Responses of Fine Root Functional Traits to Soil Nutrient Limitations in a Karst Ecosystem of Southwest China
Abstract
1. Introduction
2. Materials and Methods
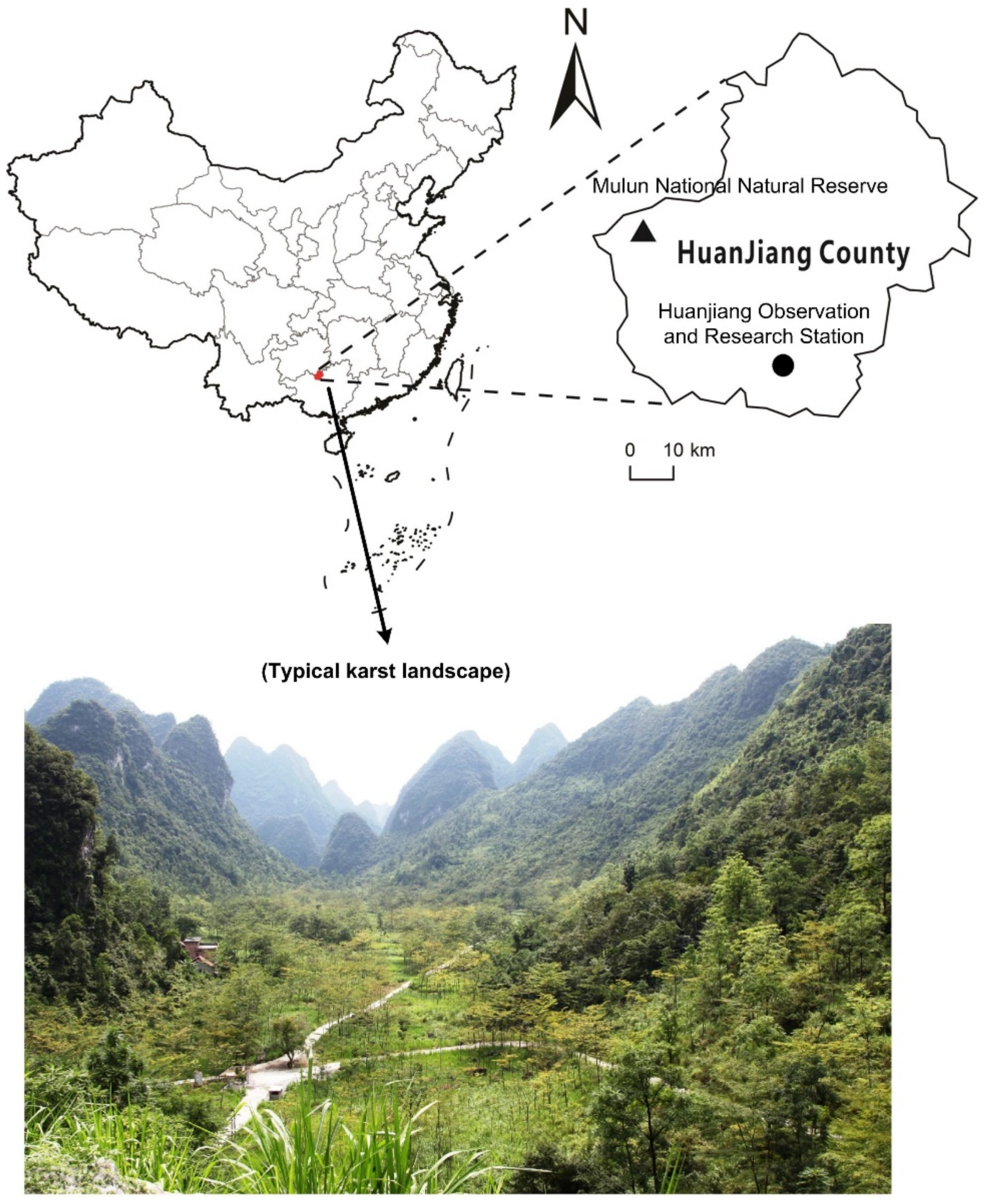
2.1. Study Site
2.2. Sampling
2.3. Laboratory Analyses
2.3.1. SRL, RT/RB, and Nroot of Fine Roots
2.3.2. Oxalic Acid Concentrations of Rhizosphere Soils
2.3.3. β-1,4-N-acetylglucosaminidase (NAG) Activities of Rhizosphere Soils
2.3.4. Soil MBC of Rhizosphere Soils
2.3.5. Chemical Properties of Rhizosphere and Bulk Soils
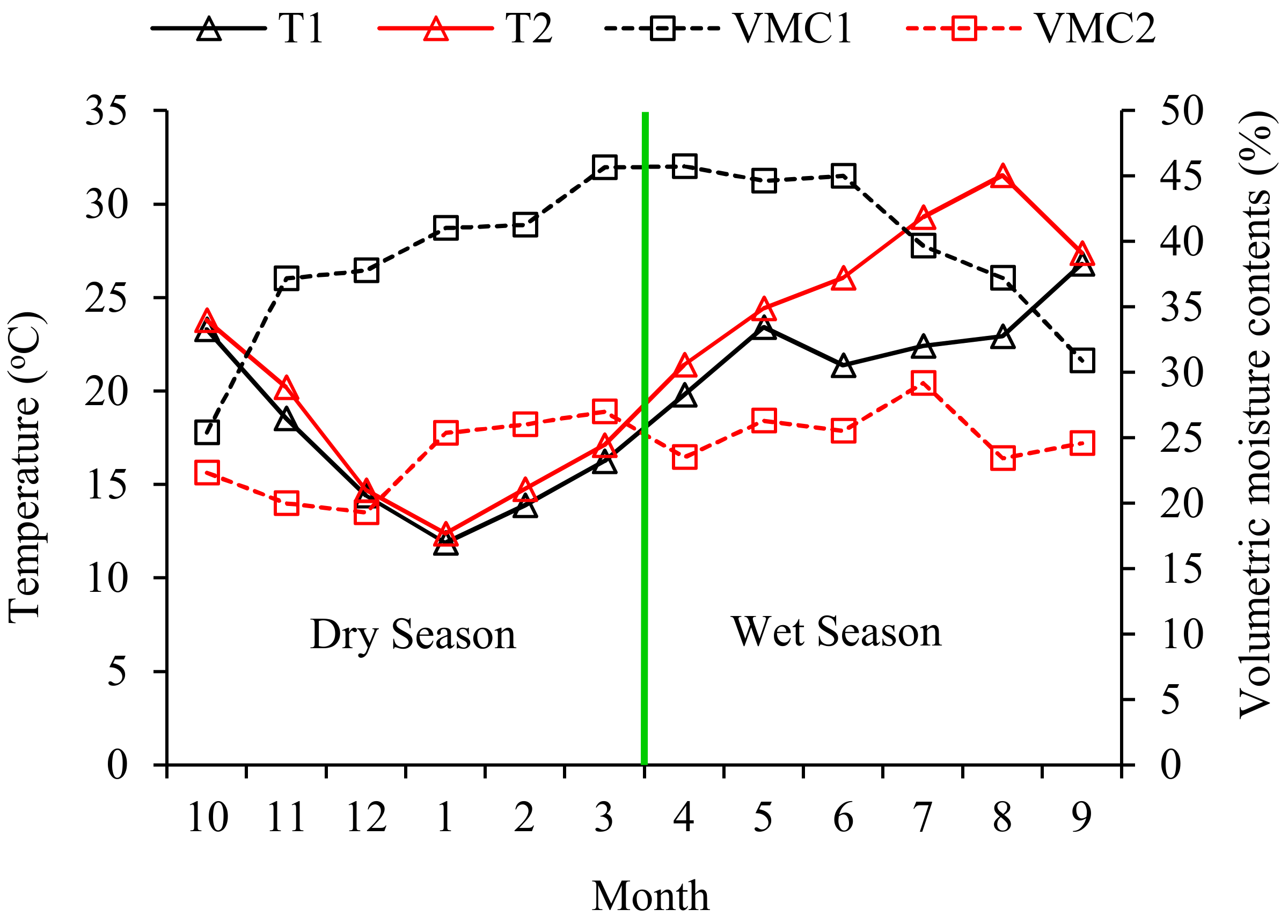
2.3.6. Soil Temperature and Moisture Contents
2.4. Statistical Analyses
3. Results
3.1. Soil Nutrient, Temperature, and Water Characteristics
3.2. Patterns of Fine Root Functional Traits
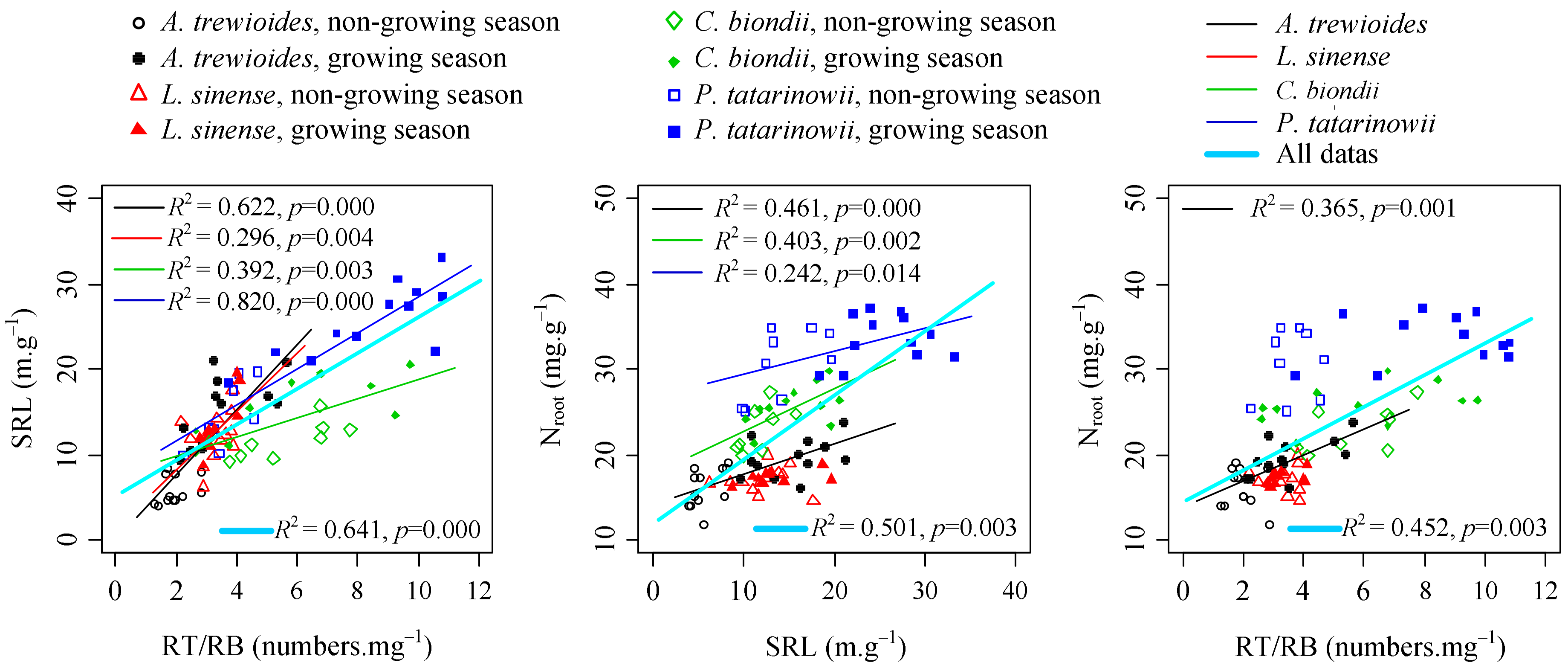
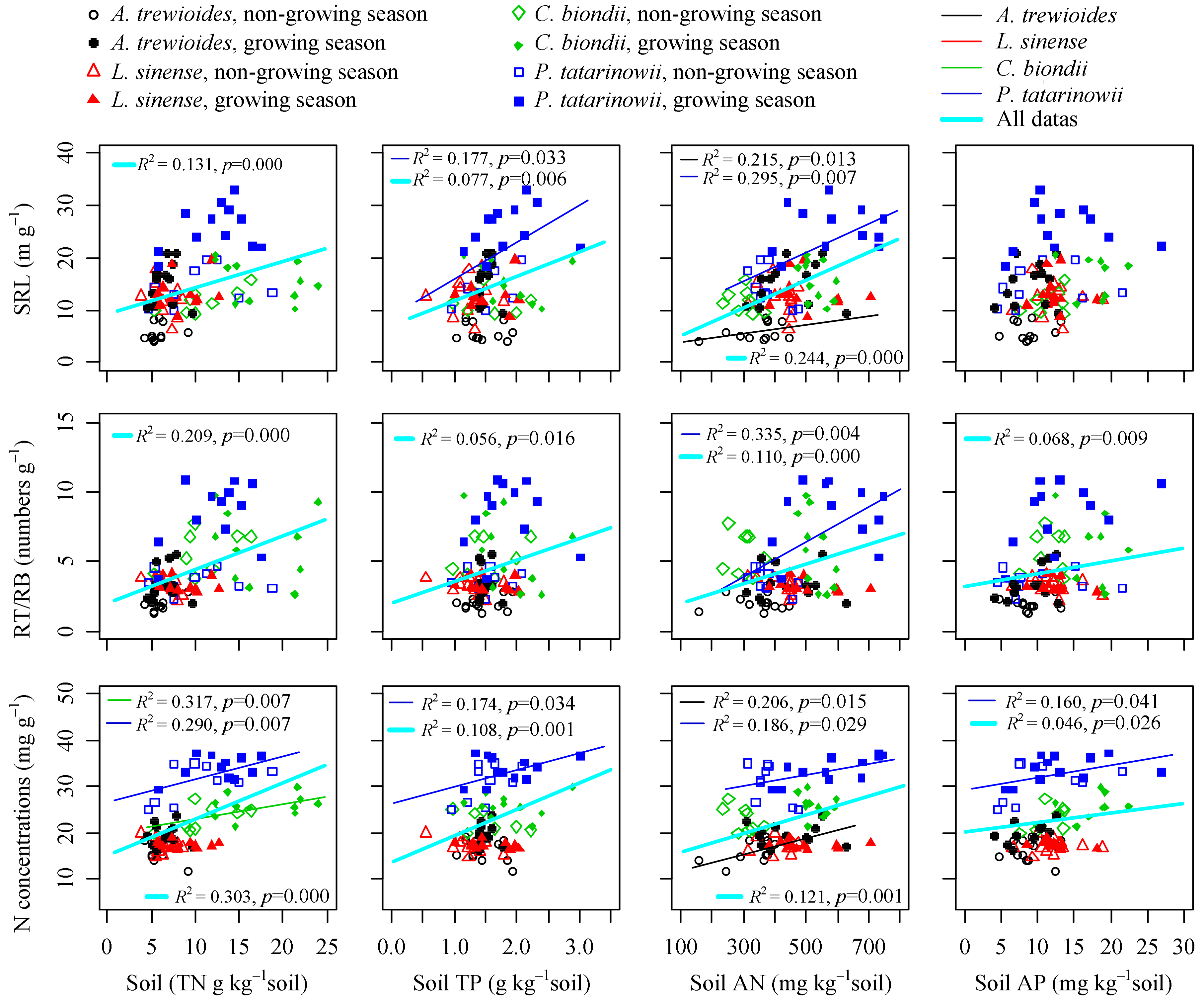
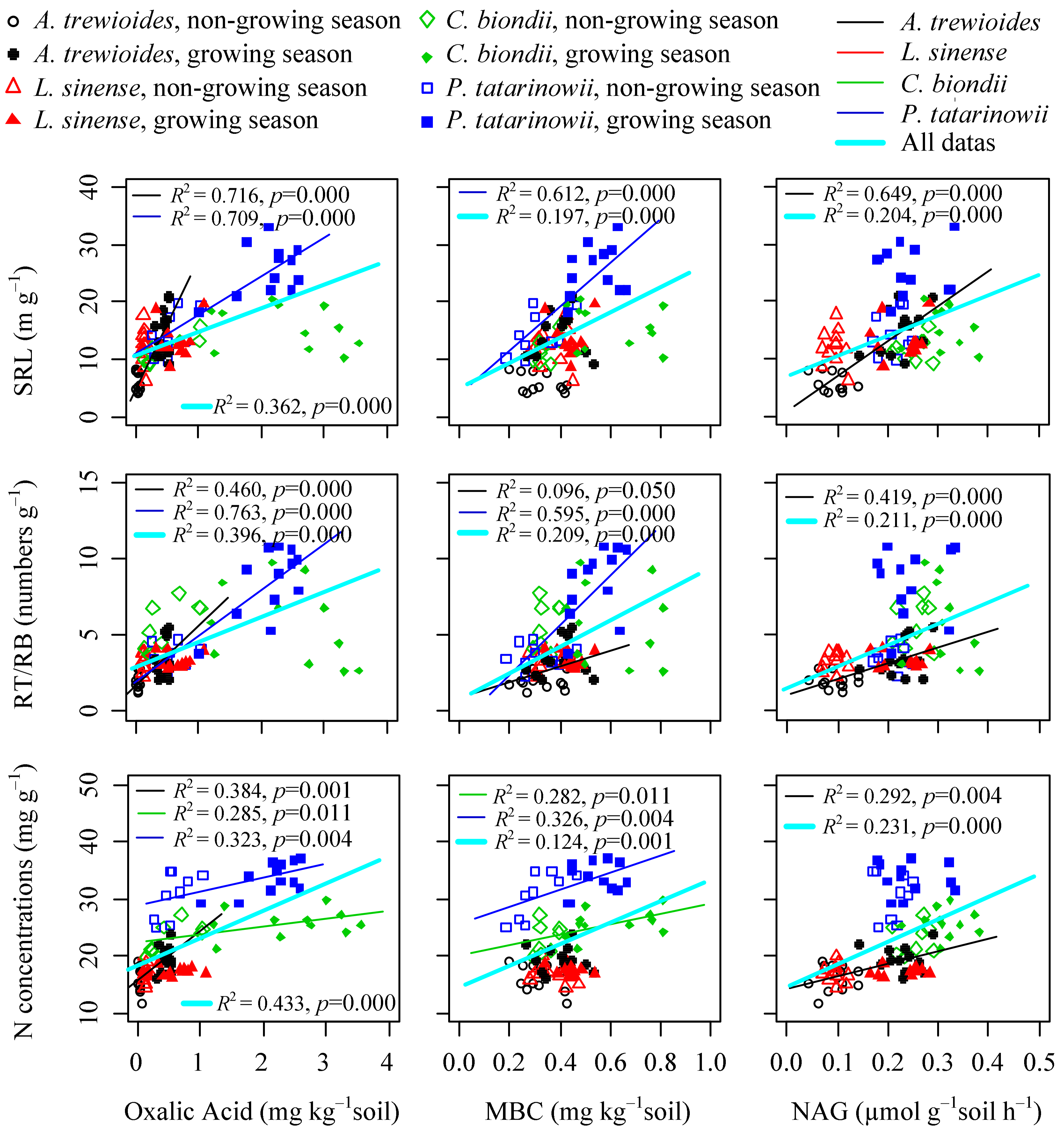
3.3. Relationships between Fine Foot Functional Fraits and Soil Biochemistry
4. Discussion
4.1. Factors Influencing Fine Root Functional Traits
4.2. Relationship between Fine Root Functional Traits and Plant Nutrient Acquisition Strategies
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yuan, D.X. Karstology of China; Geological Publishing House: Beijing, Chinese, 1994. [Google Scholar]
- Qi, X.K.; Wang, K.L.; Zhang, C.H. Effectiveness of ecological restoration projects in a karst region of southwest China assessed using vegetation succession mapping. Ecol. Eng. 2013, 54, 245–253. [Google Scholar] [CrossRef]
- Su, Z.M.; Li, X.K. The types of natural vegetation in karst region of Guangxi and its classified system. Guihaia 2003, 23, 289–293. [Google Scholar]
- Chen, H.S.; Zhang, W.; Wang, K.L.; Fu, W. Soil moisture dynamics under different land uses on karst hillslope in northwest Guangxi, China. Environ. Earth Sci. 2010, 61, 1105–1111. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, K.L.; Chen, H.S.; He, X.Y.; Zhang, J.G. Ancillary information improves kriging on soil organic carbon data for a typical karst peak cluster depression landscape. J. Sci. Food Agr. 2012, 92, 1094–1102. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.J.; Liu, Q.M.; Zhang, D.F. Karst rocky desertification in southwestern China: Geomorphology, landuse, impact and rehabilitation. Land Degrad. Dev. 2004, 15, 115–121. [Google Scholar] [CrossRef]
