AIDS Clinical Research in Spain—Large HIV Population, Geniality of Doctors, and Missing Opportunities
Abstract
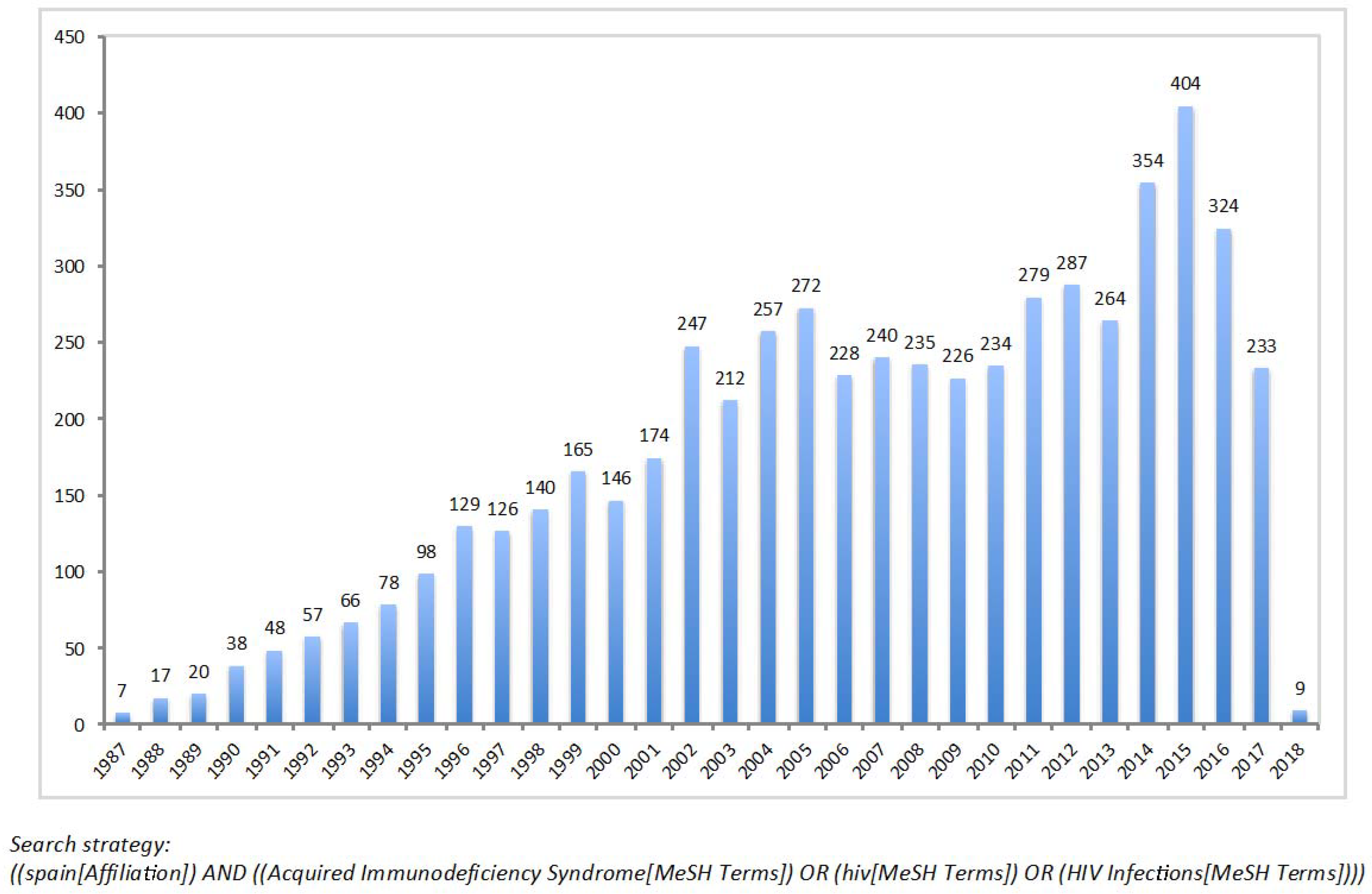
1. Introduction
2. Tuberculosis and HIV
3. Toxoplasmosis and Leishmaniasis
4. HIV-1 Variants and Spanish HIV-2 Network
5. HIV Drug Resistance Platform
6. HIV Pharmacokinetics and Pharmacogenetics
7. Lipodystrophy, Metabolic Abnormalities, and Cardiovascular Risk
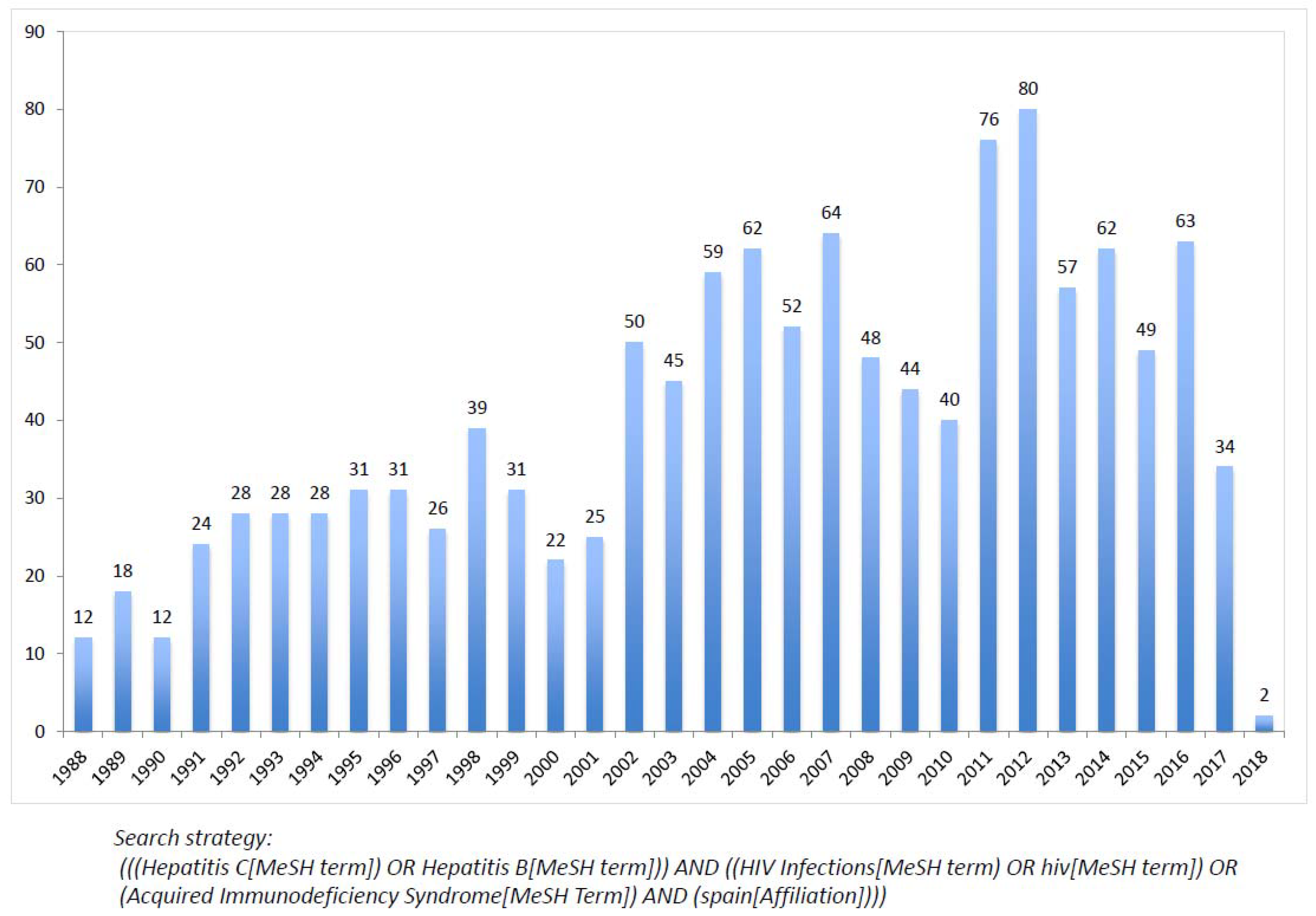
8. Non-Cirrhotic Portal Hypertension in HIV
9. Reproductive Options in HIV
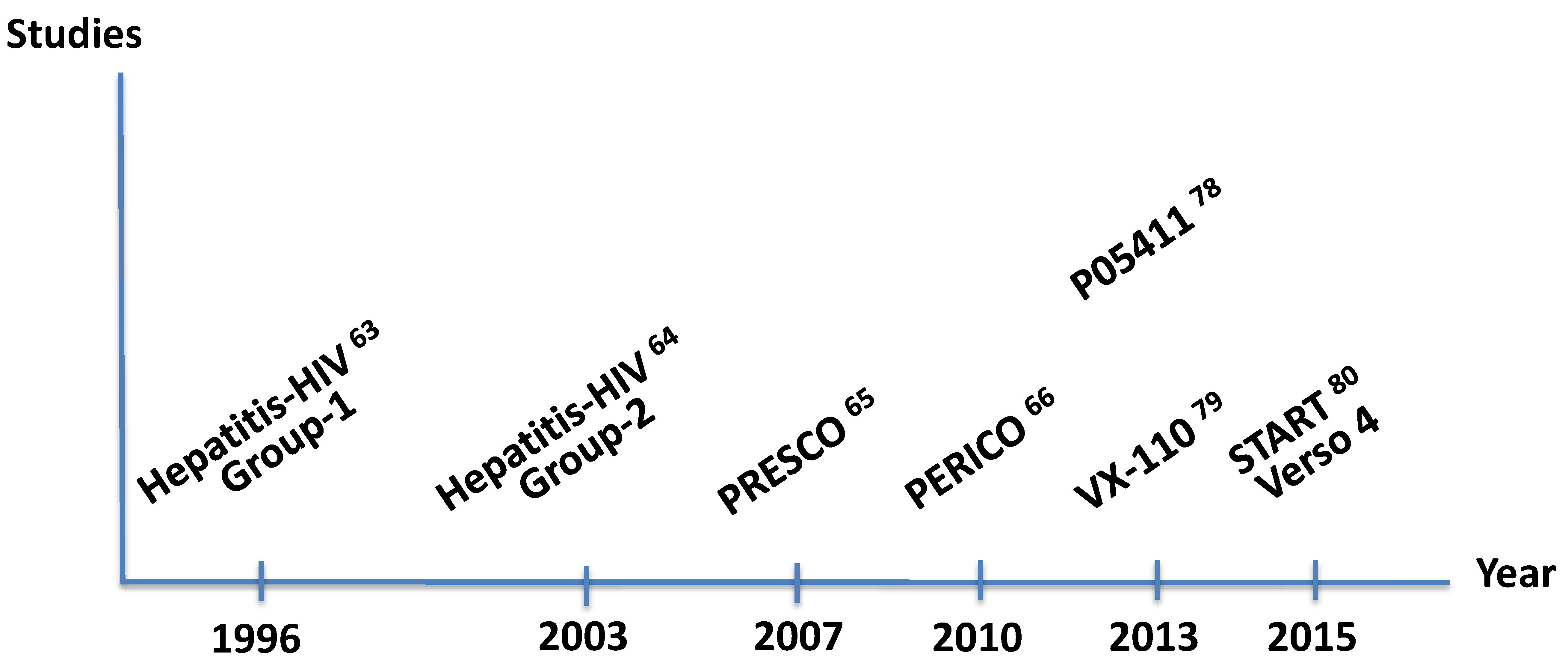
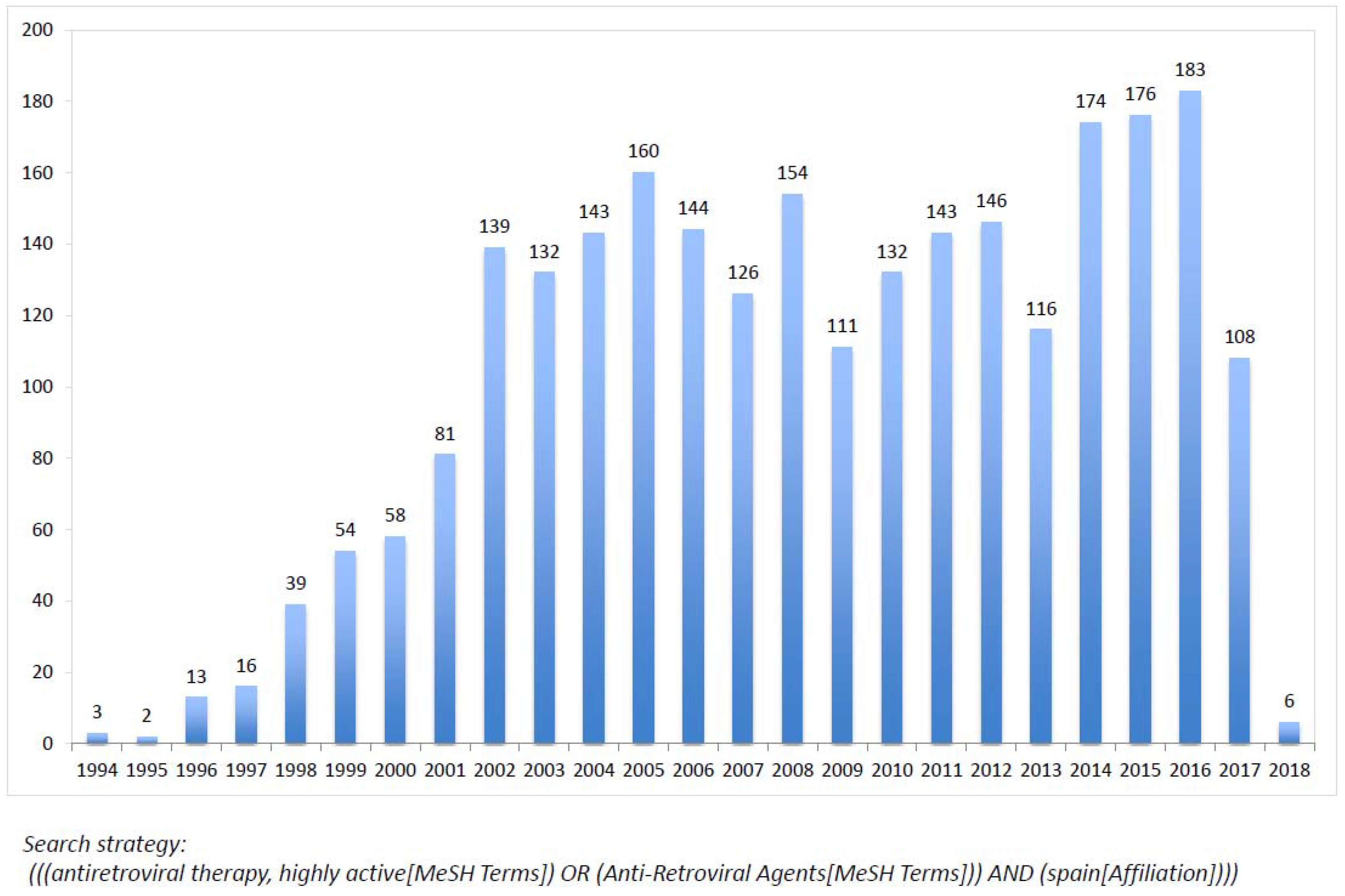
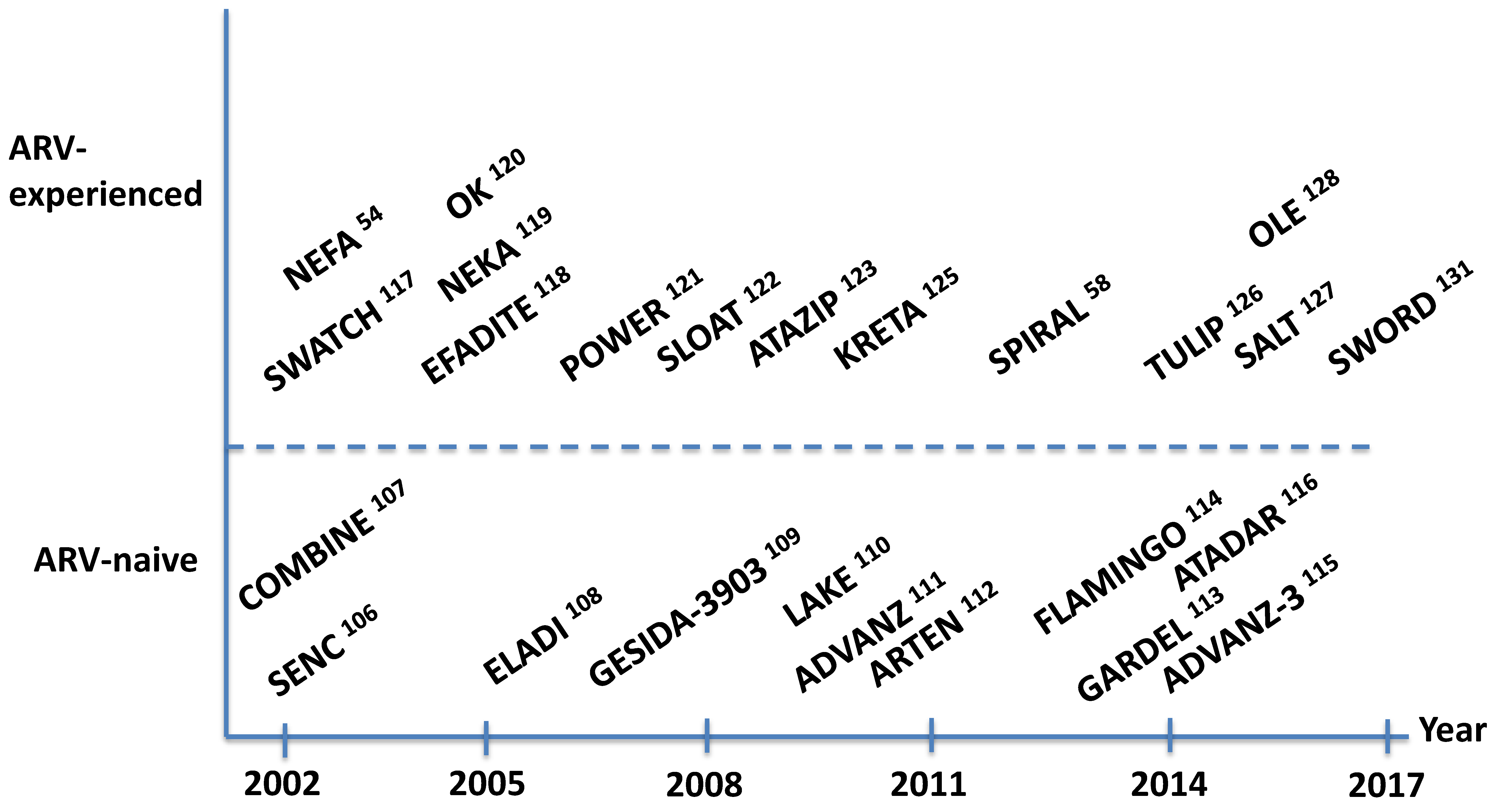
10. Antiretroviral Clinical Trials
11. HIV Immunopathogenesis and Ageing
12. HIV Vaccine Development
13. Other Spanish Contributions to HIV Research
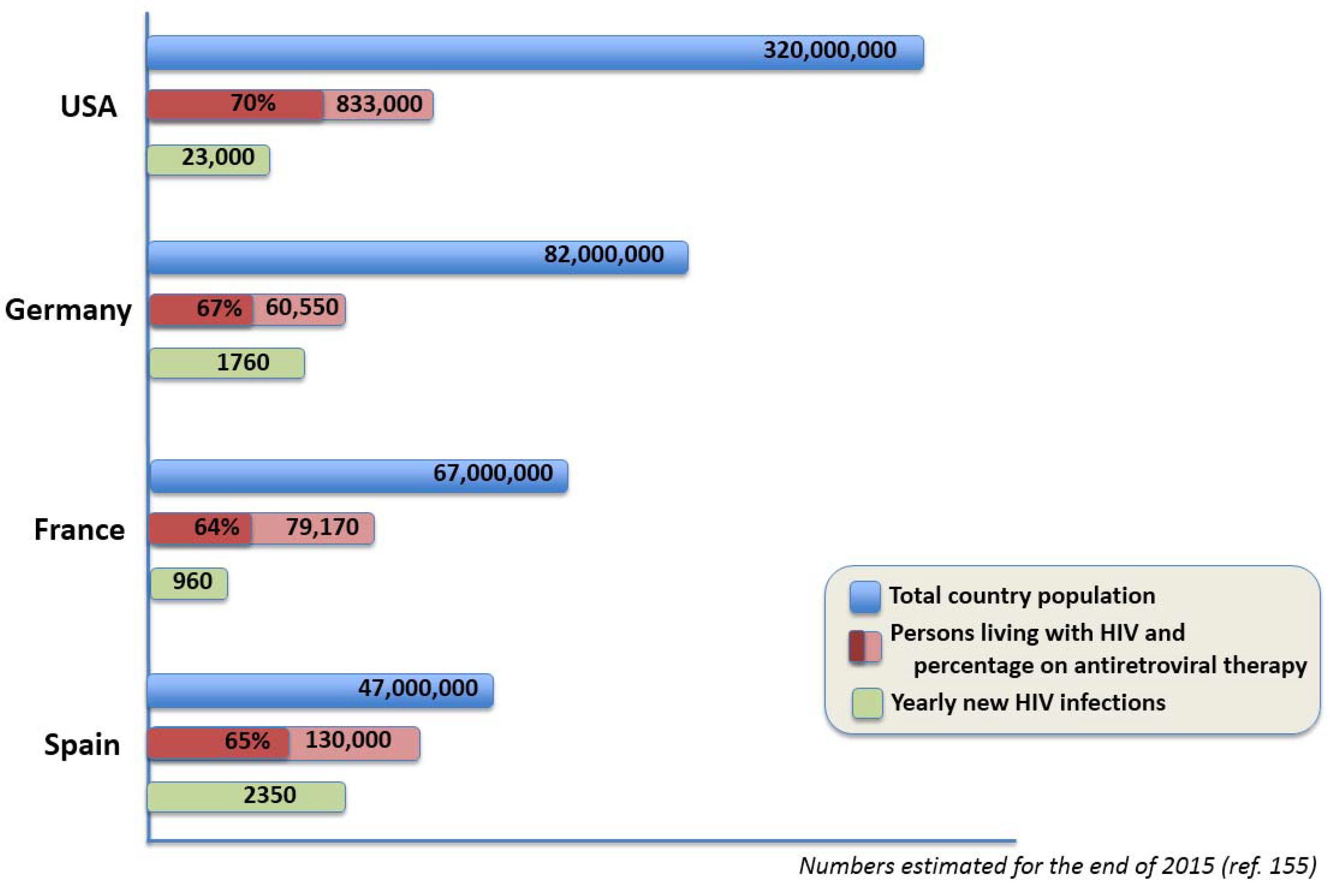
14. Current Spanish HIV Research
15. Future Challenges for Spanish HIV Research
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Lissen, E.; Wichmann, I.; Jimenez, J.M.; Andreu-Kern, F. AIDS in haemophilia patients in Spain. Lancet 1983, 321, 992–993. [Google Scholar] [CrossRef]
- Estruch, R.; Urbano, A.; Pedro-Botet, J.; Cuturi, C.; Urbano-Márquez, A.; Rozman, C. AIDS in a male residing in Barcelona. Med. Clin. 1983, 81, 645. [Google Scholar]
- Sobrino-Vegas, P.; Moreno, S.; Rubio, R.; Viciana, P.; Bernardino, J.I.; Blanco, J.R.; Bernal, E.; Asensi, V.; Pulido, F.; del Amo, J.; et al. Cohorte de la Red de Investigación en Sida, Spain. Impact of late presentation of HIV infection on short-, mid- and long-term mortality and causes of death in a multicenter national cohort: 2004–2013. J. Infect. 2016, 72, 587–596. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, L.; Bravo, M.J.; Barrio, G.; Parras, F.; Suárez, M.; Rodés, A.; Noguer, I. Lessons from the history of the HIV/AIDS epidemic among Spanish drug injectors. Clin. Infect. Dis. 2003, 37 (Suppl. S5), 410–415. [Google Scholar] [CrossRef] [PubMed]
- Ramos, J.M.; González-Alcaide, G.; Gutiérrez, F. Bibliometric analysis of the Spanish scientific production in infectious diseases and microbiology. Enferm. Infecc. Microbiol. Clin. 2016, 34, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Berenguer, J.; Moreno, S.; Laguna, F.; Vicente, T.; Adrados, M.; Ortega, A.; González-Lahoz, J.; Bouza, E. Tuberculous meningitis in patients infected with the HIV. N. Engl. J. Med. 1992, 326, 668–672. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Tor, J.; Gabarre, E.; Gros, T.; Muga, R. Multifocal splenic abscesses caused by Mycobacterium tuberculosis in HIV-infected drug users. AIDS 1991, 5, 901–902. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Multidrug-resistant tuberculosis outbreak on an HIV ward—Madrid, Spain, 1991–1995. Morb. Mortal. Wkly. Rep. 1996, 45, 330–333. [Google Scholar]
- Guerrero, A.; Cobo, J.; Fortún, J.; Navas, E.; Quereda, C.; Asensio, A.; Cañón, J.; Blazquez, J.; Gómez-Mampaso, E. Nosocomial transmission of Mycobacterium bovis resistant to 11 drugs in people with advanced HIV-1 infection. Lancet 1997, 350, 1738–1742. [Google Scholar] [CrossRef]
- Curran, A.; Falcó, V.; Pahissa, A.; Ribera, E. Management of tuberculosis in HIV-infected patients. AIDS Rev. 2012, 14, 231–246. [Google Scholar] [PubMed]
- Podzamczer, D.; Salazar, A.; Jiménez, J.; Consiglio, E.; Santín, M.; Casanova, A.; Rufí, G.; Gudiol, F. Intermittent trimethoprim-sulfamethoxazole compared with dapsone-pyrimethamine for the simultaneous primary prophylaxis of Pneumocystis pneumonia and toxoplasmosis in patients infected with HIV. Ann. Intern. Med. 1995, 122, 755–761. [Google Scholar] [CrossRef] [PubMed]
- Podzamczer, D.; Miró, J.M.; Bolao, F.; Gatell, J.M.; Cosín, J.; Sirera, G.; Domingo, P.; Laguna, F.; Santamaría, J.; Verdejo, J.; et al. Twice-weekly maintenance therapy with sulfadiazine-pyrimethamine to prevent recurrent toxoplasmic encephalitis in patients with AIDS. Ann. Intern. Med. 1995, 123, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Lejeune, M.; Miró, J.M.; De Lazzari, E.; García, F.; Claramonte, X.; Martínez, E.; Ribera, E.; Arrizabalaga, J.; Arribas, J.R.; Domingo, P.; et al. Restoration of T cell responses to toxoplasma gondii after successful combined antiretroviral therapy in patients with AIDS with previous toxoplasmic encephalitis. Clin. Infect. Dis. 2011, 52, 662–670. [Google Scholar] [CrossRef] [PubMed]
- Miro, J.M.; Lopez, J.C.; Podzamczer, D.; Peña, J.M.; Alberdi, J.C.; Martínez, E.; Domingo, P.; Cosin, J.; Claramonte, X.; Arribas, J.R.; et al. Discontinuation of primary and secondary Toxoplasma gondii prophylaxis is safe in HIV-infected patients after immunological restoration with highly active antiretroviral therapy: Results of an open, randomized, multicenter clinical trial. Clin. Infect. Dis. 2006, 43, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Dona, C.; Rodríguez-Rosado, R.; Barreiro, P.; González-Lahoz, J. Discontinuation of secondary prophylaxis for opportunistic infections in HIV-infected patients receiving highly active antiretroviral therapy. AIDS 2000, 14, 383–386. [Google Scholar] [CrossRef] [PubMed]
- Alvar, J.; Cañavate, C.; Gutiérrez-Solar, B.; Jiménez, M.; Laguna, F.; López-Vélez, R.; Molina, R.; Moreno, J. Leishmania and HIV coinfection: The first 10 years. Clin. Microbiol. Rev. 1997, 10, 298–319. [Google Scholar] [PubMed]
- Pasquau, F.; Ena, J.; Sanchez, R.; Cuadrado, J.M.; Amador, C.; Flores, J.; Benito, C.; Redondo, C.; Lacruz, J.; Abril, V.; et al. Leishmaniasis as an opportunistic infection in HIV-infected patients: Determinants of relapse and mortality in a collaborative study of 228 episodes in a Mediterranean region. Eur. J. Clin. Microbiol. Infect. Dis. 2005, 24, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Laguna, F.; García-Samaniego, J.; Soriano, V.; Gómez-Cano, M.; Laguna, F.; Muñoz, F.; Soriano, V. Gastrointestinal leishmaniasis in HIV-infected patients: Report of five cases and review. Clin. Infect. Dis. 1994, 19, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Martínez, P.; de la Vega, E.; Laguna, F.; Soriano, V.; Puente, S.; Moreno, V.; Sentchordi, M.J.; García-Aguado, C.; González-Lahoz, J. Diagnosis of visceral leishmaniasis in HIV-infected individuals using peripheral blood smears. AIDS 1993, 7, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Laguna, F.; López-Vélez, R.; Pulido, F.; Salas, A.; Torre-Cisneros, J.; Torres, E.; Medrano, F.J.; Sanz, J.; Picó, G.; Gómez-Rodrigo, J.; et al. Treatment of visceral leishmaniasis in HIV-infected patients: A randomized trial comparing meglumine antimoniate with amphotericin B. AIDS 1999, 13, 1063–1069. [Google Scholar] [CrossRef] [PubMed]
