Quality Assessment of Virus-Like Particles at Single Particle Level: A Comparative Study
Abstract
1. Introduction
2. Materials and Methods
2.1. HEK 293 Mammalian Cell Line, Culture Conditions and Transient Transfection
2.2. Sf9 Insect Cell Line, Culture Conditions and Baculovirus Infection
2.3. Electron Microscopy (EM)
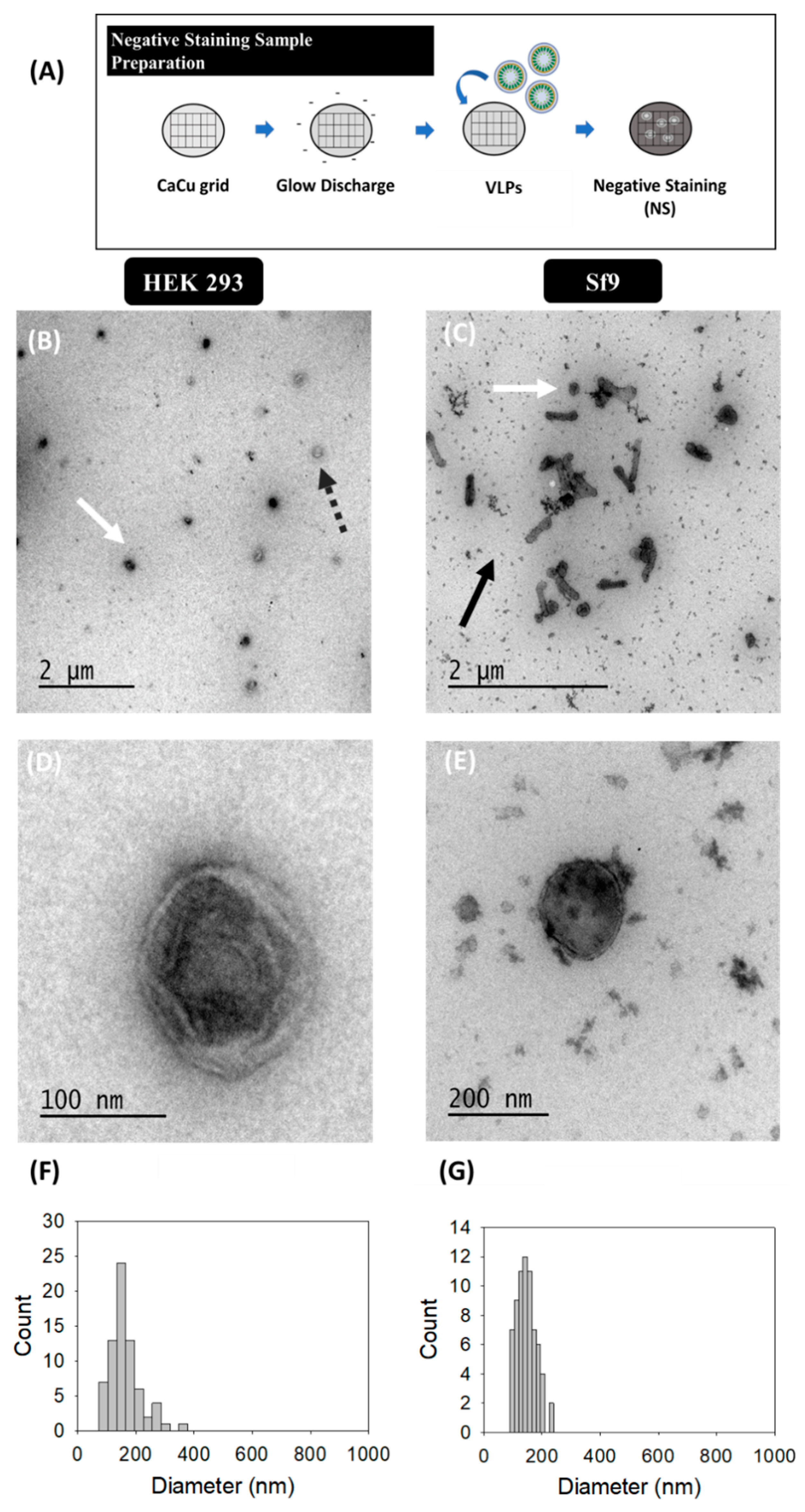
2.3.1. Transmission Electron Microscopy (TEM)-Negative Staining
2.3.2. Scanning Electron Microscopy (SEM)-Alcian Blue Staining
2.3.3. Cryogenic Transmission Electron Microscopy (cryo-TEM)
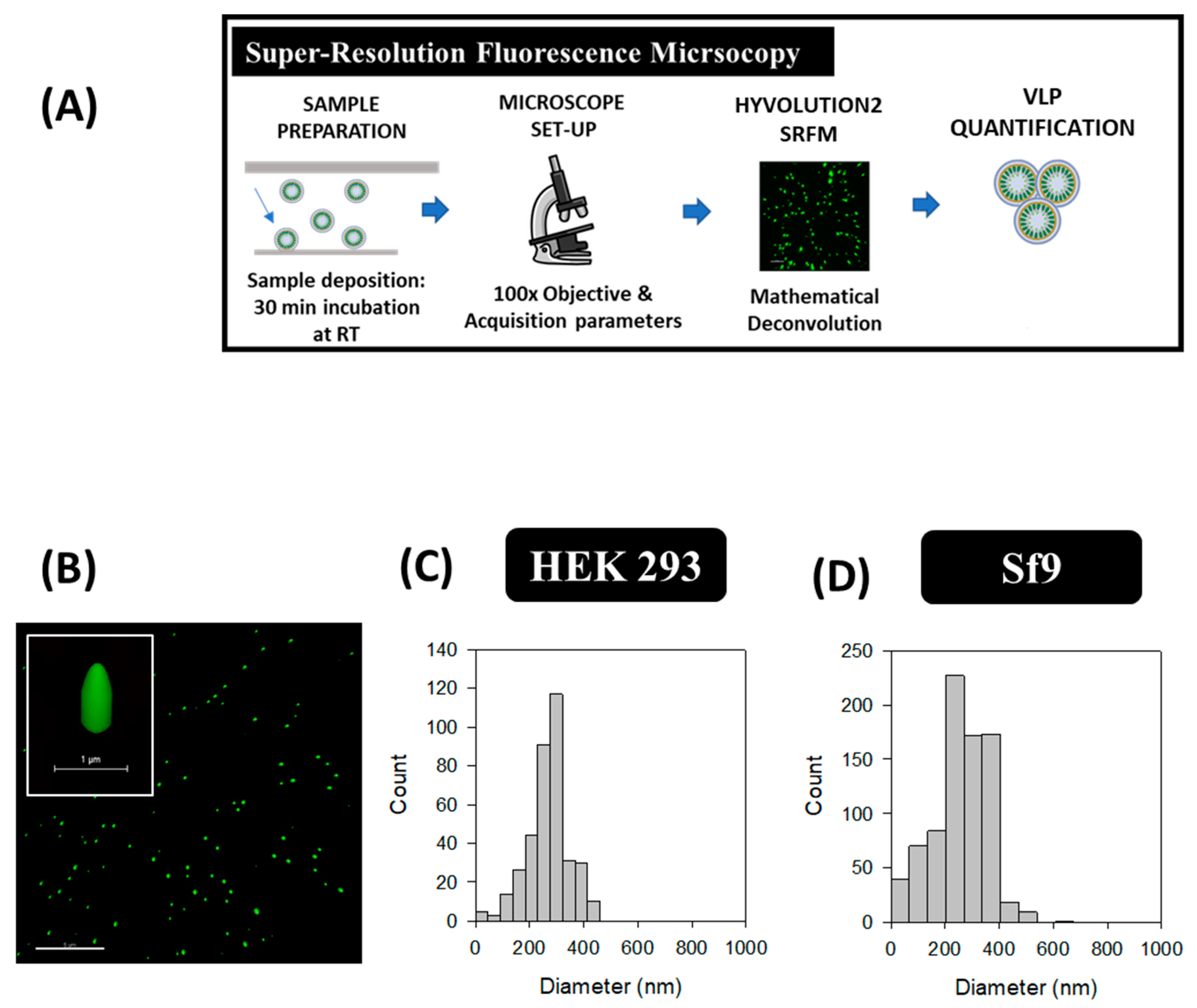
2.4. Super-Resolution Fluorescence Microscopy (SRFM)
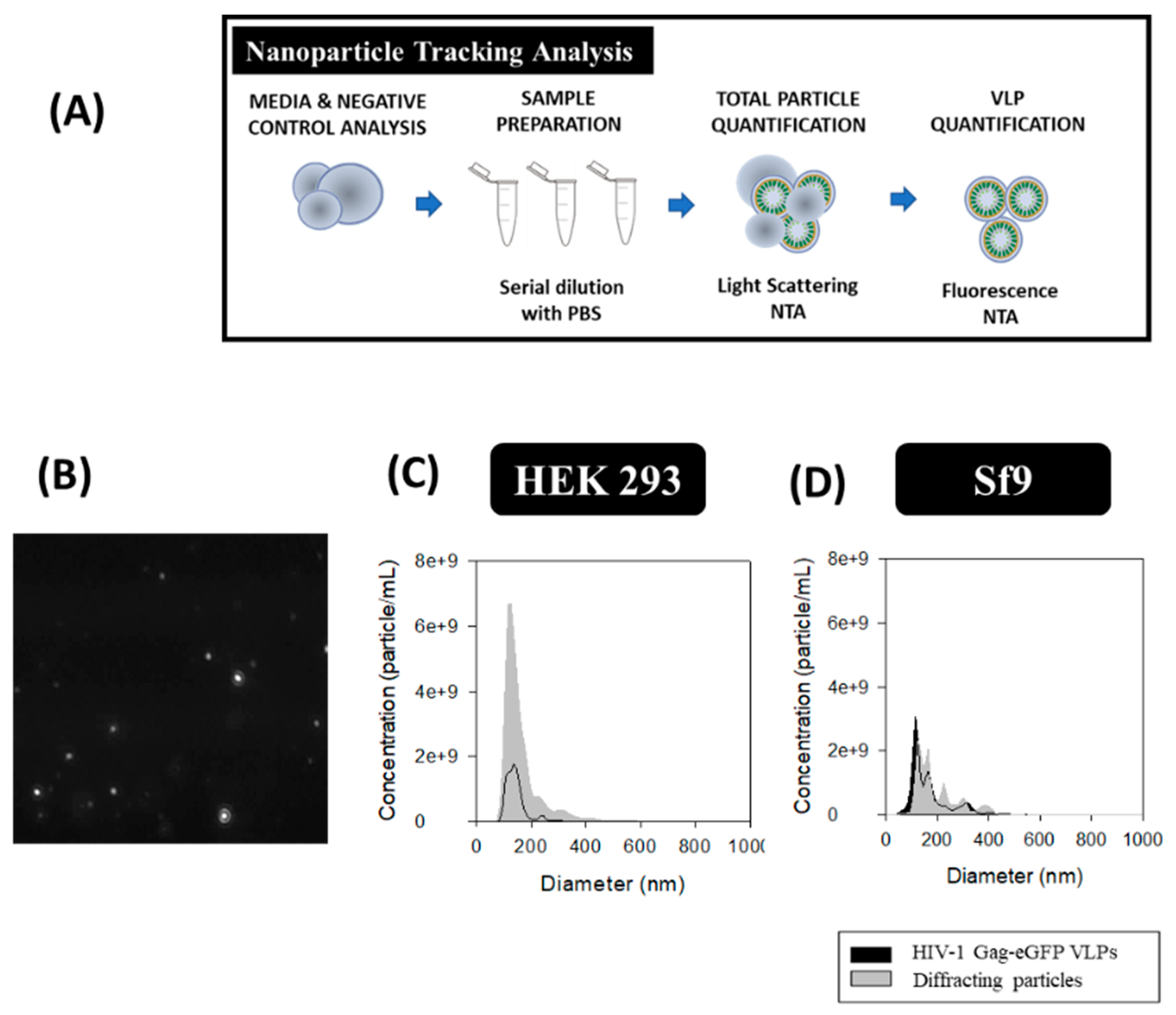
2.5. Nanoparticle Tracking Analysis (NTA)
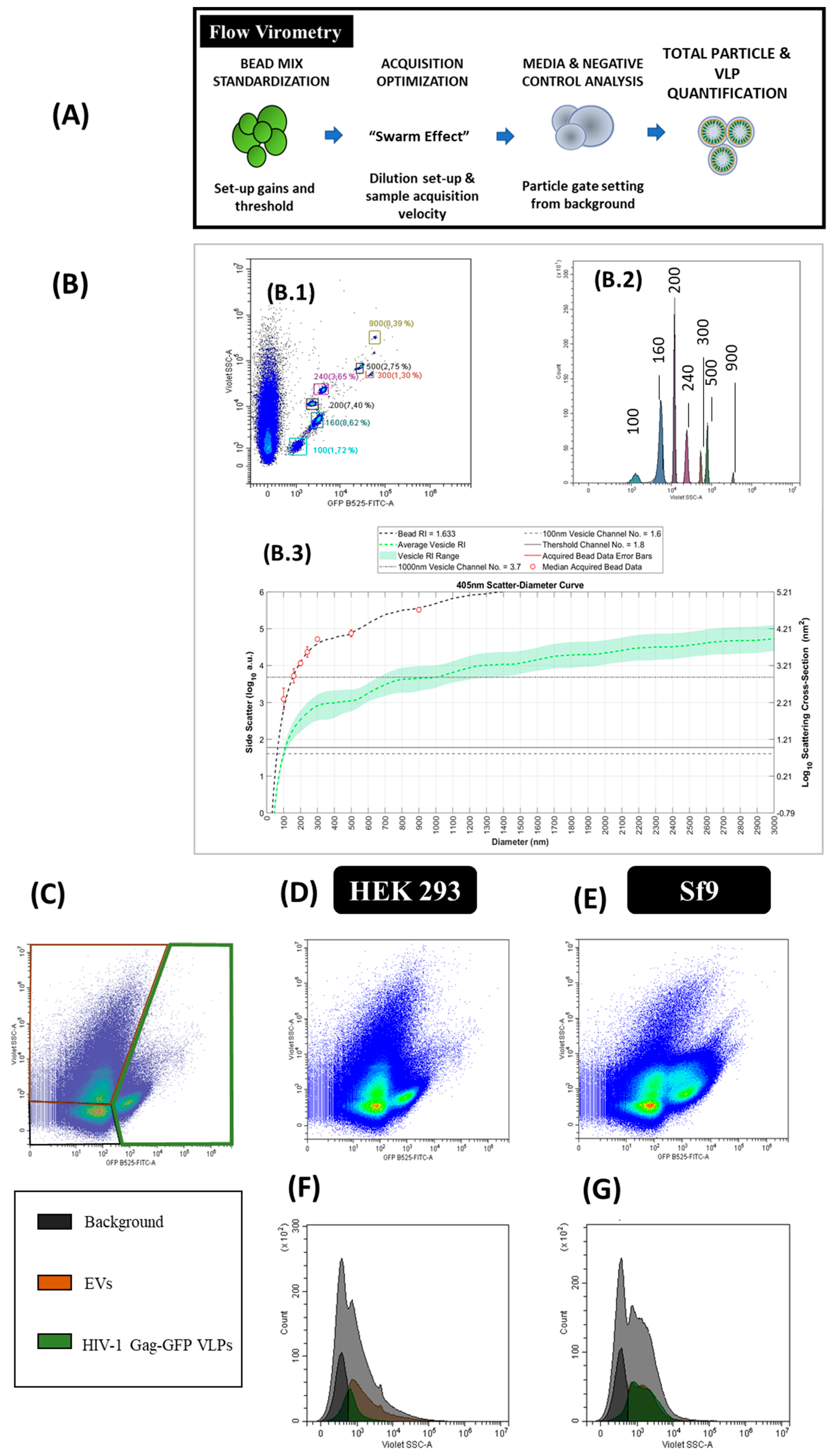
2.6. Flow Virometry
3. Results
3.1. Transmission Electron Microscopy (TEM)-Negative Staining
3.2. Scanning Electron Microscopy (SEM)-Alcian Blue Staining
3.3. Cryogenic Transmission Electron Microscopy (cryo-TEM)
3.4. Super-Resolution Fluorescence Microscopy (SRFM)
3.5. Nanoparticle Tracking Analysis (NTA)
3.6. Flow Virometry
4. Discussion
4.1. Sample Preparation and Equipment Set-up
4.2. Ultrastructural Analysis
4.3. Particle Size Distribution
4.4. Particle Concentration
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AB | Alcian Blue |
| AFM | Atomic Force Microscopy |
| BV | Baculovirus |
| CQA | Critical Quality Attributes |
| Cryo-TEM | Cryogenic Transmission Electron Microscopy |
| DLS | Dynamic Light Scattering |
| EM | Electron Microscopy |
| EV | Extracellular Vesicle |
| cBV | filled baculovirus with vesicle-like or protein content |
| LM | Lipid Membrane |
| MVB | Multivesicular Body |
| NS | Negative Staining |
| NTA | Nanoparticle Tracking Analysis |
| ODV | Occlusion-Derived Baculovirus |
| PSD | Particle Size Distribution |
| RI | Refractive Index |
| rBV | relaxed-form of baculovirus |
| RT | Room Temperature |
| SEM | Scanning Electron Microscopy |
| SRFM | Super-Resolution Fluorescence Microscopy |
| TEM | Transmission Electron Microscopy |
