Response of Black Ash Wetland Gaseous Soil Carbon Fluxes to a Simulated Emerald Ash Borer Infestation
Abstract
1. Introduction
2. Materials and Methods
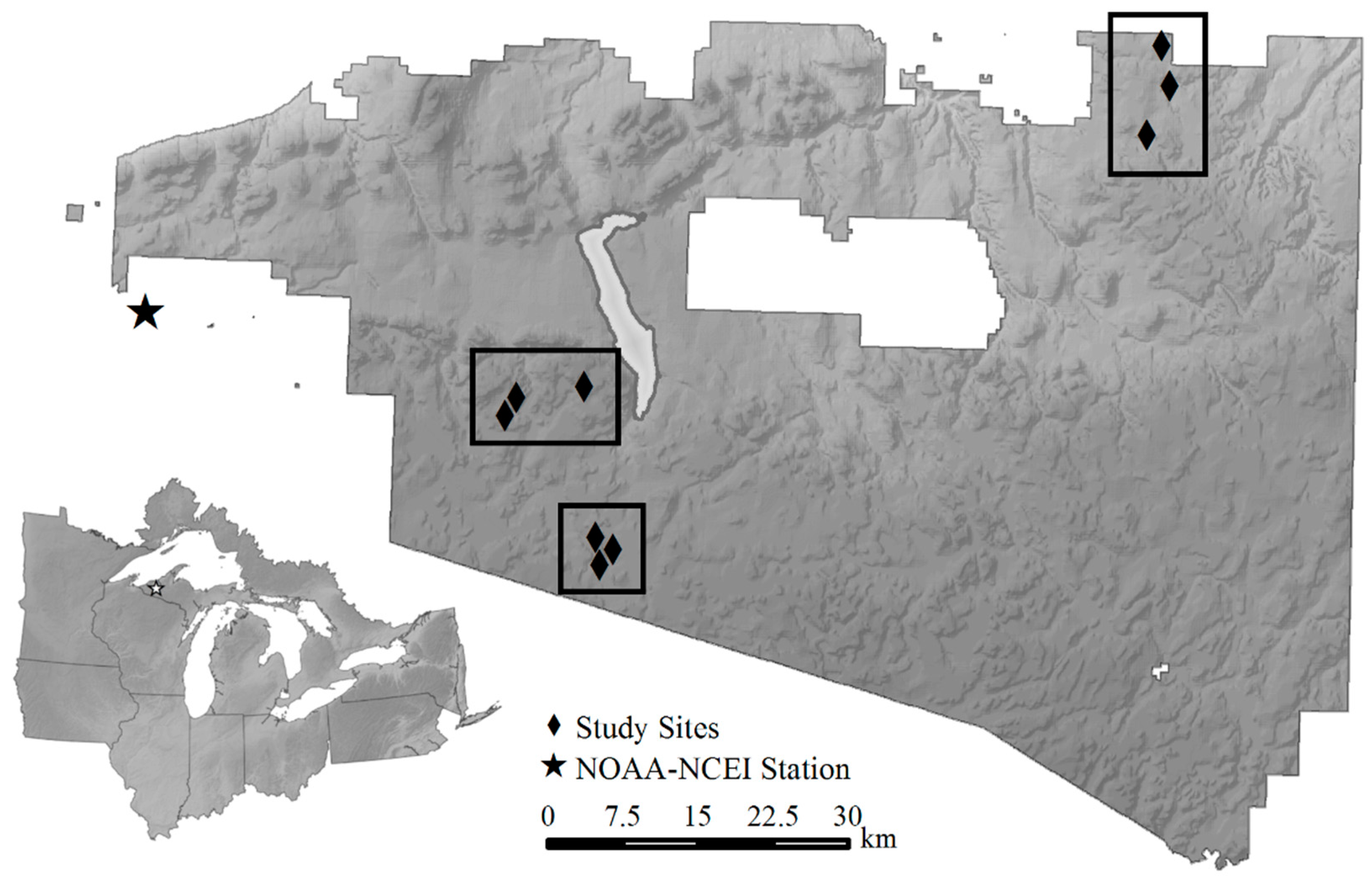
2.1. Study Area
2.2. Experimental Study Design
2.3. Instrumentation and Monitoring
2.4. Statistical Analysis
3. Results
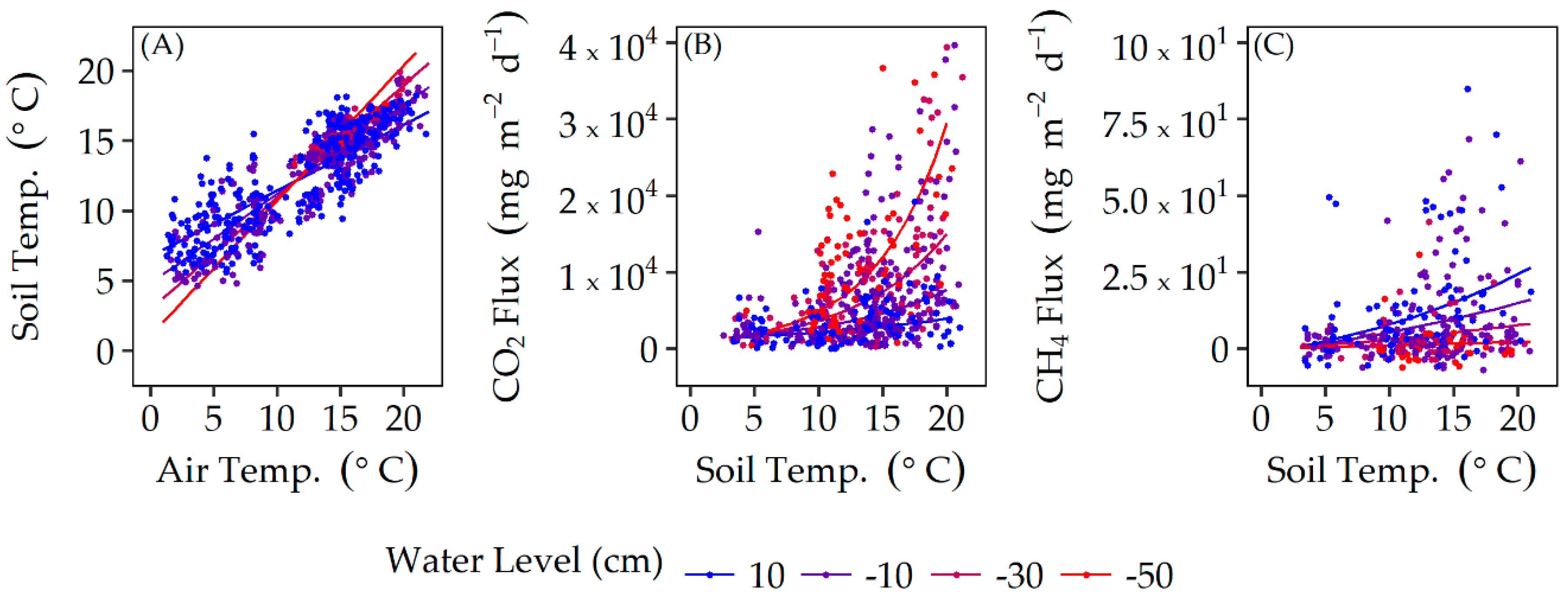
3.1. Regional Climate and Soil Temperature
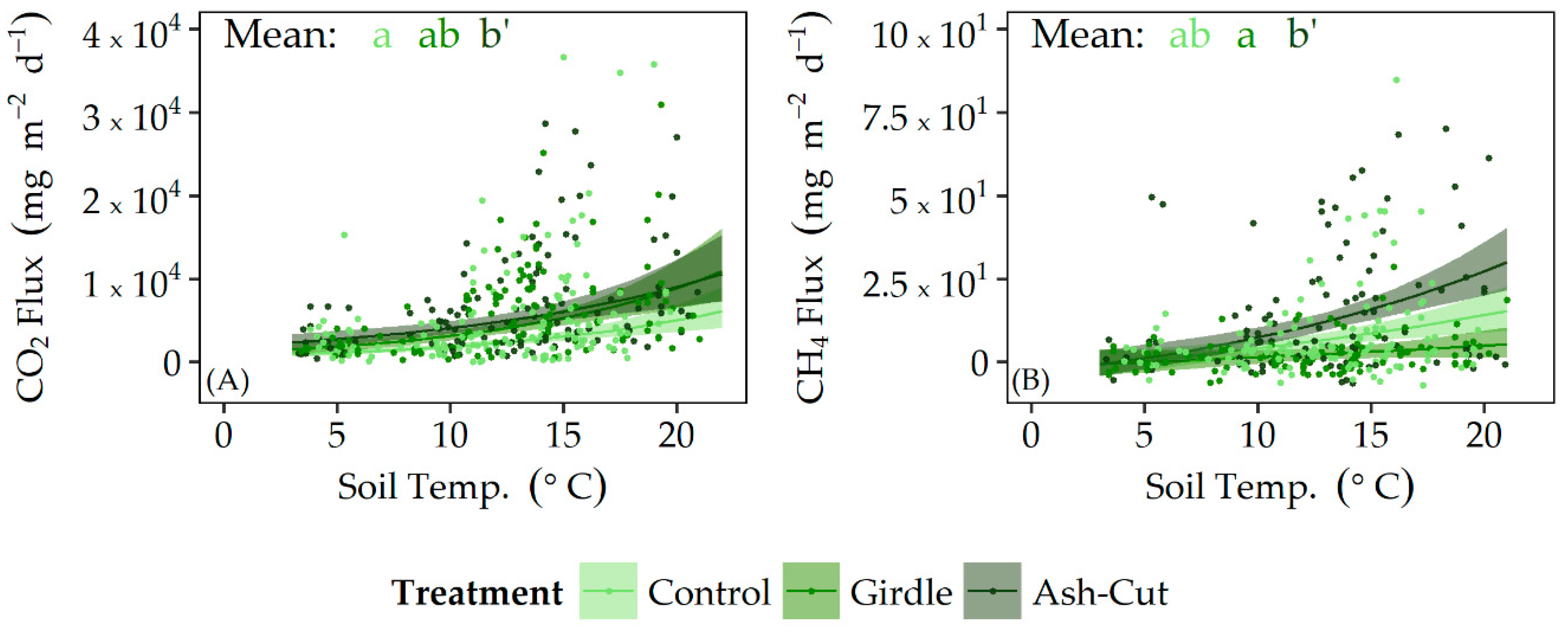
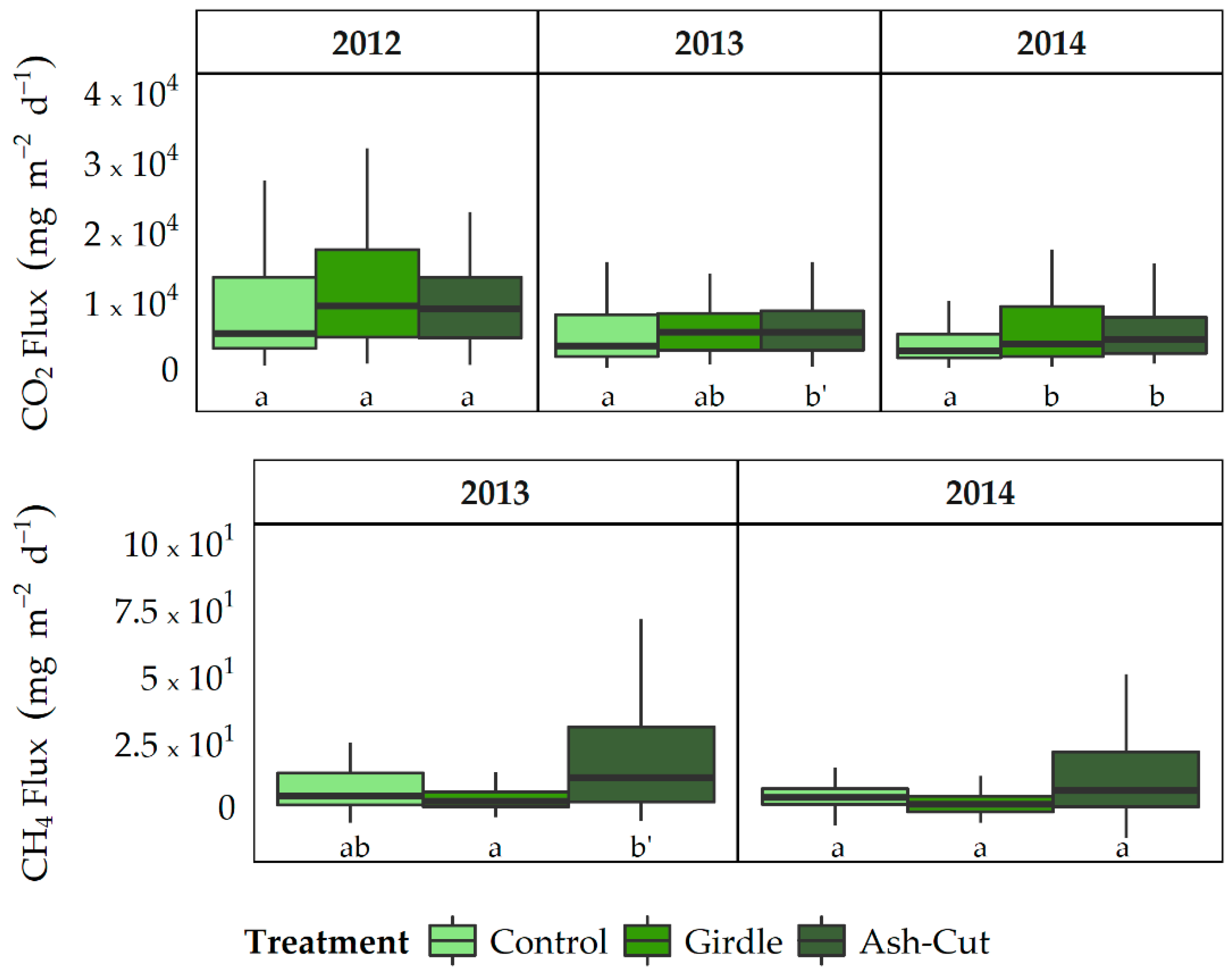
3.2. Soil CO2 and CH4 Fluxes
4. Discussion
4.1. Soil Temperature and Water Level
4.2. Responses of Soil CO2 and CH4 Fluxes to Disturbance
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- McLaughlin, J.W.; Gale, M.R.; Jurgensen, M.F.; Trettin, C.C. Soil organic matter and nitrogen cycling in response to harvesting, mechanical site preparation, and fertilization in a wetland with a mineral substrate. For. Ecol. Manag. 2000, 129, 7–23. [Google Scholar] [CrossRef]
- Sun, G.; McNulty, S.G.; Shepard, J.P.; Amatya, D.M.; Riekerk, H.; Comerford, N.B.; Skaggs, W.; Swift, L. Effects of timber management on the hydrology of wetland forests in the southern united states. For. Ecol. Manag. 2001, 143, 227–236. [Google Scholar] [CrossRef]
- MacFarlane, D.W.; Meyer, S.P. Characteristics and distribution of potential ash tree hosts for emerald ash borer. For. Ecol. Manag. 2005, 213, 15–24. [Google Scholar] [CrossRef]
- Kashian, D.M.; Witter, J.A. Assessing the potential for ash canopy tree replacement via current regeneration following emerald ash borer-caused mortality on southeastern Michigan landscapes. For. Ecol. Manag. 2011, 261, 480–488. [Google Scholar] [CrossRef]
- Flower, C.E.; Knight, K.S.; Gonzalez-Meler, M.A. Impacts of the emerald ash borer (Agrilus planipennis fairmaire) induced ash (Fraxinus spp.) mortality on forest carbon cycling and successional dynamics in the eastern united states. Biol. Invasions 2013, 15, 931–944. [Google Scholar] [CrossRef]
