Effect of Stem Diameter, Genetics, and Wood Properties on Stem Cracking in Norway Spruce
Abstract
:1. Introduction
2. Materials and Methods
2.1. Studied Trial and Measurements
2.2. Data Analysis
3. Results
3.1. Effect of DBH and Genetics
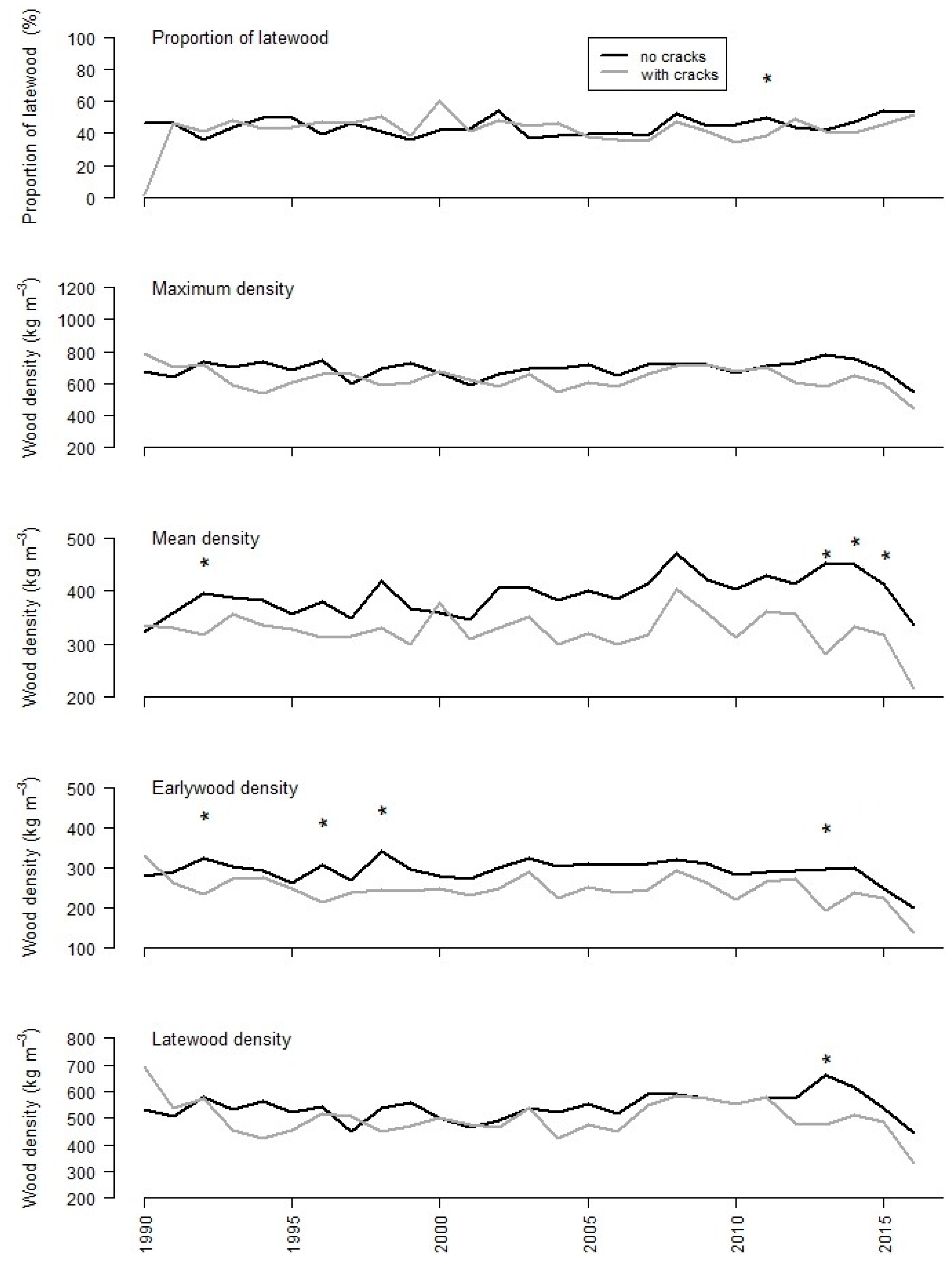
3.2. Tree-Ring Analysis
4. Discussion
Tree-Ring Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schelhaas, M.J.; Varis, S.; Schuck, A.; Nabuurs, G.J. EFISCEN Inventory Database, European Forest Institute, Joensuu, Finland. Available online: http://www.efi.int/portal/virtual_library/databases/efiscen (accessed on 7 August 2017).