| TRPS | Tunable Resistive Pulse Sensing |
| V-SSC | Violet Side Scatter |
| VLP | Virus-Like Particle |
References
- Charlton Hume, H.K.; Vidigal, J.; Carrondo, M.J.T.; Middelberg, A.P.J.; Roldão, A.; Lua, L.H.L. Synthetic biology for bioengineering virus-like particle vaccines. Biotechnol. Bioeng. 2019, 116, 919–935. [Google Scholar] [CrossRef]
- Cervera, L.; Gòdia, F.; Tarrés-Freixas, F.; Aguilar-Gurrieri, C.; Carrillo, J.; Blanco, J.; Gutiérrez-Granados, S. Production of HIV-1-based virus-like particles for vaccination: Achievements and limits. Appl. Microbiol. Biotechnol. 2019, 103, 7367–7384. [Google Scholar] [CrossRef] [PubMed]
- Lua, L.H.L.; Connors, N.K.; Sainsbury, F.; Chuan, Y.P.; Wibowo, N.; Middelberg, A.P.J. Bioengineering virus-like particles as vaccines. Biotechnol. Bioeng. 2014, 111, 425–440. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.K.; Sahni, N.; Kumru, O.S.; Joshi, S.B.; Volkin, D.B.; Russell Middaugh, C. Formulation and stabilization of recombinant protein based virus-like particle vaccines. Adv. Drug Deliv. Rev. 2015, 93, 42–55. [Google Scholar] [CrossRef] [PubMed]
- Van der Pol, E.; Coumans, F.a.W.; Grootemaat, a.E.; Gardiner, C.; Sargent, I.L.; Harrison, P.; Sturk, A.; van Leeuwen, T.G.; Nieuwland, R. Particle size distribution of exosomes and microvesicles determined by transmission electron microscopy, flow cytometry, nanoparticle tracking analysis, and resistive pulse sensing. J. Thromb. Haemost. 2014, 12, 1182–1192. [Google Scholar] [CrossRef] [PubMed]
- Akers, J.C.; Gonda, D.; Kim, R.; Carter, B.S.; Chen, C.C. Biogenesis of extracellular vesicles (EV): Exosomes, microvesicles, retrovirus-like vesicles, and apoptotic bodies. J. Neurooncol. 2013, 113, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Bosch, B.-J.; Vlak, J.M.; van Oers, M.M.; Rottier, P.J.; van Lent, J.W.M. Budded baculovirus particle structure revisited. J. Invertebr. Pathol. 2016, 134, 15–22. [Google Scholar] [CrossRef]
- Transfiguracion, J.; Manceur, A.P.; Petiot, E.; Thompson, C.M.; Kamen, A.A. Particle quantification of influenza viruses by high performance liquid chromatography. Vaccine 2015, 33, 78–84. [Google Scholar] [CrossRef]