- Laguna, F.; Videla, S.; Jiménez-Mejías, M.E.; Sirera, G.; Torre-Cisneros, J.; Ribera, E.; Prados, D.; Clotet, B.; Sust, M.; López-Vélez, R.; et al. Amphotericin B lipid complex versus meglumine antimoniate in the treatment of visceral leishmaniasis in patients infected with HIV: A randomized pilot study. J. Antimicrob. Chemother. 2003, 52, 464–468. [Google Scholar] [CrossRef] [PubMed]
- Monge-Maillo, B.; López-Vélez, R. Treatment options for visceral leishmaniasis and HIV coinfection. AIDS Rev. 2016, 18, 32–43. [Google Scholar] [PubMed]
- Treviño, A.; Soriano, V.; Rodríguez, C.; Arredondo, M.; Rivas, P.; Herrero-Mendoza, D.; Parra, P.; del Romero, J.; Anta, L.; Puente, S.; et al. Changing rate of non-B subtypes and coinfection with hepatitis B/C viruses in newly diagnosed HIV type 1 individuals in Spain. AIDS Res. Hum. Retrovir. 2011, 27, 633–638. [Google Scholar] [CrossRef] [PubMed]
- De Mendoza, C.; Garrido, C.; Poveda, E.; Corral, A.; Zahonero, N.; Treviño, A.; Anta, L.; Soriano, V. Changes in drug resistance patterns following the introduction of HIV type 1 non-B subtypes in Spain. AIDS Res. Hum. Retrovir. 2009, 25, 967–972. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Gutiérrez, M.; García-Lerma, G.; Aguilera, O.; Mas, A.; Bravo, R.; Pérez-Labad, M.L.; Baquero, M.; González-Lahoz, J. First case of HIV-1 group O infection in Spain. Vox Sang. 1996, 71, 66. [Google Scholar] [CrossRef] [PubMed]
- Rodes, B.; de Mendoza, C.; Rodgers, M.; Newell, A.; Jimenez, V.; Lopez-Brugada, R.M.; Soriano, V. Treatment response and drug resistance in patients infected with HIV type 1 group O viruses. AIDS Res. Hum. Retrovir. 2005, 21, 602–607. [Google Scholar] [CrossRef] [PubMed]
- Briz, V.; Garrido, C.; Poveda, E.; Morello, J.; Barreiro, P.; de Mendoza, C.; Soriano, V. Raltegravir and etravirine are active against HIV type 1 group O. AIDS Res. Hum. Retrovir. 2009, 25, 225–227. [Google Scholar] [CrossRef] [PubMed]
- Treviño, A.; Caballero, E.; de Mendoza, C.; Aguilera, A.; Pirón, M.; Soriano, V.; Spanish HIV-2/HTLV Study Group. The burden of neglected HIV-2 and HTLV-1 infections in Spain. AIDS Rev. 2015, 17, 212–219. [Google Scholar]
- Soriano, V.; Tor, J.; Ribera, A.; Clotet, B.; Muga, R.; Pauplana, M.; Fernandez, J.L.; Martin, J.; Foz, M. HIV-1, HIV-2 and HTLV-I infection in high-risk groups in Spain. AIDS 1989, 3, 615–616. [Google Scholar] [PubMed]
- De Mendoza, C.; Cabezas, T.; Caballero, E.; Requena, S.; Amengual, M.J.; Peñaranda, M.; Sáez, A.; Tellez, R.; Lozano, A.B.; Treviño, A.; et al. HIV-2 epidemic in Spain: Challenges and missing opportunities. AIDS 2017, 31, 1353–1364. [Google Scholar] [CrossRef] [PubMed]
- Treviño, A.; Cabezas, T.; Lozano, A.; García-Delgado, R.; Force, L.; Fernández-Montero, J.M.; de Mendoza, C.; Caballero, E.; Soriano, V. Dolutegravir for the treatment of HIV-2 infection. J. Clin. Virol. 2015, 64, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Cilla, G.; Rodés, B.; Pérez-Trallero, E.; Arrizabalaga, J.; Soriano, V. Molecular evidence of homosexual transmission of HIV type 2 in Spain. AIDS Res. Hum. Retrovir. 2001, 17, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Rodés, B.; Toro, C.; Jiménez, V.; Soriano, V. Viral response to antiretroviral therapy in a patient coinfected with HIV type 1 and type 2. Clin. Infect. Dis. 2005, 41, e19–e21. [Google Scholar] [CrossRef] [PubMed]
- Poveda, E.; Anta, L.; Blanco, J.L.; Casado, J.L.; Gutiérrez, F.; García, F.; Gómez-Sirvent, J.L.; Iribarren, J.A.; Soriano, V.; de Mendoza, C. Resistance Platform of the Spanish AIDS Research Network (ResRIS). Drug resistance mutations in HIV-infected patients in the Spanish drug resistance database failing tipranavir and darunavir therapy. Antimicrob. Agents Chemother. 2010, 54, 3018–3020. [Google Scholar] [CrossRef] [PubMed]
- Poveda, E.; Anta, L.; Blanco, J.L.; Pérez-Elías, M.J.; García, F.; Leal, M.; Ribera, E.; Gutiérrez, F.; Soriano, V.; de Mendoza, C. Resistance Platform of the Spanish AIDS Research Network (ResRIS). Etravirine resistance associated mutations in HIV-infected patients failing efavirenz or nevirapine in the Spanish antiretroviral resistance database. AIDS 2010, 24, 469–471. [Google Scholar] [CrossRef] [PubMed]
- Betancor, G.; Garriga, C.; Puertas, M.C.; Nevot, M.; Anta, L.; Blanco, J.L.; Pérez-Elías, M.J.; de Mendoza, C.; Martínez, M.A.; Martinez-Picado, J.; et al. Clinical, virological and biochemical evidence supporting the association of HIV-1 reverse transcriptase polymorphism R284K and thymidine analogue resistance mutations M41L, L210W and T215Y in patients failing tenofovir/emtricitabine therapy. Retrovirology 2012, 9, 68. [Google Scholar] [CrossRef] [PubMed]
- Anta, L.; Blanco, J.L.; Llibre, J.M.; García, F.; Pérez-Elías, M.J.; Aguilera, A.; Pérez-Romero, P.; Caballero, E.; Vidal, C.; Cañizares, A.; et al. Resistance to the most recent protease and non-nucleoside reverse transcriptase inhibitors across HIV-1 non-B subtypes. J. Antimicrob. Chemother. 2013, 68, 1994–2002. [Google Scholar] [CrossRef] [PubMed]
- Anta, L.; Llibre, J.M.; Poveda, E.; Blanco, J.L.; Alvarez, M.; Pérez-Elías, M.J.; Aguilera, A.; Caballero, E.; Soriano, V.; de Mendoza, C. Resistance Platform of the Spanish AIDS Research Network. Rilpivirine resistance mutations in HIV patients failing non-nucleoside reverse transcriptase inhibitor-based therapies. AIDS 2013, 27, 81–85. [Google Scholar] [CrossRef] [PubMed]
- De Mendoza, C.; Anta, L.; García, F.; Pérez-Elías, M.J.; Gutiérrez, F.; Llibre, J.M.; Menéndez-Arias, L.; Dalmau, D.; Soriano, V. HIV-1 genotypic drug resistance interpretation rules - 2009 Spanish guidelines. AIDS Rev. 2009, 11, 39–51. [Google Scholar] [PubMed]
