House Dust Mite Exposure Causes Increased Susceptibility of Nasal Epithelial Cells to Adenovirus Infection
Abstract
1. Introduction
2. Materials and Methods
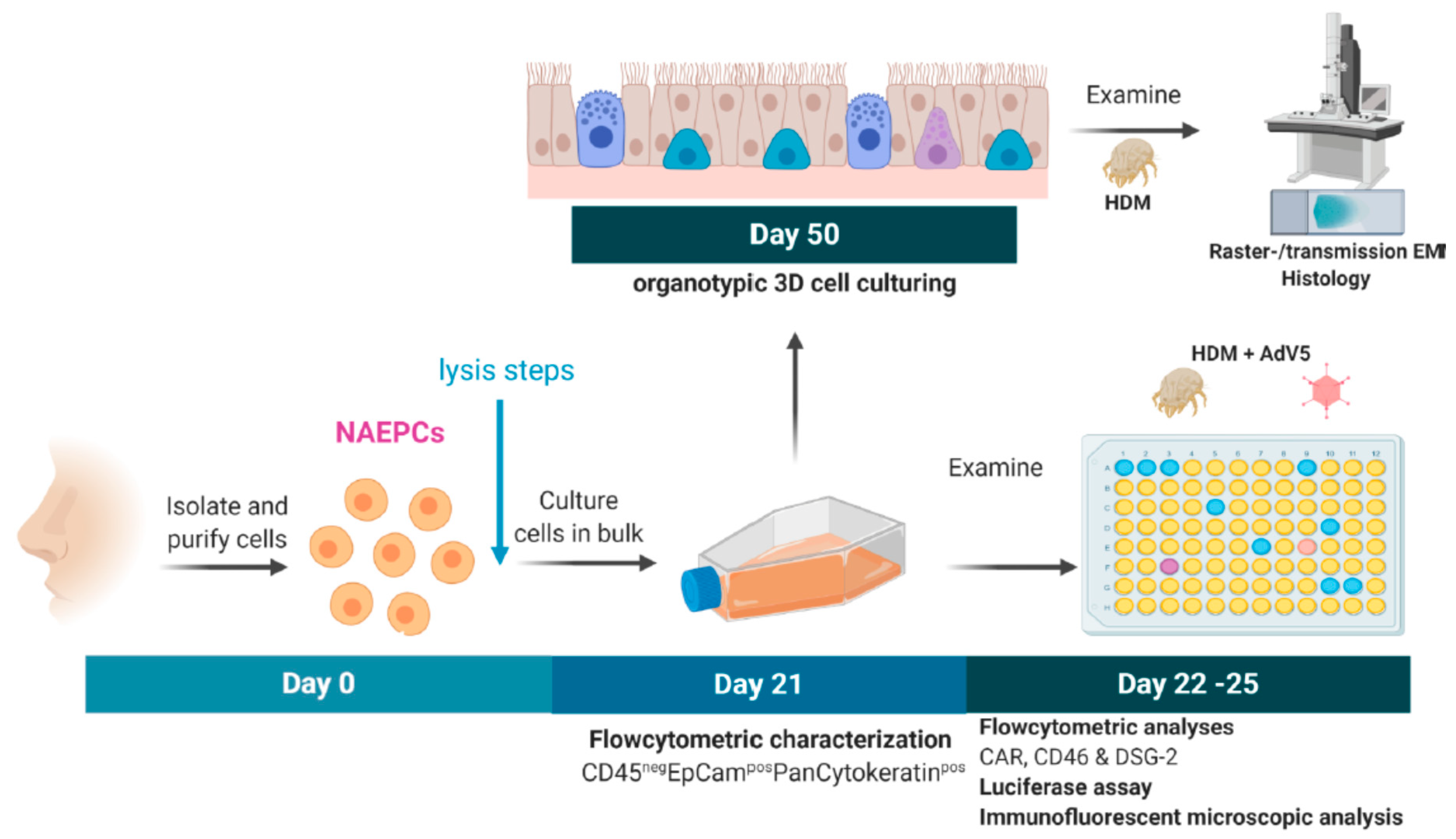
2.1. Subjects and Study Design
2.2. Technical Information
2.2.1. Culturing of NAEPCs in 2D and 3D Models
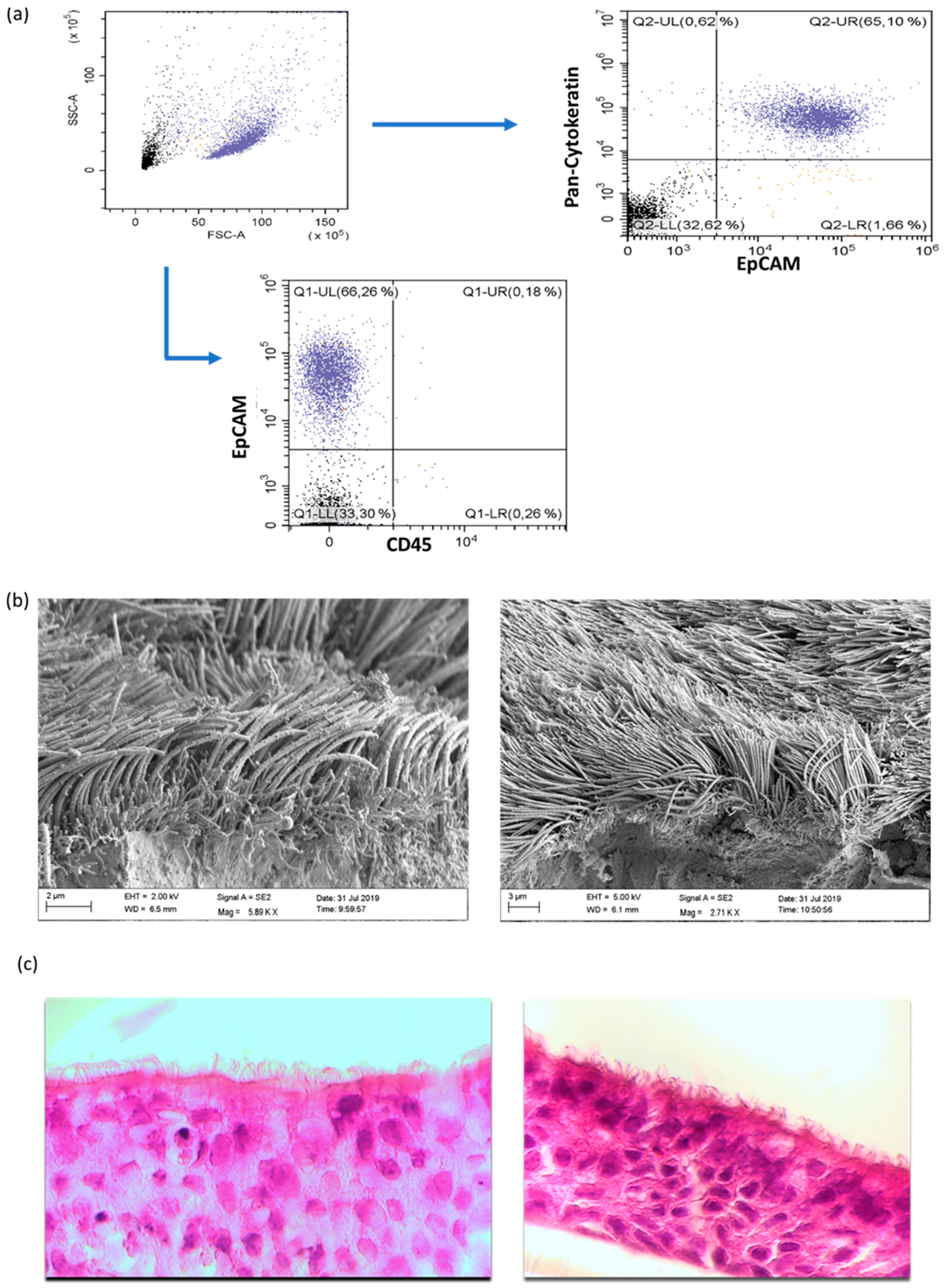
2.2.2. Flow Cytometric Characterization of NAEPCs and Adenoviral Receptors
2.2.3. Scanning Electron Microscopic Imaging of Organotypic 3D NAEPCs Cultures
2.2.4. Transmission Electron Microscopic (TEM) Imaging of Organotypic 3D NAEPCs Cultures
2.2.5. House Dust Mite Induced Exposition In Vitro
2.2.6. Vector Production and Titration
2.3. Statistical Analysis
3. Results
3.1. The Characterization of NAEPCs
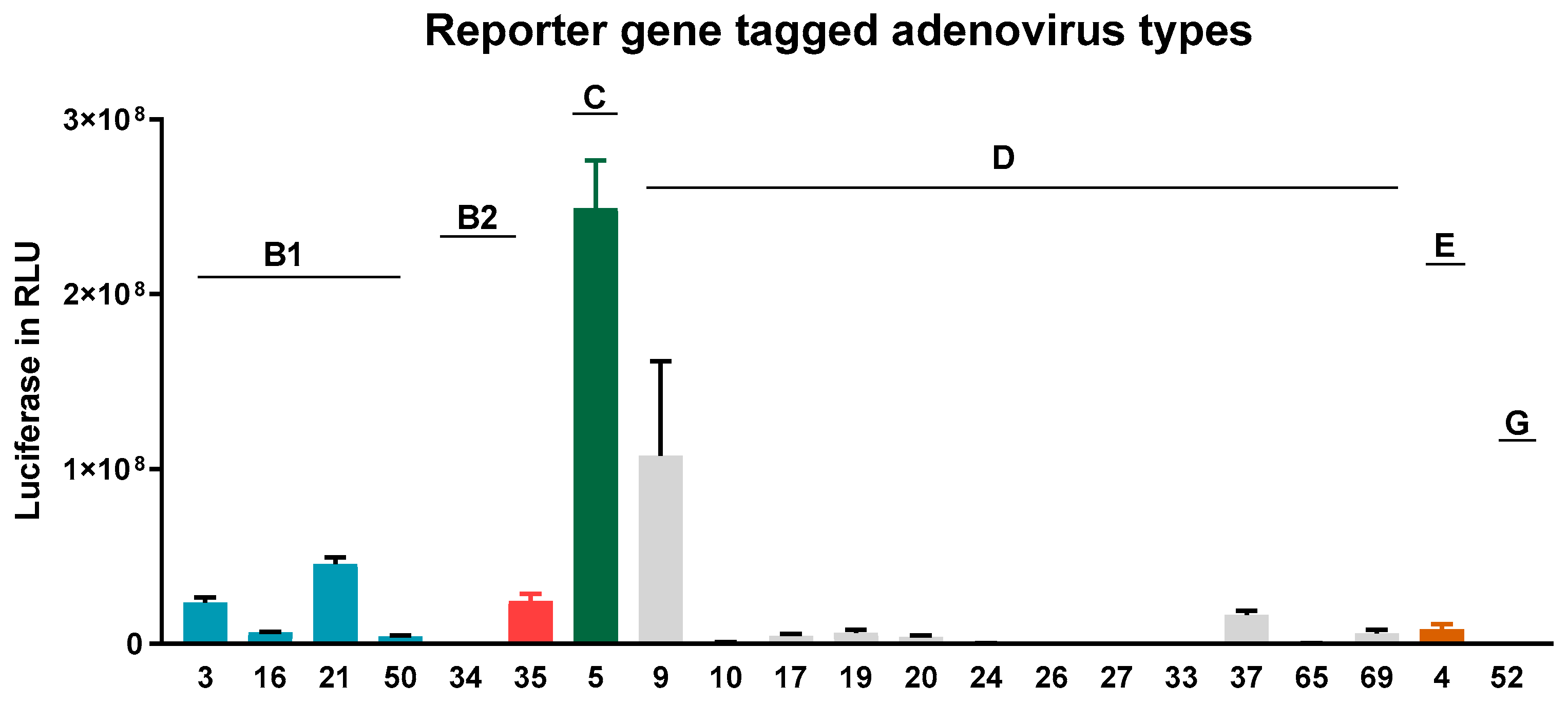
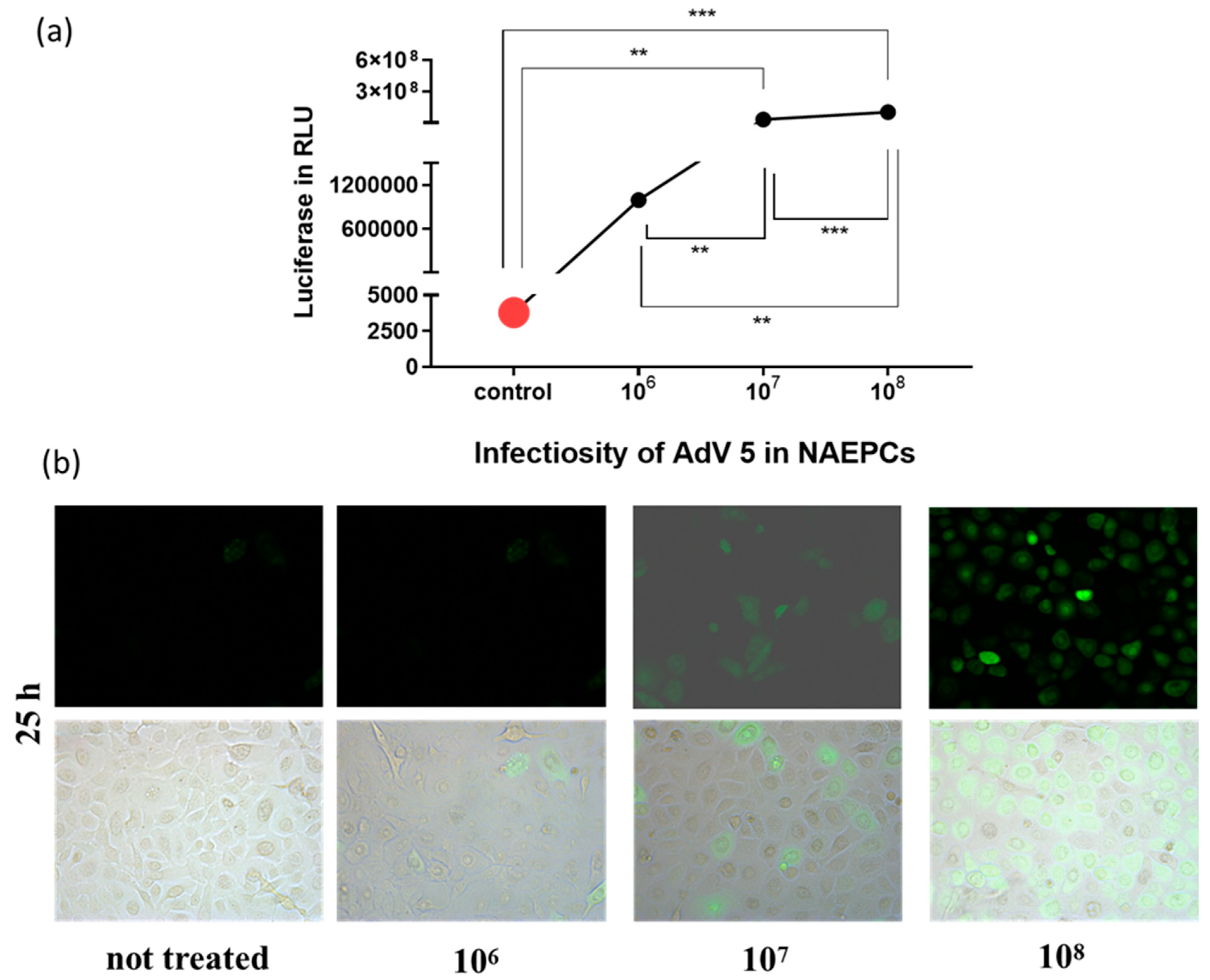
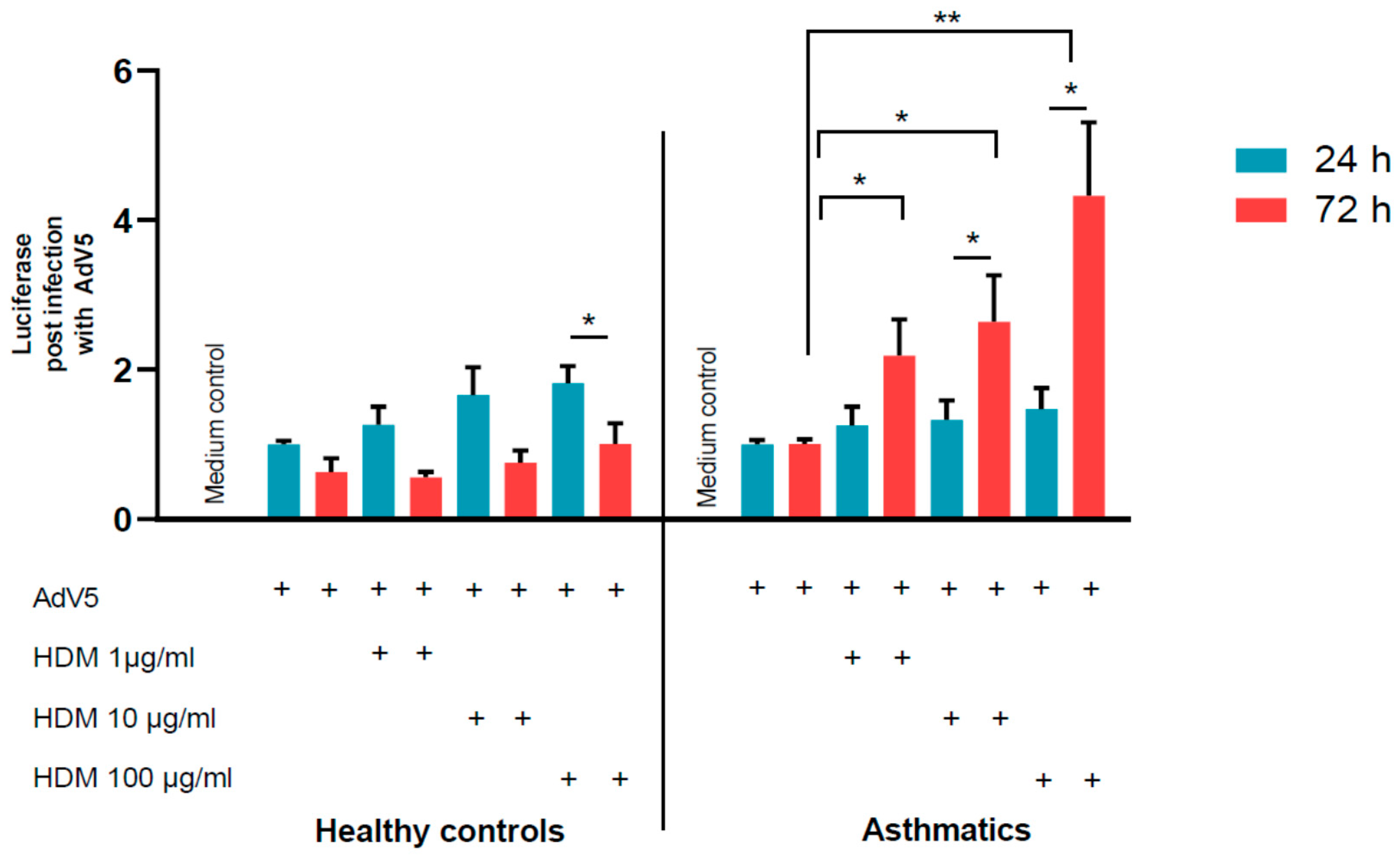
3.2. Adenovirus Transduction Rates of NAEPC
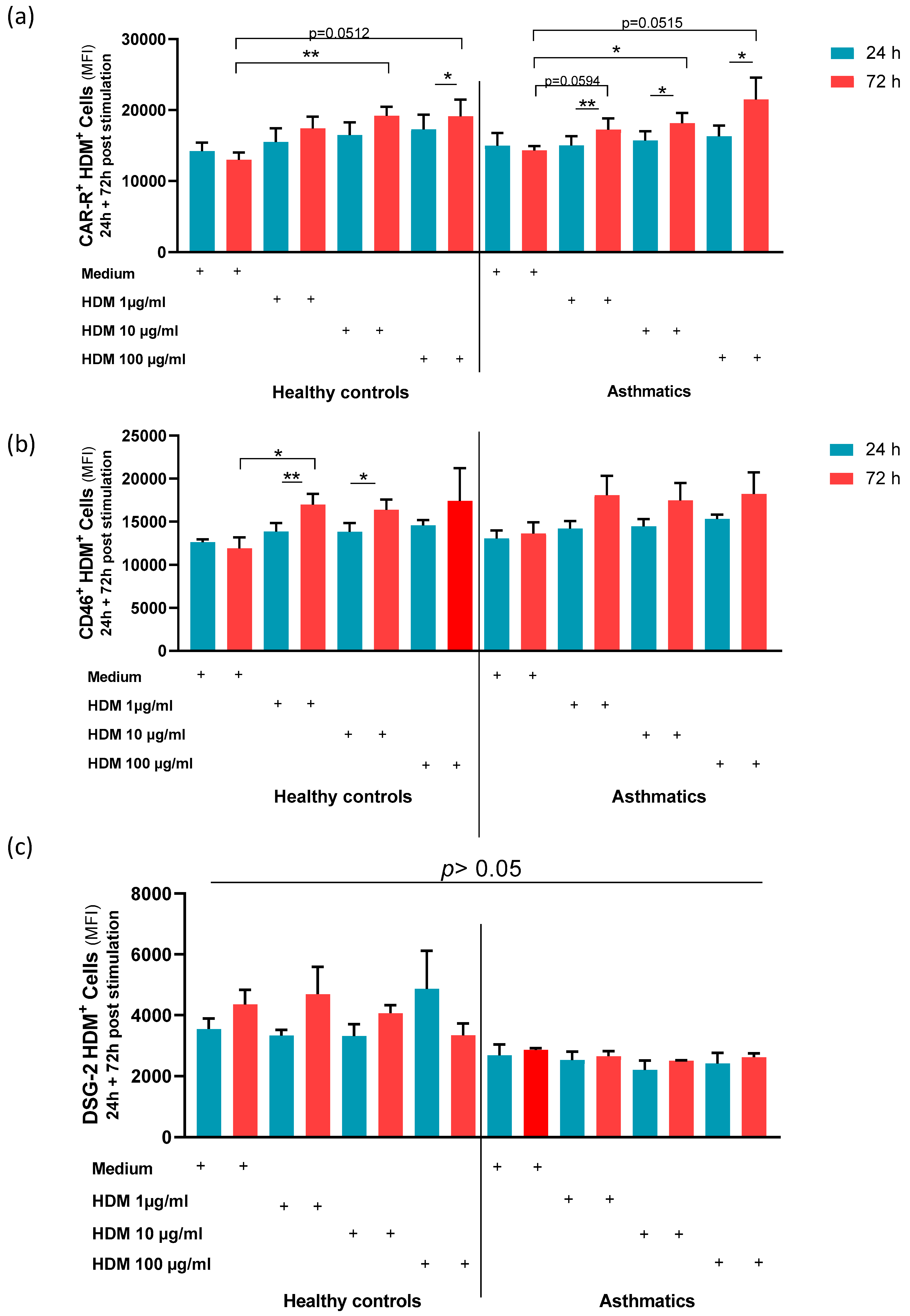
3.3. Stimulation with HDM Increased the Expression of AdV Specific Receptors
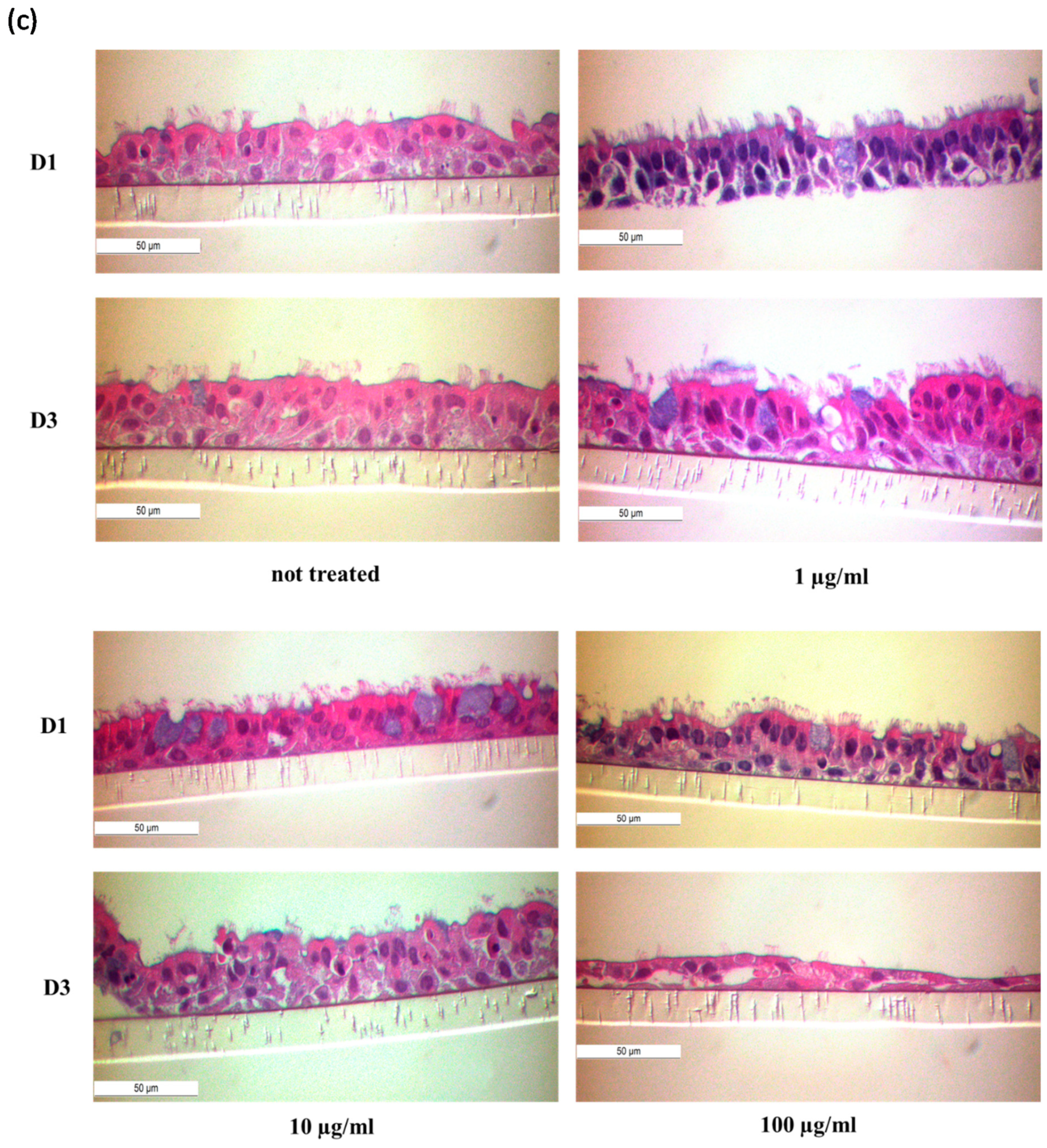
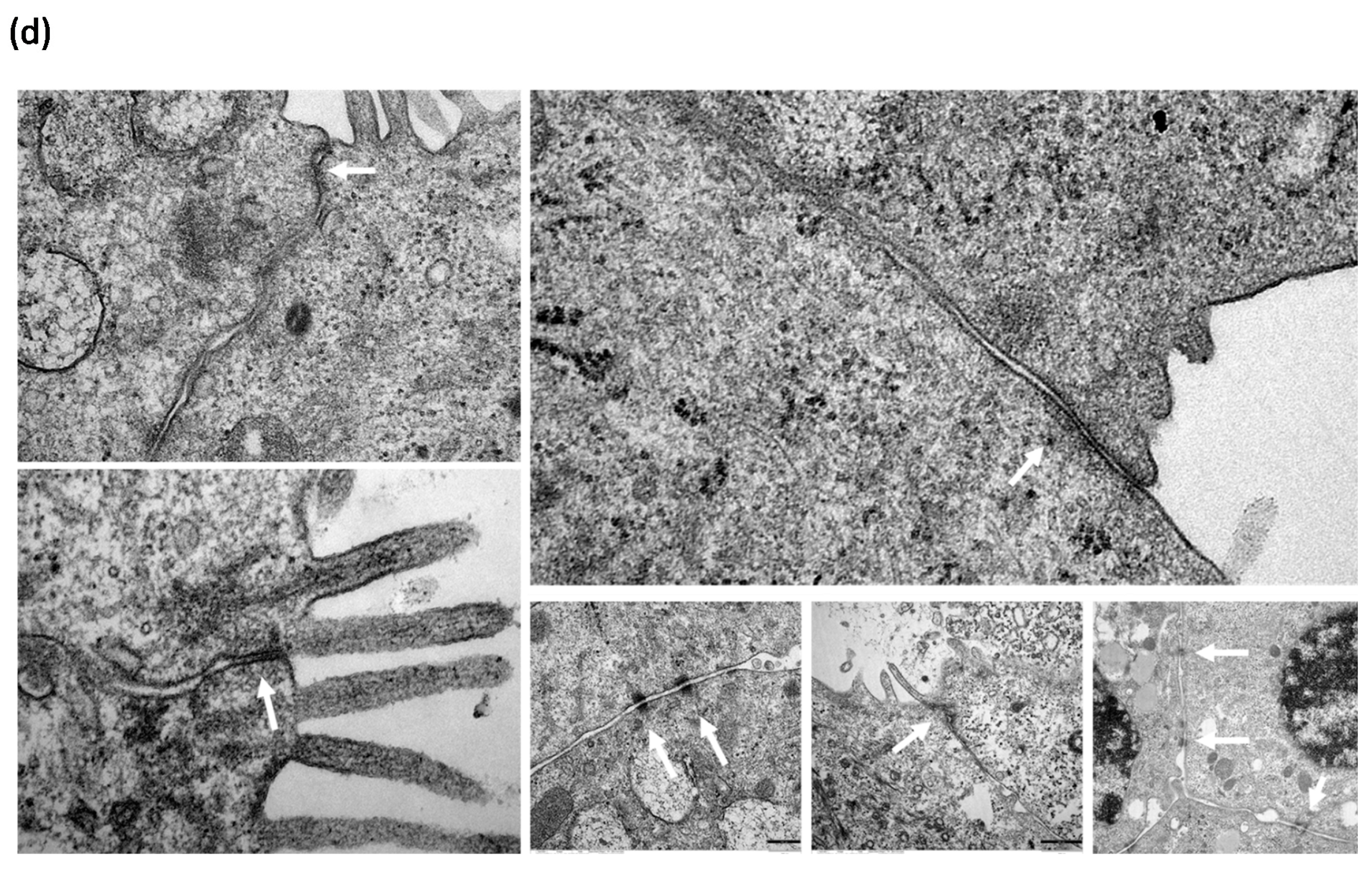
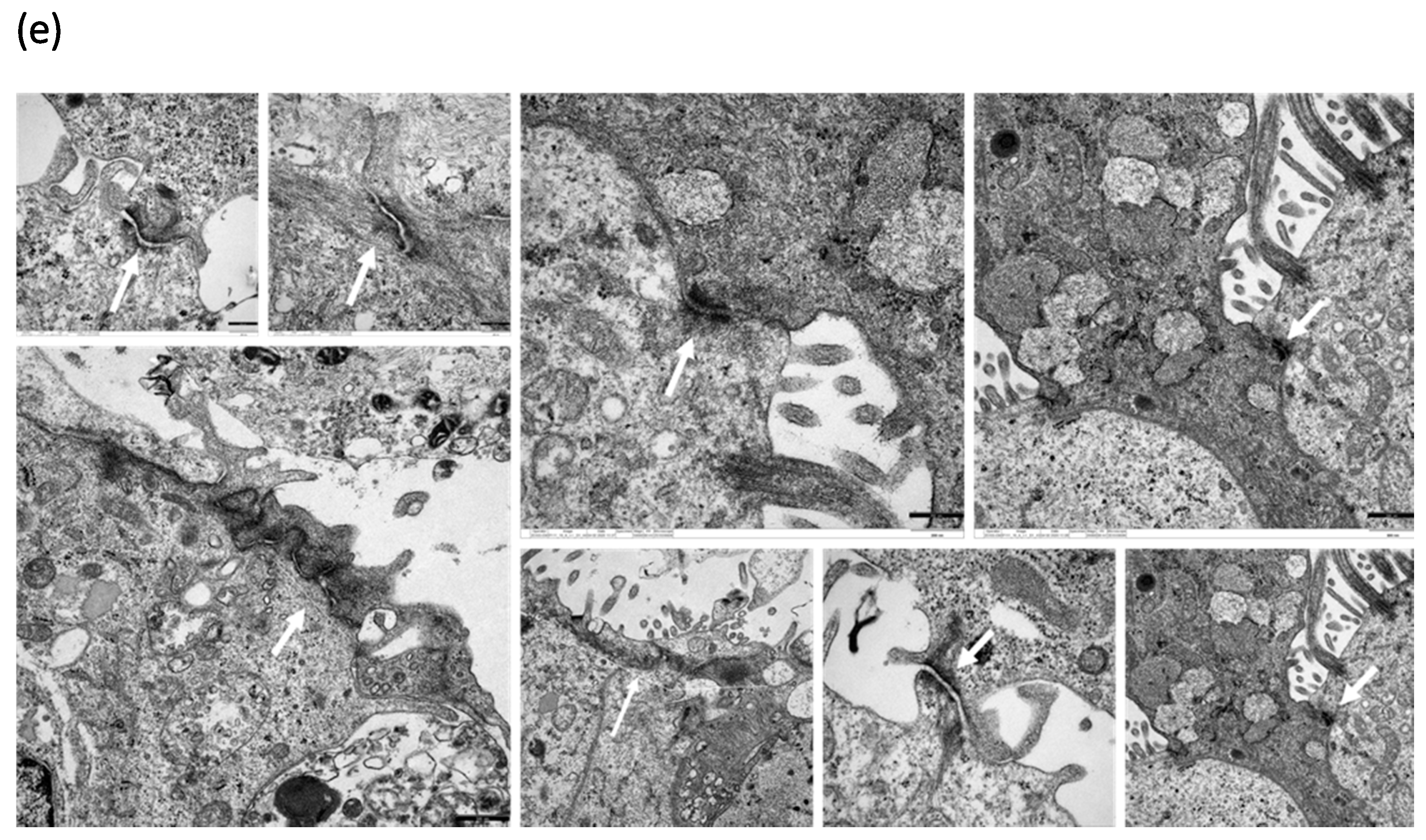