- Sydnor, T.D.; Bumgardner, M.; Todd, A. The potential economic impacts of emerald ash borer (Agrilus planipennis) on Ohio, US, Communities. Arboricult. Urban For. 2007, 33, 48–54. [Google Scholar]
- Kovacs, K.F.; Haight, R.G.; McCullough, D.G.; Mercader, R.J.; Siegert, N.W.; Liebhold, A.M. Cost of potential emerald ash borer damage in U.S. Communities, 2009–2019. Ecol. Econ. 2010, 69, 569–578. [Google Scholar] [CrossRef]
- Gandhi, K.K.; Herms, D. Direct and indirect effects of alien insect herbivores on ecological processes and interactions in forests of eastern north America. Biol. Invasions 2010, 12, 389–405. [Google Scholar] [CrossRef]
- Lovett, G.M.; Canham, C.D.; Arthur, M.A.; Weathers, K.C.; Fitzhugh, R.D. Forest ecosystem responses to exotic pests and pathogens in eastern north America. BioScience 2006, 56, 395–405. [Google Scholar] [CrossRef]
- Kimble, J.M. The Potential of U.S. Forest Soils to Sequester Carbon and Mitigate the Greenhouse Effect; CRC Press: Boca Raton, FL, USA, 2003; p. 429. [Google Scholar]
- Freeman, C.; Ostle, N.; Kang, H. An enzymic ‘latch’ on a global carbon store–A shortage of oxygen locks up carbon in peatlands by restraining a single enzyme. Nature 2001, 409, 149. [Google Scholar] [CrossRef] [PubMed]
- Gorham, E. Northern peatlands: Role in the carbon cycle and probable responses to climatic warming. Ecol. Appl. 1991, 1, 182–195. [Google Scholar] [CrossRef] [PubMed]
- Hogberg, P.; Nordgren, A.; Buchmann, N.; Taylor, A.F.S.; Ekblad, A.; Hogberg, M.N.; Nyberg, G.; Ottosson-Lofvenius, M.; Read, D.J. Large-scale forest girdling shows that current photosynthesis drives soil respiration. Nature 2001, 411, 789–792. [Google Scholar] [CrossRef] [PubMed]
- Zerva, A.; Mencuccini, M. Short-term effects of clearfelling on soil CO2, CH4, and N2O fluxes in a sitka spruce plantation. Soil Biol. Biochem. 2005, 37, 2025–2036. [Google Scholar] [CrossRef]