- De Mendoza, C.; Rodriguez, C.; Eiros, J.M.; Colomina, J.; Garcia, F.; Leiva, P.; Torre-Cisneros, J.; Agüero, J.; Pedreira, J.; Viciana, I.; et al. Antiretroviral recommendations may influence the rate of transmission of drug-resistant HIV type 1. Clin. Infect. Dis. 2005, 41, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Monge, S.; Guillot, V.; Alvarez, M.; Peña, A.; Viciana, P.; García-Bujalance, S.; Pérez-Elias, M.J.; Iribarren, J.A.; Gutiérrez, F.; Itziar-Casado, M.; et al. Analysis of transmitted drug resistance in Spain in the years 2007–2010 documents a decline in mutations to the non-nucleoside drug class. Clin. Microbiol. Infect. 2012, 18, E485–E490. [Google Scholar] [CrossRef] [PubMed]
- Monge, S.; Guillot, V.; Alvarez, M.; Chueca, N.; Stella, N.; Peña, A.; Delgado, R.; Córdoba, J.; Aguilera, A.; Vidal, C.; et al. Clinically relevant transmitted drug resistance to first line antiretroviral drugs and implications for recommendations. PLoS ONE 2014, 9, e90710. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Novoa, S.; Barreiro, P.; Rendón, A.; Jiménez-Nacher, I.; González-Lahoz, J.; Soriano, V. Influence of 516G>T polymorphisms at the gene encoding the CYP450-2B6 isoenzyme on efavirenz plasma concentrations in HIV-infected subjects. Clin. Infect. Dis. 2005, 40, 1358–1361. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Nóvoa, S.; Labarga, P.; D’Avolio, A.; Barreiro, P.; Albalate, M.; Vispo, E.; Solera, C.; Siccardi, M.; Bonora, S.; Di Perri, G.; Soriano, V. Impairment in kidney tubular function in patients receiving tenofovir is associated with higher tenofovir plasma concentrations. AIDS 2010, 24, 1064–1066. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Nóvoa, S.; Labarga, P.; Soriano, V.; Egan, D.; Albalater, M.; Morello, J.; Cuenca, L.; González-Pardo, G.; Khoo, S.; Back, D.; et al. Predictors of kidney tubular dysfunction in HIV-infected patients treated with tenofovir: A pharmacogenetic study. Clin. Infect. Dis. 2009, 48, e108–e116. [Google Scholar] [CrossRef] [PubMed]
- Barreiro, P.; Fernández-Montero, J.V.; de Mendoza, C.; Labarga, P.; Soriano, V. Pharmacogenetics of antiretroviral therapy. Expert Opin. Drug Metab. Toxicol. 2014, 10, 1119–1130. [Google Scholar] [CrossRef] [PubMed]
- Vidal, F.; Gutiérrez, F.; Gutiérrez, M.; Olona, M.; Sánchez, V.; Mateo, G.; Peraire, J.; Viladés, C.; Veloso, S.; López-Dupla, M.; et al. Pharmacogenetics of adverse effects due to antiretroviral drugs. AIDS Rev. 2010, 12, 15–30. [Google Scholar] [PubMed]
- Martinez, E.; Mocroft, A.; García-Viejo, M.A.; Pérez-Cuevas, J.B.; Blanco, J.L.; Mallolas, J.; Bianchi, L.; Conget, I.; Blanch, J.; Phillips, A.; et al. Risk of lipodystrophy in HIV-1-infected patients treated with protease inhibitors: A prospective cohort study. Lancet 2001, 357, 592–598. [Google Scholar] [CrossRef]
- Moreno, S.; Miralles, C.; Negredo, E.; Domingo, P.; Estrada, V.; Gutiérrez, F.; Lozano, F.; Martínez, E. Disorders of body fat distribution in HIV-1-infected patients. AIDS Rev. 2009, 11, 126–134. [Google Scholar] [PubMed]
- Ribera, E.; Larrousse, M.; Curran, A.; Negredo, E.; Clotet, B.; Estrada, V.; Sanz, J.; Berenguer, J.; Rubio, R.; Pulido, F.; et al. Impact of switching from zidovudine/lamivudine to tenofovir/emtricitabine on lipoatrophy: The RECOMB study. HIV Med. 2013, 14, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Domingo, P.; Gutierrez, M.; Gallego-Escuredo, J.M.; Torres, F.; Mateo, G.; Villarroya, J.; de los Santos, I.; Domingo, J.C.; Villarroya, F.; Del Rio, L.; et al. Effects of switching from stavudine to raltegravir on subcutaneous adipose tissue in HIV-infected patients with HIV/HAART-associated lipodystrophy syndrome (HALS). A clinical and molecular study. PLoS ONE 2014, 9, e89088. [Google Scholar] [CrossRef] [PubMed]
- Domingo, P.; Estrada, V.; López-Aldeguer, J.; Villaroya, F.; Martínez, E. Fat redistribution syndromes associated with HIV-1 infection and combination antiretroviral therapy. AIDS Rev. 2012, 14, 112–123. [Google Scholar] [PubMed]
- Serrano-Villar, S.; Gutiérrez, F.; Miralles, C.; Berenguer, J.; Rivero, A.; Martínez, E.; Moreno, S. HIV as a chronic disease: Evaluation and management of non-AIDS-defining conditions. Open Forum Infect. Dis. 2016, 3, ofw097. [Google Scholar] [CrossRef] [PubMed]
- Martínez, E.; Arnaiz, J.A.; Podzamczer, D.; Dalmau, D.; Ribera, E.; Domingo, P.; Knobel, H.; Riera, M.; Pedrol, E.; Force, L.; et al. Substitution of nevirapine, efavirenz, or abacavir for protease inhibitors in patients with HIV infection. N. Engl. J. Med. 2003, 349, 1036–1046. [Google Scholar] [CrossRef] [PubMed]
- Negredo, E.; Ribalta, J.; Paredes, R.; Ferré, R.; Sirera, G.; Ruiz, L.; Salazar, J.; Reiss, P.; Masana, L.; Clotet, B. Reversal of atherogenic lipoprotein profile in HIV-1 infected patients with lipodystrophy after replacing protease inhibitors by nevirapine. AIDS 2002, 16, 1383–1389. [Google Scholar] [CrossRef] [PubMed]
- Negredo, E.; Miró, O.; Rodríguez-Santiago, B.; Garrabou, G.; Estany, C.; Masabeu, A.; Force, L.; Barrufet, P.; Cucurull, J.; Domingo, P.; et al. Improvement of mitochondrial toxicity in patients receiving a nucleoside reverse-transcriptase inhibitor-sparing strategy: Results from the Multicenter Study with Nevirapine and Kaletra (MULTINEKA). Clin. Infect. Dis. 2009, 49, 892–900. [Google Scholar] [CrossRef] [PubMed]
- Podzamczer, D.; Andrade-Villanueva, J.; Clotet, B.; Taylor, S.; Rockstroh, J.; Reiss, P.; Domingo, P.; Gellermann, H.; Cairns, V.; Soriano, V. Lipid profiles for nevirapine vs. atazanavir/ritonavir, both combined with tenofovir disoproxil fumarate and emtricitabine over 48 weeks, in treatment-naïve HIV-1-infected patients (the ARTEN study). HIV Med. 2011, 12, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Martínez, E.; D’Albuquerque, P.; Llibre, J.; Gutierrez, F.; Podzamczer, D.; Antela, A.; Berenguer, J.; Domingo, P.; Moreno, X.; Perez, I.; et al. Changes in cardiovascular biomarkers in HIV-infected patients switching from ritonavir-boosted protease inhibitors to raltegravir. AIDS 2012, 26, 2315–2326. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Barreiro, P.; Sherman, K. The changing epidemiology of liver disease in HIV patients. AIDS Rev. 2013, 15, 25–31. [Google Scholar] [PubMed]
- Altice, F.; Kamarulzaman, A.; Soriano, V.; Friedland, G. Treatment of medical, psychiatric, and substance use comorbidities in people infected with HIV who use drugs. Lancet 2010, 376, 367–387. [Google Scholar] [CrossRef]