3.4. HDM Increased the Permeability between Cell–Cell Contacts through Affecting the Epithelial Barrier Integrity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Robinson, C.M.; Singh, G.; Lee, J.Y.; Dehghan, S.; Rajaiya, J.; Liu, E.B.; Yousuf, M.A.; Betensky, R.A.; Jones, M.S.; Dyer, D.W.; et al. Molecular evolution of human adenoviruses. Sci. Rep. 2013, 3, 1812. [Google Scholar] [CrossRef] [PubMed]
- Nemerow, G.; Flint, J. Lessons learned from adenovirus (1970–2019). FEBS Lett. 2019, 593, 3395–3418. [Google Scholar] [CrossRef] [PubMed]
- Davison, A.J.; Benko, M.; Harrach, B. Genetic content and evolution of adenoviruses. J. Gen. Virol. 2003, 84, 2895–2908. [Google Scholar] [CrossRef] [PubMed]
- Ghebremedhin, B. Human adenovirus: Viral pathogen with increasing importance. Eur. J. Microbiol. Immunol. 2014, 4, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Woo, Y.Y.; de Bruyne, J.A.; Nathan, A.M.; Kee, S.Y.; Chan, Y.F.; Chiam, C.W.; Eg, K.P.; Thavagnanam, S.; Sam, I.C. Epidemiology, clinical presentation and respiratory sequelae of adenovirus pneumonia in children in Kuala Lumpur, Malaysia. PLoS ONE 2018, 13, e0205795. [Google Scholar] [CrossRef] [PubMed]
- Ismail, A.M.; Zhou, X.; Dyer, D.W.; Seto, D.; Rajaiya, J.; Chodosh, J. Genomic foundations of evolution and ocular pathogenesis in human adenovirus species D. FEBS Lett. 2019, 593, 3583–3608. [Google Scholar] [CrossRef]
