Native and Invasive Woody Species Differentially Respond to Forest Edges and Forest Successional Age
Abstract
1. Introduction
2. Materials and Methods
2.1. Species Description
2.2. Site Selection and Habitat Characterization
2.3. Experimental Design
2.4. Data Analysis
3. Results
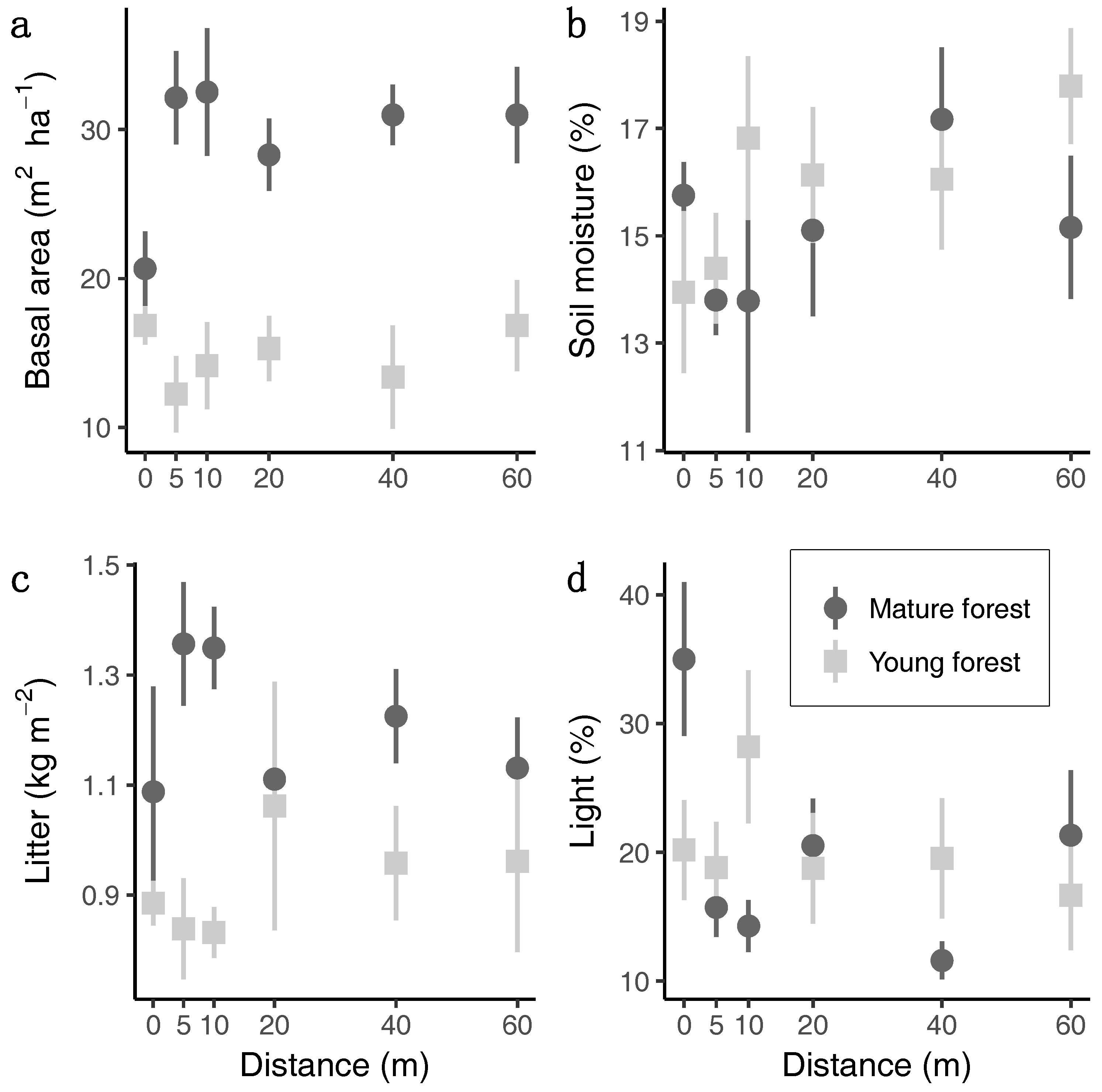
3.1. Habitat Conditions
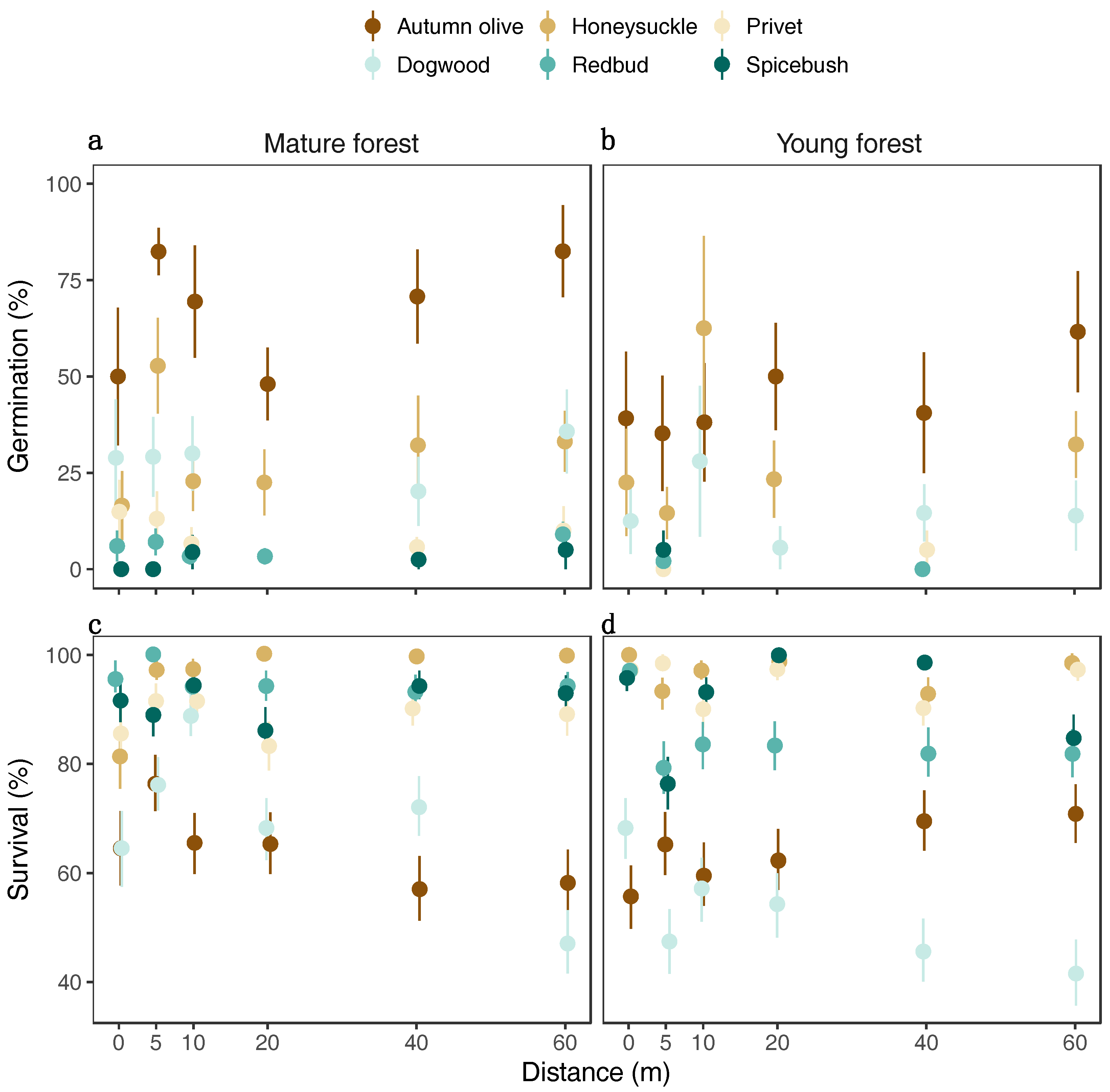
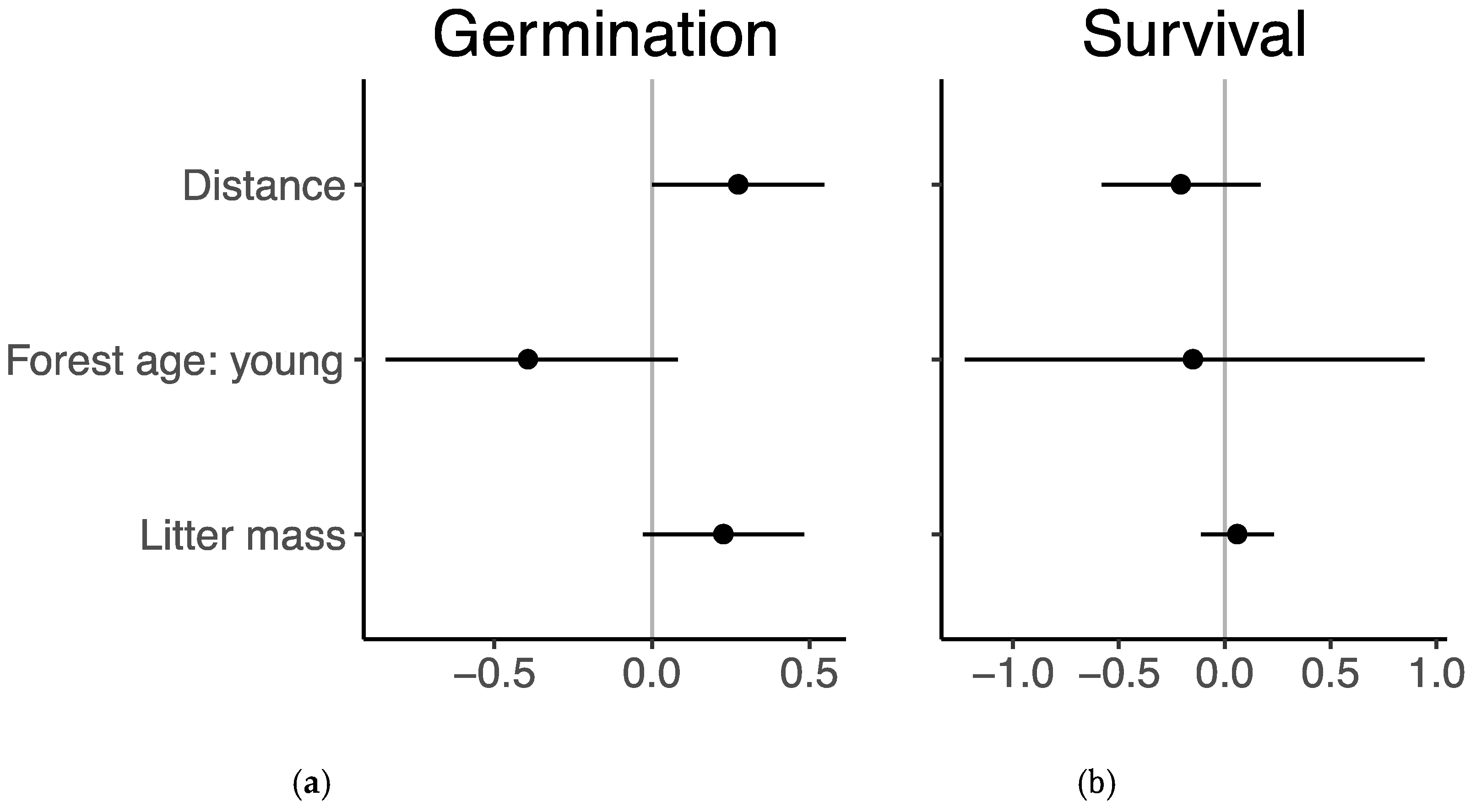
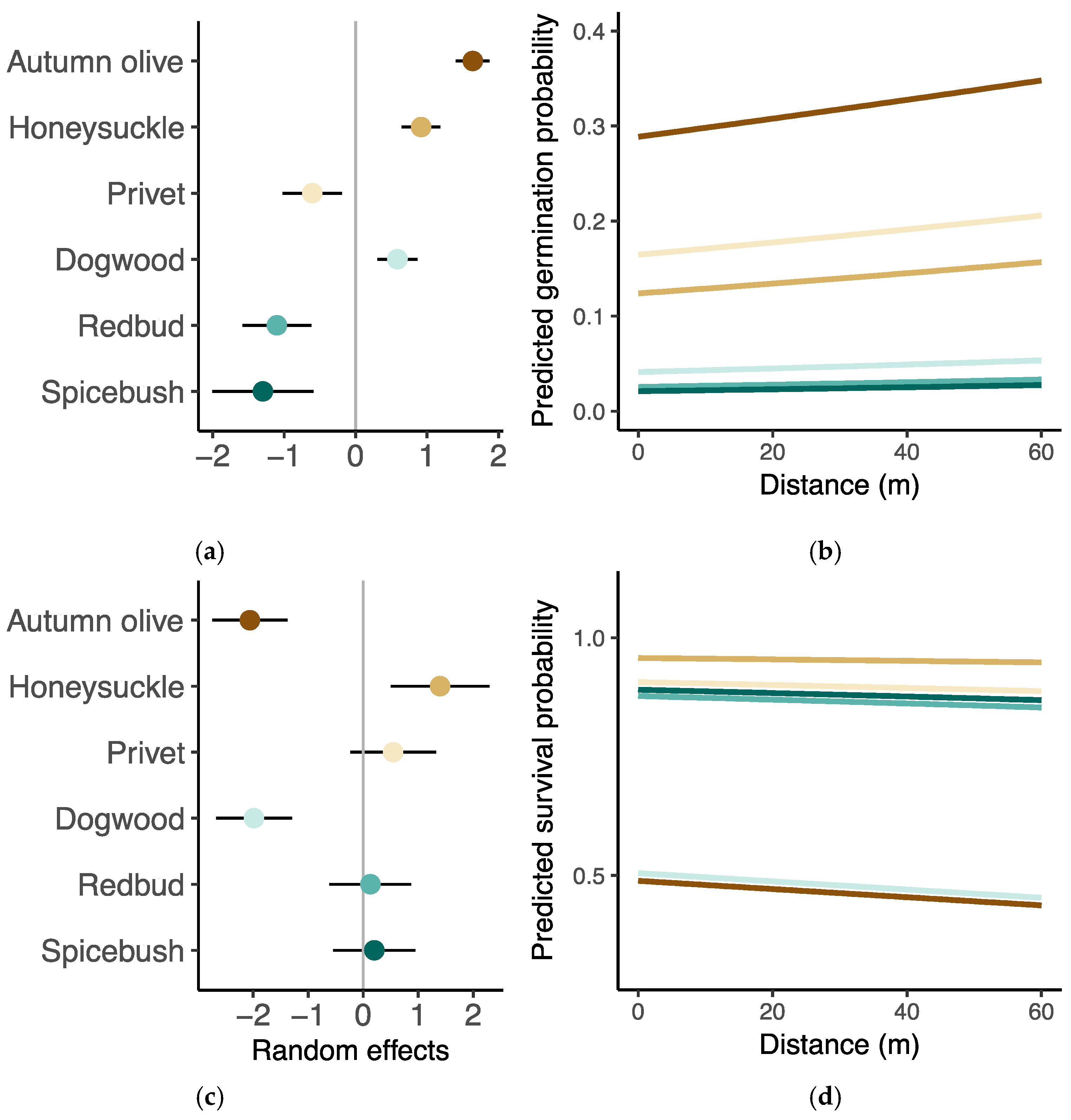
3.2. Germination and Survival
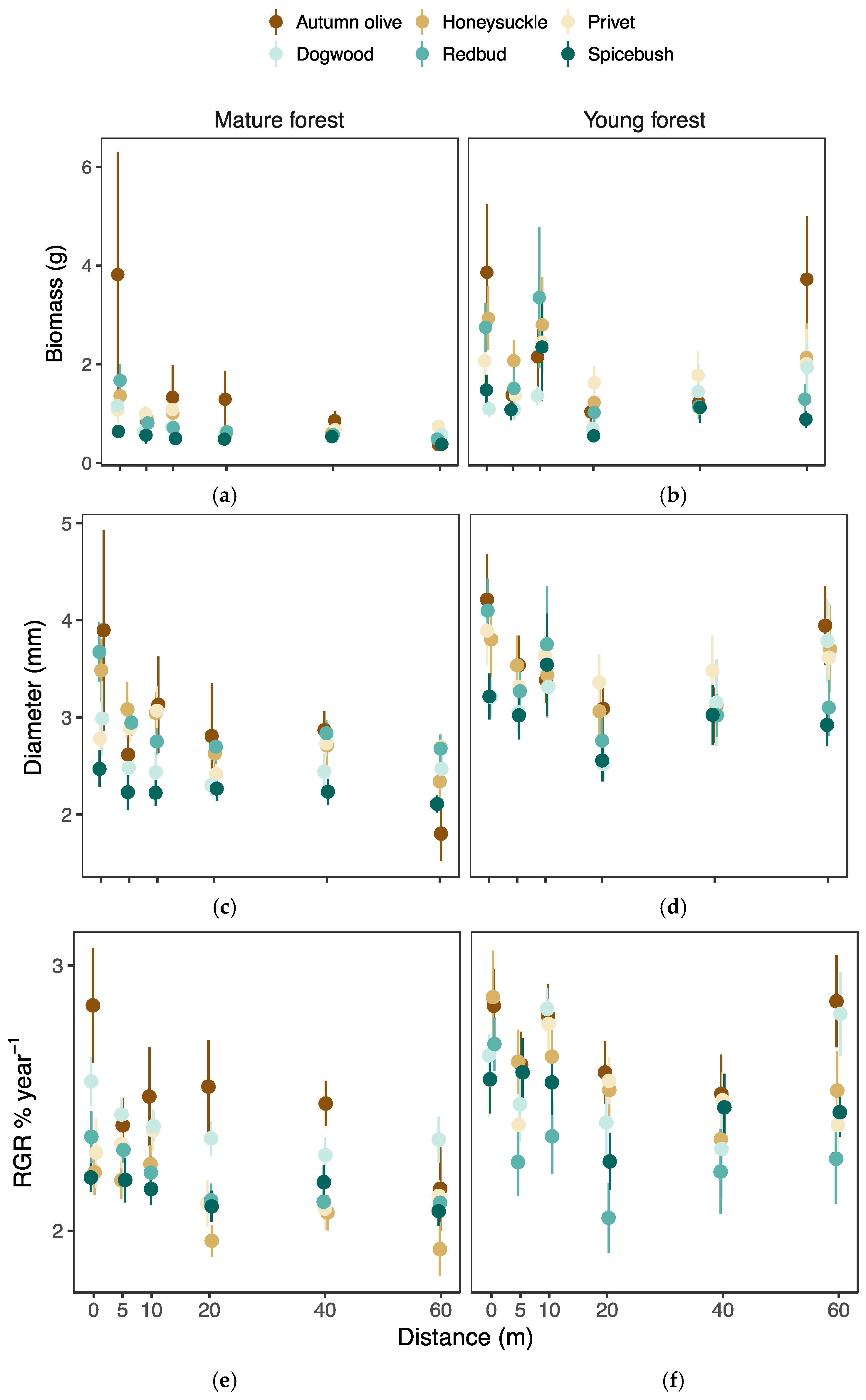
3.3. Seedling Performance
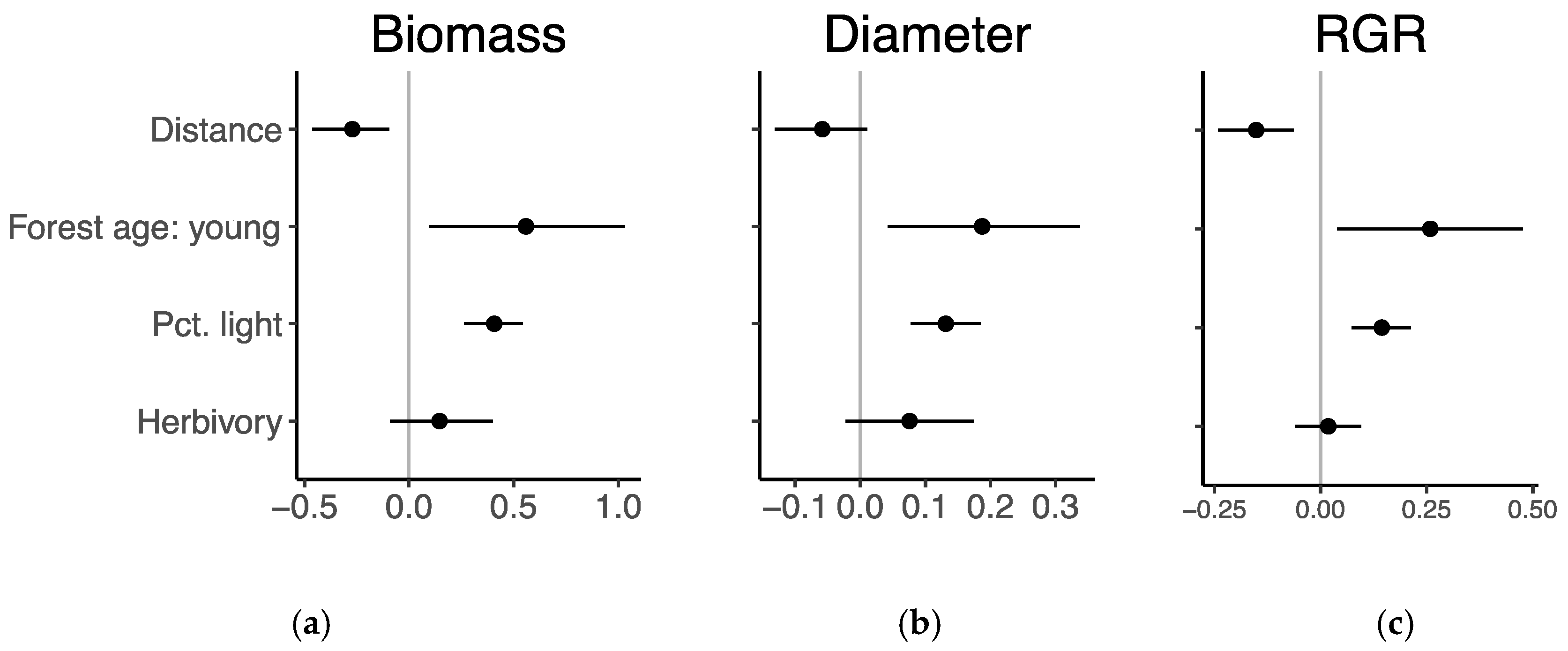
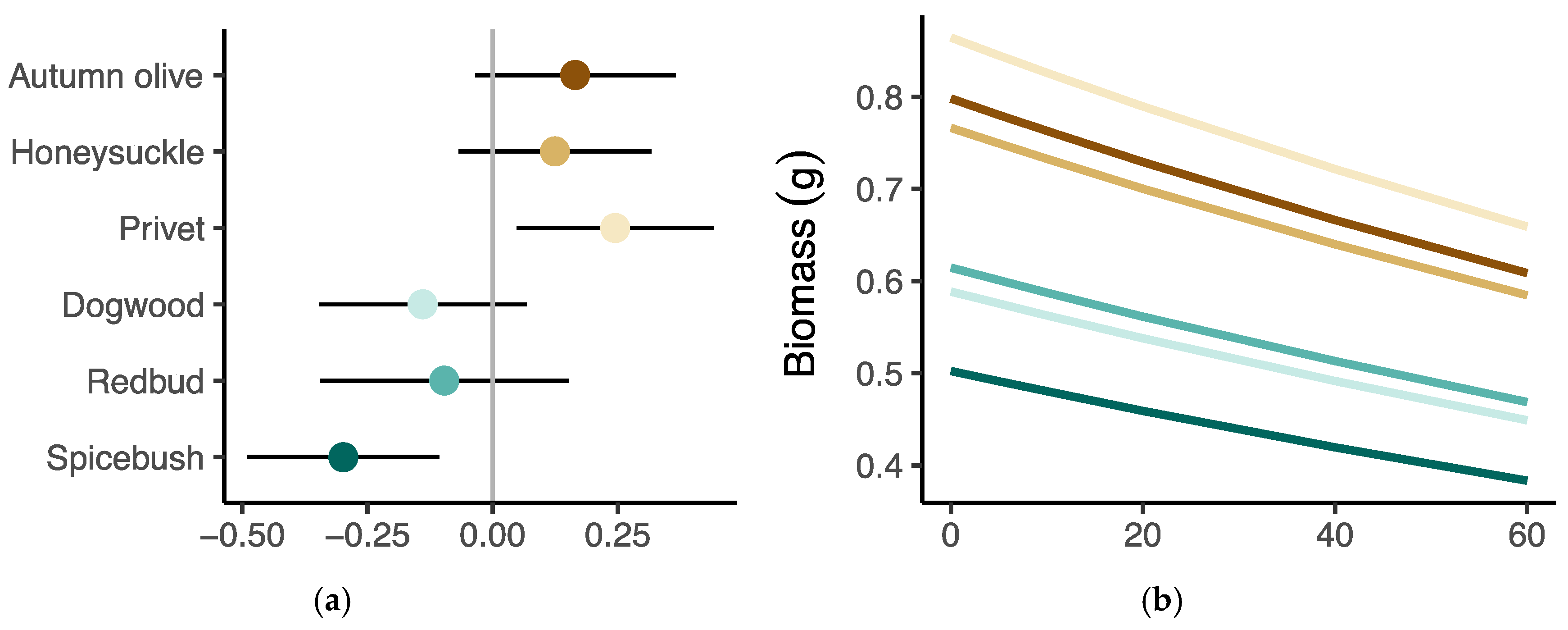
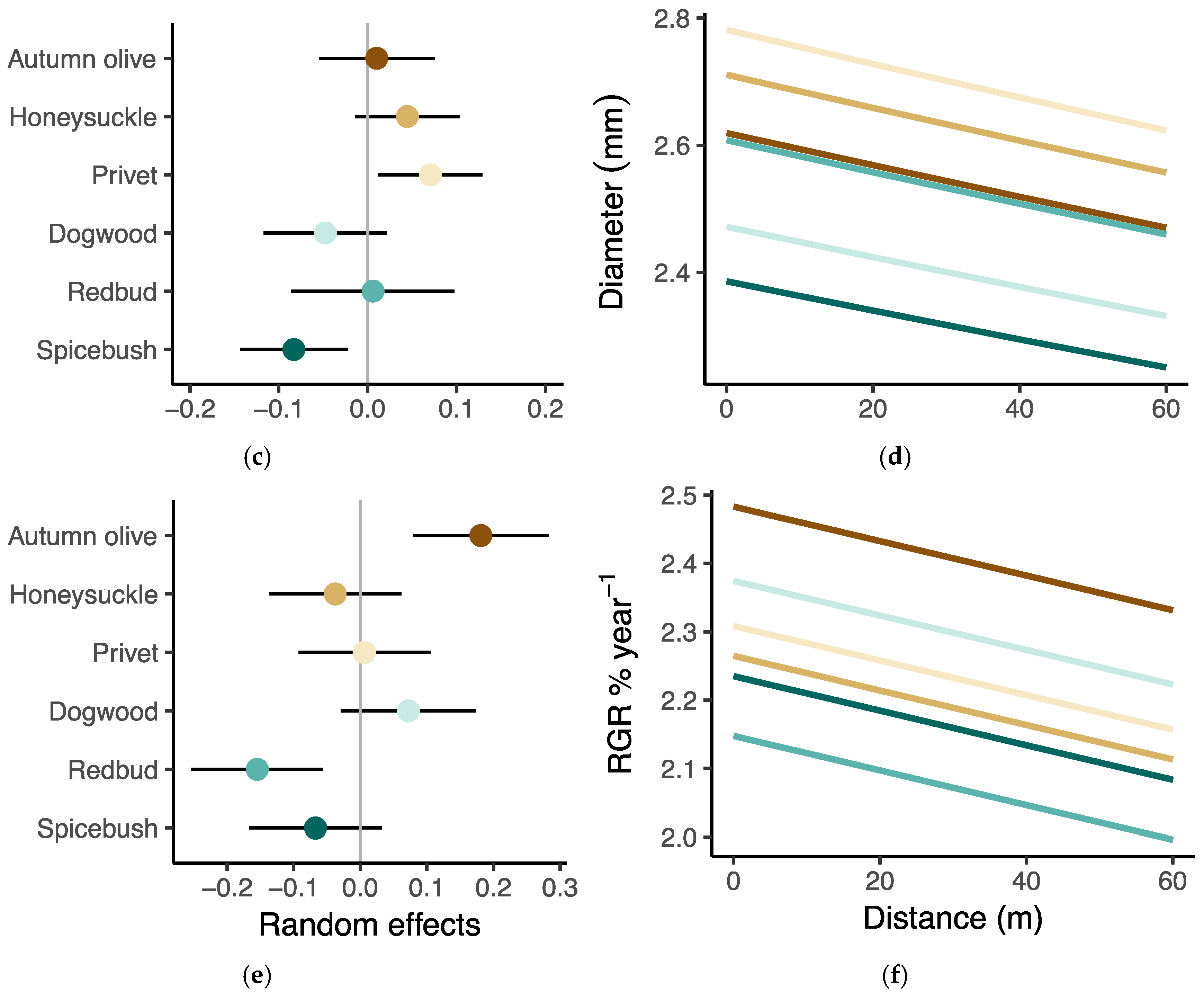
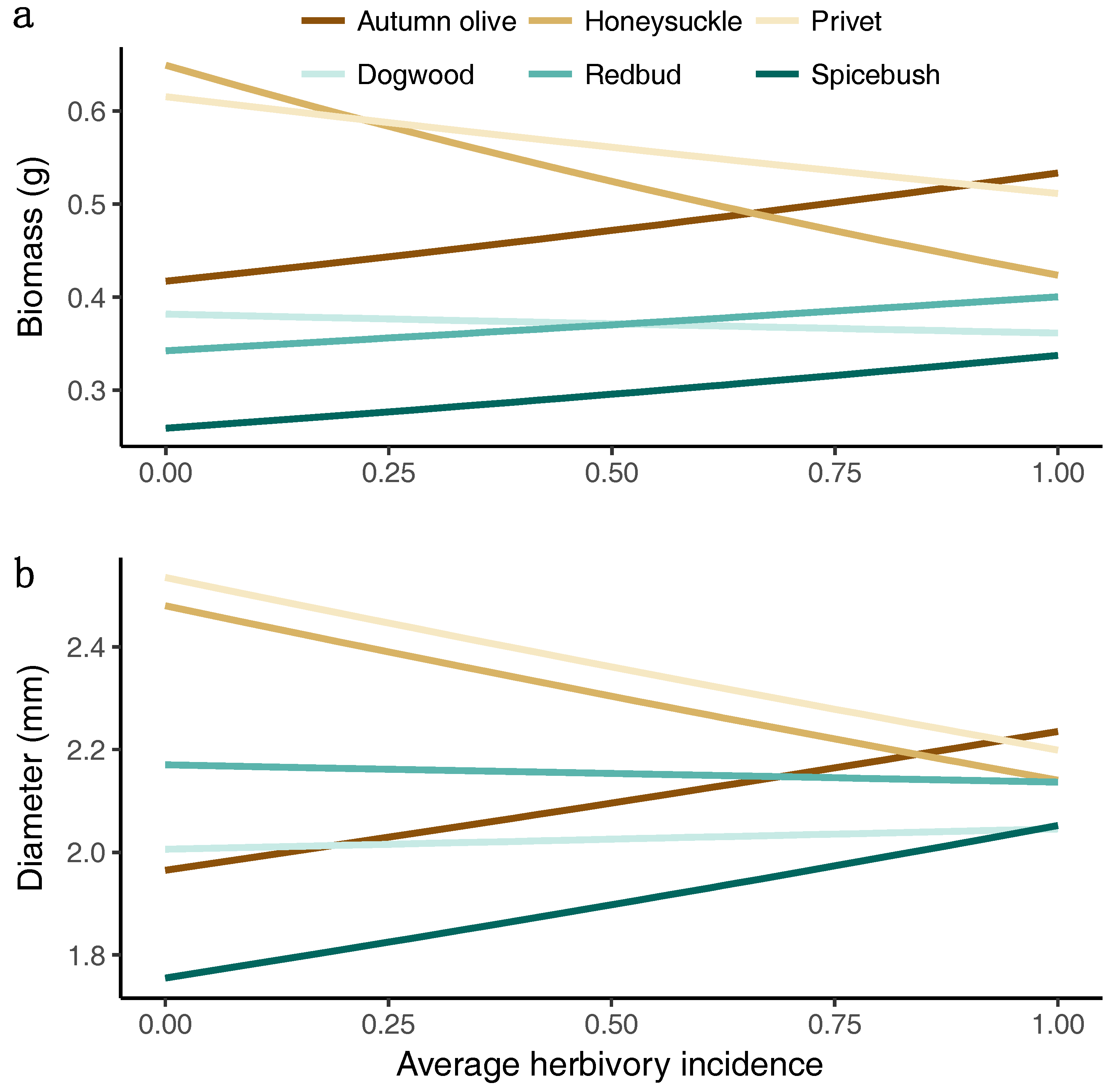
3.4. Growth Response to Herbivory
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Species | Status | Mature Forest | Young Forest | Average |
|---|---|---|---|---|
| Autumn olive | Invasive | 67.19 | 44.07 | 55.63 |
| Honeysuckle | Invasive | 29.99 | 25.34 | 27.66 |
| Privet | Invasive | 8.21 | 0.97 | 4.59 |
| Dogwood | Native | 23.85 | 12.34 | 18.09 |
| Redbud | Native | 4.77 | 0.96 | 2.86 |
| Spicebush | Native | 2.18 | 10.00 | 6.09 |
| Species | Status | Mature Forest | Young Forest | Average |
|---|---|---|---|---|
| Autumn olive | Invasive | 48.53 | 50.69 | 49.61 |
| Honeysuckle | Invasive | 95.56 | 95.14 | 95.35 |
| Privet | Invasive | 85.29 | 93.06 | 89.17 |
| Dogwood | Native | 61.03 | 41.67 | 51.35 |
| Redbud | Native | 91.91 | 80.56 | 86.23 |
| Spicebush | Native | 86.76 | 88.19 | 87.48 |
| Fixed Parts | Germination | Survival | ||
|---|---|---|---|---|
| Odds Ratio | CI | Odds Ratio | CI | |
| (Intercept) | 0.11 | 0.04–0.30 | 5.92 | 1.74–20.16 |
| Distance | 1.31 | 1.00–1.73 | 0.82 | 0.57–1.20 |