- Moore, T.R.; Dalva, M. The influence of temperature and water-table position on carbon-dioxide and methane emissions from laboratory columns of peatland soils. J. Soil Sci. 1993, 44, 651–664. [Google Scholar] [CrossRef]
- Macdonald, J.A.; Fowler, D.; Hargreaves, K.J.; Skiba, U.; Leith, I.D.; Murray, M.B. Methane emission rates from a northern wetland; response to temperature, water table and transport. Atmos. Environ. 1998, 32, 3219–3227. [Google Scholar] [CrossRef]
- Van Grinsven, M.J.; Shannon, J.P.; Davis, J.C.; Bolton, N.W.; Wagenbrenner, J.W.; Kolka, R.K.; Grant Pypker, T. Source water contributions and hydrologic responses to simulated emerald ash borer infestations in depressional black ash wetlands. Ecohydrology 2017, 10. [Google Scholar] [CrossRef]
- Slesak, R.A.; Lenhart, C.F.; Brooks, K.N.; D’Amato, A.W.; Palik, B.J. Water table response to harvesting and simulated emerald ash borer mortality in black ash wetlands in Minnesota, USA. Can. J. For. Res. 2014, 44, 961–968. [Google Scholar] [CrossRef]
- Blodau, C. Carbon cycling in peatlands—A review of processes and controls. Environ. Rev. 2002, 10, 111–134. [Google Scholar] [CrossRef]
- Nancy, D.; Narasinha, S.; Peter, W.; Shashi, V.; Elon, V.; Eville, G.; Patrick, C.; Robert, H.; Cheryl, K.; Joseph, Y.; et al. Carbon emissions from peatlands. In Peatland Biogeochemistry and Watershed Hydrology at the Marcell Experimental Forest; CRC Press: Boca Raton, FL, USA, 2011; pp. 297–347. [Google Scholar]
- McFee, W.W.; Kelly, J.M. Soil carbon in northern forested wetlands: Impacts of silvicultural practices. Carbon 1995, 1, 437–461. [Google Scholar]
- Trettin, C.C.; Jurgensen, M.F.; Grigal, D.F.; Gale, M.R.; Jeglum, J.R. Northern Forested Wetlands Ecology and Management; CRC Press: Boca Raton, FL, USA, 1996. [Google Scholar]
- Eswaran, H.; Van Den Berg, E.; Reich, P. Organic carbon in soils of the world. Soil Sci. Soc. Am. J. 1993, 57, 192–194. [Google Scholar] [CrossRef]
- Bridgham, S.; Megonigal, J.P.; Keller, J.; Bliss, N.; Trettin, C. The carbon balance of north American wetlands. Wetlands 2006, 26, 889–916. [Google Scholar] [CrossRef]
