Emerging Role of Mucosal Vaccine in Preventing Infection with Avian Influenza A Viruses
Abstract
1. Introduction
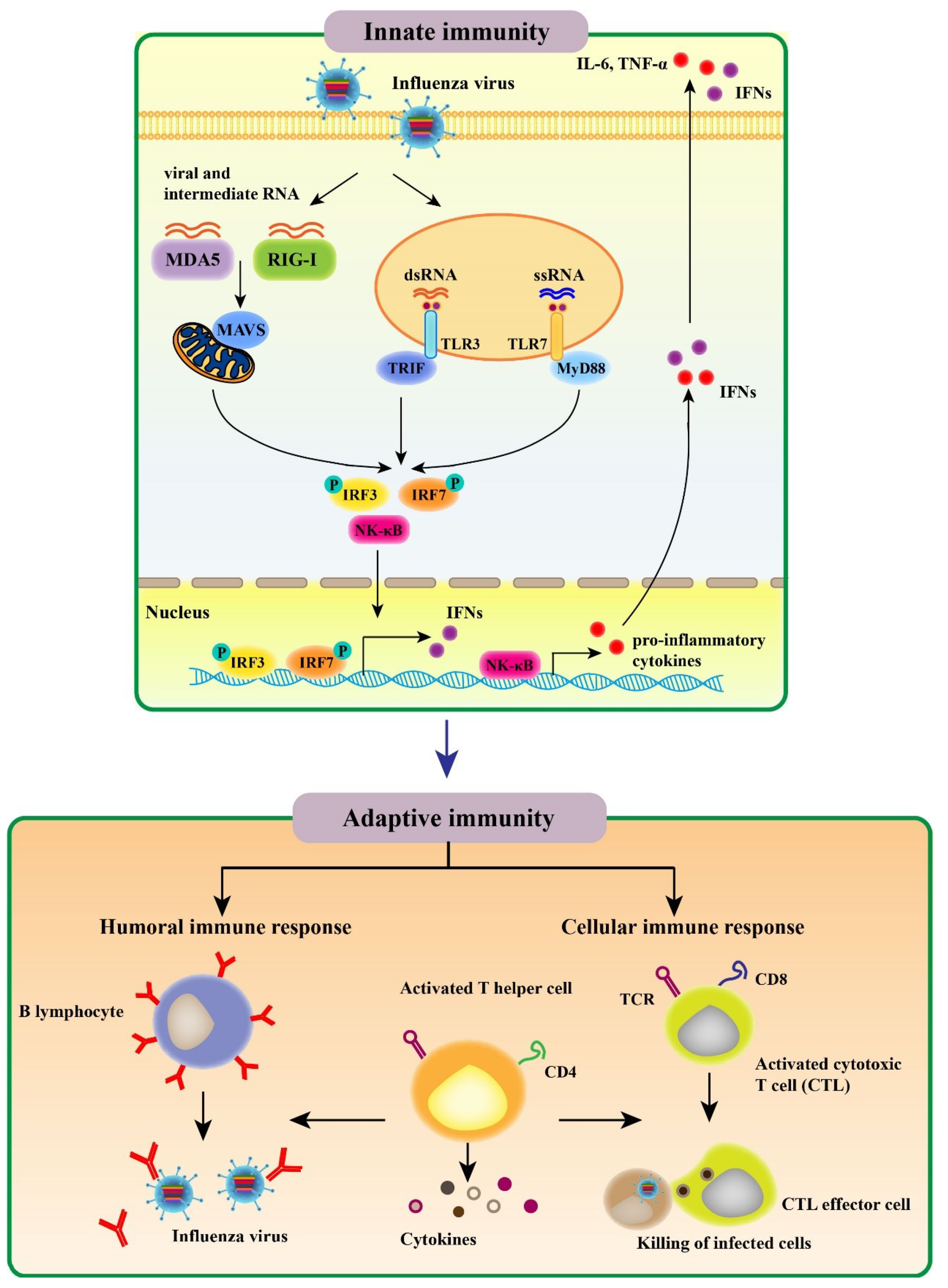
2. Host Immune Responses to Influenza Viruses
2.1. Innate Immune Response to Influenza Virus
2.2. Adaptive Immune Response to Influenza Virus
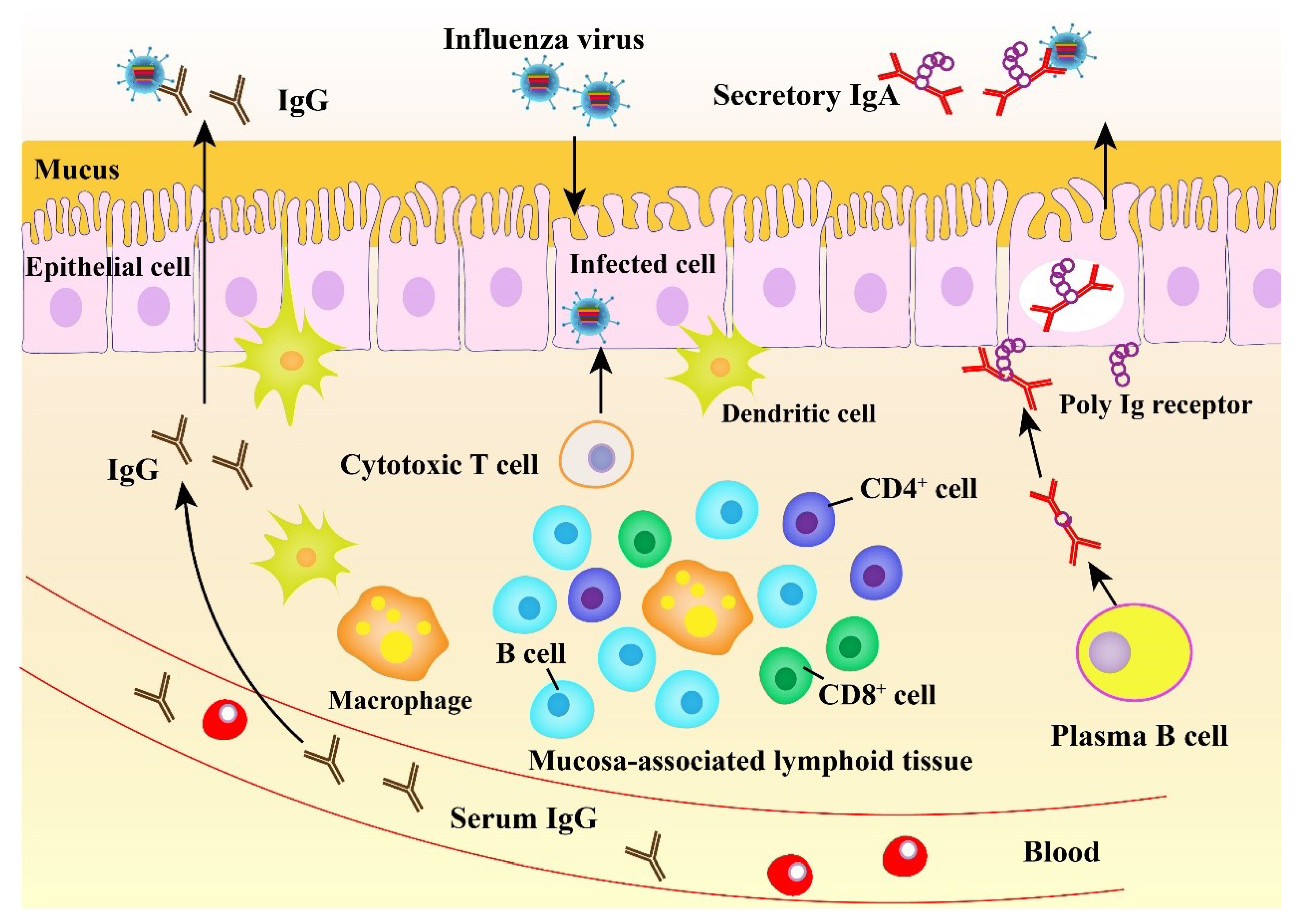
2.3. Mucosal Immune Response to Influenza Virus
3. The Importance of Mucosal Immunity against Influenza Virus Infection
4. Strategies for Mucosal Vaccination
4.1. Live Attenuated Vaccines
4.2. Mucosal Adjuvants
4.3. Mucosal DNA Vaccines
4.4. Mucosal Delivery Systems
4.4.1. Particulate Formulations
4.4.2. Live Vector Vaccines
4.5. Mucosal Vaccine Delivery Routes
4.5.1. Oral Vaccination
4.5.2. Intranasal Vaccination
5. Protective Mucosal Immunity to Newcastle Disease in Chickens: A Successful Case for Vaccine Design
6. Conclusions and Perspectives
Funding
Conflicts of Interest
References
- Yoon, S.W.; Webby, R.J.; Webster, R.G. Evolution and ecology of influenza A viruses. Curr. Top. Microbiol. Immunol. 2014, 385, 359–375. [Google Scholar] [PubMed]
- Luke, C.J.; Subbarao, K. Vaccines for pandemic influenza. Emerg. Infect. Dis. 2006, 12, 66–72. [Google Scholar] [CrossRef]
- Kobasa, D.; Kawaoka, Y. Emerging influenza viruses: Past and present. Curr. Mol. Med. 2005, 5, 791–803. [Google Scholar] [CrossRef] [PubMed]
- Tong, S.; Zhu, X.; Li, Y.; Shi, M.; Zhang, J.; Bourgeois, M.; Yang, H.; Chen, X.; Recuenco, S.; Gomez, J.; et al. New world bats harbor diverse influenza A viruses. PLoS Pathog. 2013, 9, e1003657. [Google Scholar] [CrossRef] [PubMed]
- Peiris, J.S.; de Jong, M.D.; Guan, Y. Avian influenza virus (H5N1): A threat to human health. Clin. Microbiol. Rev. 2007, 20, 243–267. [Google Scholar] [CrossRef]
- Poovorawan, Y.; Pyungporn, S.; Prachayangprecha, S.; Makkoch, J. Global alert to avian influenza virus infection: From H5N1 to H7N9. Pathog. Glob. Health 2013, 107, 217–223. [Google Scholar] [CrossRef]
- Zhang, F.; Bi, Y.; Wang, J.; Wong, G.; Shi, W.; Hu, F.; Yang, Y.; Yang, L.; Deng, X.; Jiang, S. Human infections with recently-emerging highly pathogenic H7N9 avian influenza virus in China. J. Infect. 2017, 75, 71–75. [Google Scholar] [CrossRef]
- Yuan, R.; Liang, L.; Wu, J.; Kang, Y.; Song, Y.; Zou, L.; Zhang, X.; Ni, H.; Ke, C. Human infection with an avian influenza A/H9N2 virus in Guangdong in 2016. J. Infect. 2017, 74, 422–425. [Google Scholar] [CrossRef]
- Taubenberger, J.K.; Morens, D.M. Influenza: The once and future pandemic. Public Health Rep. 2010, 125, 16–26. [Google Scholar] [CrossRef]
- Jiang, H.; Wu, P.; Uyeki, T.M.; He, J.; Deng, Z.; Xu, W.; Lv, Q.; Zhang, J.; Wu, Y.; Tsang, T.K. Preliminary epidemiologic assessment of human infections with highly pathogenic avian influenza A(H5N6) virus, China. Clin. Infect. Dis. 2017, 65, 3. [Google Scholar] [CrossRef]
- Lai, S.; Ying, Q.; Cowling, B.J.; Xiang, R.; Wardrop, N.A.; Gilbert, M.; Tsang, T.K.; Peng, W.; Feng, L.; Hui, J. Global epidemiology of avian influenza A H5N1 virus infection in humans, 1997–2015: A systematic review of individual case data. Lancet Infect. Dis. 2016, 16, e108–e118. [Google Scholar] [CrossRef]
- Iuliano, A.D.; Jang, Y.; Jones, J.; Davis, C.T.; Wentworth, D.E.; Uyeki, T.M.; Roguski, K.; Thompson, M.G.; Gubareva, L.; Fry, A.M. Increase in Human Infections with Avian Influenza A(H7N9) Virus During the Fifth Epidemic China, October 2016-February 2017. Morb. Mortal. Wkly. Rep. 2017, 66, 254. [Google Scholar] [CrossRef] [PubMed]
- Swayne, D.E. Impact of vaccines and vaccination on global control of avian influenza. Avian Dis. 2012, 56, 818–828. [Google Scholar] [CrossRef] [PubMed]
- Giovanni, C.; Adelaide, M.; Nigel, T.; Bianca, Z.; Alessandra, B.; Eleonora, M.; Mona Meherez, A.; Abdel, A.; Ilaria, C. Antigenic drift in H5N1 avian influenza virus in poultry is driven by mutations in major antigenic sites of the hemagglutinin molecule analogous to those for human influenza virus. J. Virol. 2011, 85, 8718–8724. [Google Scholar]
- Swayne, D.E.; Kapczynski, D. Strategies and challenges for eliciting immunity against avian influenza virus in birds. Immunol. Rev. 2010, 225, 314–331. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.W.; Senne, D.A.; Suarez, D.L. Effect of vaccine use in the evolution of Mexican lineage H5N2 avian influenza virus. J. Virol. 2004, 78, 8372–8381. [Google Scholar] [CrossRef] [PubMed]
- Neutra, M.R.; Kozlowski, P.A. Mucosal vaccines: The promise and the challenge. Nat. Rev. Immunol. 2006, 6, 148–158. [Google Scholar] [CrossRef]
- Cao, X. Self-regulation and cross-regulation of pattern-recognition receptor signalling in health and disease. Nat. Rev. Immunol. 2016, 16, 35–50. [Google Scholar] [CrossRef]
