Genetic Resistance to Avian Leukosis Viruses Induced by CRISPR/Cas9 Editing of Specific Receptor Genes in Chicken Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Construction of the Gene Editing CRISPR/Cas9 Vectors
2.2. DF-1 Cell Culture and Genome Editing
2.3. Determining Genome Targeting Efficiency Using the T7EI Assay
2.4. Analysis of Gene-Edited Single-Cell Clones of DF-1 Cells
2.5. Viral Propagation and Cell Infection
3. Results
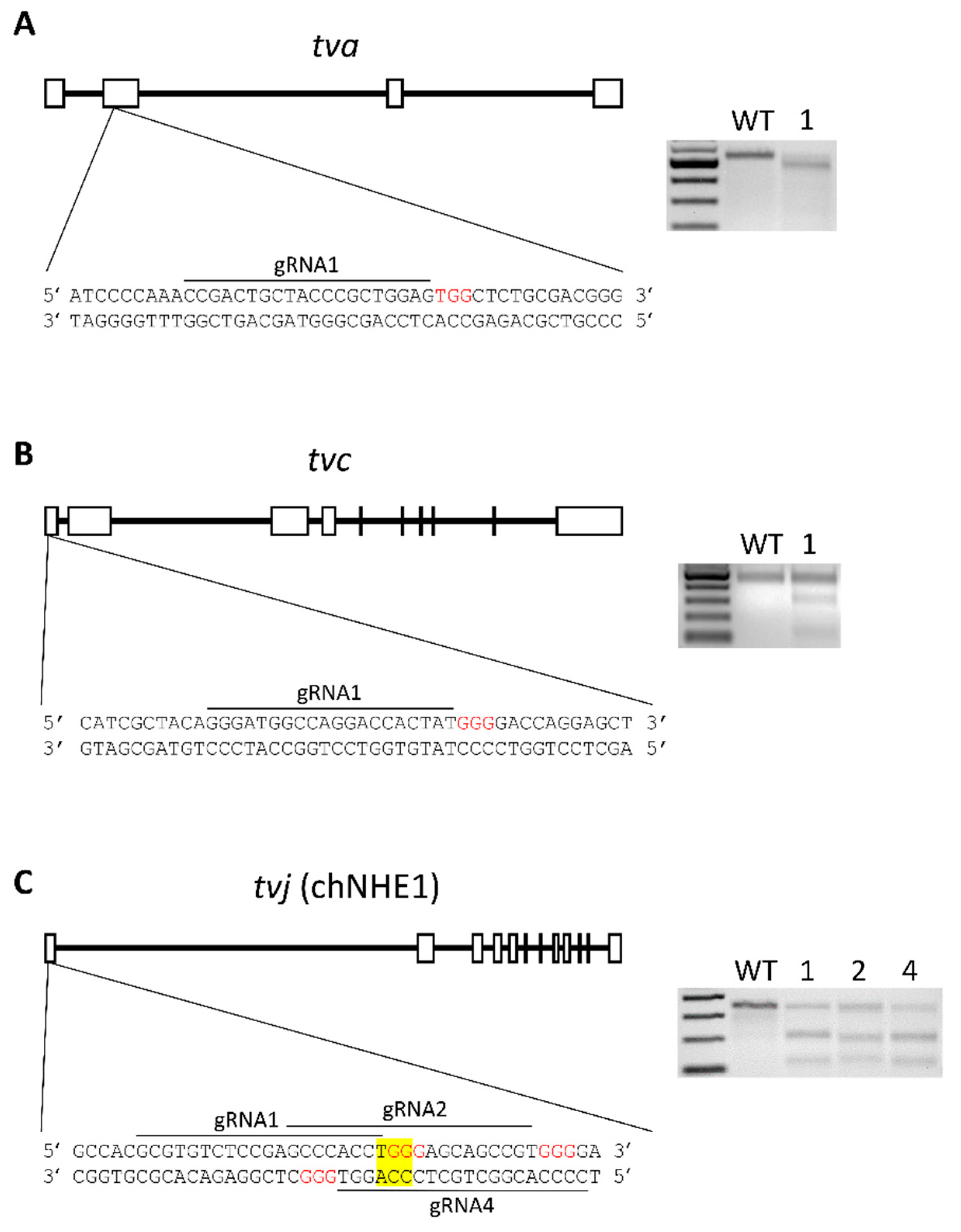
3.1. Design of Gene Editing Experiments
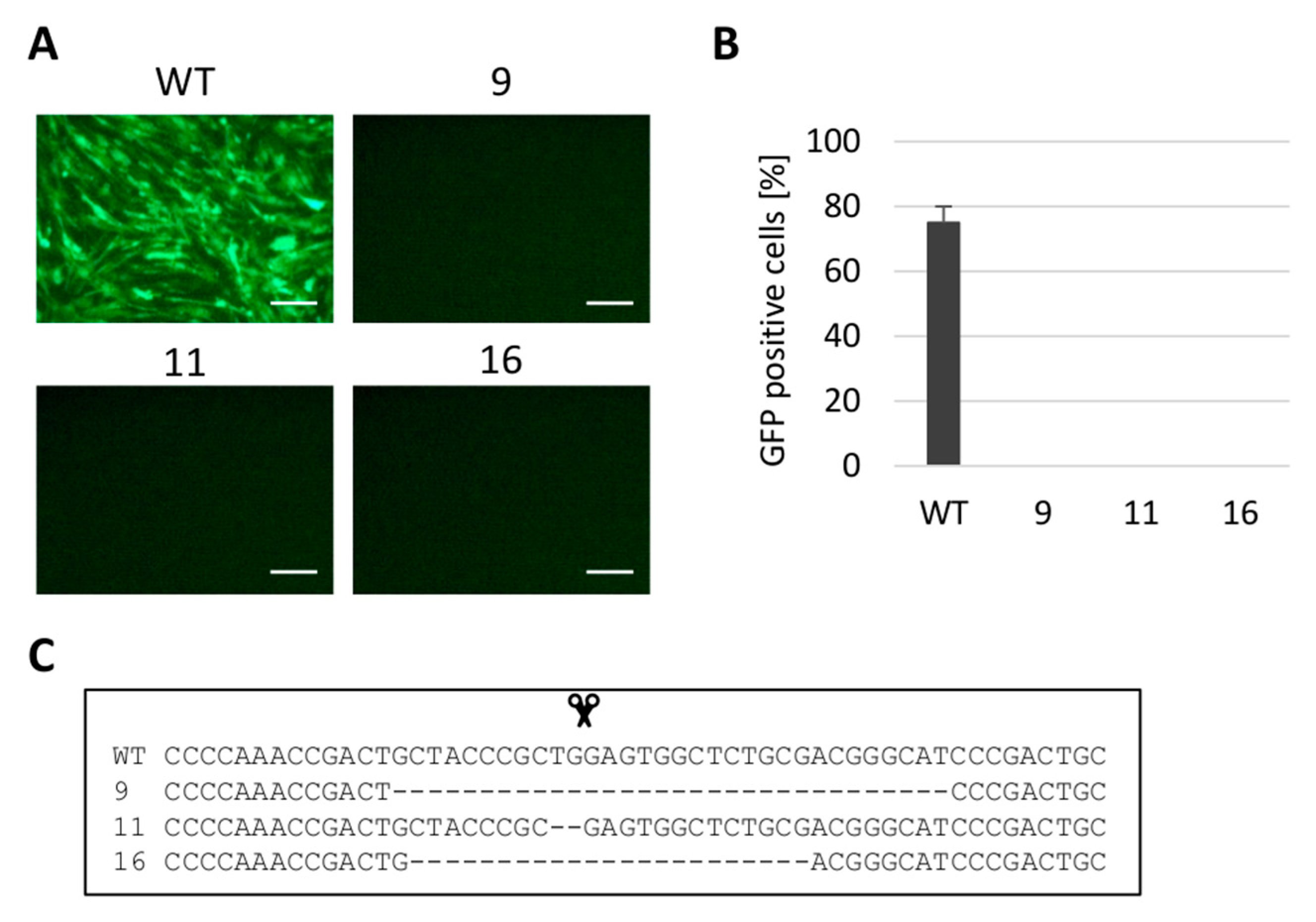
3.2. Gene Editing of tva Gene Confers the Resistance to ALV Subgroup A
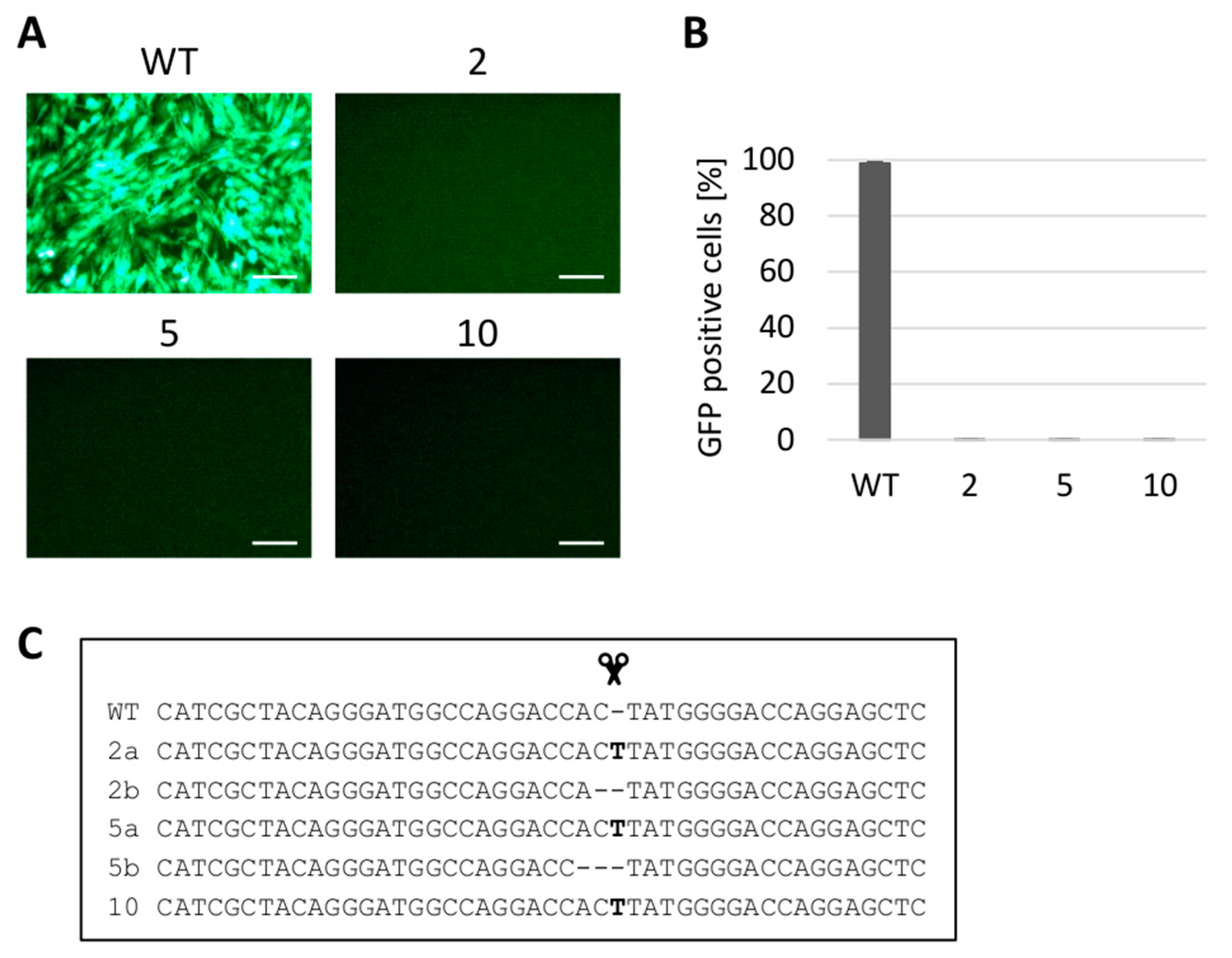
3.3. Gene Editing of tvc Gene Renders the Cells Resistant to ALV Subgroup C
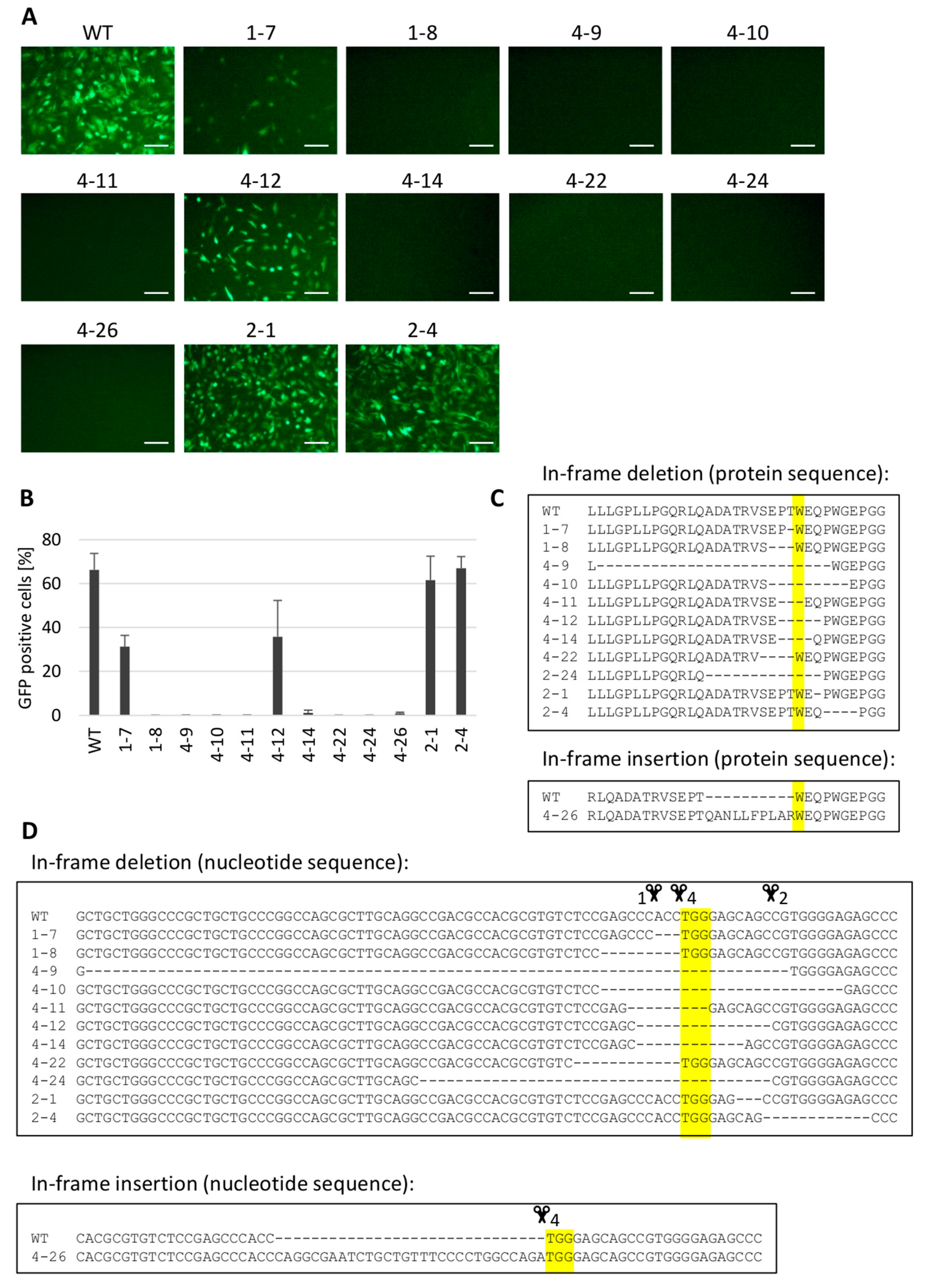
3.4. Gene Editing of chNHE1 (tvj) Renders the Cells Resistant to ALV Subgroup J
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Payne, L.N.; Nair, V. The long view: 40 years of avian leukosis research. Avian Pathol. 2012, 41, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Cui, N.; Su, S.; Chen, Z.; Zhao, X.; Cui, Z. Genomic sequence analysis and biological characteristics of a rescued clone of avian leukosis virus strain JS11C1, isolated from indigenous chickens. J. Gen. Virol. 2014, 95, 2512–2522. [Google Scholar] [CrossRef] [PubMed]
- Weiss, R.A. Cellular receptors and viral glycoproteins involved in retrovirus entry. In The Retroviridae; Levy, J.A., Ed.; Plenum Press: New York, NY, USA, 1992; Volume 2, pp. 1–108. [Google Scholar]
