Robust Model Predicts Shoot Phenology of Fraser Fir under Extreme Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Phase I: Development of Phenology Model
2.2. Phase II: Mapping of Lateral Shoot Phenology to Model
3. Results
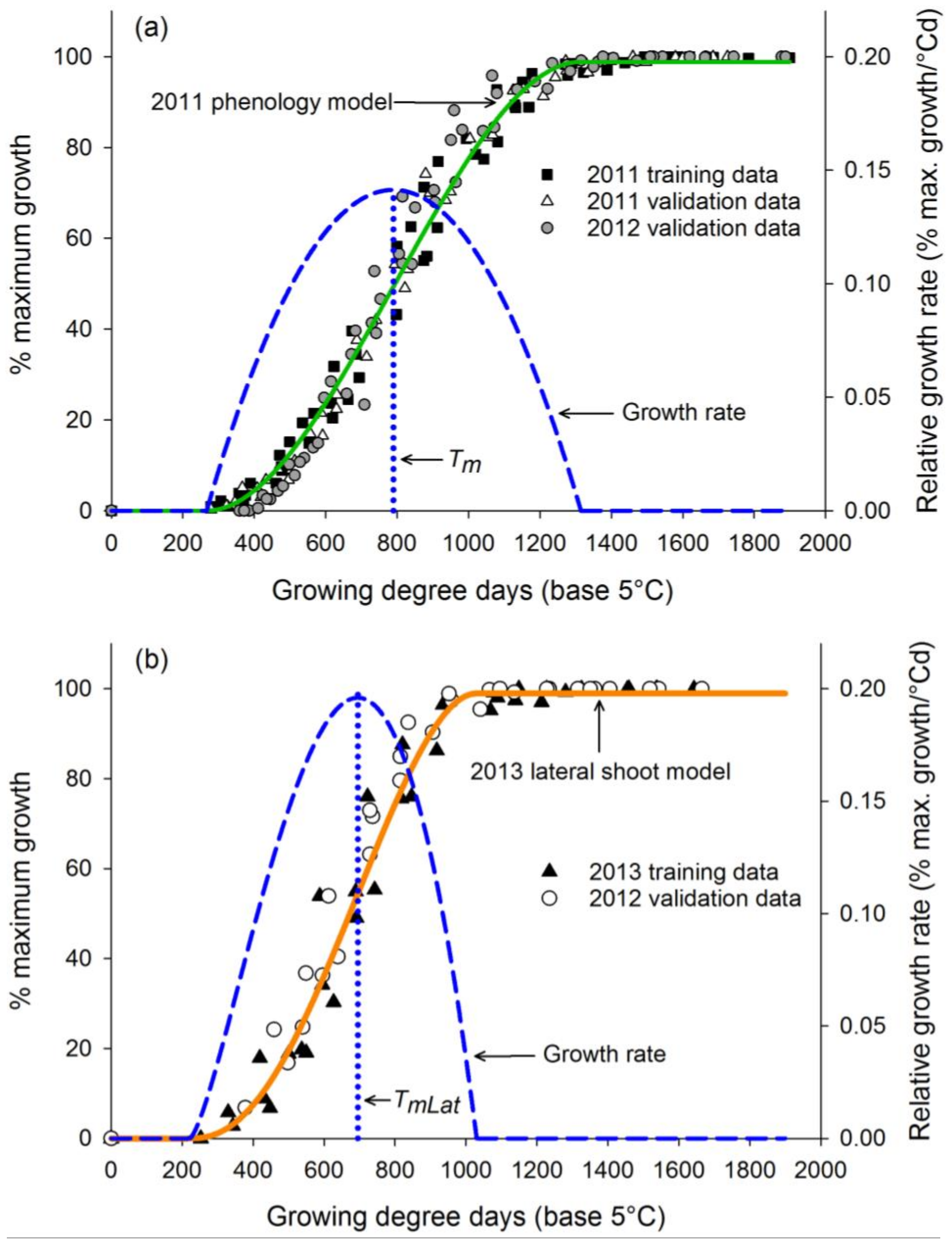
3.1. Phase I: Development of Phenology Model
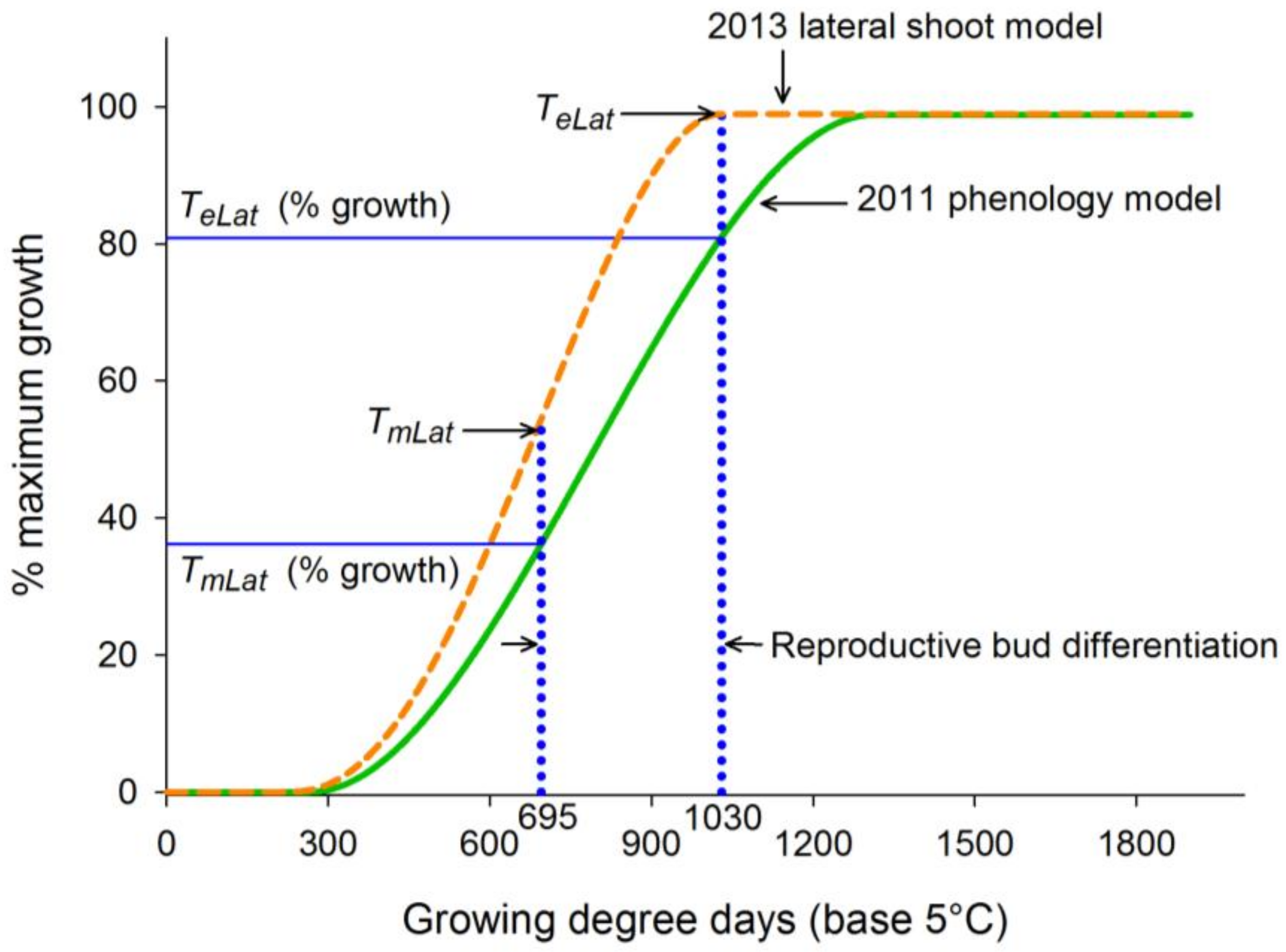
3.2. Phase II: Mapping of Lateral Shoot Phenology to Model
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pettersson, J.M.; Frampton, J.; Ronnberg, J.; Shew, D.; Benson, D.M.; Cuberta, M.A. Increased number of Phytophthora species found in Fraser fir Christmas tree plantations in the southern Appalachians. In Proceedings of the 12th International Christmas Tree Research and Extension Conference; Talgo, V., Floistad, I., Eds.; Norwegian Institute of Boieconomy Research (NIBO): Biri, Norway, 2015; p. 27. [Google Scholar]
- Reinhardt, K.; Smith, W.K. Leaf gas exchange of understory spruce-fir saplings in relict cloud forests, southern Appalachian Mountains, USA. Tree Physiol. 2008, 28, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Cory, S.T.; Wood, L.K.; Neufeld, H.S. Phenology and growth responses of Fraser fir (Abies fraseri) Christmas trees along an elevational gradient, southern Appalachian Mountains, USA. Agric. For. Meteorol. 2017, 243, 25–32. [Google Scholar] [CrossRef]
- Melillo, J.M.; Richmond, T.C.; Yohe, G.W. (Eds.) Climate Change Impacts in the United States: The Third National Climate Assessment; U.S. Global Change Research Program, Government Printing Office: Washington, DC, USA, 2014.