- Chen, X.; Liu, S.; Goraya, M.U.; Maarouf, M.; Huang, S.; Chen, J.L. Host Immune Response to Influenza A Virus Infection. Front. Immunol. 2018, 9, 320. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. The role of pattern-recognition receptors in innate immunity: Update on Toll-like receptors. Nat. Immunol. 2010, 11, 373–384. [Google Scholar] [CrossRef]
- Gupta, S.K.; Deb, R.; Dey, S.; Chellappa, M.M. Toll-like receptor-based adjuvants: Enhancing the immune response to vaccines against infectious diseases of chicken. Expert Rev. Vaccines 2014, 13, 909–925. [Google Scholar] [CrossRef] [PubMed]
- Lund, J.M.; Alexopoulou, L.; Sato, A.; Karow, M.; Adams, N.C.; Gale, N.W.; Iwasaki, A.; Flavell, R.A. Recognition of single-stranded RNA viruses by Toll-like receptor 7. Proc. Natl. Acad. Sci. USA 2004, 101, 5598–5603. [Google Scholar] [CrossRef] [PubMed]
- Pichlmair, A.; Schulz, O.; Tan, C.P.; Naslund, T.I.; Liljestrom, P.; Weber, F.; Reis e Sousa, C. RIG-I-mediated antiviral responses to single-stranded RNA bearing 5’-phosphates. Science 2006, 314, 997–1001. [Google Scholar] [CrossRef] [PubMed]
- Munir, M. TRIM proteins: Another class of viral victims. Sci. Signal. 2010, 3, jc2. [Google Scholar] [CrossRef]
- Bowie, A.G.; Unterholzner, L. Viral evasion and subversion of pattern-recognition receptor signalling. Nat. Rev. Immunol. 2008, 8, 911–922. [Google Scholar] [CrossRef] [PubMed]
- Yoneyama, M.; Onomoto, K.; Jogi, M.; Akaboshi, T.; Fujita, T. Viral RNA detection by RIG-I-like receptors. Curr. Opin. Immunol. 2015, 32, 48–53. [Google Scholar] [CrossRef]
- Hiscott, J.; Lin, R.; Nakhaei, P.; Paz, S. MasterCARD: A priceless link to innate immunity. Trends. Mol. Med. 2006, 12, 53–56. [Google Scholar] [CrossRef]
- Zou, J.; Chang, M.; Nie, P.; Secombes, C.J. Origin and evolution of the RIG-I like RNA helicase gene family. BMC Evol. Biol. 2009, 9, 85. [Google Scholar] [CrossRef]
- Franchi, L.; Eigenbrod, T.; Munoz-Planillo, R.; Nunez, G. The inflammasome: A caspase-1-activation platform that regulates immune responses and disease pathogenesis. Nat. Immunol. 2009, 10, 241–247. [Google Scholar] [CrossRef]
- Philpott, D.J.; Sorbara, M.T.; Robertson, S.J.; Croitoru, K.; Girardin, S.E. NOD proteins: Regulators of inflammation in health and disease. Nat. Rev. Immunol. 2014, 14, 9–23. [Google Scholar] [CrossRef]
- McAuley, J.L.; Tate, M.D.; MacKenzie-Kludas, C.J.; Pinar, A.; Zeng, W.; Stutz, A.; Latz, E.; Brown, L.E.; Mansell, A. Activation of the NLRP3 inflammasome by IAV virulence protein PB1-F2 contributes to severe pathophysiology and disease. PLoS Pathog. 2013, 9, e1003392. [Google Scholar] [CrossRef] [PubMed]
- Ichinohe, T.; Pang, I.K.; Iwasaki, A. Influenza virus activates inflammasomes via its intracellular M2 ion channel. Nat. Immunol. 2010, 11, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Ichinohe, T.; Lee, H.K.; Ogura, Y.; Flavell, R.; Iwasaki, A. Inflammasome recognition of influenza virus is essential for adaptive immune responses. J. Exp. Med. 2009, 206, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Allen, I.C.; Scull, M.A.; Moore, C.B.; Holl, E.K.; McElvania-TeKippe, E.; Taxman, D.J.; Guthrie, E.H.; Pickles, R.J.; Ting, J.P. The NLRP3 inflammasome mediates in vivo innate immunity to influenza A virus through recognition of viral RNA. Immunity 2009, 30, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.G.; Dash, P.; Aldridge, J.R., Jr.; Ellebedy, A.H.; Reynolds, C.; Funk, A.J.; Martin, W.J.; Lamkanfi, M.; Webby, R.J.; Boyd, K.L.; et al. The intracellular sensor NLRP3 mediates key innate and healing responses to influenza A virus via the regulation of caspase-1. Immunity 2009, 30, 566–575. [Google Scholar] [CrossRef] [PubMed]
- Krammer, F. The human antibody response to influenza A virus infection and vaccination. Nat. Rev. Immunol. 2019, 19, 383–397. [Google Scholar] [CrossRef]
- Van de Sandt, C.E.; Kreijtz, J.H.; Rimmelzwaan, G.F. Evasion of influenza A viruses from innate and adaptive immune responses. Viruses 2012, 4, 1438–1476. [Google Scholar] [CrossRef]
- Schulman, J.L.; Khakpour, M.; Kilbourne, E.D. Protective effects of specific immunity to viral neuraminidase on influenza virus infection of mice. J. Virol. 1968, 2, 778–786. [Google Scholar] [CrossRef]
- LaMere, M.W.; Lam, H.T.; Moquin, A.; Haynes, L.; Lund, F.E.; Randall, T.D.; Kaminski, D.A. Contributions of antinucleoprotein IgG to heterosubtypic immunity against influenza virus. J. Immunol. 2011, 186, 4331–4339. [Google Scholar] [CrossRef]
