Update on the Transmission of Zika Virus Through Breast Milk and Breastfeeding: A Systematic Review of the Evidence
Abstract
1. Introduction
2. Methods
2.1. Study Criteria
2.1.1. Types of Studies
2.1.2. Participants
2.1.3. Types of Exposure
2.1.4. Types of Outcomes
2.2. Search Strategy
2.3. Data Extraction and Management:
2.4. Quality of the Evidence
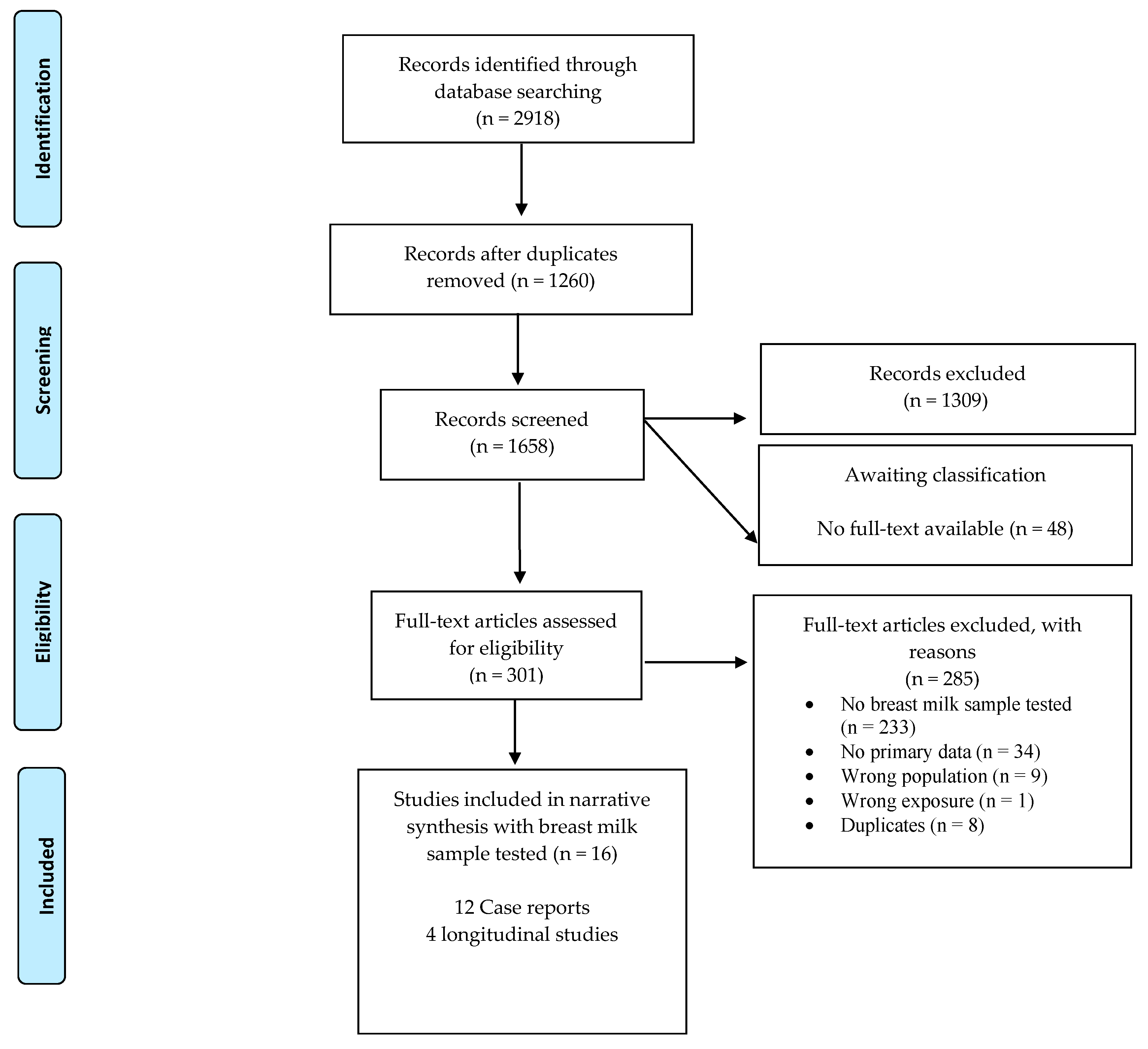
3. Results
3.1. Study Designs
3.2. Settings
3.3. Participants
3.4. Child Outcomes
3.5. Zika Virus in Breast Milk
3.6. Infant Feeding Practices
3.7. Certainty of the Evidence
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Zika Epidemiology Update 2019. Available online: https://www.who.int/emergencies/diseases/zika/zika-epidemiology-update-july-2019.pdf?ua=1 (accessed on 15 January 2021).
