Antibody-Mediated Fcγ Receptor-Based Mechanisms of HIV Inhibition: Recent Findings and New Vaccination Strategies
Abstract
:1. Introduction
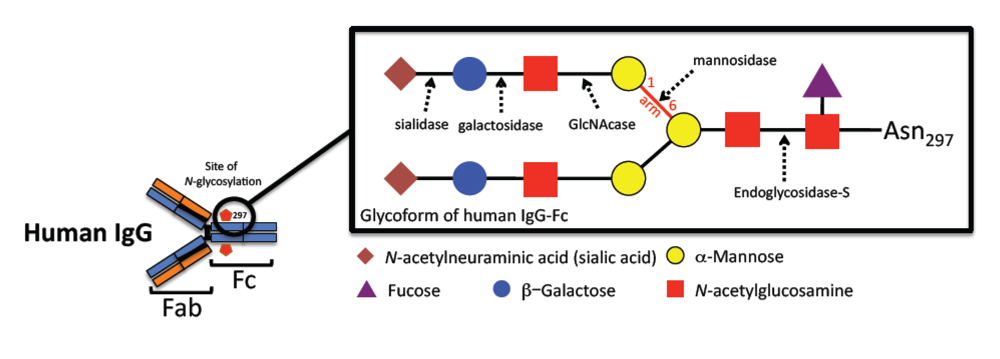
2. IgG structure and functions
| Variations in IgG-Fc glycoform | Fcγ effector binding and functions |
|---|---|
| Complete deglycosylation of IgG1: | -Loss of FcgR binding capacity |
| -Failure to initiate effector function | |
| Removal of fucose residues : | - Increase in IgG affinity (all subclasses) for FcgRIIIa |
| - Enhancement of Fc-FcgR interaction | |
| Removal of galactose residues : | - Lower levels of C1q binding to IgG-Fc |
| - Either increase or decrease in IgG affinity for FcgRs | |
| Exposure of N-acetylglucosamine residues after galactose removal : | - Increase in MBL binding |
| - Observed in some autoimmune diseases | |
| Fc glycan hydrolysis between two N-acetylglucosamine residues : | - Inhibition of the binding of IgG and C1q to FcgRs |
| - Abolition of the activation of effector and complement functions | |
| - Impairment of structural stability and functional activity | |
| Increase of the levels of sialic acid : | - Decrease in FcgRIII/IV affinity |
| - No change in IgG binding to FcgRIIb | |
| Replacement of amino acid residues interacting with galactose or N-acetylglucosamine in the 1-6 arm position : | - No influence in IgG binding to FcgRI |
| Replacement of Asn297 : | - Loss of N-linked glycosylation |
| - FcgRI affinity halved | |
| Removal of mannose residues in the 1-6 arm position : | - Impairment of FcgRs recognition by IgG |
2.1. Role of Fcγ glycosylation
2.2. Antigen-specific IgG functions
3. Broadly neutralizing antibodies against HIV
3.1. Monoclonal neutralizing IgG : IgG1 b12, 2G12, 447-52D, 2F5 and 4E10
3.2. New broadly neutralizing antibodies: recent findings
3.3. In vivoproduction of neutralizing antibodies: proof-of-concept
4. Neutralizing IgG as a hallmark of protection against HIV
4.1. Neutralizing antibodies as immune correlates of protection
4.2. Mechanism of protection-mediated by neutralizing IgG: role of FcγRs
6. Neutralizing IgA as mucosal immunity against sexual HIV transmission
7. Conclusions and Future Directions
Acknowledgments
References
- Spira, A.I.; Marx, P.A.; Patterson, B.K.; Mahoney, J.; Koup, R.A.; Wolinsky, S.M.; Ho, D.D. Cellular targets of infection and route of viral dissemination after an intravaginal inoculation of simian immunodeficiency virus into rhesus macaques. J. Exp. Med. 1996, 183, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Herbein, G.; Coaquette, A.; Perez-Bercoff, D.; Pancino, G. Macrophage activation and HIV infection: can the Trojan horse turn into a fortress? Curr. Mol. Med. 2002, 2, 723–738. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; KewalRamani, V.N. Dendritic-cell interactions with HIV: infection and viral dissemination. Nat. Rev. Immunol. 2006, 6, 859–868. [Google Scholar] [CrossRef]
- Piguet, V.; Blauvelt, A. Essential roles for dendritic cells in the pathogenesis and potential treatment of HIV disease. J. Invest. Dermatol. 2002, 119, 365–369. [Google Scholar] [CrossRef] [PubMed]
- Wilflingseder, D.; Banki, Z.; Dierich, M.P.; Stoiber, H. Mechanisms promoting dendritic cell-mediated transmission of HIV. Mol. Immunol. 2005, 42, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Larsson, M. HIV-1 and the hijacking of dendritic cells: a tug of war. Springer. Semin. Immunopathol. 2005, 26, 309–328. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.; Cordero, M.; Almeida, J.; Orfao, A. Different subsets of peripheral blood dendritic cells show distinct phenotypic and functional abnormalities in HIV-1 infection. AIDS 2005, 19, 261–271. [Google Scholar] [PubMed]
- Burrer, R.; Salmon-Ceron, D.; Richert, S.; Pancino, G.; Spiridon, G.; Haessig, S.; Roques, V.; Barre-Sinoussi, F.; Aubertin, A.M.; Moog, C. Immunoglobulin G (IgG) and IgA, but also nonantibody factors, account for in vitro neutralization of human immunodeficiency virus (HIV) type 1 primary isolates by serum and plasma of HIV-infected patients. J. Virol. 2001, 75, 5421–5424. [Google Scholar] [CrossRef] [PubMed]
- Moog, C.; Spenlehauer, C.; Fleury, H.; Heshmati, F.; Saragosti, S.; Letourneur, F.; Kirn, A.; Aubertin, A.M. Neutralization of primary human immunodeficiency virus type 1 isolates: a study of parameters implicated in neutralization in vitro. AIDS Res. Hum. Retroviruses 1997, 13, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Binley, J.M.; Wrin, T.; Korber, B.; Zwick, M.B.; Wang, M.; Chappey, C.; Stiegler, G.; Kunert, R.; Zolla-Pazner, S.; Katinger, H.; Petropoulos, C.J.; Burton, D.R. Comprehensive cross-clade neutralization analysis of a panel of anti-human immunodeficiency virus type 1 monoclonal antibodies. J. Virol. 2004, 78, 13232–13252. [Google Scholar] [CrossRef] [PubMed]
- Baba, T.W.; Liska, V.; Hofmann-Lehmann, R.; Vlasak, J.; Xu, W.; Ayehunie, S.; Cavacini, L.A.; Posner, M.R.; Katinger, H.; Stiegler, G.; Bernacky, B.J.; Rizvi, T.A.; Schmidt, R.; Hill, L.R.; Keeling, M.E.; Lu, Y.; Wright, J.E.; Chou, T.C.; Ruprecht, R.M. Human neutralizing monoclonal antibodies of the IgG1 subtype protect against mucosal simian-human immunodeficiency virus infection. Nat. Med. 2000, 6, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Mascola, J.R.; Stiegler, G.; VanCott, T.C.; Katinger, H.; Carpenter, C.B.; Hanson, C.E.; Beary, H.; Hayes, D.; Frankel, S.S.; Birx, D.L.; Lewis, M.G. Protection of macaques against vaginal transmission of a pathogenic HIV-1/SIV chimeric virus by passive infusion of neutralizing antibodies. Nat. Med. 2000, 6, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Mascola, J.R.; Lewis, M.G.; Stiegler, G.; Harris, D.; VanCott, T.C.; Hayes, D.; Louder, M.K.; Brown, C.R.; Sapan, C.V.; Frankel, S.S.; Lu, Y.; Robb, M.L.; Katinger, H.; Birx, D.L. Protection of Macaques against pathogenic simian/human immunodeficiency virus 89.6PD by passive transfer of neutralizing antibodies. J. Virol. 1999, 73, 4009–4018. [Google Scholar] [PubMed]
