Species-Rich National Forests Experience More Intense Human Modification, but Why?
Abstract
1. Introduction
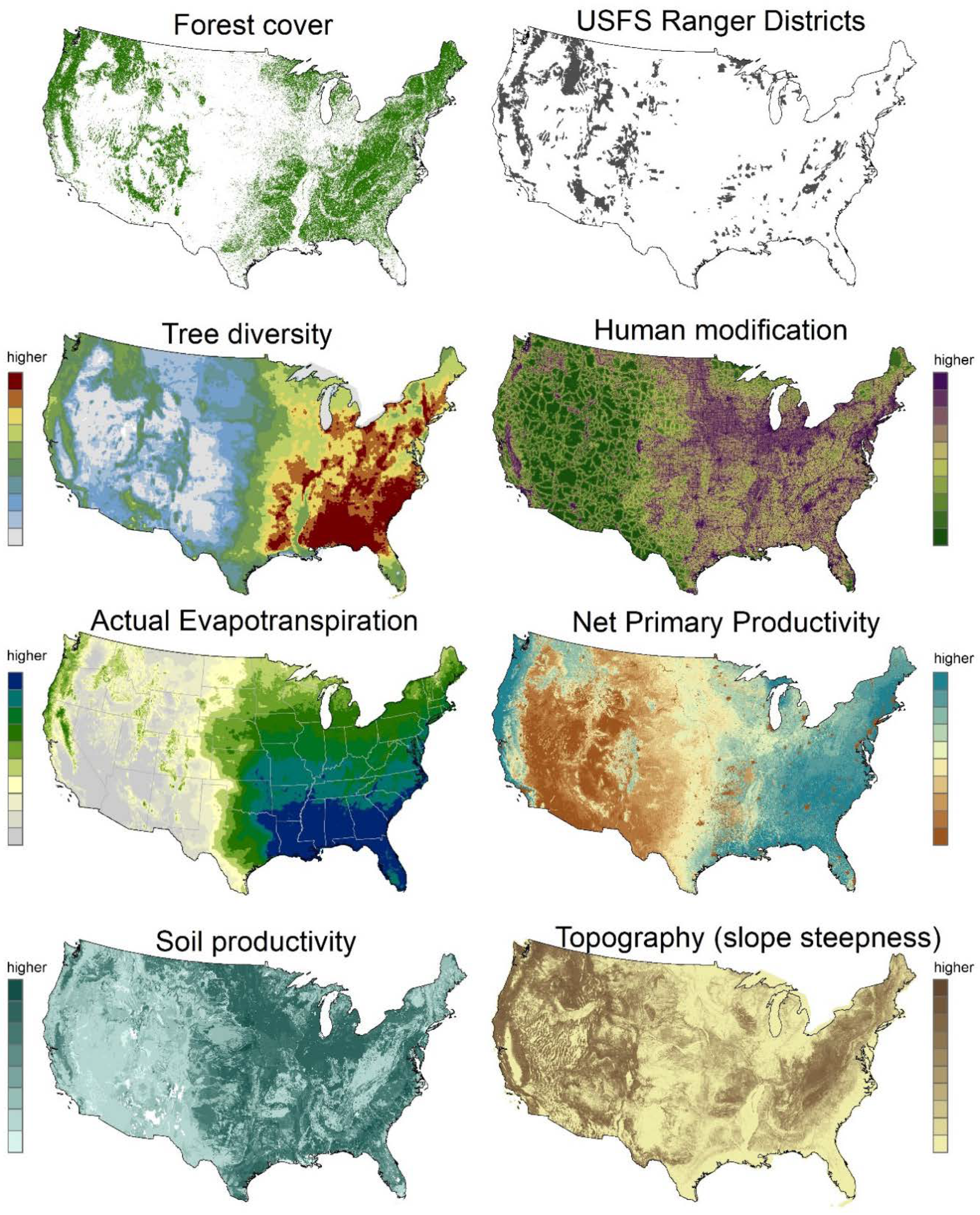
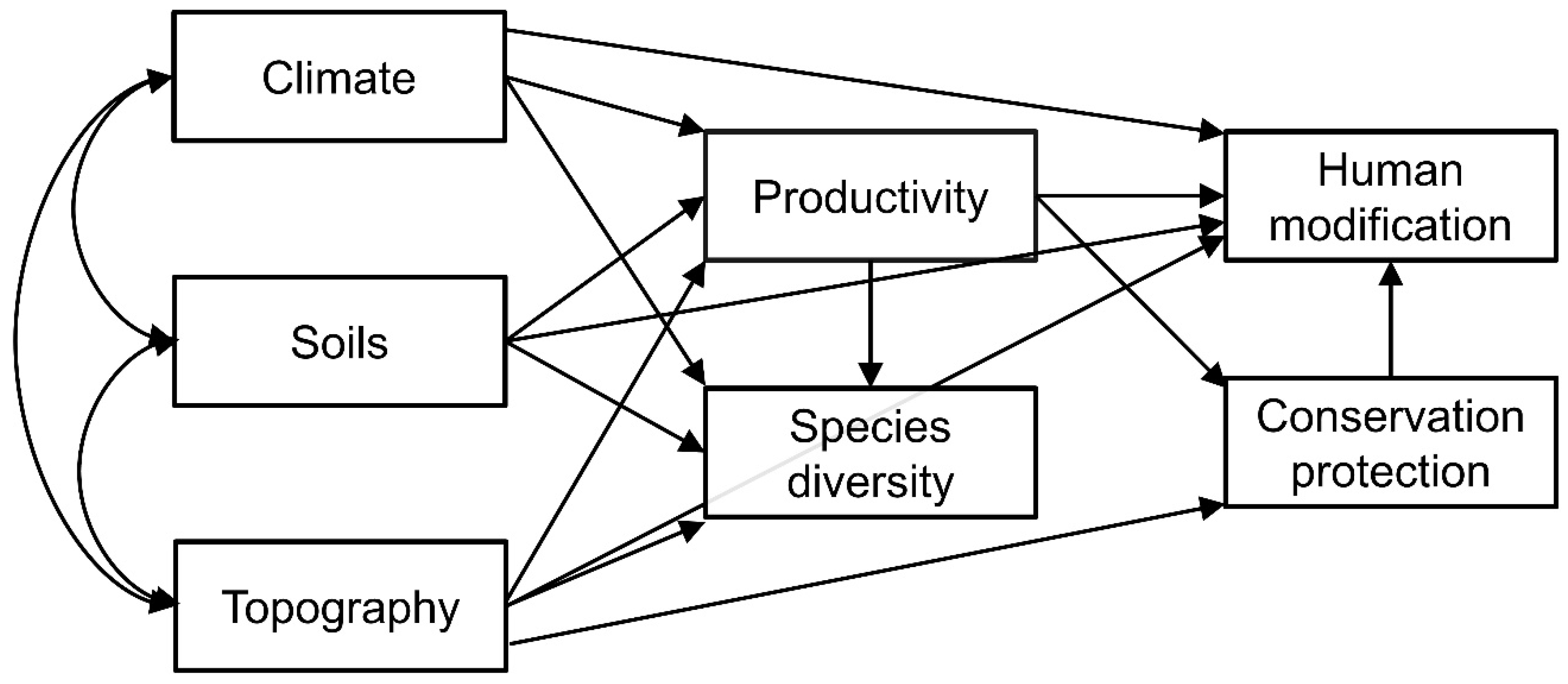
2. Materials and Methods
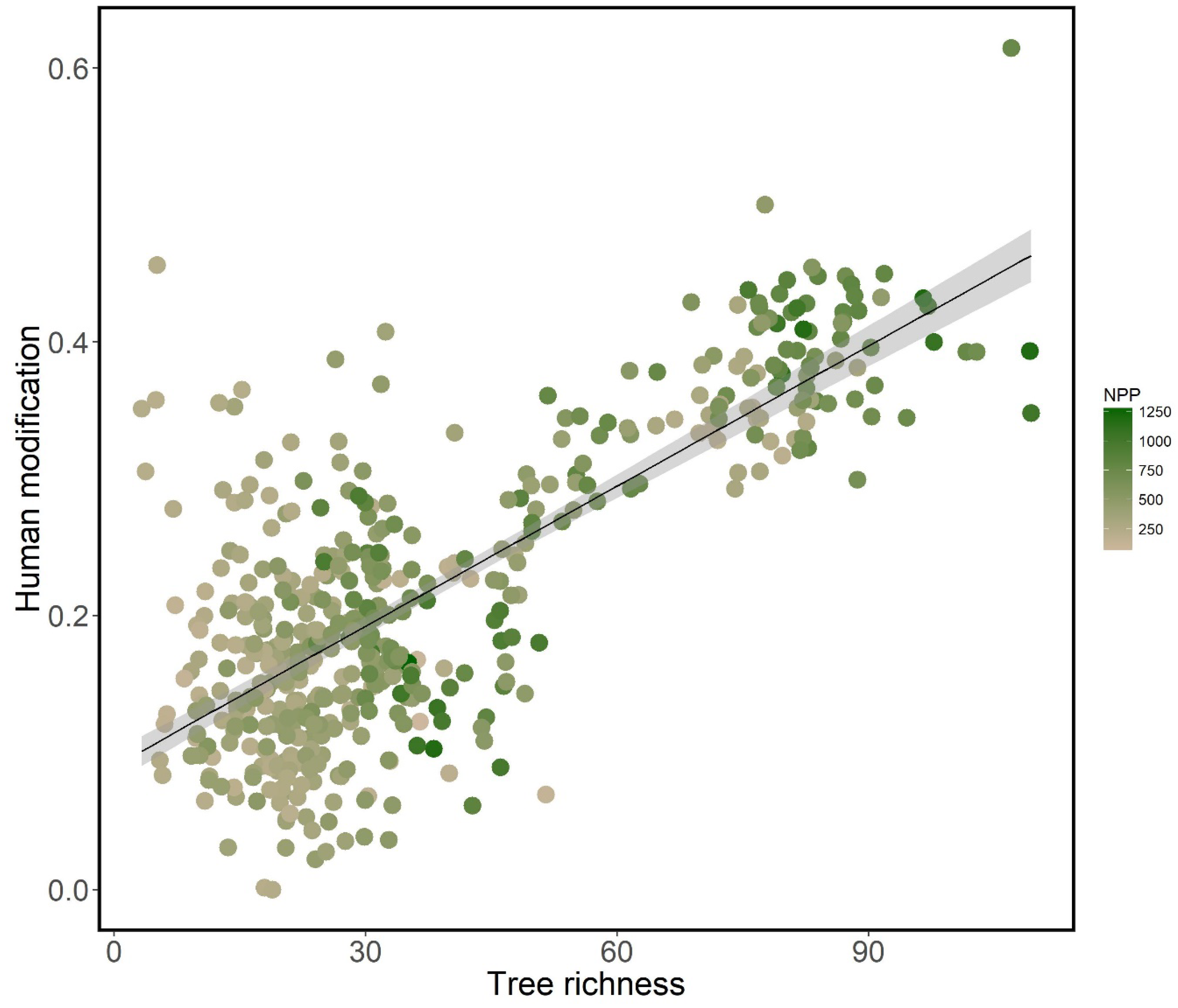
3. Results
4. Discussion
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Fine, P.V.A.; Ree, R.H. Evidence for a time-integrated species-area effect on the latitudinal gradient in tree diversity. Am. Nat. 2006, 168, 796–804. [Google Scholar] [CrossRef]
- Keil, P.; Chase, J. Integrating global patterns and drivers of tree diversity across a continuum of spatial grains. bioRxiv 2018. [Google Scholar] [CrossRef]
- Anderegg, W.R.L.; Konings, A.G.; Trugman, A.T.; Yu, K.; Bowling, D.R.; Gabbitas, R.; Karp, D.S.; Pacala, S.; Sperry, J.S.; Sulman, B.N.; et al. Hydraulic diversity of forests regulates ecosystem resilience during drought. Nature 2018, 561, 538–541. [Google Scholar] [CrossRef] [PubMed]
- Naeem, S.; Wright, J.P. Disentangling biodiversity effects on ecosystem functioning: Deriving solutions to a seemingly insurmountable problem. Ecol. Lett. 2003, 6, 567–579. [Google Scholar] [CrossRef]
