Combination Therapy with Oseltamivir and Favipiravir Delays Mortality but Does Not Prevent Oseltamivir Resistance in Immunodeficient Mice Infected with Pandemic A(H1N1) Influenza Virus
Abstract
1. Introduction
2. Materials and Methods
2.1. Virus and Cells
2.2. Antiviral Compounds
2.3. Animals
2.4. Immunosuppression Regimen
2.5. Experimental Design
2.5.1. Pandemic A(H1N1) Infection in IS Mice
2.5.2. Immunosuppression Regimen
2.5.3. Antiviral Studies with Oseltamivir, Favipiravir, or the Combination
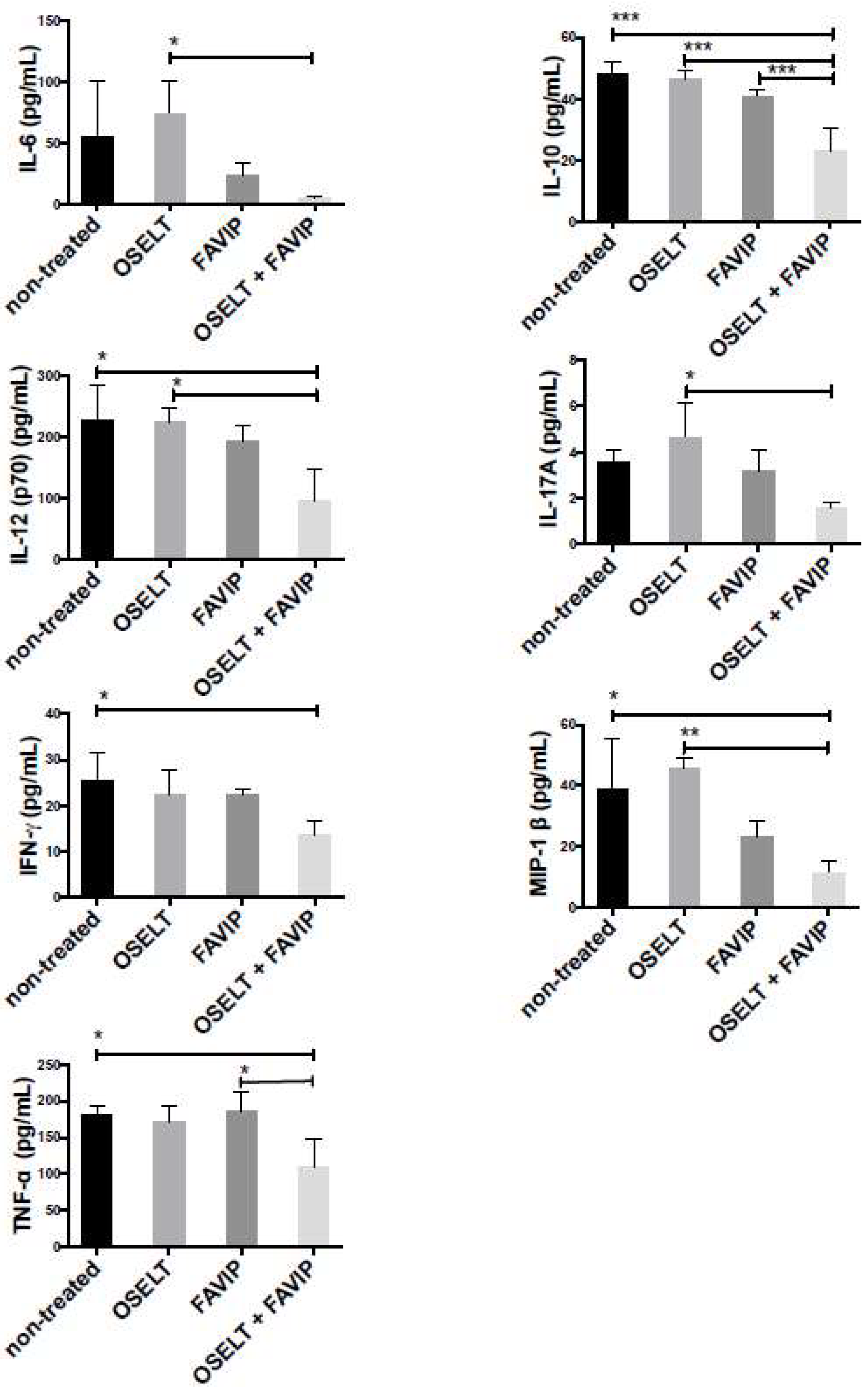
2.6. Cytokine and Chemokine Analysis
2.7. Reverse-Transcription Droplet Digital PCR (RT-ddPCR)
2.8. RT-PCR Amplification and Sanger Sequencing
2.9. Susceptibility Assays
2.10. Statistical Analyses
3. Results
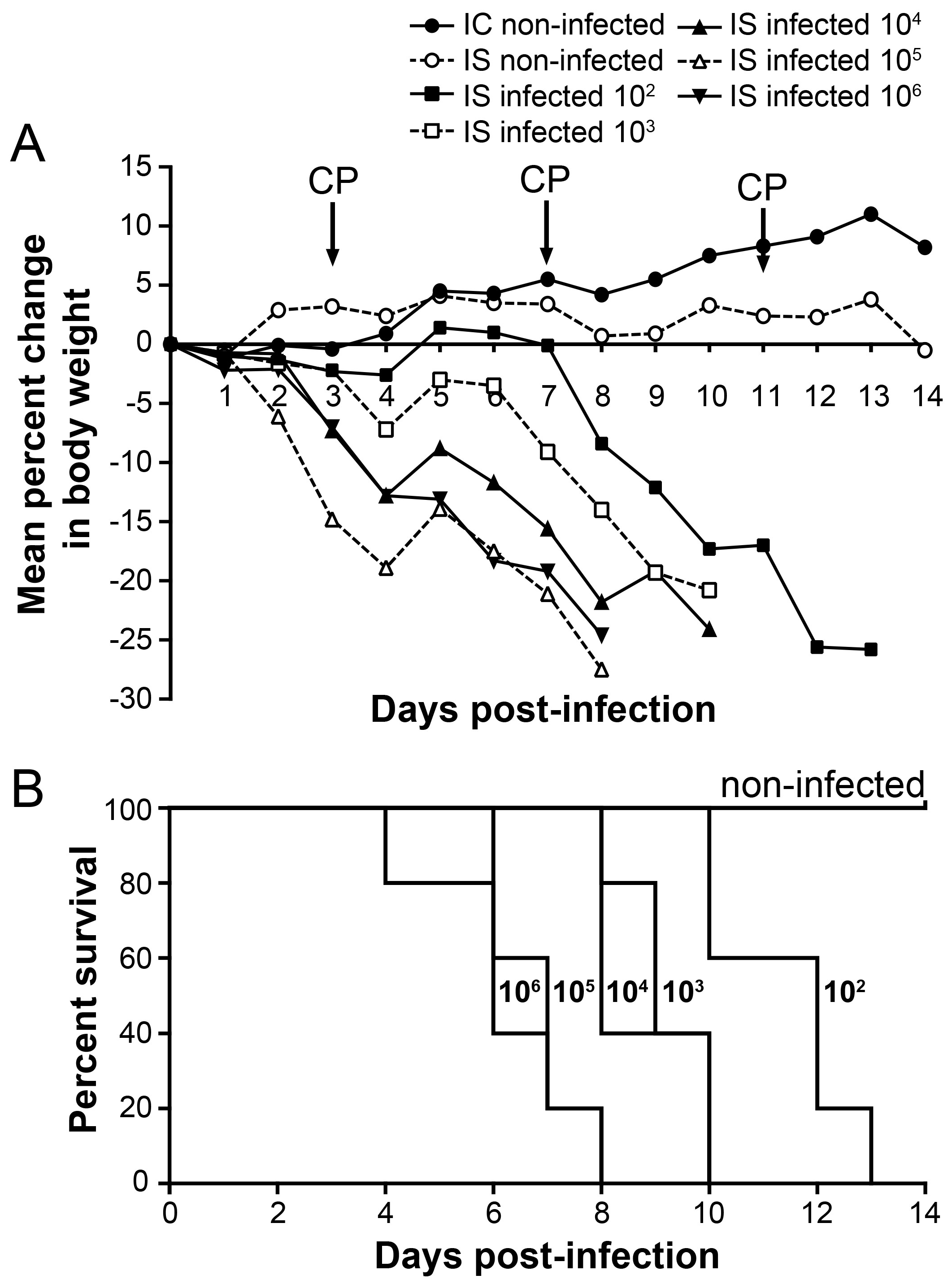
3.1. Pandemic A(H1N1) Infection in IS Mice
3.2. Immunosuppression Regimen
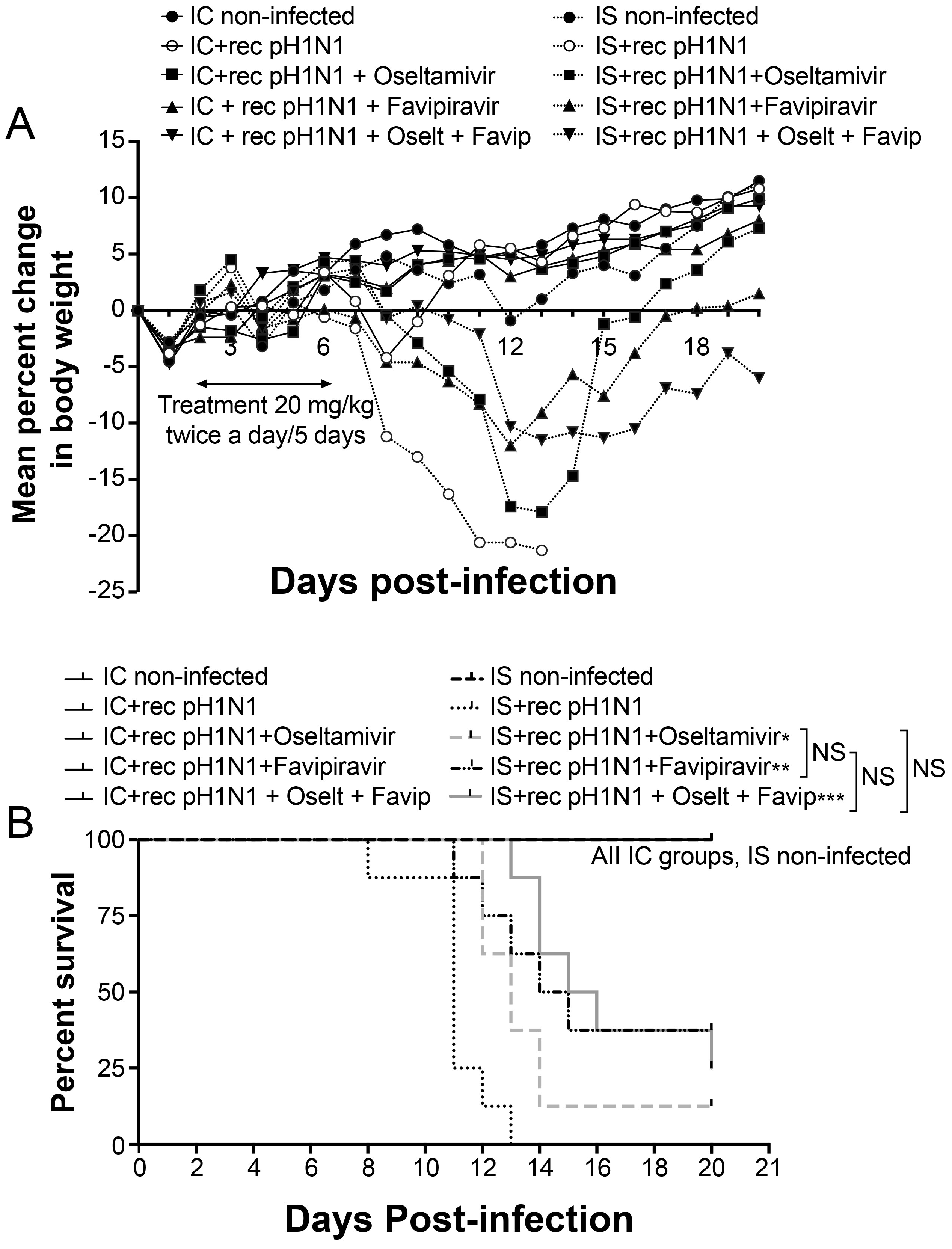
3.3. Five-Day Single and Combined Therapies in IS and IC Mice
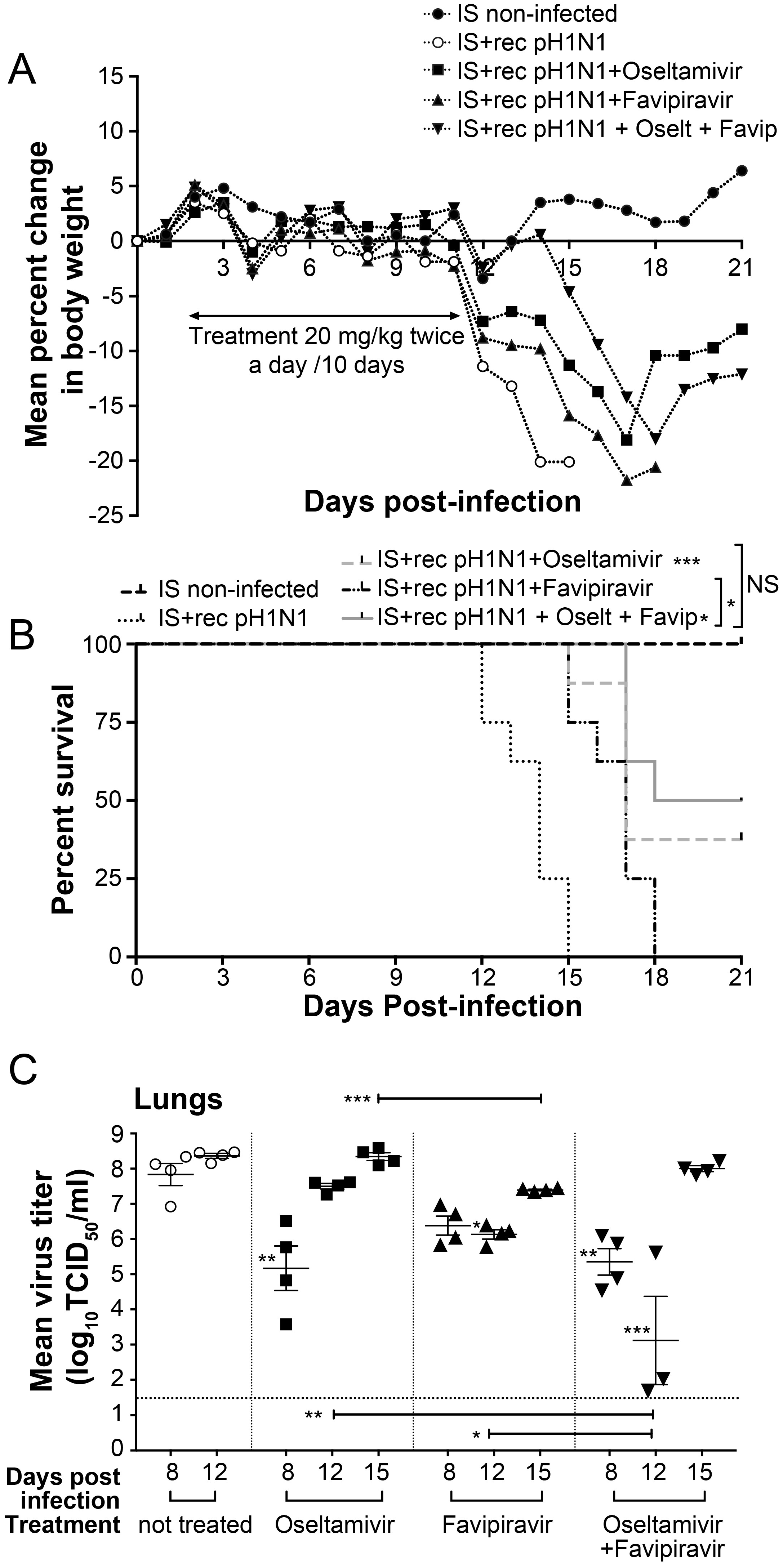
3.4. Ten-Day Single and Combined Therapies with Low Dose of Favipiravir in IS Mice
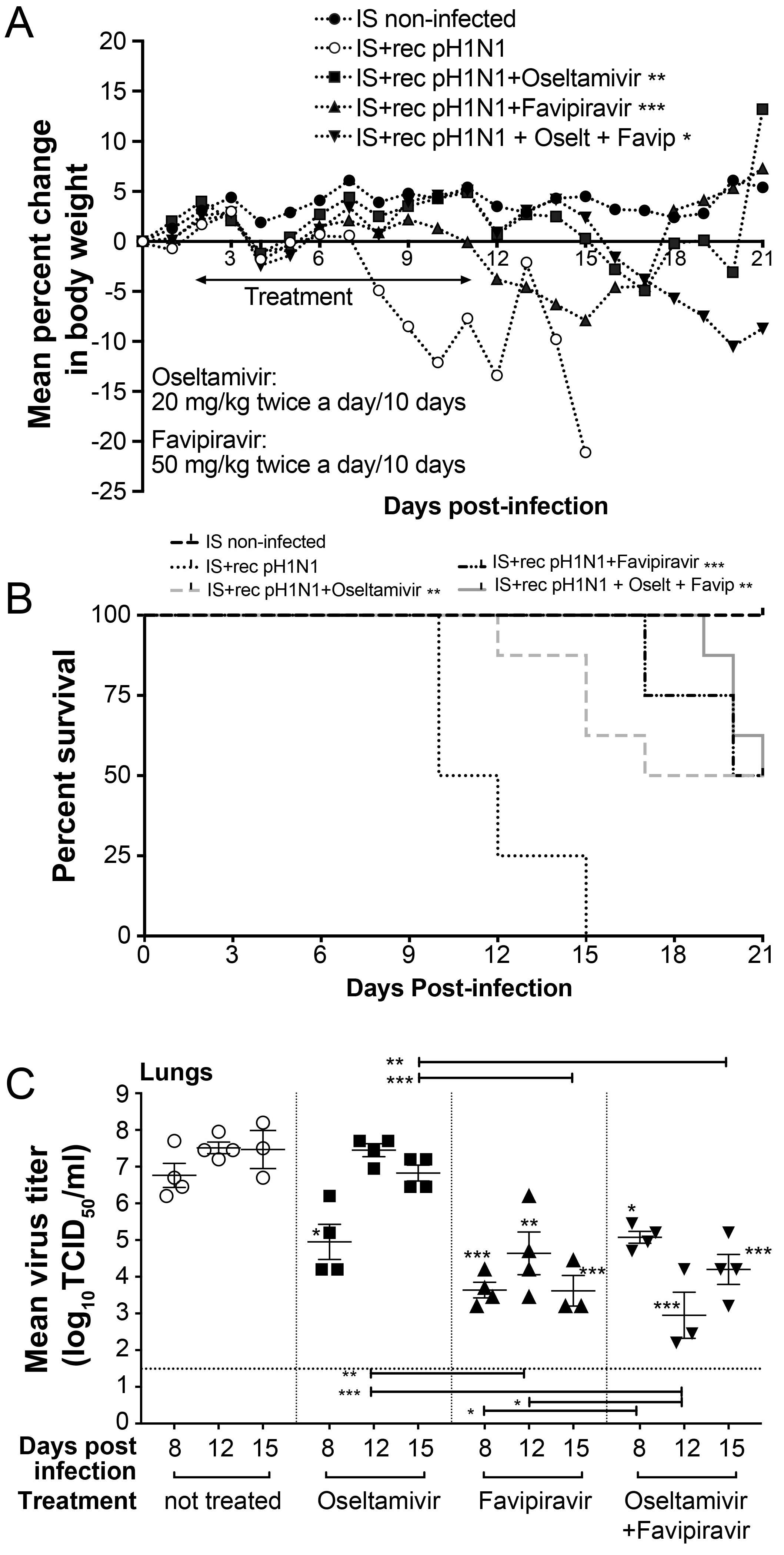
3.5. Ten-Day Single and Combined Therapies with High Dose of Favipiravir in IS Mice
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Casper, C.; Englund, J.; Boeckh, M. How I treat influenza in patients with hematologic malignancies. Blood 2010, 115, 1331–1342. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.C.; Nichol, K.L. Excess mortality due to pneumonia or influenza during influenza seasons among persons with acquired immunodeficiency syndrome. Arch. Intern. Med. 2001, 161, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Radwan, H.M.; Cheeseman, S.H.; Lai, K.K.; Ellison, I.R. Influenza in human immunodeficiency virus-infected patients during the 1997–1998 influenza season. Clin. Infect. Dis. 2000, 31, 604–606. [Google Scholar] [CrossRef] [PubMed]