| Forest age (Young) | 0.67 | 0.44–1.03 | 0.87 | 0.32–2.37 |
| Litter mass | 1.25 | 0.97–1.62 | 1.08 | 0.91–1.29 |
| Light availability | 1.08 | 0.92–1.28 | ||
| Random Parts | ||||
| Variance, site_code | 0.087 | 0.710 | ||
| Variance, common_name | 1.269 | 1.514 | ||
| Fixed Parts | Biomass | Diameter | RGR | |||
|---|---|---|---|---|---|---|
| B | CI | B | CI | B | CI | |
| (Intercept) | −0.39 | −0.77–−0.01 | 0.95 | 0.83–1.07 | 2.30 | 2.12–2.49 |
| Distance | −0.27 | −0.45–−0.09 | −0.06 | −0.13–0.01 | −0.15 | −0.24–−0.06 |
| Light availability | 0.41 | 0.27–0.55 | 0.13 | 0.08–0.19 | 0.14 | 0.08–0.21 |
| Forest age (Young) | 0.56 | 0.10–1.02 | 0.19 | 0.04–0.33 | 0.26 | 0.04–0.47 |
| Herbivory incidence | 0.14 | −0.08–0.37 | 0.08 | −0.02–0.17 | 0.02 | −0.06–0.10 |
| Random Parts | ||||||
| σ2 | 0.391 | 0.059 | 0.098 | |||
| Variance, site | 0.155 | 0.015 | 0.034 | |||
| Variance, species | 0.051 | 0.004 | 0.015 | |||
References
- Davis, M.A.; Grime, J.P.; Thompson, K. Fluctuating resources in plant communities: A general theory of invasibility. J. Ecol. 2000, 88, 528–534. [Google Scholar] [CrossRef]
- Haddad, N.M.; Brudvig, L.A.; Clobert, J.; Davies, K.F.; Gonzalez, A.; Holt, R.D.; Lovejoy, T.E.; Sexton, J.O.; Austin, M.P.; Collins, C.D.; et al. Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci. Adv. 2015, 1, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yates, E.D.; Levia, D.F.; Williams, C.L. Recruitment of three non-native invasive plants into a fragmented forest in southern Illinois. For. Ecol. Manag. 2004, 190, 119–130. [Google Scholar] [CrossRef]
- Alston, K.P.; Richardson, D.M. The roles of habitat features, disturbance, and distance from putative source populations in structuring alien plant invasions at the urban/wildland interface on the Cape Peninsula, South Africa. Biol. Conserv. 2006, 132, 183–198. [Google Scholar] [CrossRef]