- Barnard, R.J.O.; Elleder, D.; Young, J.A.T. Avian sarcoma and leukosis virus-receptor interactions: From classical genetics to novel insights into virus-cell membrane fusion. Virology 2006, 344, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Bates, P.; Young, J.A.T.; Varmus, H.E. A receptor for subgroup A Rous sarcoma virus is related to the low density lipoprotein receptor. Cell 1993, 74, 1043–1051. [Google Scholar] [CrossRef]
- Young, J.A.T.; Bates, P.; Varmus, H.E. Isolation of a chicken gene that confers susceptibility to infection by subgroup A avian leukosis and sarcoma viruses. J. Virol. 1993, 67, 1811–1816. [Google Scholar] [PubMed]
- Brojatsch, J.; Naughton, J.; Rolls, M.M.; Zingler, K.; Young, J.A. CAR1, a TNFR-related protein, is a cellular receptor for cytopathic avian leukosis-sarcoma viruses and mediates apoptosis. Cell 1996, 87, 845–855. [Google Scholar] [CrossRef]
- Adkins, H.B.; Brojatsch, J.; Naughton, J.; Rolls, M.M.; Pesola, J.M.; Young, J.A.T. Identification of a cellular receptor for subgroup E avian leukosis virus. Proc. Natl. Acad. Sci. USA 1997, 94, 11617–11622. [Google Scholar] [CrossRef] [PubMed]
- Adkins, H.B.; Brojatsch, J.; Young, J.A.T. Identification and characterization of a shared TNFR-related receptor for subgroup B, D, and E avian leukosis viruses reveal cysteine residues required specifically for subgroup E viral entry. J. Virol. 2000, 74, 3572–3578. [Google Scholar] [CrossRef] [PubMed]
