Two Separate Tyrosine-Based YXXL/Φ Motifs within the Glycoprotein E Cytoplasmic Tail of Bovine Herpesvirus 1 Contribute in Virus Anterograde Neuronal Transport
Abstract
1. Introduction
2. Materials and Methods
2.1. Cells
2.2. Viruses
2.3. Primary Neuronal Cultures and Virus Infection in the Microfluidic Chamber System
2.4. Rabbit Experiments
2.5. RNA Extraction and Reverse-Transcriptase PCR (RT-PCR)
2.6. Statistical Analysis
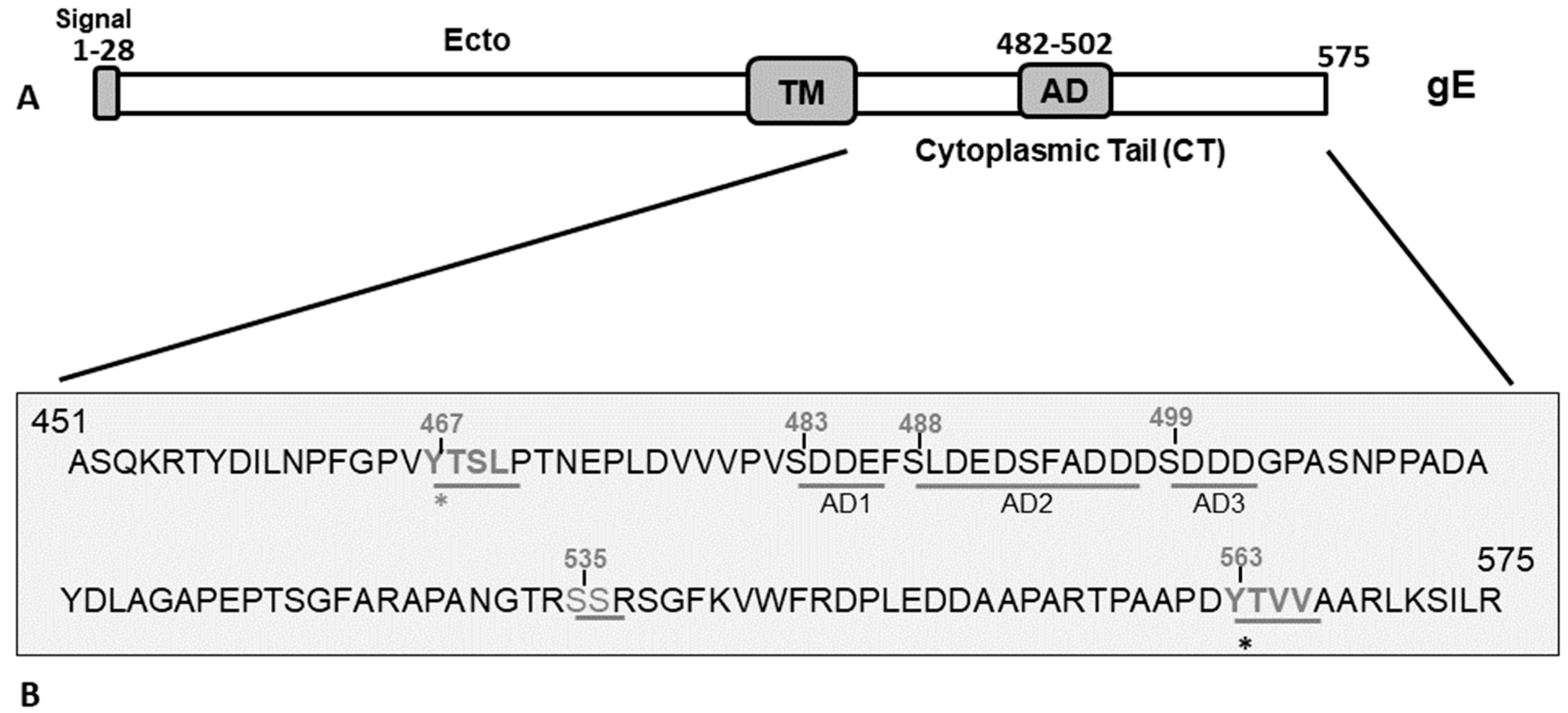
3. Results
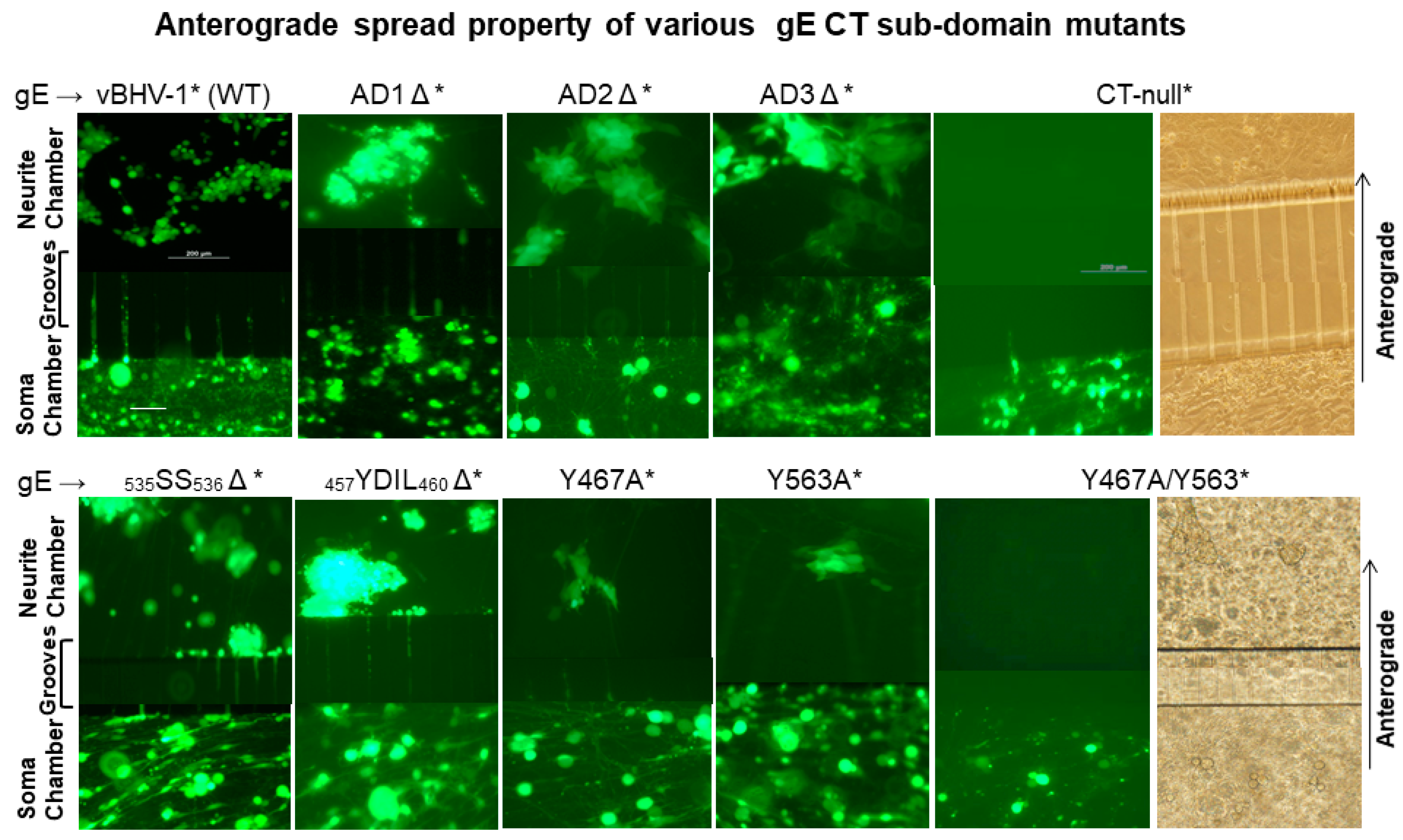
3.1. Both gE-Y467A and gE-Y563A Mutants Showed Diminished Anterograde Axonal Transport in Primary Neuron Culture, While the Double Y467A/Y563A Mutant Virus, Like the gE CT-Null Virus, Was Unable to Transport Anterogradely
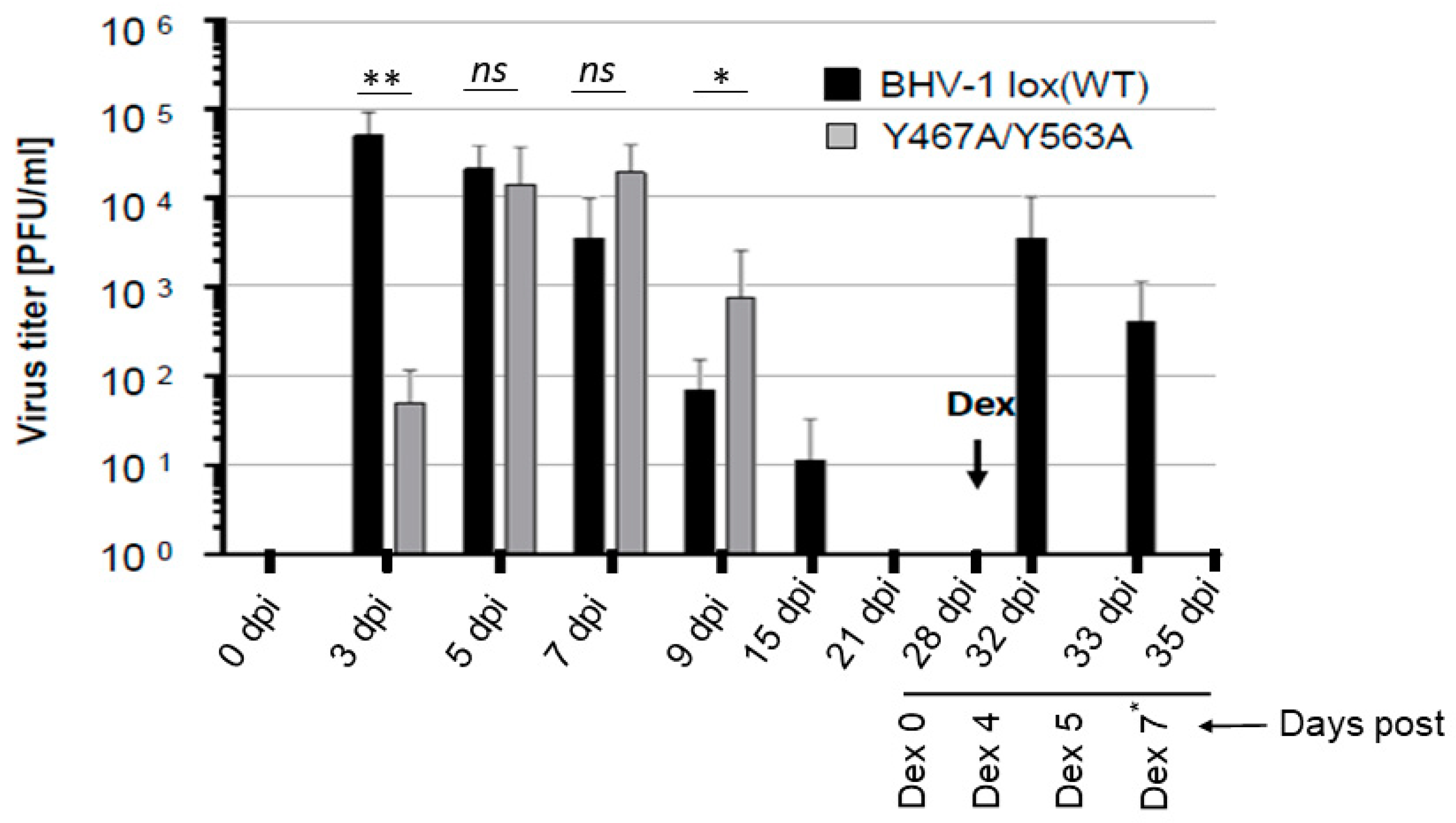
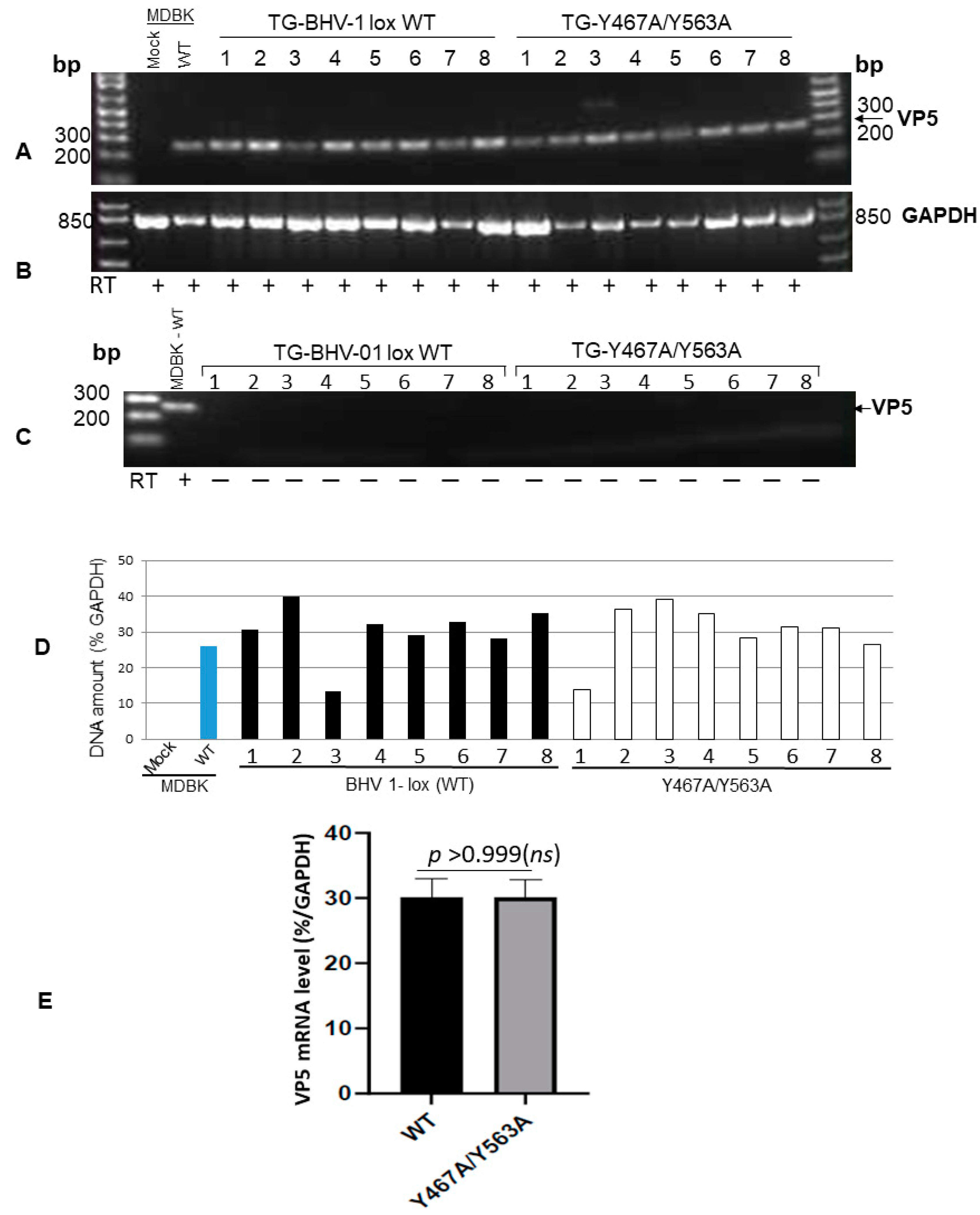
3.2. In Vivo Anterograde Neuronal Transport Property of Dual Y467A/Y563A Mutant and BHV-1 Lox (wt) Viruses in Rabbits
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jones, C.; Chowdhury, S. A review of the biology of bovine herpesvirus type 1 (BHV-1), its role as a cofactor in the bovine respiratory disease complex and development of improved vaccines. Anim. Health Res. Rev. 2007, 8, 187–205. [Google Scholar] [CrossRef] [PubMed]
- Tikoo, S.K.; Campos, M.; Babiuk, L.A. Bovine herpesvirus 1 (BHV-1): Biology, pathogenesis, and control. Adv. Virus Res. 1995, 45, 191–223. [Google Scholar] [PubMed]
