Effect of Benzothiadiazole on the Metabolome of Tomato Plants Infected by Citrus Exocortis Viroid
Abstract
1. Introduction
2. Materials and Methods
2.1. Tomato Plants and Growth Conditions
2.2. CEVd Infection Procedure
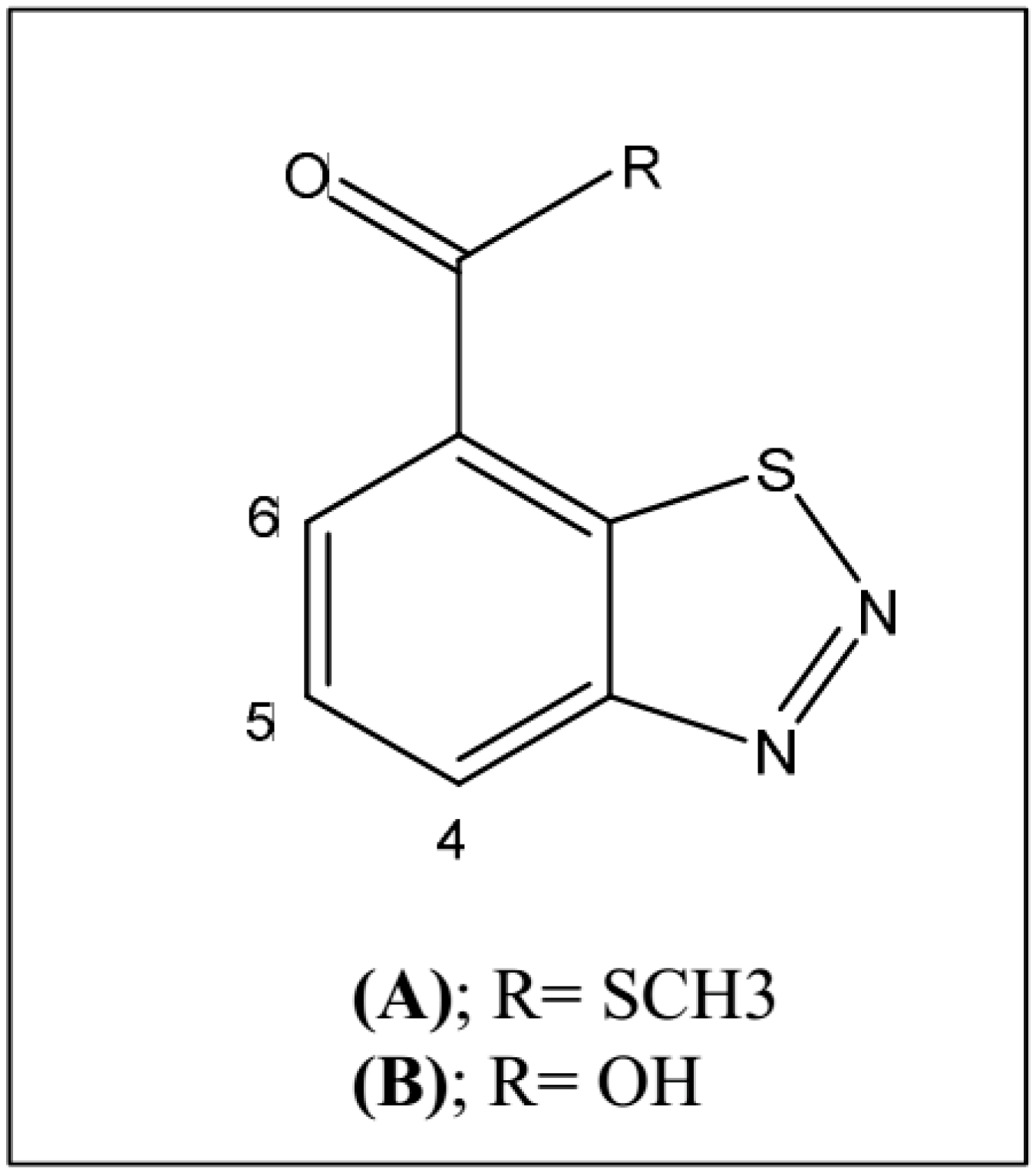
2.3. Treatments with BTH and GABA
2.4. Extraction of Metabolites and NMR Spectra Measurements
2.5. RNA Extraction and Preparation
2.6. Quantitative Real-Time Polymerase Chain Reaction (PCR) Assay
2.7. GC-MS Analysis for Primary Metabolite
2.8. Statistical Analysis
3. Results
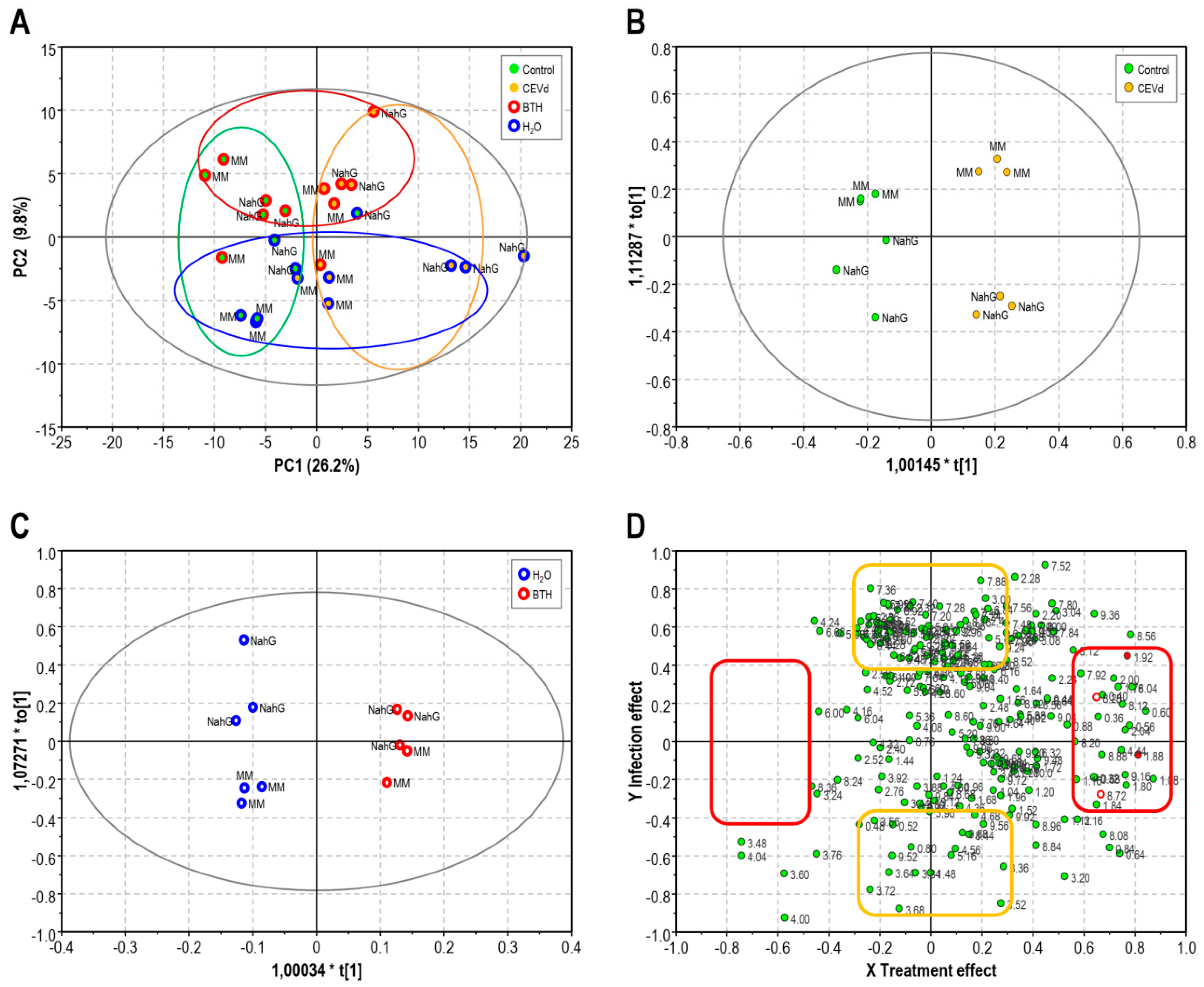
3.1. Partial Least Square of 1H-NMR Spectra of Control and BTH-treated Money Maker and NahG Tomato Plants Infected by CEVd and Mock-Inoculated
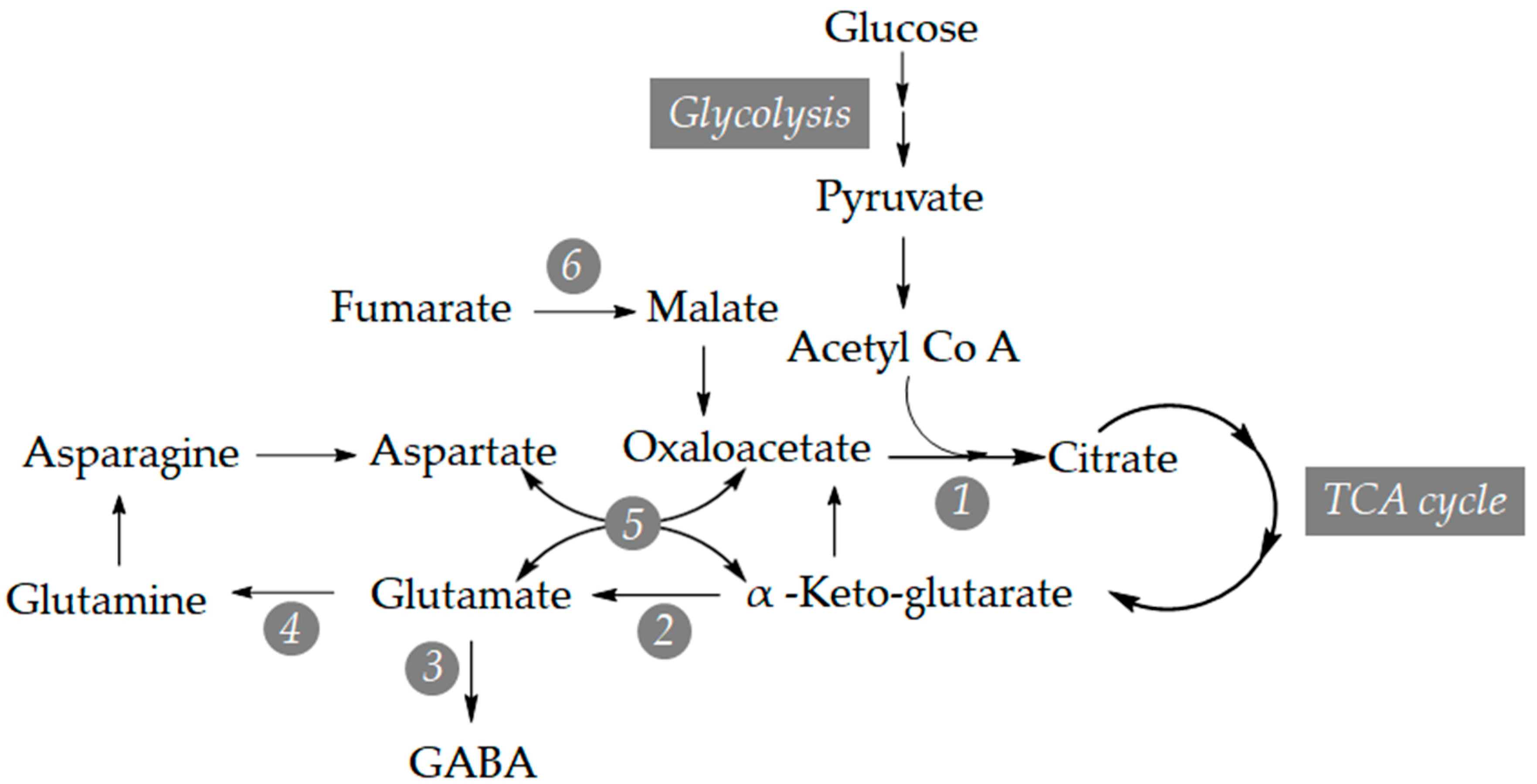
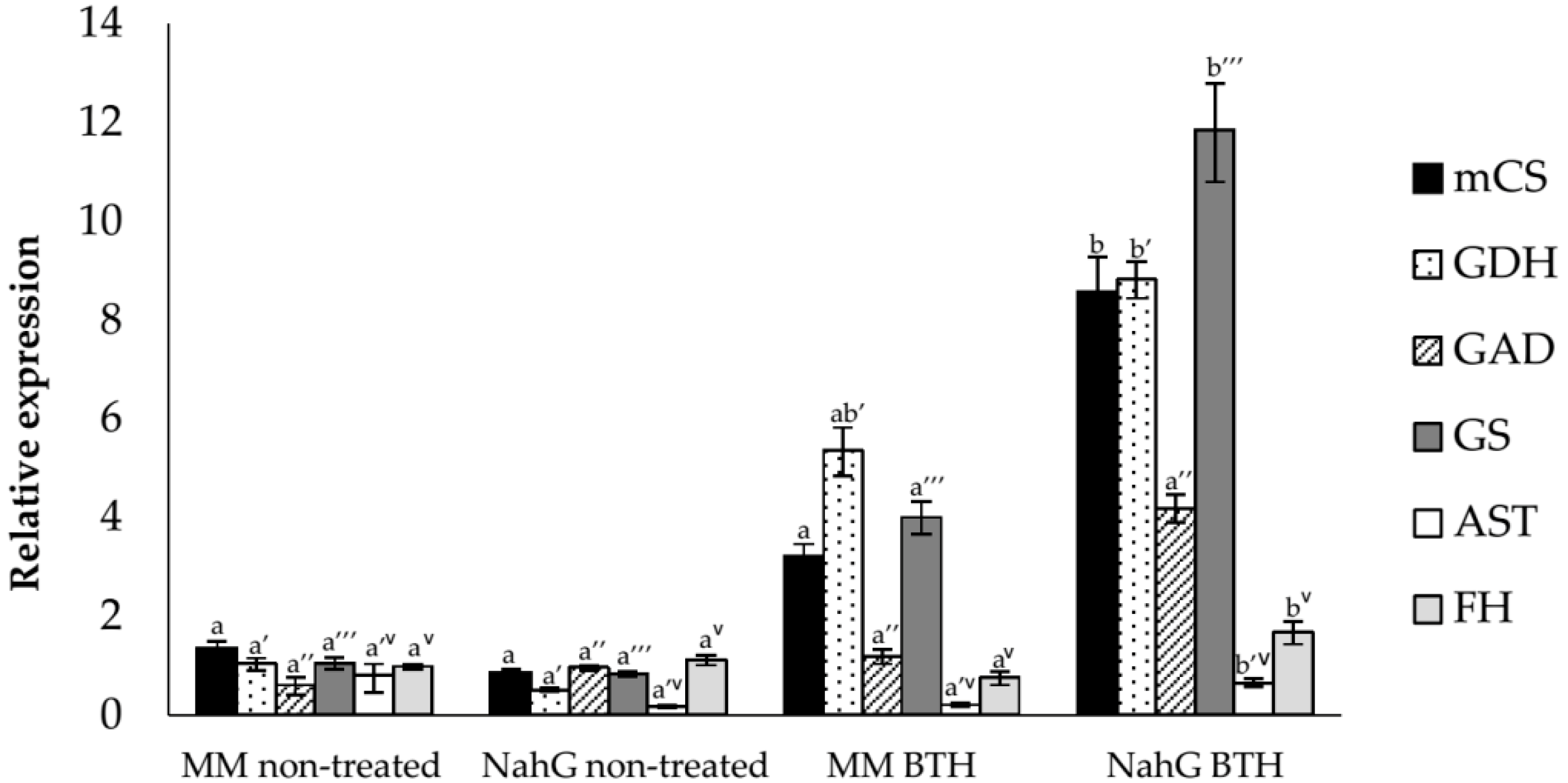
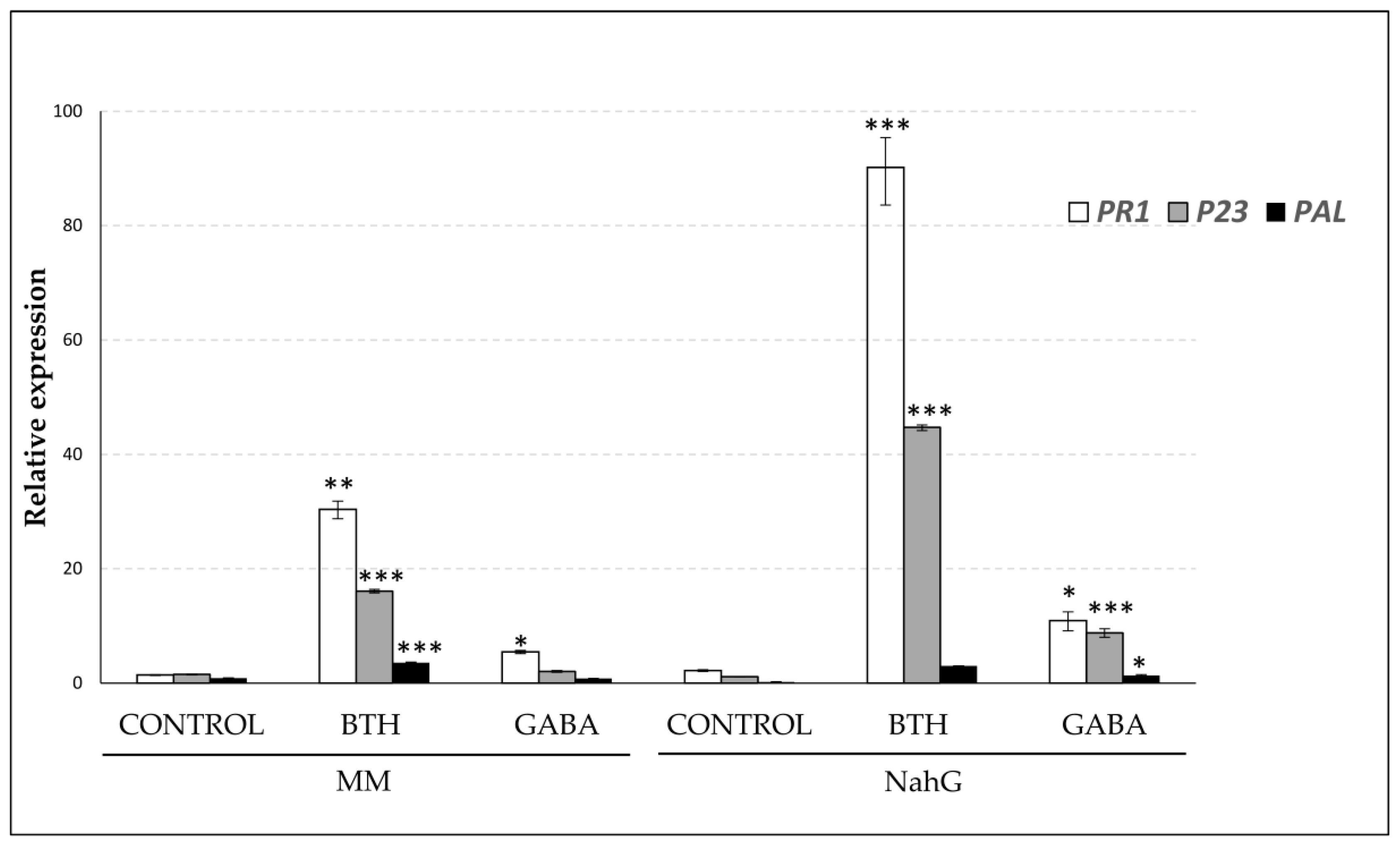
3.2. Study of the Relative Expression Levels of Tomato Genes Involved in GABA Synthesis in Control and BTH-Treated Money Maker and NahG Tomato Plants
3.3. Analysis of Primary Metabolites in Money Maker and NahG Tomato Plants after BTH Treatments
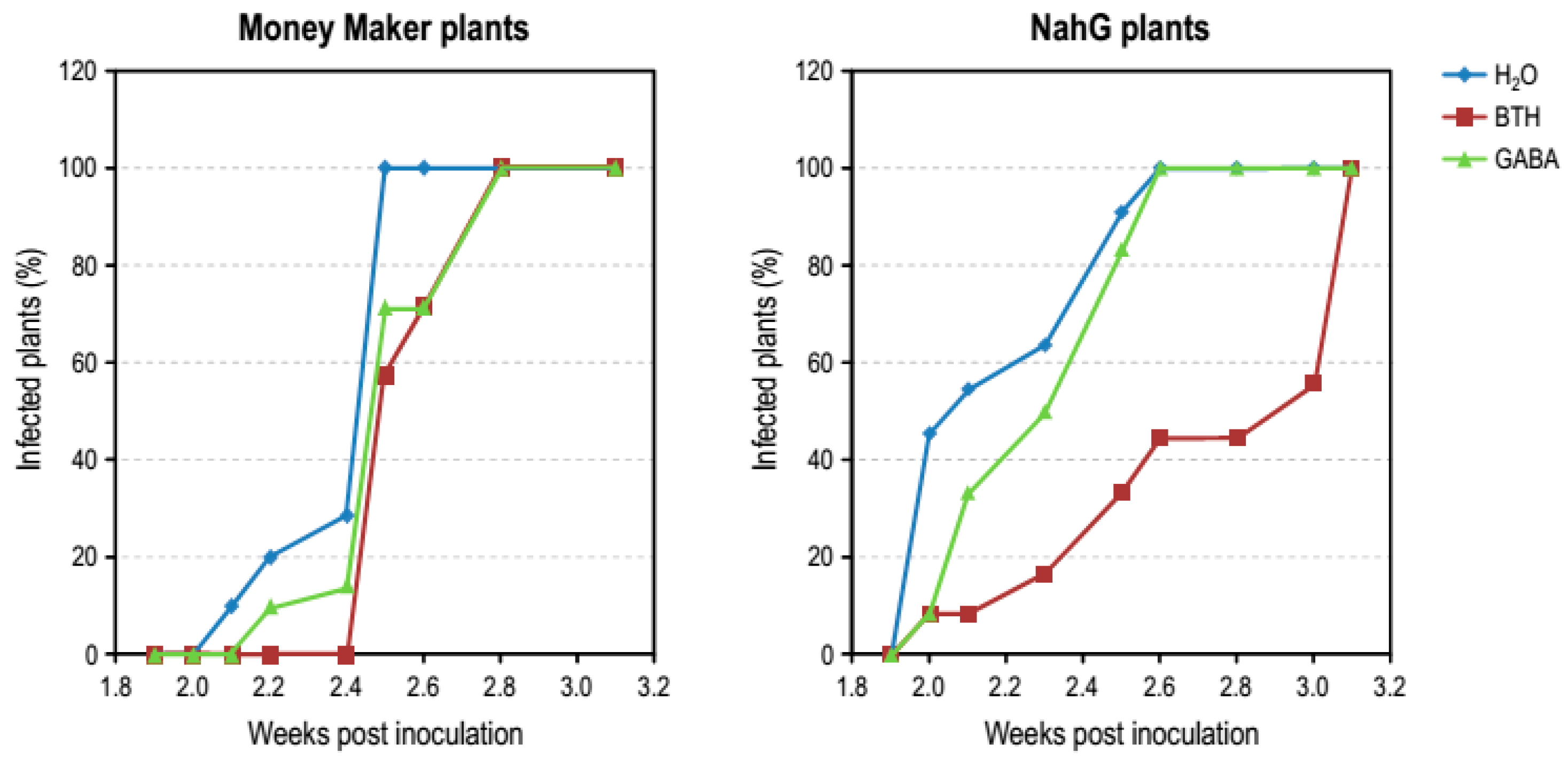
3.4. Effect of GABA Treatments in Money Maker and NahG Plants Infected by CEVd
3.5. Effect of GABA Treatments on the Expression Leves of PAL, PR1, and P23 Genes in Money Maker and NahG Plants
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mou, Z.; Fan, W.; Dong, X. Inducers of Plant Systemic Acquired Resistance Regulate NPR1 Function through Redox Changes. Cell 2003, 113, 935–944. [Google Scholar] [CrossRef]
