MERS-CoV: Understanding the Latest Human Coronavirus Threat
Abstract
1. Introduction
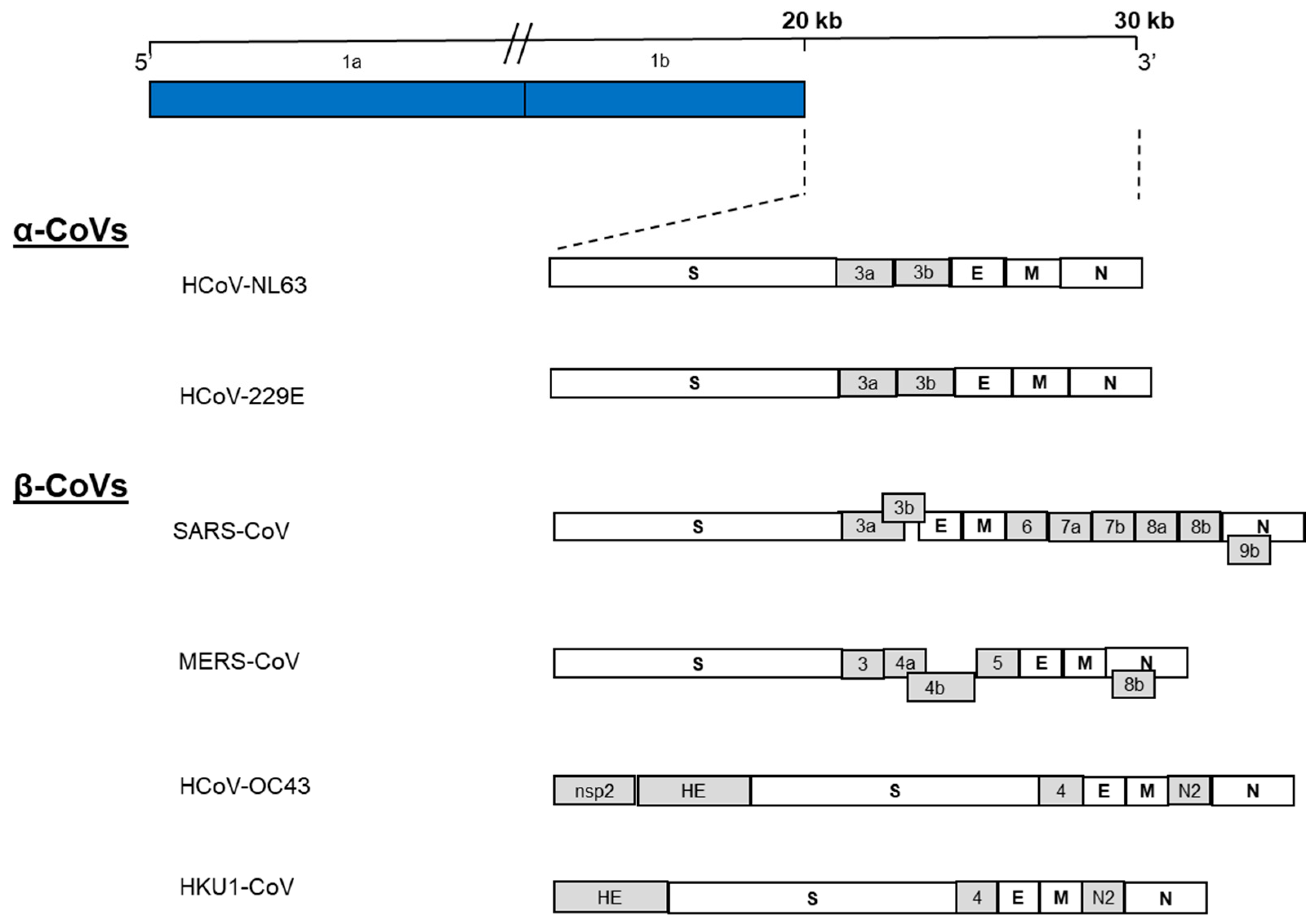
2. Genome Structure and Gene Functions
3. Clinical Features
4. Diagnosis of Infection
5. Animal Models
6. Treatment and Vaccine Development
7. Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zaki, A.M.; van Boheemen, S.; Bestebroer, T.M.; Osterhaus, A.D.; Fouchier, R.A. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N. Engl. J. Med. 2012, 367, 1814–1820. [Google Scholar] [CrossRef] [PubMed]
- De Groot, R.J.; Baker, S.C.; Baric, R.S.; Brown, C.S.; Drosten, C.; Enjuanes, L.; Fouchier, R.A.; Galiano, M.; Gorbalenya, A.E.; Memish, Z.A.; et al. Middle East respiratory syndrome coronavirus (MERS-CoV): Announcement of the Coronavirus Study Group. J. Virol. 2013, 87, 7790–7792. [Google Scholar] [CrossRef] [PubMed]
- WHO Middle East Respiratory Syndrome Coronavirus (MERS-CoV). Available online: http://www.who.int/emergencies/mers-cov/en/ (accessed on 23 May 2016).
- Gautret, P. Middle East Respiratory Syndrome (MERS) coronavirus. What travel health advice should be given to Hajj pilgrims? Travel Med. Infect. Dis. 2013, 11, 263–265. [Google Scholar] [CrossRef] [PubMed]
- Warnes, S.L.; Little, Z.R.; Keevil, C.W. Human Coronavirus 229E Remains Infectious on Common Touch Surface Materials. MBio 2015, 6, e01697-15. [Google Scholar] [CrossRef] [PubMed]
- Pas, S.D.; Koopmans, M.P.; Niedrig, M. Clinical implications of and lessons learnt from external assessment of Mers-CoV diagnostics. Expert Rev. Mol. Diagn. 2016, 16, 7–9. [Google Scholar] [CrossRef] [PubMed]
- Bin, S.Y.; Heo, J.Y.; Song, M.S.; Lee, J.; Kim, E.H.; Park, S.J.; Kwon, H.I.; Kim, S.M.; Kim, Y.I.; Si, Y.J.; et al. Environmental Contamination and Viral Shedding in MERS Patients During MERS-CoV Outbreak in South Korea. Clin. Infect. Dis. 2016, 62, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Kraaij-Dirkzwager, M.; Timen, A.; Dirksen, K.; Gelinck, L.; Leyten, E.; Groeneveld, P.; Jansen, C.; Jonges, M.; Raj, S.; Thurkow, I.; et al. Middle East respiratory syndrome coronavirus (MERS-CoV) infections in two returning travellers in the Netherlands, May 2014. Euro Surveill. 2014, 19, 20817. [Google Scholar] [CrossRef] [PubMed]
- Van Boheemen, S.; de Graaf, M.; Lauber, C.; Bestebroer, T.M.; Raj, V.S.; Zaki, A.M.; Osterhaus, A.D.; Haagmans, B.L.; Gorbalenya, A.E.; Snijder, E.J.; et al. Genomic characterization of a newly discovered coronavirus associated with acute respiratory distress syndrome in humans. MBio 2012, 3, e00473-12. [Google Scholar] [CrossRef] [PubMed]
- Wernery, U.; El Rasoul, I.H.; Wong, E.Y.; Joseph, M.; Chen, Y.; Jose, S.; Tsang, A.K.; Patteril, N.A.; Chen, H.; Elizabeth, S.K.; et al. A phylogenetically distinct Middle East respiratory syndrome coronavirus detected in a dromedary calf from a closed dairy herd in Dubai with rising seroprevalence with age. Emerg. Microbes Infect. 2015, 4, e74. [Google Scholar] [CrossRef] [PubMed]
- Smits, S.L.; Raj, V.S.; Pas, S.D.; Reusken, C.B.; Mohran, K.; Farag, E.A.; Al-Romaihi, H.E.; AlHajri, M.M.; Haagmans, B.L.; Koopmans, M.P. Reliable typing of MERS-CoV variants with a small genome fragment. J. Clin. Virol. 2015, 64, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Wernery, U.; Lau, S.K.; Woo, P.C. Genomics and zoonotic infections: Middle East respiratory syndrome. Rev. Sci. Tech. 2016, 35, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Wang, Y.; Wang, W.; Nie, K.; Zhao, Y.; Su, J.; Deng, Y.; Zhou, W.; Li, Y.; Wang, H.; et al. Complete Genome Sequence of Middle East Respiratory Syndrome Coronavirus (MERS-CoV) from the First Imported MERS-CoV Case in China. Genome Announc. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Cho, Y.J.; Kim, D.W.; Yang, J.S.; Kim, H.; Park, S.; Han, Y.W.; Yun, M.R.; Lee, H.S.; Kim, A.R.; et al. Complete Genome Sequence of Middle East Respiratory Syndrome Coronavirus KOR/KNIH/002_05_2015, Isolated in South Korea. Genome Announc. 2015, 3, e00787-15. [Google Scholar] [CrossRef] [PubMed]
- Kandeil, A.; Shehata, M.M.; El Shesheny, R.; Gomaa, M.R.; Ali, M.A.; Kayali, G. Complete Genome Sequence of Middle East Respiratory Syndrome Coronavirus Isolated from a Dromedary Camel in Egypt. Genome Announc. 2016, 4, e00309-16. [Google Scholar] [CrossRef] [PubMed]
- Thornbrough, J.M.; Jha, B.K.; Yount, B.; Goldstein, S.A.; Li, Y.; Elliott, R.; Sims, A.C.; Baric, R.S.; Silverman, R.H.; Weiss, S.R. Middle East Respiratory Syndrome Coronavirus NS4b Protein Inhibits Host RNase L Activation. MBio 2016, 7, e00258. [Google Scholar] [CrossRef] [PubMed]
- Menachery, V.D.; Mitchell, H.D.; Cockrell, A.S.; Gralinski, L.E.; Yount, B.L., Jr.; Graham, R.L.; McAnarney, E.T.; Douglas, M.G.; Scobey, T.; Beall, A.; et al. MERS-CoV Accessory ORFs Play Key Role for Infection and Pathogenesis. MBio 2017, 8, e00665-17. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhang, L.; Geng, H.; Deng, Y.; Huang, B.; Guo, Y.; Zhao, Z.; Tan, W. The structural and accessory proteins M, ORF 4a, ORF 4b, and ORF 5 of Middle East respiratory syndrome coronavirus (MERS-CoV) are potent interferon antagonists. Protein Cell 2013, 4, 951–961. [Google Scholar] [CrossRef] [PubMed]
- Scobey, T.; Yount, B.L.; Sims, A.C.; Donaldson, E.F.; Agnihothram, S.S.; Menachery, V.D.; Graham, R.L.; Swanstrom, J.; Bove, P.F.; Kim, J.D.; et al. Reverse genetics with a full-length infectious cDNA of the Middle East respiratory syndrome coronavirus. Proc. Natl. Acad. Sci. USA 2013, 110, 16157–16162. [Google Scholar] [CrossRef] [PubMed]
- Almazan, F.; DeDiego, M.L.; Sola, I.; Zuniga, S.; Nieto-Torres, J.L.; Marquez-Jurado, S.; Andres, G.; Enjuanes, L. Engineering a replication-competent, propagation-defective Middle East respiratory syndrome coronavirus as a vaccine candidate. MBio 2013, 4, e00650-13. [Google Scholar] [CrossRef] [PubMed]
- Mielech, A.M.; Kilianski, A.; Baez-Santos, Y.M.; Mesecar, A.D.; Baker, S.C. MERS-CoV papain-like protease has deISGylating and deubiquitinating activities. Virology 2014, 450–451, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Sastre, A.; Biron, C.A. Type 1 interferons and the virus-host relationship: A lesson in detente. Science 2006, 312, 879–882. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Sastre, A. Antiviral response in pandemic influenza viruses. Emerg. Infect. Dis. 2006, 12, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Randall, R.E.; Goodbourn, S. Interferons and viruses: An interplay between induction, signalling, antiviral responses and virus countermeasures. J. Gen. Virol. 2008, 89 Pt 1, 1–47. [Google Scholar] [CrossRef] [PubMed]
- Taylor, K.E.; Mossman, K.L. Recent advances in understanding viral evasion of type I interferon. Immunology 2013, 138, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Totura, A.L.; Baric, R.S. SARS coronavirus pathogenesis: Host innate immune responses and viral antagonism of interferon. Curr. Opin. Virol. 2012, 2, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Niemeyer, D.; Zillinger, T.; Muth, D.; Zielecki, F.; Horvath, G.; Suliman, T.; Barchet, W.; Weber, F.; Drosten, C.; Muller, M.A. Middle East respiratory syndrome coronavirus accessory protein 4a is a type I interferon antagonist. J. Virol. 2013, 87, 12489–12495. [Google Scholar] [CrossRef] [PubMed]