- Soriano, V.; Gallego, L. Treating hepatitis C in injection drug users. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 568–569. [Google Scholar] [CrossRef] [PubMed]
- Castellares, C.; Barreiro, P.; Martín-Carbonero, L.; Labarga, P.; Vispo, M.E.; Casado, R.; Galindo, L.; García-Gascó, P.; García-Samaniego, J.; Soriano, V. Liver cirrhosis in HIV-infected patients: Prevalence, aetiology and clinical outcome. J. Viral Hepat. 2008, 15, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; García-Samaniego, J.; Bravo, R.; González, J.; Castro, A.; Castilla, J.; Martínez-Odriozola, P.; Colmenero, M.; Carballo, E.; Suárez, D.; et al. Interferon alpha for the treatment of chronic hepatitis C in patients infected with Human Immunodeficiency Virus. Clin. Infect. Dis. 1996, 23, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Olmeda, M.; Núñez, M.; Romero, M.; González, J.; Castro, A.; Arribas, J.R.; Pedreira, J.; Barreiro, P.; García-Samaniego, J.; Martín-Carbonero, L.; et al. Pegylated IFN-alpha2b plus ribavirin as therapy for chronic hepatitis C in HIV-infected patients. AIDS 2003, 17, 1023–1028. [Google Scholar] [CrossRef] [PubMed]
- Núñez, M.; Miralles, C.; Berdún, M.A.; Losada, E.; Aguirrebengoa, K.; Ocampo, A.; Arazo, P.; Cervantes, M.; de los Santos, I.; San Joaquín, I.; et al. Role of weight-based ribavirin dosing and extended duration of therapy in chronic hepatitis C in HIV-infected patients: The PRESCO trial. AIDS Res. Hum. Retrovir. 2007, 23, 972–982. [Google Scholar] [CrossRef] [PubMed]
- Labarga, P.; Barreiro, P.; da Silva, A.; Guardiola, J.M.; Rubio, R.; Aguirrebengoa, K.; Miralles, P.; Portu, J.; Téllez, M.J.; Morano, L.; et al. Comparison of high ribavirin induction versus standard ribavirin dosing, plus peginterferon-α for the treatment of chronic hepatitis C in HIV-infected patients: The PERICO trial. J. Infect. Dis. 2012, 206, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Mocroft, A.; Peters, L.; Rockstroh, J.; Antunes, F.; Kirkby, N.; de Wit, S.; d’Arminio Monforte, A.; Flisiak, R.; Lundgren, J. EuroSIDA: Predictors of hepatitis B virus genotype and viraemia in HIV-infected patients with chronic hepatitis B in Europe. J. Antimicrob. Chemother. 2010, 65, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Mocroft, A.; Rockstroh, J.; Ledergerber, B.; Knysz, B.; Chaplinskas, S.; Peters, L.; Karlsson, A.; Katlama, C.; Toro, C.; et al. Spontaneous viral clearance, viral load, and genotype distribution of hepatitis C virus (HCV) in HIV-infected patients with anti-HCV antibodies in Europe. J. Infect. Dis. 2008, 198, 1337–1344. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Grint, D.; d’Arminio Monforte, A.; Horban, A.; Leen, C.; Poveda, E.; Antunes, F.; de Wit, S.; Lundgren, J.; Rockstroh, J.; et al. Hepatitis delta in HIV-infected individuals in Europe. AIDS 2011, 25, 1987–1992. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Puoti, M.; Peters, M.; Benhamou, Y.; Sulkowski, M.; Zoulim, F.; Mauss, S.; Rockstroh, J. Care of HIV patients with chronic hepatitis B: Updated recommendations from the HIV-Hepatitis B Virus International Panel. AIDS 2008, 22, 1399–1410. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Puoti, M.; Sulkowski, M.; Cargnel, A.; Benhamou, Y.; Peters, M.; Mauss, S.; Bräu, N.; Hatzakis, A.; Pol, S.; Rockstroh, J. Care of patients coinfected with HIV and hepatitis C virus: 2007 updated recommendations from the HCV-HIV International Panel. AIDS 2007, 21, 1073–1089. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Puoti, M.; Garcia-Gascó, P.; Rockstroh, J.; Benhamou, Y.; Barreiro, P.; McGovern, B. Antiretroviral drugs and liver injury. AIDS. 2008, 22, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Labarga, P.; Soriano, V.; Vispo, M.E.; Pinilla, J.; Martin-Carbonero, L.; Castellares, C.; Casado, R.; Maida, I.; Garcia-Gasco, P.; Barreiro, P. Hepatotoxicity of antiretroviral drugs is reduced after successful treatment of chronic hepatitis C in HIV-infected patients. J. Infect. Dis. 2007, 196, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Rallón, N.; Naggie, S.; Benito, J.M.; Medrano, J.; Restrepo, C.; Goldstein, D.; Shianna, K.; Vispo, E.; Thompson, A.; McHutchison, J.; Soriano, V. Association of a single nucleotide polymorphism near the interleukin-28B gene with response to hepatitis C therapy in HIV/hepatitis C virus-coinfected patients. AIDS 2010, 24, F23–F29. [Google Scholar] [CrossRef] [PubMed]
- Barreiro, P.; Pineda, J.A.; Rallon, N.; Naggie, S.; Martín-Carbonero, L.; Neukam, K.; Rivero, A.; Benito, J.M.; Caruz, A.; Vispo, E.; et al. Influence of interleukin-28B single-nucleotide polymorphisms on progression to liver cirrhosis in HIV-HCV-coinfected patients receiving antiretroviral therapy. J. Infect. Dis. 2011, 203, 1629–1636. [Google Scholar] [CrossRef] [PubMed]
- Barreiro, P.; Martín-Carbonero, L.; Núñez, M.; Rivas, P.; Morente, A.; Simarro, N.; Labarga, P.; González-Lahoz, J.; Soriano, V. Predictors of liver fibrosis in HIV-infected patients with chronic hepatitis C virus (HCV) infection: Assessment using transient elastometry and the role of HCV genotype 3. Clin. Infect. Dis. 2006, 42, 1032–1039. [Google Scholar] [CrossRef] [PubMed]
- Medrano, J.; Neukam, K.; Rallón, N.; Rivero, A.; Resino, S.; Naggie, S.; Caruz, A.; Calvino, A.; Macías, J.; Benito, J.M.; et al. Modeling the probability of sustained virological response to therapy with pegylated interferon plus ribavirin in patients coinfected with hepatitis C virus and HIV. Clin. Infect. Dis. 2010, 51, 1209–1216. [Google Scholar] [CrossRef] [PubMed]
- Sulkowski, M.; Pol, S.; Mallolas, J.; Fainboim, H.; Cooper, C.; Slim, J.; Rivero, A.; Mak, C.; Thompson, S.; Howe, A.; et al. Boceprevir versus placebo with pegylated interferon alfa-2b and ribavirin for treatment of hepatitis C virus genotype 1 in patients with HIV: A randomised, double-blind, controlled phase 2 trial. Lancet Infect. Dis. 2013, 13, 597–605. [Google Scholar] [CrossRef]
- Sulkowski, M.; Sherman, K.; Dieterich, D.; Bsharat, M.; Mahnke, L.; Rockstroh, J.; Gharakhanian, S.; McCallister, S.; Henshaw, J.; Girard, P.M.; et al. Combination therapy with telaprevir for chronic HCV-1 in patients with HIV. Ann. Intern. Med. 2013, 159, 86–96. [Google Scholar] [PubMed]