- Steppert, P.; Burgstaller, D.; Klausberger, M.; Tover, A.; Berger, E.; Jungbauer, A. Quantification and characterization of virus-like particles by size-exclusion chromatography and nanoparticle tracking analysis. J. Chromatogr. A 2017, 1487, 89–99. [Google Scholar] [CrossRef]
- Gutiérrez-Granados, S.; Cervera, L.; Gòdia, F.; Carrillo, J.; Segura, M.M. Development and validation of a quantitation assay for fluorescently tagged HIV-1 virus-like particles. J. Virol. Methods 2013, 193, 85–95. [Google Scholar] [CrossRef]
- Venereo-Sanchez, A.; Gilbert, R.; Simoneau, M.; Caron, A.; Chahal, P.; Chen, W.; Ansorge, S.; Li, X.; Henry, O.; Kamen, A. Hemagglutinin and neuraminidase containing virus-like particles produced in HEK-293 suspension culture: An effective influenza vaccine candidate. Vaccine 2016, 34, 3371–3380. [Google Scholar] [CrossRef] [PubMed]
- Heider, S.; Metzner, C. Quantitative real-time single particle analysis of virions. Virology 2014, 462–463, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Tang, L. Atomic cryo-EM structures of viruses. Curr. Opin. Struct. Biol. 2017, 46, 122–129. [Google Scholar] [CrossRef]
- Popov, V.L.; Tesh, R.B.; Weaver, S.C.; Vasilakis, N.; Popov, V.L.; Tesh, R.B.; Weaver, S.C.; Vasilakis, N. Electron Microscopy in Discovery of Novel and Emerging Viruses from the Collection of the World Reference Center for Emerging Viruses and Arboviruses (WRCEVA). Viruses 2019, 11, 477. [Google Scholar] [CrossRef]
- De Carlo, S.; Harris, J.R. Negative staining and cryo-negative staining of macromolecules and viruses for TEM. Micron 2011, 42, 117–131. [Google Scholar] [CrossRef]
- Kalsoom, A.; Khan, S.A.; Khan, S.B.; Asiri, A.M. Chapter 4. Scanning Electron Microscopy: Principle and Applications in Nanomaterials Characterization. In Handbook of Materials Characterization; Sharma, S.K., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 113–146. ISBN 978-3-319-92954-5. [Google Scholar]
