Patterns in Ectomycorrhizal Diversity, Community Composition, and Exploration Types in European Beech, Pine, and Spruce Forests
Abstract
1. Introduction
2. Materials and Methods
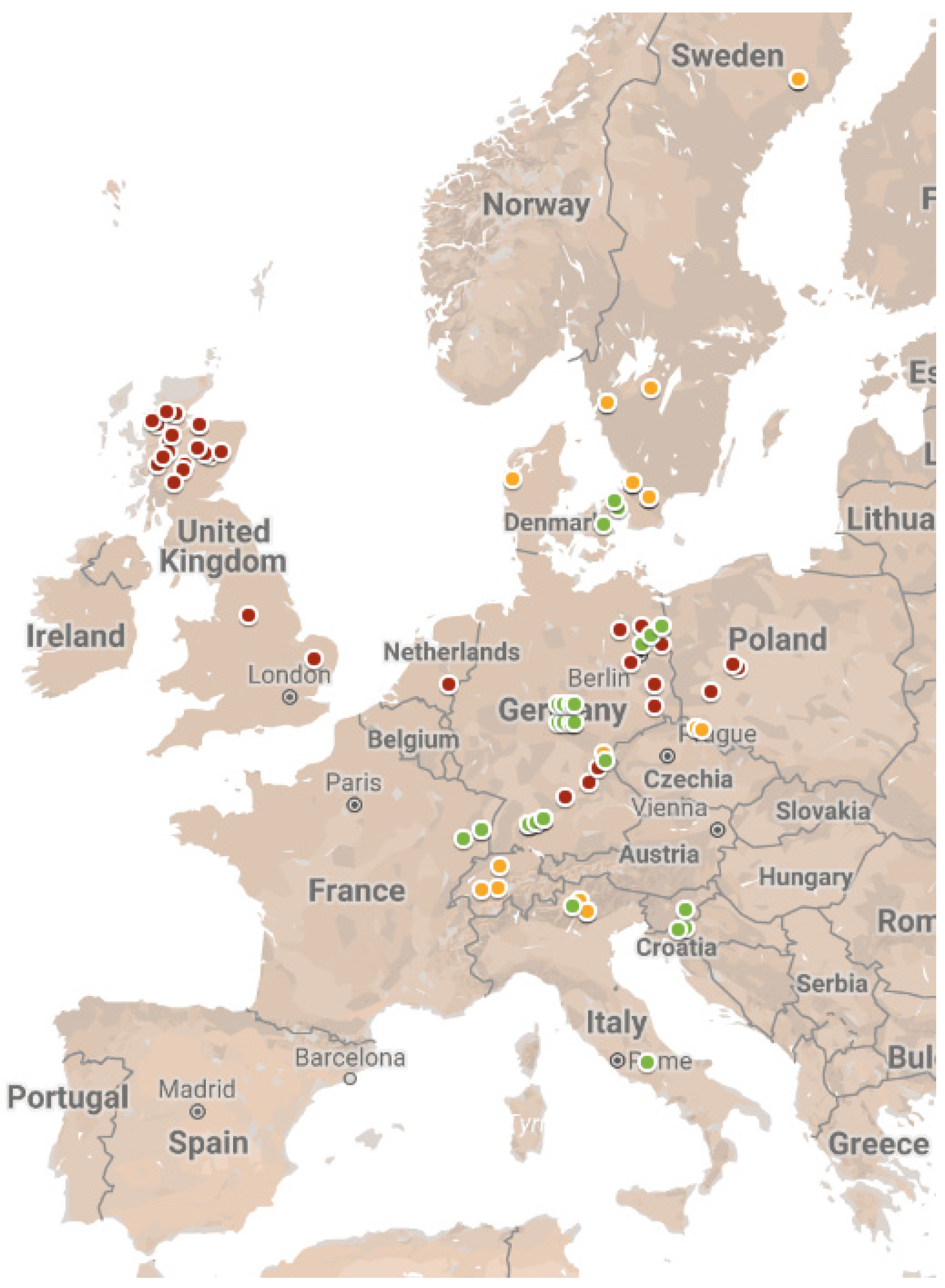
2.1. Data Collection
2.2. Data Analysis
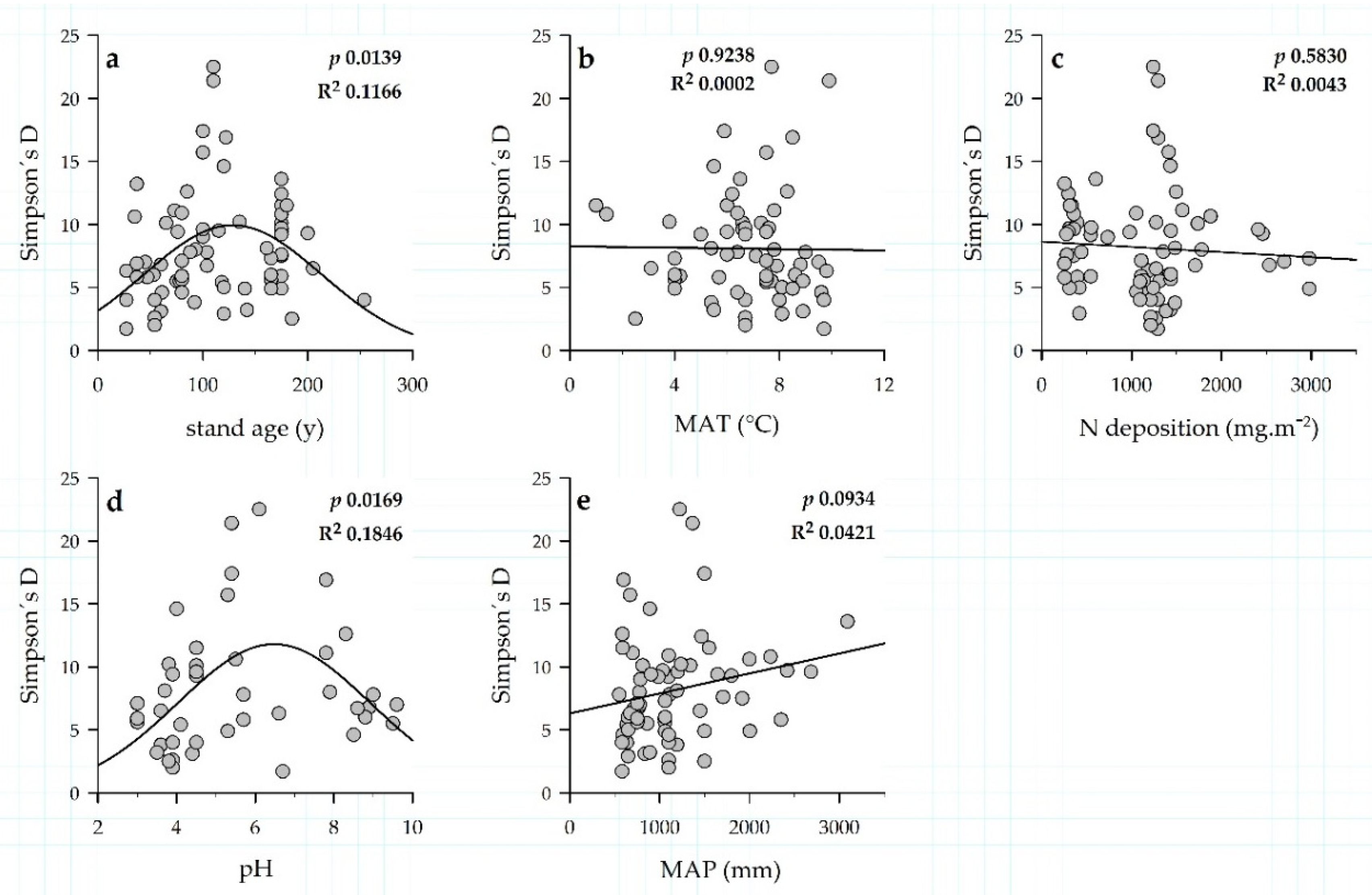
3. Results
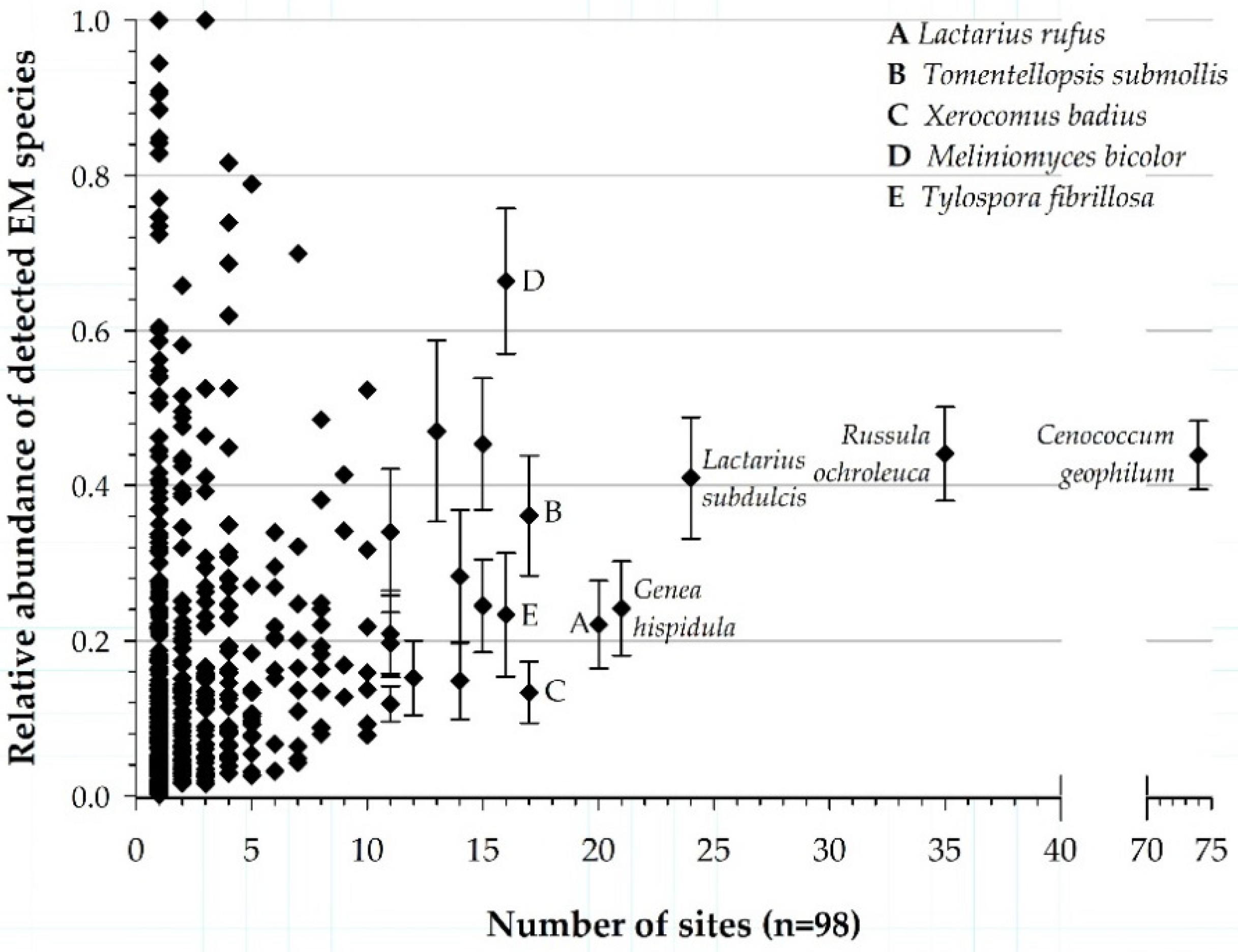
3.1. Patterns in Frequency and Abundance of EM Taxa
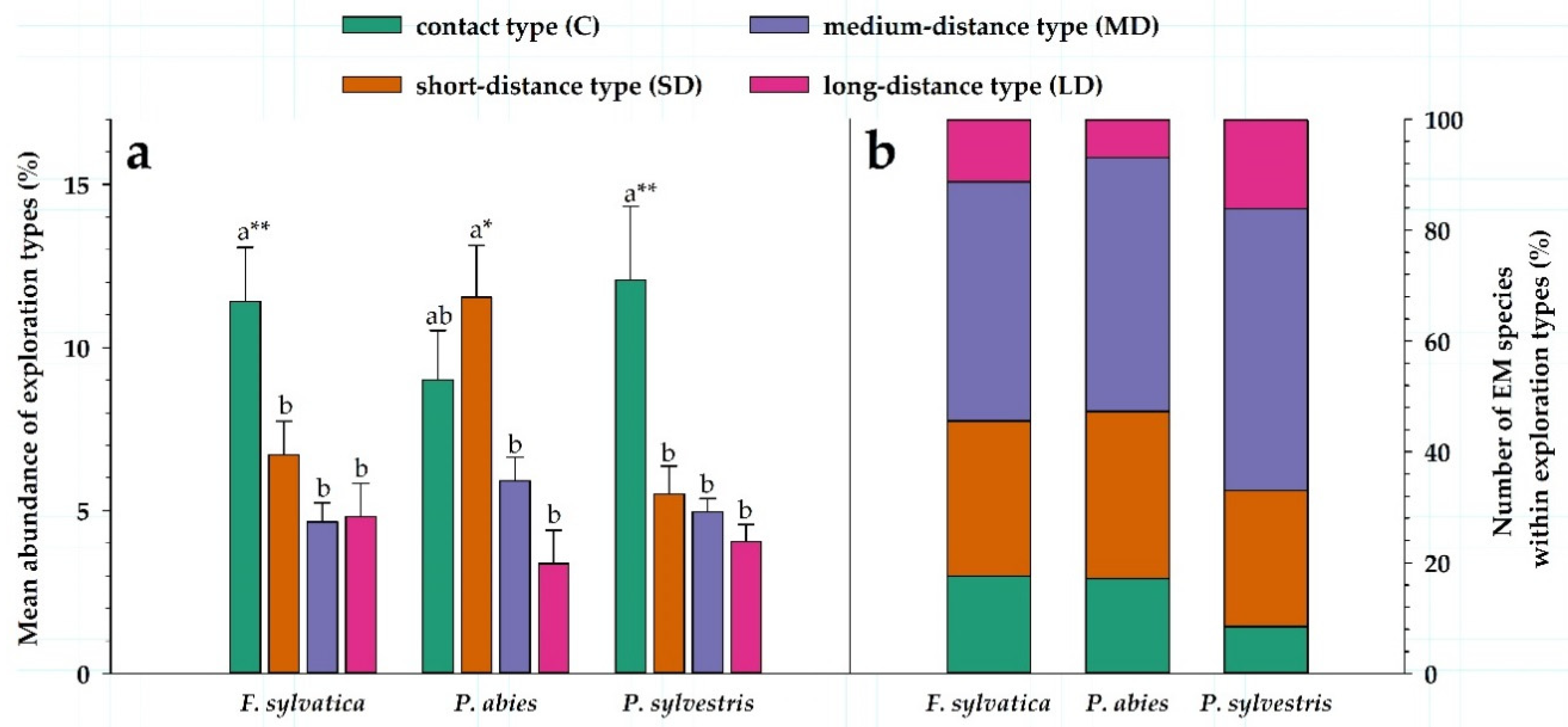
3.2. Patterns in Soil Exploration of EM Fungi
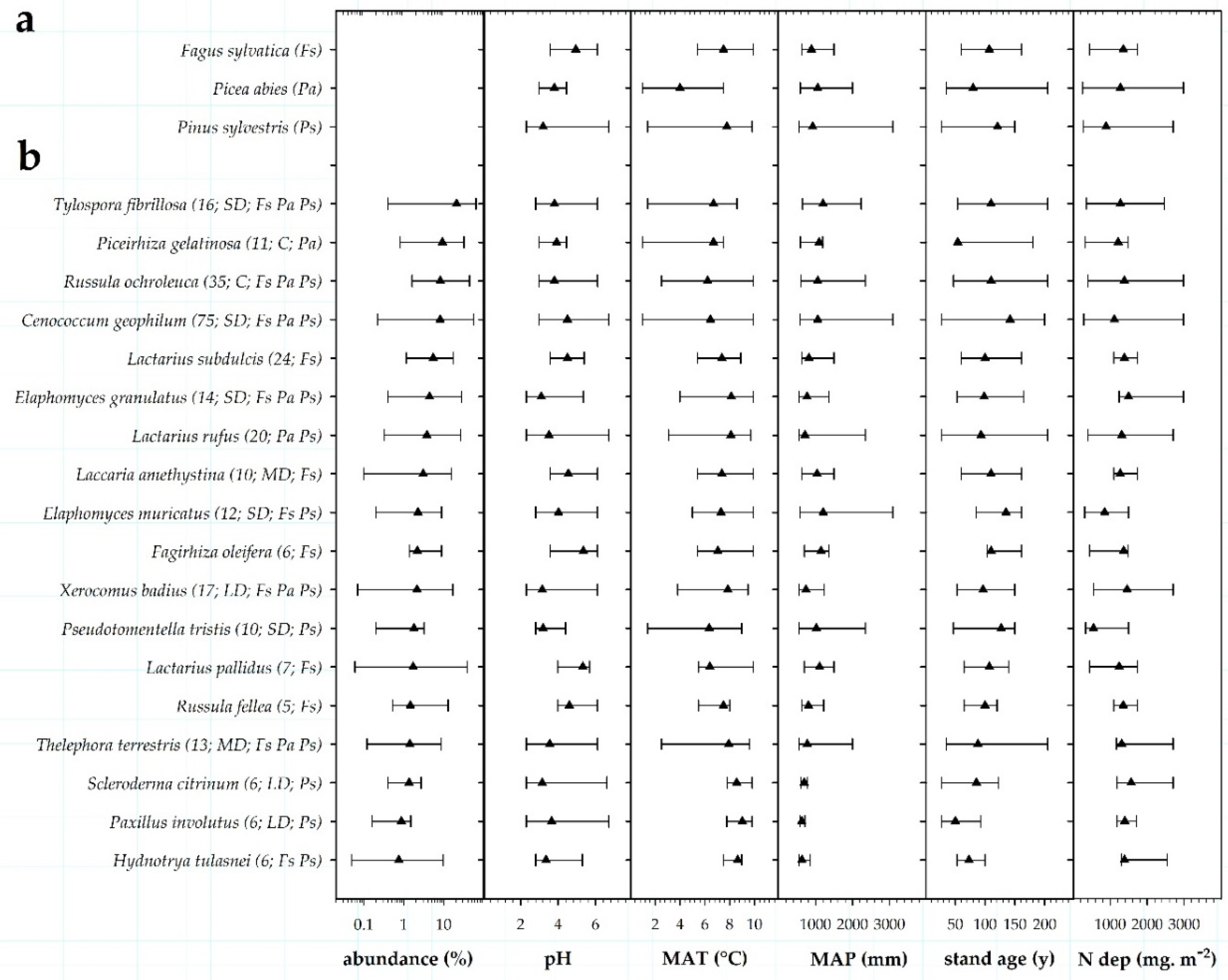
3.3. Environmental Drivers of EM Communities and Exploration Types of EM Fungi
3.4. Environmental Ranges of EM Fungal Taxa
3.5. EM Taxa Richness and Evenness
4. Discussion
4.1. Environmental Drivers of EM Communities and Exploration Types
4.2. Patterns in Frequency and Abundance of EM Taxa and Exploration Types
4.3. Limitations of the Study
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Finlay, R.D. Ecological aspects of mycorrhizal symbiosis: With special emphasis on the functional diversity of interactions involving the extraradical mycelium. J. Exp. Bot. 2008, 59, 1115–1126. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.; Read, D. Mycorrhizal Symbiosis, 3rd ed.; Academic Press: San Diego, CA, USA, 2008. [Google Scholar]
- Simard, S.W.; Jones, M.D.; Durall, D.M. Carbon and nutrient fluxes within and between mycorrhizal plants. In Mycorrhizal Ecology; Springer: Berlin, Germany, 2003; pp. 33–74. [Google Scholar]
- Van der Heijden, M.G.; Bardgett, R.D.; Van Straalen, N.M. The unseen majority: Soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol. Lett. 2008, 11, 296–310. [Google Scholar] [CrossRef] [PubMed]
- Hobbie, J.E.; Hobbie, E.A. 15N in symbiotic fungi and plants estimates nitrogen and carbon flux rates in arctic tundra. Ecology 2006, 87, 816–822. [Google Scholar] [CrossRef]