- Fitchett, J.M.; Grab, S.W.; Thompson, D.I. Plant phenology and climate change: Progress in methodological approaches and application. Prog. Phys. Geogr. 2015, 39, 460–482. [Google Scholar] [CrossRef]
- Mallozzi, S.D. Integrated Pest Management Field Guide For Christmas Trees: Douglas Fir, True Firs, Spruce; Cornell University Cooperative Extension, Dutchess County: Millbrook, NY, USA, 2007. [Google Scholar]
- Fondren, K.M.; McCullough, D.G. Phenology and density of balsam twig aphid, Mindarus abietinus Koch (Homoptera: Aphididae) in relation to bud break, shoot damage, and value of fir Christmas trees. J. Econ. Entomol. 2003, 96, 1760–1769. [Google Scholar] [CrossRef] [PubMed]
- Doherty, J.-F.; Guay, J.-F.; Cloutier, C. Temperature-manipulated dynamics and phenology of Mindarus abietinus (Hemiptera: Aphididae) in commercial Christmas tree plantations in Québec, Canada. Can. Entomol. 2017, 149, 801–812. [Google Scholar] [CrossRef][Green Version]
- Sidebottom, J.L. The 2001 Pest Management Survey; North Carolina Cooperative Extension Service: Raleigh, NC, USA, 2001. [Google Scholar]
- Owen, J. Calibrating a Backpack Sprayer for Chemical Mowing; CropLife Foundation: Washington, DC, USA, 2005. [Google Scholar]
- Hinesley, L.E.; Derby, S.A. Growth of Fraser fir Christmas trees in response to annual shearing. HortScience 2004, 39, 1644–1646. [Google Scholar]
- Cannell, M.G.R.; Smith, R.I. Climatic warming, spring budburst and forest damage on trees. J. Appl. Ecol. 1986, 23, 177–191. [Google Scholar] [CrossRef]
- Vitasse, Y.; Lenz, A.; Körner, C. The interaction between freezing tolerance and phenology in temperate deciduous trees. Front. Plant Sci. 2014, 5, 541. [Google Scholar] [CrossRef] [PubMed]
- Owens, J.N.; Blake, M.D. Forest Tree Seed Production. A Review of the Literature and Recommendations for Future Research; Petawawa National Forestry Institute, Canadian Forestry Service, Agricultural Canada: Chalk River, ON, Canada, 1985; Volume PI-X-53.
- Owens, J. Bud development in grand fir (Abies grandis). Can. J. For. Res. 1984, 14, 575–588. [Google Scholar] [CrossRef]
- Owens, J.N.; Molder, M. Vegetative bud development and cone differentiation in Abies amabilis. Can. J. Bot. 1977, 55, 992–1008. [Google Scholar] [CrossRef]
- Owens, J.; Singh, H. Vegetative bud development and the time and method of cone initiation in subalpine fir (Abies lasiocarpa). Can. J. Bot. 1982, 60, 2249–2262. [Google Scholar] [CrossRef]
- Arista, M.; Talavera, S. Phenology and anatomy of the reproductive phase of Abies pinsapo Boiss. (Pinaceae). Bot. J. Linn. Soc. 1994, 116, 223–234. [Google Scholar] [CrossRef]
- Pharis, R.P.; Kuo, C.G. Physiology of gibberellins in conifers. Can. J. For. Res. 1977, 7, 299–325. [Google Scholar] [CrossRef]
- Ross, S.; Pharis, R. Promotion of flowering in tree crops: Different mechanisms and techniques, with special reference to conifers. In Attributes of Trees as Crop Plants; Cannell, M.G.R., Jackson, J.E., Eds.; Institute of Terrestrial Ecology, Natural Envrionment Research Council: Huntingdon, UK, 1985; pp. 383–389. ISBN 0-904282-83-X. [Google Scholar]
- Philippe, G.; Lee, S.; Schute, G.; Heois, B. Flower stimulation is cost-effective in Douglas-fir seed orchards. Forestry 2004, 77, 279–286. [Google Scholar] [CrossRef]
- Solberg, S. Summer drought: A driver for crown condition and mortality of Norway spruce in Norway. For. Pathol. 2004, 34, 93–104. [Google Scholar] [CrossRef]
- Smith, R.; Greenwood, M. Effects of cone-induction treatments on black spruce (Picea mariana) current-year needle development and gas exchange properties. Tree Physiol. 1997, 17, 407–414. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Johnsen, O.; Haug, G.; Daehlen, O. Effects of heat treatment, timing of heat treatment, and gibberellin A4/7 on flowering in potted Picea abies grafts. Scand. J. For. Res. 1994, 9, 333–340. [Google Scholar] [CrossRef]