- Bellés, J.M.; Garro, R.; Pallás, V.; Fayos, J.; Rodrigo, I.; Conejero, V. Accumulation of gentisic acid as associated with systemic infections but not with the hypersensitive response in plant-pathogen interactions. Planta 2006, 223, 500–511. [Google Scholar] [CrossRef] [PubMed]
- Bellés, J.M.; Garro, R.; Fayos, J.; Navarro, P.; Primo, J.; Conejero, V. Gentisic acid as a pathogen-inducible signal, additional to salicylic acid for activation of plant defenses in tomato. Mol. Plant-Microbe Interact. 1999, 12, 227–235. [Google Scholar] [CrossRef]
- Brading, P.A.; Hammond-Kosack, K.E.; Parr, A.; Jones, J.D.G. Salicylic acid is not required for Cf-2- and Cf-9-dependent resistance of tomato to Cladosporium fulvum. Plant J. 2000, 23, 305–318. [Google Scholar] [CrossRef]
- López-Gresa, M.P.; Lisón, P.; Yenush, L.; Conejero, V.; Rodrigo, I.; Bellés, J.M. Salicylic Acid Is Involved in the Basal Resistance of Tomato Plants to Citrus Exocortis Viroid and Tomato Spotted Wilt Virus. PLoS ONE 2016, 11, e0166938. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, L.; Lawton, K.; Ruess, W.; Masner, P.; Specker, N.; Rella, M.G.; Meier, B.; Dincher, S.; Staub, T.; Uknes, S.; et al. A benzothiadiazole derivative induces systemic acquired resistance in tobacco. Plant J. 1996, 10, 61–70. [Google Scholar] [CrossRef]