- Khanna, N.; Steffen, I.; Studt, J.D.; Schreiber, A.; Lehmann, T.; Weisser, M.; Fluckiger, U.; Gratwohl, A.; Halter, J.; Hirsch, H.H. Outcome of influenza infections in outpatients after allogeneic hematopoietic stem cell transplantation. Transpl. Infect. Dis. 2009, 11, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Baz, M.; Abed, Y.; McDonald, J.; Boivin, G. Characterization of multidrug-resistant influenza A/H3N2 viruses shed during 1 year by an immunocompromised child. Clin. Infect. Dis. 2006, 43, 1555–1561. [Google Scholar] [CrossRef] [PubMed]
- Baz, M.; Abed, Y.; Papenburg, J.; Bouhy, X.; Hamelin, M.E.; Boivin, G. Emergence of oseltamivir-resistant pandemic H1N1 virus during prophylaxis. N. Engl. J. Med. 2009, 361, 2296–2297. [Google Scholar] [CrossRef] [PubMed]
- L’Huillier, A.G.; Abed, Y.; Petty, T.J.; Cordey, S.; Thomas, Y.; Bouhy, X.; Schibler, M.; Simon, A.; Chalandon, Y.; van Delden, C.; et al. E119D Neuraminidase Mutation Conferring Pan-Resistance to Neuraminidase Inhibitors in an A(H1N1)pdm09 Isolate From a Stem-Cell Transplant Recipient. J. Infect. Dis. 2015, 212, 1726–1734. [Google Scholar] [CrossRef] [PubMed]
- Eshaghi, A.; Shalhoub, S.; Rosenfeld, P.; Li, A.; Higgins, R.R.; Stogios, P.J.; Savchenko, A.; Bastien, N.; Li, Y.; Rotstein, C.; et al. Multiple influenza A (H3N2) mutations conferring resistance to neuraminidase inhibitors in a bone marrow transplant recipient. Antimicrob. Agents Chemother. 2014, 58, 7188–7197. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Galvin, H.D.; Haw, T.Y.; Nutsford, A.N.; Husain, M. Drug resistance in influenza A virus: The epidemiology and management. Infect. Drug Resist. 2017, 10, 121–134. [Google Scholar] [CrossRef] [PubMed]
- Furuta, Y.; Takahashi, K.; Fukuda, Y.; Kuno, M.; Kamiyama, T.; Kozaki, K.; Nomura, N.; Egawa, H.; Minami, S.; Watanabe, Y.; et al. In vitro and in vivo activities of anti-influenza virus compound T-705. Antimicrob. Agents Chemother. 2002, 46, 977–981. [Google Scholar] [CrossRef] [PubMed]
