
- Martin, R.R.; Constable, F.; Tzanetakis, I.E. Quarantine regulations and the impact of modern detection methods. Ann. Rev. Phytopathol. 2016, 54, 189–205. [Google Scholar] [CrossRef]
- Maina, S.; Zheng, L.; Rodoni, B.C. Targeted genome sequencing (TG-Seq) approaches to detect plant viruses. Viruses 2021, 13, 583. [Google Scholar] [CrossRef]
- Piper, A.M.; Batovska, J.; Cogan, N.O.; Weiss, J.; Cunningham, J.P.; Rodoni, B.C.; Blacket, M.J. Prospects and challenges of implementing DNA metabarcoding for high-throughput insect surveillance. GigaScience 2019, 8, giz092. [Google Scholar] [CrossRef]
- Macfadyen, S.; Hardie, D.C.; Fagan, L.; Stefanova, K.; Perry, K.D.; DeGraaf, H.E.; Holloway, J.; Spafford, H.; Umina, P.A. Reducing insecticide use in broad-acre grains production: An Australian study. PLoS ONE 2014, 9, e89119. [Google Scholar] [CrossRef]
- Herron, G.A.; Powis, K.; Rophail, J. Insecticide resistance in Aphis gossypii Glover (Hemiptera: Aphididae), a serious threat to Australian cotton. Aust. J. Entomol. 2001, 40, 85–91. [Google Scholar] [CrossRef]
- Fritts, D.A.; Michels, G.J., Jr.; Rush, C.M. The effects of planting date and insecticide treatments on the incidence of high plains disease in corn. Plant Dis. 1999, 83, 1125–1128. [Google Scholar] [CrossRef]
- Silva, A.X.; Bacigalupe, L.D.; Luna-Rudloff, M.; Figueroa, C.C. Insecticide resistance mechanisms in the green peach aphid Myzus persicae (Hemiptera: Aphididae) II: Costs and Benefits. PLoS ONE 2012, 7, e36810. [Google Scholar] [CrossRef]
- Kliot, A.; Ghanim, M. Fitness costs associated with insecticide resistance. Pest Manag. Sci. 2012, 68, 1431–1437. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.S.; Cordeiro, E.M.; Troczka, B.J.; Pym, A.; Mackisack, J.; Mathers, T.C.; Duarte, A.; Legeai, F.; Robin, S.; Bielza, P.; et al. Global patterns in genomic diversity underpinning the evolution of insecticide resistance in the aphid crop pest Myzus persicae. Commun. Biol. 2021, 4, 847. [Google Scholar] [CrossRef]
- Bass, C.; Puinean, A.M.; Andrews, M.; Cutler, P.; Daniels, M.; Elias, J.; Paul, V.L.; Crossthwaite, A.J.; Denholm, I.; Field, L.M.; et al. Mutation of a nicotinic acetylcholine receptor β subunit is associated with resistance to neonicotinoid insecticides in the aphid Myzus persicae. BMC NeuroSci. 2011, 12, 51. [Google Scholar] [CrossRef]
- McGrane, L.; Noakes, F.; Umina, P.A.; Maino, J.; Lye, J.; Arthur, A. Pesticide Resistance in Australian Grain Regions—Lessons to be Learnt. GRDC Research Updates. 2021. Available online: https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2021/02/pesticide-resistance-in-australian-grain-regions-lessons-to-be-learnt (accessed on 6 September 2021).
- Herron, G.A.; Langfield, B.J.; Bogema, D.R.; Chen, Y. Baseline susceptibility and cross-resistance in Aphis gossypii Glover (Aphididae: Hemiptera) to phorate and sulfoxaflor. Aust. Entomol. 2014, 53, 32–35. [Google Scholar] [CrossRef][Green Version]
- Ma, K.; Tang, Q.; Zhang, B.; Liang, P.; Wang, B.; Gao, X. Overexpression of multiple cytochrome P450 genes associated with sulfoxaflor resistance in Aphis gossypii Glover. Pestic. Biochem. Physiol. 2019, 157, 204–210. [Google Scholar] [CrossRef]