- Görlach, J.; Volrath, S.; Knauf-Beiter, G.; Hengy, G.; Beckhove, U.; Kogel, K.H.; Oostendorp, M.; Staub, T.; Ward, E.; Kessmann, H.; et al. Benzothiadiazole, a novel class of inducers of systemic acquired resistance, activates gene expression and disease resistance in wheat. Plant Cell 1996, 8, 629–643. [Google Scholar] [CrossRef]
- Lawton, K.A.; Friedrich, L.; Hunt, M.; Weymann, K.; Delaney, T.; Kessmann, H.; Staub, T.; Ryals, J. Benzothiadiazole induces disease resistance in Arabidopsis by activation of the systemic acquired resistance signal transduction pathway. Plant J. 1996, 10, 71–82. [Google Scholar]
- Benhamou, N.; Belanger, R.R. Benzothiadiazole-mediated induced resistance to Fusarium oxysporum f. sp. radicis-lycopersici in tomato. Plant Physiol. 1998, 118, 1203–1212. [Google Scholar] [CrossRef]
- Louws, F.J.; Wilson, M.; Campbell, H.L.; Cuppels, D.A.; Jones, J.B.; Shoemaker, P.B.; Sahin, F.; Miller, S.A. Field Control of Bacterial Spot and Bacterial Speck of Tomato Using a Plant Activator. Plant Dis. 2001, 85, 481–488. [Google Scholar] [CrossRef]
- Li, X.; Bi, Y.; Wang, J.; Dong, B.; Li, H.; Gong, D.; Zhao, Y.; Tang, Y.; Yu, X.; Shang, Q. BTH treatment caused physiological, biochemical and proteomic changes of muskmelon (Cucumis melo L.) fruit during ripening. J. Proteom. 2015, 120, 179–193. [Google Scholar] [CrossRef]
- Hien Dao, T.T.; Puig, R.C.; Kim, H.K.; Erkelens, C.; Lefeber, A.W.M.; Linthorst, H.J.M.; Choi, Y.H.; Verpoorte, R. Effect of benzothiadiazole on the metabolome of Arabidopsis thaliana. Plant Physiol. Biochem. 2009, 47, 146–152. [Google Scholar] [CrossRef]