- Moser, W.K.; Fan, Z.; Hansen, M.H.; Crosby, M.K.; Fan, S.X. Invasibility of three major non-native invasive shrubs and associated factors in Upper Midwest U.S. forest lands. For. Ecol. Manag. 2016, 379, 195–205. [Google Scholar] [CrossRef]
- Brothers, T.S.; Spingarn, A. Forest Fragmentation and Alien Plant Invasion of Central Indiana Old-Growth Forests. Conserv. Biol. 1992, 6, 91–100. [Google Scholar] [CrossRef]
- Hansen, M.J.; Clevenger, A.P. The influence of disturbance and habitat on the presence of non-native plant species along transport corridors. Biol. Conserv. 2005, 125, 249–259. [Google Scholar] [CrossRef]
- Von Der Lippe, M.; Kowarik, I. Long-Distance Dispersal of Plants by Vehicles as a Driver of Plant Invasions. Conserv. Biol. 2007, 21, 986–996. [Google Scholar] [CrossRef] [PubMed]
- Rew, L.J.; Brummer, T.J.; Pollnac, F.W.; Larson, C.D.; Taylor, K.T.; Taper, M.L.; Fleming, J.D.; Balbach, H.E. Hitching a ride: Seed accrual rates on different types of vehicles. J. Environ. Manag. 2018, 206, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Simberloff, D. The Role of Propagule Pressure in Biological Invasions. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 81–102. [Google Scholar] [CrossRef]
- Damschen, E.I.; Baker, D.V.; Bohrer, G.; Nathan, R.; Orrock, J.L.; Turner, J.R.; Brudvig, L.A.; Haddad, N.M.; Levey, D.J.; Tewksbury, J.J. How fragmentation and corridors affect wind dynamics and seed dispersal in open habitats. Proc. Natl. Acad. Sci. USA 2014. [Google Scholar] [CrossRef] [PubMed]
- Murcia, C. Edge effects in fragmented forests: Implications for conservation. Trends Ecol. Evol. 1995, 10, 58–62. [Google Scholar] [CrossRef]