- Jones, C. Alphaherpesvirus latency: Its role in disease and survival of the virus in nature. Adv. Virus Res. 1998, 51, 81–133. [Google Scholar] [PubMed]
- Brum, M.C.; Coats, C.; Sangena, R.B.; Doster, A.; Jones, C.; Chowdhury, S.I. Bovine herpesvirus type 1 (BoHV-1) anterograde neuronal transport from trigeminal ganglia to nose and eye requires glycoprotein E. J. Neurovirol. 2009, 15, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Butchi, N.B.; Jones, C.; Perez, S.; Doster, A.; Chowdhury, S.I. Envelope protein Us9 is required for the anterograde transport of bovine herpesvirus type 1 from trigeminal ganglia to nose and eye upon reactivation. J. Neurovirol. 2007, 13, 384–388. [Google Scholar] [CrossRef]
- Brideau, A.D.; Card, J.P.; Enquist, L.W. Role of pseudorabies virus Us9, a type II membrane protein, in infection of tissue culture cells and the rat nervous system. J. Virol. 2000, 74, 834–845. [Google Scholar] [CrossRef]
- Tomishima, M.J.; Enquist, L.W. A conserved alpha-herpesvirus protein necessary for axonal localization of viral membrane proteins. J. Cell Biol. 2001, 154, 741–752. [Google Scholar] [CrossRef]
- Mettenleiter, T.C. Aujeszky’s disease (pseudorabies) virus: The virus and molecular pathogenesis--state of the art, June 1999. Vet. Res. 2000, 31, 99–115. [Google Scholar] [CrossRef]