- Furuta, Y.; Takahashi, K.; Shiraki, K.; Sakamoto, K.; Smee, D.F.; Barnard, D.L.; Gowen, B.B.; Julander, J.G.; Morrey, J.D. T-705 (favipiravir) and related compounds: Novel broad-spectrum inhibitors of RNA viral infections. Antiviral. Res. 2009, 82, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Furuta, Y.; Gowen, B.B.; Takahashi, K.; Shiraki, K.; Smee, D.F.; Barnard, D.L. Favipiravir (T-705), a novel viral RNA polymerase inhibitor. Antiviral. Res. 2013, 100, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Smith, L.K.; Rajwanshi, V.K.; Kim, B.; Deval, J. The ambiguous base-pairing and high substrate efficiency of T-705 (Favipiravir) Ribofuranosyl 5’-triphosphate towards influenza A. virus polymerase. PLoS ONE 2013, 8, e68347. [Google Scholar] [CrossRef] [PubMed]
- Tarbet, E.B.; Vollmer, A.H.; Hurst, B.L.; Barnard, D.L.; Furuta, Y.; Smee, D.F. In vitro activity of favipiravir and neuraminidase inhibitor combinations against oseltamivir-sensitive and oseltamivir-resistant pandemic influenza A. (H1N1) virus. Arch. Virol. 2014, 159, 1279–1291. [Google Scholar] [CrossRef] [PubMed]
- Govorkova, E.A.; Webster, R.G. Combination chemotherapy for influenza. Viruses 2010, 2, 1510–1529. [Google Scholar] [CrossRef] [PubMed]
- Kiso, M.; Lopes, T.J.S.; Yamayoshi, S.; Ito, M.; Yamashita, M.; Nakajima, N.; Hasegawa, H.; Neumann, G.; Kawaoka, Y. Combination Therapy With Neuraminidase and Polymerase Inhibitors in Nude Mice Infected With Influenza Virus. J. Infect. Dis. 2018, 217, 887–896. [Google Scholar] [CrossRef] [PubMed]
- Pizzorno, A.; Bouhy, X.; Abed, Y.; Boivin, G. Generation and characterization of recombinant pandemic influenza A(H1N1) viruses resistant to neuraminidase inhibitors. J. Infect. Dis. 2011, 203, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Hurd, J.; Heath, R.B. Effect of cyclophosphamide on infections in mice caused by virulent and avirulent strains of influenza virus. Infect. Immun. 1975, 11, 886–889. [Google Scholar] [PubMed]
- Mastino, A.; Grelli, S.; Premrov, M.G.; Favalli, C. Susceptibility to influenza A virus infection in mice immunosuppressed with cyclophosphamide. J. Chemother. 1991, 3, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Sidwell, R.W.; Bailey, K.W.; Morrey, J.D.; Wong, M.H.; Baldwin, T.J.; Smee, D.F. Inhibition of influenza virus infections in immunosuppressed mice with orally administered peramivir (BCX-1812). Antiviral. Res. 2003, 60, 17–25. [Google Scholar] [CrossRef]