- Elleder, D.; Stepanets, V.; Melder, D.C.; Šenigl, F.; Geryk, J.; Pajer, P.; Plachý, J.; Hejnar, J.; Svoboda, J.; Federspiel, M.J. The receptor for the subgroup C avian sarcoma and leukosis viruses, Tvc, is related to mammalian butyrophilins, members of the immunoglobulin superfamily. J. Virol. 2005, 79, 10408–10419. [Google Scholar] [CrossRef] [PubMed]
- Chai, N.; Bates, P. Na/H exchanger type 1 is a receptor for pathogenic subgroup J avian leukosis virus. Proc. Natl. Acad. Sci. USA 2006, 103, 5531–5536. [Google Scholar] [CrossRef] [PubMed]
- Klucking, S.; Adkins, H.B.; Young, J.A.T. Resistance to infection by subgroups B, D, and E avian sarcoma and leukosis viruses is explained by a premature stop codon within a resistance allele of the tvb receptor gene. J. Virol. 2002, 76, 7918–7921. [Google Scholar] [CrossRef] [PubMed]
- Elleder, D.; Melder, D.C.; Trejbalová, K.; Svoboda, J.; Federspiel, M.J. Two different molecular defects in the Tva receptor gene explain the resistance of two tvar lines of chickens to infection by subgroup A avian sarcoma and leukosis viruses. J. Virol. 2004, 78, 13489–13500. [Google Scholar] [CrossRef] [PubMed]
- Kučerová, D.; Plachý, J.; Reinišová, M.; Šenigl, F.; Trejbalová, K.; Geryk, J.; Hejnar, J. Nonconserved tryptophan 38 of the cell surface receptor for subgroup J avian leukosis virus discriminates sensitive from resistant avian species. J. Virol. 2013, 87, 8399–8407. [Google Scholar] [CrossRef] [PubMed]