- Sharma, M.; Mishra, B.; Saikia, U.N.; Bahl, A.; Ratho, R.K.; Talwar, K.K. Role of coxsackievirus and adenovirus receptor (CAR) expression and viral load of adenovirus and enterovirus in patients with dilated cardiomyopathy. Arch. Virol. 2016, 161, 87–94. [Google Scholar] [CrossRef]
- Gu, J.; Su, Q.Q.; Zuo, T.T.; Chen, Y.B. Adenovirus diseases: A systematic review and meta-analysis of 228 case reports. Infection 2020. [Google Scholar] [CrossRef]
- Potter, R.N.; Cantrell, J.A.; Mallak, C.T.; Gaydos, J.C. Adenovirus-associated deaths in US military during postvaccination period, 1999–2010. Emerg. Infect. Dis. 2012, 18, 507–509. [Google Scholar] [CrossRef]
- Zheng, X.Y.; Xu, Y.J.; Guan, W.J.; Lin, L.F. Regional, age and respiratory-secretion-specific prevalence of respiratory viruses associated with asthma exacerbation: A literature review. Arch. Virol. 2018, 163, 845–853. [Google Scholar] [CrossRef]
- Pech, M.; Weckmann, M.; Konig, I.R.; Franke, A.; Heinsen, F.A.; Oliver, B.; Ricklefs, I.; Fuchs, O.; Rabe, K.; Hansen, G.E.; et al. Rhinovirus infections change DNA methylation and mRNA expression in children with asthma. PLoS ONE 2018, 13, e0205275. [Google Scholar] [CrossRef] [PubMed]