- Munkhbayar, O.; Liu, N.; Li, M.; Qiu, X. First report of voltage-gated sodium channel M918V and molecular diagnostics of nicotinic acetylcholine receptor R81T in the cotton aphid. J. Appl. Entomol. 2021, 145, 261–269. [Google Scholar] [CrossRef]
- Hopkinson, J.; Pumpa, S.; van Brunschot, S.; Fang, C.; Frese, M.; Tay, W.T.; Walsh, T. Insecticide resistance status of Bemisia tabaci MEAM1 (Hemiptera: Aleyrodidae) in Australian cotton production valleys. Aust. Entomol. 2020, 59, 202–214. [Google Scholar] [CrossRef]
- Jones, C.M.; Daniels, M.; Andrews, M.; Slater, R.; Lind, R.J.; Gorman, K.; Williamson, M.S.; Denholm, I. Age-specific expression of a P450 monooxygenase (CYP6CM1) correlates with neonicotinoid resistance in Bemisia tabaci. Pestic. Biochem. Physiol. 2011, 101, 53–58. [Google Scholar] [CrossRef]
- Guo, L.; Lv, H.; Tan, D.; Liang, N.; Guo, C.; Chu, D. Resistance to insecticides in the field and baseline susceptibility to cyclaniliprole of whitefly Bemisia tabaci (Gennadius) in China. Crop Prot. 2020, 130, 105065. [Google Scholar] [CrossRef]
- Longhurst, C.; Babcock, J.M.; Denholm, I.; Gorman, K.; Thomas, J.D.; Sparks, T.C. Cross-resistance relationships of the sulfoximine insecticide sulfoxaflor with neonicotinoids and other insecticides in the whiteflies Bemisia tabaci and Trialeurodes vaporariorum. Pest Manag. Sci. 2013, 69, 809–813. [Google Scholar] [CrossRef] [PubMed]
- Herron, G.A.; Gunning, R.V.; Cottage, E.L.; Borzatta, V.; Gobbi, C. Spinosad resistance, esterase isoenzymes and temporal synergism in Frankliniella occidentalis (Pergande) in Australia. Pesticide Biochem. Physiol. 2014, 114, 32–37. [Google Scholar] [CrossRef]
- Zhang, L.; Brown, H. Control of Melon Thrips, Thrips palmi. 2008. Available online: http://www.nt.gov.au/d/Content/File/p/Plant_Pest/753.pdf (accessed on 6 September 2021).
- Umina, P.A.; Reidy-Crofts, J.; Babineau, M.; Maino, J.L.; Edwards, O.R. Susceptibility of the bird cherry-oat aphid, Rhopalosiphum padi (Hemiptera: Aphididae), to four insecticides. Aust. Entomol. 2020, 59, 838–844. [Google Scholar] [CrossRef]
- Chen, M.; Han, Z.; Qiao, X.; Qu, M. Mutations in acetylcholinesterase genes of Rhopalosiphum padi resistant to organophosphate and carbamate insecticides. Genome 2007, 50, 172–179. [Google Scholar] [CrossRef]
- Wang, K.; Bai, J.; Zhao, J.; Su, S.; Liu, L.; Han, Z.; Chen, M. Super-kdr mutation M918L and multiple cytochrome P450s associated with the resistance of Rhopalosiphum padi to pyrethroid. Pest Manag. Sci. 2020, 76, 2809–2817. [Google Scholar] [CrossRef] [PubMed]
- Walsh, L.; Ferrari, E.; Foster, S.; Gaffney, M.T. Evidence of pyrethroid tolerance in the bird cherry-oat aphid Rhopalosiphum padi in Ireland. Outlooks Pest Manag. 2020, 31, 5–9. [Google Scholar] [CrossRef]
- Hallmann, C.A.; Sorg, M.; Jongejans, E.; Siepel, H.; Hofland, N.; Schwan, H.; Stenmans, W.; Müller, A.; Sumser, H.; Hörren, T.; et al. More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 2017, 12, e0185809. [Google Scholar] [CrossRef]
- Wood, T.J.; Goulson, D. The environmental risks of neonicotinoid pesticides: A review of the evidence post 2013. Environ. Sci. Pollut. Res. 2017, 24, 17285–17325. [Google Scholar] [CrossRef]