- Cregg, B.M.; O’Donnell, J.; Koelling, M.R. Precocious cone production in Fraser fir. In Proceedings of the 6th International Christmas Tree Research and Extension Conference, Hendersonville, NC, USA, 14–19 September 2003; pp. 98–105. [Google Scholar]
- Owen, J. Using herbicides to interrupt cone development on Fraser fir. In Proceedings of the 12th International Christmas Tree Research and Extension Conference; Talgo, V., Floistad, I.S., Eds.; Norwegian Institute of Boieconomy Research (NIBO): Biri, Norway, 2015; p. 41. [Google Scholar]
- Powell, G.R. Biennial strobilus production in balsam fir: A review of its morphogenesis and a discussion of its apparent physiological basis. Can. J. For. Res 1977, 7, 547–555. [Google Scholar] [CrossRef]
- Sala, A.; Hopping, K.; McIntire, E.J.B.; Delzon, S.; Crone, E.E. Masting in whitebark pine (Pinus albicaulis) depletes stored nutrients. New Phytol. 2012, 196, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Crain, B.A.; Cregg, B.M. Gibberellic acid inhibitors control height growth and cone production in Abies fraseri. Scand. J. For. Res. 2017, 32, 391–396. [Google Scholar] [CrossRef]
- Crain, B.A.; Cregg, B.M. Using irrigation and mulch to control cone production in Fraser fir (Abies fraseri). Scand. J. For. Res. 2017, 32, 384–390. [Google Scholar] [CrossRef]
- Kramer, K.; Leinonen, I.; Loustau, D. The importance of phenology for the evaluation of impact of climate change on growth of boreal, temperate and Mediterranean forests ecosystems: An overview. Int. J. Biometeorol. 2000, 44, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Owens, J.N.; Molder, M.; Langer, H. Bud development in Picea glauca. I. Annual growth cycle of vegetative buds and shoot elongation as they relate to date and temperature sums. Can. J. Bot. 1977, 55, 2728–2745. [Google Scholar] [CrossRef]
- Trudgill, D.L.; Honek, A.; Li, D.; van Straalen, N.M. Thermal time—Concepts and utility. Ann. Appl. Biol. 2005, 146, 1–14. [Google Scholar] [CrossRef]
- USDA NRCS Web Soil Survey. Available online: http://websoilsurvey.nrcs.usda.gov/ (accessed on 26 February 2016).
- NOAA 1981–2010 U.S. Climate Normals. Available online: http://weatherdb.com/ (accessed on 26 February 2016).
- Yin, X.; Goudriaan, J.; Lantinga, E.A.; Vos, J.; Spiertz, H.J. A flexible sigmoid function of determinate growth. Ann. Bot. 2003, 91, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Shi, P.-J.; Chen, L.; Hui, C.; Grissino-Mayer, H.D. Capture the time when plants reach their maximum body size by using the beta sigmoid growth equation. Ecol. Modell. 2016, 320, 177–181. [Google Scholar] [CrossRef]
- Auzanneau, J.; Huyghe, C.; Escobar-Gutiérrez, A.J.; Julier, B.; Gastal, F.; Barre, P. Association study between the gibberellic acid insensitive gene and leaf length in a Lolium perenne L. synthetic variety. BMC Plant Biol. 2011, 11, 183. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Gramatica, P. On the development and validation of QSAR models. In Computational Toxicology; Reisfeld, B., Mayeno, A., Eds.; Springer: New York, NY, USA, 2013; Volume 930, pp. 499–526. [Google Scholar]
- Frank, I.E.; Todeschini, R. The Data Analysis Handbook; Elsevier Science B.V.: Amsterdam, The Netherlands, 1994; ISBN 008086841X. [Google Scholar]
- NOAA National Temperature and Precipitation Maps. Available online: https://www.ncdc.noaa.gov/temp-and-precip/us-maps/6/201202?products[]=statewidetavgrank#us-maps-select (accessed on 27 March 2018).