- Vanderven, H.A.; Ana-Sosa-Batiz, F.; Jegaskanda, S.; Rockman, S.; Laurie, K.; Barr, I.; Chen, W.; Wines, B.; Hogarth, P.M.; Lambe, T.; et al. What Lies Beneath: Antibody Dependent Natural Killer Cell Activation by Antibodies to Internal Influenza Virus Proteins. EBioMedicine 2016, 8, 277–290. [Google Scholar] [CrossRef]
- Ingulli, E.; Mondino, A.; Khoruts, A.; Jenkins, M.K. In vivo detection of dendritic cell antigen presentation to CD4(+) T cells. J. Exp. Med. 1997, 185, 2133–2141. [Google Scholar] [CrossRef] [PubMed]
- Lukens, M.V.; Kruijsen, D.; Coenjaerts, F.E.; Kimpen, J.L.; van Bleek, G.M. Respiratory syncytial virus-induced activation and migration of respiratory dendritic cells and subsequent antigen presentation in the lung-draining lymph node. J. Virol. 2009, 83, 7235–7243. [Google Scholar] [CrossRef] [PubMed]
- Pape, K.A.; Khoruts, A.; Mondino, A.; Jenkins, M.K. Inflammatory cytokines enhance the in vivo clonal expansion and differentiation of antigen-activated CD4+ T cells. J. Immunol. 1997, 159, 591–598. [Google Scholar] [PubMed]
- Zhu, J.; Paul, W.E. Heterogeneity and plasticity of T helper cells. Cell. Res. 2010, 20, 4–12. [Google Scholar] [CrossRef] [PubMed]
- Belz, G.T.; Wodarz, D.; Diaz, G.; Nowak, M.A.; Doherty, P.C. Compromised influenza virus-specific CD8(+)-T-cell memory in CD4(+)-T-cell-deficient mice. J. Virol. 2002, 76, 12388–12393. [Google Scholar] [CrossRef] [PubMed]
- Okoye, I.S.; Wilson, M.S. CD4+ T helper 2 cells--microbial triggers, differentiation requirements and effector functions. Immunology 2011, 134, 368–377. [Google Scholar] [CrossRef]
- Strutt, T.M.; McKinstry, K.K.; Dibble, J.P.; Winchell, C.; Kuang, Y.; Curtis, J.D.; Huston, G.; Dutton, R.W.; Swain, S.L. Memory CD4+ T cells induce innate responses independently of pathogen. Nat. Med. 2010, 16, 558–564, 1p following 564. [Google Scholar] [CrossRef]
- Teijaro, J.R.; Turner, D.; Pham, Q.; Wherry, E.J.; Lefrancois, L.; Farber, D.L. Cutting edge: Tissue-retentive lung memory CD4 T cells mediate optimal protection to respiratory virus infection. J. Immunol. 2011, 187, 5510–5514. [Google Scholar] [CrossRef]
- Brown, D.M.; Dilzer, A.M.; Meents, D.L.; Swain, S.L. CD4 T cell-mediated protection from lethal influenza: Perforin and antibody-mediated mechanisms give a one-two punch. J. Immunol. 2006, 177, 2888–2898. [Google Scholar] [CrossRef]
- He, L.; Gu, W.; Wang, M.; Chang, X.; Sun, X.; Zhang, Y.; Lin, X.; Yan, C.; Fan, W.; Su, P.; et al. Extracellular matrix protein 1 promotes follicular helper T cell differentiation and antibody production. Proc. Natl. Acad. Sci. USA 2018, 115, 8621–8626. [Google Scholar] [CrossRef]
- Ho, A.W.S.; Nayana, P.; Richard John, B.; Moyar Qing, G.; Xilei, D.; Paul Edward, H.; Chuin, L.F.; Kok Loon, W.; Brendon John, H.; Macary, P.A. Lung CD103+ dendritic cells efficiently transport influenza virus to the lymph node and load viral antigen onto MHC class I for presentation to CD8 T cells. J. Immunol. 2011, 187, 6011–6021. [Google Scholar] [CrossRef]
- Pipkin, M.E.; Sacks, J.A.; Cruz-Guilloty, F.; Lichtenheld, M.G.; Bevan, M.J.; Rao, A. Interleukin-2 and Inflammation Induce Distinct Transcriptional Programs that Promote the Differentiation of Effector Cytolytic T Cells. Immunity 2010, 32, 91–103. [Google Scholar] [CrossRef]
- Nakanishi, Y.; Lu, B.; Gerard, C.; Iwasaki, A. CD8(+) T lymphocyte mobilization to virus-infected tissue requires CD4(+) T-cell help. Nature 2009, 462, 510–513. [Google Scholar] [CrossRef]
- Andrade, F. Non-cytotoxic antiviral activities of granzymes in the context of the immune antiviral state. Immunol. Rev. 2010, 235, 128–146. [Google Scholar] [CrossRef]
- Van, D.R.; Bovenschen, N. Cell death-independent functions of granzymes: Hit viruses where it hurts. Rev. Med. Virol. 2011, 21, 301–314. [Google Scholar]
- Allie, S.R.; Randall, T.D. Pulmonary immunity to viruses. Clin. Sci. (Lond) 2017, 131, 1737–1762. [Google Scholar] [CrossRef] [PubMed]
- Grant, E.J.; Quinones-Parra, S.M.; Clemens, E.B.; Kedzierska, K. Human influenza viruses and CD8(+) T cell responses. Curr. Opin. Virol. 2016, 16, 132–142. [Google Scholar] [CrossRef] [PubMed]
- Pulendran, B.; Maddur, M.S. Innate immune sensing and response to influenza. Curr Top. Microbiol. Immunol. 2015, 386, 23–71. [Google Scholar] [PubMed]
- Ito, R.; Ozaki, Y.A.; Yoshikawa, T.; Hasegawa, H.; Sato, Y.; Suzuki, Y.; Inoue, R.; Morishima, T.; Kondo, N.; Sata, T. Roles of anti-hemagglutinin IgA and IgG antibodies in different sites of the respiratory tract of vaccinated mice in preventing lethal influenza pneumonia. Vaccine 2003, 21, 2362–2371. [Google Scholar] [CrossRef]