- Mascola, J.R. Passive transfer studies to elucidate the role of antibody-mediated protection against HIV-1. Vaccine 2002, 20, 1922–1925. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, Y.; Igarashi, T.; Haigwood, N.L.; Sadjadpour, R.; Donau, O.K.; Buckler, C.; Plishka, R.J.; Buckler-White, A.; Martin, M.A. Transfer of neutralizing IgG to macaques 6 h but not 24 h after SHIV infection confers sterilizing protection: implications for HIV-1 vaccine development. Proc. Natl. Acad. Sci. U S A 2003, 100, 15131–15136. [Google Scholar] [CrossRef] [PubMed]
- Zhuge, W.; Jia, F.; Adany, I.; Narayan, O.; Stephens, E.B. Plasmas from lymphocyte- and macrophage-tropic SIVmac-infected macaques have antibodies with a broader spectrum of virus neutralization activity in macrophage versus lymphocyte cultures. Virology 1997, 227, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Ruppach, H.; Nara, P.; Raudonat, I.; Elanjikal, Z.; Rubsamen-Waigmann, H.; Dietrich, U. Human immunodeficiency virus (HIV)-positive sera obtained shortly after seroconversion neutralize autologous HIV type 1 isolates on primary macrophages but not on lymphocytes. J. Virol. 2000, 74, 5403–5411. [Google Scholar] [CrossRef] [PubMed]
- Ganesh, L.; Leung, K.; Lore, K.; Levin, R.; Panet, A.; Schwartz, O.; Koup, R.A.; Nabel, G.J. Infection of specific dendritic cells by CCR5-tropic human immunodeficiency virus type 1 promotes cell-mediated transmission of virus resistant to broadly neutralizing antibodies. J. Virol. 2004, 78, 11980–11987. [Google Scholar] [CrossRef] [PubMed]
- Frankel, S.S.; Steinman, R.M.; Michael, N.L.; Kim, S.R.; Bhardwaj, N.; Pope, M.; Louder, M.K.; Ehrenberg, P.K.; Parren, P.W.; Burton, D.R.; Katinger, H.; VanCott, T.C.; Robb, M.L.; Birx, D.L.; Mascola, J.R. Neutralizing monoclonal antibodies block human immunodeficiency virus type 1 infection of dendritic cells and transmission to T cells. J. Virol. 1998, 72, 9788–9794. [Google Scholar] [PubMed]
- Wen, M.; Yang, H.; Zhou, P. “Unconventional” neutralizing activity of antibodies against HIV. Virologica Sinica 2007, 2, 462–470. [Google Scholar] [CrossRef]
- Holl, V.; Hemmerter, S.; Burrer, R.; Schmidt, S.; Bohbot, A.; Aubertin, A.M.; Moog, C. Involvement of Fc gamma RI (CD64) in the mechanism of HIV-1 inhibition by polyclonal IgG purified from infected patients in cultured monocyte-derived macrophages. J. Immunol. 2004, 173, 6274–6283. [Google Scholar] [PubMed]
- Holl, V.; Peressin, M.; Schmidt, S.; Decoville, T.; Zolla-Pazner, S.; Aubertin, A.M.; Moog, C. Efficient inhibition of HIV-1 replication in human immature monocyte-derived dendritic cells by purified anti-HIV-1 IgG without induction of maturation. Blood 2006, 107, 4466–4474. [Google Scholar] [CrossRef] [PubMed]
- Holl, V.; Peressin, M.; Decoville, T.; Schmidt, S.; Zolla-Pazner, S.; Aubertin, A.M.; Moog, C. Nonneutralizing antibodies are able to inhibit human immunodeficiency virus type 1 replication in macrophages and immature dendritic cells. J. Virol. 2006, 80, 6177–6181. [Google Scholar] [CrossRef] [PubMed]
- Nimmerjahn, F.; Ravetch, J.V. Fc-receptors as regulators of immunity. Adv. Immunol. 2007, 96, 179–204. [Google Scholar] [PubMed]
- Nimmerjahn, F.; Ravetch, J.V. Fcgamma receptors as regulators of immune responses. Nat. Rev. Immunol. 2008, 8, 34–47. [Google Scholar] [CrossRef]
- Jefferis, R.; Lund, J. Interaction sites on human IgG-Fc for FcgammaR: current models. Immunol. Lett. 2002, 82, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Krapp, S.; Mimura, Y.; Jefferis, R.; Huber, R.; Sondermann, P. Structural analysis of human IgG-Fc glycoforms reveals a correlation between glycosylation and structural integrity. J. Mol. Biol. 2003, 325, 979–989. [Google Scholar] [CrossRef] [PubMed]
- Arnold, J.N.; Wormald, M.R.; Sim, R.B.; Rudd, P.M.; Dwek, R.A. The impact of glycosylation on the biological function and structure of human immunoglobulins. Annu. Rev. Immunol. 2007, 25, 21–50. [Google Scholar] [CrossRef] [PubMed]
- Scanlan, C.N.; Burton, D.R.; Dwek, R.A. Making autoantibodies safe. Proc. Natl. Acad. Sci. U S A 2008, 105, 4081–4082. [Google Scholar] [CrossRef] [PubMed]
- Nimmerjahn, F.; Ravetch, J.V. Divergent immunoglobulin g subclass activity through selective Fc receptor binding. Science 2005, 310, 1510–1512. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, Y.; Nimmerjahn, F.; Ravetch, J.V. Anti-inflammatory activity of immunoglobulin G resulting from Fc sialylation. Science 2006, 313, 670–673. [Google Scholar] [CrossRef] [PubMed]
- Guo, N.; Liu, Y.; Masuda, Y.; Kawagoe, M.; Ueno, Y.; Kameda, T.; Sugiyama, T. Repeated immunization induces the increase in fucose content on antigen-specific IgG N-linked oligosaccharides. Clin. Biochem. 2005, 38, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Takai, T. Roles of Fc receptors in autoimmunity. Nat. Rev. Immunol. 2002, 2, 580–592. [Google Scholar] [PubMed]