- Dieterich, D.; Nelson, M.; Soriano, V.; Arastéh, K.; Guardiola, J.M.; Rockstroh, J.; Bhagani, S.; Laguno, M.; Tural, C.; Ingiliz, P.; et al. Faldaprevir and pegylated interferon α-2a/ribavirin in individuals co-infected with hepatitis C virus genotype-1 and HIV. AIDS 2015, 29, 571–581. [Google Scholar] [PubMed]
- Soriano, V.; Peters, M.; Zeuzem, S. New Therapies for Hepatitis C Virus Infection. Clin. Infect. Dis. 2009, 48, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Plaza, Z.; Aguilera, A.; Mena, A.; Vispo, E.; Sierra-Enguita, R.; Tomé, S.; Pedreira, J.; Rodriguez, C.; Barreiro, P.; del Romero, J.; et al. Influence of HIV infection on response to tenofovir in patients with chronic hepatitis B. AIDS 2013, 27, 2219–2224. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Montero, J.V.; Vispo, E.; Barreiro, P.; Sierra-Enguita, R.; de Mendoza, C.; Labarga, P.; Soriano, V. Hepatitis delta is a major determinant of liver decompensation events and death in HIV-infected patients. Clin. Infect. Dis. 2014, 58, 1549–1553. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Sherman, K.; Barreiro, P. Hepatitis delta and HIV infection. AIDS 2017, 31, 875–884. [Google Scholar] [CrossRef] [PubMed]
- Macías, J.; Pineda, J.A.; Real, L. Non-alcoholic fatty liver disease in HIV infection. AIDS Rev. 2017, 19, 35–46. [Google Scholar] [PubMed]
- Miro, J.M.; Montejo, M.; Castells, L.; Rafecas, A.; Moreno, S.; Agüero, F.; Abradelo, M.; Miralles, P.; Torre-Cisneros, J.; Pedreira, J.; et al. Outcome of HCV/HIV-coinfected liver transplant recipients: A prospective and multicenter cohort study. Am. J. Transplant. 2012, 12, 1866–1876. [Google Scholar] [CrossRef] [PubMed]
- Agüero, F.; Forner, A.; Manzardo, C.; Valdivieso, A.; Blanes, M.; Barcena, R.; Rafecas, A.; Castells, L.; Abradelo, M.; Torre-Cisneros, J.; et al. HIV infection does not worsen prognosis of liver transplantation for hepatocellular carcinoma. Hepatology 2016, 63, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Agüero, F.; Forner, A.; Valdivieso, A.; Blanes, M.; Barcena, R.; Manzardo, C.; Rafecas, A.; Castells, L.; Abradelo, M.; Barrera-Baena, P.; et al. HIV-infected liver transplant recipients with incidental hepatocellular carcinoma: A prospective multicenter nationwide cohort study. Liver Transplant. 2017, 23, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Soriano, V.; Labarga, P.; de Mendoza, C.; Fernández-Montero, J.V.; Esposito, I.; Benítez-Gutiérrez, L.; Peña, J.M.; Barreiro, P. New hepatitis C therapies for special patient populations. Expert Opin. Pharmacother. 2016, 17, 217–229. [Google Scholar] [CrossRef] [PubMed]
- Maida, I.; Núñez, M.; Ríos, M.J.; Martín-Carbonero, L.; Sotgiu, G.; Toro, C.; Rivas, P.; Barreiro, P.; Mura, M.S.; Babudieri, S.; et al. Severe liver disease associated with prolonged exposure to antiretroviral drugs. J. Acquir. Immune Defic. Syndr. 2006, 42, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Maida, I.; Garcia-Gasco, P.; Sotgiu, G.; Rios, M.J.; Vispo, M.E.; Martin-Carbonero, L.; Barreiro, P.; Mura, M.S.; Babudieri, S.; Albertos, S.; et al. Antiretroviral-associated portal hypertension: A new clinical condition? Prevalence, predictors and outcome. Antivir. Ther. 2008, 13, 103–107. [Google Scholar] [PubMed]
- Vispo, E.; Moreno, A.; Maida, I.; Barreiro, P.; Cuevas, A.; Albertos, S.; Soriano, V. Non-cirrhotic portal hypertension in HIV-infected patients: Unique clinical and pathological findings. AIDS 2010, 24, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Vispo, E.; Maida, I.; Barreiro, P.; Moreno, V.; Soriano, V. Upper gastrointestinal bleeding may unmask didanosine-associated portal hepatopathy in HIV/HCV co-infected patients. HIV Clin. Trials 2008, 9, 440–444. [Google Scholar] [CrossRef] [PubMed]
- Vispo, E.; Cevik, M.; Rockstroh, J.; Barreiro, P.; Nelson, M.; Scourfield, A.; Boesecke, C.; Wasmuth, J.C.; Soriano, V.; European Network of Clinical Trials (NEAT). Genetic determinants of idiopathic non-cirrhotic portal hypertension in HIV-infected patients. Clin. Infect. Dis. 2013, 56, 1117–1122. [Google Scholar] [CrossRef] [PubMed]
- Barreiro, P.; del Romero, J.; Leal, M.; Hernando, V.; Asencio, R.; de Mendoza, C.; Labarga, P.; Núñez, M.; Ramos, J.T.; González-Lahoz, J.; et al. Natural pregnancies in HIV-serodiscordant couples receiving successful antiretroviral therapy. J. Acquir. Immune Defic. Syndr. 2006, 43, 324–326. [Google Scholar] [CrossRef] [PubMed]
- Barreiro, P.; Castilla, J.A.; Labarga, P.; Soriano, V. Is natural conception a valid option for HIV-serodiscordant couples? Hum. Reprod. 2007, 22, 2353–2358. [Google Scholar] [CrossRef] [PubMed]
- Del Romero, J.; Baza, M.B.; Río, I.; Jerónimo, A.; Vera, M.; Hernando, V.; Rodríguez, C.; Castilla, J. Natural conception in HIV-serodiscordant couples with the infected partner in suppressive antiretroviral therapy: A prospective cohort study. Medicine 2016, 95, e4398. [Google Scholar] [CrossRef] [PubMed]
- Das, M.; Chu, P.L.; Santos, G.; Scheer, S.; Vittinghoff, E.; McFarland, W.; Colfax, G. Decreases in community viral load are accompanied by reductions in new HIV infections in San Francisco. PLoS ONE 2010, 5, e11068. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Montero, J.V.; Barreiro, P.; Del Romero, J.; Soriano, V. Antiretroviral drugs for pre-exposure prophylaxis of HIV infection. AIDS Rev. 2012, 14, 54–61. [Google Scholar] [PubMed]
- Alaei, K.; Paynter, C.; Juan, S.; Alaei, A. Using PrEP, losing condoms? PrEP promotion may undermine safe sex. AIDS 2016, 30, 2753–2756. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Sancho, A.; Barreiro, P.; Castellares, C.; Labarga, P.; Ramos, B.; Garcia-Samaniego, J.; Gutiérrez, M.; Soriano, V. Outbreak of syphilis, but not of acute hepatitis, C.; among HIV-infected homosexual men in Madrid. HIV Clin. Trials 2007, 8, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, C.; Plaza, Z.; Vispo, E.; de Mendoza, C.; Barreiro, P.; Fernández-Montero, J.V.; Labarga, P.; Poveda, E.; Soriano, V. Scaling up epidemics of acute hepatitis C and syphilis in HIV-infected men who have sex with men in Spain. Liver Int. 2013, 33, 1357–1362. [Google Scholar] [CrossRef] [PubMed]
- Boyd, M.A.; Donovan, B.; Prestage, G.; Chen, M.; Petoumenos, K.; Gray, R.; Guy, R.; Rogers, G.; Bourne, C.; Klausner, J. Is it time to rethink syphilis control? Clin. Infect. Dis. 2015, 60, 325–326. [Google Scholar] [CrossRef] [PubMed]