- Westin, J.; Haapanen, M. Norway spruce—Picea abies (L.) Karst. In Best Practice for Tree Breeding in Europe; Mullin, T.J., Lee, S.J., Eds.; Skogforsk: Uppsala, Sweden, 2013; pp. 29–47. [Google Scholar]
- Aarrestad, P.A.; Myking, T.; Stabbetorp, O.E.; Tollefsrud, M.M. Foreign Norway Spruce (Picea abies) Provenances in Norway and Effects on Biodiversity; Norsk institutt for naturforskning: Trondheim, Norway, 2014. [Google Scholar]
- Dzerina, B.; Girdziusas, S.; Lazdina, D.; Lazdins, A.; Jansons, J.; Neimane, U.; Jansons, Ā. Influence of spot mounding on height growth and tending of Norway spruce: Case study in Latvia. For. Stud. 2016, 65, 24–33. [Google Scholar] [CrossRef]
- Neimane, U.; Katrevics, J.; Sisenis, L.; Purins, M.; Luguza, S.; Adamovics, A. Intra-annual dynamics of height growth of Norway spruce in Latvia. Agron. Res. 2016, 14, 853–861. [Google Scholar]
- Haapanen, M.; Jansson, G.; Nielsen, U.B.; Steffenrem, A.; Stener, L.-G. The Status of Tree Breeding and Its Potential for Improving Biomass Production: A Review of Breeding Activities and Genetic Gains in Scandinavia and Finland; SkogForsk: Uppsala, Sweden, 2015. [Google Scholar]
- Jansons, Ā.; Donis, J.; Danusevičius, D.; Baumanis, I. Differential analysis for next breeding cycle for Norway spruce in Latvia. Balt. For. 2015, 21, 285–297. [Google Scholar]
- Lindner, M.; Maroschek, M.; Netherer, S.; Kremer, A.; Barbati, A.; Garcia-Gonzalo, J.; Seidl, R.; Delzon, S.; Corona, P.; Kolström, M.; et al. Climate change impacts, adaptive capacity, and vulnerability of European forest ecosystems. For. Ecol. Manag. 2010, 259, 698–709. [Google Scholar] [CrossRef]
- Avotniece, Z.; Rodinov, V.; Lizuma, L.; Briede, A.; Kļaviņš, M. Trends in the frequency of extreme climate events in Latvia. Balt. 2010, 23, 135–148. [Google Scholar]
- Dietrichson, J.; Rognerud, P.A.; Haveraaen, O.; Skroppa, T. Stem cracks in Norway spruce (Picea abies (L.) Karst.). Rep. Nor. For. Res. Inst. 1985, 38, 1–32. [Google Scholar]
- Caspari, C.O.; Sachsse, H. Cracking of Norway spruce. Forst Holz 1990, 45, 685–688. (In German) [Google Scholar]
- Grabner, M.; Cherubini, P.; Rozenberg, P.; Hannrup, B. Summer drought and low earlywood density induce intra-annual radial cracks in conifers. Scand. J. For. Res. 2006, 21, 151–157. [Google Scholar] [CrossRef]
- Persson, A. Stem cracks in Norway spruce in southern Scandinavia: Causes and consequences. Ann. For. Sci. 1994, 51, 315–327. [Google Scholar] [CrossRef]
- Vasiliauskas, R.; Juška, E.; Stenlid, J.; Vasiliauskas, A. Clonal differences and relations between diameter growth, stem cracks and fungi in a 36-year-old clonal seed orchard of Norway spruce (Picea abies (L.) Karst.). Silvae Genet. 2001, 50, 227–233. [Google Scholar]