- Freeman, C.; Kim, S.Y.; Lee, S.H.; Kang, H. Effects of elevated atmospheric CO2 concentrations on soil microorganisms. J. Microbiol. 2004, 42, 267–277. [Google Scholar] [PubMed]
- Blodau, C.; Basiliko, N.; Moore, T.R. Carbon turnover in peatland mesocosms exposed to different water table levels. Biogeochemistry 2004, 67, 331–351. [Google Scholar] [CrossRef]
- Dalva, M.; Moore, T.R.; Arp, P.; Clair, T.A. Methane and soil and plant community respiration from wetlands, Kejimkujik National Park, Nova Scotia: Measurements, predictions, and climatic change. J. Geophys. Res. Atmos. 2001, 106, 2955–2962. [Google Scholar] [CrossRef]
- Segers, R. Methane production and methane consumption: A review of processes underlying wetland methane fluxes. Biogeochemistry 1998, 41, 23–51. [Google Scholar] [CrossRef]
- Aller, R.C. Bioturbation and remineralization of sedimentary organic-matter—Effects of redox oscillation. Chem. Geol. 1994, 114, 331–345. [Google Scholar] [CrossRef]
- van Hulzen, J.B.; Segers, R.; van Bodegom, P.M.; Leffelaar, P.A. Temperature effects on soil methane production: An explanation for observed variability. Soil Biol. Biochem. 1999, 31, 1919–1929. [Google Scholar] [CrossRef]
- Fissore, C.; Giardina, C.P.; Kolka, R.K.; Trettin, C.C. Soil organic carbon quality in forested mineral wetlands at different mean annual temperature. Soil Biol. Biochem. 2009, 41, 458–466. [Google Scholar] [CrossRef]
- Hagerty, S.B.; Van Groenigen, K.J.; Allison, S.D.; Hungate, B.A.; Schwartz, E.; Koch, G.W.; Kolka, R.K.; Dijkstra, P. Accelerated microbial turnover but constant growth efficiency with warming in soil. Nat. Clim. Chang. 2014, 4, 903–906. [Google Scholar] [CrossRef]
- Daulat, W.E.; Clymo, R.S. Effects of temperature and watertable on the efflux of methane from peatland surface cores. Atmos. Environ. 1998, 32, 3207–3218. [Google Scholar] [CrossRef]