- Barreiro, P. More pre-exposure prophylaxis for rising HIV infection? AIDS Rev. 2014, 16, 118–119. [Google Scholar] [PubMed]
- Soriano, V. Transmission of multi-drug resistant HIV-1 despite antiretroviral prophylaxis. AIDS Rev. 2017, 19, 54–55. [Google Scholar] [PubMed]
- Núñez, M.; Soriano, V.; Martín-Carbonero, L.; Barrios, A.; Barreiro, P.; Blanco, F.; García-Benayas, T.; González-Lahoz, J. SENC (Spanish efavirenz vs. nevirapine comparison) trial: A randomized, open-label study in HIV-infected naive individuals. HIV Clin. Trials 2002, 3, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Podzamczer, D.; Ferrer, E.; Consiglio, E.; Gatell, J.M.; Perez, P.; Perez, J.L.; Luna, E.; González, A.; Pedrol, E.; Lozano, L.; et al. A randomized clinical trial comparing nelfinavir or nevirapine associated to zidovudine/ lamivudine in HIV-infected naive patients (the Combine Study). Antivir. Ther. 2002, 7, 81–90. [Google Scholar] [PubMed]
- Sánchez-Conde, M.; Palacios, R.; Sanz, J.; Rodríguez-Novoa, S.; Rivas, P.; Santos, J.; Sola, J.; Asensi, V.; de Mendoza, C.; Estrada, V.; et al. Efficacy and safety of a once daily regimen with efavirenz, lamivudine, and didanosine, with and without food, as initial therapy for HIV Infection: The ELADI study. AIDS Res. Hum. Retrovir. 2007, 23, 1237–1241. [Google Scholar] [CrossRef] [PubMed]
- Berenguer, J.; González, J.; Ribera, E.; Domingo, P.; Santos, J.; Miralles, P.; Ribas, M.A.; Asensi, V.; Gimeno, J.L.; Pérez-Molina, J.A.; et al. Didanosine, lamivudine, and efavirenz versus zidovudine, lamivudine, and efavirenz for the initial treatment of HIV type 1 infection: Final analysis (48 weeks) of a prospective, randomized, non-inferiority clinical trial, GESIDA 3903. Clin. Infect. Dis. 2008, 47, 1083–1092. [Google Scholar] [CrossRef] [PubMed]
- Echeverria, P.; Negredo, E.; Carosi, G.; Gálvez, J.; Gómez, J.L.; Ocampo, A.; Portilla, J.; Prieto, A.; López, J.C.; Rubio, R.; et al. Similar antiviral efficacy and tolerability between efavirenz and lopinavir/ritonavir, administered with abacavir/lamivudine (Kivexa), in antiretroviral-naive patients: A 48-week, multicentre, randomized study (Lake Study). Antivir. Res. 2010, 85, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Miró, J.M.; Manzardo, C.; Pich, J.; Domingo, P.; Ferrer, E.; Arribas, J.R.; Ribera, E.; Arrizabalaga, J.; Loncá, M.; Cruceta, A.; et al. Immune reconstitution in severely immunosuppressed antiretroviral-naive HIV type 1-infected patients using a non-nucleoside reverse transcriptase inhibitor-based or a boosted protease inhibitor-based antiretroviral regimen: Three-year results (The Advanz Trial): A randomized, controlled trial. AIDS Res. Hum. Retrovir. 2010, 26, 747–757. [Google Scholar] [PubMed]
- Soriano, V.; Arastéh, K.; Migrone, H.; Lutz, T.; Opravil, M.; Andrade-Villanueva, J.; Antunes, F.; Di Perri, G.; Podzamczer, D.; Taylor, S.; et al. Nevirapine versus atazanavir/ritonavir, each combined with tenofovir disoproxil fumarate/emtricitabine, in antiretroviral-naive HIV-1 patients: The ARTEN trial. Antivir. Ther. 2011, 16, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Cahn, P.; Andrade-Villanueva, J.; Arribas, J.R.; Gatell, J.M.; Lama, J.R.; Norton, M.; Patterson, P.; Sierra Madero, J.; Sued, O.; Figueroa, M.I.; et al. Dual therapy with lopinavir and ritonavir plus lamivudine versus triple therapy with lopinavir and ritonavir plus two nucleoside reverse transcriptase inhibitors in antiretroviral-therapy-naive adults with HIV-1 infection: 48 week results of the randomised, open label, non-inferiority GARDEL trial. Lancet Infect. Dis. 2014, 14, 572–580. [Google Scholar] [PubMed]
- Clotet, B.; Feinberg, J.; van Lunzen, J.; Khuong-Josses, M.A.; Antinori, A.; Dumitru, I.; Pokrovskiy, V.; Fehr, J.; Ortiz, R.; Saag, M.; et al. Once-daily dolutegravir versus darunavir plus ritonavir in antiretroviral-naive adults with HIV-1 infection (FLAMINGO): 48 week results from the randomised open-label phase 3b study. Lancet 2014, 383, 2222–2231. [Google Scholar] [CrossRef]
- Miro, J.M.; Manzardo, C.; Ferrer, E.; Loncà, M.; Guardo, A.C.; Podzamczer, D.; Domingo, P.; Curran, A.; Clotet, B.; Cruceta, A.; et al. Immune reconstitution in severely immunosuppressed antiretroviral-naive HIV-1-infected patients starting efavirenz, lopinavir-ritonavir, or atazanavir-ritonavir plus tenofovir/emtricitabine: Final 48-week results (the Advanz-3 trial). J. Acquir. Immune Defic. Syndr. 2015, 69, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Martinez, E.; Gonzalez-Cordon, A.; Ferrer, E.; Domingo, P.; Negredo, E.; Gutierrez, F.; Portilla, J.; Curran, A.; Podzamczer, D.; Ribera, E.; et al. Differential body composition effects of protease inhibitors recommended for initial treatment of HIV infection: A randomized clinical trial. Clin. Infect. Dis. 2015, 60, 811–820. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Picado, J.; Negredo, E.; Ruiz, L.; Shintani, A.; Fumaz, C.R.; Zala, C.; Domingo, P.; Vilaró, J.; Llibre, J.M.; Viciana, P.; et al. Alternation of antiretroviral drug regimens for HIV infection. A randomized, controlled trial. Ann. Intern. Med. 2003, 139, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Barrios, A.; Negredo, E.; Domingo, P.; Estrada, V.; Labarga, P.; Asensi, V.; Morales, D.; Santos, J.; Clotet, B.; Soriano, V. Simplification therapy with once-daily didanosine, tenofovir and efavirenz in HIV-1-infected adults with viral suppression receiving a more complex antiretroviral regimen: Final results of the EFADITE trial. Antivir. Ther. 2005, 10, 825–832. [Google Scholar] [PubMed]
- Negredo, E.; Moltó, J.; Burger, D.; Côté, H.; Miró, O.; Ribalta, J.; Martínez, E.; Puig, J.; Ruiz, L.; Salazar, J.; et al. Lopinavir/ritonavir plus nevirapine as a nucleoside-sparing approach in antiretroviral-experienced patients (NEKA study). J. Acquir. Immune Defic. Syndr. 2005, 38, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Arribas, J.R.; Pulido, F.; Delgado, R.; Lorenzo, A.; Miralles, P.; Arranz, A.; González, J.; Cepeda, C.; Hervás, R.; Paño, J.R.; et al. Lopinavir/ritonavir as single-drug therapy for maintenance of HIV-1 viral suppression: 48-week results of a randomized, controlled, open-label, proof-of-concept pilot clinical trial (OK Study). J. Acquir. Immune Defic. Syndr. 2005, 40, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Clotet, B.; Bellos, N.; Molina, J.M.; Cooper, D.; Goffard, J.C.; Lazzarin, A.; Wöhrmann, A.; Katlama, C.; Wilkin, T.; Haubrich, R.; et al. Efficacy and safety of darunavir-ritonavir at week 48 in treatment-experienced patients with HIV-1 infection in POWER 1 and 2: A pooled subgroup analysis of data from two randomised trials. Lancet 2007, 369, 1169–1178. [Google Scholar] [CrossRef]
