Hepatitis A Strain Linked to the European Outbreaks During Gay Events between 2016 and 2017, Identified in a Brazilian Homosexual Couple in 2017
Abstract
1. Introduction
2. Materials and Methods
Investigation
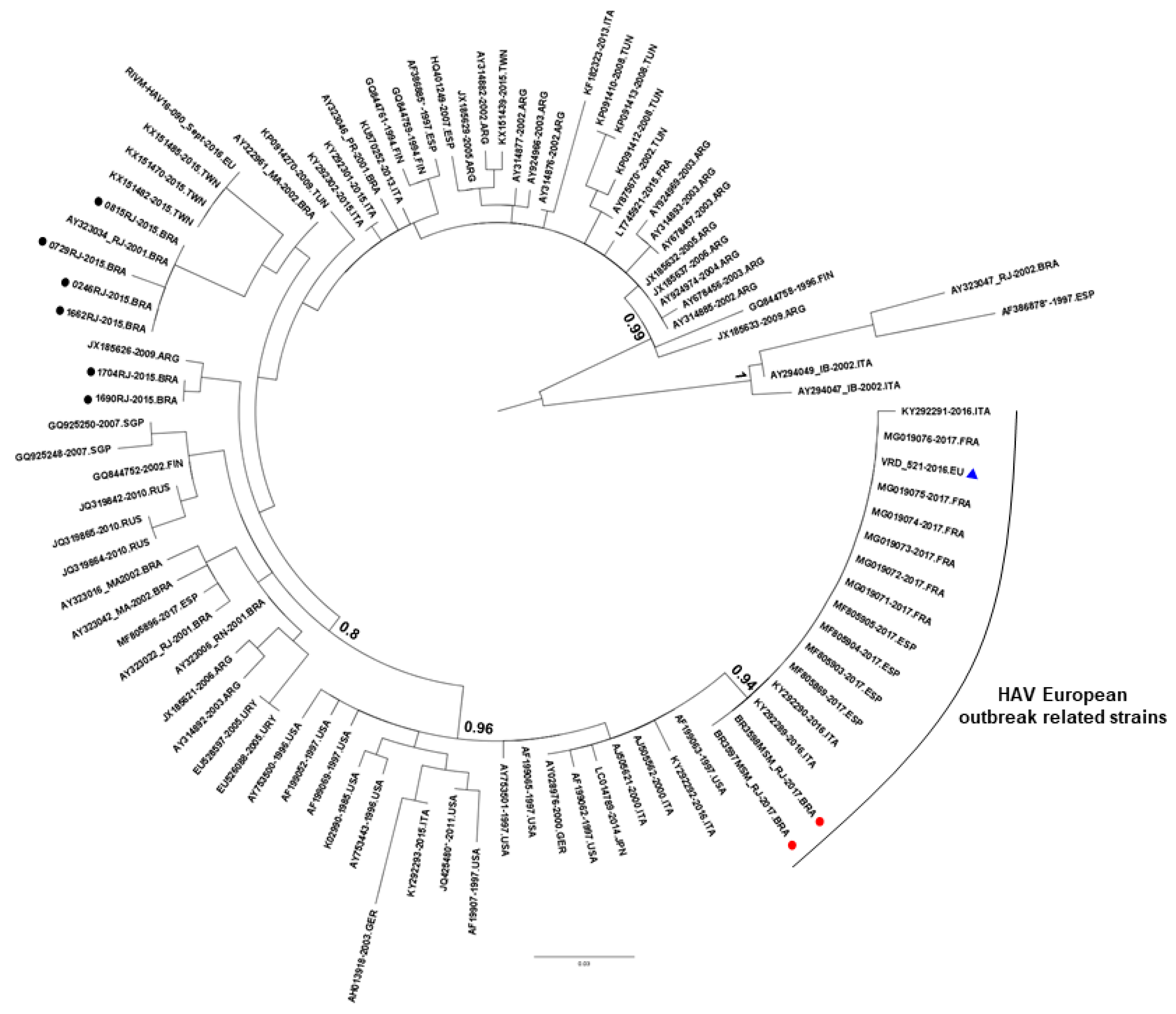