- Gibbons, D.; Morrissey, C.; Mineau, P. A review of the direct and indirect effects of neonicotinoids and fipronil on vertebrate wildlife. Environ. Sci. Pollut. Res. 2015, 22, 103–118. [Google Scholar] [CrossRef]
- Siviter, H.; Brown, M.J.F.; Leadbeater, E. Sulfoxaflor exposure reduces bumblebee reproductive success. Nature 2018, 561, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Al Naggar, Y.; Baer, B. Consequences of a short time exposure to a sublethal dose of flupyradifurone (Sivanto) pesticide early in life on survival and immunity in the honeybee (Apis mellifera). Sci. Rep. 2019, 9, 19753. [Google Scholar] [CrossRef]
- Lundin, O.; Malsher, G.; Högfeldt, C.; Bommarco, R. Pest management and yield in spring oilseed rape without neonicotinoid seed treatments. Crop Prot. 2020, 137, 105261. [Google Scholar] [CrossRef]
- McNamara, L.; Gauthier, K.; Walsh, L.; Thébaud, G.; Gaffney, M.; Jacquot, E. Management of yellow dwarf disease in Europe in a post-neonicotinoid agriculture. Pest Manag. Sci. 2020, 76, 2276–2285. [Google Scholar] [CrossRef]
- Kalinina, N.O.; Khromov, A.; Love, A.J.; Taliansky, M.E. CRISPR applications in plant virology: Virus resistance and beyond. Phytopathology 2020, 110, 18–28. [Google Scholar] [CrossRef]
- Conyers, M.; van der Rijt, V.; Oates, A.; Poile, G.; Kirkegaard, J.; Kirkby, C. The strategic use of minimum tillage within conservation agriculture in southern New South Wales, Australia. Soil Tillage Res. 2019, 193, 17–26. [Google Scholar] [CrossRef]
- Davis, J.A.; Radcliffe, E.B.; Ragsdale, D.W. Effects of high and fluctuating temperatures on Myzus persicae (Hemiptera: Aphididae). Environ. Entomol. 2006, 35, 1461–1468. [Google Scholar] [CrossRef]
- Nancarrow, N.; Constable, F.E.; Finlay, K.J.; Freeman, A.J.; Rodoni, B.C.; Trebicki, P.; Vassiliadis, S.; Yen, A.L.; Luck, J.E. The effect of elevated temperature on barley yellow dwarf virus-PAV in wheat. Virus Res. 2014, 186, 97–103. [Google Scholar] [CrossRef]
- Trębicki, P.; Nancarrow, N.; Cole, E.; Bosque-Pérez, N.A.; Constable, F.E.; Freeman, A.J.; Rodoni, B.C.; Yen, A.L.; Luck, J.E.; Fitzgerald, G.J. Virus disease in wheat predicted to increase with a changing climate. Glob. Chang. Biol. 2015, 21, 3511–3519. [Google Scholar] [CrossRef] [PubMed]
- Trębicki, P.; Vandegeer, R.K.; Bosque-Pérez, N.A.; Powell, K.S.; Dader, B.; Freeman, A.J.; Yen, A.L.; Fitzgerald, G.J.; Luck, J.E. Virus infection mediates the effects of elevated CO2 on plants and vectors. Sci. Rep. 2016, 6, 22785. [Google Scholar] [CrossRef]
- Trębicki, P.; Nancarrow, N.; Bosque-Pérez, N.A.; Rodoni, B.C.; Aftab, M.; Freeman, A.J.; Yen, A.L.; Fitzgerald, G.J. Virus incidence in wheat increases under elevated CO2: A 4-year study of yellow dwarf viruses from a free air carbon dioxide facility. Virus Rep. 2017, 241, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Australian Bureau of Statistics. Australian Farming and Farmers. 4102.0 Australian Social Trends. 2012. Available online: https://www.abs.gov.au/AUSSTATS/abs@.nsf/Lookup/4102.0Main+Features10Dec+2012 (accessed on 7 September 2021).
- ABC Rural. CPSU Says Funding and Staff Cuts to Department of Agriculture and Food Western Australia Is Threatening the Department’s Future Viability. 2015. Available online: https://www.abc.net.au/news/rural/2015-05-21/cpsu-toni-walkington-dafwa-budget-staff-cuts/6487790 (accessed on 7 September 2021).