- Kettunen, A.; Kaitala, V.; Lehtinen, A.; Lohila, A.; Alm, J.; Silvola, J.; Martikainen, P.J. Methane production and oxidation potentials in relation to water table fluctuations in two boreal mires. Soil Biol. Biochem. 1999, 31, 1741–1749. [Google Scholar] [CrossRef]
- Peters, V.; Conrad, R. Sequential reduction processes and initiation of CH4 production upon flooding of oxic upland soils. Soil Biol. Biochem. 1996, 28, 371–382. [Google Scholar] [CrossRef]
- Moore, T.R.; Heyes, A.; Roulet, N.T. Methane emissions from wetlands, southern hudson-bay lowland. J Geophys. Res. Atmos. 1994, 99, 1455–1467. [Google Scholar] [CrossRef]
- Amaral, J.A.; Knowles, R. Methane metabolism in a temperate swamp. Appl. Environ. Microbiol. 1994, 60, 3945–3951. [Google Scholar] [PubMed]
- Shannon, R.D.; White, J.R. A three-year study of controls on methane emissions from two Michigan peatlands. Biogeochemistry 1994, 27, 35–60. [Google Scholar] [CrossRef]
- Ballantyne, D.; Hribljan, J.; Pypker, T.; Chimner, R. Long-term water table manipulations alter peatland gaseous carbon fluxes in northern Michigan. Wetl. Ecol. Manag. 2014, 22, 35–47. [Google Scholar] [CrossRef]
- Johnson, C.P.; Pypker, T.G.; Hribljan, J.A.; Chimner, R.A. Open top chambers and infrared lamps: A comparison of heating efficacy and CO2/CH4 dynamics in a northern Michigan peatland. Ecosystems 2013, 16, 736–748. [Google Scholar] [CrossRef]
- Prasad, A.M.; Iverson, L.R. Little’s Range and FIA Importance Value Database for 135 Eastern US Tree Species; Northeastern Research Station: Delaware, OH, USA, 2003. [Google Scholar]
- Davis, J.C.; Shannon, J.P.; Bolton, N.W.; Kolka, R.K.; Pypker, T. Vegetation responses to simulated emerald ash borer infestation in Fraxinus nigra dominated wetlands of Upper Michigan, USA. Can. J. For. Res. 2017, 47, 319–330. [Google Scholar] [CrossRef]
- Soil Survey Staff. Web Soil Survey. Natural Resources Conservation Service, United States Department of Agriculture. Available online: http://websoilsurvey.Nrcs.Usda.Gov/ (accessed on 14 November 2014).