- Cadenasso, M.L.; Pickett, S.T.A. Effect of Edge Structure on the Flux of Species into Forest Interiors. Conserv. Biol. 2001, 15, 91–97. [Google Scholar] [CrossRef]
- Harper, K.A.; Macdonald, S.E.; Burton, P.J.; Chen, J.; Brosofske, K.D.; Saunders, S.C.; Euskirchen, E.S.; Roberts, D.; Jaiteh, M.S.; Esseen, P.A. Edge Influence on Forest Structure and Composition in Fragmented Landscapes. Conserv. Biol. 2005, 19, 768–782. [Google Scholar] [CrossRef]
- Wirth, R.; Meyer, S.T.; Leal, I.R.; Tabarelli, M. Plant Herbivore Interactions at the Forest Edge. In Progress in Botany; Lüttge, U., Beyschlag, W., Murata, J., Eds.; Springer: Berlin, Germany, 2008; Volume 69, pp. 423–448. ISBN 978-3-540-72953-2. [Google Scholar]
- De Carvalho Guimarães, C.D.; Viana, J.P.R.; Cornelissen, T. A Meta-Analysis of the Effects of Fragmentation on Herbivorous Insects. Environ. Entomol. 2014, 43, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Matlack, G.R. Microenvironment variation within and among forest edge sites in the eastern United States. Biol. Conserv. 1993, 66, 185–194. [Google Scholar] [CrossRef]
- Funk, J.L.; McDaniel, S. Altering Light Availability to Restore Invaded Forest: The Predictive Role of Plant Traits. Restor. Ecol. 2010, 18, 865–872. [Google Scholar] [CrossRef]
- Flory, S.L.; Clay, K. Invasive shrub distribution varies with distance to roads and stand age in eastern deciduous forests in Indiana, USA. Plant Ecol. 2006, 184, 131–141. [Google Scholar] [CrossRef]
- Levine, J.M.; Adler, P.B.; Yelenik, S.G. A meta-analysis of biotic resistance to exotic plant invasions. Ecol. Lett. 2004, 7, 975–989. [Google Scholar] [CrossRef]
- Rejmanek, M.; Richardson, D.M. What Attributes Make Some Plant Species More Invasive? Ecology 1996, 77, 1655–1661. [Google Scholar] [CrossRef]
