Abstract
Trypodendron laeve Eggers, 1939 is a species of ambrosia beetle much less known than the other three Trypodendron species occurring in Europe. Its status (native or alien) in Central Europe has been a subject of debate over the past two decades. In Romania, the species was discovered in 2008 and the aim of the research presented in this paper was to investigate its distribution in the Carpathians, mainly at high altitudes (>800 m), in tree stands with Norway spruce (Picea abies [L.] H. Karst). Panel intercept traps baited with synthetic pheromone for Trypodendron lineatum (Olivier, 1795) were used in the spring of 2015, at 31 locations. Adults of T. laeve were caught in 20 of them. Additional observations were made within some studies using similar baits and T. laeve specimens were caught in eight locations. T. laeve was always trapped together with T. lineatum, and at some locations also together with T. domesticum (Linnaeus, 1758) and T. signatum (Fabricius, 1787). In all traps, fewer specimens of T. laeve were caught compared to T. lineatum. The species has a widespread distribution in the mountain regions, within forests composed of native tree species and generally located far away from commercial routes. There, it occurs together with other native species of the same taxonomic genus. It seems to be more abundant at high altitudes, but overall its populations are less abundant than those of T. lineatum.
1. Introduction
The species of the genus Trypodendron Stephens, 1830 are ambrosia beetles, which make breeding galleries in wood, but feed on symbiotic fungi [1]. They infest the sapwood of weak, dying, and newly dead trees, logs, and stumps [2]. The beetles locate the suitable hosts using the ethanol released from wood undergoing anaerobic fermentation as a guiding cue [3]. However, they are able to distinguish broadleaved species from conifers. Trypodendron species living on conifers use the monoterpenes, mainly α-pinene, to recognize their hosts, the ethanol and α-pinene acting synergistically in attracting the beetles [4,5,6]. Then, the beetles aggregate by means of aggregation pheromones [7,8]. In fact, conifer living species use lineatin along with the two host volatiles as a very effective aggregation signal [9,10], while the adults of T. domesticum appear to be repelled by terpenes [5,11]. The tunnels excavated by insects and fungal staining can cause important economic loss through the degradation of the infested logs. In addition, some ambrosia beetle species attack apparently healthy trees [12] and consequently are important economic pests [13].
According to the latest reference work on bark beetles, the Holarctic region contains 13 species of Trypodendron [14], with four in Europe including T. domesticum (Linnaeus, 1758), T. signatum (Fabricius, 1787), T. lineatum (Olivier, 1795), and T. laeve Eggers, 1939 [15]. The first two species occur on broadleaved trees, with the second one seeming to prefer the oaks (Quercus spp.) [2]. In the Palaearctic region, T. lineatum was found on many species of Picea A. Dietr., 1824, Pinus L. 1753, Abies Mill. 1754, Larix Mill. 1754, and Cedrus Trew 1757 [16], while in North America, it is also occasionally found attacking species of Alnus Mill., Betula L. 1753, Acer L. 1753, and Malus Mill. 1754 [13,17]. The host tree species of T. laeve are less known. It was found in logs or dead trees of Picea abies (L.) H. Karst. 1881 [18,19,20] and Pinus sylvestris L. 1753 [20,21]. Picea obovata Ledeb. 1833 and P. jezoensis (Siebold & Zucc.) Carr. are also between its hosts [16].
While T. lineatum and the European species associated with broadleaved trees are quite well studied, T. laeve is less known, because its taxonomic status was only recently clarified. It was firstly described in Japan [22] and secondly (a few years later) in Norway, under the name Trypodendron piceum A. Strand, 1946 [18]. However, during the next four decades, the species was not reported in other parts of Europe. The standard taxonomic literature and identification keys used during that time, such as Balachowsky [23], Stark [24], Nunberg [25], and Pfeffer [26], did not include T. laeve or T. piceum. Consequently, T. laeve was “forgotten” or overlooked, particularly by applied entomologists. Later, Grüne [27] did not refer to T. laeve or T. piceum at all in the illustrated key of the European scolytids, and Schedl [28] regarded both names as synonyms for T. lineatum. Even Annila et al. [29] did not distinguish between T. piceum and T. lineatum [30], although it was known that the species was present in the Nordic countries.
Eventually, Holzschuh [19], comparing some specimens captured in 1982–1983 that looked different from those of T. lineatum, with specimens from the Schedl’s collection of scolytids, identified those specimens as belonging to T. laeve and drew attention to the fact that they differ from T. lineatum both in the conformation of male genitalia and in the femur colour, suggesting the treatment of T. laeve as a distinct species. Pfeffer [31] mistakenly synonymised T. piceum Strand with T. proximum (Niijima, 1909), but it was corrected in Pfeffer [16,32], where he presents T. laeve as a separate species and the name T. piceum as a synonym for T. laeve, as Wood [33] suggested. Due to the original mistake in Pfeffer [31], T. laeve (=T. piceum) was mentioned by Martikainen [20] under the name T. proximum, but Mandelshtam and Popovichev [34] demonstrated that they are different species. This clarifies the taxonomic status of the T. laeve species in Europe, but not its origin.
In the 1980s, in Austria, various species of Siberian insects were reported, and Holzschuh [19] assumed that T. laeve is also an alien species that came to Europe with imported wood from Russia. The idea was reiterated in later works [35,36,37] and some authors mentioned this species as an invasive one in Europe [38,39]. However, the information gathered until the year 2000 about the distribution of the species in Fennoscandia led Martikainen [20] to reconsider the status of the species. Consequently, Kenis [38] regarded the species to be native to Scandinavia but alien to central Europe, where its distribution was less known. Apart from the above-mentioned reports from Austria, there were few reports from Germany [40] and the Czech Republic [41,42].