- Reinišová, M.; Plachý, J.; Kučerová, D.; Šenigl, F.; Vinkler, M.; Hejnar, J. Genetic Diversity of NHE1, Receptor for Subgroup J Avian Leukosis Virus, in Domestic Chicken and Wild Anseriform Species. PLoS ONE 2016, 11, 0150589. [Google Scholar] [CrossRef] [PubMed]
- Plachý, J.; Reinišová, M.; Kučerová, D.; Šenigl, F.; Stepanets, V.; Hron, T.; Trejbalová, K.; Elleder, D.; Hejnar, J. Identification of New World Quails Susceptible to Infection with Avian Leukosis Virus Subgroup J. J. Virol. 2017, 91, e02002. [Google Scholar] [CrossRef] [PubMed]
- Reinišová, M.; Šenigl, F.; Yin, X.; Plachý, J.; Geryk, J.; Elleder, D.; Svoboda, J.; Federspiel, M.J.; Hejnar, J. A single-amino-acid substitution in the TvbS1 receptor results in decreased susceptibility to infection by avian sarcoma and leukosis virus subgroups B and D and resistance to infection by subgroup E in vitro and in vivo. J. Virol. 2008, 82, 2097–2105. [Google Scholar] [CrossRef] [PubMed]
- Reinišová, M.; Plachý, J.; Trejbalová, K.; Šenigl, F.; Kučerová, D.; Geryk, J.; Svoboda, J.; Hejnar, J. Intronic deletions that disrupt mRNA splicing of the tva receptor gene result in decreased susceptibility to infection by avian sarcoma and leukosis virus subgroup A. J. Virol. 2012, 86, 2021–2030. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Lee, K.Y.; Park, Y.H.; Choi, H.J.; Yao, Y.; Nair, V.; Han, J.Y. Acquisition of resistance to avian leukosis virus subgroup B through mutations on tvb cysteine-rich domains in DF-1 chicken fibroblasts. Vet. Res. 2017, 48, 48. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Lee, K.Y.; Jung, K.M.; Park, K.J.; Lee, K.O.; Suh, J.Y.; Yao, Y.; Nair, V.; Han, J.Y. Precise gene editing of chicken Na+/H+ exchange type 1 (chNHE1) confers resistance to avian leukosis virus subgroup J (ALV-J). Dev. Comp. Immunol. 2017, 77, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Ran, F.A.; Hsu, P.D.; Wright, J.; Agarwala, V.; Scott, D.A.; Zhang, F. Genome engineering using the CRISPR-Cas9 system. Nat. Protoc. 2013, 8, 2281–2308. [Google Scholar] [CrossRef] [PubMed]
- Hsu, P.D.; Scott, D.A.; Weinstein, J.A.; Ran, F.A.; Konermann, S.; Agarwala, V.; Li, Y.; Fine, E.J.; Wu, X.; Shalem, O.; et al. DNA targeting specificity of RNA-guided Cas9 nucleases. Nat. Biotechnol. 2013, 31, 827–832. [Google Scholar] [CrossRef] [PubMed]
- Himly, M.; Foster, D.N.; Bottoli, I.; Iacovoni, J.S.; Vogt, P.K. The DF-1 chicken fibroblast cell line: Transformation induced by diverse oncogenes and cell death resulting from infection by avian leukosis viruses. Virology 1998, 248, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Federspiel, M.J.; Hughes, S.H. Retroviral gene delivery. Methods Cell Biol. 1997, 52, 67–177. [Google Scholar]
- Schusser, B.; Collarini, E.J.; Yi, H.; Izquierdo, S.M.; Fesler, J.; Pedersen, D.; Klasing, K.C.; Kaspers, B.; Harriman, W.D.; van de Lavoir, M.C.; et al. Immunoglobulin knockout chickens via efficient homologous recombination in primordial germ cells. Proc. Natl. Acad. Sci. USA 2013, 110, 20170–20175. [Google Scholar] [CrossRef] [PubMed]