- Fridley, J.D. Extended leaf phenology and the autumn niche in deciduous forest invasions. Nature 2012, 485, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Funk, J.L. The physiology of invasive plants in low-resource environments. Conserv. Physiol. 2013, 1. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, K.A.; Dukes, J.S. Plant invasion across space and time: Factors affecting nonindigenous species success during four stages of invasion. New Phytol. 2007, 176, 256–273. [Google Scholar] [CrossRef] [PubMed]
- Keane, R.M.; Crawley, M.J. Exotic plant invasions and the enemy release hypothesis. Trends Ecol. Evol. 2002, 17, 164–170. [Google Scholar] [CrossRef]
- Mitchell, C.E.; Power, A.G. Release of invasive plants from fungal and viral pathogens. Nature 2003, 421, 625–627. [Google Scholar] [CrossRef] [PubMed]
- Rouget, M.; Richardson, D.M. Inferring Process from Pattern in Plant Invasions: A Semimechanistic Model Incorporating Propagule Pressure and Environmental Factors. Am. Nat. 2003, 162, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Dreiss, L.M.; Volin, J.C. Influence of leaf phenology and site nitrogen on invasive species establishment in temperate deciduous forest understories. For. Ecol. Manag. 2013, 296, 1–8. [Google Scholar] [CrossRef]
- Kaufman, S.R.; Kaufman, W. Invasive Plants: A Guide to Identification and the Impacts and Control of Common North American Species; Stackpole Books: Mechanicsburg, PA, USA, 2013. [Google Scholar]
- Flory, S.L.; Clay, K. Effects of roads and forest successional age on experimental plant invasions. Biol. Conserv. 2009, 142, 2531–2537. [Google Scholar] [CrossRef]
- Naumann, J.C.; Bissett, S.N.; Young, D.R.; Edwards, J.; Anderson, J.E. Diurnal patterns of photosynthesis, chlorophyll fluorescence, and PRI to evaluate water stress in the invasive species, Elaeagnus umbellata Thunb. Trees 2010, 24, 237–245. [Google Scholar] [CrossRef]