Currently, the species is also reported as being present in Estonia, Finland, Latvia, Norway, Poland, Slovakia, Sweden, Switzerland, the Central European Territory and North European Territory of Russia, China, and Japan [15], as well as from Romania [43] and the Russian Far East [44]. However, while the species distribution in the Fennoscandia is well documented [20,21,30,45,46,47,48,49,50,51], in Central and Eastern Europe, the knowledge is quite poor, although some studies have been done [43,52,53]. Consequently, the objective of the research presented in this paper was to determine the species distribution in Romania.
2. Materials and Methods
2.1. Field Research
To determine the species distribution of T. laeve in Romania, in the spring of 2015, with the help of field forestry personnel, 78 pheromone traps were placed in 31 locations situated along the Carpathian Mountains chain (Table 1).

Table 1.
Location of study sites and tree stand characteristics where pheromone traps were set up.
Trapping locations were chosen to be—generally—at altitudes above 800 m, within pure Norway spruce (Picea abies) stands or mixed stands of spruce and other native species, mainly silver fir (Abies alba), European beech (Fagus sylvatica L.), sycamore (Acer pseudoplatanus L.), and European larch (Larix decidua), most of them aged over 75–80 years. The selected sites were tree stands located at 50–100 m from the places where harvested logs were stored in 2014 before transport to a mill or nearby in one-year old clear-cut areas, but far from the main rail or car transport routes and woodworking factories.
At each location, three traps were placed in general, but at some points, there were only one to two traps (Table 2). The pheromone traps were of the Intercept® type. They were set up within the stand, at 10–15 m from the stand edge and at a distance of 50–100 m from each other.

Table 2.
Captures of Trypodendon species at the 31 sites monitored in 2015.
The traps were primed with pheromone lures whose composition was optimized to attract beetles of T. lineatum, like in other studies [19,20,35,51,52], because there are no commercial products designed to attract T. laeve. The lures contained diluted lineatin in methylbutenol, ethanol, and alpha-pinene. This mixture diffused through a polyethylene film at a rate of 30 mg/day at 20 °C. The pheromone lures and traps were provided by the Research Institute in Chemistry “Raluca Ripan” Cluj-Napoca.
Since February 2015 and the first ten days of March 2015 were warmer than normal, the installation of the traps in the forest was scheduled for March, so that they would be operational before the maximum daily temperature reached 13 °C, but in the second half of March and the beginning of April, the weather had become very variable, including snowing periods, and in many cases, the installation of traps was postponed for April or even May.
The traps were kept in the field for four to eight weeks and the captures were generally checked weekly, and in some cases every two weeks. The collected insects were preserved in ethanol until their identification.
Additional data on the presence of the T. laeve species in other sites was obtained from the processing of biological material captured in eight other studies using the same type of pheromone, and in one case (at Borca, Neamţ county), a flying beetle collected on logs.
2.2. Determining the Date of the Commencement of the Flight and the Number of Missed Flight Days
Given the way in which field traps were installed, for a fair interpretation of the results, it was necessary to indirectly determine whether they were set up before or after the beginning of flight, and the respective number of missed flight-friendly days.
For this purpose, the maximum daily air temperature (Tmax) values for January–May 2015 have been extracted from the E-OBS data base [54] version 15.0, for the sites where the traps had been installed. The average altitude of the area covered by the grid cell (0.25° × 0.25°) corresponding to each site has been obtained from the same data base.
Then, the maximum daily temperature was corrected for the difference between the elevation of the field location and the mean elevation of the grid cell, taking into account a mean thermal gradient for the maximum daily temperature during the spring of 0.86 °C/100 m for the Eastern Carpathians, 0.92 °C/100 m for the Southern Carpathians, and 0.79 °C/100 m for the Western Carpathians [55].
Daily corrected maximum temperatures (Tmax.correct) were then compared to the thermal threshold (13 °C) at which, according to the data published by Martikainen [20], the flight of T. laeve species begins. The first day of the year when this threshold was reached was considered to be the start date of the flight.
If the trap installation was made after that date, the time interval between the start of the flight and the day of the trap installation was considered a delay time, expressed in days of delay. In the period of delay, the maximum daily temperature was often below the thermal threshold and the insects did not fly. Subtracting from the total number of days of delay those in which the maximum corrected temperature was below 13 °C resulted in the number of days actually missed and for those days, the average maximum temperature (Tmax.aver) was computed.
The same procedure was used for T. lineatum for which a temperature threshold of 15 °C [20] was taken into account.
2.3. Studies of Collection Material
In order to find out if there are specimens of T. laeve in the country collections, the main collections of Scolytinae in the country have been checked, namely those from the Museum of Natural Sciences Suceava—“Ştefan Negru” collection, presented in the catalogue published by Vasiliu et al. [56]; from the Brukenthal Museum in Sibiu—a collection presented by Negru [57]; and from the Faculty of Biology of Babeş-Bolyai University in Cluj-Napoca—the non-catalogued Orest Mark’s collection.
Species identification was done using the key published by Pfeffer [16].