- Arhipova, N.; Jansons, A.; Zaluma, A.; Gaitnieks, T.; Vasaitis, R. Bark stripping of Pinus contorta caused by moose and deer: Wounding patterns, discoloration of wood, and associated fungi. Can. J. For. Res. 2015, 45, 1434–1438. [Google Scholar] [CrossRef]
- Burneviča, N.; Jansons, Ā.; Zaļuma, A.; Kļaviņa, D.; Jansons, J.; Gaitnieks, T. Fungi inhabiting bark stripping wounds made by large game on stems of Picea abies (L.) Karst. in Latvia. Balt. For. 2016, 22, 2–7. [Google Scholar]
- Baders, E.; Donis, J.; Snepsts, G.; Adamovics, A.; Jansons, A. Pruning effect on Norway spruce (Picea abies (L.) Karst.) growth and quality. For. Stud. 2017, 66, 33–48. [Google Scholar] [CrossRef]
- Napola, J. The success of the origins of Eastern and Mid-European Norway spruce in southern Finland. Work. Pap. Finn. For. Res. 2014, 288, 77. (In Finnish) [Google Scholar]
- Napola, M.; Napola, J. Observations and results of stem damages in the experimental Norway spruce plantations. Work. Pap. Finn. For. Res. 2014, 296, 51. (In Finnish) [Google Scholar]
- Zeltiņš, P.; Katrevičs, J.; Gailis, A.; Maaten, T.; Jansons, J.; Jansons, Ā. Stem cracks of Norway spruce (Picea abies (L.) Karst.) provenances in Western Latvia. For. Stud. 2016, 65, 57–63. [Google Scholar] [CrossRef]
- Harris, I.; Jones, P.D.; Osborn, T.J.; Lister, D.H. Updated high-resolution grids of monthly climatic observations—The CRU TS3.10 Dataset. Int. J. Climatol. 2014, 34, 623–642. [Google Scholar] [CrossRef] [Green Version]
- Spiecker, H.; Hansen, N.; Schinker, M.G. High-Frequency Densitometry—A New Method for the Rapid Evaluation of Wood Density Variations. IAWA J. 2003, 24, 231–239. [Google Scholar]
- Gilmour, A.R.; Anderson, R.D.; Rae, A.L. The analysis of binomial data by a generalised linear mixed model. Biometrika 1985, 72, 593–599. [Google Scholar] [CrossRef]
- Falconer, D.S.; Longman, T.F.C.M. Introduction to Quantitative Genetics, 4th ed.; Longman Group Ltd.: London, UK, 1996. [Google Scholar]
- Christensen, R.H.B. Ordinal—Regression Models for Ordinal Data. R Package Version 2015.6-28. 2015. Available online: https://cran.r-project.org/web/packages/ordinal/ordinal.pdf (accessed on 21 February 2018).
- Grissino-Mayer, H.D. Evaluating crossdating accuracy: A manual and tutorial for the computer program COFECHA. Tree-Ring Res. 2001, 57, 205–221. [Google Scholar]
- Jansons, Ā.; Matisons, R.; Krišāns, O.; Džeriņa, B.; Zeps, M. Effect of initial fertilization on 34-year increment and wood properties of Norway spruce in Latvia. Silva Fenn. 2016, 50, 1346. [Google Scholar] [CrossRef]