- Lieurance, D.; Landsbergen, K. The influence of light habitat on the physiology, biomass allocation, and fecundity of the invasive shrub Amur honeysuckle (Lonicera maackii, Caprifoliaceae). J. Torrey Bot. Soc. 2016, 143, 415–426. [Google Scholar] [CrossRef]
- Wang, H.H.; Grant, W.E. Determinants of Chinese and European Privet (Ligustrum sinense and Ligustrum vulgare) Invasion and Likelihood of Further Invasion in Southern U.S. Forestlands. Invasive Plant Sci. Manag. 2012, 5, 454–463. [Google Scholar] [CrossRef]
- Pisula, N.L.; Meiners, S.J. Relative allelopathic potential of invasive plant species in a young disturbed woodland. J. Torrey Bot. Soc. 2010, 137, 81–87. [Google Scholar] [CrossRef]
- Montti, L.; Ayup, M.M.; Aragón, R.; Qi, W.; Ruan, H.; Fernández, R.; Casertano, S.A.; Zou, X. Herbivory and the success of Ligustrum lucidum: Evidence from a comparison between native and novel ranges. Aust. J. Bot. 2016, 64, 181–192. [Google Scholar] [CrossRef]
- Dorning, M.; Cipollini, D. Leaf and Root Extracts of the Invasive Shrub, Lonicera maackii, Inhibit Seed Germination of Three Herbs with No Autotoxic Effects. Plant Ecol. 2006, 184, 287–296. [Google Scholar] [CrossRef]
- Lieurance, D.; Cipollini, D. Exotic Lonicera species both escape and resist specialist and generalist herbivores in the introduced range in North America. Biol. Invasions 2013, 15, 1713–1724. [Google Scholar] [CrossRef]
- Nesom, G.L. Taxonomic Overview of Ligustrum (Oleaceae) Naturalizaed in North America North of Mexico. Phytologia 2009, 91, 467–482. [Google Scholar]