2.4. Data Processing
Since our primary goal was to determine whether T. laeve is present in the locations where traps were installed, the results are given as the total number of captures per site. However, in order to facilitate the interpretation of data on T. laeve captures, taking into account the captures of T. lineatum, a species collected at all sites and which uses the same substrate with T. laeve [18,19,20], the potential correlation between the captures of these species was analysed. For this analysis, only the places with T. laeve captures were taken into account. Because the data were not normally distributed (Shapiro-Wilk test), even after their log or square root transformation, Spearman’s rank order correlation was run.
To understand if T. laeve responds differently than T. lineatum to the pheromone we used, a fact that could have affected T. laeve captures, the proportion of males in the total catches of the two species was analysed. It has been assumed that the sex ratio in nature for both species is the same, 1:1, as is known for T. lineatum [58]. Only locations where at least 30 specimens of each species were captured were considered, and the proportions have been calculated for each place. Testing for the difference between the two proportions was done using the Z-test [59], because theoretically, both sexes have the same chance of being captured.
A significance level of 0.05 was taken into account both for correlation analysis and for the comparison of two proportions. Statistical calculations were made with XLSTAT 2012 version (Addinsoft: Paris, France).
2.5. Maps
The distribution map for each Trypodendron species was obtained in ArcMap 10.2.2 software (Esri: Redlands, CA, USA). The projected coordinate system used was GCS WGS 1984.
3. Results
At the 31 sites where traps were installed to detect T. laeve in the spring of 2015, a total of 30,175 specimens of Trypodendron were captured, of which 863 (2.9%) were T. laeve, 28,041 (92.9%) T. lineatum, 1149 (3.8%) T. domesticum, and 122 (0.4%) T. signatum (Table 2).
T. laeve was trapped at 20 places, while T. lineatum was captured at 31, T. domesticum at 29, and T. signatum at seven places (Figure 1, Figure 2, Figure 3 and Figure 4).
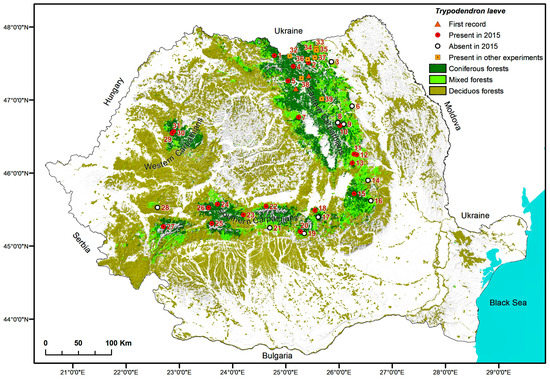
Figure 1.
Sampling locations surveyed for the occurrence of Trypodendron laeve in 2015 and locations where it was found on other occasions mentioned in this study.
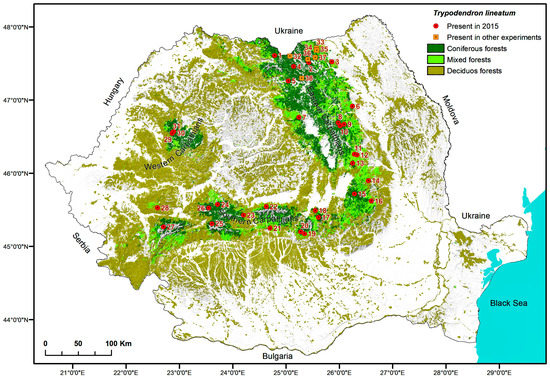
Figure 2.
Sampling locations where Trypodendron lineatum was found in 2015 or on other occasions mentioned in this study.
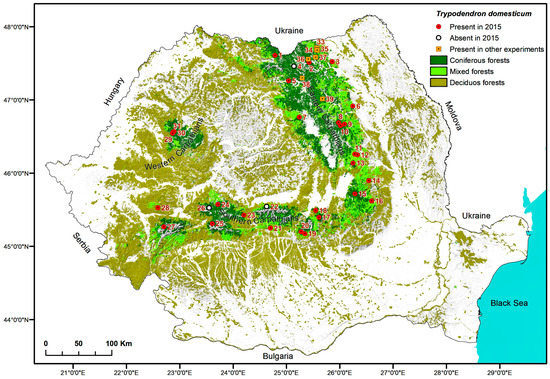
Figure 3.
Sampling locations where Trypodendron domesticum was found in 2015 or on other occasions mentioned in this study.
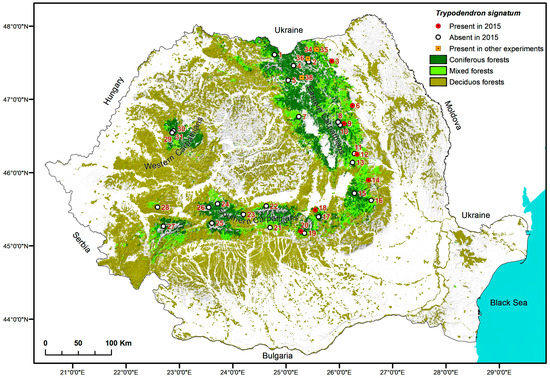
Figure 4.
Sampling locations where Trypodendron signatum was found in 2015 or on other occasions mentioned in this study.
There was a strong positive and statistically significant correlation (rs = 0.6225, p = 0.0041) between T. lineatum and T. laeve captures.
The number of T. laeve captures varied greatly from one place to another, being between 0.33 and 72.3 beetles/trap, with most of them in Cârlibaba (25.1%) and Poiana Braşov (20.6%), where the traps were set up in the spring of 2015 sufficiently early in relation to the altitude of the place and the evolution of the weather; in both cases, only one day of flight was lost.