- Matthews, K.L.; Coleman, C.M.; van der Meer, Y.; Snijder, E.J.; Frieman, M.B. The ORF4b-encoded accessory proteins of Middle East respiratory syndrome coronavirus and two related bat coronaviruses localize to the nucleus and inhibit innate immune signalling. J. Gen. Virol. 2014, 95 Pt 4, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Lokugamage, K.G.; Narayanan, K.; Nakagawa, K.; Terasaki, K.; Ramirez, S.I.; Tseng, C.T.; Makino, S. Middle East Respiratory Syndrome Coronavirus nsp1 Inhibits Host Gene Expression by Selectively Targeting mRNAs Transcribed in the Nucleus while Sparing mRNAs of Cytoplasmic Origin. J. Virol. 2015, 89, 10970–10981. [Google Scholar] [CrossRef] [PubMed]
- Bailey-Elkin, B.A.; Knaap, R.C.; Johnson, G.G.; Dalebout, T.J.; Ninaber, D.K.; van Kasteren, P.B.; Bredenbeek, P.J.; Snijder, E.J.; Kikkert, M.; Mark, B.L. Crystal structure of the Middle East respiratory syndrome coronavirus (MERS-CoV) papain-like protease bound to ubiquitin facilitates targeted disruption of deubiquitinating activity to demonstrate its role in innate immune suppression. J. Biol. Chem. 2014, 289, 34667–34682. [Google Scholar] [CrossRef] [PubMed]
- Arabi, Y.M.; Arifi, A.A.; Balkhy, H.H.; Najm, H.; Aldawood, A.S.; Ghabashi, A.; Hawa, H.; Alothman, A.; Khaldi, A.; Al Raiy, B. Clinical course and outcomes of critically ill patients with Middle East respiratory syndrome coronavirus infection. Ann. Intern. Med. 2014, 160, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Senga, M.; Arabi, Y.M.; Fowler, R.A. Clinical spectrum of the Middle East respiratory syndrome coronavirus (MERS-CoV). J. Infect. Public Health 2017, 10, 191–194. [Google Scholar] [CrossRef] [PubMed]
- Zumla, A.; Hui, D.S.; Perlman, S. Middle East respiratory syndrome. Lancet 2015, 386, 995–1007. [Google Scholar] [CrossRef]
- Assiri, A.; Al-Tawfiq, J.A.; Al-Rabeeah, A.A.; Al-Rabiah, F.A.; Al-Hajjar, S.; Al-Barrak, A.; Flemban, H.; Al-Nassir, W.N.; Balkhy, H.H.; Al-Hakeem, R.F.; et al. Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: A descriptive study. Lancet Infect. Dis. 2013, 13, 752–761. [Google Scholar] [CrossRef]
- Assiri, A.; McGeer, A.; Perl, T.M.; Price, C.S.; Al Rabeeah, A.A.; Cummings, D.A.; Alabdullatif, Z.N.; Assad, M.; Almulhim, A.; Makhdoom, H.; et al. Hospital outbreak of Middle East respiratory syndrome coronavirus. N. Engl. J. Med. 2013, 369, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Hijawi, B.; Abdallat, M.; Sayaydeh, A.; Alqasrawi, S.; Haddadin, A.; Jaarour, N.; Alsheikh, S.; Alsanouri, T. Novel coronavirus infections in Jordan, April 2012: Epidemiological findings from a retrospective investigation. East. Mediterr. Health J. 2013, 19 (Suppl. S1), S12–S18. [Google Scholar] [PubMed]
- Drosten, C.; Muth, D.; Corman, V.M.; Hussain, R.; Al Masri, M.; HajOmar, W.; Landt, O.; Assiri, A.; Eckerle, I.; Al Shangiti, A.; et al. An observational, laboratory-based study of outbreaks of middle East respiratory syndrome coronavirus in Jeddah and Riyadh, kingdom of Saudi Arabia, 2014. Clin. Infect. Dis. 2015, 60, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Guery, B.; Poissy, J.; el Mansouf, L.; Sejourne, C.; Ettahar, N.; Lemaire, X.; Vuotto, F.; Goffard, A.; Behillil, S.; Enouf, V.; et al. Clinical features and viral diagnosis of two cases of infection with Middle East Respiratory Syndrome coronavirus: A report of nosocomial transmission. Lancet 2013, 381, 2265–2272. [Google Scholar] [CrossRef]
- Memish, Z.A.; Zumla, A.I.; Al-Hakeem, R.F.; Al-Rabeeah, A.A.; Stephens, G.M. Family cluster of Middle East respiratory syndrome coronavirus infections. N. Engl. J. Med. 2013, 368, 2487–2494. [Google Scholar] [CrossRef] [PubMed]
- Al-Tawfiq, J.A.; Hinedi, K.; Ghandour, J.; Khairalla, H.; Musleh, S.; Ujayli, A.; Memish, Z.A. Middle East respiratory syndrome coronavirus: A case-control study of hospitalized patients. Clin. Infect. Dis. 2014, 59, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Oboho, I.K.; Tomczyk, S.M.; Al-Asmari, A.M.; Banjar, A.A.; Al-Mugti, H.; Aloraini, M.S.; Alkhaldi, K.Z.; Almohammadi, E.L.; Alraddadi, B.M.; Gerber, S.I.; et al. 2014 MERS-CoV outbreak in Jeddah—A link to health care facilities. N. Engl. J. Med. 2015, 372, 846–854. [Google Scholar] [CrossRef] [PubMed]
- Drosten, C.; Meyer, B.; Muller, M.A.; Corman, V.M.; Al-Masri, M.; Hossain, R.; Madani, H.; Sieberg, A.; Bosch, B.J.; Lattwein, E.; et al. Transmission of MERS-coronavirus in household contacts. N. Engl. J. Med. 2014, 371, 828–835. [Google Scholar] [CrossRef] [PubMed]
- Kupferschmidt, K. INFECTIOUS DISEASE. Camel vaccine offers hope to stop MERS. Science 2015, 350, 1453. [Google Scholar] [CrossRef] [PubMed]
- Al-Dorzi, H.M.; Alsolamy, S.; Arabi, Y.M. Critically ill patients with Middle East respiratory syndrome coronavirus infection. Crit. Care 2016, 20, 65. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Pringle, K.; Kumar, A.; Dearth, S.; Liu, L.; Lovchik, J.; Perez, O.; Pontones, P.; Richards, S.; Yeadon-Fagbohun, J.; et al. Clinical and laboratory findings of the first imported case of Middle East respiratory syndrome coronavirus to the United States. Clin. Infect. Dis. 2014, 59, 1511–1518. [Google Scholar] [CrossRef] [PubMed]
- Browne, A.; St-Onge Ahmad, S.; Beck, C.R.; Nguyen-van-Tam, J.S. The roles of transportation and transportation hubs in the propagation of influenza and coronaviruses: A systematic review. J. Travel Med. 2016, 23. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, M.S.; Khalid, H.S.; Muddathir, A.E.; El-Tahir, K.; Khan, A.A.; Algadir, H.A.; Osman, W.J.; Siddiqui, N.A. Effect of some plants’ extracts used in Sudanese folkloric medicines on carrageenan-induced inflammation. Pak. J. Pharm. Sci. 2015, 28, 159–165. [Google Scholar] [PubMed]
- Corman, V.M.; Albarrak, A.M.; Omrani, A.S.; Albarrak, M.M.; Farah, M.E.; Almasri, M.; Muth, D.; Sieberg, A.; Meyer, B.; Assiri, A.M.; et al. Viral Shedding and Antibody Response in 37 Patients With Middle East Respiratory Syndrome Coronavirus Infection. Clin. Infect. Dis. 2016, 62, 477–483. [Google Scholar] [CrossRef] [PubMed]
- The WHO MERS-CoV Research Group. State of Knowledge and Data Gaps of Middle East Respiratory Syndrome Coronavirus (MERS-CoV) in Humans. PLoS Curr. 2013, 5. [Google Scholar] [CrossRef]
- Berry, M.; Fielding, B.C.; Gamieldien, J. Potential Broad Spectrum Inhibitors of the Coronavirus 3CLpro: A Virtual Screening and Structure-Based Drug Design Study. Viruses 2015, 7, 6642–6660. [Google Scholar] [CrossRef] [PubMed]
- Al-Abdallat, M.M.; Payne, D.C.; Alqasrawi, S.; Rha, B.; Tohme, R.A.; Abedi, G.R.; Al Nsour, M.; Iblan, I.; Jarour, N.; Farag, N.H.; et al. Hospital-associated outbreak of Middle East respiratory syndrome coronavirus: A serologic, epidemiologic, and clinical description. Clin. Infect. Dis. 2014, 59, 1225–1233. [Google Scholar] [CrossRef] [PubMed]