- Alston, M. Who is down on the farm? Social aspects of Australian agriculture in the 21st century. Agric. Hum. Values 2004, 21, 37–46. [Google Scholar] [CrossRef]
- Kellock, A. Red-leaf virus—A newly recognized virus disease of subterranean clover (Trifolium subterraneum L.). Aust. J. Agric. Res. 1971, 22, 615–624. [Google Scholar] [CrossRef]
- Severtson, D.; Congdon, B.S.; Valentine, C. Apps, traps and LAMP’s: ‘Smart’ improvements to pest and disease management. In 2018 Grains Research Updates; GRDC: Perth, WA, Australia, 2018. [Google Scholar]
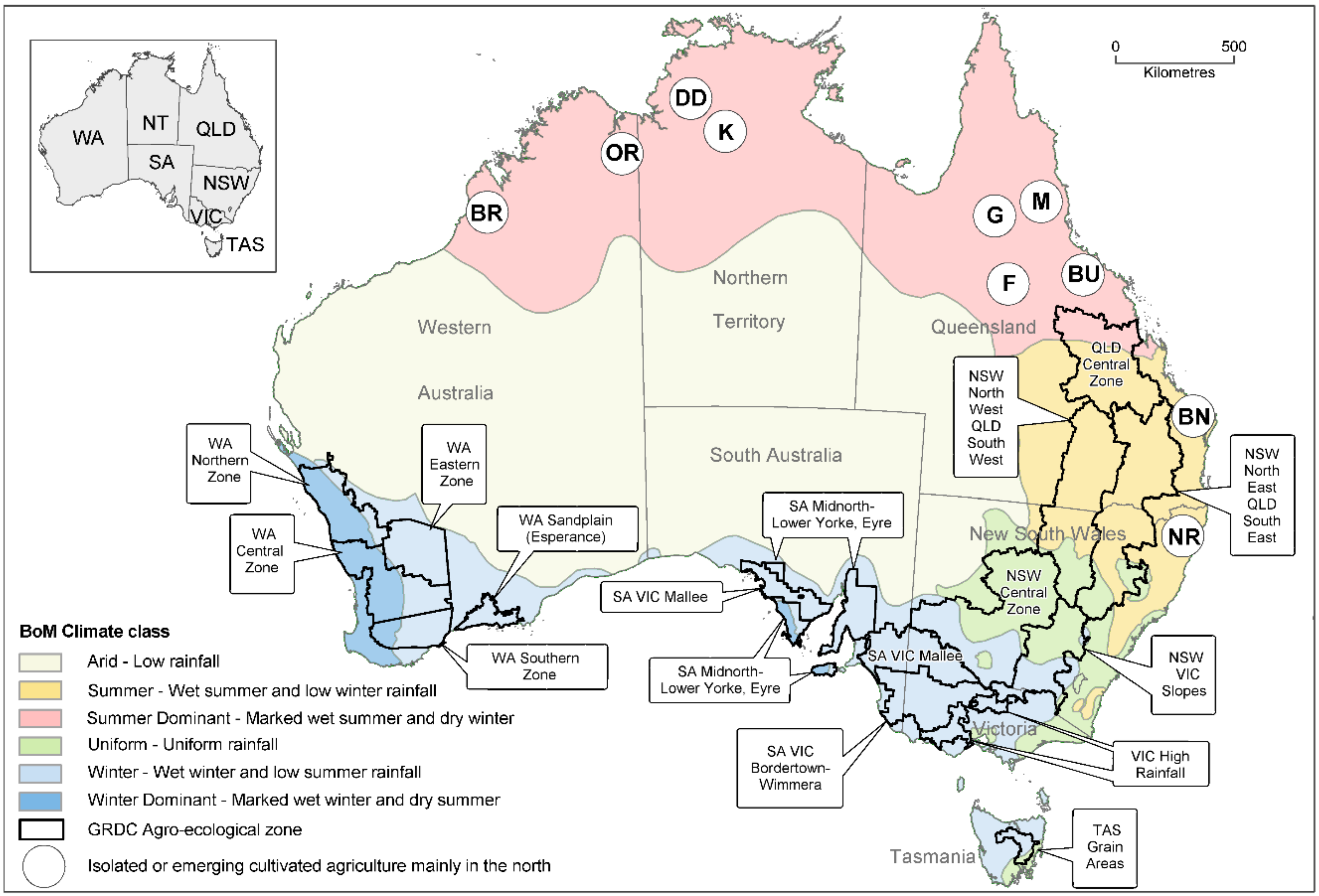
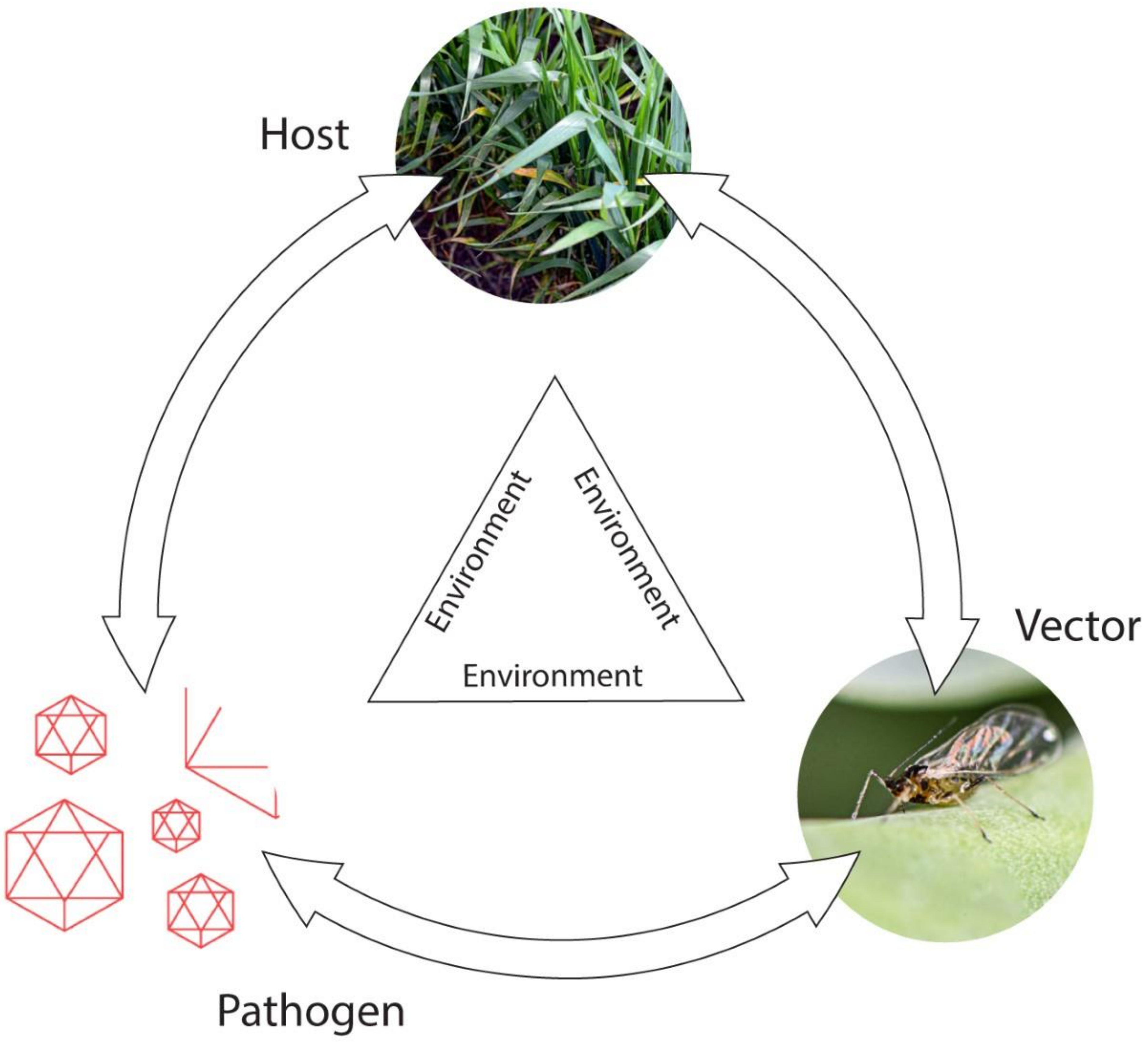





| Method | Measure | Selectivity | Primary Source (Xo) | Rate of Spread (r) | |||
|---|---|---|---|---|---|---|---|
| Low | High | External | Internal | Early | Late | ||
| Phytosanitary, against virus | General hygiene | + | − | + | + | + | − |
| Roguing | + | + | − | + | + | − | |
| Healthy seed, seed sieving, seed therapy | + | − | − | + | + | − | |
| Phytosanitary, against vector and virus | Weed control, soil solarization, fallowing | + | − | − | + | + | − |
| Cultural | Isolation, safe planting distances | + | − | + | − | + | − |
| Planting upwind, non-host barrier, large field size, windbreaks | + | − | + | − | + | − | |
| Manipulate sowing date to avoid peak vector numbers | + | − | − | − | + | − | |
| Mini7mum tillage, groundcover, mulches, reflective surfaces | + | − | − | − | + | − | |
| Early canopy cover, high plant density, narrow row spacing | + | − | + | + | − | + | |
| Early harvest, early maturing cultivar | + | − | − | − | − | + | |
| Crop and weed free periods, single and phased rotations | + | − | + | + | + | − | |
| Chemical, against vector | Specific, regular foliar applications (PTO) | − | + | − | − | + | + |
| General, regular foliar applications (PTO) | + | − | − | − | + | + | |
| Specific, seed dressings (PTO) | − | + | − | − | + | − | |
| General, seed dressings (PTO) | + | − | − | − | + | − | |
| Specific, in furrow or soil before or directly after planting (PTO) | − | + | − | − | + | − | |
| General, in furrow or soil before or directly after planting (PTO) | + | − | − | − | + | − | |
| Natural host resistance, against virus | Partial, polygenic | − | + | − | − | + | (+) |
| Strain-specific, single gene, resistance-breaking strain absent | − | + | − | − | + | + | |
| Extreme resistance, single gene | − | + | − | − | + | + | |
| Natural host resistance, against vector | Partial, polygenic | − | + | − | − | + | (+) |
| Specific, single gene, resistance-breaking vector race absent | − | + | − | − | + | (+) | |
| Crop Type | Virus | Virus Genus | Crops Affected | Main Foliage Symptoms | Vector | Seed Transmission | Maximum % Yield Loss Recorded | Seed Quality Defect | Region Recorded in a |
|---|---|---|---|---|---|---|---|---|---|
| Cereal | Barley yellow dwarf virus (BYDV) | Luteovirus | Barley, wheat, oats, triticale, rye | Chlorosis, reddening, stunting | Aphids | No | 80% | Shriveled grain, reduced seed size, weight and protein content | STE, MED, TE |
| Cereal yellow dwarf virus (CYDV) | Polerovirus | Barley, wheat, oats, triticale, rye | Chlorosis, reddening, stunting | Aphids | No | No data | No data | STE, MED, TE | |
