Molecular Characterization of Viruses from Clinical Respiratory Samples Producing Unidentified Cytopathic Effects in Cell Culture
Abstract
:1. Introduction
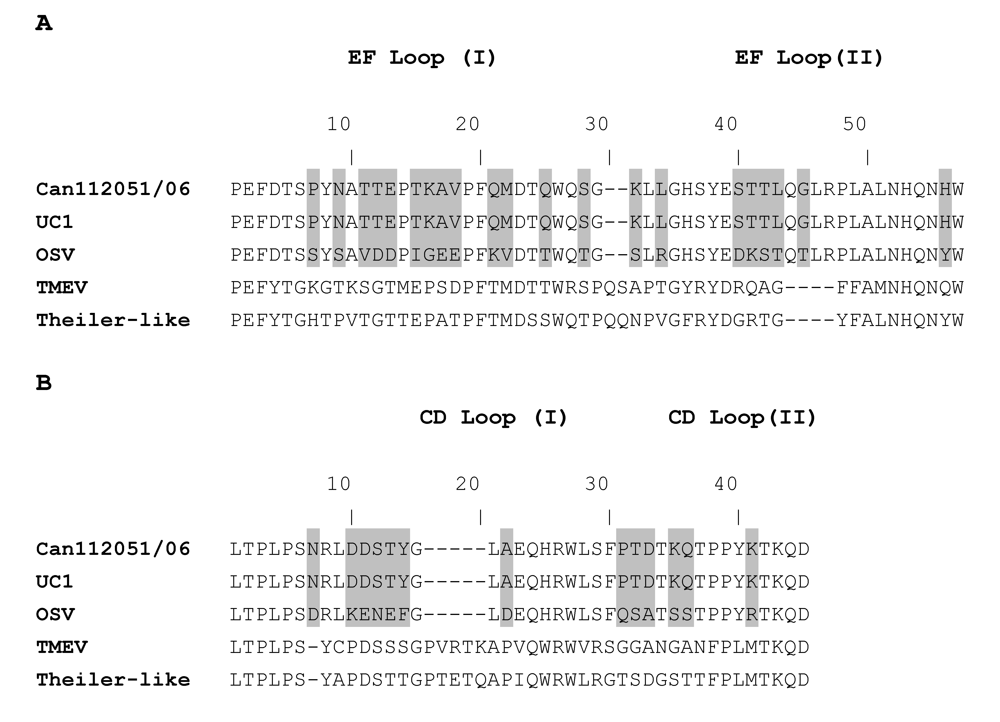
2. Results and Discussion
| Isolate/year | Specimen | Virus | Time to CPE |
|---|---|---|---|
| Can90775/2003 | NPA | Influenza C | 19 days |
| Can101516/2005 | BAL | HPeV-3 | 6 days |
| Can101519/2005 | BAL | HPeV-3 | 6 days |
| Can112051/2006 | NPA | Cardiovirus Saffold | 4 days |
| Can116603/2006 | NPA | Cardiovirus Saffold | 4 days |
| Can116604/2006 | NPA | Cardiovirus Saffold | 4 days |
3. Experimental Section
4. Conclusions
References
- Abed, Y.; Boivin, G. Treatment of respiratory virus infections. Antivir. Res. 2006, 70, 1–16. [Google Scholar] [CrossRef]
- Ruiz, M.; Ewig, S.; Marcos, M. Etiology of community-acquired pneumonia: impact of age, comorbidity, and severity. Am. J. Respir. Crit. Care Med. 1999, 160, 397–405. [Google Scholar] [PubMed]
- Davies, H.D.; Matlow, A.; Petric, M.; Glazier, R.; Wang, E.E. Prospective comparative study of viral, bacterial and atypical organisms identified in pneumonia and bronchiolitis in hospitalized Canadian infants. Pediatr. Infect. Dis. J. 1996, 15, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Allander, T.; Tammi, M.T.; Eriksson, M.; Bjerkner, A.; Tiveljung-Lindell, A. Cloning of a human parvovirus by molecular screening of respiratory tract samples. Proc. Natl. Acad. Sci. USA. 2005, 102, 12891–12896. [Google Scholar] [CrossRef]
- Jones, M.S.; Kapoor, A.; Lukashov, V.L.; Simmonds, P.; Hecht, F.; Delwart, E. New DNA viruses identified in patients with acute viral infection syndrome. J. Virol. 2005, 79, 8230–8236. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.S.; Lukashov, V.V.; Ganac, R.D.; Schnurr, D.P. Discovery of a novel human picornavirus in a stool sample from a pediatric patient presenting with fever of unknown origin. J. Clin. Microbiol. 2007, 45, 2144–2150. [Google Scholar] [CrossRef] [PubMed]
- Gouarin, S.; Vabret, A.; Dina, J.; Petitjean, J.; Brouard, J.; Cuvillon-Nimal, D.; Freymuth, F. Study of influenza C infection in France. J. Med. Virol. 2008, 80, 1441–1446. [Google Scholar] [CrossRef] [PubMed]
- Harval, H.; Simmonds, P. Human parechoviruses: Biology, epidemiology and clinical significance. J. Clin. Virol. 2009, 45, 1–9. [Google Scholar] [CrossRef]
- Abed, Y.; Boivin, G. Human parechovirus infections in Canada. Emerg. Infect. Dis. 2006, 12, 969–975. [Google Scholar] [PubMed]
- Boivin, G.; Abed, Y.; Boucher F.D. Human parechovirus-3 and neonatal infections. Emerg. Infect. Dis. 2005, 11, 103–105. [Google Scholar] [PubMed]
- Benschop, K.S.; Schinkel, J.; Minnaar, R.P.; Pajkrt, D.; Spanjerberg, L.; Kraakman, H.C. Human parechovirus infections in Dutch children and the association between serotype and disease severity. Clin. Infect. Dis. 2006, 15, 204–210. [Google Scholar] [CrossRef]