- Regnault, A.; Lankar, D.; Lacabanne, V.; Rodriguez, A.; Thery, C.; Rescigno, M.; Saito, T.; Verbeek, S.; Bonnerot, C.; Ricciardi-Castagnoli, P.; Amigorena, S. Fcgamma receptor-mediated induction of dendritic cell maturation and major histocompatibility complex class I-restricted antigen presentation after immune complex internalization. J. Exp. Med. 1999, 189, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Tyler, D.S.; Nastala, C.L.; Stanley, S.D.; Matthews, T.J.; Lyerly, H.K.; Bolognesi, D.P.; Weinhold, K.J. GP120 specific cellular cytotoxicity in HIV-1 seropositive individuals. Evidence for circulating CD16+ effector cells armed in vivo with cytophilic antibody. J. Immunol. 1989, 142, 1177–1182. [Google Scholar] [PubMed]
- Lanier, L.L.; Phillips, J.H.; Hackett Jr., J.; Tutt, M.; Kumar, V. Natural killer cells: definition of a cell type rather than a function. J. Immunol. 1986, 137, 2735–2739. [Google Scholar] [PubMed]
- Jewett, A.; Giorgi, J.V.; Bonavida, B. Antibody-dependent cellular cytotoxicity against HIV-coated target cells by peripheral blood monocytes from HIV seropositive asymptomatic patients. J. Immunol. 1990, 145, 4065–4071. [Google Scholar] [PubMed]
- Willey, S.; Aasa-Chapman, M.M. Humoral immunity to HIV-1: neutralisation and antibody effector functions. Trends Microbiol. 2008, 16, 596–604. [Google Scholar] [CrossRef] [PubMed]
- Karagiannis, S.N.; Bracher, M.G.; Hunt, J.; McCloskey, N.; Beavil, R.L.; Beavil, A.J.; Fear, D.J.; Thompson, R.G.; East, N.; Burke, F.; Moore, R.J.; Dombrowicz, D.D.; Balkwill, F.R.; Gould, H.J. IgE-antibody-dependent immunotherapy of solid tumors: cytotoxic and phagocytic mechanisms of eradication of ovarian cancer cells. J. Immunol. 2007, 179, 2832–2843. [Google Scholar] [PubMed]
- Karagiannis, S.N.; Wang, Q.; East, N.; Burke, F.; Riffard, S.; Bracher, M.G.; Thompson, R.G.; Durham, S.R.; Schwartz, L.B.; Balkwill, F.R.; Gould, H.J. Activity of human monocytes in IgE antibody-dependent surveillance and killing of ovarian tumor cells. Eur. J. Immunol. 2003, 33, 1030–1040. [Google Scholar] [CrossRef] [PubMed]
- Zwick, M.B.; Labrijn, A.F.; Wang, M.; Spenlehauer, C.; Saphire, E.O.; Binley, J.M.; Moore, J.P.; Stiegler, G.; Katinger, H.; Burton, D.R.; Parren, P.W. Broadly neutralizing antibodies targeted to the membrane-proximal external region of human immunodeficiency virus type 1 glycoprotein gp41. J. Virol. 2001, 75, 10892–10905. [Google Scholar] [CrossRef] [PubMed]
- Buchacher, A.; Predl, R.; Strutzenberger, K.; Steinfellner, W.; Trkola, A.; Purtscher, M.; Gruber, G.; Tauer, C.; Steindl, F.; Jungbauer, A.; et al. Generation of human monoclonal antibodies against HIV-1 proteins; electrofusion and Epstein-Barr virus transformation for peripheral blood lymphocyte immortalization. AIDS Res. Hum. Retroviruses 1994, 10, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Trkola, A.; Dragic, T.; Arthos, J.; Binley, J.M.; Olson, W.C.; Allaway, G.P.; Cheng-Mayer, C.; Robinson Jr., J.; Maddon, P.J.; Moore, J.P. CD4-dependent, antibody-sensitive interactions between HIV-1 and its co-receptor CCR-5. Nature 1996, 384, 184–187. [Google Scholar] [CrossRef] [PubMed]
- Conley, A.J.; Gorny, M.K.; Kessler III, J.A.; Boots, L.J.; Ossorio-Castro, M.; Koenig, S.; Lineberger, D.W.; Emini, E.A.; Williams, C.; Zolla-Pazner, S. Neutralization of primary human immunodeficiency virus type 1 isolates by the broadly reactive anti-V3 monoclonal antibody, 447-52D. J. Virol. 1994, 68, 6994–7000. [Google Scholar] [PubMed]
- Burton, D.R.; Barbas, C.F.; Persson, M.A.; Koenig, S.; Chanock, R.M.; Lerner, R.A. A large array of human monoclonal antibodies to type 1 human immunodeficiency virus from combinatorial libraries of asymptomatic seropositive individuals. Proc. Natl. Acad. Sci. USA 1991, 88, 10134–10137. [Google Scholar] [CrossRef]
- Gray, E.S.; Madiga, M.C.; Moore, P.L.; Mlisana, K.; Karim, S.S.; Binley, J.M.; Shaw, G.M.; Mascola, J.R.; Morris, L. Broad HIV-1 neutralization mediated by plasma antibodies against the gp41 membrane proximal external region. J. Virol. 2009. [Google Scholar] [CrossRef]
- Gray, E.S.; Taylor, N.; Wycuff, D.; Moore, P.L.; Tomaras, G.D.; Wibmer, C.K.; Puren, A.; DeCamp, A.; Gilbert, P.B.; Wood, B.; Montefiori, D.C.; Binley, J.M.; Shaw, G.M.; Haynes, B.F.; Mascola, J.R.; Morris, L. Antibody specificities associated with neutralization breadth in plasma from human immunodeficiency virus type 1 subtype C-infected blood donors. J. Virol. 2009, 83, 8925–8937. [Google Scholar] [CrossRef] [PubMed]
- Walker, L.M.; Phogat, S.K.; Chan-Hui, P.Y.; Wagner, D.; Phung, P.; Goss, J.L.; Wrin, T.; Simek, M.D.; Fling, S.; Mitcham, J.L.; Lehrman, J.K.; Priddy, F.H.; Olsen, O.A.; Frey, S.M.; Hammond, P.W.; Miiro, G.; Serwanga, J.; Pozniak, A.; McPhee, D.; Manigart, O.; Mwananyanda, L.; Karita, E.; Inwoley, A.; Jaoko, W.; Dehovitz, J.; Bekker, L.G.; Pitisuttithum, P.; Paris, R.; Allen, S.; Kaminsky, S.; Zamb, T.; Moyle, M.; Koff, W.C.; Poignard, P.; Burton, D.R. Broad and Potent Neutralizing Antibodies from an African Donor Reveal a New HIV-1 Vaccine Target. Science 2009, 326, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Saphire, E.O.; Parren, P.W.; Pantophlet, R.; Zwick, M.B.; Morris, G.M.; Rudd, P.M.; Dwek, R.A.; Stanfield, R.L.; Burton, D.R.; Wilson, I.A. Crystal structure of a neutralizing human IGG against HIV-1: a template for vaccine design. Science 2001, 293, 1155–1159. [Google Scholar] [CrossRef] [PubMed]