- Gállego Ossul, I. Estructura y propiedades de las placas de cromatina de los cromosomas metafásicos: Estudio mediante técnicas de microscopía TEM, AFM y Espectroscopia de Fuerza Atómica; Universitat Autònoma de Barcelona: Bellaterra, Spain, 2010. [Google Scholar]
- Michen, B.; Graule, T. Isoelectric points of viruses. J. Appl. Microbiol. 2010, 109, 388–397. [Google Scholar] [CrossRef]
- Deregibus, M.C.; Figliolini, F.; D’antico, S.; Manzini, P.M.; Pasquino, C.; De Lena, M.; Tetta, C.; Brizzi, M.F.; Camussi, G. Charge-based precipitation of extracellular vesicles. Int. J. Mol. Med. 2016, 38, 1359–1366. [Google Scholar] [CrossRef]
- Murata, K.; Wolf, M. Cryo-electron microscopy for structural analysis of dynamic biological macromolecules. Biochim. Biophys. Acta Gen. Subj. 2018, 1862, 324–334. [Google Scholar] [CrossRef]
- Thompson, R.F.; Walker, M.; Siebert, C.A.; Muench, S.P.; Ranson, N.A. An introduction to sample preparation and imaging by cryo-electron microscopy for structural biology. Methods 2016, 100, 3–15. [Google Scholar] [CrossRef]
- Hanne, J.; Zila, V.; Heilemann, M.; Müller, B.; Kräusslich, H.-G. Super-resolved insights into human immunodeficiency virus biology. FEBS Lett. 2016, 590, 1858–1876. [Google Scholar] [CrossRef] [PubMed]
- Godin, A.G.; Lounis, B.; Cognet, L. Super-resolution Microscopy Approaches for Live Cell Imaging. Biophysj 2014, 107, 1777–1784. [Google Scholar] [CrossRef] [PubMed]
- González-Domínguez, I.; Puente-Massaguer, E.; Cervera, L.; Gòdia, F. Quantification of the HIV-1 virus-like particle production process by super-resolution imaging: From VLP budding to nanoparticle analysis. 2020. Under review. [Google Scholar]
- Puente-Massaguer, E.; Lecina, M.; Gòdia, F. Application of advanced quantification techniques in nanoparticle-based vaccine development with the Sf9 cell baculovirus expression system. Vaccine 2020, 38, 1849–1859. [Google Scholar] [CrossRef] [PubMed]
- Borlinghaus, R.T.; Kappel, C. HyVolution—The smart path to confocal super-resolution. Nat. Methods 2016, 13, i. [Google Scholar] [CrossRef]