- Husak, P.J.; Kuo, T.; Enquist, L.W. Pseudorabies virus membrane proteins gI and gE facilitate anterograde spread of infection in projection-specific neurons in the rat. J. Virol. 2000, 74, 10975–10983. [Google Scholar] [CrossRef]
- Ch’ng, T.H.; Enquist, L.W. Efficient axonal localization of alphaherpesvirus structural proteins in cultured sympathetic neurons requires viral glycoprotein E. J. Virol. 2005, 79, 8835–8846. [Google Scholar] [CrossRef]
- Chowdhury, S.I.; Coats, J.; Neis, R.A.; Navarro, S.M.; Paulsen, D.B.; Feng, J.M. A bovine herpesvirus type 1 mutant virus with truncated glycoprotein E cytoplasmic tail has defective anterograde neuronal transport in rabbit dorsal root ganglia primary neuronal cultures in a microfluidic chamber system. J. Neurovirol. 2010, 16, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.F.; Brum, M.C.S.; Doster, A.; Jones, C.; Chowdhury, S.I. A bovine herpesvirus type 1 mutant virus specifying a carboxyl-terminal truncation of glycoprotein E is defective in anterograde neuronal transport in rabbits and calves. J. Virol. 2008, 82, 7432–7442. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.I.; Ross, C.S.; Lee, B.J.; Hall, V.; Chu, H.J. Construction and characterization of a glycoprotein E gene-deleted bovine herpesvirus type 1 recombinant. Am. J. Vet. Res. 1999, 60, 227–232. [Google Scholar] [PubMed]