- Scanlan, C.N.; Pantophlet, R.; Wormald, M.R.; Ollmann Saphire, E.; Stanfield, R.; Wilson, I.A.; Katinger, H.; Dwek, R.A.; Rudd, P.M.; Burton, D.R. The broadly neutralizing anti-human immunodeficiency virus type 1 antibody 2G12 recognizes a cluster of alpha1-->2 mannose residues on the outer face of gp120. J. J. Virol. 2002, 76, 7306–7321. [Google Scholar] [CrossRef] [PubMed]
- Sanders, R.W.; Vesanen, M.; Schuelke, N.; Master, A.; Schiffner, L.; Kalyanaraman, R.; Paluch, M.; Berkhout, B.; Maddon, P.J.; Olson, W.C.; Lu, M.; Moore, J.P. Stabilization of the soluble, cleaved, trimeric form of the envelope glycoprotein complex of human immunodeficiency virus type 1. J. Virol. 2002, 76, 8875–8889. [Google Scholar] [CrossRef] [PubMed]
- Sanders, R.W.; Venturi, M.; Schiffner, L.; Kalyanaraman, R.; Katinger, H.; Lloyd, K.O.; Kwong, P.D.; Moore, J.P. The mannose-dependent epitope for neutralizing antibody 2G12 on human immunodeficiency virus type 1 glycoprotein gp120. J. Virol. 2002, 76, 7293–7305. [Google Scholar] [CrossRef] [PubMed]
- Trkola, A.; Purtscher, M.; Muster, T.; Ballaun, C.; Buchacher, A.; Sullivan, N.; Srinivasan, K.; Sodroski, J.; Moore, J.P.; Katinger, H. Human monoclonal antibody 2G12 defines a distinctive neutralization epitope on the gp120 glycoprotein of human immunodeficiency virus type 1. J. Virol. 1996, 70, 1100–1108. [Google Scholar] [PubMed]
- Trkola, A. HIV-host interactions: vital to the virus and key to its inhibition. Curr. Opin. Microbiol. 2004, 7, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Hessell, A.J.; Rakasz, E.G.; Poignard, P.; Hangartner, L.; Landucci, G.; Forthal, D.N.; Koff, W.C.; Watkins, D.I.; Burton, D.R. Broadly neutralizing human anti-HIV antibody 2G12 is effective in protection against mucosal SHIV challenge even at low serum neutralizing titers. PLoS Pathog. 2009, 5, e1000433. [Google Scholar] [CrossRef] [PubMed]
- Haynes, B.F.; Fleming, J.; St Clair, E.W.; Katinger, H.; Stiegler, G.; Kunert, R.; Robinson, J.; Scearce, R.M.; Plonk, K.; Staats, H.F.; Ortel, T.L.; Liao, H.X.; Alam, S.M. Cardiolipin polyspecific autoreactivity in two broadly neutralizing HIV-1 antibodies. Science 2005, 308, 1906–1908. [Google Scholar] [CrossRef] [PubMed]
- Haynes, B.F.; Moody, M.A.; Verkoczy, L.; Kelsoe, G.; Alam, S.M. Antibody polyspecificity and neutralization of HIV-1: a hypothesis. Hum. Antibodies 2005, 14, 59–67. [Google Scholar] [PubMed]
- Kunert, R.; Steinfellner, W.; Purtscher, M.; Assadian, A.; Katinger, H. Stable recombinant expression of the anti HIV-1 monoclonal antibody 2F5 after IgG3/IgG1 subclass switch in CHO cells. Biotechnol. Bioeng. 2000, 67, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Scharf, O.; Golding, H.; King, L.R.; Eller, N.; Frazier, D.; Golding, B.; Scott, D.E. Immunoglobulin G3 from polyclonal human immunodeficiency virus (HIV) immune globulin is more potent than other subclasses in neutralizing HIV type 1. J. Virol. 2001, 75, 6558–6565. [Google Scholar] [CrossRef] [PubMed]
- Perez, L.G.; Costa, M.R.; Todd, C.A.; Haynes, B.F.; Montefiori, D.C. Utilization of immunoglobulin G Fc receptors by human immunodeficiency virus type 1: a specific role for antibodies against the membrane-proximal external region of gp41. J. Virol. 2009, 83, 7397–7410. [Google Scholar] [CrossRef] [PubMed]
- Hessell, A.J.; Hangartner, L.; Hunter, M.; Havenith, C.E.; Beurskens, F.J.; Bakker, J.M.; Lanigan, C.M.; Landucci, G.; Forthal, D.N.; Parren, P.W.; Marx, P.A.; Burton, D.R. Fc receptor but not complement binding is important in antibody protection against HIV. Nature 2007, 449, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Svehla, K.; Louder, M.K.; Wycuff, D.; Phogat, S.; Tang, M.; Migueles, S.A.; Wu, X.; Phogat, A.; Shaw, G.M.; Connors, M.; Hoxie, J.; Mascola, J.R.; Wyatt, R. Analysis of neutralization specificities in polyclonal sera derived from human immunodeficiency virus type 1-infected individuals. J. Virol. 2009, 83, 1045–1059. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Parks, R.J.; Montefiori, D.C.; Kirchherr, J.L.; Keele, B.F.; Decker, J.M.; Blattner, W.A.; Gao, F.; Weinhold, K.J.; Hicks, C.B.; Greenberg, M.L.; Hahn, B.H.; Shaw, G.M.; Haynes, B.F.; Tomaras, G.D. In vivo gp41 antibodies targeting the 2F5 monoclonal antibody epitope mediate human immunodeficiency virus type 1 neutralization breadth. J. Virol. 2009, 83, 3617–3625. [Google Scholar] [CrossRef] [PubMed]
- Sather, D.N.; Armann, J.; Ching, L.K.; Mavrantoni, A.; Sellhorn, G.; Caldwell, Z.; Yu, X.; Wood, B.; Self, S.; Kalams, S.; Stamatatos, L. Factors associated with the development of cross-reactive neutralizing antibodies during human immunodeficiency virus type 1 infection. J. Virol. 2009, 83, 757–769. [Google Scholar] [CrossRef] [PubMed]
- Gustchina, E.; Louis, J.M.; Frisch, C.; Ylera, F.; Lechner, A.; Bewley, C.A.; Clore, G.M. Affinity maturation by targeted diversification of the CDR-H2 loop of a monoclonal Fab derived from a synthetic naive human antibody library and directed against the internal trimeric coiled-coil of gp41 yields a set of Fabs with improved HIV-1 neutralization potency and breadth. Virology 2009, 393, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.R.; Schnepp, B.C.; Zhang, J.; Connell, M.J.; Greene, S.M.; Yuste, E.; Desrosiers, R.C.; Reed Clark, K. Vector-mediated gene transfer engenders long-lived neutralizing activity and protection against SIV infection in monkeys. Nat. Med. 2009, 15, 901–906. [Google Scholar] [CrossRef] [PubMed]