Where the traps were placed before the flight or at most four days favourable to flight were lost, and the maximum daily temperature of those days did not exceed, on average, 15 °C, the ratio between the number of T. laeve and T. lineatum (caught during the T. laeve flight period) was, with only two exceptions, greater than 1:25.0. Where more than four flight-favourable days have been missed, the number of T. laeve captures has considerably decreased both in absolute terms and in relation to T. lineatum captures. In the places where more than 10 flight-favourable days have been lost, at most one to three beetles have been caught (e.g., at Doda Pilii and Padiş_2).
T. laeve was not captured at Soveja, Padiş_1, and Predeal, where the traps were installed at the latest time.
Some beetles of T. laeve were also caught in other locations (Table 3 and Figure 1), in traps baited with synthetic pheromone for T. lineatum, but—in the most cases—captures were very low compared to those of T. lineatum. However, in Putna, at 850 m above sea level, the captures of T. laeve were higher than those in Carlibaba and Poiana Brasov.

Table 3.
Captures of Trypodendron species in other studies that have been conducted with synthetic pheromone of T. lineatum.
The proportion of males was 45.2–81.7% and 32.6–50.7% in T. lineatum and T. laeve captures, respectively, at the locations monitored in 2015. At all locations, except one (Bădeni), the male proportion in T. laeve captures was statistically significantly lower than in T. lineatum captures (z = 2.26–5.56; p < 0.05). In one of the eight additional studies using traps with similar baits (Putna, in 2017), the male proportions were 78.0–84.4% in T. laeve captures and 76.8–78.4% in T. lineatum captures, and the differences between proportions were not statistically significant (z = 0.18–1.10; p > 0.05).
No specimens of T. laeve were found in the coleopteran collections analysed in the study.
4. Discussion
Adults of T. laeve were captured along the entire Carpathian Mountains chain in Romania and in nine of the sampling locations the catches were substantial (more than 30 individuals). At some sites, the catches were very low, and in one third of all sampling sites, no specimens of T. laeve were trapped.
Considering that T. laeve fly very early and the intense flight takes only about three weeks [21,37,51], it is quite normal for the number of specimens to be smaller where the traps have not functioned throughout the flight period (as was the case for 23 of 31 places), or even to capture nothing where the traps have been set up after the end of the flight (e.g., in Predeal).
On the other hand, the abundance of Trypodendron individuals in a given location is dependent on the abundance of the ephemeral substrate in which these species develop, varying with it. Where the host resource is reduced, the populations decline, while they increase where the available host habitats increase [13,51,60]. Consequently, relatively small or missing T. laeve captures from some places where the installation of the traps was not delayed (Lunca Ilvei, Moroeni) or where only few favourable days for flight were lost (Sălătruc, Dărmăneşti, Paltin, Brateş_1–3, Almaş, Cacica) could be due to the poverty of the suitable breeding material (wind–thrown trees, stumps, coniferous trees killed by bark beetles). This is suggested by T. lineatum’s low captures from the same places, with the captures of the two species (T. laeve and T. lineatum) being closely correlated according to our results and to data from other studies [61], because both species colonize coniferous wood. The results from Putna confirm this hypothesis. While the traps at the elevations of 650 m and 750 m were set up in tree stands with only a few stumps (all older than one year), at an 850 m altitude, the traps were located at about 50 m apart from the logs (about 100 cubic meters) abandoned in the forest from the previous spring (2014) and many more beetles have been captured.
It is known that T. laeve hibernates in tree bark and wood [20], while T. lineatum hibernates in soil [13], which allows the first species to fly when the soil is still covered with snow. This gives T. laeve a competitive advantage over T. lineatum in occupying the available substrate [62], but only when the soil is covered by snow or when the warming in the spring does not proceed very quickly, but gradually. If warming is very fast, there is no delay or only a very short one between the dates when the maximum daily temperature reaches 13 °C and 15 °C when the flight of T. laeve and T. lineatum respectively starts [20]. This happened in 13 of the places where traps were set up in 2015. The almost simultaneous beginning of the flight of the two species was observed in Austria at low altitudes by Krehan and Holzschuh [37] and in southern Sweden in the nemo-boreal zone, where there is little or no snow in winter, by Öhrn et al. [51]. Even in many parts of the very north or high altitude forests, the beginning of the flight is more or less the same for both species (Torstein Kvamme, personal communication). In this context, T. laeve is also disadvantaged by the fact that the adults of this species apparently do not produce a sister-brood [20,51], as is the case with T. lineatum [13,63].
The above-mentioned issues could explain why T. lineatum was captured in a much larger number (at least 3.2 times more) than T. laeve, even if only the flight time of T. laeve is taken into account and only a maximum of one to two days of T. laeve flight were missed. However, it is possible that the number of captures has also been influenced to a certain extent by a possible differential response of the two species to the synthetic pheromone used in the traps, as Öhrn et al. [51] suggested for their study. If the natural sex ratio in the case of the two species is the same, the different proportions of males in the total captures indicate quite a different response of the two species to the synthetic pheromone used in this study. Such differences have also been reported by Krehan and Holzschuh [37], Martikainen [20], and Lukášová and Holuša [61]. The fact that the two species do not respond in the same way to olfactory stimuli was also evidenced by Kvamme [21], who found that T. lineatum adults (especially the males) were attracted to alpha-pinene released at a rate of 1.2–1.4 mg/h, while those of T. laeve were not.