- Besnard, M.; Eyrolle-Guignot, D.; Guillemette-Artur, P.; Lastère, S.; Bost-Bezeaud, F.; Marcelis, L.; Abadie, V.; Garel, C.; Moutard, M.L.; Jouannic, J.M.; et al. Congenital cerebral malformations and dysfunction in fetuses and newborns following the 2013 to 2014 Zika virus epidemic in French Polynesia. Eurosurveillance 2016, 21, 30181. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.A.; Staples, J.E.; Dobyns, W.B.; Pessoa, A.; Ventura, C.V.; Da Fonseca, E.B.; Ribeiro, E.M.; Ventura, L.O.; Neto, N.N.; Arena, J.F.; et al. Characterizing the Pattern of Anomalies in Congenital Zika Syndrome for Pediatric Clinicians. JAMA Pediatr. 2017, 171, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Pires, P.; Jungmann, P.; Galvão, J.M.; Hazin, A.; Menezes, L.; Ximenes, R.; Tonni, G.; Júnior, E.A. Neuroimaging findings associated with congenital Zika virus syndrome: Case series at the time of first epidemic outbreak in Pernambuco State, Brazil. Child Nerv. Syst. 2018, 34, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Werner, H.; Sodré, D.; Hygino, C.; Guedes, B.; Fazecas, T.; Nogueira, R.; Daltro, P.; Tonni, G.; Lopes, J.; Júnior, E.A. First-trimester intrauterine Zika virus infection and brain pathology: Prenatal and postnatal neuroimaging findings. Prenat. Diagn. 2016, 36, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Duffy, M.R.; Chen, T.-H.; Hancock, W.T.; Powers, A.M.; Kool, J.L.; Lanciotti, R.S.; Pretrick, M.; Marfel, M.; Holzbauer, S.; DuBray, C.; et al. Zika Virus Outbreak on Yap Island, Federated States of Micronesia. N. Engl. J. Med. 2009, 360, 2536–2543. [Google Scholar] [CrossRef] [PubMed]
- Musso, D.; Bossin, H.; Mallet, H.P.; Besnard, M.; Broult, J.; Baudouin, L.; Levi, J.E.; Sabino, E.C.; Ghawche, F.; Lanteri, M.C.; et al. Zika virus in French Polynesia 2013–14: Anatomy of a completed outbreak. Lancet Infect. Dis. 2018, 18, e172–e182. [Google Scholar] [CrossRef]
- Metsky, H.C.; Matranga, C.B.; Wohl, S.; Schaffner, S.F.; Freije, C.A.; Winnicki, S.M.; West, K.; Quigley, J.E.; Baniecki, M.L.; Gladden-Young, A.; et al. Zika virus evolution and spread in the Americas. Nature 2017, 546, 411–415. [Google Scholar] [CrossRef]
- Dyer, O. Zika virus spreads across Americas as concerns mount over birth defects. BMJ 2015, 351, h6983. [Google Scholar] [CrossRef]
- Besnard, M.; Lastère, S.; Teissier, A.; Cao-Lormeau, V.M.; Musso, D. Evidence of perinatal transmission of Zika virus, French Polynesia, December 2013 and February 2014. Eurosurveillance 2014, 19, 20751. [Google Scholar] [CrossRef]
- Oliveira Melo, A.S.; Malinger, G.; Ximenes, R.; Szejnfeld, P.O.; Alves Sampaio, S.; Bispo de Filippis, A.M. Zika virus intrauterine infection causes fetal brain abnormality and microcephaly: Tip of the iceberg? Ultrasound Obstet. Gynecol. 2016, 47, 6–7. [Google Scholar] [CrossRef]
- WHO. Zika Virus: World Health Organization. 2018. Available online: https://www.who.int/news-room/fact-sheets/detail/zika-virus (accessed on 15 January 2021).