- Suzuki, T.; Kawaguchi, A.; Ainai, A.; Tamura, S.; Ito, R.; Multihartina, P.; Setiawaty, V.; Pangesti, K.N.; Odagiri, T.; Tashiro, M.; et al. Relationship of the quaternary structure of human secretory IgA to neutralization of influenza virus. Proc. Natl. Acad. Sci. USA 2015, 112, 7809–7814. [Google Scholar] [CrossRef]
- Brandtzaeg, P. Induction of secretory immunity and memory at mucosal surfaces. Vaccine 2007, 25, 5467–54844. [Google Scholar] [CrossRef] [PubMed]
- Tamura, S.; Funato, H.; Hirabayashi, Y.; Suzuki, Y.; Nagamine, T.; Aizawa, C.; Kurata, T. Cross-protection against influenza A virus infection by passively transferred respiratory tract IgA antibodies to different hemagglutinin molecules. Eur. J. Immunol. 1991, 21, 1337–1344. [Google Scholar] [CrossRef] [PubMed]
- Sano, K.; Ainai, A.; Suzuki, T.; Hasegawa, H. Intranasal inactivated influenza vaccines for the prevention of seasonal influenza epidemics. Expert. Rev. Vaccines 2018, 17, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Asahi-Ozaki, Y.; Yoshikawa, T.; Iwakura, Y.; Suzuki, Y.; Tamura, S.; Kurata, T.; Sata, T. Secretory IgA antibodies provide cross-protection against infection with different strains of influenza B virus. J. Med. Virol. 2004, 74, 328–335. [Google Scholar] [CrossRef]
- Yel, L. Selective IgA Deficiency. J. Clin. Immunol. 2010, 30, 10–16. [Google Scholar] [CrossRef]
- Bemark, M.; Boysen, P.; Lycke, N.Y. Induction of gut IgA production through T cell-dependent and T cell-independent pathways. Ann. N. Y. Acad. Sci. 2012, 1247, 97–116. [Google Scholar] [CrossRef]
- Van Ginkel, F.W.; Nguyen, H.H.; McGhee, J.R. Vaccines for mucosal immunity to combat emerging infectious diseases. Emerg. Infect. Dis. 2000, 6, 123–132. [Google Scholar] [CrossRef]
- Spalding, D.M.; Williamson, S.I.; Koopman, W.J.; McGhee, J.R. Preferential induction of polyclonal IgA secretion by murine Peyer’s patch dendritic cell-T cell mixtures. J. Exp. Med. 1984, 160, 941–946. [Google Scholar] [CrossRef]
- Rajao, D.S.; Perez, D.R. Universal Vaccines and Vaccine Platforms to Protect against Influenza Viruses in Humans and Agriculture. Front. Microbiol. 2018, 9, 123. [Google Scholar] [CrossRef]
- Soema, P.C.; Kompier, R.; Amorij, J.P.; Kersten, G.F. Current and next generation influenza vaccines: Formulation and production strategies. Eur. J. Pharm. Biopharm. 2015, 94, 251–263. [Google Scholar] [CrossRef]
- Vincent, A.L.; Ciacci-Zanella, J.R.; Lorusso, A.; Gauger, P.C.; Zanella, E.L.; Kehrli, M.E.K., Jr.; Janke, B.H.; Lager, K.M. Efficacy of inactivated swine influenza virus vaccines against the 2009 A/H1N1 influenza virus in pigs. Vaccine 2010, 28, 2782–2787. [Google Scholar] [CrossRef] [PubMed]
- Neurath, A.R.; Rubin, B.A.; Sillaman, J.; Tint, H. The effect of nonaqueous solvents on the quaternary structure of viruses: A procedure for the simultaneous concentration, purification and disruption of influenza viruses. Microbios 1971, 4, 145–150. [Google Scholar] [PubMed]
- Morokutti, A.; Muster, T.; Ferko, B. Intranasal vaccination with a replication-deficient influenza virus induces heterosubtypic neutralising mucosal IgA antibodies in humans. Vaccine 2014, 32, 1897–1900. [Google Scholar] [CrossRef] [PubMed]
- Ross, K.F.; Herzberg, M.C. Autonomous immunity in mucosal epithelial cells: Fortifying the barrier against infection. Microbes. Infect. 2016, 18, 387–398. [Google Scholar] [CrossRef] [PubMed]
- Tamura, S.; Tanimoto, T.; Kurata, T. Mechanisms of broad cross-protection provided by influenza virus infection and their application to vaccines. Jpn. J. Infect. Dis. 2005, 58, 195–207. [Google Scholar]
- Hasegawa, H.; Ichinohe, T.; Ainai, A.; Tamura, S.; Kurata, T. Development of mucosal adjuvants for intranasal vaccine for H5N1 influenza viruses. Ther. Clin. Risk. Manag. 2009, 5, 125–132. [Google Scholar] [CrossRef]
- Tamura, S.; Yamanaka, A.; Shimohara, M.; Tomita, T.; Komase, K.; Tsuda, Y.; Suzuki, Y.; Nagamine, T.; Kawahara, K.; Danbara, H.; et al. Synergistic action of cholera toxin B subunit (and Escherichia coli heat-labile toxin B subunit) and a trace amount of cholera whole toxin as an adjuvant for nasal influenza vaccine. Vaccine 1994, 12, 419–426. [Google Scholar] [CrossRef]
- Haredy, A.M.; Takenaka, N.; Yamada, H.; Sakoda, Y.; Okamatsu, M.; Yamamoto, N.; Omasa, T.; Ohtake, H.; Mori, Y.; Kida, H.; et al. An MDCK cell culture-derived formalin-inactivated influenza virus whole-virion vaccine from an influenza virus library confers cross-protective immunity by intranasal administration in mice. Clin. Vaccine Immunol. 2013, 20, 998–1007. [Google Scholar] [CrossRef]