- Smee, D.; Burger, R.; Warren, R.; Bailey, K.; Sidwell, R. An Immunosuppressed Mouse Model of Lethal Murine Gammaherpesvirus 68 Infection for Studying Potential Treatment of Epstein-Barr Virus Infection in Man. Antivir. Chem. Chemother. 1997, 8, 573–581. [Google Scholar] [CrossRef]
- Berd, D.; Maguire, J.H.C.; Mastrangelo, M.J. Immunopotentiation by cyclophosphamide and other cytotoxic agents. In Immune Modulation Agents and Their Mechanisms; Fenichel, R.L., Chirigos, M.A., Eds.; Marcel Dekker: New York, NY, USA, 1984; pp. 39–61. [Google Scholar]
- Hatakeyama, S.; Sakai-Tagawa, Y.; Kiso, M.; Goto, H.; Kawakami, C.; Mitamura, K.; Sugaya, N.; Suzuki, Y.; Kawaoka, Y. Enhanced expression of an α2,6-linked sialic acid on MDCK cells improves isolation of human influenza viruses and evaluation of their sensitivity to a neuraminidase inhibitor. J. Clin. Microbiol. 2005, 43, 4139–4146. [Google Scholar] [CrossRef] [PubMed]
- Baz, M.; Boonnak, K.; Paskel, M.; Santos, C.; Powell, T.; Townsend, A.; Subbarao, K. Nonreplicating influenza A virus vaccines confer broad protection against lethal challenge. MBio 2015, 6, e01487-15. [Google Scholar] [CrossRef] [PubMed]
- Reed, L.; Muench, H. A simple method of estimating fifty percent endpoints. Am. J. Hyg. 1938, 27, 493–497. [Google Scholar]
- Taylor, S.C.; Carbonneau, J.; Shelton, D.N.; Boivin, G. Optimization of Droplet Digital PCR from RNA and DNA extracts with direct comparison to RT-qPCR: Clinical implications for quantification of Oseltamivir-resistant subpopulations. J. Virol. Methods 2015, 224, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Baz, M.; Abed, Y.; Boivin, G. Characterization of drug-resistant recombinant influenza A/H1N1 viruses selected in vitro with peramivir and zanamivir. Antiviral. Res. 2007, 74, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Stephen, P.; Baz, M.; Boivin, G.; Lin, S.X. Structural Insight into NS5 of Zika Virus Leading to the Discovery of MTase Inhibitors. J. Am. Chem. Soc. 2016, 138, 16212–16215. [Google Scholar] [CrossRef] [PubMed]