- Trefil, P.; Aumann, D.; Koslová, A.; Mucksová, J.; Benešová, B.; Kalina, J.; Wurmser, C.; Fries, R.; Elleder, D.; Schusser, B.; et al. Male fertility restored by transplanting primordial germ cells into testes: A new way towards efficient transgenesis in chicken. Sci. Rep. 2017, 7, 14246. [Google Scholar] [CrossRef] [PubMed]
- Whitworth, K.M.; Rowland, R.R.; Ewen, C.L.; Trible, B.R.; Kerrigan, M.A.; Cino-Ozuna, A.G.; Samuel, M.S.; Lightner, J.E.; McLaren, D.G.; Mileham, A.J.; et al. Gene-edited pigs are protected from porcine reproductive and respiratory syndrome virus. Nat. Biotechnol. 2016, 34, 20–22. [Google Scholar] [CrossRef] [PubMed]
- Boddicker, N.J.; Bjorkquist, A.; Rowland, R.R.; Lunney, J.K.; Reecy, J.M.; Dekkers, J.C. Genome-wide association and genomic prediction for host response to porcine reproductive and respiratory syndrome virus infection. Genet. Sel. Evol. 2014, 46, 18. [Google Scholar] [CrossRef] [PubMed]
- Counillon, L.; Bouret, Y.; Marchiq, I.; Pouysségur, J. Na(+)/H(+) antiporter (NHE1) and lactate/H(+) symporters (MCTs) in pH homeostasis and cancer metabolism. Biochim. Biophys. Acta 2016, 1863, 2465–2480. [Google Scholar] [CrossRef] [PubMed]
- Denker, S.P.; Barber, D.L. Cell migration requires both ion translocation and cytoskeletal anchoring by the Na-H exchanger NHE1. J. Cell Biol. 2002, 159, 1087–1096. [Google Scholar] [CrossRef] [PubMed]
- Cardone, R.A.; Casavola, V.; Reshkin, S.J. The role of disturbed pH dynamics and the Na+/H+ exchanger in metastasis. Nat. Rev. Cancer 2005, 5, 786–795. [Google Scholar] [CrossRef] [PubMed]