- WHO. Prioritizing Diseases for Research and Development in Emergency Contexts 2020. Available online: https://www.who.int/activities/prioritizing-diseases-for-research-and-development-in-emergency-contexts (accessed on 15 January 2021).
- Counotte, M.J.; Kim, C.R.; Wang, J.; Bernstein, K.; Deal, C.D.; Broutet, N.J.N.; Low, N. Sexual transmission of Zika virus and other flaviviruses: A living systematic review. PLoS Med. 2018, 15, e1002611. [Google Scholar] [CrossRef] [PubMed]
- Colt, S.; Garcia-Casal, M.N.; Peña-Rosas, J.P.; Finkelstein, J.L.; Rayco-Solon, P.; Weise Prinzo, Z.C.; Mehta, S. Transmission of Zika virus through breast milk and other breastfeeding-related bodily-fluids: A systematic review. PLoS Negl. Trop. Dis. 2017, 11, e0005528. [Google Scholar] [CrossRef] [PubMed]
- Pierson, T.C.; Diamond, M.S. The emergence of Zika virus and its new clinical syndromes. Nature 2018, 560, 573–581. [Google Scholar] [CrossRef] [PubMed]
- WHO. Laboratory Testing for Zika Virus Infection: Interim Guidance: World Health Organization. 2016. Available online: https://apps.who.int/iris/handle/10665/204671 (accessed on 15 January 2021).
- Zorrilla, C.D.; García, I.G.; Fragoso, L.G.; De La Vega, A. Zika Virus Infection in Pregnancy: Maternal, Fetal, and Neonatal Considerations. J. Infect. Dis. 2017, 216, S891–S896. [Google Scholar] [CrossRef]
- Reynolds, M.R.; Jones, A.M.; Petersen, E.E.; Lee, E.H.; Rice, M.E.; Bingham, A.; Ellington, S.R.; Evert, N.; Reagan-Steiner, S.; Oduyebo, T.; et al. Vital Signs: Update on Zika Virus-Associated Birth Defects and Evaluation of All U.S. Infants with Congenital Zika Virus Exposure—U.S. Zika Pregnancy Registry, 2016. MMWR Morb. Mortal Wkly Rep. 2017, 66, 366–373. [Google Scholar] [CrossRef]
- Alvarado, M.G.; Schwartz, D.A. Zika Virus Infection in Pregnancy, Microcephaly, and Maternal and Fetal Health: What We Think, What We Know, and What We Think We Know. Arch. Pathol. Lab. Med. 2017, 141, 26–32. [Google Scholar] [CrossRef]
- CDC. Zika and Pregnancy: Congenital Zika Syndrome & Other Birth Defects: Centers for Disease Control and Prevention. 2020; (updated 26 April 2020). Available online: https://www.cdc.gov/pregnancy/zika/testing-follow-up/zika-syndrome-birth-defects.html (accessed on 15 January 2021).
- Werner, H.; Fazecas, T.; Guedes, B.; Dos Santos, J.L.; Daltro, P.; Tonni, G.; Campbell, S.; Júnior, E.A. Intrauterine Zika virus infection and microcephaly: Correlation of perinatal imaging and three-dimensional virtual physical models. Ultrasound Obstet. Gynecol. 2016, 47, 657–660. [Google Scholar] [CrossRef]
- Teixeira, F.M.E.; Pietrobon, A.J.; Oliveira, L.D.M.; Oliveira, L.M.D.S.; Sato, M.N. Maternal-Fetal Interplay in Zika Virus Infection and Adverse Perinatal Outcomes. Front. Immunol. 2020, 11, 175. [Google Scholar] [CrossRef]
- WHO. Infant Feeding in Areas of Zika Virus Transmission Guideline: World Health Organization. 2016. (updated 25 February 2016). Available online: https://www.who.int/nutrition/publications/guidelines/infantfeeding_zikavirus_transmission/en/ (accessed on 15 January 2021).
