Molecular Characterization of Newcastle Disease Viruses Isolated from Chickens in Tanzania and Ghana
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolates from Tanzania and Samples from Ghana
2.2. Partial F Gene Amplification
2.3. Library Preparation and Sequencing
2.4. Phylogenetic Analysis
3. Results
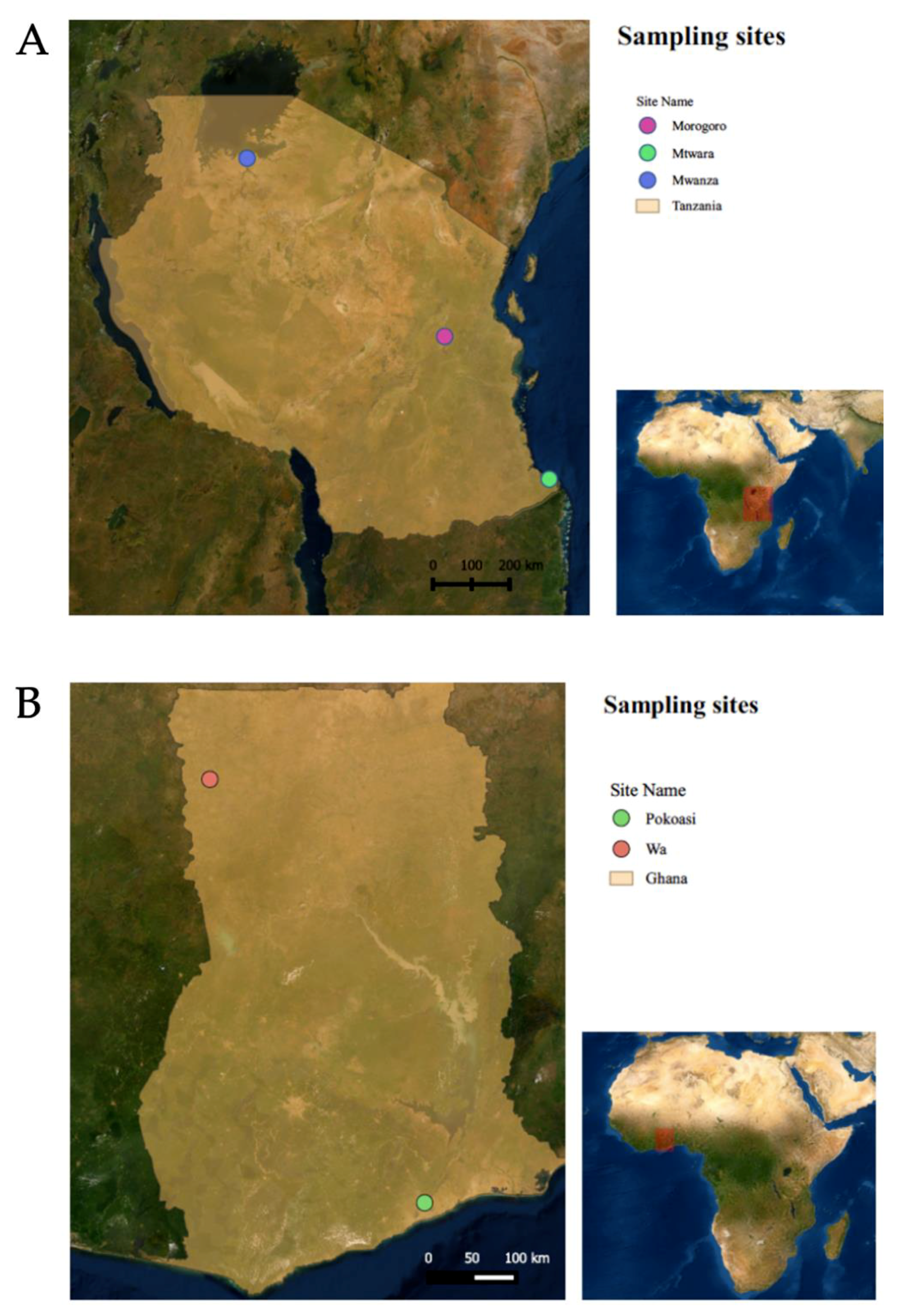
3.1. Geographic Distribution of the Sequences
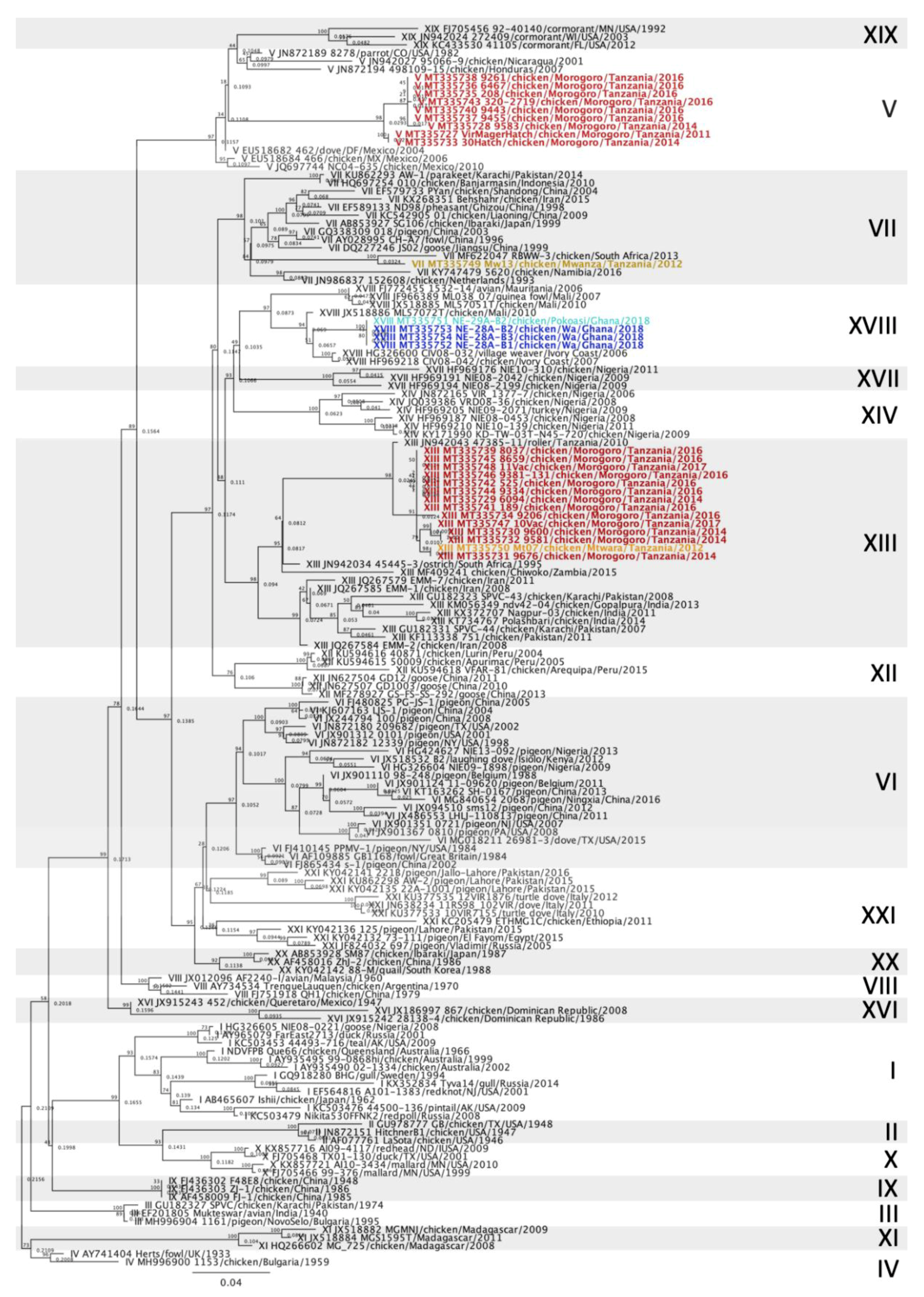
3.2. Newcastle Disease Virus Pilot Tree
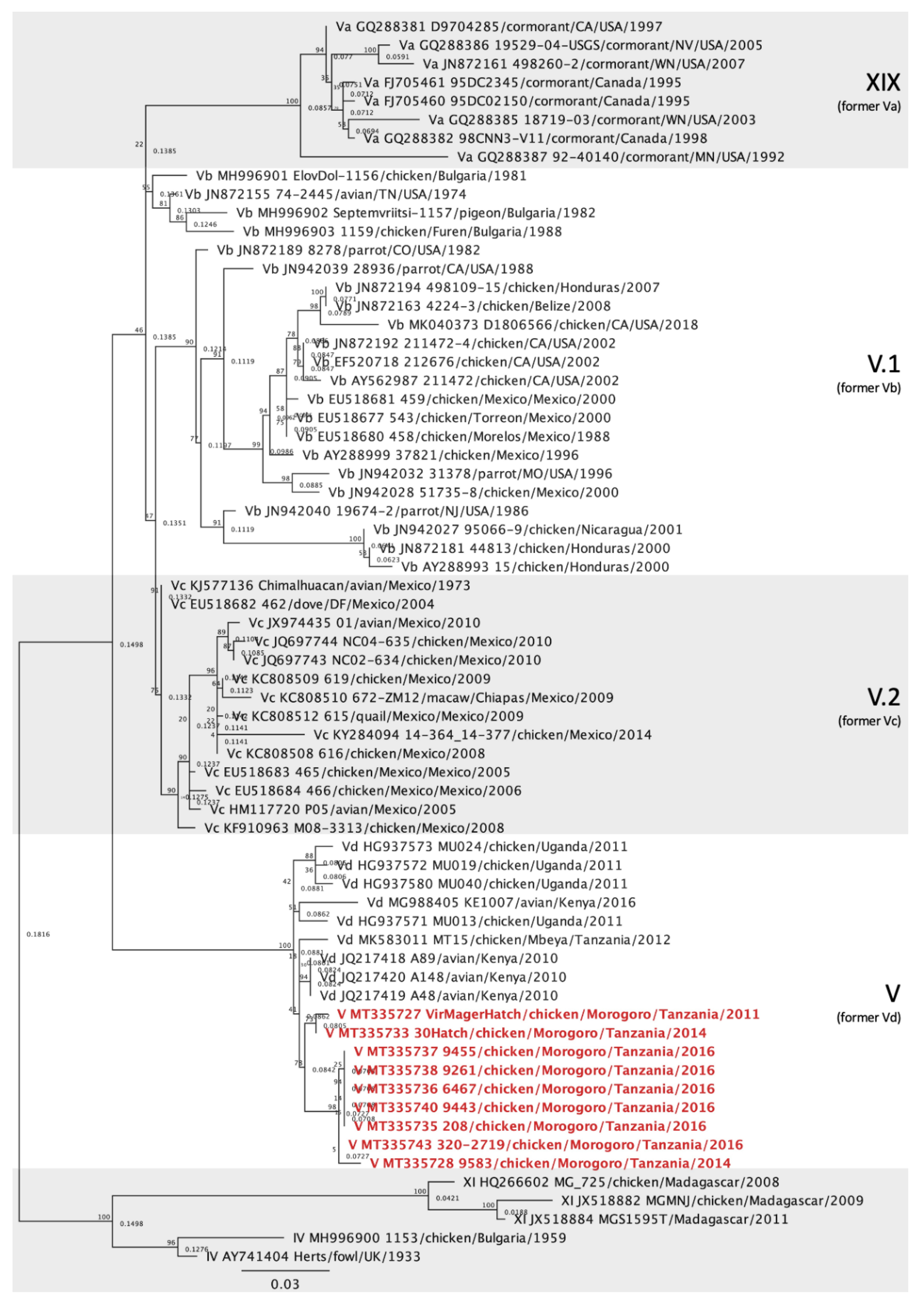
3.3. Genotype V Isolates
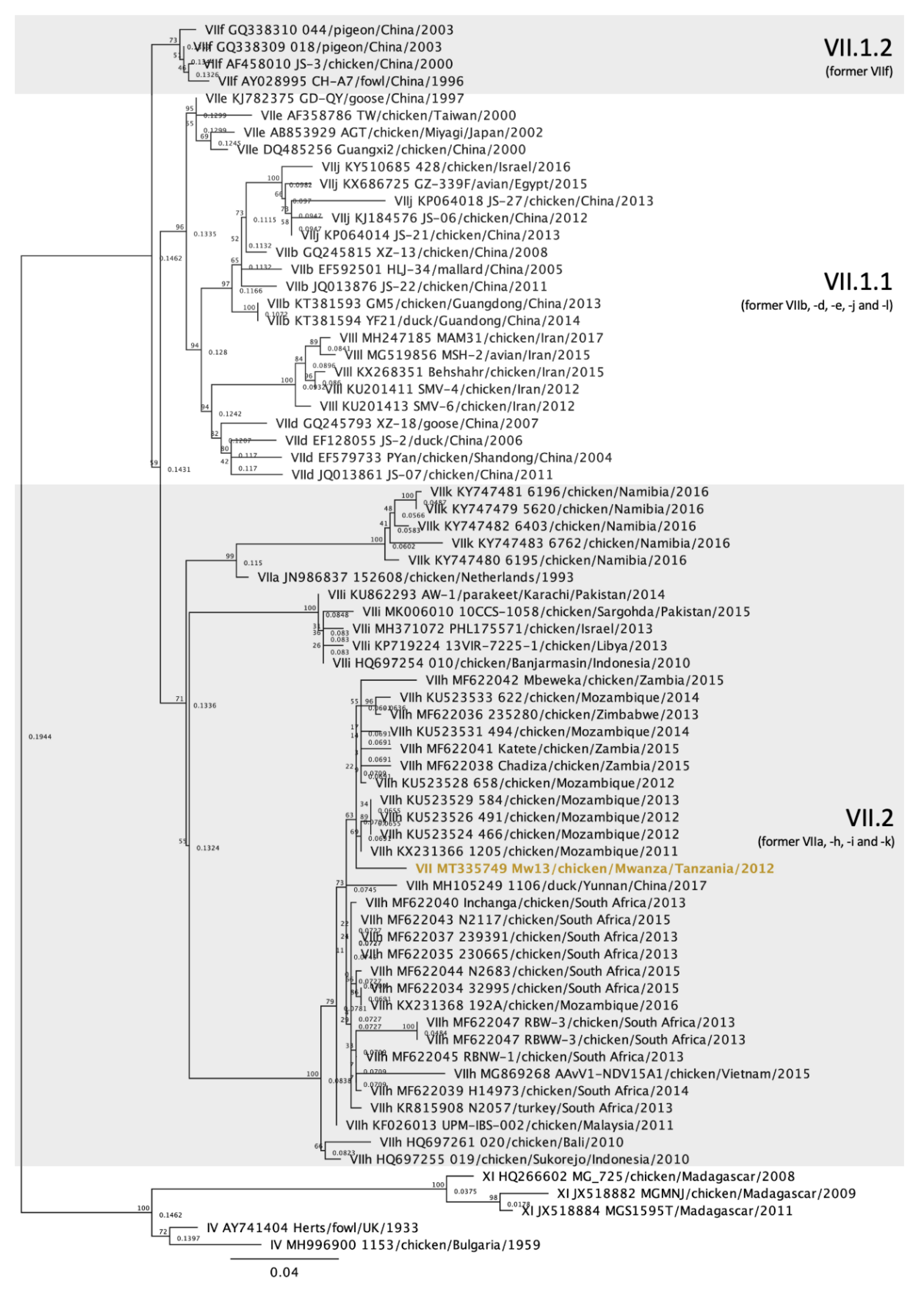
3.4. Genotype VII Isolate
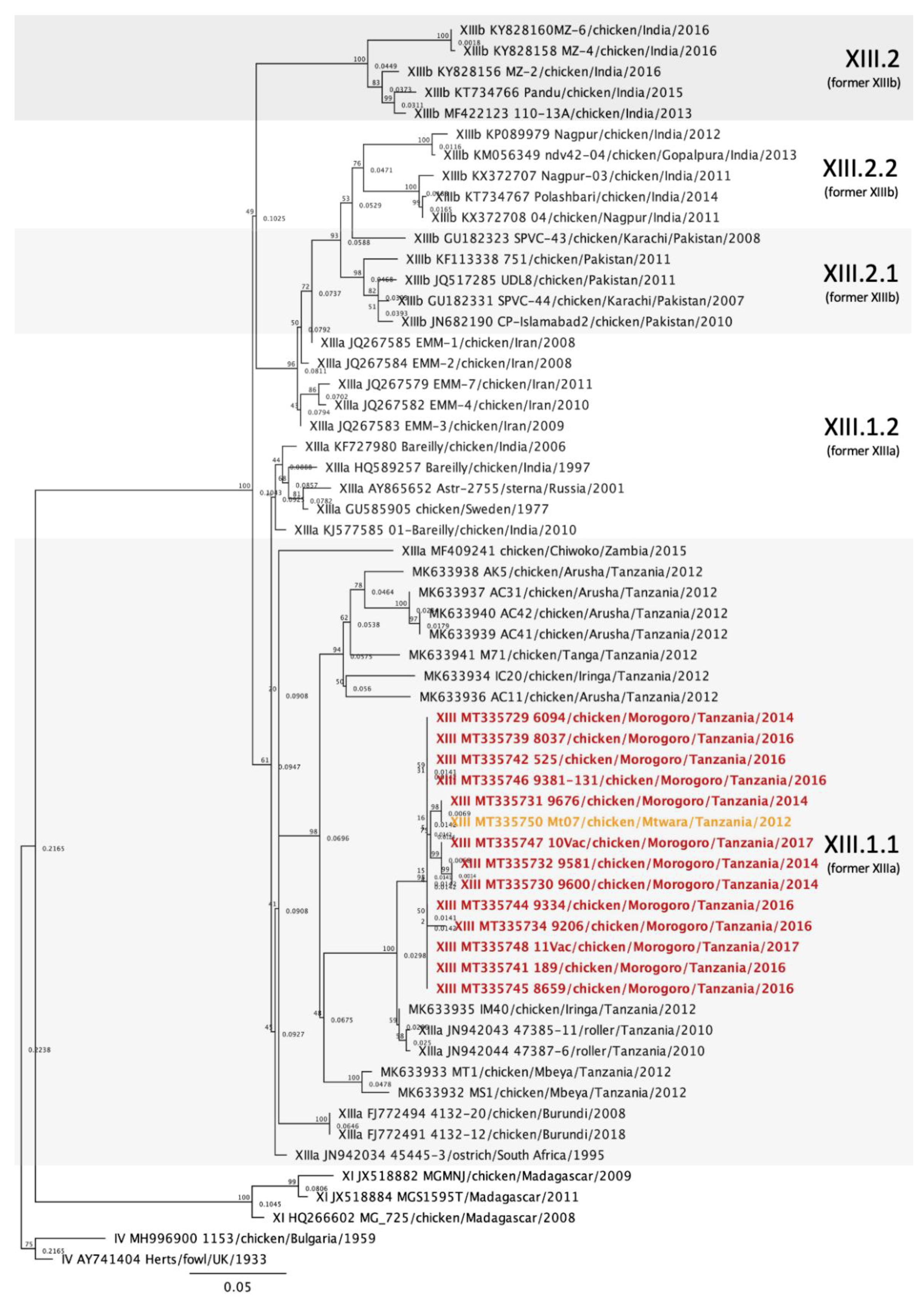
3.5. Genotype XIII Isolates
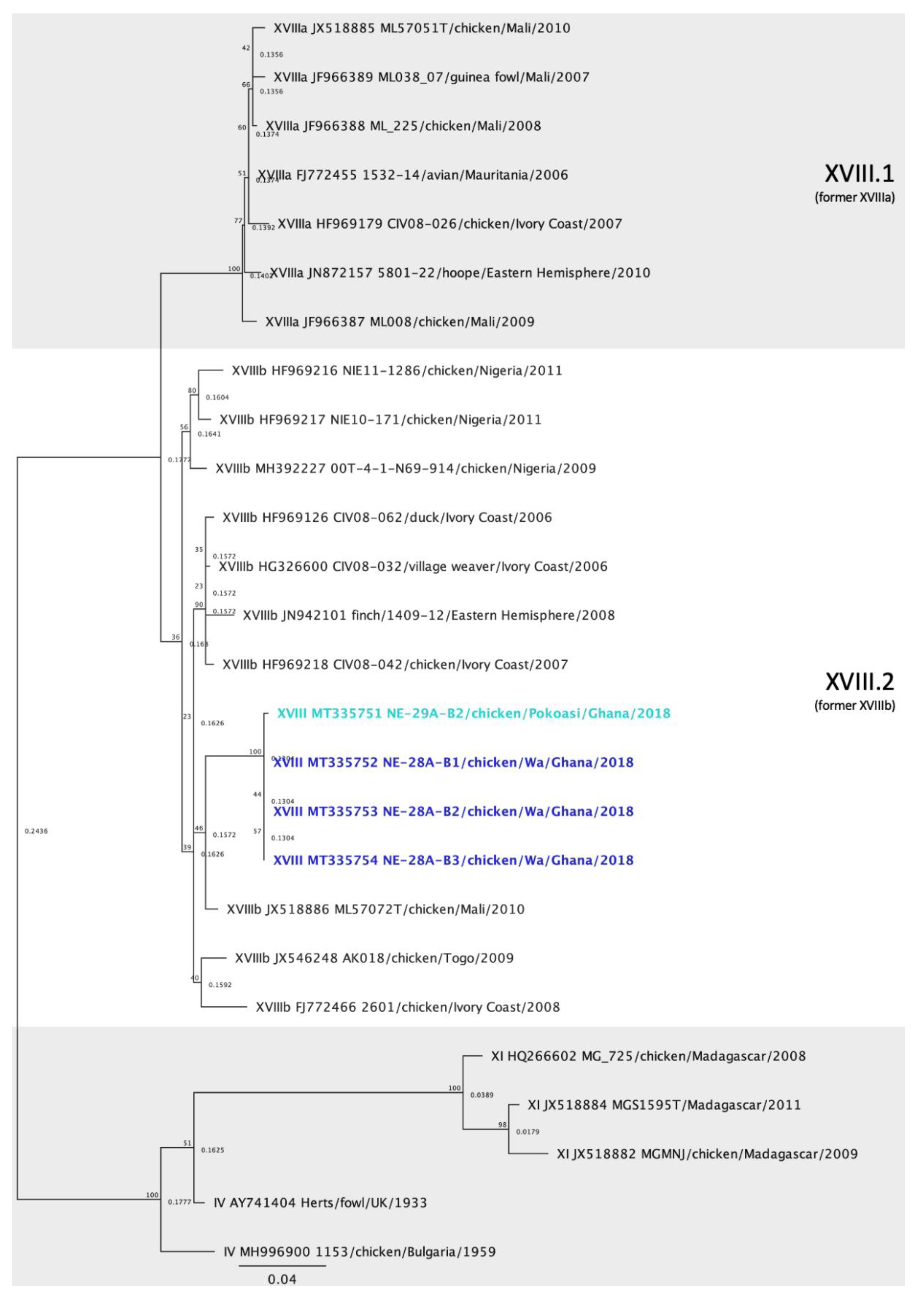
3.6. Genotype XVIII Isolates
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Suarez, D.L. Newcastle Disease, other Avian Paramyxoviruses, and Avian Metapneumovirus Infections. In Diseases of Poultry, 14th ed.; Swayne, D.E., Ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2020; pp. 111–166. [Google Scholar]