- Loreau, M.; Naeem, S.; Inchausti, P.; Bengtsson, J.; Grime, J.P.; Hector, A.; Hooper, D.U.; Huston, M.A.; Raffaelli, D.; Schmid, B.; et al. Biodiversity and ecosystem functioning: Current knowledge and future challenges. Science 2001, 294, 804–808. [Google Scholar] [CrossRef] [PubMed]
- Haddad, N.M.; Brudvig, L.A.; Clobert, J.; Davies, K.F.; Gonzalez, A.; Holt, R.D.; Lovejoy, T.E.; Sexton, J.O.; Austin, M.P.; Collins, C.D.; et al. Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci. Adv. 2015, 1, e1500052. [Google Scholar] [CrossRef]
- Venter, O.; Sanderson, E.W.; Magrach, A.; Allan, J.R.; Beher, J.; Jones, K.R.; Possingham, H.P.; Laurance, W.F.; Wood, P.; Fekete, B.M.; et al. Sixteen years of change in the global terrestrial human footprint and implications for biodiversity conservation. Nat. Commun. 2016, 7, 1–11. [Google Scholar] [CrossRef]
- Sanderson, E.W.; Jaiteh, M.; Levy, M.; Redford, K.H.; Wannebo, A.V.; Woolmer, G. The human footprint and the last of the wild. Bioscience 2002, 52, 891–904. [Google Scholar] [CrossRef]