- GRADEpro. GRADEproGDT: GRADEpro Guideline Development Tool [Software]. McMaster University (developed by Evidence Prime, Inc.). 2015. Available online: www.gradepro.org (accessed on 15 January 2021).
- Besnard, M.; Dub, T.; Gérardin, P. Outcomes for 2 Children after Peripartum Acquisition of Zika Virus Infection, French Polynesia, 2013–2014. Emerg. Infect. Dis. 2017, 23, 1421–1423. [Google Scholar] [CrossRef]
- Dupont-Rouzeyrol, M.; Biron, A.; O’Connor, O.; Huguon, E.; Descloux, E. Infectious Zika viral particles in breastmilk. Lancet 2016, 387, 1051. [Google Scholar] [CrossRef]
- Cavalcanti, M.G.; Cabral-Castro, M.J.; Gonçalves, J.L.; Santana, L.S.; Pimenta, E.S.; Peralta, J.M. Zika virus shedding in human milk during lactation: An unlikely source of infection? Int. J. Infect. Dis. 2017, 57, 70–72. [Google Scholar] [CrossRef] [PubMed]
- Sotelo, J.R.; Sotelo, A.B.; Sotelo, F.J.; Doi, A.M.; Pinho, J.R.; de Cassia Oliveira, R.; Bezerra, A.M.; Deutsch, A.D.; Villas-Boas, L.S.; Felix, A.C.; et al. Persistence of Zika Virus in Breast Milk after Infection in Late Stage of Pregnancy. Emerg. Infect Dis. 2017, 23, 856–857. [Google Scholar] [CrossRef] [PubMed]
- Giovanetti, M.; de Jesus, J.G.; de Maia, M.L.; Junior, J.X.; Amarante, M.C.; Viana, P.; Barreto, F.K.; De Cerqueira, E.M.; Santos, N.P.; Falcão, M.B.; et al. Genetic evidence of Zika virus in mother’s breast milk and body fluids of a newborn with severe congenital defects. Clin. Microbiol. Infect. 2018, 24, 1112. [Google Scholar] [CrossRef] [PubMed]
- Blohm, G.M.; Lednicky, J.A.; Márquez, M.; White, S.K.; Loeb, J.C.; Pacheco, C.A.; Nolan, D.J.; Paisie, T.; Salemi, M.; Rodríguez-Morales, A.J.; et al. Complete Genome Sequences of Identical Zika virus Isolates in a Nursing Mother and Her Infant. Genome Announc. 2017, 5, e00231-17. [Google Scholar] [CrossRef] [PubMed]
- Blohm, G.M.; Lednicky, J.A.; Márquez, M.; White, S.K.; Loeb, J.C.; A Pacheco, C.; Nolan, D.J.; Paisie, T.; Salemi, M.; Rodríguez-Morales, A.J.; et al. Evidence for Mother-to-Child Transmission of Zika Virus Through Breast Milk. Clin. Infect. Dis. 2018, 66, 1120–1121. [Google Scholar] [CrossRef] [PubMed]
- Buathong, R.; Wacharapluesadee, S.; Ruchiseesarod, C.; Joyjinda, Y.; Sae-Liang, N.; Kanjanasombut, H.; Suwanchairob, O.; Siriarayapon, P.; Hemachudha, T. Breast milk and Zika virus infection in pregnancy, Thailand 2016–2017. Am. J. Trop. Med. Hyg. 2017, 97, 444. [Google Scholar]
- Hemachudha, P.; Wacharapluesadee, S.; Buathong, R.; Petcharat, S.; Bunprakob, S.; Ruchiseesarod, C.; Roeksomtawin, P.; Hemachudha, T. Lack of Transmission of Zika Virus Infection to Breastfed Infant. Clin. Med. Insights: Case Rep. 2019, 12. [Google Scholar] [CrossRef] [PubMed]
- Rodó, C.; Suy, A.; Sulleiro, E.; Soriano-Arandes, A.; Maiz, N.; García-Ruiz, I.; Arévalo, S.; Rando, A.; Anton, A.; Méndez, É.V.; et al. Pregnancy outcomes after maternal Zika virus infection in a non-endemic region: Prospective cohort study. Clin. Microbiol. Infect. 2019, 25, 633.e5–633.e9. [Google Scholar] [CrossRef]