- NOAA-NCEI. National Oceanic and Atomospheric Administration. National Centers for Environmental Information: Asheville, NC, USA. Available online: https://www.ncei.noaa.gov/ (accessed on 15 November 2017).
- McCullough, D.G.; Schneeberger, N.F.; Katovich, S.A.; Siegert, N.W. Pest Alert: Emerald Ash Borer. USDA For. Serv., Northeastern Area, State & Private Forestry: Newtown Square, PA, USA, 2015. NA-PR-02-04. Available online: http://www.na. fs.fed.us/spfo/pubs/pest_al/eab/eab.pdf (accessed on 5 December 2017).
- Tuittila, E.-S.; Komulainen, V.-M.; Vasander, H.; Nykänen, H.; Martikainen, P.J.; Laine, J. Methane dynamics of a restored cut-away peatland. Glob. Chang. Biol. 2000, 6, 569–581. [Google Scholar] [CrossRef]
- Carroll, P.; Crill, P. Carbon balance of a temperate poor fen. Glob. Biogeochem. Cycles 1997, 11, 349–356. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing [Computer Software]; R: Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using LME4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Skoog, D.A.; Holler, J.; Crouch, S.R. Liquid chromatography. In Principles of Instrumental Analysis, 6th ed.; Thomson Cengage Learning: Boston, MA, USA, 2006; pp. 816–855. [Google Scholar]
- Hewett, P.; Ganser, G.H. A comparison of several methods for analyzing censored data. Ann. Occup. Hyg. 2007, 51, 611–632. [Google Scholar] [PubMed]
- Millard, S.P. Envstats, an R Package for Environmental Statistics; Springer Science & Business Media: Berlin, Germany, 2013. [Google Scholar]
- Nakagawa, S.; Schielzeth, H. A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol. Evol. 2013, 4, 133–142. [Google Scholar] [CrossRef]
- Lenth, R.V. Lsmeans: Least-Squares Means. R Package Version 2013, Volume 2. Available online: https://www.jstatsoft.org/article/view/v069i01/0 (accessed on 18 October 2016).
- Halekoh, U.; Højsgaard, S.; Højsgaard, M.S.; Matrix, I. Package ‘pbkrtest’. 2014. Available online: https://cran.r-project.org/web/packages/pbkrtest/index.html (accessed on 23 September 2016).
- Gałecki, A.; Burzykowski, T. Linear Mixed-Effects Models Using R; Springer: New York, NY, USA, 2013. [Google Scholar]
- Fox, J. Effect displays in R for generalised linear models. J. Stat. Softw. 2003, 8, 1–27. [Google Scholar] [CrossRef]
- Wickham, H.; Chang, W. Ggplot2: An Implementation of the Grammar of Graphics. 2015. Available online: http://ftp.auckland.ac.nz/software/CRAN/src/contrib/Descriptions/ggplot.html (accessed on 11 August 2016).