- Aplet, G.; Thomson, J.; Wilbert, M. Indicators of wildness: Using attributes of the land to assess the context of wilderness. In Proceedings of the Wilderness Science in a Time of Change, Missoula, MT, USA, 23–27 May 1999; McCool, S.F., Cole, D.N., Borrie, W.T., O’Laughlin, J., Eds.; 2000; RMRS-P-15, pp. 89–98. [Google Scholar]
- Watson, J.E.M.; Shanahan, D.F.; Marco, M.D.; Allan, J.; Laurance, W.F.; Sanderson, E.W.; Mackey, B.; Venter, O. Catastrophic declines in wilderness areas undermine global environment targets. Curr. Biol. 2016, 26, 1–6. [Google Scholar] [CrossRef]
- Pimm, S.L.; Jenkins, C.N.; Abell, R.; Brooks, T.M.; Gittleman, J.L.; Joppa, L.N.; Raven, P.H.; Roberts, C.M.; Sexton, J.O. The biodiversity of species and their rates of extinction, distribution, and protection. Science 2014, 344, 1246752. [Google Scholar] [CrossRef]
- Wilcove, D.S.; Rothstein, D.; Dubow, J.; Phillips, A.; Losos, E. Quantifying threats to imperiled species in the United States. Bioscience 1998, 48, 607–615. [Google Scholar] [CrossRef]
- Huston, M. Biological and Diversity, Economics, and Soils. Science 1993, 262, 1676–1680. [Google Scholar] [CrossRef] [PubMed]