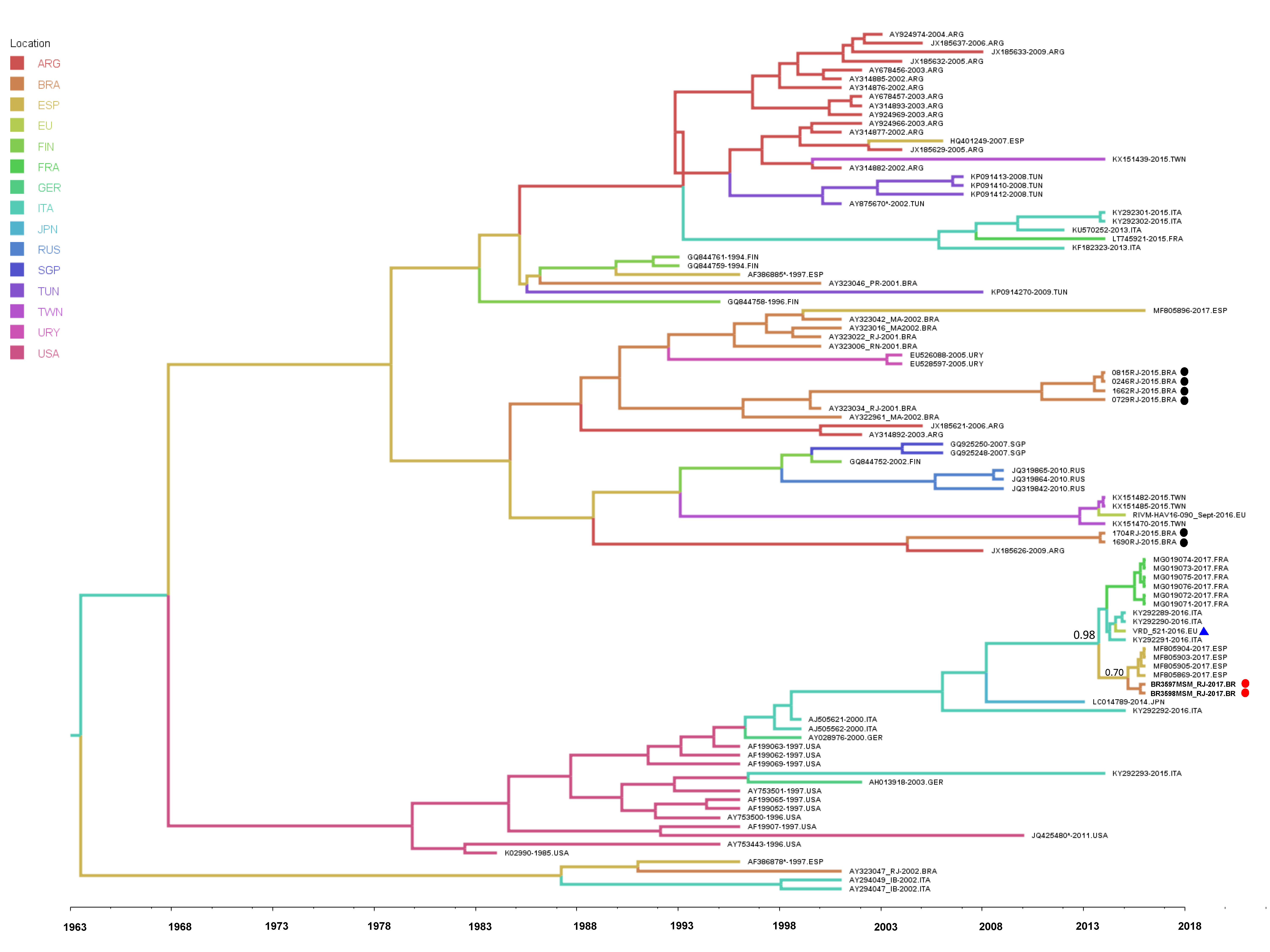
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Centers for Disease Control and Prevention (CDC). Hepatitis A Information. 2017. Available online: https://www.cdc.gov/hepatitis/hav/index.htm (accessed on 10 August 2018).