- Pryor, S.C.; Scavia, D.; Downer, C.; Gaden, M.; Iverson, L.; Nordstrom, R.; Patz, J.; Robertson, G.P. Ch. 18: Mid-west. In Climate Change Impacts in the United States: The Third National Climate Assessment; Melillo, J.M., Richmond, T.C., Yohe, G.W., Eds.; U.S. Global Change Research Program, Government Printing Office: Washington, DC, USA, 2014; pp. 418–440. [Google Scholar]
- Enviroweather Fraser fir Growth Model. Available online: http://enviroweather.msu.edu/run.php?mod=c_ffg (accessed on 5 March 2016).
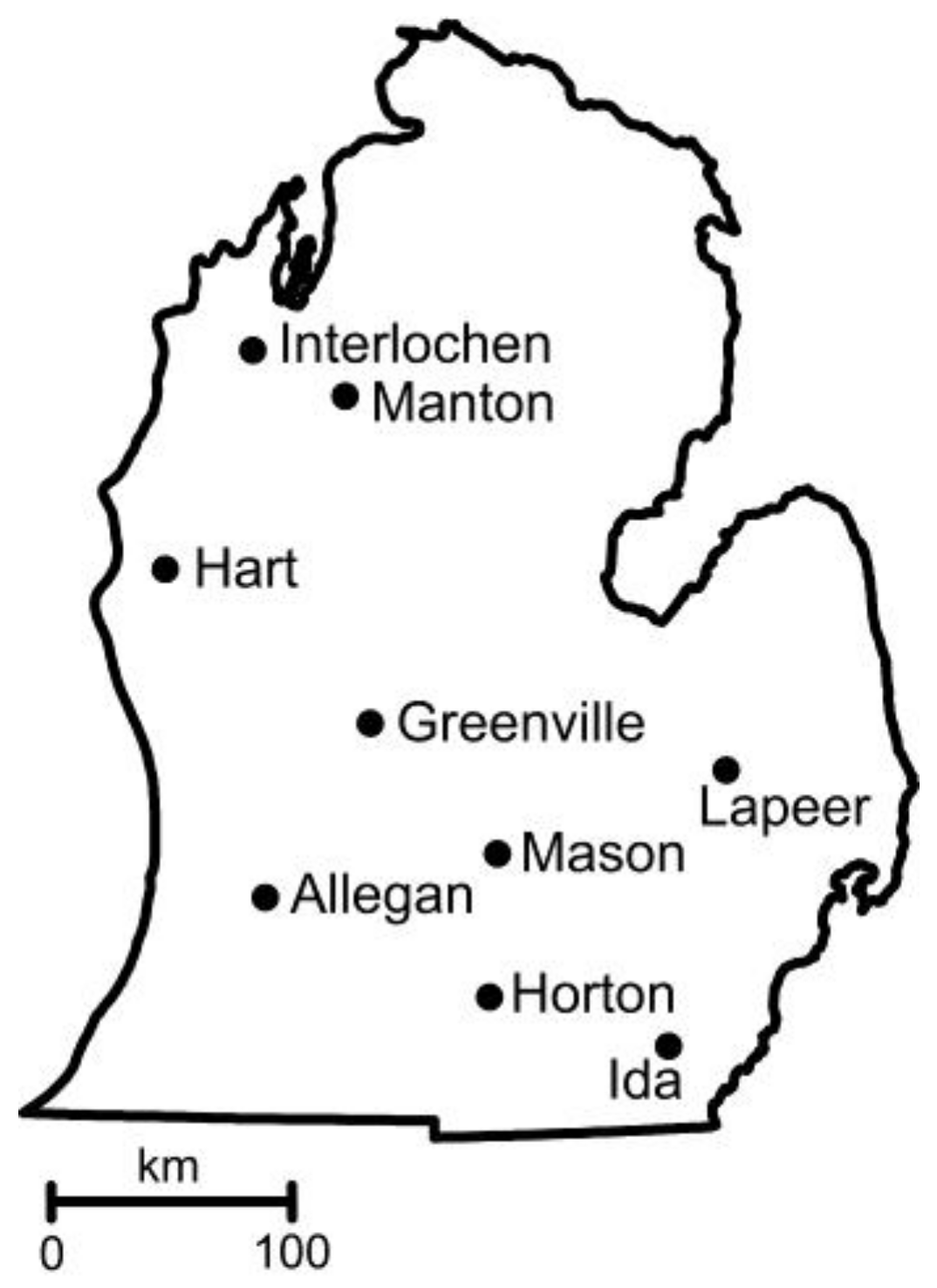
| Coordinates (Decimal Degrees) | Weather Station a | Soil b | Temp. c (June) | Precip. d (June) | |||
|---|---|---|---|---|---|---|---|
| City | Latitude | Longitude | (km) | Type (slope, %) | pH | (°C) | (cm) |
| Allegan | 42.5475 | −85.7796 | 30.32 | Ockley loam (1–6) | 5.8 | 11.4 (20.2) | 92.7 (10.7) |
| Greenville | 43.1806 | −85.1394 | 17.72 | Tekenink-Spinks loamy sands (6–12) | 5.7 | 10.9 (20.2) | 98.6 (9.4) |
| Hart † | 43.7391 | −86.2144 | 11.7 | Spinks loamy fine sand (6–12) | 6.2 | 9.2 (18.0) | 88.1 (8.1) |
| Horton † | 42.0760 | −84.4830 | 31.7 | Hillsdale-Riddles sandy loams (6–12) | 6.2 | 9.1 (19.7) | 203.2 (21.1) |
| Ida † | 41.8846 | −83.6237 | 8.2 | Oakville fine sand (0–6) | 6.5 | 10.5 (20.3) | 89.7 (9.1) |
| Interlochen | 44.5605 | −85.7194 | 35.53 | Karlin loamy sand (6–12) | 5.8 | 8.9 (17.7) | 84.1 (8.1) |
| Lapeer | 42.9964 | −83.3133 | 8.9 | Miami loam (2–6) | 6.1 | 9.9 (19.3) | 82.6 (8.6) |