- Högberg, M.N.; Briones, M.J.; Keel, S.G.; Metcalfe, D.B.; Campbell, C.; Midwood, A.J.; Thornton, B.; Hurry, V.; Linder, S.; Näsholm, T. Quantification of effects of season and nitrogen supp. ly on tree below-ground carbon transfer to ectomycorrhizal fungi and other soil organisms in a boreal pine forest. New Phytol. 2010, 187, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Clemmensen, K.; Bahr, A.; Ovaskainen, O.; Dahlberg, A.; Ekblad, A.; Wallander, H.; Stenlid, J.; Finlay, R.; Wardle, D.; Lindahl, B. Roots and associated fungi drive long-term carbon sequestration in boreal forest. Science 2013, 339, 1615–1618. [Google Scholar] [CrossRef] [PubMed]
- Heinemeyer, A.; Hartley, I.P.; Evans, S.P.; Carreira de la Fuente, J.A.; Ineson, P. Forest soil CO2 flux: Uncovering the contribution and environmental responses of ectomycorrhizas. Glob. Chang. Biol. 2007, 13, 1786–1797. [Google Scholar] [CrossRef]
- Hobbie, E.A. Carbon allocation to ectomycorrhizal fungi correlates with belowground allocation in culture studies. Ecology 2006, 87, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, A.; Comandini, O.; Kuyper, T.W. Ectomycorrhizal fungal diversity: Seperating the wheat from the chaff. Fungal Divers. 2008, 33, 1–45. [Google Scholar]
- Tedersoo, L.; Bahram, M.; Toots, M.; Diedhiou, A.G.; Henkel, T.W.; Kjøller, R.; Morris, M.H.; Nara, K.; Nouhra, E.; Peay, K.G. Towards global patterns in the diversity and community structure of ectomycorrhizal fungi. Mol. Ecol. 2012, 21, 4160–4170. [Google Scholar] [CrossRef] [PubMed]
- Tedersoo, L.; Bahram, M.; Põlme, S.; Kõljalg, U.; Yorou, N.S.; Wijesundera, R.; Ruiz, L.V.; Vasco-Palacios, A.M.; Thu, P.Q.; Suija, A. Global diversity and geography of soil fungi. Science 2014, 346, 1256688. [Google Scholar] [CrossRef] [PubMed]
- Tedersoo, L.; Nara, K. General latitudinal gradient of biodiversity is reversed in ectomycorrhizal fungi. New Phytol. 2010, 185, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Kjøller, R. Disproportionate abundance between ectomycorrhizal root tips and their associated mycelia. FEMS Microbiol. Ecol. 2006, 58, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Pena, R.; Offermann, C.; Simon, J.; Naumann, P.S.; Geßler, A.; Holst, J.; Dannenmann, M.; Mayer, H.; Kögel-Knabner, I.; Rennenberg, H.; et al. Girdling affects ectomycorrhizal fungal (EMF) diversity and reveals functional differences in emf community composition in a beech forest. App. Lied Environ. Microbiol. 2010, 76, 1831–1841. [Google Scholar] [CrossRef] [PubMed]