- Huston, M.A.; Wolverton, S. The global distribution of net primary production: Resolving the paradox. Ecol. Monogr. 2009, 79, 343–377. [Google Scholar] [CrossRef]
- Belote, R.T.; Prisley, S.; Jones, R.H.; Fitzpatrick, M.; de Beurs, K. Forest productivity and tree diversity relationships depend on ecological context within mid-Atlantic and Appalachian forests (USA). For. Ecol. Manag. 2011, 261, 1315–1324. [Google Scholar] [CrossRef]
- Jenkins, C.N.; Pimm, S.L.; Joppa, L.N. Global patterns of terrestrial vertebrate diversity and conservation. Proc. Natl. Acad. Sci. USA 2013, 110, E2602–E2610. [Google Scholar] [CrossRef] [PubMed]
- Belote, R.T.; Aplet, G.H. Land protection and timber harvesting along productivity and diversity gradients in the Northern Rocky Mountains. Ecosphere 2014, 5. [Google Scholar] [CrossRef]
- Jenkins, C.N.; Van Houtan, K.S.; Pimm, S.L.; Sexton, J.O. US protected lands mismatch biodiversity priorities. Proc. Natl. Acad. Sci. USA 2015, 112, 5081–5086. [Google Scholar] [CrossRef] [PubMed]
- Little, E.L. Atlas of United States Trees; U.S. Department of Agriculture Forest Service: Washington, DC, USA, 1971.