- Bourguignon, L.Y.; Singleton, P.A.; Diedrich, F.; Stern, R.; Gilad, E. CD44 interaction with Na+-H+ exchanger (NHE1) creates acidic microenvironments leading to hyaluronidase-2 and cathepsin B activation and breast tumor cell invasion. J. Biol. Chem. 2004, 279, 26991–27007. [Google Scholar] [CrossRef] [PubMed]
- Shrode, L.D.; Gan, B.S.; D’Souza, S.J.; Orlowski, J.; Grinstein, S. Topological analysis of NHE1, the ubiquitous Na+/H+ exchanger using chymotryptic cleavage. Am. J. Physiol. 1998, 275, C431–C439. [Google Scholar] [CrossRef] [PubMed]
- Counillon, L.; Pouysségur, J.; Reithmeier, R.A. The Na+/H+ exchanger NHE-1 possesses N- and O-linked glycosylation restricted to the first N-terminal extracellular domain. Biochemistry 1994, 33, 10463–10469. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.; Zhang, Y.; Yu, M.; Ren, C.; Gao, Y.; Yun, B.; Liu, Y.; Wang, Y.; Qi, X.; Liu, C.; et al. Residues 28 to 39 of the Extracellular Loop 1 of Chicken Na+/H+ Exchanger Type I Mediate Cell Binding and Entry of Subgroup J Avian Leukosis Virus. J. Virol. 2018, 92, e01627-17. [Google Scholar] [CrossRef] [PubMed]

| Oligonucleotide Name | Sequence (5′–3′) |
|---|---|
| TVA-gRNA1a | CACCGCCGACTGCTACCCGCTGGAG |
| TVA-gRNA1b | AAACCTCCAGCGGGTAGCAGTCGGC |
| TVC-gRNA1a | CACCGGGATGGCCAGGACCACTATG |
| TVC-gRNA1b | AAACATAGTGGTCCTGGCCATCCCG |
| TVJ-gRNA1a | CACCGCGTGTCTCCGAGCCCACCT |
| TVJ-gRNA1b | AAACAGGTGGGCTCGGAGACACGC |