In the case of a very low population density in some places, the lack of T. laeve captures may also be the result of other factors, such as the low sampling effort (up to three traps and in some places only one) or the inappropriate placement of some traps, so that in five places, only one or two of three traps captured adults of this species. Observations made in other studies have highlighted the fact that traps that are not seen due to the foliage of young trees (with branches close to the ground) capture much less insects than those that are not surrounded by obstacles (Olenici, unpublished data). On the other hand, Öhrn et al. [51] noted that both T. lineatum and T. laeve were captured in larger numbers in traps placed in shade than in the sun-exposed traps. This may be the result of beetles responding better to olfactory stimuli under relatively still conditions [64], which they find inside the forest rather than in open settings [65].
Of the Trypodendron species which develop in the wood of broadleaved trees, it is worth noting that, although in many places the traps were located in coniferous stands, T. domesticum was found almost everywhere, suggesting that it is a common species in Romania, like T. lineatum. On the other hand, T. signatum was collected from a much smaller number of places.
It seems that T. signatum is a rare species compared to T. domesticum, previously being reported in Romania from only a few places (Haţeg, Huluzu in Latoriţa Mountains, Sibiu, Braşov, Şumuleu Ciuc, Mihăileni, Frumoasa—Harghita, Tazlău—Neamţ, and Remeţi—Maramureş) [56,66,67,68].
The lack of specimens of T. laeve in the collections of Scolytinae analysed by us may be due to several reasons. First of all, the scolytids have been much less collected and studied in Romania than in the countries with a rich entomological tradition. Secondly, entomological excursions and insect sampling are not usually done so early, when T. laeve is flying. Even nowadays, it seems unusual for many people, including practitioners, to search for insects during the winter months or when snow is still on the ground. Thirdly, until recently, when there were no pheromone lures, collecting bark and ambrosia beetles by an axe and chisel was a more difficult and uncertain task than today, especially for rare species. Eloquent is the fact that almost all the data obtained after 1980 on the presence of T. laeve in different places were obtained using traps baited with synthetic attractants [19,21,35,37,44,52,53]. Even in countries with a very rich entomological tradition, there is relatively little historical data. Specimens of T. laeve were found as early as the 19th century or the beginning of the 20th century only in Sweden and Finland [30], while in Germany, there is a single historical record which dates back to 1953 [40].
Summarizing the above, one can say that T. laeve is a widespread species in the Carpathian Mountains, where it coexists with at least two other indigenous species: T. lineatum and T. domesticum. It has a continuous range, accompanying the Norway spruce even where it has been extended into the altitudinal belt of beech forests, but appears to be more abundant in the spruce forests at high altitudes.
5. Conclusions
T. laeve has a widespread distribution in the Carpathian Mountains and it seems that the species is more abundant at high altitudes. Overall, its populations are less abundant than those of T. lineatum.
Author Contributions
N.O. conceived and designed the study; I.V. prepared the pheromone baits and tested them to establish the release rate; M.-L.D. and G.I. dealt with fieldwork, collected all the data about the places where the traps were installed, and verified the three entomological collections; N.O. identified the beetles and M.K. verified the correctness of the identifications; N.O. wrote the paper and M.-L.D. prepared the maps. All co-authors assisted the lead author in writing and revising the manuscript.
Funding
This research was partly funded by Romanian Ministry of Research and Innovation grant number PN/2018-18040110 for N.O.; M.-L.D. was supported by a grant of the Romanian National Authority for Scientific Research and Innovation, CNCS/CCCDI—UEFISCDI, project number PN-III-P2-2. l-BG-2016-0376, within PNCDI III. M.K. was partly supported by the Ministry of Agriculture of the Czech Republic (Resolution RO0117; 6779/2017-MZE-14151).