- Farnsworth, A.; Johnson, D.C. Herpes simplex virus gE/gI must accumulate in the trans-Golgi network at early times and then redistribute to cell junctions to promote cell-cell spread. J. Virol. 2006, 80, 3167–3179. [Google Scholar] [CrossRef] [PubMed]
- Tirabassi, R.S.; Enquist, L.W. Role of envelope protein gE endocytosis in the pseudorabies virus life cycle. J. Virol. 1998, 72, 4571–4579. [Google Scholar] [CrossRef]
- Tirabassi, R.S.; Enquist, L.W. Mutation of the YXXL endocytosis motif in the cytoplasmic tail of pseudorabies virus gE. J. Virol. 1999, 73, 2717–2728. [Google Scholar] [CrossRef]
- Yezid, H.; Pannhorst, K.; Wei, H.; Chowdhury, S.I. Bovine herpesvirus 1 (BHV-1) glycoprotein E endocytosis/recycling from apical membrane to trans-Golgi network and redistribution to basolateral surface are regulated by two seerate YXXL/Φ motifs within the cytoplasmic tail which together promote efficient virus cell-to-cell spread. Virology 2020, 548, 136–151. [Google Scholar]
- Chowdhury, S.I.; Ludwig, H.; Buhk, H.J. Molecular biological characterization of equine herpesvirus type 1 (EHV-1) isolates from ruminant hosts. Virus Res. 1988, 11, 127–139. [Google Scholar] [CrossRef]
- Gruffat, H.; Marchione, R.; Manet, E. Herpesvirus Late Gene Expression: A Viral-Specific Pre-initiation Complex Is Key. Front. Microbiol. 2016, 7, 869. [Google Scholar] [CrossRef]