- Akbarshahi, H.; Menzel, M.; Ramu, S.; Mahmutovic Persson, I.; Bjermer, L.; Uller, L. House dust mite impairs antiviral response in asthma exacerbation models through its effects on TLR3. Allergy 2018, 73, 1053–1063. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.M.; Platts-Mills, T.A.E. Home Environmental Interventions for House Dust Mite. J. Allergy Clin. Immunol. Pract. 2018, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Xepapadaki, P.; Papadopoulos, N.G. Childhood asthma and infection: Virus-induced exacerbations as determinants and modifiers. Eur. Respir. J. 2010, 36, 438–445. [Google Scholar] [CrossRef]
- Papadopoulos, N.G.; Christodoulou, I.; Rohde, G.; Agache, I.; Almqvist, C.; Bruno, A.; Bonini, S.; Bont, L.; Bossios, A.; Bousquet, J.; et al. Viruses and bacteria in acute asthma exacerbations—A GA(2) LEN-DARE systematic review. Allergy 2011, 66, 458–468. [Google Scholar] [CrossRef]
- Vroling, A.B.; Jonker, M.J.; Breit, T.M.; Fokkens, W.J.; van Drunen, C.M. Comparison of expression profiles induced by dust mite in airway epithelia reveals a common pathway. Allergy 2008, 63, 461–467. [Google Scholar] [CrossRef]
- Vroling, A.B.; Jonker, M.J.; Luiten, S.; Breit, T.M.; Fokkens, W.J.; van Drunen, C.M. Primary nasal epithelium exposed to house dust mite extract shows activated expression in allergic individuals. Am. J. Respir. Cell. Mol. Biol. 2008, 38, 293–299. [Google Scholar] [CrossRef]