Acknowledgments
We acknowledge the E-OBS dataset from the EU-FP6 project ENSEMBLES (http://ensembles-eu.metoffice.com) and the data providers in the ECA&D project (http://www.ecad.eu). We wish to express our thanks to the staff of the forest districts for assistance in fieldwork at different sites. We are grateful to the four anonymous reviewers whose comments and suggestions on a previous version of the manuscript have greatly contributed to the improvement of the work. Tudor Stăncioiu kindly revised the English.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
References
- Raffa, K.F.; Grégoire, J.-C.; Staffan Lindgren, B. Natural history and ecology of bark beetles. In Bark Beetles: Biology and Ecology of Native and Invasive Species; Vega, F.E., Hofstetter, R.W., Eds.; Elsevier: San Diego, CA, USA, 2015; pp. 1–40. [Google Scholar]
- Postner, M. Scolytidae (=Ipidae), Borkenkäfer. In Die Forstschädlinge Europas. 2. Band; Schwenke, W., Ed.; Paul Parey: Berlin, Germany; Hamburg, Germany, 1974; pp. 334–482. [Google Scholar]
- Moeck, H.A. Ethanol as the primary attractant for the ambrosia beetle Trypodendron lineatum (Coleoptera: Scolytidae). Can. Entomol. 1970, 102, 985–995. [Google Scholar] [CrossRef]
- Bauer, J.; Vité, J. Host selection by Trypodendron lineatum. Naturwissenschaften 1975, 62, 539. [Google Scholar] [CrossRef]
- Nijholt, W.; Shonherr, J. Chemical response behavior of scolytids in West Germany and Western Canada. Can. For. Serv. Bi-Mon. Res. Note 1976, 32, 31–32. [Google Scholar]
- Kohnle, U. Untersuchungen über die Pheromonsysteme sekundärer Borkenkäfer (Col., Scolytidae). Z. Angew. Entomol. 1985, 100, 197–218. [Google Scholar] [CrossRef]
- Klimetzek, D.; Vité, J.; König, E. Über das Verhalten mitteleuropäischer Trypodendron-Arten gegenüber natürlichen und synthetischen Lockstoffen. Allg. For. Jagd-Zeit. 1981, 152, 64–70. [Google Scholar]
- Schurig, V.; Weber, R.; Klimetzek, D.; Kohnle, U.; Mori, K. Enantiomeric composition of ‘lineatin’ in three sympatric ambrosia beetles. Naturwissenschaften 1982, 69, 602–603. [Google Scholar] [CrossRef]
- Vité, J.; Bakke, A. Synergism between chemical and physical stimuli in host colonization by an ambrosia beetle. Naturwissenschaften 1979, 66, 528–529. [Google Scholar] [CrossRef]
- Shore, T.L.; Lindgren, B.S. Effect of ethanol and α-pinene on response of ambrosia beetle, Trypodendron lineatum, to lineatin-baited funnel and drainpipe traps. J. Chem. Ecol. 1996, 22, 2187. [Google Scholar] [CrossRef] [PubMed]
- Byers, J.A. Attraction of bark beetles, Tomicus piniperda, Hylurgops palliatus, and Trypodendron domesticum and other insects to short-chain alcohols and monoterpenes. J. Chem. Ecol. 1992, 18, 2385–2402. [Google Scholar] [CrossRef] [PubMed]
- Kühnholz, S.; Borden, J.H.; Uzunovic, A. Secondary ambrosia beetles in apparently healthy trees: Adaptations, potential causes and suggested research. Integr. Pest Manag. Rev. 2001, 6, 209–219. [Google Scholar] [CrossRef]
- Borden, J.H. The striped ambrosia beetle. In Dynamics of Forest Insect Populations: Patterns, Causes, Implications; Berryman, A.A., Ed.; Springer: Boston, MA, USA, 1988; pp. 579–596. [Google Scholar]
- Smith, S.M.; Hulcr, J. Scolytus and other economically important bark and ambrosia beetles. In Bark Beetles: Biology and Ecology of Native and Invasive Species; Vega, F.E., Hofstetter, R.W., Eds.; Elsevier: San Diego, CA, USA, 2015; pp. 495–531. [Google Scholar]
- Knížek, M. Scolytinae. In Catalogue of Palearctic Coleoptera; Löbl, I., Smetana, A., Eds.; Apollo Books: Stenstrup, Danmark, 2011; Volume 7, p. 373. [Google Scholar]
- Pfeffer, A. Zentral- und Westpaläarktische Borken- und Kernkäfer: (Coleoptera: Scolytidae, Platypodidae); Pro Entomologia, c/o Naturhistorisches Museum: Basel, Switzerland, 1995; p. 310. [Google Scholar]
- Lindgren, B.S. Trypodendron lineatum (Olivier) (Coleoptera: Scolytidae) breeding in bigleaf maple, Acer mocrophyllum. J. Entomol. Soc. B. C. 1986, 83, 44. [Google Scholar]
- Strand, A. Seven new species of coleoptera from Norway. Nor. Entomol. Tidskr. 1946, 7, 168–172. [Google Scholar]
- Holzschuh, C. Ein neuer, gefährlicher Nutzholzborkenkäfer in Österreich. Forstsch.-Aktuell Wien 1990, 3, 2. [Google Scholar]
- Martikainen, P. Flight period and ecology of Trypodendron proximum (Niijima) (Col., Scolytidae) in Finland. J. Appl. Entomol. 2000, 124, 57–62. [Google Scholar] [CrossRef]
- Kvamme, T. Trypodendron piceum Strand (Col., Scolytidae): Flight period and response to synthetic pheromones. Fauna Nor. Ser. B 1988, 35, 65–70. [Google Scholar]