- Mavromara-Nazos, P.; Roizman, B. Activation of herpes simplex virus 1 gamma 2 genes by viral DNA replication. Virology 1987, 161, 593–598. [Google Scholar] [CrossRef]
- Miller, C.S.; Danaher, R.J.; Jacob, R.J. Molecular aspects of herpes simplex virus I latency, reactivation, and recurrence. Crit. Rev. Oral Biol. Med. 1998, 9, 541–562. [Google Scholar] [CrossRef] [PubMed]
- Ikonen, E.; Simons, K. Protein and lipid sorting from the trans-Golgi network to the plasma membrane in polarized cells. Semin. Cell Dev. Biol. 1998, 9, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Bonifacino, J.S.; Dell’Angelica, E.C. Molecular bases for the recognition of tyrosine-based sorting signals. J. Cell Biol. 1999, 145, 923–926. [Google Scholar] [CrossRef]
- Keller, P.; Simons, K. Post-Golgi biosynthetic trafficking. J. Cell Sci. 1997, 110, 3001–3009. [Google Scholar]
- Hambleton, S.; Gershon, M.D.; Gershon, A.A. The role of the trans-Golgi network in varicella zoster virus biology. Cell Mol. Life Sci. 2004, 61, 3047–3056. [Google Scholar] [CrossRef] [PubMed]
- Hirst, J.; Irving, C.; Borner, G.H. Adaptor protein complexes AP-4 and AP-5: New players in endosomal trafficking and progressive spastic paraplegia. Traffic 2013, 14, 153–164. [Google Scholar] [CrossRef]
- Robinson, M.S. Adaptable adaptors for coated vesicles. Trends Cell Biol. 2004, 14, 167–174. [Google Scholar] [CrossRef]