- Bruns, T.D. Thoughts on the processes that maintain local species diversity of ectomycorrhizal fungi. In The Significance and Regulation of Soil Biodiversity; Springer: Berlin, Germany, 1995; pp. 63–73. [Google Scholar]
- Courty, P.-E.; Munoz, F.; Selosse, M.A.; Duchemin, M.; Criquet, S.; Ziarelle, F.; Buée, M.; Plassard, C.; Taudière, A.; Garbaye, J. Into the functional ecology of ectomycorrhizal communities: Environmental filtering of enzymatic activities. J. Ecol. 2016, 104, 1585–1598. [Google Scholar]
- Courty, P.E.; Pritsch, K.; Schloter, M.; Hartmann, A.; Garbaye, J. Activity profiling of ectomycorrhiza communities in two forest soils using multiple enzymatic tests. New Phytol. 2005, 167, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Courty, P.-E.; Pouysegur, R.; Buée, M.; Garbaye, J. Laccase and phosphatase activities of the dominant ectomycorrhizal types in a lowland oak forest. Soil Biol. Biochem. 2006, 38, 1219–1222. [Google Scholar] [CrossRef]
- Gorissen, A.; Kuyper, T.W. Fungal species-specific responses of ectomycorrhizal Scots pine (Pinus sylvestris) to elevated CO2. New Phytol. 2000, 146, 163–168. [Google Scholar] [CrossRef]
- Agerer, R.; Rambold, G. Deemy—An Information System for Characterization and Determination of Ectomycorrhizae. Available online: www.deemy.de (accessed on 15 November 2016).
- Agerer, R. Exploration types of ectomycorrhizae. Mycorrhiza 2001, 11, 107–114. [Google Scholar] [CrossRef]
- Pena, R.; Lang, C.; Lohaus, G.; Boch, S.; Schall, P.; Schöning, I.; Ammer, C.; Fischer, M.; Polle, A. Phylogenetic and functional traits of ectomycorrhizal assemblages in top soil from different biogeographic regions and forest types. Mycorrhiza 2016, 27, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Tedersoo, L.; Smith, M.E. Lineages of ectomycorrhizal fungi revisited: Foraging strategies and novel lineages revealed by sequences from belowground. Fungal Biol. Rev. 2013, 27, 83–99. [Google Scholar] [CrossRef]
- Fernandez, C.W.; Langley, J.A.; Chapman, S.; McCormack, M.L.; Koide, R.T. The decomposition of ectomycorrhizal fungal necromass. Soil Biol. Biochem. 2016, 93, 38–49. [Google Scholar] [CrossRef]