| Manton † | 44.3986 | −85.2875 | 0.5 | Montcalm-Graycalm complex (0–6) | 5.8 | 9.0 (18.4) | 87.4 (8.9) |
| Mason † | 42.6661 | −84.4482 | 3.4 | Capac loam (0–4) | 6.6 | 10.4 (19.8) | 81.8 (8.9) |
| SDEC or SDEP a | Fitted Parameters b | ||||||
|---|---|---|---|---|---|---|---|
| n | R2 | Cm | Tb | Te | Tm | ||
| 2011 phenology model training | 57 | 0.9889 | 0.0398 | 0.1414 | 266.42 | 1314.78 | 789.28 |
| 2011 validation (spatial) | 44 | 0.9930 | 0.0323 | ||||
| 2012 validation (temporal) | 73 | 0.9775 | 0.0575 | ||||
| 2013 lateral shoot model training | 36 | 0.9735 | 0.0601 | 0.1960 | 221.28 | 1030.11 | 694.83 |
| 2012 validation (temporal) | 30 | 0.9663 | 0.0556 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crain, B.A.; Nzokou, P.; O’Donnell, J.; Bishop, B.; Cregg, B.M. Robust Model Predicts Shoot Phenology of Fraser Fir under Extreme Conditions. Forests 2018, 9, 193. https://doi.org/10.3390/f9040193
Crain BA, Nzokou P, O’Donnell J, Bishop B, Cregg BM. Robust Model Predicts Shoot Phenology of Fraser Fir under Extreme Conditions. Forests. 2018; 9(4):193. https://doi.org/10.3390/f9040193
Chicago/Turabian StyleCrain, Brent A., Pascal Nzokou, Jill O’Donnell, Beth Bishop, and Bert M. Cregg. 2018. "Robust Model Predicts Shoot Phenology of Fraser Fir under Extreme Conditions" Forests 9, no. 4: 193. https://doi.org/10.3390/f9040193
APA StyleCrain, B. A., Nzokou, P., O’Donnell, J., Bishop, B., & Cregg, B. M. (2018). Robust Model Predicts Shoot Phenology of Fraser Fir under Extreme Conditions. Forests, 9(4), 193. https://doi.org/10.3390/f9040193