- Lewis, A.D.; Chen, R.; Montefiori, D.C.; Johnson, P.R.; Clark, K.R. Generation of neutralizing activity against human immunodeficiency virus type 1 in serum by antibody gene transfer. J. Virol. 2002, 76, 8769–8775. [Google Scholar] [CrossRef] [PubMed]
- Scheid, J.F.; Mouquet, H.; Feldhahn, N.; Seaman, M.S.; Velinzon, K.; Pietzsch, J.; Ott, R.G.; Anthony, R.M.; Zebroski, H.; Hurley, A.; Phogat, A.; Chakrabarti, B.; Li, Y.; Connors, M.; Pereyra, F.; Walker, B.D.; Wardemann, H.; Ho, D.; Wyatt, R.T.; Mascola, J.R.; Ravetch, J.V.; Nussenzweig, M.C. Broad diversity of neutralizing antibodies isolated from memory B cells in HIV-infected individuals. Nature 2009, 458, 636–640. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, J.E.; Kuroda, M.J.; Santra, S.; Simon, M.A.; Lifton, M.A.; Lin, W.; Khunkhun, R.; Piatak, M.; Lifson, J.D.; Grosschupff, G.; Gelman, R.S.; Racz, P.; Tenner-Racz, K.; Mansfield, K.A.; Letvin, N.L.; Montefiori, D.C.; Reimann, K.A. Effect of humoral immune responses on controlling viremia during primary infection of rhesus monkeys with simian immunodeficiency virus. J. Virol. 2003, 77, 2165–2173. [Google Scholar] [CrossRef] [PubMed]
- Pilgrim, A.K.; Pantaleo, G.; Cohen, O.J.; Fink, L.M.; Zhou, J.Y.; Zhou, J.T.; Bolognesi, D.P.; Fauci, A.S.; Montefiori, D.C. Neutralizing antibody responses to human immunodeficiency virus type 1 in primary infection and long-term-nonprogressive infection. J. Infect. Dis. 1997, 176, 924–932. [Google Scholar] [PubMed]
- Carotenuto, P.; Looij, D.; Keldermans, L.; de Wolf, F.; Goudsmit, J. Neutralizing antibodies are positively associated with CD4+ T-cell counts and T-cell function in long-term AIDS-free infection. AIDS 1998, 12, 1591–1600. [Google Scholar] [CrossRef] [PubMed]
- Montefiori, D.C.; Hill, T.S.; Vo, H.T.; Walker, B.D.; Rosenberg, E.S. Neutralizing antibodies associated with viremia control in a subset of individuals after treatment of acute human immunodeficiency virus type 1 infection. J. Virol. 2001, 75, 10200–10207. [Google Scholar] [CrossRef] [PubMed]
- Barouch, D.H. Challenges in the development of an HIV-1 vaccine. Nature 2008, 455, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Shibata, S.; Nagase, M.; Yoshida, S.; Kawarazaki, W.; Kurihara, H.; Tanaka, H.; Miyoshi, J.; Takai, Y.; Fujita, T. Modification of mineralocorticoid receptor function by Rac1 GTPase: implication in proteinuric kidney disease. Nat. Med. 2008, 14, 1370–1376. [Google Scholar] [CrossRef] [PubMed]
- Parren, P.W.; Marx, P.A.; Hessell, A.J.; Luckay, A.; Harouse, J.; Cheng-Mayer, C.; Moore, J.P.; Burton, D.R. Antibody protects macaques against vaginal challenge with a pathogenic R5 simian/human immunodeficiency virus at serum levels giving complete neutralization in vitro. J. Virol. 2001, 75, 8340–8347. [Google Scholar] [CrossRef] [PubMed]
- Hessell, A.J.; Poignard, P.; Hunter, M.; Hangartner, L.; Tehrani, D.M.; Bleeker, W.K.; Parren, P.W.; Marx, P.A.; Burton, D.R. Effective, low-titer antibody protection against low-dose repeated mucosal SHIV challenge in macaques. Nat. Med. 2009. [Google Scholar] [PubMed]
- Burton, D.R.; Montefiori, D.C. The antibody response in HIV-1 infection. AIDS 1997, 11, S87–98. [Google Scholar] [PubMed]
- Robert-Guroff, M. IgG surfaces as an important component in mucosal protection. Nat. Med. 2000, 6, 129–130. [Google Scholar] [CrossRef]
- Haigwood, N.L.; Hirsch, V.M. Blocking and tackling HIV. Nat. Med. 2009, 15, 841–842. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.M.; Nybakken, G.E.; Thompson, B.S.; Engle, M.J.; Marri, A.; Fremont, D.H.; Diamond, M.S. Antibodies against West Nile Virus nonstructural protein NS1 prevent lethal infection through Fc gamma receptor-dependent and -independent mechanisms. J. Virol. 2006, 80, 1340–1351. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.M.; Thompson, B.S.; Fremont, D.H.; Diamond, M.S. Antibody recognition of cell surface-associated NS1 triggers Fc-gamma receptor-mediated phagocytosis and clearance of West Nile Virus-infected cells. J. Virol. 2007, 81, 9551–9555. [Google Scholar] [CrossRef] [PubMed]
- Schlesinger, J.J.; Foltzer, M.; Chapman, S. The Fc portion of antibody to yellow fever virus NS1 is a determinant of protection against YF encephalitis in mice. Virology 1993, 192, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Ferrantelli, F.; Rasmussen, R.A.; Buckley, K.A.; Li, P.L.; Wang, T.; Montefiori, D.C.; Katinger, H.; Stiegler, G.; Anderson, D.C.; McClure, H.M.; Ruprecht, R.M. Complete protection of neonatal rhesus macaques against oral exposure to pathogenic simian-human immunodeficiency virus by human anti-HIV monoclonal antibodies. J. Infect. Dis. 2004, 189, 2167–2173. [Google Scholar] [CrossRef] [PubMed]
- Takeda, A.; Tuazon, C.U.; Ennis, F.A. Antibody-enhanced infection by HIV-1 via Fc receptor-mediated entry. Science 1988, 242, 580–583. [Google Scholar] [PubMed]
- Homsy, J.; Meyer, M.; Tateno, M.; Clarkson, S.; Levy, J.A. The Fc and not CD4 receptor mediates antibody enhancement of HIV infection in human cells. Science 1989, 244, 1357–1360. [Google Scholar] [PubMed]
- Trischmann, H.; Davis, D.; Lachmann, P.J. Lymphocytotropic strains of HIV type 1 when complexed with enhancing antibodies can infect macrophages via Fc gamma RIII, independently of CD4. AIDS Res. Hum. Retroviruses 1995, 11, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Montefiori, D.C. Role of complement and Fc receptors in the pathogenesis of HIV-1 infection. Springer Semin. Immunopathol. 1997, 18, 371–390. [Google Scholar] [CrossRef] [PubMed]