- Hobbie, E.A.; Agerer, R. Nitrogen isotopes in ectomycorrhizal sporocarps correspond to belowground exploration types. Plant Soil 2010, 327, 71–83. [Google Scholar] [CrossRef]
- Tedersoo, L.; Naadel, T.; Bahram, M.; Pritsch, K.; Buegger, F.; Leal, M.; Kõljalg, U.; Põldmaa, K. Enzymatic activities and stable isotope patterns of ectomycorrhizal fungi in relation to phylogeny and exploration types in an afrotropical rain forest. New Phytol. 2012, 195, 832–843. [Google Scholar] [CrossRef] [PubMed]
- Koide, R.T.; Fernandez, C.; Malcolm, G. Determining place and process: Functional traits of ectomycorrhizal fungi that affect both community structure and ecosystem function. New Phytol. 2014, 201, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Morgado, L.N.; Semenova, T.A.; Welker, J.M.; Walker, M.D.; Smets, E.; Geml, J. Summer temperature increase has distinct effects on the ectomycorrhizal fungal communities of moist tussock and dry tundra in arctic Alaska. Glob. Chang. Biol. 2015, 21, 959–972. [Google Scholar] [CrossRef] [PubMed]
- Deslipp, E.J.; Hartmann, M.; Mohn, W.; Simard, S. Long-term experimental manipulation of climate alters the ectomycorrhizal community of Betula nana in arctic tundra. Glob. Chang. Biol. 2011, 17, 1625–1636. [Google Scholar] [CrossRef]
- Nickel, U.T.; Weikl, F.; Kerner, R.; Schäfer, C.; Kallenbach, C.; Munch, J.C.; Pritsch, K. Quantitative losses vs. Qualitative stability of ectomycorrhizal community responses to 3 years of experimental summer drought in a beech–spruce forest. Glob. Chang. Biol. 2017, e560–e576. [Google Scholar] [CrossRef] [PubMed]
- Gehring, C.A.; Theimer, T.C.; Whitham, T.G.; Keim, P. Ectomycorrhizal fungal community structure of pinyon pines growing in two environmental extremes. Ecology 1998, 79, 1562–1572. [Google Scholar] [CrossRef]
- Gordon, G.J.; Gehring, C.A. Molecular characterization of pezizalean ectomycorrhizas associated with pinyon pine during drought. Mycorrhiza 2011, 21, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Lilleskov, E.A.; Fahey, T.J.; Horton, T.R.; Lovett, G.M. Belowground ectomycorrhizal fungal community change over a nitrogen deposition gradient in Alaska. Ecology 2002, 83, 104–115. [Google Scholar] [CrossRef]