- De Quental, O.B.; França, E.L.; Honório-França, A.C.; Morais, T.C.; Daboin, B.E.; Bezerra, I.M.; Komninakis, S.V.; de Abreu, L.C. Zika Virus Alters the Viscosity and Cytokines Profile in Human Colostrum. J. Immunol. Res. 2019, 2019, 9020519. [Google Scholar] [CrossRef]
- Mello, A.S.; Bertozzi, A.P.A.P.; Rodrigues, M.M.D.; Gazeta, R.E.; Moron, A.F.; Soriano-Arandes, A.; Sarmento, S.G.P.; Vedovello, D.; Silva, A.C.B.; Fajardo, T.C.G.; et al. Development of Secondary Microcephaly After Delivery: Possible Consequence of Mother-Baby Transmission of Zika Virus in Breast Milk. Am. J. Case Rep. 2019, 20, 723–725. [Google Scholar] [CrossRef]
- Tozetto-Mendoza, T.R.; Avelino-Silva, V.I.; Fonseca, S.; Claro, I.M.; Paula, A.V.D.; Levin, A.S.; Sabino, E.C.; Mendes-Correa, M.C.; Figueiredo, W.M.; Felix, A.C.; et al. Zika virus infection among symptomatic patients from two healthcare centers in Sao Paulo State, Brazil: Prevalence, clinical characteristics, viral detection in body fluids and serodynamics. Rev. Inst. Med. Trop. Sao Paulo 2019, 61. [Google Scholar] [CrossRef] [PubMed]
- Villamil-Gómez, W.E.; Guijarro, E.; Castellanos, J.; Rodríguez-Morales, A.J. Congenital Zika syndrome with prolonged detection of Zika virus RNA. J. Clin. Virol. 2017, 95, 52–54. [Google Scholar] [CrossRef] [PubMed]
- Desclaux, A.; de Lamballerie, X.; Leparc-Goffart, I.; Vilain-Parcé, A.; Coatleven, F.; Fleury, H.; Malvy, D. Probable sexually transmitted zika virus infection in a pregnant woman. N. Engl. J. Med. 2018, 378, 1458–1460. [Google Scholar] [CrossRef]
- Hayashi, S.; Kimura, H.; Oshiro, M.; Kato, Y.; Yasuda, A.; Suzuki, C.; Watanabe, Y.; Morishima, T.; Hayakawa, M. Transmission of cytomegalovirus via breast milk in extremely premature infants. J. Perinatol. 2011, 31, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Read, J.S.; The Breastfeeding and HIV International Transmission Study Groupa. Late postnatal transmission of HIV-1 in breast-fed children: An individual patient data meta-analysis. J. Infect. Dis. 2004, 189, 2154–2166. [Google Scholar]
- Coutsoudis, A.; Pillay, K.; Kuhn, L.; Spooner, E.; Tsai, W.-Y.; Coovadia, H.M. Method of feeding and transmission of HIV-1 from mothers to children by 15 months of age: Prospective cohort study from Durban, South Africa. AIDS 2001, 15, 379–387. [Google Scholar] [CrossRef]
- Vogels, C.B.; Rückert, C.; Cavany, S.M.; Perkins, T.A.; Ebel, G.D.; Grubaugh, N.D. Arbovirus coinfection and co-transmission: A neglected public health concern? PLoS Biol. 2019, 17, e3000130. [Google Scholar] [CrossRef]
- Mann, T.Z.; Haddad, L.; Williams, T.R.; Hills, S.L.; Read, J.S.; Dee, D.L.; Dziuban, E.J.; Pérez-Padilla, J.; Jamieson, D.J.; Honein, M.A.; et al. Breast milk transmission of flaviviruses in the context of Zika virus: A systematic review. Paediatr. Périnat. Epidemiol. 2018, 32, 358–368. [Google Scholar] [CrossRef]
- Sampieri, C.L.; Montero, H. Breastfeeding in the time of Zika: A systematic literature review. PeerJ 2019, 7, e6452. [Google Scholar] [CrossRef]