- Ison, M.G.; Gubareva, L.V.; Atmar, R.L.; Treanor, J.; Hayden, F.G. Recovery of drug-resistant influenza virus from immunocompromised patients: A case series. J. Infect. Dis. 2006, 193, 760–764. [Google Scholar] [CrossRef] [PubMed]
- Carr, S.; Ilyushina, N.A.; Franks, J.; Adderson, E.E.; Caniza, M.; Govorkova, E.A.; Webster, R.G. Oseltamivir-resistant influenza A and B viruses pre- and postantiviral therapy in children and young adults with cancer. Pediatr. Infect. Dis. J. 2011, 30, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Kossyvakis, A.; Mentis, A.A.; Tryfinopoulou, K.; Pogka, V.; Kalliaropoulos, A.; Antalis, E.; Lytras, T.; Meijer, A.; Tsiodras, S.; Karakitsos, P.; et al. Antiviral susceptibility profile of influenza A viruses; keep an eye on immunocompromised patients under prolonged treatment. Eur. J. Clin. Microbiol. Infect. Dis. 2017, 36, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Beigel, J.H.; Bao, Y.; Beeler, J.; Manosuthi, W.; Slandzicki, A.; Dar, S.M.; Panuto, J.; Beasley, R.L.; Perez-Patrigeon, S.; Suwanpimolkul, G.; et al. Oseltamivir, amantadine, and ribavirin combination antiviral therapy versus oseltamivir monotherapy for the treatment of influenza: A multicentre, double-blind, randomised phase 2 trial. Lancet Infect. Dis. 2017, 17, 1255–1265. [Google Scholar] [CrossRef]
- Hayden, F.G.; Treanor, J.J.; Fritz, R.S.; Lobo, M.; Betts, R.F.; Miller, M.; Kinnersley, N.; Mills, R.G.; Ward, P.; Straus, S.E. Use of the oral neuraminidase inhibitor oseltamivir in experimental human influenza: Randomized controlled trials for prevention and treatment. JAMA 1999, 282, 1240–1246. [Google Scholar] [CrossRef] [PubMed]