- ICTV. International Committee on Taxonomy of Viruses. Available online: https://talk.ictvonline.org/ictv-reports/ictv_online_report/negative-sense-rna-viruses/mononegavirales/w/paramyxoviridae (accessed on 4 May 2020).
- Acheson, N.H.; Kolakofsky, D.; Richardson, C. Paramyxoviruses and Rhabdoviruses. In Fundamentals of Molecular Virology; Acheson, N.H., Ed.; Wiley: Hoboken, NJ, USA, 2011; pp. 173–187. [Google Scholar]
- Diel, D.G.; da Silva, L.H.; Liu, H.; Wang, Z.; Miller, P.J.; Afonso, C.L. Genetic diversity of avian paramyxovirus type 1: Proposal for a unified nomenclature and classification system of Newcastle disease virus genotypes. Infect. Genet. Evol. 2012, 12, 1770–1779. [Google Scholar] [CrossRef] [PubMed]
- Dimitrov, K.M.; Abolnik, C.; Afonso, C.L.; Albina, E.; Bahl, J.; Berg, M.; Briand, F.X.; Brown, I.H.; Choi, K.S.; Chvala, I.; et al. Updated unified phylogenetic classification system and revised nomenclature for Newcastle disease virus. Infect. Genet. Evol. 2019, 74, 103917. [Google Scholar] [CrossRef] [PubMed]