- Zamora, J.L.R.; Aguilar, H.C. Flow virometry as a tool to study viruses. Methods 2017, 134–135, 87–97. [Google Scholar] [CrossRef]
- Maguire, C.M.; Rösslein, M.; Wick, P.; Prina-Mello, A. Characterisation of particles in solution—A perspective on light scattering and comparative technologies. Sci. Technol. Adv. Mater. 2018, 19, 732–745. [Google Scholar] [CrossRef]
- Kramberger, P.; Ciringer, M.; Štrancar, A.; Peterka, M. Evaluation of nanoparticle tracking analysis for total virus particle determination. Virol. J. 2012, 9, 265. [Google Scholar] [CrossRef]
- Aguilar, P.P.; González-Domínguez, I.; Schneider, T.A.; Gòdia, F.; Cervera, L.; Jungbauer, A. At-line multi-angle light scattering detector for faster process development in enveloped virus-like particle purification. J. Sep. Sci. 2019, 42, 2640–2648. [Google Scholar] [CrossRef]
- González-Domínguez, I.; Grimaldi, N.; Cervera, L.; Ventosa, N.; Gòdia, F. Impact of physicochemical properties of DNA/PEI complexes on transient transfection of mammalian cells. N. Biotechnol. 2019, 49, 88–97. [Google Scholar] [CrossRef]
- Nolan, J.P.; Duggan, E. Analysis of Individual Extracellular Vesicles by Flow Cytometry. In Flow Cytometry Protocols, Methods in Molecular Biology; Hawley, T.S., Hawley, R.G., Eds.; Springer Science+Business Media LLC: Berlin, Germany, 2018; Volume 1678, pp. 79–92. ISBN 978-1-4939-7346-0. [Google Scholar]
- Welsh, J.A.; Horak, P.; Wilkinson, J.S.; Ford, V.J.; Jones, J.C.; Smith, D.; Holloway, J.A.; Englyst, N.A. FCM PASS Software Aids Extracellular Vesicle Light Scatter Standardization. Cytom. Part A 2019. [Google Scholar] [CrossRef]
- McVey, M.J.; Spring, C.M.; Kuebler, W.M. Improved resolution in extracellular vesicle populations using 405 instead of 488 nm side scatter. J. Extracell. Vesicles 2018, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Charlton Hume, H.K.; Lua, L.H.L. Platform technologies for modern vaccine manufacturing. Vaccine 2017, 35, 4480–4485. [Google Scholar] [CrossRef]