- Robinson, M.S.; Bonifacino, J.S. Adaptor-related proteins. Curr. Opin. Cell Biol. 2001, 13, 444–453. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yezid, H.; Lay, C.T.; Pannhorst, K.; Chowdhury, S.I. Two Separate Tyrosine-Based YXXL/Φ Motifs within the Glycoprotein E Cytoplasmic Tail of Bovine Herpesvirus 1 Contribute in Virus Anterograde Neuronal Transport. Viruses 2020, 12, 1025. https://doi.org/10.3390/v12091025
Yezid H, Lay CT, Pannhorst K, Chowdhury SI. Two Separate Tyrosine-Based YXXL/Φ Motifs within the Glycoprotein E Cytoplasmic Tail of Bovine Herpesvirus 1 Contribute in Virus Anterograde Neuronal Transport. Viruses. 2020; 12(9):1025. https://doi.org/10.3390/v12091025
Chicago/Turabian StyleYezid, Hocine, Christian T. Lay, Katrin Pannhorst, and Shafiqul I. Chowdhury. 2020. "Two Separate Tyrosine-Based YXXL/Φ Motifs within the Glycoprotein E Cytoplasmic Tail of Bovine Herpesvirus 1 Contribute in Virus Anterograde Neuronal Transport" Viruses 12, no. 9: 1025. https://doi.org/10.3390/v12091025
APA StyleYezid, H., Lay, C. T., Pannhorst, K., & Chowdhury, S. I. (2020). Two Separate Tyrosine-Based YXXL/Φ Motifs within the Glycoprotein E Cytoplasmic Tail of Bovine Herpesvirus 1 Contribute in Virus Anterograde Neuronal Transport. Viruses, 12(9), 1025. https://doi.org/10.3390/v12091025