- Laurence, J.; Saunders, A.; Early, E.; Salmon, J.E. Human immunodeficiency virus infection of monocytes: relationship to Fc-gamma receptors and antibody-dependent viral enhancement. Immunology 1990, 70, 338–343. [Google Scholar] [PubMed]
- Jouault, T.; Chapuis, F.; Olivier, R.; Parravicini, C.; Bahraoui, E.; Gluckman, J.C. HIV infection of monocytic cells: role of antibody-mediated virus binding to Fc-gamma receptors. AIDS 1989, 3, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Robinson, W.E.; Montefiori, D.C.; Mitchell, W.M. A human immunodeficiency virus type 1 (HIV-1) infection-enhancing factor in seropositive sera. Biochem. Biophys. Res. Commun. 1987, 149, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Kozlowski, P.A.; Black, K.P.; Shen, L.; Jackson, S. High prevalence of serum IgA HIV-1 infection-enhancing antibodies in HIV-infected persons. Masking by IgG. J. Immunol. 1995, 154, 6163–6173. [Google Scholar] [PubMed]
- Stoiber, H.; Soederholm, A.; Wilflingseder, D.; Gusenbauer, S.; Hildgartner, A.; Dierich, M.P. Complement and antibodies: a dangerous liaison in HIV infection? Vaccine 2008, 26, I79–85. [Google Scholar] [CrossRef] [PubMed]
- Hidajat, R.; Xiao, P.; Zhou, Q.; Venzon, D.; Summers, L.E.; Kalyanaraman, V.S.; Montefiori, D.C.; Robert-Guroff, M. Correlation of vaccine-elicited systemic and mucosal nonneutralizing antibody activities with reduced acute viremia following intrarectal simian immunodeficiency virus SIVmac251 challenge of rhesus macaques. J. Virol. 2009, 83, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Perez-Bercoff, D.; David, A.; Sudry, H.; Barre-Sinoussi, F.; Pancino, G. Fcgamma receptor-mediated suppression of human immunodeficiency virus type 1 replication in primary human macrophages. J. Virol. 2003, 77, 4081–4094. [Google Scholar] [CrossRef] [PubMed]
- Connor, R.I.; Dinces, N.B.; Howell, A.L.; Romet-Lemonne, J.L.; Pasquali, J.L.; Fanger, M.W. Fc receptors for IgG (Fc gamma Rs) on human monocytes and macrophages are not infectivity receptors for human immunodeficiency virus type 1 (HIV-1): studies using bispecific antibodies to target HIV-1 to various myeloid cell surface molecules, including the Fc gamma R. Proc. Natl. Acad. Sci. U S A 1991, 88, 9593–9597. [Google Scholar] [CrossRef] [PubMed]
- Wilflingseder, D.; Banki, Z.; Garcia, E.; Pruenster, M.; Pfister, G.; Muellauer, B.; Nikolic, D.S.; Gassner, C.; Ammann, C.G.; Dierich, M.P.; Piguet, V.; Stoiber, H. IgG opsonization of HIV impedes provirus formation in and infection of dendritic cells and subsequent long-term transfer to T cells. J. Immunol. 2007, 178, 7840–7848. [Google Scholar] [PubMed]
- Choudhry, V.; Zhang, M.Y.; Harris, I.; Sidorov, I.A.; Vu, B.; Dimitrov, A.S.; Fouts, T.; Dimitrov, D.S. Increased efficacy of HIV-1 neutralization by antibodies at low CCR5 surface concentration. Biochem. Biophys. Res. Commun. 2006, 348, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Forthal, D.N.; Landucci, G.; Phan, T.B.; Becerra, J. Interactions between natural killer cells and antibody Fc result in enhanced antibody neutralization of human immunodeficiency virus type 1. J. Virol. 2005, 79, 2042–2049. [Google Scholar] [CrossRef] [PubMed]
- Forthal, D.N.; Gilbert, P.B.; Landucci, G.; Phan, T. Recombinant gp120 vaccine-induced antibodies inhibit clinical strains of HIV-1 in the presence of Fc receptor-bearing effector cells and correlate inversely with HIV infection rate. J. Immunol. 2007, 178, 6596–6603. [Google Scholar] [PubMed]
- Montefiori, D.C.; Morris, L.; Ferrari, G.; Mascola, J.R. Neutralizing and other antiviral antibodies in HIV-1 infection and vaccination. Curr. Opin. HIV AIDS 2007, 2, 169–176. [Google Scholar] [CrossRef]
- Connick, E.; Marr, D.G.; Zhang, X.Q.; Clark, S.J.; Saag, M.S.; Schooley, R.T.; Curiel, T.J. HIV-specific cellular and humoral immune responses in primary HIV infection. AIDS Res. Hum. Retroviruses 1996, 12, 1129–1140. [Google Scholar] [CrossRef] [PubMed]
- Rook, A.H.; Lane, H.C.; Folks, T.; McCoy, S.; Alter, H.; Fauci, A.S. Sera from HTLV-III/LAV antibody-positive individuals mediate antibody-dependent cellular cytotoxicity against HTLV-III/LAV-infected T cells. J. Immunol. 1987, 138, 1064–1067. [Google Scholar] [PubMed]
- Lyerly, H.K.; Reed, D.L.; Matthews, T.J.; Langlois, A.J.; Ahearne, P.A.; Petteway Jr., S.R.; Weinhold, K.J. Anti-GP 120 antibodies from HIV seropositive individuals mediate broadly reactive anti-HIV ADCC. AIDS Res. Hum. Retroviruses 1987, 3, 409–422. [Google Scholar] [CrossRef]
- Forthal, D.N.; Landucci, G.; Daar, E.S. Antibody from patients with acute human immunodeficiency virus (HIV) infection inhibits primary strains of HIV type 1 in the presence of natural-killer effector cells. J. Virol. 2001, 75, 6953–6961. [Google Scholar] [CrossRef] [PubMed]
- Forthal, D.N.; Landucci, G. In vitro reduction of virus infectivity by antibody-dependent cell-mediated immunity. J. Immunol. Methods 1998, 220, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Baum, L.L.; Cassutt, K.J.; Knigge, K.; Khattri, R.; Margolick, J.; Rinaldo, C.; Kleeberger, C.A.; Nishanian, P.; Henrard, D.R.; Phair, J. HIV-1 gp120-specific antibody-dependent cell-mediated cytotoxicity correlates with rate of disease progression. J. Immunol. 1996, 157, 2168–2173. [Google Scholar] [PubMed]