- Vogt, T. Phenylpropanoid biosynthesis. Mol. Plant 2010, 3, 2–20. [Google Scholar] [CrossRef]
- Katz, V.A.; Thulke, O.U.; Conrath, U. A Benzothiadiazole Primes Parsley Cells for Augmented Elicitation of Defense Responses. Plant Physiol. 1998, 117, 1333–1339. [Google Scholar] [CrossRef]
- Iriti, M.; Rossoni, M.; Borgo, M.; Faoro, F. Benzothiadiazole Enhances Resveratrol and Anthocyanin Biosynthesis in Grapevine, Meanwhile Improving Resistance to Botrytis cinerea. J. Agric. Food Chem. 2004, 52, 4406–4413. [Google Scholar] [CrossRef]
- Verpoorte, R.; Choi, Y.; Kim, H. NMR-based metabolomics at work in phytochemistry. Phytochem. Rev. 2007, 6, 3–14. [Google Scholar] [CrossRef]
- López-Gresa, M.P.; Maltese, F.; Bellés, J.M.; Conejero, V.; Kim, H.K.; Choi, Y.H.; Verpoorte, R. Metabolic response of tomato leaves upon different plant-pathogen interactions. Phytochem. Anal. 2010, 21, 89–94. [Google Scholar] [CrossRef]
- López-Gresa, M.P.; Lisón, P.; Kim, H.K.; Choi, Y.H.; Verpoorte, R.; Rodrigo, I.; Conejero, V.; Bellés, J.M. Metabolic fingerprinting of Tomato Mosaic Virus infected Solanum lycopersicum. J. Plant Physiol. 2012, 169, 1586–1596. [Google Scholar] [CrossRef]
- Shelp, B.J.; Bozzo, G.G.; Trobacher, C.P.; Zarei, A.; Deyman, K.L.; Brikis, C.J. Hypothesis/review: Contribution of putrescine to 4-aminobutyrate (GABA) production in response to abiotic stress. Plant Sci. 2012, 193–194, 130–135. [Google Scholar] [CrossRef]