- Takashita, E.; Ejima, M.; Ogawa, R.; Fujisaki, S.; Neumann, G.; Furuta, Y.; Kawaoka, Y.; Tashiro, M.; Odagiri, T. Antiviral susceptibility of influenza viruses isolated from patients pre- and post-administration of favipiravir. Antiviral Res. 2016, 132, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Tamura, D.; DeBiasi, R.L.; Okomo-Adhiambo, M.; Mishin, V.P.; Campbell, A.P.; Loechelt, B.; Wiedermann, B.L.; Fry, A.M.; Gubareva, L.V. Emergence of Multidrug-Resistant Influenza A(H1N1)pdm09 Virus Variants in an Immunocompromised Child Treated With Oseltamivir and Zanamivir. J. Infect. Dis. 2015, 212, 1209–1213. [Google Scholar] [CrossRef] [PubMed]
- Ison, M.G.; Mishin, V.P.; Braciale, T.J.; Hayden, F.G.; Gubareva, L.V. Comparative activities of oseltamivir and A-322278 in immunocompetent and immunocompromised murine models of influenza virus infection. J. Infect. Dis. 2006, 193, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Van der Vries, E.; Stittelaar, K.J.; van Amerongen, G.; Veldhuis Kroeze, E.J.; de Waal, L.; Fraaij, P.L.; Meesters, R.J.; Luider, T.M.; van der Nagel, B.; Koch, B.; et al. Prolonged influenza virus shedding and emergence of antiviral resistance in immunocompromised patients and ferrets. PLoS Pathog. 2013, 9, e1003343. [Google Scholar] [CrossRef] [PubMed]
- Stevaert, A.; Naesens, L. The Influenza Virus Polymerase Complex: An Update on Its Structure, Functions, and Significance for Antiviral Drug Design. Med. Res. Rev. 2016, 36, 1127–1173. [Google Scholar] [CrossRef] [PubMed]
| NAI Assay (Oseltamivir) | PRA Assay (Favipiravir) | ddPCR (Mut 275Y) | |||||
|---|---|---|---|---|---|---|---|
| Mouse # | Days of Treatment | IC50 (nM) | Mean IC50 (nM) | IC50 (µM) | Mean IC50 (µM) | % | Mean % |
| Not treated | |||||||
| 1 | 12 | 0.3 | 0.3 ± 0.1 | 13.8 | 10.9 ± 1.7 | 0.0 | 0.0 ± 0.0 |
| 2 | 0.2 | 10.5 | 0.0 | ||||
| 3 | 0.3 | 9.5 | 0.0 | ||||
| 4 | 0.4 | 9.9 | 0.0 | ||||
| Oseltamivir 20 mg/kg/BID | |||||||
| 1 | 8 | 0.5 | 0.6 ± 0.0 | ND | ND | 0.0 | 1.1 ± 1.0 |
| 2 | 0.6 | ND | 2.3 | ||||
| 3 | 0.6 | ND | 0.1 | ||||
| 4 | 0.6 | ND | 1.9 | ||||
| 1 | 12 | 0.4 | 0.5 ± 0.0 | ND | ND | 0.5 | 5.8 ± 8.3 |
| 2 | 0.4 | ND | 0.5 | ||||
| 3 | 0.5 | ND | 20.1 | ||||
| 4 | 0.5 | ND | 2.0 | ||||
| 1 | 15 | 0.4 | 0.3 ± 0.1 | ND | ND | 5.5 | 1.8 ± 2.2 |