- Eggers, H. Japanische Borkenkäfer II. Arb. morph. taxon. Ent. Berlin-Dahlem 1939, 6, 114–123. [Google Scholar]
- Balachowsky, A.S. Faune de France 50. Coléoptères, Scolytides; Paul Lechevalier: Paris, France, 1949; p. 320. [Google Scholar]
- Stark, V.N. Zhestkokrylye, Koroedy. Fauna SSSR; Akademia Nauk SSSR: Moskva, Russia; Leningrad, Russia, 1952; p. 463. [Google Scholar]
- Nunberg, M. Klucze do Oznaczania Owadów Polski, Cz. Xix Chrząszcze—Coleoptera, Zeszyt 99–100, Korniki—Scolytidae, Wyrynniki—Platypodidae; Panstwowe Wydawnictwo Naukowe: Warszawa, Poland, 1954; p. 106. [Google Scholar]
- Pfeffer, A. Kůrovci—Scolytoidea. FAUNA Čsr, Svazek 6; ČSAV: Praha, Czech Republic, 1955; p. 324. [Google Scholar]
- Grüne, S. Handbuch zur Bestimmung der Europäischen Borkenkäfer: Brief Illustrated Key to European Bark Beetles; M. & H. Schaper: Hannover, Germany, 1979; p. 182. [Google Scholar]
- Schedl, K.E. 91. Familie: Scolytidae (Borken-und Ambrosiakäfer). In Die Käfer Mitteleuropas; Freude, H., Harde, K.W., Lohse, G.A., Eds.; Spektrum Akademischer Verlag: Berlin/Heidelberg, Germany, 1981; Volume 10, pp. 34–99. [Google Scholar]
- Annila, E.; Bakke, A.; Bejer-Petersen, B.; Lekander, B. Flight period and brood emergence in Trypodendron lineatum (Oliv.) (Coleoptera, Scolytidae) in the Nordic countries. Commun. Inst. For. Fenn. 1972, 76, 1–28. [Google Scholar]
- Lindelöw, Å. Aktuellt om svenska barkborrar (Coleoptera; Curculionidae, Scolytinae). Entomol. Tidskr. 2010, 131, 97–104. [Google Scholar]
- Pfeffer, A. Taxonomischer Status einer Arten der Gattung Xyloterus Erichson (Coleoptera, Scolytidae). Acta Entomol. Bohemoslov. 1989, 86, 129–136. [Google Scholar]
- Pfeffer, A. 91. Familie: Scolytidae. In Die Käfer Mitteleuropas; Lohse, G.A., Lucht, W.H., Eds.; Springer Spektrum: Berlin/Heidelberg, Germany, 1994; Volume 14. [Google Scholar]
- Wood, S.L. Nomenclatural changes and new species of Scolytidae (Coleoptera), Part IV. Great Basin Nat. 1989, 49, 167–185. [Google Scholar]
- Mandelshtam, M.Y.; Popovichev, B.G. Annotated list of bark beetles (Coleoptera, Scolytidae) of Leningrad province. Ėntomol. Obozr. 2000, 79, 599–618. [Google Scholar]
- Holzschuh, C. Ergebnisse von Untersuchungen über die Einschleppung von Borkenkäfern an Holzlager- und Umschlagplätzen. Forstsch.-Aktuell Wien 1990, 5, 7–8. [Google Scholar]
- Holzschuh, C. Forstschädlinge, die in den letzten fünfzig Jahren in Österreich eingewandert sind oder eingeschleppt wurden. Stapfia Lienz 1995, 37, 129–141. [Google Scholar]
- Krehan, H.; Holzschuh, C. Trypodendron laeve—Vorkommen in Österreich. Forstsch.-Aktuell Wien 1999, 23/24, 6–8. [Google Scholar]
- Kenis, M. Insects—Insecta. In Invasive Alien Species in Switzerland—An Inventory of Alien Species and Their Threat to Biodiversity and Economy in Switzerland; Wittenberg, R., Kenis, M., Blick, T., Hänggi, A., Gassmann, A., Weber, E., Eds.; Federal Office for the Environment (FOEN): Bern, Switzerland, 2006; pp. 71–100. [Google Scholar]
- DAISE. Handbook of Alien Species in Europe; Springer: Berlin/Heidelberg, Germany, 2009; p. 399. [Google Scholar]
- Köhler, F.; Klausnitzer, B. Verzeichnis der Käfer Deutschlands. Ent. Nachr. Ber. (Dresden) Beiheft 1998, 4, 1–185. [Google Scholar]
- Zelený, J.; Doležal, P. Kůrovcoviti brouci (Scolytidae, Coleoptera) na smrku na Šumavě. Akt. Šumav. Výzkumu 2004, 2, 221–223. [Google Scholar]
- Zelený, J. Nejčastější kůrovcovití na smrku na Šumavě. Lesn. Práce 2000, 80, 258–259. [Google Scholar]
- Olenici, N.; Knížek, M.; Olenici, V.; Duduman, M.-L.; Biriş, I.-A. First report of three scolytid species (Coleoptera: Curculionidae, Scolytinae) in Romania. Ann. For. Res. 2014, 57, 87. [Google Scholar] [CrossRef]
- Sweeney, J.D.; Silk, P.; Grebennikov, V.; Mandelshtam, M. Efficacy of semiochemical-baited traps for detection of Scolytinae species (Coleoptera: Curculionidae) in the Russian Far East. Eur. J. Entomol. 2016, 113, 84–97. [Google Scholar] [CrossRef]
- Lekander, B.; Bejer-Petersen, B.; Kangas, E.; Bakke, A. The distribution of bark beetles in the Nordic countries. Acta Entomol. Fenn. 1977, 32, 1–37. [Google Scholar]
- Muona, J. Four species of beetles new to Finland. Not. Entomol. 1989, 69, 195–197. [Google Scholar]
- Muona, J. Tarkennuksia eräiden kuoriaislajien esiintymiseen Suomessa ja Venäjän Karjalassa (Coleoptera). Sahlbergia 1994, 1, 7–10. [Google Scholar]
- Martikainen, P.; Siitonen, J.; Kaila, L.; Punttila, P. Intensity of forest management and bark beetles in non-epidemic conditions: A comparison between Finnish and Russian Karelia. J. Appl. Entomol. 1996, 120, 257–264. [Google Scholar] [CrossRef]