- Yu, C.; Zeng, L.; Sheng, K.; Chen, F.; Zhou, T.; Zheng, X.; Yu, T. γ-Aminobutyric acid induces resistance against Penicillium expansum by priming of defence responses in pear fruit. Food Chem. 2014, 159, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Bolton, M.D. Primary metabolism and plant defense—Fuel for the fire. Mol. Plant Microbe Interact. 2009, 22, 487–497. [Google Scholar] [CrossRef]
- Seifi, H.S.; Curvers, K.; de Vleesschauwer, D.; Delaere, I.; Aziz, A.; Hofte, M. Concurrent overactivation of the cytosolic glutamine synthetase and the GABA shunt in the ABA-deficient sitiens mutant of tomato leads to resistance against Botrytis cinerea. New Phytol. 2013, 199, 490–504. [Google Scholar] [CrossRef]
- Oldroyd, G.E.D.; Staskawicz, B.J. Genetically engineered broad-spectrum disease resistance in tomato. Proc. Natl. Acad. Sci. USA 1998, 95, 10300–10305. [Google Scholar] [CrossRef]
- Roessner, U.; Wagner, C.; Kopka, J.; Trethewey, R.N.; Willmitzer, L. Technical advance: Simultaneous analysis of metabolites in potato tuber by gas chromatography-mass spectrometry. Plant J. 2000, 23, 131–142. [Google Scholar] [CrossRef]
- Bellés, J.M.; López-Gresa, M.P.; Fayos, J.; Pallás, V.; Rodrigo, I.; Conejero, V. Induction of cinnamate 4-hydroxylase and phenylpropanoids in virus-infected cucumber and melon plants. Plant Sci. 2008, 174, 524–533. [Google Scholar] [CrossRef]