- Geml, J.; Morgado, L.N.; Semenova, T.A.; Welker, J.M.; Walker, M.D.; Smets, E. Long-term warming alters richness and composition of taxonomic and functional groups of arctic fungi. FEMS Microbiol. Ecol. 2015, 91, fiv095. [Google Scholar] [CrossRef] [PubMed]
- Treseder, K.K.; Marusenko, Y.; Romero-Olivares, A.L.; Maltz, M.R. Experimental warming alters potential function of the fungal community in boreal forest. Glob. Chang. Biol. 2016, 22, 3395–3404. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, Y.; Sakai, A.; Hattori, M.; Nara, K. Strong effect of climate on ectomycorrhizal fungal composition: Evidence from range overlap between two mountains. ISME J. 2015, 9, 1870–1879. [Google Scholar] [CrossRef] [PubMed]
- Bahram, M.; Põlme, S.; Kõljalg, U.; Zarre, S.; Tedersoo, L. Regional and local patterns of ectomycorrhizal fungal diversity and community structure along an altitudinal gradient in the hyrcanian forests of northern Iran. New Phytol. 2012, 193, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Buée, M.; Courty, P.; Mignot, D.; Garbaye, J. Soil niche effect on species diversity and catabolic activities in an ectomycorrhizal fungal community. Soil Biol. Biochem. 2007, 39, 1947–1955. [Google Scholar] [CrossRef]
- Dickie, I.A.; Xu, B.; Koide, R.T. Vertical niche differentiation of ectomycorrhizal hyphae in soil as shown by T-RFLP analysis. New Phytol. 2002, 156, 527–535. [Google Scholar] [CrossRef]
- Suz, L.M.; Barsoum, N.; Benham, S.; Dietrich, H.P.; Fetzer, K.D.; Fischer, R.; García, P.; Gehrman, J.; Kristöfel, F.; Manninger, M. Environmental drivers of ectomycorrhizal communities in Europe’s temperate oak forests. Mol. Ecol. 2014, 23, 5628–5644. [Google Scholar] [CrossRef] [PubMed]
- Põlme, S.; Bahram, M.; Yamanaka, T.; Nara, K.; Dai, Y.C.; Grebenc, T.; Kraigher, H.; Toivonen, M.; Wang, P.H.; Matsuda, Y. Biogeography of ectomycorrhizal fungi associated with alders (Alnus spp.) in relation to biotic and abiotic variables at the global scale. New Phytol. 2013, 198, 1239–1249. [Google Scholar] [CrossRef] [PubMed]