- Okamoto, S.; Matsuoka, S.; Takenaka, N.; Haredy, A.M.; Tanimoto, T.; Gomi, Y.; Ishikawa, T.; Akagi, T.; Akashi, M.; Okuno, Y.; et al. Intranasal immunization with a formalin-inactivated human influenza A virus whole-virion vaccine alone and intranasal immunization with a split-virion vaccine with mucosal adjuvants show similar levels of cross-protection. Clin. Vaccine Immunol. 2012, 19, 979–990. [Google Scholar] [CrossRef]
- Maassab, H.F.; Francis, T., Jr.; Davenport, F.M.; Hennessy, A.V.; Minuse, E.; Anderson, G. Laboratory and clinical characteristics of attenuated strains of influenza virus. Bull. World. Health. Organ. 1969, 41, 589. [Google Scholar]
- Manicassamy, S.; Pulendran, B. Modulation of adaptive immunity with Toll-like receptors. Semin. Immunol. 2009, 21, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Pasetti, M.F.; Simon, J.K.; Sztein, M.B.; Levine, M.M. Immunology of gut mucosal vaccines. Immunol. Rev. 2011, 239, 125–148. [Google Scholar] [CrossRef] [PubMed]
- Lycke, N. Recent progress in mucosal vaccine development: Potential and limitations. Nat. Rev. Immunol. 2012, 12, 592–605. [Google Scholar] [CrossRef]
- Santos, J.J.S.; Obadan, A.O.; Garcia, S.C.; Carnaccini, S.; Kapczynski, D.R.; Pantin-Jackwood, M.; Suarez, D.L.; Perez, D.R. Short- and long-term protective efficacy against clade 2.3.4.4 H5N2 highly pathogenic avian influenza virus following prime-boost vaccination in turkeys. Vaccine 2017, 35, 5637–5643. [Google Scholar] [CrossRef]
- Slepushkin, A.N.; Dukova, V.S.; Kalegaeva, V.A.; Kagan, A.N.; Temriuk, E.E. Results of studying the effectiveness of a live influenza vaccine for peroral use on preschool and schoolchildren. Mikrobiol. Epidemiol. Immunobiol. 1974, 12, 24–29. [Google Scholar]
- Rudenko, L.; Yeolekar, L.; Kiseleva, I.; Isakovasivak, I. Development and approval of live attenuated influenza vaccines based on Russian master donor viruses: Process challenges and success stories. Vaccine 2016, 34, 5436–5441. [Google Scholar] [CrossRef]
- Rudenko, L.; Kiseleva, I.; Krutikova, E.; Stepanova, E.; Rekstin, A.; Donina, S.; Pisareva, M.; Grigorieva, E.; Kryshen, K.; Muzhikyan, A.; et al. Rationale for vaccination with trivalent or quadrivalent live attenuated influenza vaccines: Protective vaccine efficacy in the ferret model. PLoS ONE 2018, 13, e0208028. [Google Scholar] [CrossRef]
- Ruiz-Palacios, G.M.; Irene, P.S.; F Raúl, V.; Hector, A.; Thomas, B.; Sueann Costa, C.; Brigitte, C.; Felix, E.; Paul, G.; Innis, B.L. Safety and efficacy of an attenuated vaccine against severe rotavirus gastroenteritis. N. Engl. J. Med. 2006, 354, 11. [Google Scholar] [CrossRef]
- Kirkpatrick, B.D.; Mckenzie, R.; O’Neill, J.P.; Larsson, C.J.; Bourgeois, A.L.; Shimko, J.; Bentley, M.; Makin, J.; Chatfield, S.; Hindle, Z. Evaluation of Salmonella enterica serovar Typhi (Ty2 aroC-ssaV-) M01ZH09, with a defined mutation in the Salmonella pathogenicity island 2, as a live, oral typhoid vaccine in human volunteers. Vaccine 2006, 24, 116–123. [Google Scholar] [CrossRef]
- John, S.; Lowen, A.C.; Lindomar, P.; Matthew, A.; Alicia, S.; Randy, A.; Perez, D.R.; Adolfo, G.S.; Peter, P. Live attenuated influenza viruses containing NS1 truncations as vaccine candidates against H5N1 highly pathogenic avian influenza. J. Virol. 2009, 83, 1742. [Google Scholar]
- Langley, J.M.; Louise, F.; Robert, J.; Halperin, S.A.; Michael, K.; Laurence, C.; Shelly, M.N.; Mamadou, D.; Philippe, M.; Louis, F. Immunogenicity of heterologous H5N1 influenza booster vaccination 6 or 18 months after primary vaccination in adults: A randomized controlled clinical trial. Vaccine 2015, 33, 559–567. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nicola, P.; Susanna, E. Adjuvanted influenza vaccines. Hum. Vaccin. 2012, 8, 59–66. [Google Scholar]
- Liang, J.; Fu, J.; Kang, H.; Lin, J.; Yu, Q.; Yang, Q. Comparison of 3 kinds of Toll-like receptor ligands for inactivated avian H5N1 influenza virus intranasal immunization in chicken. Poult. Sci. 2013, 92, 2651–2660. [Google Scholar] [CrossRef] [PubMed]
- Chaung, H.C.; Cheng, L.T.; Hung, L.H.; Tsai, P.C.; Skountzou, I.; Wang, B.; Compans, R.W.; Lien, Y.Y. Salmonella flagellin enhances mucosal immunity of avian influenza vaccine in chickens. Vet. Microbiol. 2012, 157, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.A.; Gomis, S.; Dar, A.; Willson, P.J.; Babiuk, L.A.; Potter, A.; Mutwiri, G.; Tikoo, S.K. Oligodeoxynucleotides containing CpG motifs (CpG-ODN) predominantly induce Th1-type immune response in neonatal chicks. Dev. Comp. Immunol. 2008, 32, 1041–1049. [Google Scholar] [CrossRef]
- St Paul, M.; Brisbin, J.T.; Abdul-Careem, M.F.; Sharif, S. Immunostimulatory properties of Toll-like receptor ligands in chickens. Vet. Immunol. Immunopathol. 2013, 152, 191–199. [Google Scholar] [CrossRef]