- Du, Y.X.; Pan, G.X.; Li, L.Q.; Hu, Z.L.; Wang, X.Z. Leaf N/P ratio and nutrient reuse between dominant species and stands: Predicting phosphorus deficiencies in karst ecosystems, southwestern China. Environ. Earth Sci. 2011, 64, 299–309. [Google Scholar] [CrossRef]
- Yang, H.; Cao, J.H.; Sun, L.; Luan, H.N.; Hou, Y.L. Fractions and Distribution of Inorganic Phosphorus in Different Land Use Types of Karst Area. J. Soil Water Conserv. 2010, 2, 135–140. [Google Scholar]
- Zhang, X.B.; Bai, X.Y.; He, X.B. Soil creeping in the weathering crust of carbonate rocks and underground soil losses in the karst mountain areas of southwest China. Carbonates Evaporites 2011, 26, 149–153. [Google Scholar] [CrossRef]
- Pan, F.J.; Zhang, W.; Liang, Y.M.; Liu, S.J.; Wang, K.L. Increased associated effects of topography and litter and soil nutrients on soil enzyme activities and microbial biomass along vegetation successions in karst ecosystem, southwestern China. Environ. Sci. Pollut. R. 2018, 25, 16979–16990. [Google Scholar] [CrossRef] [PubMed]
- Zhou, A.P.; Xiang, W.S.; Yao, Y.F.; Huang, F.Z.; Li, X.K. Analyzing variation characteristics of vegetation net primary productivity in Guangxi. Guihaia 2014, 34, 622–628. [Google Scholar]
- Zhang, W.; Zhao, J.; Pan, F.J.; Li, D.J.; Chen, H.S.; Wang, K.L. Changes in nitrogen and phosphorus limitation during secondary succession in a karst region in southwest China. Plant Soil 2015, 391, 77–91. [Google Scholar] [CrossRef]
- Kang, M.; Wang, J.; Huang, H.W. Nitrogen limitation as a driver of genome size evolution in a group of karst plants. Sci. Rep. 2015, 5, 11636. [Google Scholar] [CrossRef] [PubMed]
- Thorley, R.M.; Taylor, L.L.; Banwart, S.A.; Leake, J.R.; Beerling, D.J. The role of forest trees and their mycorrhizal fungi in carbonate rock weathering and its significance for global carbon cycling. Plant Cell Environ. 2014, 38, 1947–1961. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.M.; He, X.Y.; Chen, C.Y.; Feng, S.Z.; Liu, L.; Chen, X.B.; Zhao, Z.W.; Su, Y.R. Influence of plant communities and soil properties during natural vegetation restoration on arbuscular mycorrhizal fungal communities in a karst region. Ecol. Eng. 2015, 82, 57–65. [Google Scholar] [CrossRef]
- Liu, L.; He, X.Y.; Wang, K.L.; Xie, Y.J.; Xie, Q.; O’Donnell, A.G.; Chen, C.Y. The Bradyrhizobium-legume symbiosis is dominant in the shrubby ecosystem of the Karst region, Southwest China. Eur. J. Soil Biol. 2015, 68, 1–8. [Google Scholar] [CrossRef]