- Theobald, D.M. A general model to quantify ecological integrity for landscape assessments and US application. Landsc. Ecol. 2013, 28, 1859–1874. [Google Scholar] [CrossRef]
- Venter, O.; Sanderson, E.W.; Magrach, A.; Allan, J.R.; Beher, J.; Jones, K.R.; Possingham, H.P.; Laurance, W.F.; Wood, P.; Fekete, B.M.; et al. Global terrestrial Human Footprint maps for 1993 and 2009. Sci. Data 2016, 160067. [Google Scholar] [CrossRef]
- Dobrowski, S.Z.; Abatzoglou, J.; Swanson, A.K.; Greenberg, J.A.; Mynsberge, A.R.; Holden, Z.A.; Schwartz, M.K. The climate velocity of the contiguous United States during the 20th century. Glob. Chang. Biol. 2013, 19, 241–251. [Google Scholar] [CrossRef]
- Stephenson, N.L. Climatic control of vegetation distribution: The role of the water balance. Am. Nat. 1990, 135, 649–670. [Google Scholar] [CrossRef]
- Zhao, M.; Heinsch, F.A.; Nemani, R.R.; Running, S.W. Improvements of the MODIS terrestrial gross and net primary production global data set. Remote Sens. Environ. 2005, 95, 164–176. [Google Scholar] [CrossRef]
- Scott, J.M.; Davis, F.W.; McGhie, R.G.; Wright, R.G.; Groves, C.; Estes, J. Nature reserves: Do they capture the full range of America’s biological diversity? Ecol. Appl. 2001, 11, 999–1007. [Google Scholar] [CrossRef]
- Aycrigg, J.L.; Davidson, A.; Svancara, L.K.; Gergely, K.J.; McKerrow, A.; Scott, J.M. Representation of ecological systems within the protected areas network of the continental United States. PLoS ONE 2013, 8, e54689. [Google Scholar] [CrossRef] [PubMed]
- The Conservation Biology Institute. PAD-US (CBI Edition) Version 2.1; The Conservation Biology Institute: Corvallis, OR, USA, 2015. [Google Scholar]
- Belote, R.T.; Jones, R.H.; Hood, S.M.; Wender, B.W. Diversity-invasibility across an experimental disturbance gradient in Appalachian forests. Ecology 2008, 89. [Google Scholar] [CrossRef]
- Grace, J.B. Structural Equation Modeling and Natural Systems; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Rosseel, Y. lavaan: An R package for structural equation modeling. J. Stat. Softw. 2012, 48, 1–36. [Google Scholar] [CrossRef]
- Barrett, P. Structural equation modelling: Adjudging model fit. Pers. Individ. Differ. 2007, 42, 815–824. [Google Scholar] [CrossRef]
- Shands, W.E.; Healy, R.G. The Lands Nobody Wanted: Policy for National Forests in the Eastern United States; Conservation Foundation: Washington, DC, USA, 1977. [Google Scholar]
- Hessburg, P.F.; Agee, J.K. An Environmental Narrative of Inland Northwest United States Forests, 1800–2000. For. Ecol. Manag. 2003, 178, 23–59. [Google Scholar] [CrossRef]
- Lieberman, B.; Gordan, E. Climate Change in Human History: Prehistory to the present; Bloomsbury Academic: London, UK, 2018; ISBN 9781472598493. [Google Scholar]