- Girard, F.; Pery, P.; Naciri, M.; Quere, P. Adjuvant effect of cholera toxin on systemic and mucosal immune responses in chickens infected with E. tenella or given recombinant parasitic antigen per os. Vaccine 1999, 17, 1516–1524. [Google Scholar] [CrossRef]
- Baptista, A.A.; Donato, T.C.; Garcia, K.C.; Goncalves, G.A.; Coppola, M.P.; Okamoto, A.S.; Sequeira, J.L.; Andreatti Filho, R.L. Immune response of broiler chickens immunized orally with the recombinant proteins flagellin and the subunit B of cholera toxin associated with Lactobacillus spp. Poult. Sci. 2014, 93, 39–45. [Google Scholar] [CrossRef]
- Lei, H.; Sheng, Z.; Ding, Q.; Chen, J.; Wei, X.; Lam, D.M.; Xu, Y. Evaluation of oral immunization with recombinant avian influenza virus HA1 displayed on the Lactococcus lactis surface and combined with the mucosal adjuvant cholera toxin subunit B. Clin. Vaccine Immunol. 2011, 18, 1046–1051. [Google Scholar] [CrossRef]
- Lalsiamthara, J.; Lee, J.H. Immunization with Salmonella Enteritidis secreting mucosal adjuvant labile toxin confers protection against wild type challenge via augmentation of CD3(+)CD4(+) T-cell proliferation and enhancement of IFN-gamma, IL-6 and IL-10 expressions in chicken. Vaccine 2017, 35, 767–773. [Google Scholar] [CrossRef]
- Katz, J.M.; Lu, X.; Young, S.A.; Galphin, J.C. Adjuvant activity of the heat-labile enterotoxin from enterotoxigenic Escherichia coli for oral administration of inactivated influenza virus vaccine. J. Infect. Dis. 1997, 175, 352–363. [Google Scholar] [CrossRef] [PubMed]
- St Paul, M.; Mallick, A.I.; Read, L.R.; Villanueva, A.I.; Parvizi, P.; Abdul-Careem, M.F.; Nagy, E.; Sharif, S. Prophylactic treatment with Toll-like receptor ligands enhances host immunity to avian influenza virus in chickens. Vaccine 2012, 30, 4524–4531. [Google Scholar] [CrossRef] [PubMed]
- Scheepers, K.; Becht, H. Protection of mice against an influenza virus infection by oral vaccination with viral nucleoprotein incorporated into immunostimulating complexes. Med. Microbiol. Immunol. 1994, 183, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Zhao, Y.; Liu, J.; Ji, X.; Meng, L.; Wang, T.; Sun, W.; Zhang, K.; Sang, X.; Yu, Z.; et al. Inclusion of membrane-anchored LTB or flagellin protein in H5N1 virus-like particles enhances protective responses following intramuscular and oral immunization of mice. Vaccine 2018, 36, 5990–5998. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shan, C.; Ming, S.; Liu, Y.; Du, Y.; Jiang, G. Immunoadjuvant effects of bacterial genomic DNA and CpG oligodeoxynucleotides on avian influenza virus subtype H5N1 inactivated oil emulsion vaccine in chicken. Res. Vet. Sci. 2009, 86, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Hung, L.H.; Tsai, P.C.; Wang, C.H.; Li, S.L.; Huang, C.C.; Lien, Y.Y.; Chaung, H.C. Immunoadjuvant efficacy of plasmids with multiple copies of a CpG motif coadministrated with avian influenza vaccine in chickens. Vaccine 2011, 29, 4668–4675. [Google Scholar] [CrossRef]
- Tateishi, K.; Fujihashi, K.; Yamamoto, N.; Hasegawa, H.; Ainai, A.; Sato, K.; Iho, S.; Yamamoto, S.; Maeyama, J.I.; Odagiri, T.; et al. CpG ODN G9.1 as a novel nasal ODN adjuvant elicits complete protection from influenza virus infection without causing inflammatory immune responses. Vaccine 2019, 37, 5382–5389. [Google Scholar] [CrossRef]
- Kang, H.; Wang, H.; Yu, Q.; Yang, Q. A novel combined adjuvant strongly enhances mucosal and systemic immunity to low pathogenic avian influenza after oral immunization in ducks. Poult. Sci. 2013, 92, 1543–1551. [Google Scholar] [CrossRef]
- Serradell, M.C.; Rupil, L.L.; Martino, R.A.; Prucca, C.G.; Carranza, P.G.; Saura, A.; Fernandez, E.A.; Gargantini, P.R.; Tenaglia, A.H.; Petiti, J.P.; et al. Efficient oral vaccination by bioengineering virus-like particles with protozoan surface proteins. Nat. Commun. 2019, 10, 361. [Google Scholar] [CrossRef]
- Onuigbo, E.; Iseghohimhen, J.; Chah, K.; Gyang, M.; Attama, A. Chitosan/alginate microparticles for the oral delivery of fowl typhoid vaccine: Innate and acquired immunity. Vaccine 2018, 36, 4973–4978. [Google Scholar] [CrossRef]
- Svindland, S.C.; Jul-Larsen, A.; Pathirana, R.; Andersen, S.; Madhun, A.; Montomoli, E.; Jabbal-Gill, I.; Cox, R.J. The mucosal and systemic immune responses elicited by a chitosan-adjuvanted intranasal influenza H5N1 vaccine. Influenza. Other. Respir. Viruses 2012, 6, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Song, S.K.; Moldoveanu, Z.; Nguyen, H.H.; Kim, E.H.; Choi, K.Y.; Kim, J.B.; Mestecky, J. Intranasal immunization with influenza virus and Korean mistletoe lectin C (KML-C) induces heterosubtypic immunity in mice. Vaccine 2007, 25, 6359–6366. [Google Scholar] [CrossRef]