- Lambrecht, B.N.; Hammad, H. The immunology of asthma. Nat. Immunol. 2015, 16, 45–56. [Google Scholar] [CrossRef]
- Zhang, W.; Fu, J.; Liu, J.; Wang, H.; Schiwon, M.; Janz, S.; Schaffarczyk, L.; von der Goltz, L.; Ehrke-Schulz, E.; Dorner, J.; et al. An Engineered Virus Library as a Resource for the Spectrum-wide Exploration of Virus and Vector Diversity. Cell Rep. 2017, 19, 1698–1709. [Google Scholar] [CrossRef]
- Maestre-Batlle, D.; Pena, O.M.; Hirota, J.A.; Gunawan, E.; Rider, C.F.; Sutherland, D.; Alexis, N.E.; Carlsten, C. Novel flow cytometry approach to identify bronchial epithelial cells from healthy human airways. Sci. Rep. 2017, 7, 42214. [Google Scholar] [CrossRef]
- Hampel, U.; Schroder, A.; Mitchell, T.; Brown, S.; Snikeris, P.; Garreis, F.; Kunnen, C.; Willcox, M.; Paulsen, F. Serum-induced keratinization processes in an immortalized human meibomian gland epithelial cell line. PLoS ONE 2015, 10, e0128096. [Google Scholar] [CrossRef] [PubMed]
- James, K.M.; Peebles, R.S., Jr.; Hartert, T.V. Response to infections in patients with asthma and atopic disease: An epiphenomenon or reflection of host susceptibility? J. Allergy Clin. Immunol. 