- Cervera, L.; Gutiérrez-Granados, S.; Martínez, M.; Blanco, J.; Gòdia, F.; Segura, M.M. Generation of HIV-1 Gag VLPs by transient transfection of HEK 293 suspension cell cultures using an optimized animal-derived component free medium. J. Biotechnol. 2013, 166, 152–165. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Granados, S.; Cervera, L.; de L.M. Segura, M.; Wölfel, J.; Gòdia, F. Optimized production of HIV-1 virus-like particles by transient transfection in CAP-T cells. Appl. Microbiol. Biotechnol. 2016, 100, 3935–3947. [Google Scholar] [CrossRef] [PubMed]
- Göttlinger, H.G. HIV-1 Gag: A Molecular Machine Driving Viral Particle Assembly and Release. In HIV Sequence Compendium 2011; Kuiken, C., Foley, B., Leitner, T., Apetrei, C., Hahn, B., Mizrachi, I., Mullins, J., Rambaut, A., Wolinsky, S., Korber, B., Eds.; Los Alamos National Laboratory, Theoretical Biology and Biophysics: Los Alamos, NM, USA, 2011; pp. 2–28, LA-UR-11-11440. [Google Scholar]
- Thompson, C.M.; Petiot, E.; Mullick, A.; Aucoin, M.G.; Henry, O.; Kamen, A.A. Critical assessment of influenza VLP production in Sf9 and HEK293 expression systems. BMC Biotechnol. 2015, 15, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Genzel, Y. Designing cell lines for viral vaccine production: Where do we stand? Biotechnol. J. 2015, 10, 728–740. [Google Scholar] [CrossRef]
- Puente-Massaguer, E.; Lecina, M.; Gòdia, F. Integrating nanoparticle quantification and statistical design of experiments for efficient HIV-1 virus-like particle production in High Five cells. Appl. Microbiol. Biotechnol. 2020, 104, 1569–1582. [Google Scholar] [CrossRef]
- De Rond, L.; Coumans, F.A.W.; Nieuwland, R.; van Leeuwen, T.G.; van der Pol, E. Deriving Extracellular Vesicle Size From Scatter Intensities Measured by Flow Cytometry. Curr. Protoc. Cytom. 2018, 86, 1–14. [Google Scholar] [CrossRef]
- Malvern Instruments Limited. Determining Fluorescence Limit of Detection with Nanoparticle Tracking Analysis (NTA). 2015. Available online: https://www.malvernpanalytical.com/en/learn/knowledge-center/application-notes/AN150507FluorescenceLOD (accessed on 17 February 2020).