- Cordel, N.; Hébert, V.; Vega, R.A.; Herrmann-Storck, C. In vivo Zika virus detection in human skin. Br. J. Dermatol. 2018, 178, e108. [Google Scholar] [CrossRef]
- Musso, D.; Roche, C.; Nhan, T.-X.; Robin, E.; Teissier, A.; Cao-Lormeau, V.-M. Detection of Zika virus in saliva. J. Clin. Virol. 2015, 68, 53–55. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wu, D.; Zhong, H.; Guan, D.; Zhang, H.; Tan, Q.; Ke, C. Presence of Zika Virus in Conjunctival Fluid. JAMA Ophthalmol. 2016, 134, 1330–1332. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.J.L.; Balne, P.K.; Leo, Y.-S.; Tong, L.; Ng, L.F.P.; Agrawal, R. Persistence of Zika virus in conjunctival fluid of convalescence patients. Sci. Rep. 2017, 7, 1–5. [Google Scholar] [CrossRef] [PubMed]
| Children Outcomes | Breast Milk Outcomes | Maternal Outcomes | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Study | Design | Children with ZIKV Infection | Blood RT-PCR | Infant Feeding Practices | RT-PCR | Culture | Viral RNA Sequencing | Confirmed ZIKV Infection | Diagnostic Essay | Country |
| Besnard 2014 [10] Besnard 2017 [26] | Case reports | Yes | Positive in blood saliva | Breastfeeding | Positive | Negative | NA | Yes | Positive RT-PCR, 5 days after symptoms onset | French Polynesia |
| Yes | Positive | Not clear | Positive | Negative | NA | Yes | Positive RT-PCR, 2 days after symptoms onset | |||
| Dupont 2016 [27] | Case report | Undetermined | Ambiguous | Breastfeeding | Positive | Positive | NA | Yes | Positive 3 days after symptoms onset | New Caledonia |
| Blohm 2017 [31] Blohm 2018 [32] | Case report | Yes | Plasma positive | Breastfed for 5 months | Positive | Positive | 99% identity with the virus isolated from the child’s urine | Yes | Negative RT-PCR, 5 days after symptoms onsetPositive for IgM and marginal IgG | Venezuela |
| Cavalcanti 2017 [28] | Case reports | No | Serum, negative | Mix-feeding | Positive | Positive | NA | Yes | Positive RT-PCR | Brazil |
| No | NA | Breastfeeding | Negative | NA | NA | Yes | Positive RT-PCR | |||
| No | Serum, Negative for ZIKV Serum, Positive for CHIKV | Breastfeeding | Negative | NA | NA | Yes | Positive RT-PCR | |||
| No | Serum, negative | Breastfeeding | Negative | NA | NA | Yes | Positive RT-PCR | |||
| Sotelo 2017 [29] | Case report | Yes | Cord blood, negative | Not reported | Positive for samples collected at 37th gestational week, at birth, 38th gestational week, and 10th weeks postpartum | Positive for colostrum and sample 10 days after birth | NA | Yes | Positive RT-PCRIgM and IgG positive | Brazil |
| Giovanetti 2018 [30] | Case report | Yes | Serum, positive | Not reported | Positive | NA | Positive for strain similarities found with newborn viral genome sequencing obtained from the newborn | Yes | IgM positive. IgG not reported | Brazil |
| Desclaux 2018 [40] | Case report | No | Negative (serum) | Not reported | Negative | NA | NA | Yes | Positive RT-PCR | France |
| Mello 2018 [37] | Case report | No | Negative in blood and saliva | Breastfeeding | Positive | NA | NA | No | Negative RT-PCR and Antibodies | Brazil |