- Corey, L.; Holmes, K.K. Sexual transmission of hepatitis A in homosexual men: Incidence and mechanism. N. Engl. J. Med. 1980, 302, 435–438. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control. Hepatitis A among homosexual men—United States, Canada, and Australia. Morb. Mortal. Wkly. Rep. 1992, 41, 155–161. [Google Scholar] [CrossRef]
- Leentvaar-Kuijpers, A.; Kool, J.L.; Veugelers, P.J.; Coutinho, R.A.; van Griensven, G.J. An outbreak of hepatitis A among homosexual men in Amsterdam, 1991–1993. Int. J. Epidemiol. 1995, 24, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Christenson, B.; Broström, C.; Böttiger, M.; Hermanson, J.; Weiland, O.; Ryd, G.; Berg, J.V.; Sjüblom, R. An epidemic outbreak of hepatitis A among homosexual men in Stockholm. Hepatitis A, a special hazard for the male homosexual subpopulation in Sweden. Am. J. Epidemiol. 1982, 116, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Henning, K.J.; Bell, E.; Braun, J.; Barker, N.D. A community-wide outbreak of hepatitis A: Risk factors for infection among homosexual and bisexual men. Am. J. Med. 1995, 99, 132–136. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control. Hepatitis a Outbreaks in the EU/EEA Mostly Affecting Men Who Have Sex with Men—Third Update, 28 June 2017; ECDC: Stockholm, Sweden, 2017; Available online: https://ecdc.europa.eu/en/publications-data/rapid-risk-assessment-hepatitis-outbreak-eueea-mostly-affecting-men-who-have-sex (accessed on 15 August 2018).
- Gozlan, Y.; Bar-Or, I.; Rakovsky, A.; Savion, M.; Amitai, Z.; Sheffer, R.; Ceder, N.; Anis, E.; Grotto, I.; Mendelson, E.; et al. Ongoing Hepatitis A among men who have sex with men (MSM) linked to outbreaks in Europe in Tel Aviv area, Israel, December 2016–June 2017. Eurosurveillance 2017, 20, 22–29. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Hepatitis A Outbreaks Mostly Affecting Men Who Have Sex with Men—European Region and the Americas. 2017. Available online: http://www.who.int/csr/don/07-june-2017-hepatitis-a/en/ (accessed on 10 August 2018).
- Chen, G.J.; Lin, K.Y.; Hung, C.C.; Chang, S.C. Hepatitis A Outbreak Among Men Who Have Sex With Men in a Country of Low Endemicity of Hepatitis A Infection. J. Infect. Dis. 2017, 215, 1339–1340. [Google Scholar] [CrossRef]
- Rivas, V.; Barrera, A.; Pino, K.; Núñez, R.; Caceres, C.J.; Lopez-Lastra, M.; Soza, A. Hepatitis A outbreak since November 2016 affecting men who have sex with men (MSM) in Chile connected to the current outbreak in MSM in Europe, situation up to October 2017. Eurosurveillance 2018, 23, 18-00060. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Epidemiological Update: Hepatitis a Outbreak in the EU/EEA Mostly Affecting Men Who Have Sex with Men, 22 December 2017; ECDC: Stockholm, Sweden, 2017; Available online: https://ecdc.europa.eu/en/news-events/epidemiological-update-hepatitis-outbreak-eueea-mostly-affecting-men-who-have-sex-men-0 (accessed on 20 November 2018).
- Governo Federal do Brasil, Ministério da Saúde. Boletim Epidemiológico de Hepatites Virais–2018. 2018. Available online: http://www.aids.gov.br/pt-br/pub/2018/boletim-epidemiologico-de-hepatites-virais-2018 (accessed on 20 August 2018).
- Governo Federal do Brasil, Ministério da Saúde. Nota Informativa nº 10/2018—Ampliação da Indicação do Uso da Vacina Hepatite A Para Pessoas Que Tenham Prática Sexual Com Contato Oral-Anal (Com Priorização de Gays e Homens Que Fazem Sexo Com Homens—HSH). Available online: http://www.aids.gov.br/pt-br/legislacao/nota-informativa-no-102018-covigcgvpdiahvsvsms (accessed on 20 August 2018).