- Clarholm, M.; Skyllberg, U.; Rosling, A. Organic acid induced release of nutrients from metal-stabilized soil organic matter—The unbutton model. Soil Biol. Biochem. 2015, 84, 168–176. [Google Scholar] [CrossRef]
- Pan, F.J.; Liang, Y.M.; Zhang, W.; Zhao, J.; Wang, K.L. Enhanced Nitrogen Availability in Karst Ecosystems by Oxalic Acid Release in the Rhizosphere. Front Plant Sci. 2016, 7, 687. [Google Scholar] [CrossRef] [PubMed]
- Fort, F.; Cruz, P.; Catrice, O.; Delbrut, A.; Luzarreta, M.; Stroia, C.; Jouany, C. Root functional trait syndromes and plasticity drive the ability of grassland Fabaceae to tolerate water and phosphorus shortage. Environ. Exp. Bot. 2015, 110, 62–72. [Google Scholar] [CrossRef]
- Valverde-Barrantes, O.J.; Smemo, K.A.; Blackwood, C.B. Fine root morphology is phylogenetically structured, but nitrogen is related to the plant economics spectrum in temperate trees. Funct. Ecol. 2015, 29, 796–807. [Google Scholar] [CrossRef]
- Helmisaari, H.S.; Makkonen, K.; Kellomaki, S.; Valtonen, E.; Malkonen, E. Below- and above-ground biomass, production and nitrogen use in Scots pine stands in eastern Finland. For. Ecol. Manag. 2002, 165, 317–326. [Google Scholar] [CrossRef]
- Montagnoli, A.; Di Iorio, A.; Terzaghi, M.; Trupiano, D.; Scippa, G.S.; Chiatante, D. Influence of soil temperature and water content on fine-root seasonal growth of European beech natural forest in Southern Alps, Italy. Eur. J. For. Res. 2014, 133, 957–968. [Google Scholar] [CrossRef]
- Wurzburger, N.; Wright, S.J. Fine-root responses to fertilization reveal multiple nutrient limitation in a lowland tropical forest. Ecology 2015, 96, 2137–2146. [Google Scholar] [CrossRef] [PubMed]
- Grassein, F.; Lemauviel-Lavenant, S.; Lavorel, S.; Bahn, M.; Bardgett, R.D.; Desclos-Theveniau, M.; Laine, P. Relationships between functional traits and inorganic nitrogen acquisition among eight contrasting European grass species. Ann. Bot.-London 2015, 115, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Pinno, B.D.; Wilson, S.D. Fine root response to soil resource heterogeneity differs between grassland and forest. Plant Ecol. 2013, 214, 821–829. [Google Scholar] [CrossRef]