- Conejero, V.; Bellés, J.M.; García-Breijo, F.; Garro, R.; Hernández-Yago, J.; Rodrigo, I.; Vera, P. Signaling in Viroid Pathogenesis. In Recognition and Response in Plant-Virus Interactions; Fraser, R.S., Ed.; Springer: Berlin/Heidelberg, Germany, 1990; Volume 41, pp. 233–261. [Google Scholar]
- Fayos, J.; Bellés, J.M.; López-Gresa, M.P.; Primo, J.; Conejero, V. Induction of gentisic acid 5-O-β-D-xylopyranoside in tomato and cucumber plants infected by different pathogens. Phytochemistry 2006, 67, 142–148. [Google Scholar] [CrossRef]
- Kinnersley, A.M.; Turano, F.J. Gamma Aminobutyric Acid (GABA) and Plant Responses to Stress. Crit. Rev. Plant Sci. 2000, 19, 479–509. [Google Scholar] [CrossRef]
- Roberts, M.R. Does GABA Act as a Signal in Plants?: Hints from Molecular Studies. Plant Signal. Behav. 2007, 2, 408–409. [Google Scholar] [CrossRef]
- Kawano, T.; Sahashi, N.; Takahashi, K.; Uozumi, N.; Muto, S. Salicylic Acid Induces Extracellular Superoxide Generation Followed by an Increase in Cytosolic Calcium Ion in Tobacco Suspension Culture: The Earliest Events in Salicylic Acid Signal Transduction. Plant Cell Physiol. 1998, 39, 721–730. [Google Scholar] [CrossRef]
- Van der Merwe, J.A.; Dubery, I.A. Benzothiadiazole inhibits mitochondrial NADH: Ubiquinone oxidoreductase in tobacco. J. Plant Physiol. 2006, 163, 877–882. [Google Scholar] [CrossRef]