- Saez-Cirion, A.; Sinet, M.; Shin, S.Y.; Urrutia, A.; Versmisse, P.; Lacabaratz, C.; Boufassa, F.; Avettand-Fenoel, V.; Rouzioux, C.; Delfraissy, J.F.; Barre-Sinoussi, F.; Lambotte, O.; Venet, A.; Pancino, G. Heterogeneity in HIV suppression by CD8 T cells from HIV controllers: association with Gag-specific CD8 T cell responses. J. Immunol. 2009, 182, 7828–7837. [Google Scholar] [CrossRef] [PubMed]
- Lambotte, O.; Ferrari, G.; Moog, C.; Yates, N.L.; Liao, H.X.; Parks, R.J.; Hicks, C.B.; Owzar, K.; Tomaras, G.D.; Montefiori, D.C.; Haynes, B.F.; Delfraissy, J.F. Heterogeneous neutralizing antibody and antibody-dependent cell cytotoxicity responses in HIV-1 elite controllers. AIDS 2009, 23, 897–906. [Google Scholar] [CrossRef] [PubMed]
- Banks, N.D.; Kinsey, N.; Clements, J.; Hildreth, J.E. Sustained antibody-dependent cell-mediated cytotoxicity (ADCC) in SIV-infected macaques correlates with delayed progression to AIDS. AIDS Res. Hum. Retroviruses 2002, 18, 1197–1205. [Google Scholar] [PubMed]
- Binley, J.M.; Clas, B.; Gettie, A.; Vesanen, M.; Montefiori, D.C.; Sawyer, L.; Booth, J.; Lewis, M.; Marx, P.A.; Bonhoeffer, S.; Moore, J.P. Passive infusion of immune serum into simian immunodeficiency virus-infected rhesus macaques undergoing a rapid disease course has minimal effect on plasma viremia. Virology 2000, 270, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Fauci, A.S.; Mavilio, D.; Kottilil, S. NK cells in HIV infection: paradigm for protection or targets for ambush. Nat. Rev. Immunol. 2005, 5, 835–843. [Google Scholar] [CrossRef]
- Ahmad, A.; Menezes, J. Antibody-dependent cellular cytotoxicity in HIV infections. Faseb J. 1996, 10, 258–266. [Google Scholar] [PubMed]
- Valentin, A.; Rosati, M.; Patenaude, D.J.; Hatzakis, A.; Kostrikis, L.G.; Lazanas, M.; Wyvill, K.M.; Yarchoan, R.; Pavlakis, G.N. Persistent HIV-1 infection of natural killer cells in patients receiving highly active antiretroviral therapy. Proc. Natl. Acad. Sci. U S A 2002, 99, 7015–7020. [Google Scholar] [CrossRef] [PubMed]
- Chung, A.; Rollman, E.; Johansson, S.; Kent, S.J.; Stratov, I. The utility of ADCC responses in HIV infection. Curr. HIV Res. 2008, 6, 515–519. [Google Scholar] [CrossRef] [PubMed]
- Forthal, D.N.; Moog, C. Fc receptor-mediated antiviral antibodies. Current Opinion in HIV and AIDS 2009, 4, 388–393. [Google Scholar] [CrossRef]
- Oliva, A.; Kinter, A.L.; Vaccarezza, M.; Rubbert, A.; Catanzaro, A.; Moir, S.; Monaco, J.; Ehler, L.; Mizell, S.; Jackson, R.; Li, Y.; Romano, J.W.; Fauci, A.S. Natural killer cells from human immunodeficiency virus (HIV)-infected individuals are an important source of CC-chemokines and suppress HIV-1 entry and replication in vitro. J. Clin. Invest. 1998, 102, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Bogers, W.M.; Davis, D.; Baak, I.; Kan, E.; Hofman, S.; Sun, Y.; Mortier, D.; Lian, Y.; Oostermeijer, H.; Fagrouch, Z.; Dubbes, R.; van der Maas, M.; Mooij, P.; Koopman, G.; Verschoor, E.; Langedijk, J.P.; Zhao, J.; Brocca-Cofano, E.; Robert-Guroff, M.; Srivastava, I.; Barnett, S.; Heeney, J.L. Systemic neutralizing antibodies induced by long interval mucosally primed systemically boosted immunization correlate with protection from mucosal SHIV challenge. Virology 2008, 382, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Nag, P.; Kim, J.; Sapiega, V.; Landay, A.L.; Bremer, J.W.; Mestecky, J.; Reichelderfer, P.; Kovacs, A.; Cohn, J.; Weiser, B.; Baum, L.L. Women with cervicovaginal antibody-dependent cell-mediated cytotoxicity have lower genital HIV-1 RNA loads. J. Infect. Dis. 2004, 190, 1970–1978. [Google Scholar] [CrossRef] [PubMed]
- Battle-Miller, K.; Eby, C.A.; Landay, A.L.; Cohen, M.H.; Sha, B.E.; Baum, L.L. Antibody-dependent cell-mediated cytotoxicity in cervical lavage fluids of human immunodeficiency virus type 1--infected women. J. Infect. Dis. 2002, 185, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Forthal, D.N.; Landucci, G.; Cole, K.S.; Marthas, M.; Becerra, J.C.; Van Rompay, K. Rhesus macaque polyclonal and monoclonal antibodies inhibit simian immunodeficiency virus in the presence of human or autologous rhesus effector cells. J. Virol. 2006, 80, 9217–9225. [Google Scholar] [CrossRef] [PubMed]
- Ranki, A.; Mattinen, S.; Yarchoan, R.; Broder, S.; Ghrayeb, J.; Lahdevirta, J.; Krohn, K. T-cell response towards HIV in infected individuals with and without zidovudine therapy, and in HIV-exposed sexual partners. AIDS 1989, 3, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, M.; Lopalco, L.; Mazzotta, F.; Lo Caputo, S.; Veas, F.; Clerici, M. The 'immunologic advantage' of HIV-exposed seronegative individuals. AIDS 2009, 23, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Mazzoli, S.; Trabattoni, D.; Lo Caputo, S.; Piconi, S.; Ble, C.; Meacci, F.; Ruzzante, S.; Salvi, A.; Semplici, F.; Longhi, R.; Fusi, M.L.; Tofani, N.; Biasin, M.; Villa, M.L.; Mazzotta, F.; Clerici, M. HIV-specific mucosal and cellular immunity in HIV-seronegative partners of HIV-seropositive individuals. Nat. Med. 1997, 3, 1250–1257. [Google Scholar] [CrossRef] [PubMed]