- Kalliokoski, T.; Pennanen, T.; Nygren, P.; Sievanen, R.; Helmisaari, H.S. Belowground interspecific competition in mixed boreal forests: Fine root and ectomycorrhiza characteristics along stand developmental stage and soil fertility gradients. Plant Soil 2010, 330, 73–89. [Google Scholar] [CrossRef]
- Beyer, F.; Hertel, D.; Jung, K.; Fender, A.C.; Leuschner, C. Competition effects on fine root survival of Fagus sylvatica and Fraxinus excelsior. For. Ecol. Manag. 2013, 302, 14–22. [Google Scholar] [CrossRef]
- Sun, Y.; Gu, J.C.; Zhuang, H.F.; Wang, Z.Q. Effects of ectomycorrhizal colonization and nitrogen fertilization on morphology of root tips in a Larix gmelinii plantation in northeastern China. Ecol. Res. 2010, 25, 295–302. [Google Scholar] [CrossRef]
- Comas, L.H.; Eissenstat, D.M. Linking fine root traits to maximum potential growth rate among 11 mature temperate tree species. Funct. Ecol. 2004, 18, 388–397. [Google Scholar] [CrossRef]
- McCormack, M.L.; Adams, T.S.; Smithwick, E.A.H.; Eissenstat, D.M. Predicting fine root lifespan from plant functional traits in temperate trees. New Phytol. 2012, 195, 823–831. [Google Scholar] [CrossRef] [PubMed]
- Nan, H.; Liu, Q.; Chen, J.; Cheng, X.; Yin, H.; Yin, C.; Zhao, C. Effects of nutrient heterogeneity and competition on root architecture of spruce seedlings: Implications for an essential feature of root foraging. PLoS ONE 2013, 8, e65650. [Google Scholar] [CrossRef] [PubMed]
- Ostonen, I.; Püttsepp, Ü.; Biel, C.; Alberton, O.; Bakker, M.R.; Lõhmus, K.; Majdi, H.; Metcalfe, D.; Olsthoorn, A.F.M.; Pronk, A.; Vanguelova, E.; et al. Specific root length as an indicator of environmental change. Plant Biosyst. 2007, 141, 426–442. [Google Scholar] [CrossRef]
- Wang, G.L.; Fahey, T.J.; Xue, S.; Liu, F. Root morphology and architecture respond to N addition in Pinus tabuliformis, west China. Oecologia 2013, 171, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Kochsiek, A.; Tan, S.; Russo, S.E. Fine root dynamics in relation to nutrients in oligotrophic Bornean rain forest soils. Plant Ecol. 2013, 214, 869–882. [Google Scholar] [CrossRef]
- Espeleta, J.F.; Donovan, L.A. Fine root demography and morphology in response to soil resources availability among xeric and mesic sandhill tree species. Funct. Ecol. 2002, 16, 113–121. [Google Scholar] [CrossRef]
- Montagnoli, A.; Terzaghi, M.; Di Iorio, A.; Scippa, G.S.; Chiatante, D. Fine-root morphological and growth traits in a Turkey-oak stand in relation to seasonal changes in soil moisture in the Southern Apennines, Italy. Ecol. Res. 2012, 27, 1015–1025. [Google Scholar] [CrossRef]