| Johnsongrass mosaic virus (JGMV) | Potyvirus | Maize, sweet corn, sorghum, pearl millet | Mosaic, reddening, necrosis, stunting | Aphids | Yes | 92% | Reduced size | TN, STE | |
| Wheat streak mosaic virus (WSMV) | Tritimovirus | Wheat | Mosaic, chlorosis, streaking, stunting | Mites | Yes | 83% | Shriveled grain | STE, MED, TE | |
| Oilseed | Cotton bunchy top virus (CBTV) | Polerovirus | Cottonseed | Mosaic, deformation, stunting | Aphids | No | 100% | No data | TN, STE |
| Tobacco streak virus (TSV) | Ilarvirus | Peanut, safflower, soybean, sunflower | Chlorosis, deformation, necrosis, stunting, plant death | Thrips | No | 70% | Reduced size | TN, STE, TE | |
| Turnip mosaic virus (TuMV) | Potyvirus | Canola, Indian mustard | Mosaic, stunting | Aphids | No | 84% | Reduced size | STE, MED, TE | |
| Turnip yellows virus (TuYV) | Polerovirus | Canola, Indian mustard | Chlorosis, reddening, stunting | Aphids | No | 46% | Fewer seeds, reduced oil content, increased erucic acid, glucosinolates | STE, MED, TE |
| Crop Type | Virus | Virus Genus | Crops Affected | Main Foliage Symptoms | Vector | Seed Transmission | Maximum % Seed Yield Loss Recorded | Seed Quality Defect | Region Recorded in a |
|---|---|---|---|---|---|---|---|---|---|
| Cereal | Barley stripe mosaic virus | Hordeivrus | Barley, wheat, rye | Stripey mosaic, sometimes necrotic | Contact only | Yes | 35% | Reduced size | STE, MED, TE |
| Barley virus G | Polerovirus | Barley, pearl millet | Mosaic, yellow striping, necrosis | Aphid | No | No data | No data | TE | |
| Barley yellow striate mosaic virus (Synonym: Cereal striate mosaic virus, maize sterile stunt virus) | Nucleorhabdovirus | Barley, maize, wheat, oats, rye | Chlorotic stipes, mosaic, reddening, stunting, sterility | Planthopper | No | No data | No data | TE, STE | |
| Cereal chlorotic mottle virus | Nucleorhabdovirus | Barley, maize, wheat, oats, rye | Chlorotic stipes, stunting and sterility | Leafhopper | No | No data | No data | TE, STE | |
| Chloris striate mosaic virus (Synonym: Wheat striate mosaic) | Mastrevirus | Barley, oats, maize, wheat | Chlorotic steaking, striate mosaic, stunting | Leafhopper | No | No data | No data | TE, STE | |
| High plains wheat mosaic virus (HPWMoV) | Emravairus | Wheat, maize | Mosaic, chlorosis, streaking, stunting | Mites | Yes | No data, | No data, | STE, MED | |
| Maize stripe virus | Tenuivirus | Maize, sweet corn, sorghum | Chlorotic stripes, apical bending, stunting, premature death | Planthopper | No | 76% | No data | TN, STE, MED, TE | |
| Maize yellow dwarf virus (MYDV) | Polerovirus | Maize, sweet corn, barley, oats, wheat | Chlorosis, reddening, stunting | Aphids | No | No data, | No data, | STE, MED, TE | |
| Sugarcane mosaic virus (SMV) | Potyvirus | Maize, sorghum, pearl millet | Mosaic, ringspots, necrosis | Aphid | No | N/A | N/A | TN, STE | |
| Oilseed | Alfalfa mosaic virus (AMV) | Alfamovirus | Soybean | Mosaic, yellow spotting, stunting | Aphid | Yes | 48% | Fewer mottled seeds | STE, MED, TE |
| Broccoli necrotic yellows virus (BNYV) | Cytorhabdovirus | Canola | Asymptomatic | Aphid | No | No data | No data | STE, TE | |