| Villamil-Gomez 2017 [39] | Case report | Yes | Positive (serum) at birth and 4, 6 months | Not reported | Positive | NA | NA | Yes | Positive RT-PCR at birth and 4, 6 months | Colombia |
| Hemachudha 2019 [34] | Case report | No | NA | Breastfeeding interrupted due to maternal diagnosis | Positive, starting 3 days before maternal symptoms and remained positive for 11 days (22 samples total). | NA | NA | Yes | Positive RT-PCR | Thailand |
| Buathong 2017(Abstract only) [33] | Cohort | No 9 from confirmed cases and 62 from probable cases | Negative | Breastfeeding interrupted due to maternal diagnosis | Positive | NA | Asian lineage identified | Yes, all 6 women | Positive (method unclear) | Thailand |
| Rodo 2019 [35] | Cohort | NA | Negative (serum) | Not reported | Negative | NA | NA | 72 of 254 women were positive | Positive (9 cases) by RT-PCR 62/71, 87.3%, positive for ZIKV IgM and/or ZIKV IgG and positive. 4 cases had positive IgM, 3 cases had positive both IgM and RT-PCR tests. 9 cases had detectable IgG levels. | Spain |
| De Quental 2019 [36] | Cohort | No | NA | Not reported | Negative | NA | NA | Yes, 20 women were infected | Positive (20 cases) by RT-PCR | Brazil |
| Tozetto-Mendoza 2019 [38] | Cohort | No | Negative cord blood, saliva, and urine. | Not reported | Positive | Positive | NA | Yes, 1 | Positive (94/235) confirmed ZIKV infection by RT-PCR in either plasma, serum, or urine. At least 7 positive cases were pregnant women. | Brazil |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Centeno-Tablante, E.; Medina-Rivera, M.; Finkelstein, J.L.; Herman, H.S.; Rayco-Solon, P.; Garcia-Casal, M.N.; Rogers, L.; Ghezzi-Kopel, K.; Zambrano Leal, M.P.; Andrade Velasquez, J.K.; et al. Update on the Transmission of Zika Virus Through Breast Milk and Breastfeeding: A Systematic Review of the Evidence. Viruses 2021, 13, 123. https://doi.org/10.3390/v13010123
Centeno-Tablante E, Medina-Rivera M, Finkelstein JL, Herman HS, Rayco-Solon P, Garcia-Casal MN, Rogers L, Ghezzi-Kopel K, Zambrano Leal MP, Andrade Velasquez JK, et al. Update on the Transmission of Zika Virus Through Breast Milk and Breastfeeding: A Systematic Review of the Evidence. Viruses. 2021; 13(1):123. https://doi.org/10.3390/v13010123
Chicago/Turabian StyleCenteno-Tablante, Elizabeth, Melisa Medina-Rivera, Julia L. Finkelstein, Heather S. Herman, Pura Rayco-Solon, Maria Nieves Garcia-Casal, Lisa Rogers, Kate Ghezzi-Kopel, Mildred P. Zambrano Leal, Joyce K. Andrade Velasquez, and et al. 2021. "Update on the Transmission of Zika Virus Through Breast Milk and Breastfeeding: A Systematic Review of the Evidence" Viruses 13, no. 1: 123. https://doi.org/10.3390/v13010123
APA StyleCenteno-Tablante, E., Medina-Rivera, M., Finkelstein, J. L., Herman, H. S., Rayco-Solon, P., Garcia-Casal, M. N., Rogers, L., Ghezzi-Kopel, K., Zambrano Leal, M. P., Andrade Velasquez, J. K., Chang Asinc, J. G., Peña-Rosas, J. P., & Mehta, S. (2021). Update on the Transmission of Zika Virus Through Breast Milk and Breastfeeding: A Systematic Review of the Evidence. Viruses, 13(1), 123. https://doi.org/10.3390/v13010123