- O’Keefe, L.C. Middle East Respiratory Syndrome Coronavirus. Workplace Health Saf. 2016. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Li, Q.; Sun, X.; Ren, Z.; He, F.; Wang, Y.; Wang, L. Recycling flue gas desulphurization (FGD) gypsum for removal of Pb(II) and Cd(II) from wastewater. J. Colloid Interface Sci. 2015, 457, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Tandi, T.E.; Choi, J.W.; Moon, J.M.; Kim, M.S. Middle East respiratory syndrome coronavirus (MERS-CoV) outbreak in South Korea, 2015: Epidemiology, characteristics and public health implications. J. Hosp. Infect. 2017, 95, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Hui, D.S.; Memish, Z.A.; Zumla, A. Severe acute respiratory syndrome vs. the Middle East respiratory syndrome. Curr. Opin. Pulm. Med. 2014, 20, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Al-Tawfiq, J.A.; Kattan, R.F.; Memish, Z.A. Middle East respiratory syndrome coronavirus disease is rare in children: An update from Saudi Arabia. World J. Clin. Pediatr. 2016, 5, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Memish, Z.A.; Al-Tawfiq, J.A.; Assiri, A.; AlRabiah, F.A.; Al Hajjar, S.; Albarrak, A.; Flemban, H.; Alhakeem, R.F.; Makhdoom, H.Q.; Alsubaie, S.; et al. Middle East respiratory syndrome coronavirus disease in children. Pediatr. Infect. Dis. J. 2014, 33, 904–906. [Google Scholar] [CrossRef] [PubMed]
- Zumla, A.; Azhar, E.I.; Arabi, Y.; Alotaibi, B.; Rao, M.; McCloskey, B.; Petersen, E.; Maeurer, M. Host-directed therapies for improving poor treatment outcomes associated with the middle east respiratory syndrome coronavirus infections. Int. J. Infect. Dis. 2015, 40, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Pinky, L.; Dobrovolny, H.M. Coinfections of the Respiratory Tract: Viral Competition for Resources. PLoS ONE 2016, 11, e0155589. [Google Scholar] [CrossRef] [PubMed]
- Calvo, C.; Garcia-Garcia, M.L.; Pozo, F.; Paula, G.; Molinero, M.; Calderon, A.; Gonzalez-Esguevillas, M.; Casas, I. Respiratory Syncytial Virus Coinfections With Rhinovirus and Human Bocavirus in Hospitalized Children. Medicine 2015, 94, e1788. [Google Scholar] [CrossRef] [PubMed]
- Furuse, Y.; Okamoto, M.; Oshitani, H. Conservation of nucleotide sequences for molecular diagnosis of Middle East respiratory syndrome coronavirus, 2015. Int. J. Infect. Dis. 2015, 40, 25–27. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wiwanitkit, V. Critical care medicine for emerging Middle East respiratory syndrome: Which point to be considered? Indian J. Crit. Care Med. 2015, 19, 528–530. [Google Scholar] [CrossRef] [PubMed]
- Eggers, M.; Eickmann, M.; Zorn, J. Rapid and Effective Virucidal Activity of Povidone-Iodine Products Against Middle East Respiratory Syndrome Coronavirus (MERS-CoV) and Modified Vaccinia Virus Ankara (MVA). Infect. Dis. Ther. 2015, 4, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chan, K.H.; Kang, Y.; Chen, H.; Luk, H.; Poon, R.W.; Chan, J.F.; Yuen, K.Y.; Xia, N.; Lau, S.K.; et al. A sensitive and specific antigen detection assay for Middle East respiratory syndrome coronavirus. Emerg. Microbes Infect. 2015, 4, e26. [Google Scholar] [CrossRef] [PubMed]
- Noorwali, A.A.; Turkistani, A.M.; Asiri, S.I.; Trabulsi, F.A.; Alwafi, O.M.; Alzahrani, S.H.; Rashid, M.M.; Hegazy, S.A.; Alzaydi, M.D.; Bawakid, K.O. Descriptive epidemiology and characteristics of confirmed cases of Middle East respiratory syndrome coronavirus infection in the Makkah Region of Saudi Arabia, March to June 2014. Ann. Saudi Med. 2015, 35, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Bakaletz, L.O. Viral-bacterial co-infections in the respiratory tract. Curr. Opin. Microbiol. 2017, 35, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Heo, J.H.; Kim, H.O.; Song, S.H.; Park, S.S.; Park, T.H.; Ahn, J.Y.; Kim, M.K.; Choi, J.P. Neurological Complications during Treatment of Middle East Respiratory Syndrome. J. Clin. Neurol. 2017, 13, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Arabi, Y.M.; Harthi, A.; Hussein, J.; Bouchama, A.; Johani, S.; Hajeer, A.H.; Saeed, B.T.; Wahbi, A.; Saedy, A.; AlDabbagh, T.; et al. Severe neurologic syndrome associated with Middle East respiratory syndrome corona virus (MERS-CoV). Infection 2015, 43, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Saad, M.; Omrani, A.S.; Baig, K.; Bahloul, A.; Elzein, F.; Matin, M.A.; Selim, M.A.; Al Mutairi, M.; Al Nakhli, D.; Al Aidaroos, A.Y.; et al. Clinical aspects and outcomes of 70 patients with Middle East respiratory syndrome coronavirus infection: A single-center experience in Saudi Arabia. Int. J. Infect. Dis. 2014, 29, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Algahtani, H.; Subahi, A.; Shirah, B. Neurological Complications of Middle East Respiratory Syndrome Coronavirus: A Report of Two Cases and Review of the Literature. Case Rep. Neurol. Med. 2016, 2016, 3502683. [Google Scholar] [CrossRef] [PubMed]
- Cha, R.H.; Yang, S.H.; Moon, K.C.; Joh, J.S.; Lee, J.Y.; Shin, H.S.; Kim, D.K.; Kim, Y.S. A Case Report of a Middle East Respiratory Syndrome Survivor with Kidney Biopsy Results. J. Korean Med. Sci. 2016, 31, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Poissy, J.; Goffard, A.; Parmentier-Decrucq, E.; Favory, R.; Kauv, M.; Kipnis, E.; Mathieu, D.; van der Werf, S.; Guery, B. Kinetics and pattern of viral excretion in biological specimens of two MERS-CoV cases. J. Clin. Virol. 2014, 61, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Drosten, C.; Seilmaier, M.; Corman, V.M.; Hartmann, W.; Scheible, G.; Sack, S.; Guggemos, W.; Kallies, R.; Muth, D.; Junglen, S.; et al. Clinical features and virological analysis of a case of Middle East respiratory syndrome coronavirus infection. Lancet Infect. Dis. 2013, 13, 745–751. [Google Scholar] [CrossRef]
- Van Doremalen, N.; Bushmaker, T.; Munster, V.J. Stability of Middle East respiratory syndrome coronavirus (MERS-CoV) under different environmental conditions. Euro Surveill. 2013, 18, 20590. [Google Scholar] [CrossRef] [PubMed]
- Corman, V.M.; Eckerle, I.; Bleicker, T.; Zaki, A.; Landt, O.; Eschbach-Bludau, M.; van Boheemen, S.; Gopal, R.; Ballhause, M.; Bestebroer, T.M.; et al. Detection of a novel human coronavirus by real-time reverse-transcription polymerase chain reaction. Euro Surveill. 2012, 17, 20285. [Google Scholar] [CrossRef] [PubMed]
- Corman, V.M.; Muller, M.A.; Costabel, U.; Timm, J.; Binger, T.; Meyer, B.; Kreher, P.; Lattwein, E.; Eschbach-Bludau, M.; Nitsche, A.; et al. Assays for laboratory confirmation of novel human coronavirus (hCoV-EMC) infections. Euro Surveill. 2012, 17, 20334. [Google Scholar] [CrossRef] [PubMed]
- De Wilde, A.H.; Raj, V.S.; Oudshoorn, D.; Bestebroer, T.M.; van Nieuwkoop, S.; Limpens, R.W.; Posthuma, C.C.; van der Meer, Y.; Barcena, M.; Haagmans, B.L.; et al. MERS-coronavirus replication induces severe in vitro cytopathology and is strongly inhibited by cyclosporin A or interferon-alpha treatment. J. Gen. Virol. 2013, 94 Pt 8, 1749–1760. [Google Scholar] [CrossRef] [PubMed]
- Leland, D.S.; Ginocchio, C.C. Role of cell culture for virus detection in the age of technology. Clin. Microbiol. Rev. 2007, 20, 49–78. [Google Scholar] [CrossRef] [PubMed]
- Mackay, I.M.; Arden, K.E. MERS coronavirus: Diagnostics, epidemiology and transmission. Virol. J. 2015, 12, 222. [Google Scholar] [CrossRef] [PubMed]