- Miller, P.J.; Dimitrov, K.M.; Williams-Coplin, D.; Peterson, M.P.; Pantin-Jackwood, M.J.; Swayne, D.E.; Suarez, D.L.; Afonso, C.L. International Biological Engagement Programs Facilitate Newcastle Disease Epidemiological Studies. Front. Public Health 2015, 3, 235. [Google Scholar] [CrossRef] [PubMed]
- Butt, S.L.; Taylor, T.L.; Volkening, J.D.; Dimitrov, K.M.; Williams-Coplin, D.; Lahmers, K.K.; Miller, P.J.; Rana, A.M.; Suarez, D.L.; Afonso, C.L.; et al. Rapid virulence prediction and identification of Newcastle disease virus genotypes using third-generation sequencing. Virol. J. 2018, 15, 179. [Google Scholar] [CrossRef] [PubMed]
- OIE. Newcastle Disease (Infection with Newcastle Disease Virus). In Manual of Diagnostic Tests and Vaccines for Terrestrial Animals: Mammals, Birds and Bees; OIE, Ed.; Office International des Epizooties: Paris, France, 2019; pp. 964–983. [Google Scholar]
- WAHIS. World Animal Health Information Database. Terrestrial: Newcastle Disease, Jan–Jun 2019. Available online: https://www.oie.int/wahis_2/public/wahid.php/Diseaseinformation/Diseasedistributionmap/index/newlang/en?disease_type_hidden=&disease_id_hidden=&selected_disease_name_hidden=&disease_type=0&disease_id_terrestrial=16&species_t=0&disease_id_aquatic=-999&species_a=0&sta_method=semesterly&selected_start_year=2019&selected_report_period=1&selected_start_month=1&date_submit=OK (accessed on 4 June 2020).
- Kitalyi, A.J. Village Chicken Production Systems in Rural Africa: Household Food Security and Gender Issues; FAO: Rome, Italy, 1998. [Google Scholar]
- Yongolo, M.G.; Christensen, H.; Handberg, K.; Minga, U.; Olsen, J.E. On the origin and diversity of Newcastle disease virus in Tanzania. Onderstepoort J. Vet. Res. 2011, 78, 312. [Google Scholar] [CrossRef] [PubMed]
- Byarugaba, D.K.; Mugimba, K.K.; Omony, J.B.; Okitwi, M.; Wanyana, A.; Otim, M.O.; Kirunda, H.; Nakavuma, J.L.; Teillaud, A.; Paul, M.C.; et al. High pathogenicity and low genetic evolution of avian paramyxovirus type I (Newcastle disease virus) isolated from live bird markets in Uganda. Virol. J. 2014, 11, 173. [Google Scholar] [CrossRef] [PubMed]
- Samuel, A.; Nayak, B.; Paldurai, A.; Xiao, S.; Aplogan, G.L.; Awoume, K.A.; Webby, R.J.; Ducatez, M.F.; Collins, P.L.; Samal, S.K. Phylogenetic and pathotypic characterization of newcastle disease viruses circulating in west Africa and efficacy of a current vaccine. J. Clin. Microbiol. 2013, 51, 771–781. [Google Scholar] [CrossRef] [PubMed]