- Montagnoli, A.; Di Iorio, A.; Terzaghi, M.; Trupiano, D.; Scippa, G.S.; Chiatante, D. Influence of soil temperature and water content on fine-root seasonal growth of European beech natural forest in Southern Alps, Italy. Eur. J. For. Res. 2014, 133, 957–968. [Google Scholar] [CrossRef]
- Montagnolia, A.; Dumroeseb, R.K.; Terzaghia, M.; Onellic, E.; Scippad, G.S.; Chiatantea, D. Seasonality of fine root dynamics and activity of root and shoot vascular cambium in a Quercus ilex L. forest (Italy). For. Ecol. Manag. 2018. [Google Scholar] [CrossRef]
- Holdaway, R.J.; Richardson, S.J.; Dickie, I.A.; Peltzer, D.A.; Coomes, D.A. Species- and community-level patterns in fine root traits along a 120,000-year soil chronosequence in temperate rain forest. J. Ecol. 2011, 99, 954–963. [Google Scholar] [CrossRef]
- Wright, I.J.; Reich, P.B.; Westoby, M.; Ackerly, D.D.; Baruch, Z.; Bongers, F.; Cavender-Bares, J.; Chapin, T.; Cornelissen, J.H.C.; Diemer, M.; et al. The worldwide leaf economics spectrum. Nature 2004, 428, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Shipley, B.; Lechowicz, M.J.; Wright, I.; Reich, P.B. Fundamental trade-offs generating the worldwide leaf economics spectrum. Ecology 2006, 87, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Ostonen, I.; Helmisaari, H.S.; Borken, W.; Tedersoo, L.; Kukumagi, M.; Bahram, M.; Lindroos, A.J.; Nojd, P.; Uri, V.; Merila, P.; et al. Fine root foraging strategies in Norway spruce forests across a European climate gradient. Glob. Change Biol. 2011, 17, 3620–3632. [Google Scholar] [CrossRef]
- Kou, L.; Guo, D.L.; Yang, H.; Gao, W.L.; Li, S.G. Growth, morphological traits and mycorrhizal colonization of fine roots respond differently to nitrogen addition in a slash pine plantation in subtropical China. Plant Soil. 2015, 391, 207–218. [Google Scholar] [CrossRef]
- Zhang, Z.H.; Hu, G.; Zhu, J.D.; Luo, D.H.; Ni, J. Spatial patterns and interspecific associations of dominant tree species in two old-growth karst forests, SW China. Ecol. Res. 2010, 25, 1151–1160. [Google Scholar] [CrossRef]
- Liu, C.C.; Liu, Y.G.; Guo, K.; Li, G.Q.; Zheng, Y.R.; Yu, L.F.; Yang, R. Comparative ecophysiological responses to drought of two shrub and four tree species from karst habitats of southwestern China. Trees-Struct. Funct. 2011, 25, 537–549. [Google Scholar] [CrossRef]