- Diamond, J.M. Guns, Germs and Steel: A Short History of Everybody for the Last 13,000 Years; Random House: New York, NY, USA, 1998; ISBN 0099302780. [Google Scholar]
- Francis, A.P.; Currie, D.J. A globally consistent richness-climate relationship for angiosperms. Am. Nat. 2003, 161, 523–536. [Google Scholar] [CrossRef]
- Dietz, M.S.; Belote, R.T.; Aplet, G.H.; Aycrigg, J.L. The world’s largest wilderness protection network after 50 years: An assessment of ecological system representation in the U.S. National Wilderness Preservation System. Biol. Conserv. 2015, 184, 431–438. [Google Scholar] [CrossRef]
- Belote, R.T.; Dietz, M.S.; Jenkins, C.N.; McKinley, P.S.; Irwin, G.H.; Fullman, T.J.; Leppi, J.C.; Aplet, G.H. Wild, connected, and diverse: Building a more resilient system of protected areas. Ecol. Appl. 2017, 27, 1050–1056. [Google Scholar] [CrossRef] [PubMed]
- Noss, R.F.; Platt, W.J.; Sorrie, B.A.; Weakley, A.S.; Means, D.B.; Costanza, J.; Peet, R.K. How global biodiversity hotspots may go unrecognized: Lessons from the North American Coastal Plain. Divers. Distrib. 2015, 21, 236–244. [Google Scholar] [CrossRef]
- Ellison, A.M.; Bank, M.S.; Clinton, B.D.; Colburn, E.A.; Elliott, K.; Ford, C.R.; Foster, D.R.; Kloeppel, B.D.; Knoepp, J.D.; Lovett, G.M.; et al. Loss of foundation species: Consequences for the structure and dynamics of forested ecosystems. Front. Ecol. Environ. 2005, 3, 479–486. [Google Scholar] [CrossRef]
- Lovett, G.M.; Canham, C.D.; Arthur, M.A.; Weathers, K.C.; Fitzhugh, R.D. Forest Ecosystem Responses to Exotic Pests and Pathogens in Eastern North America. Bioscience 2006, 56, 395–405. [Google Scholar] [CrossRef]
- Riitters, K.; Potter, K.; Iannone, B.; Oswalt, C.; Guo, Q.; Fei, S.; Riitters, K.; Potter, K.M.; Iannone, B.V.; Oswalt, C.; et al. Exposure of protected and unprotected forest to plant invasions in the eastern United States. Forests 2018, 9, 723. [Google Scholar] [CrossRef]
- Faurby, S.; Svenning, J.C. Historic and prehistoric human-driven extinctions have reshaped global mammal diversity patterns. Divers. Distrib. 2015, 21, 1155–1166. [Google Scholar] [CrossRef]
- Houghton, R.A.; Hackler, J.L. Changes in terrestrial carbon storage in the United States. 1: The roles of agriculture and forestry. Glob. Ecol. Biogeogr. 2000, 9, 125–144. [Google Scholar] [CrossRef]
- Belote, R.T.; Jones, R.H.; Wieboldt, T.F. Compositional stability and diversity of vascular plant communities following logging disturbance in Appalachian forests. Ecol. Appl. 2012, 22. [Google Scholar] [CrossRef]
- Drummond, M.A.; Loveland, T.R. Land-use pressure and a transition to forest-cover loss in the Eastern United States. Bioscience 2010, 60, 286–298. [Google Scholar] [CrossRef]