- Soriano, V.; García-Gasco, P.; Vispo, E.; Ruiz-Sancho, A.; Blanco, F.; Martín-Carbonero, L.; Rodríguez-Novoa, S.; Morello, J.; de Mendoza, C.; Rivas, P.; et al. Efficacy and safety of replacing lopinavir with atazanavir in HIV-infected patients with undetectable plasma viraemia: Final results of the SLOAT trial. J. Antimicrob. Chemother. 2008, 61, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Mallolas, J.; Podzamczer, D.; Milinkovic, A.; Domingo, P.; Clotet, B.; Ribera, E.; Gutiérrez, F.; Knobel, H.; Cosin, J.; Ferrer, E.; et al. Efficacy and safety of switching from boosted lopinavir to boosted atazanavir in patients with virological suppression receiving a LPV/r-containing HAART: The ATAZIP study. J. Acquir. Immune Defic. Syndr. 2009, 51, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Moltó, J.; Valle, M.; Santos, J.R.; Mothe, B.; Miranda, C.; Cedeño, S.; Negredo, E.; Yritia, M.; Videla, S.; Barbanoj, M.J.; et al. Treatment simplification to once daily darunavir/ritonavir guided by the darunavir inhibitory quotient in heavily pretreated HIV-infected patients. Antivir. Ther. 2010, 15, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Bernardino, J.I.; Pulido, F.; Martinez, E.; Arrizabalaga, J.; Domingo, P.; Portilla, J.; Ocampo, A.; Muñoz, J.; Torres, R.; Arribas, J. Switching to lopinavir/ritonavir with or without abacavir/lamivudine in lipoatrophic patients treated with zidovudine/abacavir/lamivudine. J. Antimicrob. Chemother. 2013, 68, 1373–1381. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.R.; Saumoy, M.; Curran, A.; Bravo, I.; Llibre, J.M.; Navarro, J.; Estany, C.; Podzamczer, D.; Ribera, E.; Negredo, E.; et al. The lipid-lowering effect of tenofovir/emtricitabine: A randomized, crossover, double-blind, placebo-controlled trial. Clin. Infect. Dis. 2015, 61, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Perez-Molina, J.A.; Rubio, R.; Rivero, A.; Pasquau, J.; Suárez-Lozano, I.; Riera, M.; Estébanez, M.; Santos, J.; Sanz-Moreno, J.; Troya, J.; et al. Dual treatment with atazanavir and ritonavir plus lamivudine versus triple treatment with atazanavir and ritonavir plus two nucleos[t]ides in patients who are HIV1 infected, on antiretroviral treatment, and virologically stable (SALT): 48 week results from a randomised, open-label, non-inferiority trial. Lancet Infect. Dis. 2015, 15, 775–784. [Google Scholar] [PubMed]
- Arribas, J.; Girard, P.; Landman, R.; Pich, J.; Mallolas, J.; Martínez-Rebollar, M.; Zamora, F.; Estrada, V.; Crespo, M.; Podzamczer, D.; et al. Dual treatment with lopinavir and ritonavir plus lamivudine versus triple treatment with lopinavir and ritonavir plus lamivudine or emtricitabine and a second nucleos(t)ide reverse transcriptase inhibitor for maintenance of HIV-1 viral suppression (OLE): A randomised, open-label, non-inferiority trial. Lancet Infect. Dis. 2015, 15, 785–792. [Google Scholar] [PubMed]
- Santos, J.R.; Llibre, J.M.; Bravo, I.; García-Rosado, D.; Cañadas, M.P.; Pérez-Álvarez, N.; Paredes, R.; Clotet, B.; Moltó, J. Efficacy and safety of treatment simplification to lopinavir/ritonavir or darunavir/ritonavir monotherapy: A randomized clinical trial. AIDS Res. Hum. Retrovir. 2016, 32, 452–455. [Google Scholar] [CrossRef] [PubMed]
- Negredo, E.; Estrada, V.; Domingo, P.; Gutiérrez, M.; Mateo, G.; Puig, J.; Bonjoch, A.; Ornelas, A.; Echeverría, P.; Estany, C.; et al. Switching from a ritonavir-boosted PI to dolutegravir as an alternative strategy in virologically suppressed HIV-infected individuals. J. Antimicrob. Chemother. 2017, 72, 844–849. [Google Scholar] [CrossRef] [PubMed]
- Llibre, J.M.; Hung, C.; Brinson, C.; Castelli, F.; Girard, P.M.; Kahl, L.; Blair, E.; Angelis, K.; Wynne, B.; Vandermeulen, K.; et al. Efficacy, safety, and tolerability of dolutegravir-rilpivirine for the maintenance of virological suppression in adults with HIV-1: Phase 3, randomised, non-inferiority SWORD-1 and SWORD-2 studies. Lancet 2018, 39, 839–849. [Google Scholar] [CrossRef]
- Real, L.; Herrero, R.; Rivero-Juárez, A.; Camacho, Á.; Macías, J.; Vic, S.; Soriano, V.; Viedma, S.; Guardiola, J.M.; Fibla, J.; et al. IFNL4 rs368234815 polymorphism is associated with innate resistance to HIV-1 infection. AIDS 2015, 29, 1895–1897. [Google Scholar] [CrossRef] [PubMed]
- Rodés, B.; Toro, C.; Paxinos, E.; Poveda, E.; Martinez-Padial, M.; Benito, J.M.; Jimenez, V.; Wrin, T.; Bassani, S.; Soriano, V. Differences in disease progression in a cohort of long-term non-progressors after more than 16 years of HIV-1 infection. AIDS 2004, 18, 1109–1116. [Google Scholar] [CrossRef] [PubMed]
- Noel, N.; Peña, R.; David, A.; Avettand-Fenoel, V.; Erkizia, I.; Jimenez, E.; Lecuroux, C.; Rouzioux, C.; Boufassa, F.; Pancino, G.; et al. Long-term spontaneous control of HIV-1 is related to low frequency of infected cells and inefficient viral reactivation. J. Virol. 2016, 90, 6148–6158. [Google Scholar] [CrossRef] [PubMed]
- Leon, A.; Perez, I.; Ruiz-Mateos, E.; Benito, J.M.; Leal, M.; Lopez-Galindez, C.; Rallon, N.; Alcami, J.; Lopez-Aldeguer, J.; Viciana, P.; et al. Rate and predictors of progression in elite and viremic HIV-1 controllers. AIDS 2016, 30, 1209–1220. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Molina, B.; Tarancon-Diez, L.; Hua, S.; Abad-Molina, C.; Rodriguez-Gallego, E.; Machmach, K.; Vidal, F.; Tural, C.; Moreno, S.; Goñi, J.M.; et al. Spanish AIDS Research Network HIV-Controllers Cohort. HLA-B*57 and IFNL4-related polymorphisms are associated with protection against HIV-1 disease progression in controllers. Clin. Infect. Dis. 2017, 64, 621–628. [Google Scholar] [PubMed]