2012, 130, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Bossios, A.; Gourgiotis, D.; Skevaki, C.L.; Saxoni-Papageorgiou, P.; Lotvall, J.; Psarras, S.; Karpathios, T.; Constandopoulos, A.G.; Johnston, S.L.; Papadopoulos, N.G. Rhinovirus infection and house dust mite exposure synergize in inducing bronchial epithelial cell interleukin-8 release. Clin. Exp. Allergy 2008, 38, 1615–1626. [Google Scholar] [CrossRef] [PubMed]
- Golebski, K.; Luiten, S.; van Egmond, D.; de Groot, E.; Roschmann, K.I.; Fokkens, W.J.; van Drunen, C.M. High degree of overlap between responses to a virus and to the house dust mite allergen in airway epithelial cells. PLoS ONE 2014, 9, e87768. [Google Scholar] [CrossRef]
- Sharma, A.; Xu, Y.; Sung, B.; Vincent, C.T.; Worgall, T.; Worgall, S. Regulation of the Coxsackie and adenovirus receptor expression is dependent on cystic fibrosis transmembrane regulator in airway epithelial cells. Cell Microbiol. 2017, 19, e12654. [Google Scholar] [CrossRef]
- Tatsuta, M.; Kan, O.K.; Ishii, Y.; Yamamoto, N.; Ogawa, T.; Fukuyama, S.; Ogawa, A.; Fujita, A.; Nakanishi, Y.; Matsumoto, K. Effects of cigarette smoke on barrier function and tight junction proteins in the bronchial epithelium: Protective role of cathelicidin LL-37. Respir. Res. 2019, 20, 251. [Google Scholar] [CrossRef]