| Capsicum chlorosis virus (CaCV) | Orthotospovirus | Peanut | Mottle, necrotic spotting, reduced internode length, terminal growing points wilt and die | Thrips | No | No data | No data | TN, STE | |
| Cauliflower mosaic virus (CaMV) | Caulimovirus | Canola, Indian mustard | Vein clearing, vein banding, chlorotic rings, mosaic | Aphids | No | 90% | Reduced size | STE, MED, TE | |
| Cowpea mild mottle virus (CpMMV) | Calarvirus | Soybean | Mosaic, leaf distortion, stunting, pod distortion, fewer pods | Whitefly | Yes | 16% | Reduced size | STE | |
| Cucumber mosaic virus (CMV) | Cucumovirus | Sunflower | Mosaic, chlorotic rings, brown streaking of petioles and stems, stunting | Aphid | No data | No data | Shriveled seed | STE | |
| Glycine mosaic virus (GlMV) | Comovirus | Soybean | Mosaic | No data | No data | No data | No data | STE | |
| Lucerne Australian latent virus (LALV) | Nepovirus | Soybean | No data | No data | Yes | No data | No data | STE, TE | |
| Passionfruit woodiness virus (PWV) | Potyvirus | Peanut, soybean | Mosaic, ringspots, leaf deformation | Aphid | No | No data | No data | STE, MED, TN | |
| Peanut mottle virus (PMoV) | Potyvirus | Peanut, soybean | Mottle, leaf distortion, mild stunting | Aphid | Yes | 40% | Reduced size, malformation, discoloration | STE | |
| Pelargonium zonate spot virus (PZSV) | Anulavirus | Sunflower | Bright chlorotic rings | None | Yes (only in tomato) | No data | No data | STE, MED | |
| Soybean mosaic virus (SoMV) | Potyvirus | Soybean | Mosaic, deformation, stunting | Aphid | Yes | 35% (none reported in Australia) | Reduced size, seed coat mottling. | STE, MED, TE | |
| Tomato spotted wilt virus (TSWV) | Orthotospovirus | Peanut | Mosaics, chlorotic or necrotic streaking or spotting, ringspots, line patterns, general necrosis, wilting, stunting | Thrips | No | 100% | Reduced size, cracking of seedcoat | STE, MED, TE, TN | |
| Watermelon mosaic virus (WMV) | Potyvirus | Sesame | N/A | Aphid | No | No data | No data | STE |
| Syndrome | Virus or Virus Complex | Virus Genus | Cereal and Oilseed Crops Affected | Vector | Current Distribution | Disease Impact |
|---|---|---|---|---|---|---|
| Barley mild mosaic disease | Barley mild mosaic virus (BaMMV) | Bymovirus | Barley | Plasmodiophorid protist (Polymyxa graminis) | Europe, East Asia | Major yield losses in some years |
| Barley yellow mosaic disease | Barley yellow mosaic virus (BaYMV) | Bymovirus | Barley | Plasmodiophorid protist (Polymyxa graminis) | Europe, Middle East, East Asia, North America | Major yield losses in some years |
| Chinese wheat mosaic disease | Chinese wheat mosaic virus (CWMV) | Furovirus | Wheat | Plasmodiophorid protist (Polymyxa graminis) | East Asia | Major yield losses in some years |
| Cotton blue disease | Cotton leafroll dwarf virus (CLRDV) | Polerovirus | Cotton | Aphid (Aphis gossypii) | Sub-Saharan Africa, Middle East, Southeast Asia, North and South America | Devastating yield losses; major deterrent to cotton cultivation. Likely losses in grain legumes. |
| Cotton leaf curl disease complex | Cotton leaf curl viruses (CLCuVs) | Begomovirus(es) | Cotton | Whitely (Bemisia tabaci) | Indian subcontinent, East Asia, Southeast Asia, North, East and West Africa | Devastating yield losses; crop failure; major deterrent to cotton cultivation |