- Ellis, E.A. Staining Sectioned Biological Specimens for Transmission Electron Microscopy: Conventional and En Bloc Stains. In Electron Microscopy: Methods and Protocols, Methods in Molecular Biology; Kuo, J., Ed.; Humana Press: Totowa, NJ, USA, 2014; pp. 57–72. ISBN 978-1-62703-776-1. [Google Scholar]
- Cervera, L.; Fuenmayor, J.; González-Domínguez, I.; Gutiérrez-Granados, S.; Segura, M.M.; Gòdia, F. Selection and optimization of transfection enhancer additives for increased virus-like particle production in HEK293 suspension cell cultures. Appl. Microbiol. Biotechnol. 2015, 99, 9935–9949. [Google Scholar] [CrossRef]
- González-Domínguez, I.; Gutiérrez-Granados, S.; Cervera, L.; Gòdia, F.; Domingo, N. Identification of HIV-1–Based Virus-like Particles by Multifrequency Atomic Force Microscopy. Biophys. J. 2016, 111, 1173–1179. [Google Scholar] [CrossRef]
- Puente-Massaguer, E.; González-Domínguez, I. (Universitat Autònoma de Barcelona, Barcelona, Spain). Preliminary experiments on HIV-1 Gag-eGFP VLP characterization by Scanning Electron Microscopy. Personal communication, 2020. [Google Scholar]
- Gencer, D.; Bayramoglu, Z.; Nalcacioglu, R.; Kleespies, R.G.; Demirbag, Z.; Demir, I. Characterisation of three Alphabaculovirus isolates from the gypsy moth, Lymantria dispar dispar (Lepidoptera: Erebidae), in Turkey. Biocontrol Sci. Technol. 2018, 28, 107–121. [Google Scholar] [CrossRef]
- Palomares, L.A.; Realpe, M.; Ramírez, O.T. An Overview of Cell Culture Engineering for the Insect Cell-Baculovirus Expression Vector System (BEVS). In Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2015; Volume 5, pp. 501–519. ISBN 978-3-319-10319-8. [Google Scholar]
- Thermo Fisher Scientific. ExpiSfTM Expression System. 2020. Available online: https://assets.thermofisher.com/TFSAssets/LSG/manuals/MAN0017532_ExpiSfExpressionSystem_UG.pdf (accessed on 17 February 2020).
- Golding, C.G.; Lamboo, L.L.; Beniac, D.R.; Booth, T.F. The scanning electron microscope in microbiology and diagnosis of infectious disease. Sci. Rep. 2016, 6, 26516. [Google Scholar] [CrossRef] [PubMed]
- Van Der Pol, E.; Hoekstra, A.G.; Sturk, A.; Otto, C.; Van Leeuwen, T.G.; Nieuwland, R. Optical and non-optical methods for detection and characterization of microparticles and exosomes. J. Thromb. Haemost. 2010, 8, 2596–2607. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, M.; Jamali, S.; Saha, I.; Daum, R.; Bendjennat, M.; Saffarian, S. Correlative iPALM and SEM resolves virus cavity and Gag lattice defects in HIV virions. Eur. Biophys. J. 2019, 48, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Welsh, J.A.; Holloway, J.A.; Wilkinson, J.S.; Englyst, N.A. Extracellular Vesicle Flow Cytometry Analysis and Standardization. Front. Cell Dev. Biol. 2017, 5, 1–7. [Google Scholar] [CrossRef]
- Yang, Z.; Sharma, A.; Qi, J.; Peng, X.; Lee, D.Y.; Hu, R.; Lin, D.; Qu, J.; Kim, J.S. Super-resolution fluorescent materials: An insight into design and bioimaging applications. Chem. Soc. Rev. 2016, 45, 4651–4667. [Google Scholar] [CrossRef]
- Rossi, C.; Kearney, B.; Olschner, S.; Williams, P.; Robinson, C.; Heinrich, M.; Zovanyi, A.; Ingram, M.; Norwood, D.; Schoepp, R. Evaluation of ViroCyt® Virus Counter for Rapid Filovirus Quantitation. Viruses 2015, 7, 857–872. [Google Scholar] [CrossRef]
- Blancett, C.D.; Fetterer, D.P.; Koistinen, K.A.; Morazzani, E.M.; Monninger, M.K.; Piper, A.E.; Kuehl, K.A.; Kearney, B.J.; Norris, S.L.; Rossi, C.A.; et al. Accurate virus quantitation using a Scanning Transmission Electron Microscopy (STEM) detector in a scanning electron microscope. J. Virol. Methods 2017, 248, 136–144. [Google Scholar] [CrossRef]
- Strauss, J.D.; Hammonds, J.E.; Yi, H.; Ding, L.; Spearman, P.; Wright, E.R. Three-Dimensional Structural Characterization of HIV-1 Tethered to Human Cells. J. Virol. 2016, 90, 1507–1521. [Google Scholar] [CrossRef]