- Köble, R.; Seufert, G. Novel Maps for Forest Tree Species in Europe. In Proceedings of the 8th European Symposium on the Physico-Chemical Behaviour of air Pollutants: A Changing Atmosphere, Torino, Italy, 17–20 September 2001; pp. 17–20. [Google Scholar]
- Read, D.J.; Leake, J.R.; Perez-Moreno, J. Mycorrhizal fungi as drivers of ecosystem processes in heathland and boreal forest biomes. Can. J. Bot. 2004, 82, 1243–1263. [Google Scholar] [CrossRef]
- Horton, T.; Bruns, T. Multiple-host fungi are the most frequent and abundant ectomycorrhizal types in a mixed stand of douglas fir (Pseudotsuga menziesii) and bishop pine (Pinus muricata). New Phytol. 1998, 139, 331–339. [Google Scholar] [CrossRef]
- Sakakibara, S.M.; Jones, M.D.; Gillespie, M.; Hagerman, S.M.; Forrest, M.E.; Simard, S.W.; Durall, D.M. A comparison of ectomycorrhiza identification based on morphotyping and PCR-RFLP analysis. Mycol. Res. 2002, 106, 868–878. [Google Scholar] [CrossRef]
- Schöpp, W.; Posch, M.; Mylona, S.; Johansson, M. Long-term development of acid deposition (1880–2030) in sensitive freshwater regions in europe. Hydrol. Earth Syst. Sci. Discuss. 2003, 7, 436–446. [Google Scholar] [CrossRef]
- Rambold, G.; Agerer, R. Deemy–the concept of a characterization and determination system for ectomycorrhizae. Mycorrhiza 1997, 7, 113–116. [Google Scholar] [CrossRef]
- Wallander, H.; Ekblad, A.; Godbold, D.; Johnson, D.; Bahr, A.; Baldrian, P.; Björk, R.; Kieliszewska-Rokicka, B.; Kjøller, R.; Kraigher, H. Evaluation of methods to estimate production, biomass and turnover of ectomycorrhizal mycelium in forests soils—A review. Soil Biol. Biochem. 2013, 57, 1034–1047. [Google Scholar] [CrossRef]
- Paliy, O.; Shankar, V. Application of multivariate statistical techniques in microbial ecology. Mol. Ecol. 2016, 25, 1032–1057. [Google Scholar] [CrossRef] [PubMed]
- R Core Team, R. A language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minichin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Wagner, H. Vegan: Community Ecology Package. Available online: http://CRAN.R-project.org/package=vegan (accessed on 3 April 2017).