- Shirato, K.; Yano, T.; Senba, S.; Akachi, S.; Kobayashi, T.; Nishinaka, T.; Notomi, T.; Matsuyama, S. Detection of Middle East respiratory syndrome coronavirus using reverse transcription loop-mediated isothermal amplification (RT-LAMP). Virol. J. 2014, 11, 139. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Whitaker, B.; Sakthivel, S.K.; Kamili, S.; Rose, L.E.; Lowe, L.; Mohareb, E.; Elassal, E.M.; Al-sanouri, T.; Haddadin, A.; et al. Real-time reverse transcription-PCR assay panel for Middle East respiratory syndrome coronavirus. J. Clin. Microbiol. 2014, 52, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; He, B.; Li, C.; Zhang, X.; Wu, W.; Yin, X.; Fan, B.; Fan, X.; Wang, J. Real-time RT-PCR for H5N1 avian influenza A virus detection. J. Med. Microbiol. 2007, 56 Pt 5, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Pang, X.; Lee, B.; Chui, L.; Preiksaitis, J.K.; Monroe, S.S. Evaluation and validation of real-time reverse transcription-pcr assay using the LightCycler system for detection and quantitation of norovirus. J. Clin. Microbiol. 2004, 42, 4679–4685. [Google Scholar] [CrossRef] [PubMed]
- Buller, R.S.; Storch, G. Evaluation of a real-time PCR assay using the LightCycler system for detection of parvovirus B19 DNA. J. Clin. Microbiol. 2004, 42, 3326–3328. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ke, G.M.; Cheng, H.L.; Ke, L.Y.; Ji, W.T.; Chulu, J.L.; Liao, M.H.; Chang, T.J.; Liu, H.J. Development of a quantitative Light Cycler real-time RT-PCR for detection of avian reovirus. J. Virol. Methods 2006, 133, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Parida, M.M. Rapid and real-time detection technologies for emerging viruses of biomedical importance. J. Biosci. 2008, 33, 617–628. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka, Y.; Matsuyama, S.; Fukushi, S.; Matsunaga, S.; Matsushima, Y.; Kuroyama, H.; Kimura, H.; Takeda, M.; Chimuro, T.; Ryo, A. Development of Monoclonal Antibody and Diagnostic Test for Middle East Respiratory Syndrome Coronavirus Using Cell-Free Synthesized Nucleocapsid Antigen. Front. Microbiol. 2016, 7, 509. [Google Scholar] [CrossRef] [PubMed]
- Bhadra, S.; Jiang, Y.S.; Kumar, M.R.; Johnson, R.F.; Hensley, L.E.; Ellington, A.D. Real-time sequence-validated loop-mediated isothermal amplification assays for detection of Middle East respiratory syndrome coronavirus (MERS-CoV). PLoS ONE 2015, 10, e0123126. [Google Scholar] [CrossRef] [PubMed]
- Abd El Wahed, A.; Patel, P.; Heidenreich, D.; Hufert, F.T.; Weidmann, M. Reverse transcription recombinase polymerase amplification assay for the detection of middle East respiratory syndrome coronavirus. PLoS Curr. 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Ha, G.; Serhan, W.; Eltahir, Y.; Yusof, M.; Hashem, F.; Elsayed, E.; Marzoug, B.; Abdelazim, A.; Al Muhairi, S. Development and validation of a rapid immunochromatographic assay for detection of Middle East respiratory syndrome coronavirus antigen in dromedary camels. J. Clin. Microbiol. 2015, 53, 1178–1182. [Google Scholar] [CrossRef] [PubMed]
- Meyer, B.; Drosten, C.; Muller, M.A. Serological assays for emerging coronaviruses: Challenges and pitfalls. Virus Res. 2014, 194, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.H.; Chan, J.F.; Tse, H.; Chen, H.; Lau, C.C.; Cai, J.P.; Tsang, A.K.; Xiao, X.; To, K.K.; Lau, S.K.; et al. Cross-reactive antibodies in convalescent SARS patients’ sera against the emerging novel human coronavirus EMC (2012) by both immunofluorescent and neutralizing antibody tests. J. Infect. 2013, 67, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Aburizaiza, A.S.; Mattes, F.M.; Azhar, E.I.; Hassan, A.M.; Memish, Z.A.; Muth, D.; Meyer, B.; Lattwein, E.; Muller, M.A.; Drosten, C. Investigation of anti-middle East respiratory syndrome antibodies in blood donors and slaughterhouse workers in Jeddah and Makkah, Saudi Arabia, fall 2012. J. Infect. Dis. 2014, 209, 243–246. [Google Scholar] [CrossRef] [PubMed]
- Perera, R.A.; Wang, P.; Gomaa, M.R.; El-Shesheny, R.; Kandeil, A.; Bagato, O.; Siu, L.Y.; Shehata, M.M.; Kayed, A.S.; Moatasim, Y.; et al. Seroepidemiology for MERS coronavirus using microneutralisation and pseudoparticle virus neutralisation assays reveal a high prevalence of antibody in dromedary camels in Egypt, June 2013. Euro Surveill. 2013, 18, 20574. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Du, L.; Ma, C.; Li, Y.; Li, L.; Poon, V.K.; Wang, L.; Yu, F.; Zheng, B.J.; Jiang, S.; et al. A safe and convenient pseudovirus-based inhibition assay to detect neutralizing antibodies and screen for viral entry inhibitors against the novel human coronavirus MERS-CoV. Virol. J. 2013, 10, 266. [Google Scholar] [CrossRef] [PubMed]
- Reusken, C.B.; Haagmans, B.L.; Muller, M.A.; Gutierrez, C.; Godeke, G.J.; Meyer, B.; Muth, D.; Raj, V.S.; Smits-de Vries, L.; Corman, V.M.; et al. Middle East respiratory syndrome coronavirus neutralising serum antibodies in dromedary camels: A comparative serological study. Lancet Infect. Dis. 2013, 13, 859–866. [Google Scholar] [CrossRef]
- Meyer, B.; Muller, M.A.; Corman, V.M.; Reusken, C.B.; Ritz, D.; Godeke, G.J.; Lattwein, E.; Kallies, S.; Siemens, A.; van Beek, J.; et al. Antibodies against MERS coronavirus in dromedary camels, United Arab Emirates, 2003 and 2013. Emerg. Infect. Dis. 2014, 20, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Reusken, C.; Mou, H.; Godeke, G.J.; van der Hoek, L.; Meyer, B.; Muller, M.A.; Haagmans, B.; de Sousa, R.; Schuurman, N.; Dittmer, U.; et al. Specific serology for emerging human coronaviruses by protein microarray. Euro Surveill. 2013, 18, 20441. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Baek, Y.H.; Kim, Y.H.; Choi, Y.K.; Song, M.S.; Ahn, J.Y. One-Pot Reverse Transcriptional Loop-Mediated Isothermal Amplification (RT-LAMP) for Detecting MERS-CoV. Front. Microbiol. 2016, 7, 2166. [Google Scholar] [CrossRef] [PubMed]
- Go, Y.Y.; Kim, Y.S.; Cheon, S.; Nam, S.; Ku, K.B.; Kim, M.; Cho, N.H.; Park, H.; Alison Lee, P.Y.; Lin, Y.C.; et al. Evaluation and Clinical Validation of Two Field-Deployable Reverse Transcription-Insulated Isothermal PCR Assays for the Detection of the Middle East Respiratory Syndrome-Coronavirus. J. Mol. Diagn. 2017, 19, 817–827. [Google Scholar] [CrossRef] [PubMed]
- Huh, H.J.; Kim, J.Y.; Kwon, H.J.; Yun, S.A.; Lee, M.K.; Ki, C.S.; Lee, N.Y.; Kim, J.W. Performance Evaluation of the PowerChek MERS (upE & ORF1a) Real-Time PCR Kit for the Detection of Middle East Respiratory Syndrome Coronavirus RNA. Ann. Lab. Med. 2017, 37, 494–498. [Google Scholar] [PubMed]
- Teengam, P.; Siangproh, W.; Tuantranont, A.; Vilaivan, T.; Chailapakul, O.; Henry, C.S. Multiplex Paper-Based Colorimetric DNA Sensor Using Pyrrolidinyl Peptide Nucleic Acid-Induced AgNPs Aggregation for Detecting MERS-CoV, MTB, and HPV Oligonucleotides. Anal. Chem. 2017, 89, 5428–5435. [Google Scholar] [CrossRef] [PubMed]
- Xiu, L.; Zhang, C.; Wu, Z.; Peng, J. Establishment and Application of a Universal Coronavirus Screening Method Using MALDI-TOF Mass Spectrometry. Front. Microbiol. 2017, 8, 1510. [Google Scholar] [CrossRef] [PubMed]
- Noh, J.Y.; Yoon, S.W.; Kim, D.J.; Lee, M.S.; Kim, J.H.; Na, W.; Song, D.; Jeong, D.G.; Kim, H.K. Simultaneous detection of severe acute respiratory syndrome, Middle East respiratory syndrome, and related bat coronaviruses by real-time reverse transcription PCR. Arch. Virol. 2017, 162, 1617–1623. [Google Scholar] [CrossRef] [PubMed]