| Cotton leaf crumple disease | Cotton leaf crumple virus (CLCrV) | Begomovirus | Cotton | Whitely (Bemisia tabaci) | Indian subcontinent, Central and North America | Major yield losses in epidemic years |
| Groundnut bud necrosis disease | Groundnut bud necrosis virus (GBNV) | Orthotospovirus | Peanut, soybean | Thrips (Thrips palmi and Frankliniella shultzei) | Indian subcontinent, Central and East Asia, Southeast Asia | Severe yield losses with early infection |
| Groundnut rosette tri-partite disease complex (GRD) | Groundnut rosette virus (GRV), groundnut rosette assistor virus (GRAV), and virus satellite | Umbravirus (GRV), Luteovirus (GRAV), virus satellite tri-partite complex | Peanut | Aphid (Aphis craccivora) | Sub-Saharan Africa and offshore islands, including Madagascar | Devastating yield losses in some years, crop failure; major deterrent to peanut cultivation |
| Peanut clump disease | Peanut clump virus (PCV), Indian peanut clump virus (IPCV) | Pecluvirus | Barley, maize, sweet corn, peanut, pearl millet, sorghum, rice, wheat | Plasmodiophorid protist (Polymyxa graminis) | Northern region of Sub-Saharan Africa (PCV), Indian subcontinent (IPCV) | Major yield losses in epidemic years |
| Maize dwarf mosaic disease | Maize dwarf mosaic virus (MDMV) | Potyvirus | Maize, sweet corn, sorghum, oats | Many aphid species (including Rhopalosiphum maidis, Myzus persicae, Aphis craccivora, Aphis gossypii, Brevicoryne brassicae and Rhopalosiphum padi. | Europe, North, Central and South America, North and Sub-Saharan Africa, Middle Est, Central and East Asia, Indian Subcontinent, Southeast Asia | Most important and damaging virus disease of sorghum worldwide. Severe losses in susceptible maize hybrids, and sweet corn |
| Maize lethal necrosis bi-partite disease complex (MLND) | Maize chlorotic mottle virus (MCMV), plus sugarcane mosaic virus (SCMV), maize dwarf mosaic virus (MDMV) or wheat streak mosaic virus (WSMV) | Machlomovirus (MCMV), plus Potyvirus (SCMV, MDMV) or Tritimovirus (WSMV) | Maize, sweet corn (MLND). Barley, maize, sweet corn, sorghum, pearl millet, wheat (MCMV). Several cereal crops (MDMV, SCMV, WSMV) | Beetle and thrips species (including the thrips Frankliniella williamsi) (MCMV). Several aphid species (SCMV, MDMV). Eriophyid mite (Aceria tosichella) (WSMV) | East and Central Africa, East Asia, Southeast Asia, North and South America, Europe | Widespread devastating yield losses, crop failure, food shortages, major deterrent to maize and sweet corn cultivation |
| Maize rough dwarf disease | Maize rough dwarf virus (MRDV) | Fijivirus | Maize, sweet corn | Planthopper (Laodelphax striatellus) | Mediterranean region and Middle East | Devastating yield losses in hybrid maize cultivars; major threat to maize and sweet corn cultivation |
| Maize streak disease | Maize streak mosaic virus (MSMV) | Mastrevirus | Maize, sweet corn | Leafhoppers (Cicadulina mbila, nine other Cicadulina species) | Sub-Saharan Africa | Widespread devastating yield losses in some years, crop failure; major deterrent to maize and sweet corn cultivation |