- Shire, N.J.; Welge, J.A.; Sherman, K.E. Efficacy of inactivated hepatitis A vaccine in HIV-infected patients: A hierarchical bayesian meta-analysis. Vaccine 2006, 24, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Aloise, R.; de Almeida, A.J.; Sion, F.S.; Morais-de-Sá, C.A.; Gaspar, A.M.; de Paula, V.S. Changes in hepatitis A virus seroepidemiology in HIV-infected Brazilian patients. Int. J. STD AIDS 2008, 19, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Bordi, L.; Rozera, G.; Scognamiglio, P.; Minosse, C.; Loffredo, M.; Antinori, A.; Narciso, P.; Ippolito, G.; Girardi, E.; Capobianchi, M.R.; et al. Monophyletic outbreak of Hepatitis A involving HIV-infected men who have sex with men, Rome, Italy 2008–2009. J. Clin. Virol. 2012, 54, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Cella, E.; Riva, E.; Angeletti, S.; Fogolari, M.; Blasi, A.; Scolamacchia, V.; Spoto, S.; Bazzardi, R.; Lai, A.; Sagnelli, C.; et al. Genotype I hepatitis A virus introduction in Italy: Bayesian phylogenetic analysis to date different epidemics. J. Med. Virol. 2018, 90, 1493–1502. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, X.Y.; Zheng, H.H.; Cao, J.Y.; Zhou, W.T.; Bi, S.L. Evolution and genetic characterization of hepatitis A virus isolates in China. Int. J. Infect. Dis. 2015, 33, 156–158. [Google Scholar] [CrossRef] [PubMed]
- Lanini, S.; Minosse, C.; Vairo, F.; Garbuglia, A.; Di Bari, V.; Agresta, A.; Rezza, G.; Puro, V.; Pendenza, A.; Loffredo, M.R.; et al. A large ongoing outbreak of hepatitis A predominantly affecting young males in Lazio, Italy; August 2016–March 2017. PLoS ONE 2017, 12, e0185428. [Google Scholar] [CrossRef] [PubMed]
- Federal Government of Brazil. Media Guide—Rio 2016 Olympic and Paralympic Games. Federal Government of Brazil, 2016. Available online: http://www.brasil2016.gov.br/en/news/federal-government-launches-journalist-guide-for-the-rio-2016-olympic-and-paralympic-games/media-guide-rio-2016-olympic (accessed on 10 August 2018).
- Faria, N.R.; Azevedo, R.D.S.D.; Kraemer, M.U.G.; Souza, R.; Cunha, M.S.; Hill, S.C.; Thézé, J.; Bonsall, M.B.; Bowden, T.A.; Rissanen, I.; et al. Zika virus in the Americas: Early epidemiological and genetic findings. Science 2016, 352, 345–349. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mello, V.M.; Lago, B.V.; Sousa, P.S.F.; Mello, F.C.A.; Souza, C.B.; Pinto, L.C.M.; Ginuino, C.F.; Fernandes, C.A.S.; Aguiar, S.F.; Villar, L.M.; et al. Hepatitis A Strain Linked to the European Outbreaks During Gay Events between 2016 and 2017, Identified in a Brazilian Homosexual Couple in 2017. Viruses 2019, 11, 281. https://doi.org/10.3390/v11030281
Mello VM, Lago BV, Sousa PSF, Mello FCA, Souza CB, Pinto LCM, Ginuino CF, Fernandes CAS, Aguiar SF, Villar LM, et al. Hepatitis A Strain Linked to the European Outbreaks During Gay Events between 2016 and 2017, Identified in a Brazilian Homosexual Couple in 2017. Viruses. 2019; 11(3):281. https://doi.org/10.3390/v11030281
Chicago/Turabian StyleMello, Vinicius M., Barbara V. Lago, Paulo S. F. Sousa, Francisco C. A. Mello, Caroline B. Souza, Laura C. M. Pinto, Cleber F. Ginuino, Carlos A. S. Fernandes, Shirlei F. Aguiar, Lívia M. Villar, and et al. 2019. "Hepatitis A Strain Linked to the European Outbreaks During Gay Events between 2016 and 2017, Identified in a Brazilian Homosexual Couple in 2017" Viruses 11, no. 3: 281. https://doi.org/10.3390/v11030281
APA StyleMello, V. M., Lago, B. V., Sousa, P. S. F., Mello, F. C. A., Souza, C. B., Pinto, L. C. M., Ginuino, C. F., Fernandes, C. A. S., Aguiar, S. F., Villar, L. M., Lampe, E., Melgaço, J. G., & Lewis-Ximenez, L. L. (2019). Hepatitis A Strain Linked to the European Outbreaks During Gay Events between 2016 and 2017, Identified in a Brazilian Homosexual Couple in 2017. Viruses, 11(3), 281. https://doi.org/10.3390/v11030281