- Simpson, E.H. Measurement of diversity. Nature 1949, 163, 688. [Google Scholar] [CrossRef]
- Clemmensen, K.E.; Michelsen, A.; Jonasson, S.; Shaver, G.R. Increased ectomycorrhizal fungal abundance after long-term fertilization and warming of two arctic tundra ecosystems. New Phytol. 2006, 171, 391–404. [Google Scholar] [CrossRef] [PubMed]
- Penton, C.R.; St Louis, D.; Cole, J.R.; Luo, Y.; Wu, L.; Schuur, E.G.; Zhou, J.; Tiedje, J.M. Fungal diversity in permafrost and tallgrass prairie soils under experimental warming conditions. App. Lied Environ. Microbiol. 2013, 79, 7063–7072. [Google Scholar] [CrossRef] [PubMed]
- Lauber, C.L.; Hamady, M.; Knight, R.; Fierer, N. Soil ph as a predictor of soil bacterial community structure at the continental scale: A pyrosequencing-based assessment. App. Lied Environ. Microbiol. 2009, 75, 5111–5120. [Google Scholar] [CrossRef] [PubMed]
- Fierer, N.; Leff, J.W.; Adams, B.J.; Nielsen, U.N.; Bates, S.T.; Lauber, C.L.; Owens, S.; Gilbert, J.A.; Wall, D.H.; Caporaso, J.G. Cross-biome metagenomic analyses of soil microbial communities and their functional attributes. Proc. Natl. Acad. Sci. USA 2012, 109, 21390–21395. [Google Scholar] [CrossRef] [PubMed]
- Tedersoo, L.; Suvi, T.; Jairus, T.; Ostonen, I.; Põlme, S. Revisiting ectomycorrhizal fungi of the genus Alnus: Differential host specificity, diversity and determinants of the fungal community. New Phytol. 2009, 182, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Erland, S.; Söderström, B. Effects of lime and ash treatments on ectomycorrhizal infection of Pinus sylvestris L. seedlings planted in a pine forest. Scand. J. For. Res. 1991, 6, 519–525. [Google Scholar] [CrossRef]
- Deacon, J.; Fleming, L. Interactions of ectomycorrhizal fungi. In Mycorrhizal Functioning: An Integrative Plant-Fungal Process; Springer: Berlin, Germany, 1992; Volume 1249. [Google Scholar]
- Nilsson, L.O.; Bååth, E.; Falkengren-Grerup, U.; Wallander, H. Growth of ectomycorrhizal mycelia and composition of soil microbial communities in oak forest soils along a nitrogen deposition gradient. Oecologia 2007, 153, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Lilleskov, E.; Fahey, T.; Lovett, G. Ectomycorrhizal fungal aboveground community change over an atmospheric nitrogen deposition gradient. Ecol. Appl. 2001, 11, 397–410. [Google Scholar] [CrossRef]
- Kjøller, R.; Nilsson, L.O.; Hansen, K.; Schmidt, I.K.; Vesterdal, L.; Gundersen, P. Dramatic changes in ectomycorrhizal community composition, root tip abundance and mycelial production along a stand-scale nitrogen deposition gradient. New Phytol. 2012, 194, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Arnolds, E. Decline of ectomycorrhizal fungi in europe. Agric. Ecosyst. Environ. 1991, 35, 209–244. [Google Scholar] [CrossRef]
- Richard, F.; Roy, M.; Shahin, O.; Sthultz, C.; Duchemin, M.; Joffre, R.; Selosse, M.-A. Ectomycorrhizal communities in a mediterranean forest ecosystem dominated by Quercus ilex: Seasonal dynamics and response to drought in the surface organic horizon. Ann. For. Sci. 2011, 68, 57–68. [Google Scholar] [CrossRef]
- Swaty, R.L.; Deckert, R.J.; Whitham, T.G.; Gehring, C.A. Ectomycorrhizal abundance and community composition shifts with drought: Predictions from tree rings. Ecology 2004, 85, 1072–1084. [Google Scholar] [CrossRef]
- Cavender-Bares, J.; Izzo, A.; Robinson, R.; Lovelock, C. Changes in ectomycorrhizal community structure on two containerized oak hosts across an experimental hydrologic gradient. Mycorrhiza 2009, 19, 133–142. [Google Scholar] [CrossRef] [PubMed]
- LoBuglio, K.F.; Berbee, M.L.; Taylor, J.W. Phylogenetic origins of the asexual mycorrhizal symbiont Cenococcum geophilum Fr. and other mycorrhizal fungi among the ascomycetes. Mol. Phylogenet. Evol. 1996, 6, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Trapp, E.J.M. Cenococcum Graniforme—Its Distribution, Ecology, Mycorrhiza Formation, And Inherent Variation. Ph.D. Thesis, University of Washington, Seattle, WA, USA, 1962. [Google Scholar]
- Haselwandter, K.; Read, D. Fungal associations of roots of dominant and sub-dominant plants in high-alpine vegetation systems with special reference to mycorrhiza. Oecologia 1980, 45, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Koide, R.; Suomi, L.; Stevens, C.; McCormick, L. Interactions between needles of pinus resinosa and ectomycorrhizal fungi. New Phytol. 1998, 140, 539–547. [Google Scholar] [CrossRef]
- Fernandez, C.W.; Nguyen, N.H.; Stefanski, A.; Han, Y.; Hobbie, S.E.; Montgomery, R.A.; Reich, P.B.; Kennedy, P.G. Ectomycorrhizal fungal response to warming is linked to poor host performance at the boreal-temperate ecotone. Glob. Chang. Biol. 2016, 23, 1598–1609. [Google Scholar] [CrossRef] [PubMed]
- Ekblad, A.; Wallander, H.; Godbold, D.; Cruz, C.; Johnson, D.; Baldrian, P.; Björk, R.; Epron, D.; Kieliszewska-Rokicka, B.; Kjøller, R. The production and turnover of extramatrical mycelium of ectomycorrhizal fungi in forest soils: Role in carbon cycling. Plant Soil 2013, 366, 1–27. [Google Scholar] [CrossRef]
- Fransson, P. Elevated CO2 impacts ectomycorrhiza-mediated forest soil carbon flow: Fungal biomass production, respiration and exudation. Fungal Ecol. 2012, 5, 85–98. [Google Scholar] [CrossRef]