- Sutton, T.C.; Subbarao, K. Development of animal models against emerging coronaviruses: From SARS to MERS coronavirus. Virology 2015, 479–480, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Haagmans, B.L.; van den Brand, J.M.; Provacia, L.B.; Raj, V.S.; Stittelaar, K.J.; Getu, S.; de Waal, L.; Bestebroer, T.M.; van Amerongen, G.; Verjans, G.M.; et al. Asymptomatic Middle East respiratory syndrome coronavirus infection in rabbits. J. Virol. 2015, 89, 6131–6135. [Google Scholar] [CrossRef] [PubMed]
- Raj, V.S.; Smits, S.L.; Provacia, L.B.; van den Brand, J.M.; Wiersma, L.; Ouwendijk, W.J.; Bestebroer, T.M.; Spronken, M.I.; van Amerongen, G.; Rottier, P.J.; et al. Adenosine deaminase acts as a natural antagonist for dipeptidyl peptidase 4-mediated entry of the Middle East respiratory syndrome coronavirus. J. Virol. 2014, 88, 1834–1838. [Google Scholar] [CrossRef] [PubMed]
- De Wit, E.; Prescott, J.; Baseler, L.; Bushmaker, T.; Thomas, T.; Lackemeyer, M.G.; Martellaro, C.; Milne-Price, S.; Haddock, E.; Haagmans, B.L.; et al. The Middle East respiratory syndrome coronavirus (MERS-CoV) does not replicate in Syrian hamsters. PLoS ONE 2013, 8, e69127. [Google Scholar] [CrossRef] [PubMed]
- Coleman, C.M.; Matthews, K.L.; Goicochea, L.; Frieman, M.B. Wild-type and innate immune-deficient mice are not susceptible to the Middle East respiratory syndrome coronavirus. J. Gen. Virol. 2014, 95 Pt 2, 408–412. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Li, K.; Wohlford-Lenane, C.; Agnihothram, S.S.; Fett, C.; Zhao, J.; Gale, M.J., Jr.; Baric, R.S.; Enjuanes, L.; Gallagher, T.; et al. Rapid generation of a mouse model for Middle East respiratory syndrome. Proc. Natl. Acad. Sci. USA 2014, 111, 4970–4975. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.S.; Garron, T.; Tao, X.; Peng, B.H.; Wakamiya, M.; Chan, T.S.; Couch, R.B.; Tseng, C.T. Generation of a transgenic mouse model of Middle East respiratory syndrome coronavirus infection and disease. J. Virol. 2015, 89, 3659–3670. [Google Scholar] [CrossRef] [PubMed]
- Pascal, K.E.; Coleman, C.M.; Mujica, A.O.; Kamat, V.; Badithe, A.; Fairhurst, J.; Hunt, C.; Strein, J.; Berrebi, A.; Sisk, J.M.; et al. Pre- and postexposure efficacy of fully human antibodies against Spike protein in a novel humanized mouse model of MERS-CoV infection. Proc. Natl. Acad. Sci. USA 2015, 112, 8738–8743. [Google Scholar] [CrossRef] [PubMed]
- Cockrell, A.S.; Yount, B.L.; Scobey, T.; Jensen, K.; Douglas, M.; Beall, A.; Tang, X.C.; Marasco, W.A.; Heise, M.T.; Baric, R.S. A mouse model for MERS coronavirus-induced acute respiratory distress syndrome. Nat. Microbiol. 2016, 2, 16226. [Google Scholar] [CrossRef] [PubMed]
- De Wit, E.; Rasmussen, A.L.; Falzarano, D.; Bushmaker, T.; Feldmann, F.; Brining, D.L.; Fischer, E.R.; Martellaro, C.; Okumura, A.; Chang, J.; et al. Middle East respiratory syndrome coronavirus (MERS-CoV) causes transient lower respiratory tract infection in rhesus macaques. Proc. Natl. Acad. Sci. USA 2013, 110, 16598–16603. [Google Scholar] [CrossRef] [PubMed]
- Falzarano, D.; de Wit, E.; Rasmussen, A.L.; Feldmann, F.; Okumura, A.; Scott, D.P.; Brining, D.; Bushmaker, T.; Martellaro, C.; Baseler, L.; et al. Treatment with interferon-alpha2b and ribavirin improves outcome in MERS-CoV-infected rhesus macaques. Nat. Med. 2013, 19, 1313–1317. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Bao, L.; Deng, W.; Xu, L.; Li, F.; Lv, Q.; Yu, P.; Chen, T.; Xu, Y.; Zhu, H.; et al. An animal model of MERS produced by infection of rhesus macaques with MERS coronavirus. J. Infect. Dis. 2014, 209, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Falzarano, D.; de Wit, E.; Feldmann, F.; Rasmussen, A.L.; Okumura, A.; Peng, X.; Thomas, M.J.; van Doremalen, N.; Haddock, E.; Nagy, L.; et al. Infection with MERS-CoV causes lethal pneumonia in the common marmoset. PLoS Pathog. 2014, 10, e1004250. [Google Scholar] [CrossRef] [PubMed]
- Gillim-Ross, L.; Subbarao, K. Emerging respiratory viruses: Challenges and vaccine strategies. Clin. Microbiol. Rev. 2006, 19, 614–636. [Google Scholar] [CrossRef] [PubMed]
- Malik, M.; Elkholy, A.A.; Khan, W.; Hassounah, S.; Abubakar, A.; Minh, N.T.; Mala, P. Middle East respiratory syndrome coronavirus: Current knowledge and future considerations. East. Mediterr. Health J. 2016, 22, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Momattin, H.; Mohammed, K.; Zumla, A.; Memish, Z.A.; Al-Tawfiq, J.A. Therapeutic options for Middle East respiratory syndrome coronavirus (MERS-CoV)—Possible lessons from a systematic review of SARS-CoV therapy. Int. J. Infect. Dis. 2013, 17, e792–e798. [Google Scholar] [CrossRef] [PubMed]
- Omrani, A.S.; Saad, M.M.; Baig, K.; Bahloul, A.; Abdul-Matin, M.; Alaidaroos, A.Y.; Almakhlafi, G.A.; Albarrak, M.M.; Memish, Z.A.; Albarrak, A.M. Ribavirin and interferon alfa-2a for severe Middle East respiratory syndrome coronavirus infection: A retrospective cohort study. Lancet Infect. Dis. 2014, 14, 1090–1095. [Google Scholar] [CrossRef]
- Widagdo, W.; Okba, N.M.A.; Stalin Raj, V.; Haagmans, B.L. MERS-coronavirus: From discovery to intervention. ONE Health 2017, 3, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Abba, Y.; Hassim, H.; Hamzah, H.; Noordin, M.M. Antiviral Activity of Resveratrol against Human and Animal Viruses. Adv. Virol. 2015, 2015, 184241. [Google Scholar] [CrossRef] [PubMed]
- Campagna, M.; Rivas, C. Antiviral activity of resveratrol. Biochem. Soc. Trans. 2010, 38 Pt 1, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Drago, L.; Nicola, L.; Ossola, F.; de Vecchi, E. In vitro antiviral activity of resveratrol against respiratory viruses. J. Chemother. 2008, 20, 393–394. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.C.; Ho, C.T.; Chuo, W.H.; Li, S.; Wang, T.T.; Lin, C.C. Effective inhibition of MERS-CoV infection by resveratrol. BMC Infect. Dis. 2017, 17, 144. [Google Scholar] [CrossRef] [PubMed]
- McBride, R.; van Zyl, M.; Fielding, B.C. The coronavirus nucleocapsid is a multifunctional protein. Viruses 2014, 6, 2991–3018. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Saito, H.; Ikeda, M.; Hokari, R.; Kato, N.; Hibi, T.; Miura, S. An antioxidant resveratrol significantly enhanced replication of hepatitis C virus. World J. Gastroenterol. 2010, 16, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Berardi, V.; Ricci, F.; Castelli, M.; Galati, G.; Risuleo, G. Resveratrol exhibits a strong cytotoxic activity in cultured cells and has an antiviral action against polyomavirus: Potential clinical use. J. Exp. Clin. Cancer Res. 2009, 28, 96. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Li, Y.; Huang, C.; Ying, L.; Xue, J.; Wu, H.; Chen, Z.; Yang, Z. Resveratrol enhances HBV replication through activating Sirt1-PGC-1alpha-PPARalpha pathway. Sci. Rep. 2016, 6, 24744. [Google Scholar] [CrossRef] [PubMed]
- De Wilde, A.H.; Falzarano, D.; Zevenhoven-Dobbe, J.C.; Beugeling, C.; Fett, C.; Martellaro, C.; Posthuma, C.C.; Feldmann, H.; Perlman, S.; Snijder, E.J. Alisporivir inhibits MERS- and SARS-coronavirus replication in cell culture, but not SARS-coronavirus infection in a mouse model. Virus Res. 2017, 228, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Anand, K.; Ziebuhr, J.; Wadhwani, P.; Mesters, J.R.; Hilgenfeld, R. Coronavirus main proteinase (3CLpro) structure: Basis for design of anti-SARS drugs. Science 2003, 300, 1763–1767. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Shin, J.S.; Shie, J.J.; Ku, K.B.; Kim, C.; Go, Y.Y.; Huang, K.F.; Kim, M.; Liang, P.H. Identification and evaluation of potent Middle East respiratory syndrome coronavirus (MERS-CoV) 3CLPro inhibitors. Antivir. Res. 2017, 141, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Berry, M.; Fielding, B.; Gamieldien, J. Human coronavirus OC43 3CL protease and the potential of ML188 as a broad-spectrum lead compound: Homology modelling and molecular dynamic studies. BMC Struct. Biol. 2015, 15, 8. [Google Scholar] [CrossRef] [PubMed]