- Walko, R.L.; Band, L.E.; Baron, J.; Kittel, T.G.; Lammers, R.; Lee, T.J.; Ojima, D.; Pielke, R.A., Sr.; Taylor, C.; Tague, C. Coupled atmosphere–biophysics–hydrology models for environmental modeling. J. Appl. Meteorol. 2000, 39, 931–944. [Google Scholar] [CrossRef]
- Kolka, R.K.; Sebestyen, S.D.; Verry, E.S.; Brooks, K.N. Peatland Biogeochemistry and Watershed Hydrology at the Marcell Experimental Forest; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2011. [Google Scholar]
- Noh, N.J.; Shannon, J.; Bolton, N.; Davis, J.; Van Grinsven, M.; Pypker, T.; Kolka, R.; Wagenbrenner, J. Carbon dioxide fluxes from coarse dead wood in black ash wetlands. Forests 2018, in press. [Google Scholar]
- Dinsmore, K.; Skiba, U.; Billett, M.; Rees, R. Effect of water table on greenhouse gas emissions from peatland mesocosms. Plant Soil 2009, 318, 229–242. [Google Scholar] [CrossRef]
- Chimner, R.A.; Cooper, D.J. Influence of water table levels on CO2 emissions in a colorado subalpine fen: An in situ microcosm study. Soil Biol. Biochem. 2003, 35, 345–351. [Google Scholar] [CrossRef]
- Leifeld, J.; Steffens, M.; Galego-Sala, A. Sensitivity of peatland carbon loss to organic matter quality. Geophys. Res. Lett. 2012, 39. [Google Scholar] [CrossRef]
- Updegraff, K.; Pastor, J.; Bridgham, S.D.; Johnston, C.A. Environmental and substrate controls over carbon and nitrogen mineralization in northern wetlands. Ecol. Appl. 1995, 5, 151–163. [Google Scholar] [CrossRef]
- Yavitt, J.B.; Williams, C.J.; Wieder, R.K. Production of methane and carbon dioxide in peatland ecosystems across north America: Effects of temperature, aeration, and organic chemistry of pent. Geomicrobiol. J. 1997, 14, 299–316. [Google Scholar] [CrossRef]
- Best, E.P.H.; Jacobs, F.H.H. The influence of raised water table levels on carbon dioxide and methane production in ditch-dissected peat grasslands in the netherlands. Ecol. Eng. 1997, 8, 129–144. [Google Scholar] [CrossRef]
- Ewel, K.C.; Cropper, W.P., Jr.; Gholz, H.L. Soil CO2 evolution in florida slash pine plantations. Ii. Importance of root respiration. Can. J. For. Res. 1987, 17, 330–333. [Google Scholar] [CrossRef]
- Hanson, P.J.; Edwards, N.T.; Garten, C.T.; Andrews, J.A. Separating root and soil microbial contributions to soil respiration: A review of methods and observations. Biogeochemistry 2000, 48, 115–146. [Google Scholar] [CrossRef]
- Edwards, N.T.; Harris, W.F. Carbon cycling in a mixed deciduous forest floor. Ecology 1977, 58, 431–437. [Google Scholar] [CrossRef]
- Burns, R.M.; Honkala, B.H. Silvics of North America; United States Department of Agriculture: Washington, DC, USA, 1990; Volume 2. [Google Scholar]
- Costilow, K.; Knight, K.; Flower, C. Disturbance severity and canopy position control the radial growth response of maple trees (Acer spp.) in forests of northwest Ohio impacted by emerald ash borer (Agrilus planipennis). Ann. For. Sci. 2017, 74, 10. [Google Scholar] [CrossRef]
- Thomas, K.L.; Benstead, J.; Davies, K.L.; Lloyd, D. Role of wetland plants in the diurnal control of CH4 and CO2 fluxes in peat. Soil Biol. Biochem. 1996, 28, 17–23. [Google Scholar] [CrossRef]
- Nave, L.; Gough, C.; Maurer, K.; Bohrer, G.; Hardiman, B.; Le Moine, J.; Munoz, A.; Nadelhoffer, K.; Sparks, J.; Strahm, B. Disturbance and the resilience of coupled carbon and nitrogen cycling in a north temperate forest. J. Geophys. Res. Biogeosci. 2011, 116. [Google Scholar] [CrossRef]
- Nuckolls, A.E.; Wurzburger, N.; Ford, C.R.; Hendrick, R.L.; Vose, J.M.; Kloeppel, B.D. Hemlock declines rapidly with hemlock woolly adelgid infestation: Impacts on the carbon cycle of southern Appalachian forests. Ecosystems 2009, 12, 179–190. [Google Scholar] [CrossRef]
- Davis, J.C. Vegetation Dynamics and Nitrogen Cycling Responses to Simulated Emerald Ash Borer Infestation in Fraxinus nigra-Dominated Wetlands of Upper Michigan, USA; Michigan Technological University: Houghton, MI, USA, 2016. [Google Scholar]
- Zehnder, A.; Stumm, W. Geochemistry and biogeochemistry of anaerobic habitats. In Biology of Anaerobic Microorganisms; John Wiley & Sons, Inc.: New York, NY, USA, 1988; pp. 1–38. [Google Scholar]
- Dunfield, P.; Knowles, R.; Dumont, R.; Moore, T.R. Methane production and consumption in temperate and subarctic peat soils: Response to temperature and PH. Soil Biol. Biochem. 1993, 25, 321–326. [Google Scholar] [CrossRef]
- Moore, T.R.; Dalva, M. Methane and carbon dioxide exchange potentials of peat soils in Aerobic and Anaerobic laboratory incubations. Soil Biol. Biochem. 1997, 29, 1157–1164. [Google Scholar] [CrossRef]
- Gauci, V.; Gowing, D.J.G.; Hornibrook, E.R.C.; Davis, J.M.; Dise, N.B. Woody stem methane emission in mature wetland alder trees. Atmos. Environ. 2010, 44, 2157–2160. [Google Scholar] [CrossRef]
- Erdmann, G.G.; Crow, T.R.; Ralph, M., Jr.; Wilson, C.D. Managing Black Ash in the Lake States; General Technical Report NC-115; US Dept. of Agriculture, Forest Service, North Central Forest Experiment Station: St. Paul, MN, USA, 1987; Volume 115.