- Buzón, M.J.; Massanella, M.; Llibre, J.M.; Esteve, A.; Dahl, V.; Puertas, M.C.; Gatell, J.M.; Domingo, P.; Paredes, R.; Sharkey, M.; et al. HIV-1 replication and immune dynamics are affected by raltegravir intensification of HAART-suppressed subjects. Nat. Med. 2010, 16, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Vidal, F.; Leal, M.; Alcamí, J.; Domingo, P. Current situation of the pharmacogenetics of immune recovery in treated HIV-infected patients. Pharmacogenomics 2014, 15, 569–572. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Picado, J.; Deeks, S. Persistent HIV-1 replication during antiretroviral therapy. Curr. Opin. HIV AIDS 2016, 11, 417–423. [Google Scholar] [CrossRef] [PubMed]
- García-Merino, I.; de Las Cuevas, N.; Jiménez, J.L.; Gallego, J.; Gómez, C.; Prieto, C.; Serramía, M.J.; Lorente, R.; Muñoz-Fernández, M.A. The Spanish HIV BioBank: A model of cooperative HIV research. Retrovirology 2009, 6, 27. [Google Scholar]
- Geiß, Y.; Dietrich, U. Catch me if you can—The race between HIV and neutralizing antibodies. AIDS Rev. 2015, 17, 107–113. [Google Scholar] [PubMed]
- García, F.; Climent, N.; Guardo, A.; Gil, C.; León, A.; Autran, B.; Lifson, J.D.; Martínez-Picado, J.; Dalmau, J.; Clotet, B.; et al. A dendritic cell-based vaccine elicits T cell responses associated with control of HIV-1 replication. Sci. Transl. Med. 2013, 5, 166ra2. [Google Scholar] [CrossRef] [PubMed]
- Mothe, B.; Climent, N.; Plana, M.; Rosàs, M.; Jiménez, J.L.; Muñoz-Fernández, M.Á.; Puertas, M.C.; Carrillo, J.; Gonzalez, N.; León, A.; et al. Safety and immunogenicity of a modified vaccinia Ankara-based HIV-1 vaccine (MVA-B) in HIV-1-infected patients alone or in combination with a drug to reactivate latent HIV-1. J. Antimicrob. Chemother. 2015, 70, 1833–1842. [Google Scholar] [CrossRef] [PubMed]
- Gómez, C.; Perdiguero, B.; García-Arriaza, J.; Cepeda, V.; Sánchez-Sorzano, C.; Mothe, B.; Jiménez, J.L.; Muñoz-Fernández, M.Á.; Gatell, J.M.; López Bernaldo de Quirós, J.C.; et al. A phase I randomized therapeutic MVA-B vaccination improves the magnitude and quality of the T cell immune responses in HIV-1-infected subjects on HAART. PLoS ONE 2015, 10, e0141456. [Google Scholar] [CrossRef] [PubMed]
- García-Arriaza, J.; Perdiguero, B.; Heeney, J.L.; Seaman, M.; Montefiori, D.; Yates, N.; Tomaras, G.; Ferrari, G.; Foulds, K.; Roederer, M.; et al. HIV/AIDS vaccine candidates based on replication-competent recombinant poxvirus NYVAC-C-KC expressing trimeric gp140 and Gag-derived VLPs or lacking the viral molecule B19 that inhibits type I interferon activate relevant HIV-1-specific B and T cell immune functions in non-human primates. J. Virol. 2017, 91, e02182-16. [Google Scholar] [PubMed]
- Soriano, V.; Gonzalez-Lahoz, J.; Guardiola, J.M. Manual del SIDA; Permanyer Publisher: Barcelona, Spain, 2011. [Google Scholar]
- Gatell, J.; Clotet, B.; Podzamczer, D.; Miró, J.M.; Mallolas, J. Guia Práctica del SIDA; Mason Publisher: Barcelona, Spain, 2016. [Google Scholar]
- Vance, D.; Cody, S. Prediction of geriatric HIV in 2030. Lancet Infect. Dis. 2015, 15, 753–754. [Google Scholar] [CrossRef]
- Brañas, F.; Jiménez, Z.; Sánchez-Conde, M.; Dronda, F.; López-Bernaldo de Quirós, J.C.; Pérez-Elías, M.J.; Miralles, P.; Ramírez, M.; Moreno, A.; Berenguer, J.; et al. Frailty and physical function in older HIV-infected patients. Age Ageing 2017, 46, 522–526. [Google Scholar] [CrossRef] [PubMed]
- Coiras, M.; López-Huertas, M.R.; Pérez-Olmeda, M.; Alcamí, J. Understanding HIV-1 latency provides clues for the eradication of long-term reservoirs. Nat. Rev. Microbiol. 2009, 7, 798–812. [Google Scholar] [CrossRef] [PubMed]
- Duarte, R.; Salgado, M.; Sánchez-Ortega, I.; Arnan, M.; Canals, C.; Domingo-Domenech, E.; Fernández-de-Sevilla, A.; González-Barca, E.; Morón-López, S.; Nogues, N.; et al. CCR5 Δ32 homozygous cord blood allogeneic transplantation in a patient with HIV: A case report. Lancet HIV 2015, 2, e236–e242. [Google Scholar] [CrossRef]
- De Blonde, J.; Sasse, A.; Del Amo, J.; Burns, F.; Delpech, V.; Cowan, S.; Levoy, M.; Keith, L.; Pharris, A.; Amato-Gauci, A.; et al. Restricted access to antiretroviral treatment for undocumented migrants: A bottle neck to control the HIV epidemic in the EU/EEA. BMC Public Health 2015, 15, 1228. [Google Scholar]
- GBD 2015 HIV Collaborators. Estimates of global, regional, and national incidence, prevalence, and mortality of HIV, 1980-2015: The Global Burden of Disease Study 2015. Lancet HIV 2016, 3, e361–e387. [Google Scholar]
- Halperin, D.; Steiner, M.; Cassell, M.; Green, E.; Hearst, N.; Kirby, D.; Gayle, H.; Cates, W. The time has come for common ground on preventing sexual transmission of HIV. Lancet 2004, 364, 1913–1915. [Google Scholar] [CrossRef]
- Katz, I.; Ehrenkranz, P.; El-Sadr, W. The global HIV epidemic: What will it take to get to the finish line? JAMA 2018, 319, 1094–1095. [Google Scholar] [CrossRef] [PubMed]
- Sacristán, J.A.; Gutierrez, J.A. Reflexiones sobre la Ciencia en España; Unión Editorial: Madrid, Spain, 2016. (In Spanish) [Google Scholar]
- Soriano, V.; Barreiro, P. Healthcare resources restrictions and HIV infection. Med. Clin. 2013, 141, 114–115. [Google Scholar] [CrossRef] [PubMed]
- Dinden, T. Over-Regulation Has Criminalized the Practice of Medicine. Available online: www.zerohedge.com (accessed on 21 February 2017).
|
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soriano, V.; Ramos, J.M.; Barreiro, P.; Fernandez-Montero, J.V. AIDS Clinical Research in Spain—Large HIV Population, Geniality of Doctors, and Missing Opportunities. Viruses 2018, 10, 293. https://doi.org/10.3390/v10060293
Soriano V, Ramos JM, Barreiro P, Fernandez-Montero JV. AIDS Clinical Research in Spain—Large HIV Population, Geniality of Doctors, and Missing Opportunities. Viruses. 2018; 10(6):293. https://doi.org/10.3390/v10060293
Chicago/Turabian StyleSoriano, Vicente, José M. Ramos, Pablo Barreiro, and Jose V. Fernandez-Montero. 2018. "AIDS Clinical Research in Spain—Large HIV Population, Geniality of Doctors, and Missing Opportunities" Viruses 10, no. 6: 293. https://doi.org/10.3390/v10060293
APA StyleSoriano, V., Ramos, J. M., Barreiro, P., & Fernandez-Montero, J. V. (2018). AIDS Clinical Research in Spain—Large HIV Population, Geniality of Doctors, and Missing Opportunities. Viruses, 10(6), 293. https://doi.org/10.3390/v10060293