- Modjarrad, K. MERS-CoV vaccine candidates in development: The current landscape. Vaccine 2016, 34, 2982–2987. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, S.H.; McElrath, M.J.; Lewis, D.J.; del Giudice, G. Challenges and responses in human vaccine development. Curr. Opin. Immunol. 2014, 28, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Honda-Okubo, Y.; Barnard, D.; Ong, C.H.; Peng, B.H.; Tseng, C.T.; Petrovsky, N. Severe acute respiratory syndrome-associated coronavirus vaccines formulated with delta inulin adjuvants provide enhanced protection while ameliorating lung eosinophilic immunopathology. J. Virol. 2015, 89, 2995–3007. [Google Scholar] [CrossRef] [PubMed]
- Enjuanes, L.; Dediego, M.L.; Alvarez, E.; Deming, D.; Sheahan, T.; Baric, R. Vaccines to prevent severe acute respiratory syndrome coronavirus-induced disease. Virus Res. 2008, 133, 45–62. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; He, Y.; Liu, S. SARS vaccine development. Emerg. Infect. Dis. 2005, 11, 1016–1020. [Google Scholar] [CrossRef] [PubMed]
- Marshall, E.; Enserink, M. Medicine. Caution urged on SARS vaccines. Science 2004, 303, 944–946. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Lu, J. Glycan arrays lead to the discovery of autoimmunogenic activity of SARS-CoV. Physiol. Genom. 2004, 18, 245–248. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Jiang, S. Vaccine design for severe acute respiratory syndrome coronavirus. Viral Immunol. 2005, 18, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.J.; Zhao, M.; Liu, K.; Xu, K.; Wong, G.; Tan, W.; Gao, G.F. T-cell immunity of SARS-CoV: Implications for vaccine development against MERS-CoV. Antivir. Res. 2017, 137, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zheng, X.; Gai, W.; Wong, G.; Wang, H.; Jin, H.; Feng, N.; Zhao, Y.; Zhang, W.; Li, N.; et al. Novel chimeric virus-like particles vaccine displaying MERS-CoV receptor-binding domain induce specific humoral and cellular immune response in mice. Antivir. Res. 2017, 140, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Zhang, N.; Tao, X.; Zhao, G.; Guo, Y.; Tseng, C.T.; Jiang, S.; Du, L.; Zhou, Y. Optimization of antigen dose for a receptor-binding domain-based subunit vaccine against MERS coronavirus. Hum. Vaccin. Immunother. 2015, 11, 1244–1250. [Google Scholar] [CrossRef] [PubMed]
- Al-Amri, S.S.; Abbas, A.T.; Siddiq, L.A.; Alghamdi, A.; Sanki, M.A.; Al-Muhanna, M.K.; Alhabbab, R.Y.; Azhar, E.I.; Li, X.; Hashem, A.M. Immunogenicity of Candidate MERS-CoV DNA Vaccines Based on the Spike Protein. Sci. Rep. 2017, 7, 44875. [Google Scholar] [CrossRef] [PubMed]
- Jiaming, L.; Yanfeng, Y.; Yao, D.; Yawei, H.; Linlin, B.; Baoying, H.; Jinghua, Y.; Gao, G.F.; Chuan, Q.; Wenjie, T. The recombinant N-terminal domain of spike proteins is a potential vaccine against Middle East respiratory syndrome coronavirus (MERS-CoV) infection. Vaccine 2017, 35, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Perlman, S.; Vijay, R. Middle East respiratory syndrome vaccines. Int. J. Infect. Dis. 2016, 47, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.C.; Agnihothram, S.S.; Jiao, Y.; Stanhope, J.; Graham, R.L.; Peterson, E.C.; Avnir, Y.; Tallarico, A.S.; Sheehan, J.; Zhu, Q.; et al. Identification of human neutralizing antibodies against MERS-CoV and their role in virus adaptive evolution. Proc. Natl. Acad. Sci. USA 2014, 111, E2018–E2026. [Google Scholar] [CrossRef] [PubMed]
- Van Doremalen, N.; Falzarano, D.; Ying, T.; de Wit, E.; Bushmaker, T.; Feldmann, F.; Okumura, A.; Wang, Y.; Scott, D.P.; Hanley, P.W.; et al. Efficacy of antibody-based therapies against Middle East respiratory syndrome coronavirus (MERS-CoV) in common marmosets. Antivir. Res. 2017, 143, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Corti, D.; Passini, N.; Lanzavecchia, A.; Zambon, M. Rapid generation of a human monoclonal antibody to combat Middle East respiratory syndrome. J. Infect. Public Health 2016, 9, 231–235. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Luke, T.; Wu, H.; Zhao, J.; Channappanavar, R.; Coleman, C.M.; Jiao, J.A.; Matsushita, H.; Liu, Y.; Postnikova, E.N.; Ork, B.L.; et al. Human polyclonal immunoglobulin G from transchromosomic bovines inhibits MERS-CoV in vivo. Sci. Transl. Med. 2016, 8, 326ra321. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.S.; Tao, X.; Algaissi, A.; Garron, T.; Narayanan, K.; Peng, B.H.; Couch, R.B.; Tseng, C.T. Immunization with inactivated Middle East Respiratory Syndrome coronavirus vaccine leads to lung immunopathology on challenge with live virus. Hum. Vaccin. Immunother. 2016, 12, 2351–2356. [Google Scholar] [CrossRef] [PubMed]
- Volz, A.; Kupke, A.; Song, F.; Jany, S.; Fux, R.; Shams-Eldin, H.; Schmidt, J.; Becker, C.; Eickmann, M.; Becker, S.; et al. Protective Efficacy of Recombinant Modified Vaccinia Virus Ankara Delivering Middle East Respiratory Syndrome Coronavirus Spike Glycoprotein. J. Virol. 2015, 89, 8651–8656. [Google Scholar] [CrossRef] [PubMed]
- Haagmans, B.L.; van den Brand, J.M.; Raj, V.S.; Volz, A.; Wohlsein, P.; Smits, S.L.; Schipper, D.; Bestebroer, T.M.; Okba, N.; Fux, R.; et al. An orthopoxvirus-based vaccine reduces virus excretion after MERS-CoV infection in dromedary camels. Science 2016, 351, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Okada, K.; Kenniston, T.; Raj, V.S.; AlHajri, M.M.; Farag, E.A.; AlHajri, F.; Osterhaus, A.D.; Haagmans, B.L.; Gambotto, A. Immunogenicity of an adenoviral-based Middle East Respiratory Syndrome coronavirus vaccine in BALB/c mice. Vaccine 2014, 32, 5975–5982. [Google Scholar] [CrossRef] [PubMed]
- Malczyk, A.H.; Kupke, A.; Prufer, S.; Scheuplein, V.A.; Hutzler, S.; Kreuz, D.; Beissert, T.; Bauer, S.; Hubich-Rau, S.; Tondera, C.; et al. A Highly Immunogenic and Protective Middle East Respiratory Syndrome Coronavirus Vaccine Based on a Recombinant Measles Virus Vaccine Platform. J. Virol. 2015, 89, 11654–11667. [Google Scholar] [CrossRef] [PubMed]
- Wirblich, C.; Coleman, C.M.; Kurup, D.; Abraham, T.S.; Bernbaum, J.G.; Jahrling, P.B.; Hensley, L.E.; Johnson, R.F.; Frieman, M.B.; Schnell, M.J. One-Health: A Safe, Efficient, Dual-Use Vaccine for Humans and Animals against Middle East Respiratory Syndrome Coronavirus and Rabies Virus. J. Virol. 2017, 91, e02040-16. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhao, J.; Mangalam, A.K.; Channappanavar, R.; Fett, C.; Meyerholz, D.K.; Agnihothram, S.; Baric, R.S.; David, C.S.; Perlman, S. Airway Memory CD4(+) T Cells Mediate Protective Immunity against Emerging Respiratory Coronaviruses. Immunity 2016, 44, 1379–1391. [Google Scholar] [CrossRef] [PubMed]