- Wang, Z.H.; Xu, W.Y. Decomposition-rate estimation of leaf litter in karst forests in China based on a mathematical model. Plant Soil 2013, 367, 563–577. [Google Scholar] [CrossRef]
- Pan, F.J.; Zhang, W.; Liu, S.J.; Li, D.J.; Wang, K.L. Leaf N:P stoichiometry across plant functional groups in the karst region of southwestern China. Trees-Struct. Funct. 2015, 29, 883–892. [Google Scholar] [CrossRef]
- Nie, Y.P.; Chen, H.S.; Wang, K.L.; Yang, J. Water source utilization by woody plants growing on dolomite outcrops and nearby soils during dry seasons in karst region of Southwest China. J. Hydrol. 2012, 420, 264–274. [Google Scholar] [CrossRef]
- Macrae, A.; Lucon, C.M.M.; Rimmer, D.L.; O’Donnell, A.G. Sampling DNA from the rhizosphere of Brassica napus to investigate rhizobacterial community structure. Plant Soil 2001, 233, 223–230. [Google Scholar] [CrossRef]
- Pregitzer, K.S.; DeForest, J.L.; Burton, A.J.; Allen, M.F.; Ruess, R.W.; Hendrick, R.L. Fine root architecture of nine North American trees. Ecol. Monogr. 2002, 72, 293–309. [Google Scholar] [CrossRef]
- Guo, D.L.; Mitchell, R.J.; Hendricks, J.J. Fine root branch orders respond differentially to carbon source-sink manipulations in a longleaf pine forest. Oecologia 2004, 140, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Kurosumi, A.; Kaneko, E.; Nakamura, Y. Degradation of reactive dyes by ozonation and oxalic acid-assimilating bacteria isolated from soil. Biodegradation 2008, 19, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Finzi, A.C.; Sinsabaugh, R.L.; Long, T.M.; Osgood, M.P. Microbial community responses to atmospheric carbon dioxide enrichment in a warm-temperate forest. Ecosystems 2006, 9, 215–226. [Google Scholar] [CrossRef]
- Saiya-Cork, K.R.; Sinsabaugh, R.L.; Zak, D.R. The effects of long term nitrogen deposition on extracellular enzyme activity in an Acer saccharum forest soil. Soil Biol. Biochem. 2002, 34, 1309–1315. [Google Scholar] [CrossRef]
- Wu, J.S.; Joergensen, R.G.; Pommerening, B.; Chaussod, R.; Brookes, P.C. Measurement of Soil Microbial Biomass C by Fumigation Extraction—An Automated Procedure. Soil Biol. Biochem. 1990, 22, 1167–1169. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2015; Available online: http://www.R-project.org/.