- Snoeck, C.J.; Owoade, A.A.; Couacy-Hymann, E.; Alkali, B.R.; Okwen, M.P.; Adeyanju, A.T.; Komoyo, G.F.; Nakoune, E.; Le Faou, A.; Muller, C.P. High genetic diversity of Newcastle disease virus in poultry in West and Central Africa: Cocirculation of genotype XIV and newly defined genotypes XVII and XVIII. J. Clin. Microbiol. 2013, 51, 2250–2260. [Google Scholar] [CrossRef] [PubMed]
- Msoffe, P.L.M.; Chiwanga, G.H.; Cardona, C.J.; Miller, P.J.; Suarez, D.L. Isolation and Characterization of Newcastle Disease Virus from Live Bird Markets in Tanzania. Avian Dis. 2019, 63, 634–640. [Google Scholar] [CrossRef] [PubMed]
- Heather, J.M.; Chain, B. The sequence of sequencers: The history of sequencing DNA. Genomics 2016, 107, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Moore, N.E.; Deng, Y.M.; Eccles, D.A.; Hall, R.J. MinION nanopore sequencing of an influenza genome. Front. Microbiol. 2015, 6, 766. [Google Scholar] [CrossRef] [PubMed]
- Dimitrov, K.M.; Sharma, P.; Volkening, J.D.; Goraichuk, I.V.; Wajid, A.; Rehmani, S.F.; Basharat, A.; Shittu, I.; Joannis, T.M.; Miller, P.J.; et al. A robust and cost-effective approach to sequence and analyze complete genomes of small RNA viruses. Virol. J. 2017, 14, 72. [Google Scholar] [CrossRef] [PubMed]
- Villegas, P. Titration of biological suspensions. In A Laboratory Manual for the Isolation, Identification and Characterization of Avian Pathogens; Williams, S.M., Dufour-Zavala, L.D.E.S., Jackwood, M.W., Lee, M.D., Lupiani, B., Reed, W.M., Spackman, E., Woolcock, P.R., Eds.; American Association of Avian Pathologists (AAAP): Jacksonville, FL, USA, 2016; pp. 355–360. [Google Scholar]
- Wise, M.G.; Suarez, D.L.; Seal, B.S.; Pedersen, J.C.; Senne, D.A.; King, D.J.; Kapczynski, D.R.; Spackman, E. Development of a real-time reverse-transcription PCR for detection of Newcastle disease virus RNA in clinical samples. J. Clin. Microbiol. 2004, 42, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef] [PubMed]