- Pornillos, O.; Higginson, D.S.; Stray, K.M.; Fisher, R.D.; Garrus, J.E.; Payne, M.; He, G.P.; Wang, H.E.; Morham, S.G.; Sundquist, W.I. HIV Gag mimics the Tsg101-recruiting activity of the human Hrs protein. J. Cell Biol. 2003, 162, 425–434. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, B.; Musier-Forsyth, K.; Mansky, L.M.; Mueller, J.D. Fluorescence fluctuation spectroscopy on viral-like particles reveals variable Gag stoichiometry. Biophys. J. 2009, 96, 1961–1969. [Google Scholar] [CrossRef] [PubMed]
- Inamdar, K.; Floderer, C.; Favard, C.; Muriaux, D. Monitoring HIV-1 Assembly in Living Cells: Insights from Dynamic and Single Molecule Microscopy. Viruses 2019, 11, 72. [Google Scholar] [CrossRef] [PubMed]
- Steppert, P.; Burgstaller, D.; Klausberger, M.; Berger, E.; Aguilar, P.P.; Schneider, T.A.; Kramberger, P.; Tover, A.; Nöbauer, K.; Razzazi-Fazeli, E.; et al. Purification of HIV-1 gag virus-like particles and separation of other extracellular particles. J. Chromatogr. A 2016, 1455, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Xiao, P.J.; Li, C.; Neumann, A.; Samulski, R.J. Quantitative 3D tracing of gene-delivery viral vectors in human cells and animal tissues. Mol. Ther. 2012, 20, 317–328. [Google Scholar] [CrossRef]
- Sarder, P.; Nehorai, A. Deconvolution methods for 3-D fluorescence microscopy images. IEEE Signal Process. Mag. 2006, 23, 32–45. [Google Scholar] [CrossRef]
- Schermelleh, L.; Ferrand, A.; Huser, T.; Eggeling, C.; Sauer, M.; Biehlmaier, O.; Drummen, G.P.C. Super-resolution microscopy demystified. Nat. Cell Biol. 2019, 21, 72–84. [Google Scholar] [CrossRef]


| VLPs | Total Nanoparticles | |||
|---|---|---|---|---|
| Camera Level | Threshold | Camera Level | Threshold | |
| HEK 293 supernatants | 16 | 4 | 10 | 4 |
| Sf9 supernatants | 16 | 3 | 8 | 3 |
| HEK 293 conditioned medium | - | - | 10 | 4 |
| Sf9 conditioned medium | - | - | 14 | 3 |
| FreeStyle culture medium | - | - | 13 | 5 |
| Sf900III culture medium | - | - | 13 | 4 |
| Technique | VLPs (109/mL) | EVs (109/mL) | |||
|---|---|---|---|---|---|
| FV | NTA | SRFM | FV | NTA | |
| HEK 293 | 0.2 ± 0.0 | 5.0 ± 0.5 * | 4.0–43.0 * | 0.2 ± 0.0 | 16.6 ± 2.9 * |
| Sf9 | 0.2 ± 0.0 | 8.6 ± 2.1 * | 6.6–74.5 * | 0.3 ± 0.0 | 9.7 ± 1.6 * |
| Fold difference (Sf9:HEK 293) | 1.5 | 1.7 | 1.7 | 1.2 | 0.6 |
| Parameter | TEM | SEM | Cryo-TEM | HyVolution2 SRFM | NTA | Flow Virometry |
|---|---|---|---|---|---|---|
| Expertise | +++ | +++ | +++++ | +++ | ++ | ++ |
| Time of measurement | ++++ | ++++ | +++++ | +++ | ++ | + |
| Resolution | Nanometric (<10 nm) [14] | Nanometric (<10 nm) [48] | Atomic [13] | Nanometric (140 nm) [26] | Nanometric (30 nm) [29] | Nanometric (100–200 nm) [5] |
| Pros | Widely used, ultrastructural analysis | Surface analysis of nanoparticle populations | Ultrastructural analysis in its native form | Direct visualization, compatible with fluorescence | Easy handling, compatible with fluorescence | Simultaneous analysis of light scattering and fluorescence labelling |
| Cons | Staining required, working under vacuum conditions | Working under vacuum conditions | Time consuming, working at cryogenic temperatures | Data analysis, convolution effect of light | Screening effect, variability due to acquisition settings | Sensitivity, inter-equipment variability |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Domínguez, I.; Puente-Massaguer, E.; Cervera, L.; Gòdia, F. Quality Assessment of Virus-Like Particles at Single Particle Level: A Comparative Study. Viruses 2020, 12, 223. https://doi.org/10.3390/v12020223
González-Domínguez I, Puente-Massaguer E, Cervera L, Gòdia F. Quality Assessment of Virus-Like Particles at Single Particle Level: A Comparative Study. Viruses. 2020; 12(2):223. https://doi.org/10.3390/v12020223
Chicago/Turabian StyleGonzález-Domínguez, Irene, Eduard Puente-Massaguer, Laura Cervera, and Francesc Gòdia. 2020. "Quality Assessment of Virus-Like Particles at Single Particle Level: A Comparative Study" Viruses 12, no. 2: 223. https://doi.org/10.3390/v12020223
APA StyleGonzález-Domínguez, I., Puente-Massaguer, E., Cervera, L., & Gòdia, F. (2020). Quality Assessment of Virus-Like Particles at Single Particle Level: A Comparative Study. Viruses, 12(2), 223. https://doi.org/10.3390/v12020223