- Mou, H.; Raj, V.S.; van Kuppeveld, F.J.; Rottier, P.J.; Haagmans, B.L.; Bosch, B.J. The receptor binding domain of the new Middle East respiratory syndrome coronavirus maps to a 231-residue region in the spike protein that efficiently elicits neutralizing antibodies. J. Virol. 2013, 87, 9379–9383. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Deng, Y.; Wen, B.; Wang, H.; Meng, X.; Lan, J.; Gao, G.F.; Tan, W. The amino acids 736–761 of the MERS-CoV spike protein induce neutralizing antibodies: Implications for the development of vaccines and antiviral agents. Viral Immunol. 2014, 27, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Lan, J.; Yao, Y.; Deng, Y.; Chen, H.; Lu, G.; Wang, W.; Bao, L.; Deng, W.; Wei, Q.; Gao, G.F.; et al. Recombinant Receptor Binding Domain Protein Induces Partial Protective Immunity in Rhesus Macaques Against Middle East Respiratory Syndrome Coronavirus Challenge. EBioMedicine 2015, 2, 1438–1446. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Channappanavar, R.; Ma, C.; Wang, L.; Tang, J.; Garron, T.; Tao, X.; Tasneem, S.; Lu, L.; Tseng, C.T.; et al. Identification of an ideal adjuvant for receptor-binding domain-based subunit vaccines against Middle East respiratory syndrome coronavirus. Cell. Mol. Immunol. 2016, 13, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Tai, W.; Zhao, G.; Sun, S.; Guo, Y.; Wang, Y.; Tao, X.; Tseng, C.K.; Li, F.; Jiang, S.; Du, L.; et al. A recombinant receptor-binding domain of MERS-CoV in trimeric form protects human dipeptidyl peptidase 4 (hDPP4) transgenic mice from MERS-CoV infection. Virology 2016, 499, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Li, Y.; Wang, L.; Zhao, G.; Tao, X.; Tseng, C.T.; Zhou, Y.; Du, L.; Jiang, S. Intranasal vaccination with recombinant receptor-binding domain of MERS-CoV spike protein induces much stronger local mucosal immune responses than subcutaneous immunization: Implication for designing novel mucosal MERS vaccines. Vaccine 2014, 32, 2100–2108. [Google Scholar] [CrossRef] [PubMed]
- Muthumani, K.; Falzarano, D.; Reuschel, E.L.; Tingey, C.; Flingai, S.; Villarreal, D.O.; Wise, M.; Patel, A.; Izmirly, A.; Aljuaid, A.; et al. A synthetic consensus anti-spike protein DNA vaccine induces protective immunity against Middle East respiratory syndrome coronavirus in nonhuman primates. Sci. Transl. Med. 2015, 7, 301ra132. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Shi, W.; Joyce, M.G.; Modjarrad, K.; Zhang, Y.; Leung, K.; Lees, C.R.; Zhou, T.; Yassine, H.M.; Kanekiyo, M.; et al. Evaluation of candidate vaccine approaches for MERS-CoV. Nat. Commun. 2015, 6, 7712. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zheng, X.; Gai, W.; Zhao, Y.; Wang, H.; Wang, H.; Feng, N.; Chi, H.; Qiu, B.; Li, N.; et al. MERS-CoV virus-like particles produced in insect cells induce specific humoural and cellular imminity in rhesus macaques. Oncotarget 2017, 8, 12686–12694. [Google Scholar] [CrossRef] [PubMed]
- Coleman, C.M.; Liu, Y.V.; Mu, H.; Taylor, J.K.; Massare, M.; Flyer, D.C.; Smith, G.E.; Frieman, M.B. Purified coronavirus spike protein nanoparticles induce coronavirus neutralizing antibodies in mice. Vaccine 2014, 32, 3169–3174. [Google Scholar] [CrossRef] [PubMed]
- Coleman, C.M.; Venkataraman, T.; Liu, Y.V.; Glenn, G.M.; Smith, G.E.; Flyer, D.C.; Frieman, M.B. MERS-CoV spike nanoparticles protect mice from MERS-CoV infection. Vaccine 2017, 35, 1586–1589. [Google Scholar] [CrossRef] [PubMed]
- Cotten, M.; Watson, S.J.; Zumla, A.I.; Makhdoom, H.Q.; Palser, A.L.; Ong, S.H.; Al Rabeeah, A.A.; Alhakeem, R.F.; Assiri, A.; Al-Tawfiq, J.A.; et al. Spread, circulation, and evolution of the Middle East respiratory syndrome coronavirus. MBio 2014, 5, e01062-13. [Google Scholar] [CrossRef] [PubMed]
- Borucki, M.K.; Lao, V.; Hwang, M.; Gardner, S.; Adney, D.; Munster, V.; Bowen, R.; Allen, J.E. Middle East Respiratory Syndrome Coronavirus Intra-Host Populations Are Characterized by Numerous High Frequency Variants. PLoS ONE 2016, 11, e0146251. [Google Scholar]
- Gardner, L.M.; Rey, D.; Heywood, A.E.; Toms, R.; Wood, J.; Travis Waller, S.; Raina MacIntyre, C. A scenario-based evaluation of the Middle East respiratory syndrome coronavirus and the Hajj. Risk Anal. 2014, 34, 1391–1400. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.I.; Kim, Y.J.; Lemey, P.; Lee, I.; Park, S.; Bae, J.Y.; Kim, D.; Kim, H.; Jang, S.I.; Yang, J.S.; et al. The recent ancestry of Middle East respiratory syndrome coronavirus in Korea has been shaped by recombination. Sci. Rep. 2016, 6, 18825. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, M.J.; Brown, J.R.; Amrine-Madsen, H. Evidence from the evolutionary analysis of nucleotide sequences for a recombinant history of SARS-CoV. Infect. Genet. Evol. 2004, 4, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.C.; Rambaut, A. Viral evolution and the emergence of SARS coronavirus. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2004, 359, 1059–1065. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.C.; Drummond, A.J. The evolutionary genetics of viral emergence. Curr. Top. Microbiol. Immunol. 2007, 315, 51–66. [Google Scholar] [PubMed]
- Worobey, M.; Han, G.Z.; Rambaut, A. A synchronized global sweep of the internal genes of modern avian influenza virus. Nature 2014, 508, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Almazan, F.; Marquez-Jurado, S.; Nogales, A.; Enjuanes, L. Engineering infectious cDNAs of coronavirus as bacterial artificial chromosomes. Methods Mol. Biol. 2015, 1282, 135–152. [Google Scholar] [PubMed]
- Fielding, B.C. Human coronavirus NL63: A clinically important virus? Future Microbiol. 2011, 6, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, E.F.; Yount, B.; Sims, A.C.; Burkett, S.; Pickles, R.J.; Baric, R.S. Systematic assembly of a full-length infectious clone of human coronavirus NL63. J. Virol. 2008, 82, 11948–11957. [Google Scholar] [CrossRef] [PubMed]
- Madani, T.A. Case definition and management of patients with MERS coronavirus in Saudi Arabia. Lancet Infect. Dis. 2014, 14, 911–913. [Google Scholar] [CrossRef]
- Hui, D.S. Super-spreading events of MERS-CoV infection. Lancet 2016, 388, 942–943. [Google Scholar] [CrossRef]
- Zumla, A.; Hui, D.S. Infection control and MERS-CoV in health-care workers. Lancet 2014, 383, 1869–1871. [Google Scholar] [CrossRef]
| Method Used for Detection | 1 Sensitivity/2 Specificity/3 Viral Target Gene | Reference |
|---|---|---|
| rtRT-PCR | 1 Sensitivity for upE is 3.4 copies per reaction (95% confidence interval (CI): 2.5–6.9 copies) or 291 copies/mL of sample. 2 No cross-reactivity was observed with coronaviruses OC43, NL63, 229E, SARS-CoV, nor with 92 clinical specimens containing common human respiratory viruses. 3 Targeting regions upstream of the E gene (upE) or within open reading frame (ORF) 1b, respectively. | [75] |
| qRT-PCR # | 1 Sensitivity to widely used upE gene as well as a ORF1a&b was introduced 2 No false-positive amplifications were obtained with other human coronaviruses or common respiratory viral pathogens or with 336 diverse clinical specimens from non-MERS-CoV cases; specimens from two confirmed MERS-CoV cases were positive with all assay signatures. 3 Two novel signatures used one that targets the MERS-CoV N gene in combination with the upE test. The other a positive test to add to an efficient MERS-CoV kit. | [81] |