- Tjoelker, M.G.; Craine, J.M.; Wedin, D.; Reich, P.B.; Tilman, D. Linking leaf and root trait syndromes among 39 grassland and savannah species. New Phytol. 2005, 167, 493–508. [Google Scholar] [CrossRef] [PubMed]
- Fornara, D.A.; Tilman, D.; Hobbie, S.E. Linkages between plant functional composition, fine root processes and potential soil N mineralization rates. J. Ecol. 2009, 97, 48–56. [Google Scholar] [CrossRef]
- Wahl, S.; Ryser, P. Root tissue structure is linked to ecological strategies of grasses. New Phytol. 2000, 148, 459–471. [Google Scholar] [CrossRef]
- Freschet, G.T.; Valverde-Barrantes, O.J.; Tucker, C.M.; Craine, J.M.; McCormack, M.L.; Violle, C.; Fort, F.; Blackwood, C.B.; Urban-Mead, K.R.; Iversen, C.M.; et al. Climate, soil and plant functional types as drivers of global fine-root trait variation. J. Ecol. 2017, 105, 1182–1196. [Google Scholar] [CrossRef]
- Helmisaari, H.S.; Ostonen, I.; Lohmus, K.; Derome, J.; Lindroos, A.J.; Merila, P.; Nojd, P. Ectomycorrhizal root tips in relation to site and stand characteristics in Norway spruce and Scots pine stands in boreal forests. Tree Physiol. 2009, 29, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Xiang, W.H.; Wu, W.; Tong, J.; Deng, X.W.; Tian, D.L.; Zhang, L.; Liu, C.; Peng, C.H. Differences in fine root traits between early and late-successional tree species in a Chinese subtropical forest. Forestry 2013, 86, 343–351. [Google Scholar] [CrossRef]
- Loiola, P.P.; Scherer-Lorenzen, M.; Batalha, M.A. The role of environmental filters and functional traits in predicting the root biomass and productivity in savannas and tropical seasonal forests. For. Ecol. Manag. 2015, 342, 49–55. [Google Scholar] [CrossRef]
- Chen, S.M.; Lin, S.; Loges, R.; Reinsch, T.; Hasler, M.; Taube, F. Independence of seasonal patterns of root functional traits and rooting strategy of a grass-clover sward from sward age and slurry application. Grass Forage Sci. 2016, 71, 607–621. [Google Scholar] [CrossRef]
- Jagodzinski, A.M.; Dyderski, M.K.; Rawlik, K.; Katna, B. Seasonal variability of biomass, total leaf area and specific leaf area of forest understory herbs reflects their life strategies. For. Ecol. Manag. 2016, 374, 71–81. [Google Scholar] [CrossRef]
- Reich, P.B.; Walters, M.B.; Tjoelker, M.G.; Vanderklein, D.; Buschena, C. Photosynthesis and respiration rates depend on leaf and root morphology and nitrogen concentration in nine boreal tree species differing in relative growth rate. Funct. Ecol. 1998, 12, 395–405. [Google Scholar] [CrossRef]
- Reich, P.B.; Tjoelker, M.G.; Walters, M.B.; Vanderklein, D.W.; Bushena, C. Close association of RGR, leaf and root morphology, seed mass and shade tolerance in seedlings of nine boreal tree species grown in high and low light. Funct. Ecol. 1998, 12, 327–338. [Google Scholar] [CrossRef]
- Pilon, R.; Picon-Cochard, C.; Bloor, J.M.G.; Revaillot, S.; Kuhn, E.; Falcimagne, R.; Balandier, P.; Soussana, J.F. Grassland root demography responses to multiple climate change drivers depend on root morphology. Plant Soil 2013, 364, 395–408. [Google Scholar] [CrossRef]
- Sebastian, N.; Erika, H.; Christian, K. Critically low soil temperatures for root growth and root morphology in three alpine plant species. Alp. Bot. 2016, 126, 11–21. [Google Scholar] [CrossRef]
- Paz, H.; Pineda-Garcia, F.; Pinzon-Perez, L.F. Root depth and morphology in response to soil drought: Comparing ecological groups along the secondary succession in a tropical dry forest. Oecologia 2015, 179, 551–561. [Google Scholar] [CrossRef] [PubMed]
- Fischer, C.; Tischer, J.; Roscher, C.; Eisenhauer, N.; Ravenek, J.; Gleixner, G.; Attinger, S.; Jensen, B.; de Kroon, H.; Mommer, L.; et al. Plant species diversity affects infiltration capacity in an experimental grassland through changes in soil properties. Plant Soil 2015, 397, 1–16. [Google Scholar] [CrossRef]
- Noguchi, K.; Nagakura, J.; Kaneko, S. Biomass and morphology of fine roots of sugi (Cryptomeria japonica) after 3 years of nitrogen fertilization. Front Plant Sci. 2013, 4, 347. [Google Scholar] [CrossRef] [PubMed]
- Eissenstat, D.M. On the relationship between specific root length and the rate of root proliferation—A field–study using citrus rootstocks. New Phytol. 1991, 118, 63–68. [Google Scholar] [CrossRef]
- Zangaro, W.; Alves, R.A.; Lescano, L.E.; Ansanelo, A.P.; Nogueira, M.A. Investment in fine roots and arbuscular mycorrhizal fungi decrease during succession in three brazilian ecosystems. Biotropica 2012, 44, 141–150. [Google Scholar] [CrossRef]