- Goraichuk, I.V.; Msoffe, P.L.M.; Chiwanga, G.H.; Dimitrov, K.M.; Afonso, C.L.; Suarez, D.L. First Complete Genome Sequence of a Subgenotype Vd Newcastle Disease Virus Isolate. Microbiol. Resour. Announc. 2019, 8, e00436-19. [Google Scholar] [CrossRef] [PubMed]
- Mapaco, L.P.; Monjane, I.V.; Nhamusso, A.E.; Viljoen, G.J.; Dundon, W.G.; Acha, S.J. Phylogenetic analysis of Newcastle disease viruses isolated from commercial poultry in Mozambique (2011–2016). Virus Genes 2016, 52, 748–753. [Google Scholar] [CrossRef] [PubMed]
- Abolnik, C.; Mubamba, C.; Wandrag, D.B.R.; Horner, R.; Gummow, B.; Dautu, G.; Bisschop, S.P.R. Tracing the origins of genotype VIIh Newcastle disease in southern Africa. Transbound. Emerg. Dis. 2018, 65, e393–e403. [Google Scholar] [CrossRef] [PubMed]
- Miller, P.J.; Haddas, R.; Simanov, L.; Lublin, A.; Rehmani, S.F.; Wajid, A.; Bibi, T.; Khan, T.A.; Yaqub, T.; Setiyaningsih, S.; et al. Identification of new sub-genotypes of virulent Newcastle disease virus with potential panzootic features. Infect. Genet. Evol. 2015, 29, 216–229. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Silva, A.P.; Aston, E.J.; Chiwanga, G.H.; Birakos, A.; Muhairwa, A.P.; Kayang, B.B.; Kelly, T.; Zhou, H.; Gallardo, R.A. Molecular Characterization of Newcastle Disease Viruses Isolated from Chickens in Tanzania and Ghana. Viruses 2020, 12, 916. https://doi.org/10.3390/v12090916
da Silva AP, Aston EJ, Chiwanga GH, Birakos A, Muhairwa AP, Kayang BB, Kelly T, Zhou H, Gallardo RA. Molecular Characterization of Newcastle Disease Viruses Isolated from Chickens in Tanzania and Ghana. Viruses. 2020; 12(9):916. https://doi.org/10.3390/v12090916
Chicago/Turabian Styleda Silva, Ana P., Emily J. Aston, Gaspar H. Chiwanga, Ashley Birakos, Amandus P. Muhairwa, Boniface B. Kayang, Terra Kelly, Huaijun Zhou, and Rodrigo A. Gallardo. 2020. "Molecular Characterization of Newcastle Disease Viruses Isolated from Chickens in Tanzania and Ghana" Viruses 12, no. 9: 916. https://doi.org/10.3390/v12090916
APA Styleda Silva, A. P., Aston, E. J., Chiwanga, G. H., Birakos, A., Muhairwa, A. P., Kayang, B. B., Kelly, T., Zhou, H., & Gallardo, R. A. (2020). Molecular Characterization of Newcastle Disease Viruses Isolated from Chickens in Tanzania and Ghana. Viruses, 12(9), 916. https://doi.org/10.3390/v12090916