- Chen, W.-T.; Chang, H.-K.; Lin, C.-C.; Yang, S.-M.; Yin, H.-S. Chicken interleukin-1β mutants are effective single-dose vaccine adjuvants that enhance mucosal immune response. Mol. Immunol 2017, 87, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Schnepf, D.; Becker, J.; Ebert, K.; Tanriver, Y.; Bernasconi, V.; Gad, H.H.; Hartmann, R.; Lycke, N.; Staeheli, P. Interferon-lambda enhances adaptive mucosal immunity by boosting release of thymic stromal lymphopoietin. Nat. Immunol. 2019, 20, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Kimoto, T.; Kim, H.; Sakai, S.; Takahashi, E.; Kido, H. Oral vaccination with influenza hemagglutinin combined with human pulmonary surfactant-mimicking synthetic adjuvant SF-10 induces efficient local and systemic immunity compared with nasal and subcutaneous vaccination and provides protective immunity in mice. Vaccine 2019, 37, 612–622. [Google Scholar]
- Coban, C.; Koyama, S.; Takeshita, F.; Akira, S.; Ishii, K.J. Molecular and cellular mechanisms of DNA vaccines. Hum. Vaccin. 2008, 4, 453–457. [Google Scholar] [CrossRef]
- Kutzler, M.A.; Weiner, D.B. DNA vaccines: Ready for prime time? Nat. Rev. Genet. 2008, 9, 776–788. [Google Scholar] [CrossRef]
- Oh, Y.K.; Kim, J.P.; Hwang, T.S.; Ko, J.J.; Kim, J.M.; Yang, J.S.; Kim, C.K. Nasal absorption and biodistribution of plasmid DNA: An alternative route of DNA vaccine delivery. Vaccine 2001, 19, 4519–4525. [Google Scholar] [CrossRef]
- Fotouhi, F.; Shaffifar, M.; Farahmand, B.; Shirian, S.; Saeidi, M.; Tabarraei, A.; Gorji, A.; Ghaemi, A. Adjuvant use of the NKT cell agonist alpha-galactosylceramide leads to enhancement of M2-based DNA vaccine immunogenicity and protective immunity against influenza A virus. Arch. Virol. 2017, 162, 1251–1260. [Google Scholar] [CrossRef]
- Kraehenbuhl, J.P. Mucosa-targeted DNA vaccination. Trends. Immunol. 2001, 22, 646–648. [Google Scholar] [CrossRef]
- Chadwick, S.; Kriegel, C.; Amiji, M. Nanotechnology solutions for mucosal immunization. Adv. Drug Deliv. Rev. 2010, 62, 394–407. [Google Scholar] [CrossRef] [PubMed]
- Fu-Shi, Q.; Aswani, V.; Compans, R.W.; Sang-Moo, K. Virus-like particle vaccine protects against 2009 H1N1 pandemic influenza virus in mice. PLoS ONE 2010, 5, e9161. [Google Scholar]
- Pushko, P.; Tretyakova, I.; Hidajat, R.; Zsak, A.; Chrzastek, K.; Tumpey, T.M.; Kapczynski, D.R. Virus-like particles displaying H5, H7, H9 hemagglutinins and N1 neuraminidase elicit protective immunity to heterologous avian influenza viruses in chickens. Virology 2017, 501, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Wang, R.; Nie, G. Applications of nanomaterials as vaccine adjuvants. Hum. Vaccin. Immunother. 2014, 10, 2761–2774. [Google Scholar] [CrossRef] [PubMed]
- Florindo, H.F.; Pandit, S.; Gonçalves, L.; Alpar, H.O.; Almeida, A.J. New approach on the development of a mucosal vaccine against strangles: Systemic and mucosal immune responses in a mouse model. Vaccine 2009, 27, 1230–1241. [Google Scholar] [CrossRef]
- Chiou, C.J.; Tseng, L.P.; Deng, M.C.; Jiang, P.R.; Tasi, S.L.; Chung, T.W.; Huang, Y.Y.; Liu, D.Z. Mucoadhesive liposomes for intranasal immunization with an avian influenza virus vaccine in chickens. Biomaterials 2009, 30, 5862–5868. [Google Scholar] [CrossRef]
- Song, M.; Hong, H.A.; Huang, J.M.; Colenutt, C.; Khang, D.D.; Nguyen, T.V.A.; Park, S.M.; Shim, B.S.; Song, H.H.; Cheon, I.S. Killed Bacillus subtilis spores as a mucosal adjuvant for an H5N1 vaccine. Vaccine 2012, 30, 3266–3277. [Google Scholar] [CrossRef]
- Eric, N.; Olivier, G.; Michel, A.; Véronique, J. Prime-boost vaccination with recombinant H5-fowlpox and Newcastle disease virus vectors affords lasting protection in SPF Muscovy ducks against highly pathogenic H5N1 influenza virus. Vaccine 2013, 31, 4121–4128. [Google Scholar]
- Liu, L.; Wang, T.; Wang, M.; Tong, Q.; Sun, Y.; Pu, J.; Sun, H.; Liu, J. Recombinant turkey herpesvirus expressing H9 hemagglutinin providing protection against H9N2 avian influenza. Virology 2019, 529, 7–15. [Google Scholar] [CrossRef]
- Kim, S.H.; Paldurai, A.; Xiao, S.; Collins, P.L.; Samal, S.K. Modified Newcastle disease virus vectors expressing the H5 hemagglutinin induce enhanced protection against highly pathogenic H5N1 avian influenza virus in chickens. Vaccine 2014, 32, 4428–4435. [Google Scholar] [CrossRef]