- Svistoonoff, S.; Creff, A.; Reymond, M.; Sigoillot-Claude, C.; Ricaud, L.; Blanchet, A.; Nussaume, L.; Desnos, T. Root tip contact with low-phosphate media reprograms plant root architecture. Nat. Genet. 2007, 39, 792–796. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Li, D.J.; Yang, L.Q.; Luo, P.; Chen, H.; Xiao, K.C.; Song, T.Q.; Zhang, W.; He, X.Y.; Chen, H.S.; et al. Rapid recuperation of soil nitrogen following agricultural abandonment in a karst area, southwest China. Biogeochemistry 2016, 129, 341–354. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Porder, S.; Houlton, B.Z.; Chadwick, O.A. Terrestrial phosphorus limitation: Mechanisms, implications, and nitrogen-phosphorus interactions. Ecol. Appl. 2010, 20, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Strom, L.; Owen, A.G.; Godbold, D.L.; Jones, D.L. Organic acid behaviour in a calcareous soil implications for rhizosphere nutrient cycling. Soil Biol. Biochem. 2005, 37, 2046–2054. [Google Scholar] [CrossRef]
| Source | Total Nitrogen (g kg−1) | Total Phosphorus (g kg−1) | Available Nitrogen (mg kg−1) | Available Phosphorus (mg kg−1) | |
|---|---|---|---|---|---|
| Dry season | |||||
| Alchornea trewioides | RS | 5.86 ± 0.35 A | 1.45 ± 0.08 A | 346.07 ± 23.59 A | 8.83 ± 0.66 A |
| BS | 4.59 ± 0.33 a | 1.38 ± 0.07 a | 329.18 ± 22.66 a | 5.19 ± 0.67 a | |
| Ligustrum sinense | RS | 6.59 ± 0.39 A | 1.24 ± 0.09 A | 422.58 ± 14.19 B | 12.24 ± 0.92 AB |
| BS | 3.86 ± 0.38 a | 1.34 ± 0.10 a | 308.14 ± 24.41 a | 6.55 ± 0.91 a | |
| Celtis biondii | RS | 10.85 ± 1.23 B | 1.54 ± 0.17 A | 425.86 ± 60.70 B | 15.30 ± 4.11 B |
| BS | 7.58 ± 0.94 b | 1.35 ± 0.12 a | 482.65 ± 37.81 b | 7.54 ± 0.85 b | |
| Pteroceltis tatarinowii | RS | 10.30 ± 1.54 B | 1.52 ± 0.11 A | 425.87 ± 38.30 B | 10.64 ± 1.93 A |
| BS | 7.02 ± 1.06 b | 1.38 ± 0.47 a | 416.99 ± 32.33 ab | 6.49 ± 1.30 a | |
| Wet season | |||||
| Alchornea trewioides | RS | 6.53 ± 0.41 A | 1.48 ± 0.03 A | 440.20 ± 28.59 A | 9.00 ± 0.83 A |
| BS | 4.96 ± 0.26 a | 1.39 ± 0.04 a | 347.25 ± 15.92 a | 5.63 ± 0.32 a | |
| Ligustrum sinense | RS | 8.61 ± 0.72 A | 1.52 ± 0.10 A | 501.32 ± 28.87 A | 12.37 ± 0.70 A |
| BS | 5.66 ± 0.57 a | 1.42 ± 0.11 a | 334.47 ± 21.55 a | 7.44 ± 0.67 a | |
| Celtis biondii | RS | 17.61 ± 1.33 C | 1.86 ± 0.15 B | 847.34 ± 50.52 C | 25.52 ± 4.10 B |
| BS | 11.73 ± 0.62 b | 1.68 ± 0.12 b | 759.88 ± 48.81 c | 26.42 ± 4.19 c | |
| Pteroceltis tatarinowii | RS | 12.21 ± 1.11 B | 1.84 ± 0.15 B | 586.53 ± 36.96 B | 13.31 ± 1.72 A |
| BS | 10.10 ± 1.11 b | 1.68 ± 0.14 b | 547.42 ± 44.14 b | 16.12 ± 2.77 b |
| SRL (m g−1) | RT/RB (Number mg−1) | Nroot (mg g−1) | |
|---|---|---|---|
| Dry season | |||
| Alchornea trewioides (shrub) | 5.78 ± 0.48 a | 1.95 ± 0.14 a | 16.12 ± 0.67 a |
| Ligustrum sinense (shrub) | 12.08 ± 0.88 b | 3.30 ± 0.17 b | 17.01 ± 0.43 a |
| Celtis biondii (tree) | 11.70 ± 0.79 b | 5.72 ± 0.53 c | 22.97 ± 0.95 b |
| Pteroceltis tatarinowii (tree) | 14.40 ± 1.23 b | 3.60 ± 0.26 b | 30.68 ± 1.35 b |
| Wet season | |||
| Alchornea trewioides (shrub) | 15.19 ± 1.17 a | 3.47 ± 0.35 a | 19.70 ± 0.65 a |
| Ligustrum sinense (shrub) | 13.32 ± 0.98 a | 3.30 ± 0.15 a | 17.30 ± 0.23 a |
| Celtis biondii (tree) | 15.63 ± 1.14 a | 5.76 ± 0.80 b | 25.80 ± 0.72 b |
| Pteroceltis tatarinowii (tree) | 25.64 ± 1.27 b | 8.41 ± 0.66 c | 33.60 ± 0.80 c |
| Response Variable | Linear Mixed Models | AIC | BIC | T Value | Significant Difference |
|---|---|---|---|---|---|
| SRL | Null model | 558.81 | 566.21 | 4.84 | a |
| Species model | 538.97 | 553.77 | 3.56 | b | |
| Season model | 526.35 | 536.21 | 3.99 | b | |
| Species * Season model | 494.11 | 511.37 | 3.14 | c | |
| RT/RB | Null model | 373.02 | 380.42 | 2.96 | a |
| Species model | 368.20 | 382.99 | 2.09 | b | |
| Season model | 358.95 | 368.81 | 2.50 | b | |
| Species * Season model | 353.26 | 370.52 | 1.60 | c | |
| Nroot | Null model | 494.46 | 501.86 | 4.15 | a |
| Species model | 430.58 | 445.38 | 2.98 | b | |
| Season model | 487.63 | 497.50 | 3.99 | c | |
| Species * Season model | 413.68 | 430.94 | 3.25 | d |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, F.; Liang, Y.; Wang, K.; Zhang, W. Responses of Fine Root Functional Traits to Soil Nutrient Limitations in a Karst Ecosystem of Southwest China. Forests 2018, 9, 743. https://doi.org/10.3390/f9120743
Pan F, Liang Y, Wang K, Zhang W. Responses of Fine Root Functional Traits to Soil Nutrient Limitations in a Karst Ecosystem of Southwest China. Forests. 2018; 9(12):743. https://doi.org/10.3390/f9120743
Chicago/Turabian StylePan, Fujing, Yueming Liang, Kelin Wang, and Wei Zhang. 2018. "Responses of Fine Root Functional Traits to Soil Nutrient Limitations in a Karst Ecosystem of Southwest China" Forests 9, no. 12: 743. https://doi.org/10.3390/f9120743
APA StylePan, F., Liang, Y., Wang, K., & Zhang, W. (2018). Responses of Fine Root Functional Traits to Soil Nutrient Limitations in a Karst Ecosystem of Southwest China. Forests, 9(12), 743. https://doi.org/10.3390/f9120743