- McEwan, R.W.; Birchfield, M.K.; Schoergendorfer, A.; Arthur, M.A. Leaf Phenology and Freeze Tolerance of the Invasive Shrub Amur Honeysuckle and Potential Native Competitors. J. Torrey Bot. Soc. 2009, 136, 212–220. [Google Scholar] [CrossRef]
- Jenkins, M.A.; White, P.S. Cornus florida L. Mortality and Understory Composition Changes in Western Great Smoky Mountains National Park. J. Torrey Bot. Soc. 2002, 129, 194. [Google Scholar] [CrossRef]
- Niesenbaum, R.A. The Effects of Light Environment on Herbivory and Growth in the Dioecious Shrub Lindera benzoin (Lauraceae). Am. Midl. Nat. 1992, 128, 270. [Google Scholar] [CrossRef]
- Luken, J.O.; Kuddes, L.M.; Tholemeier, T.C.; Haller, D.M. Comparative Responses of Lonicera maackii (Amur honeysuckle) and Lindera benzoin (Spicebush) to Increased Light. Am. Midl. Nat. 1997, 138, 331. [Google Scholar] [CrossRef]
- Holzmueller, E.; Jose, S.; Jenkins, M.; Camp, A.; Long, A. Dogwood Anthracnose in Eastern Hardwood Forests: What Is Known and What Can Be Done? J. For. 2006, 104, 21–26. [Google Scholar]
- Burns, R.M.; Honkala, B.H. Silvics of North America; United States Department of Agriculture: Washington, DC, USA, 1990; Volume 1–2.
- Lieth, H.; Radford, J.S. Phenology, Resource Management, and Synagraphic Computer Mapping. BioScience 1971, 21, 62–70. [Google Scholar] [CrossRef]
- Herrera, C.M. Microclimate and Individual Variation in Pollinators: Flowering Plants are More than Their Flowers. Ecology 1995, 76, 1516–1524. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 1–51. [Google Scholar] [CrossRef]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82, 1–26. [Google Scholar] [CrossRef]
- R: A Language and Environment for Statistical Computing. Available online: http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.470.5851&rep=rep1&type=pdf (accessed on 15 September 2017).