| 2 | 0.2 | ND | 0.3 | ||||
| 3 | 0.2 | ND | 0.1 | ||||
| 4 | 0.4 | ND | 1.4 | ||||
| Favipiravir 20 mg/kg/BID | |||||||
| 1 | 8 | NA | NA | ND | ND | NA | NA |
| 2 | NA | ND | NA | ||||
| 3 | NA | ND | NA | ||||
| 4 | NA | ND | NA | ||||
| 1 | 12 | NA | NA | ND | ND | NA | ND |
| 2 | NA | ND | NA | ||||
| 3 | NA | ND | NA | ||||
| 4 | ND | ND | ND | ||||
| 1 | 15 | NA | NA | 12.8 | 12.0 ± 1.0 | NA | NA |
| 2 | NA | 11.5 | NA | ||||
| 3 | NA | 10.6 | NA | ||||
| 4 | NA | 13 | NA | ||||
| Oseltamivir 20 mg/kg + Favipiravir 20 mg/kg/BID | |||||||
| 1 | 8 | 0.5 | 0.6 ± 0.1 | ND | ND | 0.4 | 1.1 ± 1.0 |
| 2 | 0.4 | ND | 0.1 | ||||
| 3 | 0.6 | ND | 1.2 | ||||
| 4 | 0.8 | ND | 2.7 | ||||
| 1 | 12 | 0.7 | 0.6 ± 0.1 | ND | ND | 7.3 | 2.9 ± 3.2 |
| 2 | 0.5 | ND | 1.4 | ||||
| 3 | 0.6 | ND | 0.00 | ||||
| 4 | ND* | ND* | ND* | ||||
| 1 | 15 | 1.1 | 0.8 ± 0.3 | 11.9 | 12.3 ± 1.4 | 19.1 | 15.6 ± 9.6 |
| 2 | 0.8 | 10.3 | 16.9 | ||||
| 3 | 0.3 | 13 | 0.1 | ||||
| 4 | 0.8 | 14 | 26.3 | ||||
| NAI Assay (Oseltamivir) | PRA Assay (Favipiravir) | ddPCR (Mut 275Y) | |||||
|---|---|---|---|---|---|---|---|
| Mouse # | Days of Treatment | IC50 (nM) | Mean IC50 (nM) | IC50 (µM) | Mean IC50 (µM) | % | Mean % |
| Not treated | |||||||
| 1 | 15 | 0.3 | 0.4 ± 0.1 | 11.5 | 10.3 ± 1.3 | 0.0 | 0.0 ± 0.0 |
| 2 | 0.5 | 8.5 | 0.0 | ||||
| 3 | 0.3 | 10.9 | 0.0 | ||||
| 4 | ND* | ND * | ND * | ||||
| Oseltamivir 20 mg/kg/BID | |||||||
| 1 | 8 | 0.5 | 0.6 ± 0.1 | ND | ND | 5.0 | 3.2 ± 3.2 |
| 2 | 0.4 | ND | 7.6 | ||||
| 3 | 0.7 | ND | 0.0 | ||||
| 4 | 0.6 | ND | 0.4 | ||||
| 1 | 12 | 0.5 | 0.6 ± 0.1 | ND | ND | 1.3 | 1.9 ± 2.0 |
| 2 | 0.7 | ND | 5.4 | ||||
| 3 | 0.5 | ND | 0.7 | ||||
| 4 | 0.7 | ND | 0.3 | ||||
| 1 | 15 | 0.4 | 0.5 ± 0.1 | ND | ND | 0.3 | 0.1 ± 0.1 |
| 2 | 0.4 | ND | 0.0 | ||||
| 3 | 0.6 | ND | 0.2 | ||||
| 4 | 0.6 | ND | 0.0 | ||||
| Favipiravir 50 mg/kg/BID | |||||||
| 1 | 8 | NA | NA | ND | ND | NA | NA |
| 2 | NA | ND | NA | ||||
| 3 | NA | ND | NA | ||||
| 4 | NA | ND | NA | ||||
| 1 | 12 | NA | NA | ND | ND | NA | NA |