| TVJ-gRNA2a | CACCGCCCACCTGGGAGCAGCCGT |
| TVJ-gRNA2b | AAACACGGCTGCTCCCAGGTGGGC |
| TVJ-gRNA4a | CACCGCCCCACGGCTGCTCCCAGGT |
| TVJ-gRNA4b | AAACACCTGGGAGCAGCCGTGGGGC |
| TVA-fw | GCATGGTGCGGTTGTTGGAG |
| TVA-rv | CTGTGCCGCCGGCGGTGGGC |
| TVC-fw | CTCGCTGGCAGAGCCAGGAC |
| TVC-rv | CAAAATGTGGCCCTGATGAAGA |
| TVJ-fw | CGGCTCCCTCCGCCATG |
| TVJ-rv | TCATCAGGCAGGCCAGCAGGAT |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koslová, A.; Kučerová, D.; Reinišová, M.; Geryk, J.; Trefil, P.; Hejnar, J. Genetic Resistance to Avian Leukosis Viruses Induced by CRISPR/Cas9 Editing of Specific Receptor Genes in Chicken Cells. Viruses 2018, 10, 605. https://doi.org/10.3390/v10110605
Koslová A, Kučerová D, Reinišová M, Geryk J, Trefil P, Hejnar J. Genetic Resistance to Avian Leukosis Viruses Induced by CRISPR/Cas9 Editing of Specific Receptor Genes in Chicken Cells. Viruses. 2018; 10(11):605. https://doi.org/10.3390/v10110605
Chicago/Turabian StyleKoslová, Anna, Dana Kučerová, Markéta Reinišová, Josef Geryk, Pavel Trefil, and Jiří Hejnar. 2018. "Genetic Resistance to Avian Leukosis Viruses Induced by CRISPR/Cas9 Editing of Specific Receptor Genes in Chicken Cells" Viruses 10, no. 11: 605. https://doi.org/10.3390/v10110605
APA StyleKoslová, A., Kučerová, D., Reinišová, M., Geryk, J., Trefil, P., & Hejnar, J. (2018). Genetic Resistance to Avian Leukosis Viruses Induced by CRISPR/Cas9 Editing of Specific Receptor Genes in Chicken Cells. Viruses, 10(11), 605. https://doi.org/10.3390/v10110605