- Excoffon, K.J.; Gansemer, N.D.; Mobily, M.E.; Karp, P.H.; Parekh, K.R.; Zabner, J. Isoform-specific regulation and localization of the coxsackie and adenovirus receptor in human airway epithelia. PLoS ONE 2010, 5, e9909. [Google Scholar] [CrossRef]
- Tsai, Y.G.; Wen, Y.S.; Wang, J.Y.; Yang, K.D.; Sun, H.L.; Liou, J.H.; Lin, C.Y. Complement regulatory protein CD46 induces autophagy against oxidative stress-mediated apoptosis in normal and asthmatic airway epithelium. Sci. Rep. 2018, 8, 12973. [Google Scholar] [CrossRef]
- Thavagnanam, S.; Parker, J.C.; McBrien, M.E.; Skibinski, G.; Shields, M.D.; Heaney, L.G. Nasal epithelial cells can act as a physiological surrogate for paediatric asthma studies. PLoS ONE 2014, 9, e85802. [Google Scholar] [CrossRef]
- Vanders, R.L.; Hsu, A.; Gibson, P.G.; Murphy, V.E.; Wark, P.A.B. Nasal epithelial cells to assess in vitro immune responses to respiratory virus infection in pregnant women with asthma. Respir. Res. 2019, 20, 259. [Google Scholar] [CrossRef]
- Cardenas, A.; Sordillo, J.E.; Rifas-Shiman, S.L.; Chung, W.; Liang, L.; Coull, B.A.; Hivert, M.F.; Lai, P.S.; Forno, E.; Celedon, J.C.; et al. The nasal methylome as a biomarker of asthma and airway inflammation in children. Nat. Commun. 