| 2 | NA | ND | NA | ||||
| 3 | NA | ND | NA | ||||
| 4 | NA | ND | NA | ||||
| 1 | 15 | NA | NA | 13.1 | 12.3 ± 0.7 | NA | NA |
| 2 | NA | 12.4 | NA | ||||
| 3 | NA | 11.5 | NA | ||||
| 4 | ND* | ND * | ND * | ||||
| Oseltamivir 20 mg/kg + Favipiravir 50 mg/kg/BID | |||||||
| 1 | 8 | 0.7 | 0.6 ± 0.1 | ND | ND | 0.1 | 7.0 ± 6.7 |
| 2 | 0.5 | ND | 16.1 | ||||
| 3 | 0.6 | ND | 1.1 | ||||
| 4 | 0.5 | ND | 10.7 | ||||
| 1 | 12 | 0.7 | 0.7 ± 0.1 | ND | ND | 2.7 | 5.6 ± 3.2 |
| 2 | ND* | ND * | ND * | ||||
| 3 | 0.6 | ND | 4.1 | ||||
| 4 | 0.7 | ND | 10.1 | ||||
| 1 | 15 | 0.9 | 0.8 ± 0.1 | 12.6 | 11.0 ± 1.2 | 1.0 | 6.4 ± 6.1 |
| 2 | 0.9 | 11.6 | 16.4 | ||||
| 3 | 0.7 | 10.4 | 6.5 | ||||
| 4 | 0.8 | 9.5 | 1.7 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baz, M.; Carbonneau, J.; Rhéaume, C.; Cavanagh, M.-H.; Boivin, G. Combination Therapy with Oseltamivir and Favipiravir Delays Mortality but Does Not Prevent Oseltamivir Resistance in Immunodeficient Mice Infected with Pandemic A(H1N1) Influenza Virus. Viruses 2018, 10, 610. https://doi.org/10.3390/v10110610
Baz M, Carbonneau J, Rhéaume C, Cavanagh M-H, Boivin G. Combination Therapy with Oseltamivir and Favipiravir Delays Mortality but Does Not Prevent Oseltamivir Resistance in Immunodeficient Mice Infected with Pandemic A(H1N1) Influenza Virus. Viruses. 2018; 10(11):610. https://doi.org/10.3390/v10110610
Chicago/Turabian StyleBaz, Mariana, Julie Carbonneau, Chantal Rhéaume, Marie-Hélène Cavanagh, and Guy Boivin. 2018. "Combination Therapy with Oseltamivir and Favipiravir Delays Mortality but Does Not Prevent Oseltamivir Resistance in Immunodeficient Mice Infected with Pandemic A(H1N1) Influenza Virus" Viruses 10, no. 11: 610. https://doi.org/10.3390/v10110610
APA StyleBaz, M., Carbonneau, J., Rhéaume, C., Cavanagh, M.-H., & Boivin, G. (2018). Combination Therapy with Oseltamivir and Favipiravir Delays Mortality but Does Not Prevent Oseltamivir Resistance in Immunodeficient Mice Infected with Pandemic A(H1N1) Influenza Virus. Viruses, 10(11), 610. https://doi.org/10.3390/v10110610