- R Core Team R: A Language and Environment for Statistical Computing 2016. Available online: http://www.R-project.org/. (accessed on 22 February 2018).
- Persson, A. How genotype and silviculture interact in forming timber properties. Silva Fenn. 1994, 28, 275–282. [Google Scholar] [CrossRef] [Green Version]
- Hannrup, B.; Cahalan, C.; Chantre, G.; Grabner, M.; Karlsson, B.; Le Bayon, I.; Jones, G.L.; Müller, U.; Pereira, H.; Rodrigues, J.C.; et al. Genetic Parameters of Growth and Wood Quality Traits in Picea abies. Scand. J. For. Res. 2004, 19, 14–29. [Google Scholar] [CrossRef]
- Giertych, M. Genetics. In Biology and Ecology of Norway Spruce; Tjoelker, M.G., Boratyński, A., Bugała, W., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 115–155. [Google Scholar]
- Persson, B.; Persson, A. Variation in stem properties in a IUFRO 1964/1968 Picea abies provenance experiment in southern Sweden. Silvae Genet. 1997, 46, 94–101. [Google Scholar]
- Eriksson, G.; Andersson, S.; Schelander, B. Lovande tillvaxt hos introducerade granprovenienser i en kombinerad klon-och froplantsplantage i norra Uppland. Sver Skogvardsforb Tidskr 1975, 3, 277–286. [Google Scholar]
- Suvanto, S.; Nöjd, P.; Henttonen, H.M.; Beuker, E.; Mäkinen, H. Geographical patterns in the radial growth response of Norway spruce provenances to climatic variation. Agric. For. Meteorol. 2016, 222, 10–20. [Google Scholar] [CrossRef] [Green Version]
- Haapanen, M. Impact of family-by-trial interaction on the utility of progeny testing methods for scots pine. Silvae Genet. 1996, 45, 130–135. [Google Scholar]
- Haapanen, M.; Velling, P.; Annala, M.-L. Progeny trial estimates of genetic parameters for growth and quality traits in Scots pine. Silva Fenn. 1997, 31, 3–12. [Google Scholar] [CrossRef]
- Molteberg, D.; Høibø, O. Development and variation of wood density, kraft pulp yield and fibre dimensions in young Norway spruce (Picea abies). Wood Sci. Technol. 2006, 40, 173–189. [Google Scholar] [CrossRef]
- Irbe, I.; Sable, I.; Noldt, G.; Grinfelds, U.; Jansons, A.; Treimanis, A.; Koch, G. Wood and tracheid properties of Norway spruce (Picea abies [L.] Karst.) clones grown on former agricultural land in Latvia. Balt. For. 2015, 21, 114–123. [Google Scholar]
- Mattheck, C. Wood—The internal optimization of trees. Arboric. J. 1995, 19, 97–110. [Google Scholar] [CrossRef]
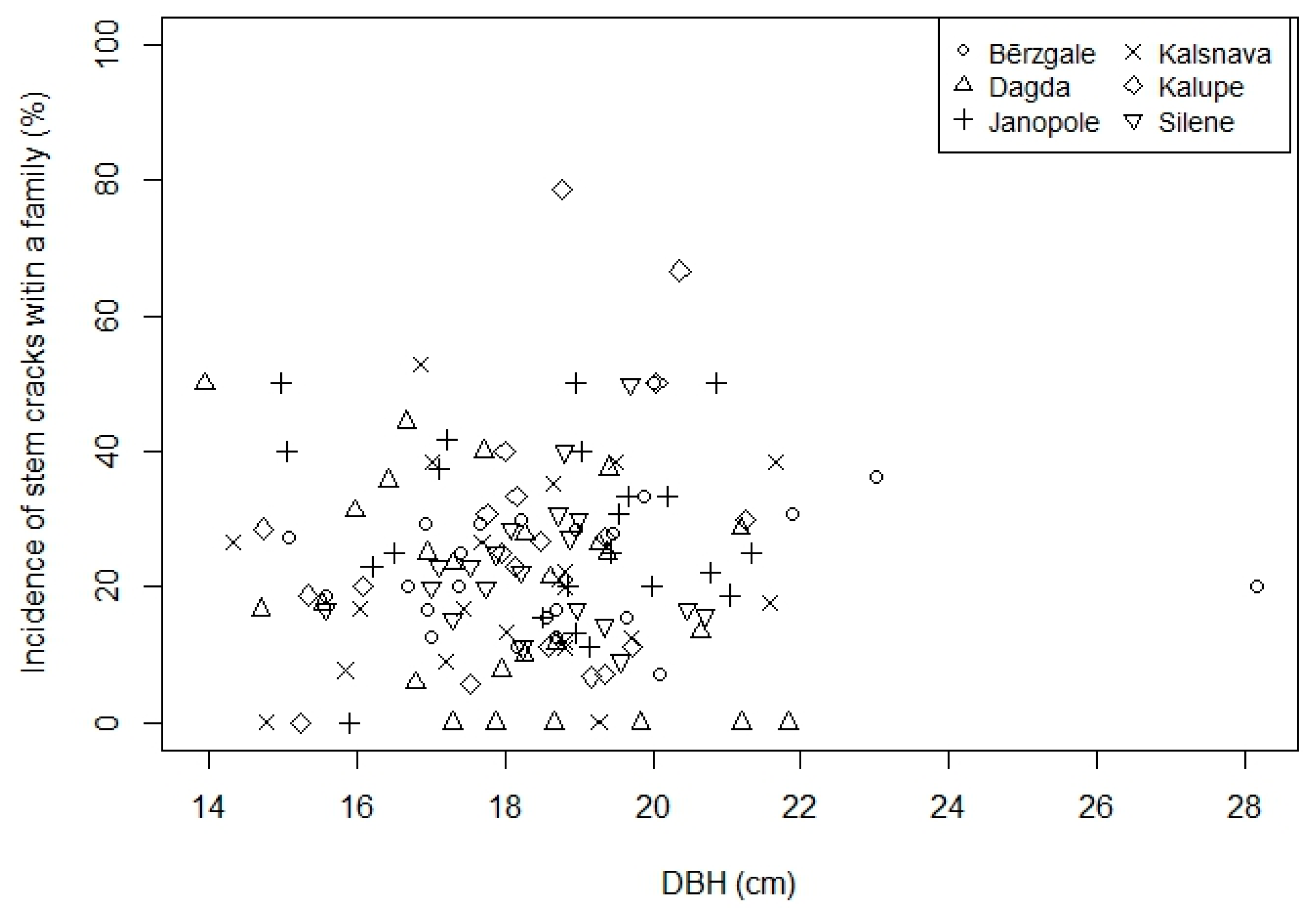
| Provenance | Geographic Coordinates | No of Families | No of Trees with Cracking Score | Mean Diameter at Breast Height ± SD (cm) | Incidence of Stem Cracks (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 5 | without Stem Cracks | with Stem Cracks | ||||