- Wendehenne, D.; Durner, J.; Chen, Z.; Klessig, D.F. Benzothiadiazole, an inducer of plant defenses, inhibits catalase and ascorbate peroxidase. Phytochemistry 1998, 47, 651–657. [Google Scholar] [CrossRef]
- Song, H.; Xu, X.; Wang, H.; Wang, H.; Tao, Y. Exogenous gamma-aminobutyric acid alleviates oxidative damage caused by aluminium and proton stresses on barley seedlings. J. Sci. Food Agric. 2010, 90, 1410–1416. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Duan, B.; Li, C.; Tang, Q.; Li, X.; Wei, M.; Chen, Y.; Li, J. γ-Aminobutyric acid delays senescence of blueberry fruit by regulation of reactive oxygen species metabolism and phenylpropanoid pathway. Sci. Hortic-Amsterdam 2018, 240, 303–309. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, P.; Chen, Z.; Gu, Z.; Yang, R. GABA enhances physio-biochemical metabolism and antioxidant capacity of germinated hulless barley under NaCl stress. J. Plant Physiol. 2018, 231, 192–201. [Google Scholar] [CrossRef]
- Aghdam, M.S.; Kakavand, F.; Rabiei, V.; Zaare-Nahandi, F.; Razavi, F. γ-Aminobutyric acid and nitric oxide treatments preserve sensory and nutritional quality of cornelian cherry fruits during postharvest cold storage by delaying softening and enhancing phenols accumulation. Sci. Hortic-Amsterdam 2019, 246, 812–817. [Google Scholar] [CrossRef]
- Bown, A.W.; Shelp, B.J. Plant GABA: Not Just a Metabolite. Trends Plant Sci. 2016, 21, 811–813. [Google Scholar] [CrossRef]
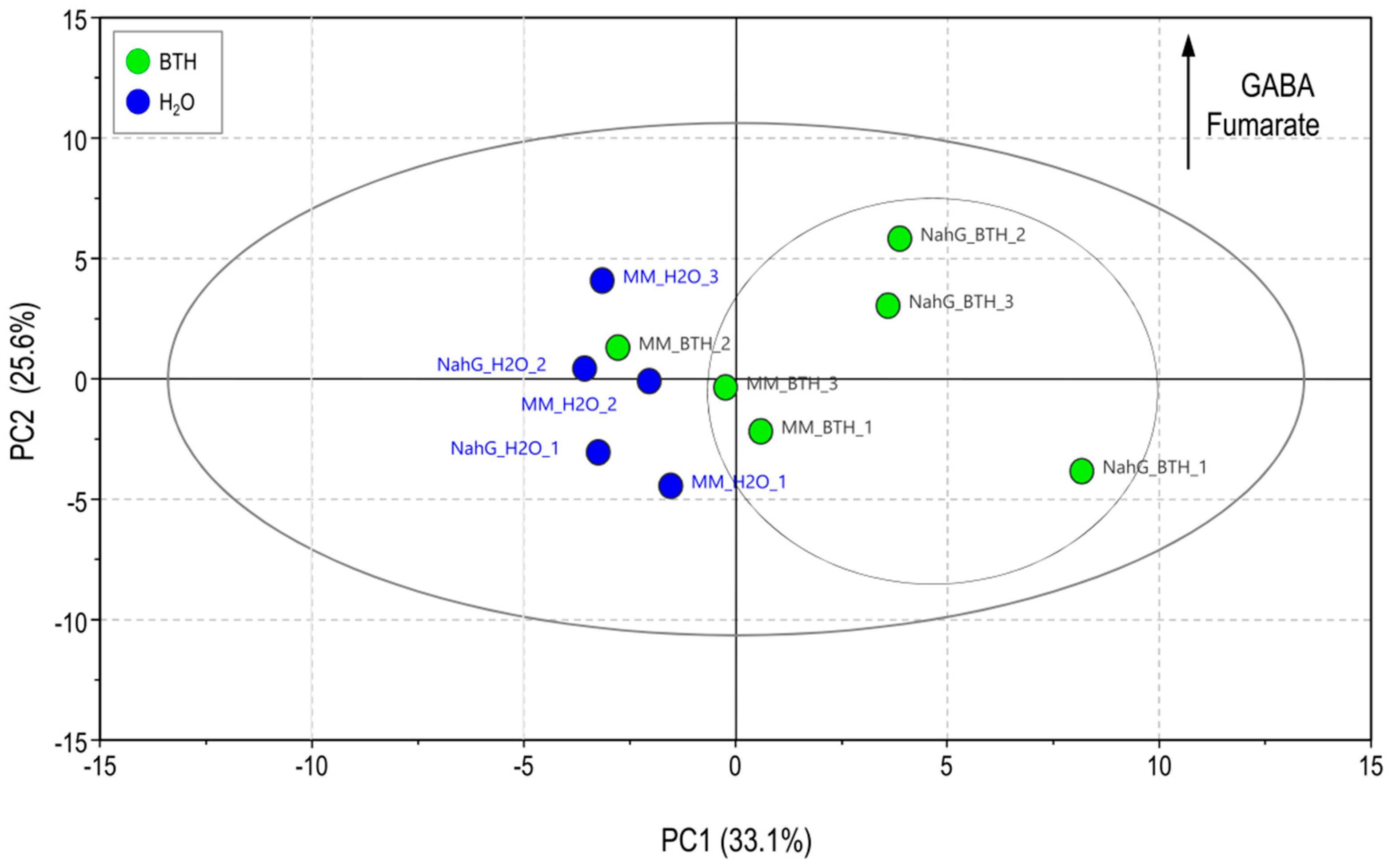

| BTH Treated vs Non-Treated Plants | ||