- Flower, C.E.; Gonzalez-Meler, M.A. Responses of temperate forest productivity to insect and pathogen disturbances. Annu. Rev. Plant Biol. 2015, 66, 547–569. [Google Scholar] [CrossRef] [PubMed]
- Looney, C.E.; D’Amato, A.W.; Palik, B.J.; Slesak, R.A. Canopy treatment influences growth of replacement tree species in Fraxinus nigra forests threatened by the emerald ash borer in Minnesota, USA. Can. J. For. Res. 2016, 47, 183–192. [Google Scholar] [CrossRef]
- Bolton, N.; Shannon, J.; Davis, J.; Grinsven, M.V.; Noh, N.J.; Schooler, S.; Kolka, R.; Pypker, T.; Wagenbrenner, J. Methods to improve survival and growth of planted alternative species seedlings in black ash ecosystems threatened by emerald ash borer. Forests 2018, 9, 146. [Google Scholar] [CrossRef]
- Iverson, L.; Knight, K.S.; Prasad, A.; Herms, D.A.; Matthews, S.; Peters, M.; Smith, A.; Hartzler, D.M.; Long, R.; Almendinger, J. Potential species replacements for black ash (Fraxinus nigra) at the confluence of two threats: Emerald ash borer and a changing climate. Ecosystems 2016, 19, 248–270. [Google Scholar] [CrossRef]
- Zehetgruber, B.; Kobler, J.; Dirnböck, T.; Jandl, R.; Seidl, R.; Schindlbacher, A. Intensive ground vegetation growth mitigates the carbon loss after forest disturbance. Plant Soil 2017, 420, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Hayhoe, K.; VanDorn, J.; Croley, T., II; Schlegal, N.; Wuebbles, D. Regional climate change projections for Chicago and the US great lakes. J. Great Lakes Res. 2010, 36, 7–21. [Google Scholar] [CrossRef]
| LMM | Predictors | AIC | PCV | RSE | ||
|---|---|---|---|---|---|---|
| Soil Temp. | Null: x = | 0.97 | 0.73 | 1763 | – | 0.62 |
| Full: x = | 0.98 | 0.73 | 1225 | 0.6% | 0.39 | |
| CO2 Flux | Null: x = | 0.34 | 0.14 | 1564 | – | 2.48 |
| Full: x = | 0.51 | 0.31 | 1489 | 0.2% | 2.27 | |
| CH4 Flux | Null: x = | 0.49 | 0.05 | 621 | – | 0.86 |
| Full: x = | 0.50 | 0.10 | 653 | 0.1% | 0.85 |
| Gas | Study Period | Treatment | df | Mean Flux (mg m−2 d−1) | SE (mg m−2 d−1) | Mean Test | n | Rank Mean | KW Test |
|---|---|---|---|---|---|---|---|---|---|
| CO2 | Pre | Control | 18.9 | 3.0 × 103 | 1.26 | a | 10 | 13.5 | a |
| Girdle | 16.5 | 4.0 × 103 | 1.25 | a | 10 | 16.5 | a | ||
| Ash-Cut | 30.9 | 5.1 × 103 | 1.32 | a | 10 | 16.5 | a | ||
| Post | Control | 6.2 | 3.1 × 103 | 1.18 | a | 24 | 30.8 | a | |
| Girdle | 7.5 | 5.7 × 103 | 1.19 | b’ | 24 | 37.4 | ab | ||
| Ash-Cut | 8.7 | 6.7 × 103 | 1.24 | b | 24 | 41.3 | b’ | ||
| CH4 | Post | Control | 5.3 | 6.6 | 1.10 | ab | 24 | 33.3 | a |
| Girdle | 5.5 | 2.4 | 1.11 | a | 24 | 26.6 | a | ||
| Ash-Cut | 5.9 | 11.9 | 1.11 | b’ | 24 | 47.6 | b |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van Grinsven, M.; Shannon, J.; Bolton, N.; Davis, J.; Noh, N.J.; Wagenbrenner, J.; Kolka, R.; Pypker, T. Response of Black Ash Wetland Gaseous Soil Carbon Fluxes to a Simulated Emerald Ash Borer Infestation. Forests 2018, 9, 324. https://doi.org/10.3390/f9060324
Van Grinsven M, Shannon J, Bolton N, Davis J, Noh NJ, Wagenbrenner J, Kolka R, Pypker T. Response of Black Ash Wetland Gaseous Soil Carbon Fluxes to a Simulated Emerald Ash Borer Infestation. Forests. 2018; 9(6):324. https://doi.org/10.3390/f9060324
Chicago/Turabian StyleVan Grinsven, Matthew, Joseph Shannon, Nicholas Bolton, Joshua Davis, Nam Jin Noh, Joseph Wagenbrenner, Randall Kolka, and Thomas Pypker. 2018. "Response of Black Ash Wetland Gaseous Soil Carbon Fluxes to a Simulated Emerald Ash Borer Infestation" Forests 9, no. 6: 324. https://doi.org/10.3390/f9060324
APA StyleVan Grinsven, M., Shannon, J., Bolton, N., Davis, J., Noh, N. J., Wagenbrenner, J., Kolka, R., & Pypker, T. (2018). Response of Black Ash Wetland Gaseous Soil Carbon Fluxes to a Simulated Emerald Ash Borer Infestation. Forests, 9(6), 324. https://doi.org/10.3390/f9060324