| Oat mosaic disease | Oat mosaic virus (OMV) | Bymovirus | Oats | Plasmodiophorid protist (Polymyxa graminis) | Europe, North America | Major yield losses in some years |
| Oat golden stripe disease | Oat golden stripe virus (OGSV) | Furovirus | Oats | Plasmodiophorid protist (Polymyxa graminis) | Europe | Major yield losses in some years |
| Rice hoja blanca disease | Rice hoja blanca virus (RHBV) | Tenuivirus | Rice | Planthopper (Sogatodes orizicola) | South, Central and North America | Devastating yield losses in some years, crop failure |
| Rice tungro bi-partite disease complex | Rice tungro bacilliform virus (RTBV),plus rice tungro spherical virus (RTSV) | Tungrovirus (RTBV), Waikavirus (RTSV) | Rice | Several leafhopper vector species transmit both viruses, Nephotettix virescens most efficient vector | Southeast Asia, East Asia (China),the Indian subcontinent | Widespread devastating yield losses, famine. Major deterrent to rice cultivation |
| Rice yellow mottle disease | Rice yellow mottle virus (RYMV) | Sobemovirus | Rice | Contact, beetles, mammals soil and water | East, Central and West Africa, Madagascar | Widespread devastating yield losses; major deterrent to rice cultivation |
| Soil-borne cereal mosaic disease | Soil-borne cereal mosaic virus (SBCMV) | Furovirus | Wheat, rye, triticale | Plasmodiophorid protist Polymyxa graminis | Europe | Major yield losses in some years |
| Soil-borne wheat mosaic disease | Soil-borne wheat mosaic virus (SBWMV) | Furovirus | Wheat, barley, rye, triticale | Plasmodiophorid protist Polymyxa graminis | Europe, New Zealand, Asia, South America, North America | Major yield losses in some years |
| Wheat yellow mosaic disease | Wheat yellow mosaic virus (WYMV) | Bymovirus | Wheat | Plasmodiophorid protist (Polymyxa graminis) | Asia | Major yield losses in some years |
| Wheat spindle streak mosaic disease | Wheat spindle streak mosaic virus (WSSMV) | Bymovirus | Wheat, rye, triticale | Plasmodiophorid protist (Polymyxa graminis) | North America, Europe | Major yield losses in some years |
| Triticum mosaic disease | Triticum mosaic virus (TriMV) | Poacevirus | Barley, wheat | Eryophid mite (Aceria tosichella) | North America | Major losses in mixed infection due to synergistic interaction with wheat streak mosaic virus |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jones, R.A.C.; Sharman, M.; Trębicki, P.; Maina, S.; Congdon, B.S. Virus Diseases of Cereal and Oilseed Crops in Australia: Current Position and Future Challenges. Viruses 2021, 13, 2051. https://doi.org/10.3390/v13102051
Jones RAC, Sharman M, Trębicki P, Maina S, Congdon BS. Virus Diseases of Cereal and Oilseed Crops in Australia: Current Position and Future Challenges. Viruses. 2021; 13(10):2051. https://doi.org/10.3390/v13102051
Chicago/Turabian StyleJones, Roger A. C., Murray Sharman, Piotr Trębicki, Solomon Maina, and Benjamin S. Congdon. 2021. "Virus Diseases of Cereal and Oilseed Crops in Australia: Current Position and Future Challenges" Viruses 13, no. 10: 2051. https://doi.org/10.3390/v13102051
APA StyleJones, R. A. C., Sharman, M., Trębicki, P., Maina, S., & Congdon, B. S. (2021). Virus Diseases of Cereal and Oilseed Crops in Australia: Current Position and Future Challenges. Viruses, 13(10), 2051. https://doi.org/10.3390/v13102051