- Alberton, O.; Kuyper, T.W. Ectomycorrhizal fungi associated with pinus sylvestris seedlings respond differently to increased carbon and nitrogen availability: Implications for ecosystem responses to global change. Glob. Chang. Biol. 2009, 15, 166–175. [Google Scholar] [CrossRef]
- Lamhamedi, M.S.; Bernier, P.Y.; André-Fortin, J. Hydraulic conductance and soil water potential at the soil–root interface of pinus pinaster seedlings inoculated with different dikaryons of pisolithus sp. Tree Physiol. 1992, 10, 231–244. [Google Scholar] [CrossRef] [PubMed]
- Lilleskov, E.; Hobbie, E.; Horton, T. Conservation of ectomycorrhizal fungi: Exploring the linkages between functional and taxonomic responses to anthropogenic n deposition. Fungal Ecol. 2011, 4, 174–183. [Google Scholar] [CrossRef]
- Cox, F.; Barsoum, N.; Lilleskov, E.A.; Bidartondo, M.I. Nitrogen availability is a primary determinant of conifer mycorrhizas across complex environmental gradients. Ecol. Lett. 2010, 13, 1103–1113. [Google Scholar] [CrossRef] [PubMed]
- Van Breemen, N.; Driscoll, C.; Mulder, J. Acidic deposition and internal proton sources in acidification of soils and waters. Nature 1984, 307, 599. [Google Scholar] [CrossRef]
- Erland, S.; Taylor, A.F. Diversity of ecto-mycorrhizal fungal communities in relation to the abiotic environment. In Mycorrhizal Ecology; Springer: Berlin, Germany, 2002; pp. 163–200. [Google Scholar]
- Jonsson, L.M.; Nilsson, M.C.; Wardle, D.A.; Zackrisson, O. Context dependent effects of ectomycorrhizal species richness on tree seedling productivity. Oikos 2001, 93, 353–364. [Google Scholar] [CrossRef]
- Taylor, A.F. Fungal diversity in ectomycorrhizal communities: Sampling effort and species detection. In Diversity and Integration in Mycorrhizas; Springer: Berlin, Germany, 2002; pp. 19–28. [Google Scholar]
- Rosinger, C.; Sandén, H.; Godbold, D.L. Ectomycorrhizal diversity in beech dominated stands in Central Europe. In Soil Biological Communities and Ecosystem Resilience; Springer: Berlin, Germany, 2017; pp. 143–156. [Google Scholar]
- Lindahl, B.D.; Nilsson, R.H.; Tedersoo, L.; Abarenkov, K.; Carlsen, T.; Kjøller, R.; Kõljalg, U.; Pennanen, T.; Rosendahl, S.; Stenlid, J. Fungal community analysis by high-throughput sequencing of amplified markers—A user’s guide. New Phytol. 2013, 199, 288–299. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.H.; Smith, D.; Peay, K.; Kennedy, P. Parsing ecological signal from noise in next generation amplicon sequencing. New Phytol. 2015, 205, 1389–1393. [Google Scholar] [CrossRef] [PubMed]
- Buée, M.; Vairelles, D.; Garbaye, J. Year-round monitoring of diversity and potential metabolic activity of the ectomycorrhizal community in a beech (Fagus silvatica) forest subjected to two thinning regimes. Mycorrhiza 2005, 15, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Yachi, S.; Loreau, M. Biodiversity and ecosystem productivity in a fluctuating environment: The insurance hypothesis. Proc. Natl. Acad. Sci. USA 1999, 96, 1463–1468. [Google Scholar] [CrossRef] [PubMed]
- Sterk, M.; Gort, G.; Klimkowska, A.; Van Ruijven, J.; Van Teeffelen, A.; Wamelink, G. Assess ecosystem resilience: Linking response and effect traits to environmental variability. Ecol. Indic. 2013, 30, 21–27. [Google Scholar] [CrossRef]
- Pritsch, K.; Garbaye, J. Enzyme secretion by ECM fungi and exploitation of mineral nutrients from soil organic matter. Ann. For. Sci. 2011, 68, 25–32. [Google Scholar] [CrossRef]
- Phillips, L.A.; Ward, V.; Jones, M.D. Ectomycorrhizal fungi contribute to soil organic matter cycling in sub-boreal forests. ISME J. 2014, 8, 699–713. [Google Scholar] [CrossRef] [PubMed]
- Clemmensen, K.E.; Finlay, R.D.; Dahlberg, A.; Stenlid, J.; Wardle, D.A.; Lindahl, B.D. Carbon sequestration is related to mycorrhizal fungal community shifts during long-term succession in boreal forests. New Phytol. 2015, 205, 1525–1536. [Google Scholar] [CrossRef] [PubMed]
- Bahram, M.; Peay, K.G.; Tedersoo, L. Local-scale biogeography and spatiotemporal variability in communities of mycorrhizal fungi. New Phytol. 2015, 205, 1454–1463. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosinger, C.; Sandén, H.; Matthews, B.; Mayer, M.; Godbold, D.L. Patterns in Ectomycorrhizal Diversity, Community Composition, and Exploration Types in European Beech, Pine, and Spruce Forests. Forests 2018, 9, 445. https://doi.org/10.3390/f9080445
Rosinger C, Sandén H, Matthews B, Mayer M, Godbold DL. Patterns in Ectomycorrhizal Diversity, Community Composition, and Exploration Types in European Beech, Pine, and Spruce Forests. Forests. 2018; 9(8):445. https://doi.org/10.3390/f9080445
Chicago/Turabian StyleRosinger, Christoph, Hans Sandén, Bradley Matthews, Mathias Mayer, and Douglas L. Godbold. 2018. "Patterns in Ectomycorrhizal Diversity, Community Composition, and Exploration Types in European Beech, Pine, and Spruce Forests" Forests 9, no. 8: 445. https://doi.org/10.3390/f9080445
APA StyleRosinger, C., Sandén, H., Matthews, B., Mayer, M., & Godbold, D. L. (2018). Patterns in Ectomycorrhizal Diversity, Community Composition, and Exploration Types in European Beech, Pine, and Spruce Forests. Forests, 9(8), 445. https://doi.org/10.3390/f9080445