- Kaul, R.; Trabattoni, D.; Bwayo, J.J.; Arienti, D.; Zagliani, A.; Mwangi, F.M.; Kariuki, C.; Ngugi, E.N.; MacDonald, K.S.; Ball, T.B.; Clerici, M.; Plummer, F.A. HIV-1-specific mucosal IgA in a cohort of HIV-1-resistant Kenyan sex workers. AIDS 1999, 13, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Hirbod, T.; Kaul, R.; Reichard, C.; Kimani, J.; Ngugi, E.; Bwayo, J.J.; Nagelkerke, N.; Hasselrot, K.; Li, B.; Moses, S.; MacDonald, K.S.; Broliden, K. HIV-neutralizing immunoglobulin A and HIV-specific proliferation are independently associated with reduced HIV acquisition in Kenyan sex workers. AIDS 2008, 22, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Devito, C.; Hinkula, J.; Kaul, R.; Kimani, J.; Kiama, P.; Lopalco, L.; Barass, C.; Piconi, S.; Trabattoni, D.; Bwayo, J.J.; Plummer, F.; Clerici, M.; Broliden, K. Cross-clade HIV-1-specific neutralizing IgA in mucosal and systemic compartments of HIV-1-exposed, persistently seronegative subjects. J. Acquir. Immune Defic. Syndr. 2002, 30, 413–420. [Google Scholar] [PubMed]
- Hirbod, T.; Kaldensjo, T.; Lopalco, L.; Klareskog, E.; Andersson, S.; Uberti-Foppa, C.; Ferrari, D.; Manghi, M.; Andersson, J.; Lore, K.; Broliden, K. Abundant and superficial expression of C-type lectin receptors in ectocervix of women at risk of HIV infection. J. Acquir. Immune Defic. Syndr. 2009, 51, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Piacentini, L.; Fenizia, C.; Naddeo, V.; Clerici, M. Not just sheer luck! Immune correlates of protection against HIV-1 infection. Vaccine 2008, 26, 3002–3007. [Google Scholar] [CrossRef] [PubMed]
- Broliden, K.; Hinkula, J.; Devito, C.; Kiama, P.; Kimani, J.; Trabbatoni, D.; Bwayo, J.J.; Clerici, M.; Plummer, F.; Kaul, R. Functional HIV-1 specific IgA antibodies in HIV-1 exposed, persistently IgG seronegative female sex workers. Immunol. Lett. 2001, 79, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Beyrer, C.; Artenstein, A.W.; Rugpao, S.; Stephens, H.; VanCott, T.C.; Robb, M.L.; Rinkaew, M.; Birx, D.L.; Khamboonruang, C.; Zimmerman, P.A.; Nelson, K.E.; Natpratan, C. Epidemiologic and biologic characterization of a cohort of human immunodeficiency virus type 1 highly exposed, persistently seronegative female sex workers in northern Thailand. Chiang Mai HEPS Working Group. J. Infect. Dis. 1999, 179, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Hirbod, T.; Broliden, K. Mucosal immune responses in the genital tract of HIV-1-exposed uninfected women. J. Intern. Med. 2007, 262, 44–58. [Google Scholar] [CrossRef] [PubMed]
- Buchacz, K.; Parekh, B.S.; Padian, N.S.; van der Straten, A.; Phillips, S.; Jonte, J.; Holmberg, S.D. HIV-specific IgG in cervicovaginal secretions of exposed HIV-uninfected female sexual partners of HIV-infected men. AIDS Res. Hum. Retroviruses 2001, 17, 1689–1693. [Google Scholar] [PubMed]
- Lo Caputo, S.; Trabattoni, D.; Vichi, F.; Piconi, S.; Lopalco, L.; Villa, M.L.; Mazzotta, F.; Clerici, M. Mucosal and systemic HIV-1-specific immunity in HIV-1-exposed but uninfected heterosexual men. AIDS 2003, 17, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, C.; VanCott, T.; Bosire, R.; Bermudez, C.; Mbori-Ngacha, D.; Lohman-Payne, B.; Nduati, R.; Otieno, P.; John-Stewart, G. Salivary human immunodeficiency virus (HIV)-1-specific immunoglobulin A in HIV-1-exposed infants in Kenya. Clin. Exp. Immunol. 2008, 153, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Duprat, C.; Mohammed, Z.; Datta, P.; Stackiw, W.; Ndinya-Achola, J.O.; Kreiss, J.K.; Holmes, K.K.; Plummer, F.A.; Embree, J.E. Human immunodeficiency virus type 1 IgA antibody in breast milk and serum. Pediatr. Infect. Dis. J. 1994, 13, 603–608. [Google Scholar] [PubMed]
- Ghys, P.D.; Belec, L.; Diallo, M.O.; Ettiegne-Traore, V.; Becquart, P.; Maurice, C.; Nkengasong, J.N.; Coulibaly, I.M.; Greenberg, A.E.; Laga, M.; Wiktor, S.Z. Cervicovaginal anti-HIV antibodies in HIV-seronegative female sex workers in Abidjan, Cote d'Ivoire. AIDS 2000, 14, 2603–2608. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.; Pean, P.; Lopalco, L.; Nouhin, J.; Phoung, V.; Ly, N.; Vermisse, P.; Henin, Y.; Barre-Sinoussi, F.; Burastero, S.E.; Reynes, J.M.; Carcelain, G.; Pancino, G. HIV-specific antibodies but not t-cell responses are associated with protection in seronegative partners of HIV-1-infected individuals in Cambodia. J. Acquir. Immune Defic. Syndr. 2006, 42, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Tudor, D.; Derrien, M.; Diomede, L.; Drillet, A.S.; Houimel, M.; Moog, C.; Reynes, J.M.; Lopalco, L.; Bomsel, M. HIV-1 gp41-specific monoclonal mucosal IgAs derived from highly exposed but IgG-seronegative individuals block HIV-1 epithelial transcytosis and neutralize CD4(+) cell infection: an IgA gene and functional analysis. Mucosal Immunol. 2009, 2, 412–426. [Google Scholar] [CrossRef]
- Walker, B.D.; Burton, D.R. Toward an AIDS vaccine. Science 2008, 320, 760–764. [Google Scholar] [CrossRef] [PubMed]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Share and Cite
Holl, V.; Peressin, M.; Moog, C. Antibody-Mediated Fcγ Receptor-Based Mechanisms of HIV Inhibition: Recent Findings and New Vaccination Strategies. Viruses 2009, 1, 1265-1294. https://doi.org/10.3390/v1031265
Holl V, Peressin M, Moog C. Antibody-Mediated Fcγ Receptor-Based Mechanisms of HIV Inhibition: Recent Findings and New Vaccination Strategies. Viruses. 2009; 1(3):1265-1294. https://doi.org/10.3390/v1031265
Chicago/Turabian StyleHoll, Vincent, Maryse Peressin, and Christiane Moog. 2009. "Antibody-Mediated Fcγ Receptor-Based Mechanisms of HIV Inhibition: Recent Findings and New Vaccination Strategies" Viruses 1, no. 3: 1265-1294. https://doi.org/10.3390/v1031265
APA StyleHoll, V., Peressin, M., & Moog, C. (2009). Antibody-Mediated Fcγ Receptor-Based Mechanisms of HIV Inhibition: Recent Findings and New Vaccination Strategies. Viruses, 1(3), 1265-1294. https://doi.org/10.3390/v1031265