| Bērzgale | 56°36′ N, 27°30′ E | 26 | 210 | 20 | 12 | 13 | 12 | 7 | 18.2 ± 4.63 | 19.8 ± 5.66 | 23.4 |
| Dagda | 56°6′ N, 27°30′ E | 29 | 263 | 31 | 16 | 14 | 2 | 6 | 18.1 ± 4.08 | 18.2 ± 4.82 | 20.8 |
| Janopole | 56°23′ N, 27°18′ E | 26 | 178 | 16 | 19 | 12 | 8 | 14 | 18.2 ± 4.33 | 20.7 ± 4.68 | 27.9 |
| Kalsnava | 55°42′ N, 26°41′ E | 24 | 227 | 17 | 18 | 10 | 8 | 12 | 17.9 ± 4.06 | 19.1 ± 4.57 | 22.3 |
| Kalupe | 56°6′ N, 26°30′ E | 24 | 202 | 18 | 15 | 8 | 11 | 15 | 17.9 ± 4.05 | 18.8 ± 4.60 | 24.9 |
| Silene | 56°42′ N, 25°53′ E | 23 | 202 | 22 | 10 | 10 | 5 | 13 | 18.2 ± 4.47 | 19.9 ± 4.72 | 22.9 |
| Total | 152 | 1282 | 124 | 90 | 67 | 46 | 67 | 18.1 ± 4.27 | 19.4 ± 4.84 | 23.5 | |
| Component | Estimate (Standard Error) |
|---|---|
| Additive genetic (family) variance | 0.074 (0.077) |
| Variance of additive genetic variance | 0.006 |
| Family × block variance | 0.013 (0.130) |
| Environmental variance | 3.29 |
| Individual-tree narrow sense heritability | 0.088 (0.091) |
| Family mean heritability | 0.198 (0.206) |
| MIN | MAX | MEAN | SD | DIFF | ||
|---|---|---|---|---|---|---|
| Latewood proportion | ||||||
| no cracks | 0.36 | 0.54 | 0.44 | 0.05 | 0.79 | |
| cracks | 0.34 | 0.60 | 0.44 | 0.06 | ||
| Maximum density (kg m−3) | ||||||
| no cracks | 594 | 783 | 696 | 45.4 | <0.01 | |
| cracks | 539 | 716 | 633 | 53.0 | ||
| Mean density (kg m−3) | ||||||
| no cracks | 345 | 471 | 398 | 32.9 | <0.01 | |
| cracks | 282 | 402 | 330 | 27.4 | ||
| Mean earlywood density (kg m−3) | ||||||
| no cracks | 250 | 341 | 297 | 21.0 | <0.01 | |
| cracks | 194 | 292 | 247 | 23.6 | ||
| Mean latewood density (kg m−3) | ||||||
| no cracks | 449 | 661 | 547 | 46.0 | <0.01 | |
| cracks | 422 | 586 | 501 | 49.6 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeltiņš, P.; Katrevičs, J.; Gailis, A.; Maaten, T.; Bāders, E.; Jansons, Ā. Effect of Stem Diameter, Genetics, and Wood Properties on Stem Cracking in Norway Spruce. Forests 2018, 9, 546. https://doi.org/10.3390/f9090546
Zeltiņš P, Katrevičs J, Gailis A, Maaten T, Bāders E, Jansons Ā. Effect of Stem Diameter, Genetics, and Wood Properties on Stem Cracking in Norway Spruce. Forests. 2018; 9(9):546. https://doi.org/10.3390/f9090546
Chicago/Turabian StyleZeltiņš, Pauls, Juris Katrevičs, Arnis Gailis, Tiit Maaten, Endijs Bāders, and Āris Jansons. 2018. "Effect of Stem Diameter, Genetics, and Wood Properties on Stem Cracking in Norway Spruce" Forests 9, no. 9: 546. https://doi.org/10.3390/f9090546
APA StyleZeltiņš, P., Katrevičs, J., Gailis, A., Maaten, T., Bāders, E., & Jansons, Ā. (2018). Effect of Stem Diameter, Genetics, and Wood Properties on Stem Cracking in Norway Spruce. Forests, 9(9), 546. https://doi.org/10.3390/f9090546