- Abed, Y.; Boivin, G. New Saffold cardioviruses in 3 children, Canada. Emerg. Infect. Dis. 2008, 14, 834–836. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.Y.; Greninger, A.L.; Kanada, K.; Kwok, T.; Fischer, K.F.; Runckel, C. Identification of cardioviruses related to Theiler’s murine encephalomyelitis virus in human infections. Proc. Natl. Acad. Sci. USA. 2008, 105, 14124–14129. [Google Scholar] [CrossRef]
- King, A.M.Q.; Brown, F.; Christian, P.; Hovi, T.; Hyypia, T.; Knowles, N.J. Family Picornaviridae. In Virus Taxonomy; Van Regenmortel, M.H.V., Fauquet, C.M., Bishop, D.H.L., Carstens, E.B., Estes, M.K., Lemon, S.M., Maniloff, J., Mayo, M.A., McGeoch, D.J., Pringle, C.R., Wickner, R.B., Eds.; Academic Press: San Diego, CA, USA, 2000. [Google Scholar]
- Blinkova, O.; Kapoor, A.; Victoria, J.; Jones, M.; Wolfe, N.; Naeem, A.; Shaukat, S.; Sharif, S.; Alam, M.M.; Angez, M.; Zaidi, S.; Delwart, E.L. Cardioviruses are genetically diverse and cause common enteric infections in South Asian children. J. Virol. 2009, 83, 4631–4641. [Google Scholar] [CrossRef] [PubMed]
- Jnaoui, K.; Minet, M.; Michiels, T. Mutations that affect the tropism of DA and GDVII strains of Theiler’s virus in vitro influence sialic acid binding and pathogenicity. J. Virol. 2002, 76, 8138–8147. [Google Scholar] [CrossRef] [PubMed]
- Greninger, A.L.; Runckel, C.; Chiu, C.Y.; Haggerty, T.; Parsonnet, J.; Ganem, D.; DeRisi, J.L. The complete genome of klassevirus-a novel picornavirus in pediatric stool. Virol. J. 2009, 6, 82. [Google Scholar] [CrossRef]
- Wang, D.; Coscoy, L.; Zylberberg, M.; Avila, P.C.; Boushey, H.A.; Ganem, D.; DeRisi, J.L. Microarray-based detection and genotyping of viral pathogens. Proc. Natl. Acad. Sci. USA. 2002, 99, 15687–15692. [Google Scholar] [CrossRef]
- Boivin, G.; De Serres, G.; Cote, S.; Gilca, R.; Abed, Y.; Rochette, L. Human metapneumovirus infections in hospitalized children. Emerg. Infect. Dis. 2003, 9, 634–640. [Google Scholar] [CrossRef] [PubMed]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Abed, Y.; Boivin, G. Molecular Characterization of Viruses from Clinical Respiratory Samples Producing Unidentified Cytopathic Effects in Cell Culture. Viruses 2009, 1, 84-90. https://doi.org/10.3390/v1020084
Abed Y, Boivin G. Molecular Characterization of Viruses from Clinical Respiratory Samples Producing Unidentified Cytopathic Effects in Cell Culture. Viruses. 2009; 1(2):84-90. https://doi.org/10.3390/v1020084
Chicago/Turabian StyleAbed, Yacine, and Guy Boivin. 2009. "Molecular Characterization of Viruses from Clinical Respiratory Samples Producing Unidentified Cytopathic Effects in Cell Culture" Viruses 1, no. 2: 84-90. https://doi.org/10.3390/v1020084
APA StyleAbed, Y., & Boivin, G. (2009). Molecular Characterization of Viruses from Clinical Respiratory Samples Producing Unidentified Cytopathic Effects in Cell Culture. Viruses, 1(2), 84-90. https://doi.org/10.3390/v1020084