2019, 10, 3095. [Google Scholar] [CrossRef] [PubMed]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aydin, M.; Naumova, E.A.; Paulsen, F.; Zhang, W.; Gopon, F.; Theis, C.; Lutz, S.; Ehrke-Schulz, E.; Arnold, W.H.; Wirth, S.; et al. House Dust Mite Exposure Causes Increased Susceptibility of Nasal Epithelial Cells to Adenovirus Infection. Viruses 2020, 12, 1151. https://doi.org/10.3390/v12101151
Aydin M, Naumova EA, Paulsen F, Zhang W, Gopon F, Theis C, Lutz S, Ehrke-Schulz E, Arnold WH, Wirth S, et al. House Dust Mite Exposure Causes Increased Susceptibility of Nasal Epithelial Cells to Adenovirus Infection. Viruses. 2020; 12(10):1151. https://doi.org/10.3390/v12101151
Chicago/Turabian StyleAydin, Malik, Ella A. Naumova, Friedrich Paulsen, Wenli Zhang, Felix Gopon, Christian Theis, Sören Lutz, Eric Ehrke-Schulz, Wolfgang H. Arnold, Stefan Wirth, and et al. 2020. "House Dust Mite Exposure Causes Increased Susceptibility of Nasal Epithelial Cells to Adenovirus Infection" Viruses 12, no. 10: 1151. https://doi.org/10.3390/v12101151
APA StyleAydin, M., Naumova, E. A., Paulsen, F., Zhang, W., Gopon, F., Theis, C., Lutz, S., Ehrke-Schulz, E., Arnold, W. H., Wirth, S., & Ehrhardt, A. (2020). House Dust Mite Exposure Causes Increased Susceptibility of Nasal Epithelial Cells to Adenovirus Infection. Viruses, 12(10), 1151. https://doi.org/10.3390/v12101151