| RT-Sequence-Validated-LAMP Assays | 1 Could detect 0.02 to 0.2 plaque forming units (PFU) (5 to 50 PFU/mL) of MERS-CoV in infected cell culture supernatants. 2 Did not cross-react with common human respiratory pathogens. | [88] |
| RT-LAMP | 1 Capable of detecting as few as 3.4 copies of MERS-CoV RNA; Assay exhibited sensitivity similar to that of MERS-CoV real-time RT-PCR. 2 No cross-reaction to other respiratory viruses. 3 Assay designed to amplify the MERS-CoV gene | [80] |
| rt-RPA | 1 Highly sensitive, is able to detect 10 MERS-CoV RNA copies with a more rapid detection time than MERS-RT-PCR. 2 No cross-reaction to other respiratory viruses including HCoVs. 3 Assay designed to amplify the partial nucleocapsid gene of MERS-CoV | [89] |
| mAb Test | 1 Rapid detection and cost effective ELISA 2 High specificity used to detect the MERS-CoV nucleocapsid protein | [87] |
| Immuno-chromotagraphic tool | 1 Highly sensitive, 2 No cross reactivity with other respiratory pathogens observed in vitro and in silico 3 Detects recombinant MERS-CoV N protein | [90] |
| Immunofluorescence Assay | 1 Highly sensitive, antigen based detection 2 Cross reactivity seen with convalescent SARS patient (sera) 3 Assay used both whole virus and S1 portion of the spike protein | [91,92,93] |
| ppNT Assay | 1 Highly sensitive, more sensitive that MNT test 2 Lack of MERS neutralizing activity indicated high specificity by this assay. No cross reactivity seen with SARS-CoV 3 Assay was designed for two different genes used: a codon optimized spike gene and a HIV/MERS pseudoparticle was generated | [94,95] |
| MNT Test | 1 Highly sensitive; less so than ppNT assay 2 Highly specific, as SARS-CoV antigen was not detected compared to MERS-CoV. 3 Test designed to detect IgG antibodies generated when using the RBD of the S1 subunit of the spike gene | [94,96,97] |
| Protein Microarray | 1 Highly sensitive assay using protein microarray technology to detect IgG and IgM antibodies 2 No cross reactivity seen with sera of patients that had been exposed to four common HCoVs. 3 Assay designed to use the S1 receptor-binding subunit of the spike protein of MERS and SARS as antigens. | [98] |
| One pot RT-LAMP | 1 Capable of detecting four viral copies MERS within 60 min 2 No cross-reaction to the other acute respiratory disease viruses (influenza type A virus (H1N1 and H3N2), influenza type B virus, HCoV-229E, and human metapneumovirus) 3 Six sets of primers designed specifically to amplify the MERS-CoV genes | [99] |
| RT-iiPCR assays | 1 Could detect 3.7 × 10−1 plaque forming units (PFU) of MERS-CoV in infected cell culture supernatants and sputum samples. 2 Viral nucleic acids extracted from infected cultures that contained HCoV-229E, HCoV-OC43, FIPV, influenza type A and B virus strains yielded negative results, indicating no cross reactivity. 3 Targeting regions upstream of the E gene (upE) or within open reading frame (ORF) 1b | [100] |
| Powerchek MERS Assay | 1 95% limits of detection of assay for the upE and ORF1a were 16.2 copies/μL and 8.2 copies/μL, respectively. 2 No cross reactivity with other respiratory pathogens observed in vitro and in silico 3 Targeting regions upstream of the E gene (upE) or within open reading frame (ORF) 1b | [101] |
| acpcPNA-AgNP aggregation assay | 1 Probe designed for targets makes this assay highly specific. Limit of detection found to be 1.53 nM 2 Cross reactivity with other CoVs was not evaluated 3 Synthetic oligonucleotides were designed to target MERS | [102] |
| mCoV-MS | 1 Highly sensitive, multiplex PCR based to target specific genes in HcoVs 2 Cross reactivity with other respiratory pathogens was not evaluated 3 Targeting regions upstream of the E gene (upE) or within open reading frame (ORF) 1b | [103] |
| Duplex-RT-PCR method | 1 Highly sensitive, simultaneous detection of MERS and SARS viruses. 2 Cross reactivity with other respiratory pathogens was not evaluated 3 Primers and probes that target the conserved spike S2 region of SARS-CoV, MERS-CoV, and their related bat CoVs were used | [104] |
| Vaccine Categories | Target Antigen | Immunization | Animal Model | Immunogenicity | Stage of Development | Reference |
|---|---|---|---|---|---|---|
| Anti-MERS-CoV monoclonal antibodies | Surface (S) glycoprotein | Passive | marmosets | Animals developed pneumonia, high viral titre detected in lungs | Preclinical: in vivo, efficacy stage | [149,150,151] |
| Human polyclonal anti-MERS-CoV antibodies | Virus structural proteins | Passive | Ad5-hDPP4-transduced mouse | Nab developed to reduce viral titres post exposure | Preclinical: in vivo, efficacy stage | [152] |
| Inactivated virion vaccines | MERS-CoV | Active | hDPP4-transgenic mice | Nab produced without adjuvant, T-cell response not done | Preclinical: in vivo, efficacy stage | [153] |
| Live attenuated vaccines (deleted E protein; mutated in nsp14) | rMERS-CoV-∆E | Active | Not tested | Not indicated | Preclinical development: in vitro | [20] |
| Recombinant viral vectors (MVA, Adenovirus, Parainfluenza virus, Measles, Rabies) | S and SolS proteins | Active | Ad/hDPP4-mice Camels | Nab in mice, antigen specific humoral and in some case T cell immune responses | Preclinical: in vitro, efficacy stage | [145,154,155,156,157,158] |
| Replicon particles (e.g., Venezuelan (VRP-S) | S protein | Active | Ad/hDPP4-mice mice | Nab produced, mice developed progressive pneumonia with virus replication detected in airways | Preclinical: in vivo, efficacy stage | [110,159] |
| Subunit vaccines RBDs rRBDs RBDs-Fc rNTDs | S/S1protein with various amino acid residues | Active | -hDPP4-transgenic-Ad5-hDPP4 mice Rabbit NHPs | High mucosal and humoral immune response, strong Nab in mice and rabbits. Good T-cell response in mice. Tg-Mice protected from MERS-CoV | Preclinical: in vitro, efficacy stage | [147,160,161,162,163,164,165] |
| DNA vaccines | S protein | Active | NHP:Rhesus Macaques Camels Mice | Cellular immune response and Nab response in mice, NHPs and camels. | Phase 1 clinical trials | [166] |
| DNA prime/Protein-boost Vaccines | S and S1 protein | Active | NHP:Rhesus Macaques Mice | Nab response seen in mice and NHPs | Preclinical: in vitro, efficacy stage | [167] |
| VLPs | S, M, E | Active | NHP:Rhesus Macaques | Virus specific Nab and IgG antibody response against the RBD | Preclinical: in vivo, efficacy stage | [168] |
| Nanoparticle vaccine | S protein | Active | Mice | Nab with the presence of adjuvant (M1 and Alum) | Preclinical: in vivo, efficacy stage | [169,170] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chafekar, A.; Fielding, B.C. MERS-CoV: Understanding the Latest Human Coronavirus Threat. Viruses 2018, 10, 93. https://doi.org/10.3390/v10020093
Chafekar A, Fielding BC. MERS-CoV: Understanding the Latest Human Coronavirus Threat. Viruses. 2018; 10(2):93. https://doi.org/10.3390/v10020093
Chicago/Turabian StyleChafekar, Aasiyah, and Burtram C. Fielding. 2018. "MERS-CoV: Understanding the Latest Human Coronavirus Threat" Viruses 10, no. 2: 93. https://doi.org/10.3390/v10020093
APA StyleChafekar, A., & Fielding, B. C. (2018). MERS-CoV: Understanding the Latest Human Coronavirus Threat. Viruses, 10(2), 93. https://doi.org/10.3390/v10020093