|---|---|---|
| Money Maker | NahG | |
| Mock-inoculated | GABA hydrolyzed-BTH | GABA 1 trigonelline |
| CEVd-inoculated | malic acid aspartic acid trigonelline | caffeic acid 1 ferulic acid 1 citric acid 1 malic acid 1 aspartic acid threonine alanine α-glucose trigonelline |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Gresa, M.P.; Payá, C.; Rodrigo, I.; Bellés, J.M.; Barceló, S.; Hae Choi, Y.; Verpoorte, R.; Lisón, P. Effect of Benzothiadiazole on the Metabolome of Tomato Plants Infected by Citrus Exocortis Viroid. Viruses 2019, 11, 437. https://doi.org/10.3390/v11050437
López-Gresa MP, Payá C, Rodrigo I, Bellés JM, Barceló S, Hae Choi Y, Verpoorte R, Lisón P. Effect of Benzothiadiazole on the Metabolome of Tomato Plants Infected by Citrus Exocortis Viroid. Viruses. 2019; 11(5):437. https://doi.org/10.3390/v11050437
Chicago/Turabian StyleLópez-Gresa, María Pilar, Celia Payá, Ismael Rodrigo, José María Bellés, Susana Barceló, Young Hae Choi, Robert Verpoorte, and Purificación Lisón. 2019. "Effect of Benzothiadiazole on the Metabolome of Tomato Plants Infected by Citrus Exocortis Viroid" Viruses 11, no. 5: 437. https://doi.org/10.3390/v11050437
APA StyleLópez-Gresa, M. P., Payá, C., Rodrigo, I., Bellés, J. M., Barceló, S., Hae Choi, Y., Verpoorte, R., & Lisón, P. (2019). Effect of Benzothiadiazole on the Metabolome of Tomato Plants Infected by Citrus Exocortis Viroid. Viruses, 11(5), 437. https://doi.org/10.3390/v11050437