- Cho, Y.; Lamichhane, B.; Nagy, A.; Chowdhury, I.R.; Samal, S.K.; Kim, S.H. Co-expression of the Hemagglutinin and Neuraminidase by Heterologous Newcastle Disease Virus Vectors Protected Chickens against H5 Clade 2.3.4.4 HPAI Viruses. Sci. Rep. 2018, 8, 16854. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Liu, X.; Jiao, X.; Liu, X. Newcastle disease virus (NDV) recombinant expressing the hemagglutinin of H7N9 avian influenza virus protects chickens against NDV and highly pathogenic avian influenza A (H7N9) virus challenges. Vaccine 2017, 35, 6585. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, T.T. The mucosal immune system. Parasite Immunol. 2003, 25, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Shakya, A.K.; Chowdhury, M.Y.E.; Tao, W.; Gill, H.S. Mucosal vaccine delivery: Current state and a pediatric perspective. J. Control. Release 2016, 240, 394–413. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.M. Immunogenicity and efficacy of oral vaccines in developing countries: Lessons from a live cholera vaccine. BMC Biol. 2010, 8, 129. [Google Scholar] [CrossRef]
- Per, B. Potential of nasopharynx-associated lymphoid tissue for vaccine responses in the airways. Am. J. Respir. Crit. Care Med. 2011, 183, 1595–1604. [Google Scholar]
- Mall, M.A. Role of cilia, mucus, and airway surface liquid in mucociliary dysfunction: Lessons from mouse models. J. Aerosol. Med. Pulm. Drug Deliv. 2008, 21, 13–24. [Google Scholar] [CrossRef]
- Scott, L.J.; Carter, N.J.; Curran, M.P. Live Attenuated Influenza Vaccine (Fluenz™). Drugs 2012, 14, 271–279. [Google Scholar] [CrossRef]
- Tomoda, T.; Morita, H.; Kurashige, T.; Maassab, H.F. Prevention of influenza by the intranasal administration of cold-recombinant, live-attenuated influenza virus vaccine: Importance of interferon-gamma production and local IgA response. Vaccine 1995, 13, 185–190. [Google Scholar] [CrossRef]
- Belshe, R.; Lee, M.S.; Walker, R.E.; Stoddard, J.; Mendelman, P.M. Safety, immunogenicity and efficacy of intranasal, live attenuated influenza vaccine. Expert. Rev. Vaccines 2004, 3, 643–654. [Google Scholar] [CrossRef]
- Rauw, F.; Gardin, Y.; Palya, V.; Borm, S.V.; Gonze, M.; Lemaire, S.; Berg, T.V.D.; Lambrecht, B. Humoral, cell-mediated and mucosal immunity induced by oculo-nasal vaccination of one-day-old SPF and conventional layer chicks with two different live Newcastle disease vaccines. Vaccine 2009, 27, 3631–3642. [Google Scholar] [CrossRef] [PubMed]
| Type of Adjuvant | Composition | Target | Delivery Route | Immune Responses |
|---|---|---|---|---|
| Enterotoxins | Cholera toxin | GM1 | Nasal or oral | Increased specific IgA and IgG [97] |
| the subunit B of cholera toxin | GM1 | Nasal or oral | Increased specific IgA, IgG and T cell response [98,99] | |
| Mutant Escherichia coli heat-labile enterotoxin | GM1 and other gangliosides | Nasal or oral | Increased S-IgA, IgG and T cell response, enhanced IFN-γ, IL-6 and IL-10 cytokine secretion [100,101] | |
| TLR ligands | Poly I:C | TLR3 | Nasal | Decreased oropharyngeal and cloacal virus shedding [93,102] |
| Lipopolysaccharide | TLR4 | Nasal or oral | Decreased oropharyngeal and cloacal virus shedding [102,103] | |
| Flagellin | TLR5 | Nasal or oral | Increased IgY and IgA, protection of lethal viral challenge [94,104] | |
| CpG | TLR9/TL21 | Nasal or oral | Increased S-IgA, IgG and IFN-γ [105,106,107,108] | |
| Variant-specific surface proteins | TLR4 | Oral | Increased IgA, IgG and IFN-γ, protection of viral challenge [109] | |
| Mucoadhesives | Chitosan | Tight junctions | Nasal or oral | Increased IgG and IFN-γ, 100% protection for fowl typhoid [110,111] |
| Lectins | M cells | Nasal | IgG and IgA induction, induced heterosubtypic immunity [112] | |
| Cytokines | chicken interleukin-1 beta | IL-1R | Nasal | Increased specific IgA, S-IgA and IFN-γ [113] |
| IFN-λ | M cells | Nasal | IgG1 and IgA induction, protection of viral challenge [114] | |
| Synthetic adjuvant | SF-10 | Dendritic cells | Oral | Increased specific IgA, S-IgA, IgG and cytokine production [115] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, T.; Wei, F.; Liu, J. Emerging Role of Mucosal Vaccine in Preventing Infection with Avian Influenza A Viruses. Viruses 2020, 12, 862. https://doi.org/10.3390/v12080862
Wang T, Wei F, Liu J. Emerging Role of Mucosal Vaccine in Preventing Infection with Avian Influenza A Viruses. Viruses. 2020; 12(8):862. https://doi.org/10.3390/v12080862
Chicago/Turabian StyleWang, Tong, Fanhua Wei, and Jinhua Liu. 2020. "Emerging Role of Mucosal Vaccine in Preventing Infection with Avian Influenza A Viruses" Viruses 12, no. 8: 862. https://doi.org/10.3390/v12080862
APA StyleWang, T., Wei, F., & Liu, J. (2020). Emerging Role of Mucosal Vaccine in Preventing Infection with Avian Influenza A Viruses. Viruses, 12(8), 862. https://doi.org/10.3390/v12080862