- Martikainen, P.; Siitonen, J.; Kaila, L.; Punttila, P.; Rauh, J. Bark beetles (Coleoptera, Scolytidae) and associated beetle species in mature managed and old-growth boreal forests in southern Finland. For. Ecol. Manag. 1999, 116, 233–245. [Google Scholar] [CrossRef]
- Voolma, K.; Mandelshtam, M.J.; Shcherbakov, A.N.; Yakovlev, E.B.; Õunap, H.; Süda, I.; Popovichev, B.G.; Sharapa, T.V.; Galasjeva, T.V.; Khairetdinov, R.R. Distribution and spread of bark beetles (Coleoptera: Scolytidae) around the gulf of Finland: A comparative study with notes on rare species of Estonia, Finland and North-Western Russia. Entomol. Fenn. 2004, 15, 198–210. [Google Scholar]
- Öhrn, P.; Lindelöw, Å.; Långström, B. Flight activity of the ambrosia beetles Trypodendron laeve and Trypodendron lineatum in relation to temperature in southern Sweden. In Biotic Risks and Climate Change in Forests; Delb, H., Pontuali, S., Eds.; Berichte Freiburger Forstliche Forschung, Forest Research Institute of Baden–Württemberg: Freiburg, Germany, 2011; Volume 89, pp. 86–90. [Google Scholar]
- Lukášová, K.; Knížek, M.; Holuša, J.; Čejka, M.; Kacprzyk, M. Is the bark beetle Trypodendron laeve (Coleoptera: Curculionidae: Scolytinae) an alien pest in the Czech Republic and Poland? Pol. J. Ecol. 2012, 60, 789–795. [Google Scholar]
- Witkowski, R.; Załuska, M.T.; Buchholz, L.; Mazur, A. Nowe dane o występowaniu Trypodendron laeve Eggers, 1939 (Coleoptera, Curculionidae, Scolytinae) w Polsce. Acta Sci. Pol. Silv. Colendar. Rat. Ind. Lignar. 2015, 14, 81–86. [Google Scholar]
- Haylock, M.R.; Hofstra, N.; Klein Tank, A.M.G.; Klok, E.J.; Jones, P.D.; New, M. A European daily high-resolution gridded data set of surface temperature and precipitation for 1950–2006. J. Geophys. Res. Atmos. 2008, 113. [Google Scholar] [CrossRef]
- Micu, D.M.; Dumitrescu, A.; Cheval, S.; Birsan, M.-V. Climate of the Romanian Carpathians: Variability and Trends; Springer: London, UK, 2014; p. 213. [Google Scholar]
- Vasiliu, M.; Zaharia, D.; Ignat, C. Catalogul scolitidelor din colecţia “Ştefan Negru” a Muzeului Judeţean Suceava (Coleoptera, Scloytoidea). In Studii şi Comunicări, Ştiinţele Naturii; Muzeul Judeţean: Suceava, Romania, 1978; pp. 37–58. [Google Scholar]
- Negru, Ș. Les Scolytoides (Coleoptera, Scolytoidea) de la collection scientifique du Musée Brukenthal—Sibiu. Trav. Mus. d’Hist. Nat. Grigore Antipa Buchar. 1966, 6, 397–405. [Google Scholar]
- Borden, J.; Fockler, C. Emergence and orientation behavior of brood Trypodendron lineatum (Coleoptera: Scolytidae). J. Entomol. Soc. B. C. 1973, 70, 34–38. [Google Scholar]
- Zar, J.H. Biostatistical Analysis, 5th ed.; Pearson Prentice Hall: Upper Saddle River, NJ, USA, 2010. [Google Scholar]
- Park, J.; Reid, M.L. Distribution of a bark beetle, Trypodendron lineatum, in a harvested landscape. For. Ecol. Manag. 2007, 242, 236–242. [Google Scholar] [CrossRef]
- Lukášová, K.; Holuša, J. Comparison of Trypodendron lineatum, T. domesticum and T. laeve (Coleoptera: Curculionidae) flight activity in Central Europe. J. For. Sci. 2014, 60, 382–387. [Google Scholar] [CrossRef]
- Bussler, H.; Schmidt, O. Remarks on the taxonomy, distribution and ecology of Trypodendron laeve Eggers, 1939 (Coleoptera: Scolytidae). Nachrichtenbl. Bayer. Entomol. 2008, 57, 62–65. [Google Scholar]
- Klimetzek, D.; Vité, J.P. Tierische Schädlinge. In Die Fichte; Schmidt-Vogt, H., Ed.; Krankheiten, Schäden, Fichtensterben; Paul Parey: Berlin, Germany, 1989; Band II/2; pp. 40–133. [Google Scholar]
- Salom, S.M.; McLean, J.A. Flight behavior of scolytid beetle in response to semiochemicals at different wind speeds. J. Chem. Ecol. 1991, 17, 647–661. [Google Scholar] [CrossRef] [PubMed]
- Salom, S.M.; McLean, J.A. Environmental influences on dispersal of Trypodendron lineatum (Coleoptera: Scolytidae). Environ. Entomol. 1991, 20, 565–576. [Google Scholar] [CrossRef]
- Petri, K. Siebenbürgens Käferfauna auf Grund ihrer Erforschung bis zum Jahre 1911; Drotleff: Hermannstadt, Romania, 1912; p. 376. [Google Scholar]
- Merkl, O. Data to the knowledge on the beetle fauna of Maramures, Romania (Coleoptera). Stud. Univ. Vasile Goldis Ser. Stiintele Vietii. 2008, 18, 243–311. [Google Scholar]
- Kocs, I. A Magyar Természettudományi Múzeum Bogárgyűjteményében Található, Székelyföldön gyűjtött ormányosalkatú bogarak fajlistája (Coleoptera: Curculionoidea). Acta Siculica 2010, 2010, 105–121. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).