- Gelman, A. Scaling regression inputs by dividing by two standard deviations. Stat. Med. 2008, 27, 2865–2873. [Google Scholar] [CrossRef] [PubMed]
- Delgado, J.D.; Arroyo, N.L.; Arévalo, J.R.; Fernández-Palacios, J.M. Edge effects of roads on temperature, light, canopy cover, and canopy height in laurel and pine forests (Tenerife, Canary Islands). Landsc. Urban Plan. 2007, 81, 328–340. [Google Scholar] [CrossRef]
- Van Kleunen, M.; Weber, E.; Fischer, M. A meta-analysis of trait differences between invasive and non-invasive plant species. Ecol. Lett. 2010, 13, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Funk, J.L.; Vitousek, P.M. Resource-use efficiency and plant invasion in low-resource systems. Nature 2007, 446, 1079–1081. [Google Scholar] [CrossRef] [PubMed]
- Miller-Rushing Abraham, J.; Primack Richard, B. Global warming and flowering times in thoreau’s concord: A community perspective. Ecology 2008, 89, 332–341. [Google Scholar] [CrossRef]
- Brym, Z.T.; Lake, J.K.; Allen, D.; Ostling, A. Plant functional traits suggest novel ecological strategy for an invasive shrub in an understorey woody plant community: Plant functional traits and invasion. J. Appl. Ecol. 2011, 48, 1098–1106. [Google Scholar] [CrossRef]
- Dechoum, M.S.; Zenni, R.D.; Castellani, T.T.; Zalba, S.M.; Rejmánek, M. Invasions across secondary forest successional stages: Effects of local plant community, soil, litter, and herbivory on Hovenia dulcis seed germination and seedling establishment. Plant Ecol. 2015, 216, 823–833. [Google Scholar] [CrossRef]
- Jo, I.; Fridley, J.D.; Frank, D.A. Linking above- and belowground resource use strategies for native and invasive species of temperate deciduous forests. Biol. Invasions 2015, 17, 1545–1554. [Google Scholar] [CrossRef]
- Bartuszevige, A.M.; Gorchov, D.L.; Raab, L. The relative importance of landscape and community features in the invasion of an exotic shrub in a fragmented landscape. Ecography 2006, 29, 213–222. [Google Scholar] [CrossRef]
- Moore, M.R.; Buckley, D.S.; Klingeman, W.E.; Saxton, A.M. Distribution and growth of autumn olive in a managed forest landscape. For. Ecol. Manag. 2013, 310, 589–599. [Google Scholar] [CrossRef]
- Ward, R.W. Extent and Dispersal Rates of Chinese Privet (Ligustrum sinense) Invasion on the Upper Oconee River Floodplain, North Georgia. Southeast. Geogr. 2002, 42, 29–48. [Google Scholar] [CrossRef]
- Merriam, R.W.; Feil, E. The Potential Impact of an Introduced Shrub on Native Plant Diversity and Forest Regeneration. 5. Biol. Invasions 2002, 4, 369–373. [Google Scholar] [CrossRef]
- Riitters, K.; Potter, K.; Iannone, B.V.; Oswalt, C.; Fei, S.; Guo, Q. Landscape correlates of forest plant invasions: A high-resolution analysis across the eastern United States. Divers. Distrib. 2018, 24, 274–284. [Google Scholar] [CrossRef]
- Benitez-Malvido, J.; Lázaro, A.; Ferraz, I.D.K. Effect of Distance to Edge and Edge Interaction on Seedling Regeneration and Biotic Damage in Tropical Rain Forest Fragments: A Long-Term Experiment. J. Ecol. 2018. [Google Scholar] [CrossRef]
- Davidson, A.M.; Jennions, M.; Nicotra, A.B. Do invasive species show higher phenotypic plasticity than native species and, if so, is it adaptive? A meta-analysis. Ecol. Lett. 2011, 14, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Flory, S.L.; Bauer, J.; Phillips, R.P.; Clay, K. Effects of a non-native grass invasion decline over time. J. Ecol. 2017, 105, 1475–1484. [Google Scholar] [CrossRef]
- Luke Flory, S.; Clay, K. Pathogen accumulation and long-term dynamics of plant invasions. J. Ecol. 2013, 101, 607–613. [Google Scholar] [CrossRef]
- Stricker, K.B.; Harmon, P.F.; Goss, E.M.; Clay, K.; Luke Flory, S. Emergence and accumulation of novel pathogens suppress an invasive species. Ecol. Lett. 2016, 19, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Boyce, R.L.; Brossart, S.N.; Bryant, L.A.; Fehrenbach, L.A.; Hetzer, R.; Holt, J.E.; Parr, B.; Poynter, Z.; Schumacher, C.; Stonebraker, S.N.; et al. The beginning of the end? Extensive dieback of an open-grown Amur honeysuckle stand in northern Kentucky, USA. Biol. Invasions 2014, 16, 2017–2023. [Google Scholar] [CrossRef]
- Dudt, J.F.; Shure, D.J. The Influence of Light and Nutrients on Foliar Phenolics and Insect Herbivory. Ecology 1994, 75, 86–98. [Google Scholar] [CrossRef]
- Lieurance, D.; Cipollini, D. Environmental influences on growth and defence responses of the invasive shrub, Lonicera maackii, to simulated and real herbivory in the juvenile stage. Ann. Bot. 2013, 112, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Meiners, S.J.; Handel, S.N.; Pickett, S.T.A. Tree seedling establishment under insect herbivory: Edge effects and inter- annual variation. Plant Ecol. 2000, 151, 161–170. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dillon, W.W.; Lieurance, D.; Hiatt, D.T.; Clay, K.; Flory, S.L. Native and Invasive Woody Species Differentially Respond to Forest Edges and Forest Successional Age. Forests 2018, 9, 381. https://doi.org/10.3390/f9070381
Dillon WW, Lieurance D, Hiatt DT, Clay K, Flory SL. Native and Invasive Woody Species Differentially Respond to Forest Edges and Forest Successional Age. Forests. 2018; 9(7):381. https://doi.org/10.3390/f9070381
Chicago/Turabian StyleDillon, Whalen W., Deah Lieurance, Drew T. Hiatt, Keith Clay, and S. Luke Flory. 2018. "Native and Invasive Woody Species Differentially Respond to Forest Edges and Forest Successional Age" Forests 9, no. 7: 381. https://doi.org/10.3390/f9070381
APA StyleDillon, W. W., Lieurance, D., Hiatt, D. T., Clay, K., & Flory, S. L. (2018). Native and Invasive Woody Species Differentially Respond to Forest Edges and Forest Successional Age. Forests, 9(7), 381. https://doi.org/10.3390/f9070381