- Theobald, D.M.; Zachmann, L.J.; Dickson, B.G.; Gray, M.E.; Albano, C.M.; Landau, V.; Harrison-Atlas, D. The Disappearing West: Description of the Approach, Data, and Analytical Methods Used to Estimate Natural Land Loss in the Western U.S.; Conservation Science Partners: Truckee, CA, USA, 2016. [Google Scholar]
- Belote, R.T.; Irwin, G.H. Quantifying the national significance of local areas for regional conservation planning: North Carolina’s Mountain Treasures. Land 2017, 6, 35. [Google Scholar] [CrossRef]
- Kareiva, P.; Marvier, M. Conserving biodiversity coldspots. Am. Sci. 2003, 91, 344–351. [Google Scholar] [CrossRef]
- Watson, J.E.M.; Venter, O.; Lee, J.; Jones, K.R.; Robinson, J.G.; Possingham, H.P.; Allan, J.R. Protect the last of the wild. Nature 2018, 563, 27–30. [Google Scholar] [CrossRef] [PubMed]
- LaManna, J.A.; Belote, R.T.; Burkle, L.A.; Catano, C.P.; Myers, J.A. Negative density dependence mediates biodiversity-productivity relationships across scales. Nat. Ecol. Evol. 2017, 1. [Google Scholar] [CrossRef] [PubMed]
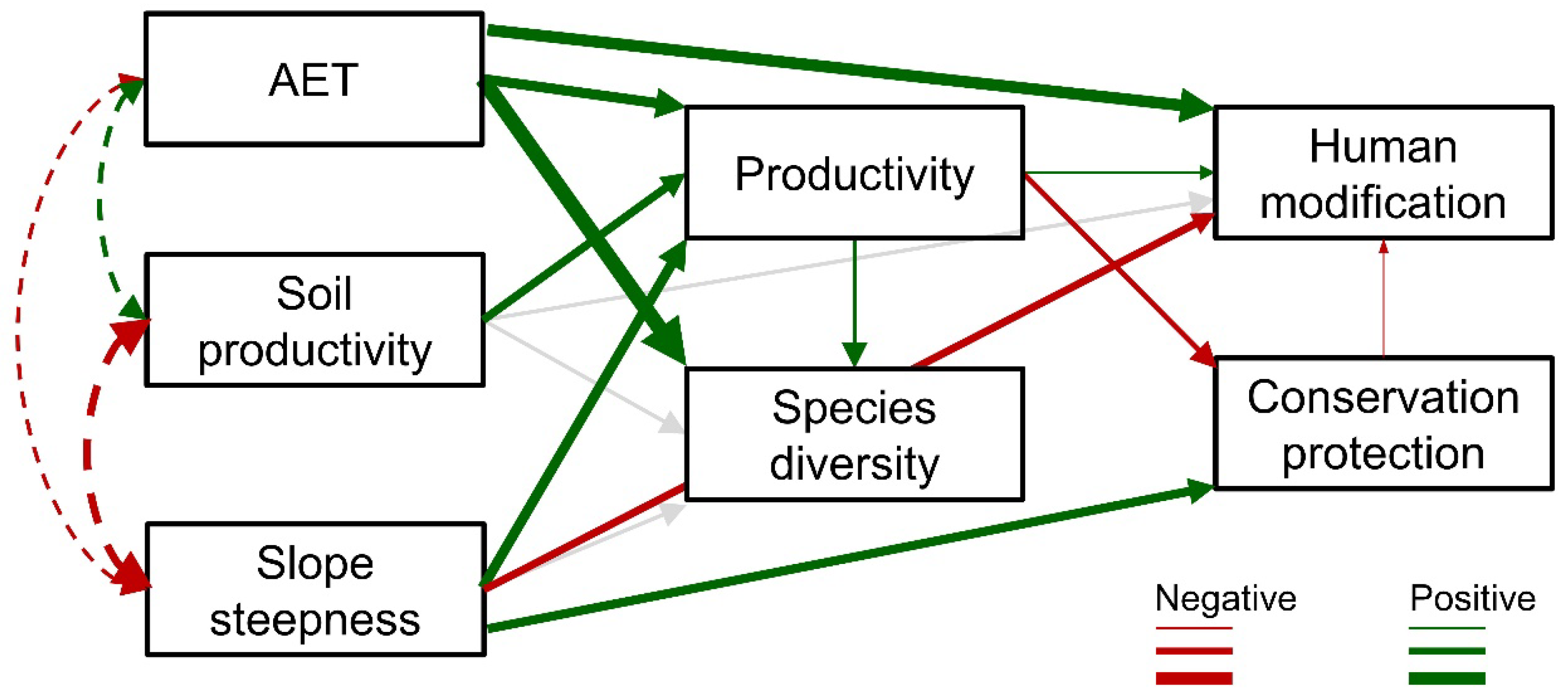
| Estimate | Standard Error | z-Value | p-Value | |
|---|---|---|---|---|
| Human modification | ||||
| Conservation protection | −0.069 | 0.028 | −2.425 | 0.015 |
| Slope steepness | −0.248 | 0.045 | −5.54 | <0.001 |
| Productivity | 0.096 | 0.033 | 2.892 | 0.004 |
| AET | 0.594 | 0.034 | 17.274 | <0.001 |
| Soil productivity | −0.004 | 0.042 | −0.095 | 0.925 |
| Conservation protection | ||||
| Productivity | −0.194 | 0.041 | −4.719 | <0.001 |
| Slope steepness | 0.375 | 0.041 | 9.118 | <0.001 |
| Productivity | ||||
| AET | 0.472 | 0.042 | 11.175 | <0.001 |
| Slope steepness | 0.489 | 0.055 | 8.82 | <0.001 |
| Soil productivity | 0.339 | 0.056 | 6.081 | <0.001 |
| Species richness | ||||
| Productivity | 0.133 | 0.023 | 5.79 | <0.001 |
| AET | 0.843 | 0.024 | 35.066 | <0.001 |
| Soil productivity | 0.007 | 0.029 | 0.227 | 0.82 |
| Slope steepness | 0.019 | 0.03 | 0.617 | 0.537 |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belote, R.T. Species-Rich National Forests Experience More Intense Human Modification, but Why? Forests 2018, 9, 753. https://doi.org/10.3390/f9120753
Belote RT. Species-Rich National Forests Experience More Intense Human Modification, but Why? Forests. 2018; 9(12):753. https://doi.org/10.3390/f9120753
Chicago/Turabian StyleBelote, R. Travis. 2018. "Species-Rich National Forests Experience More Intense Human Modification, but Why?" Forests 9, no. 12: 753. https://doi.org/10.3390/f9120753
APA StyleBelote, R. T. (2018). Species-Rich National Forests Experience More Intense Human Modification, but Why? Forests, 9(12), 753. https://doi.org/10.3390/f9120753




