Climate Warming Alters Age-Dependent Growth Sensitivity to Temperature in Eurasian Alpine Treelines
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Tree Species
2.2. Field Sampling and Dendrochronological Methods
2.3. Climate‒Growth Analysis and Projected Scenarios
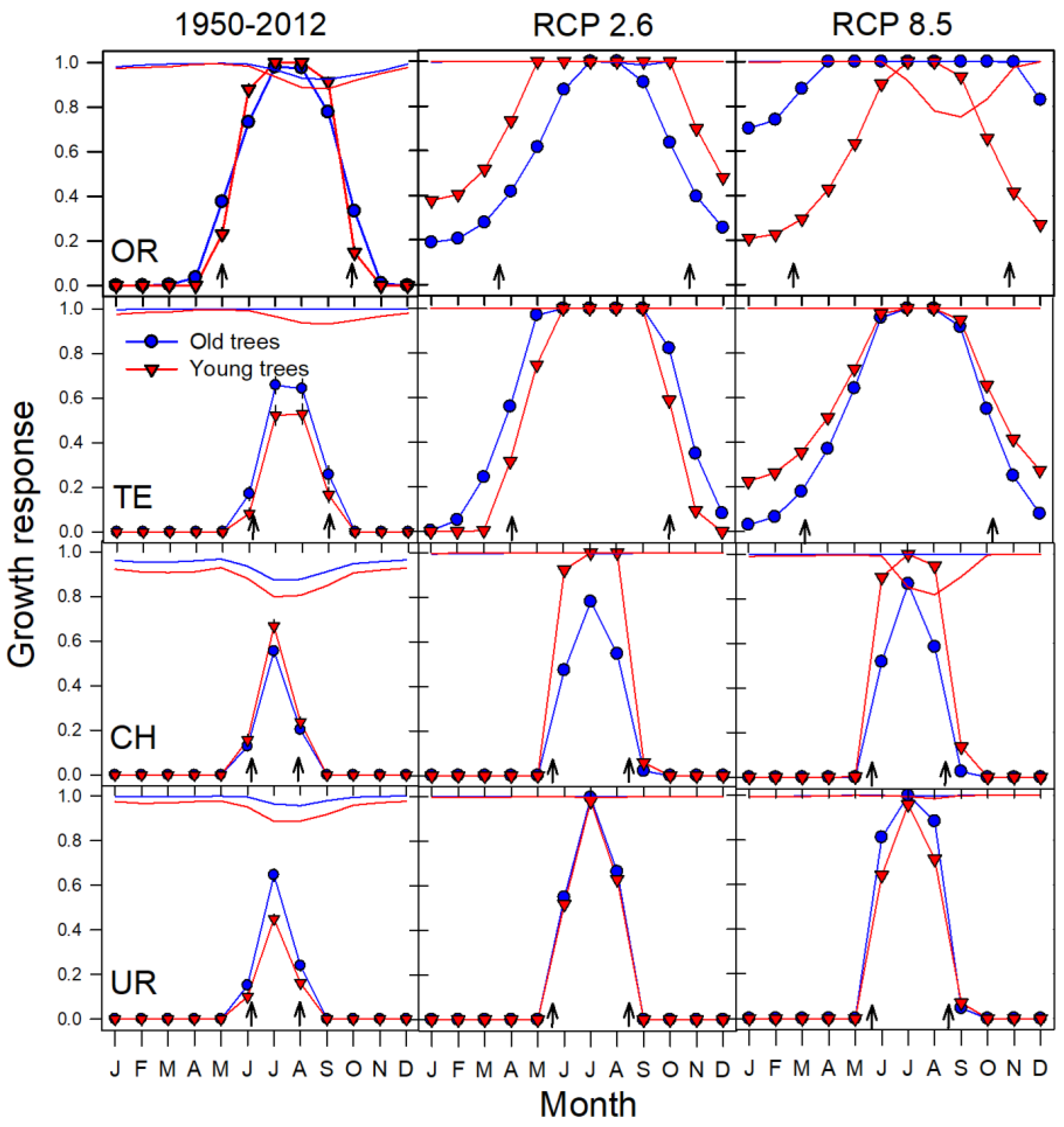
2.4. Temperature-Driven Growth Projections Based on the VS-Lite Model
3. Results
3.1. Age-Related Growth Responsiveness to Temperature
3.2. Process-Based Modelling of Tree Growth at Treeline
3.3. Growth Projections at Treeline Based on Climate‒Growth Relationships
3.4. Age-Related Changes in the Projections of Climatic Thresholds
4. Discussion
4.1. Growth Responsiveness to Rising Temperature at the Treeline
4.2. Climate Warming Influences Growth at Treeline Depending on Tree Age
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Albright, W.L.; Peterson, D.L. Tree growth and climate in the Pacific Northwest, North America: A broad-scale analysis of changing growth environments. J. Biogeogr. 2013, 40, 2119–2133. [Google Scholar] [CrossRef]
- Camarero, J.J.; Gutiérrez, E. Pace and pattern of recent treeline dynamics: Response of ecotones to climatic variability in the Spanish Pyrenees. Clim. Chang. 2004, 63, 181–200. [Google Scholar] [CrossRef]
- Harsch, M.A.; Hulme, P.E.; McGlone, M.S.; Duncan, R.P. Are treelines advancing? A global meta-analysis of treeline response to climate warming. Ecol. Lett. 2009, 12, 1040–1049. [Google Scholar] [CrossRef] [PubMed]
- Hallinger, M.; Manthey, M.; Wilmking, M. Establishing a missing link: Warm summers and winter snow cover promote shrub expansion into alpine tundra in Scandinavia. New Phytol. 2010, 186, 890–899. [Google Scholar] [CrossRef] [PubMed]
- Matías, L.; Jump, A.S. Asymmetric changes of growth and reproductive investment herald altitudinal and latitudinal range shifts of two woody species. Glob. Chang. Biol. 2015, 21, 882–896. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, J.; Körner, C. A climate-based model to predict potential treeline position around the globe. Alpine Bot. 2014, 124, 1–12. [Google Scholar] [CrossRef]
- Camarero, J.J.; Linares, J.C.; García-Cervigón, A.I.; Batllori, E.; Martínez, I.; Gutiérrez, E. Back to the future: The responses of alpine treelines to climate warming are constrained by the current ecotone structure. Ecosystems 2017, 20, 683–700. [Google Scholar] [CrossRef]
- Liu, B.; Wang, Y.; Zhu, H.; Liang, E.; Camarero, J.J. Topography and age mediate the growth responses of Smith fir to climate warming in the southeastern Tibetan Plateau. Int. J. Climatol. 2016, 60, 1577–1587. [Google Scholar] [CrossRef] [PubMed]
- Fritts, H.C. Tree Rings and Climate; Blackburn Press: Caldwell/London, UK; New York, NY, USA, 2001; ISBN 0-12-268450-8. [Google Scholar]
- Szeicz, J.M.; MacDonald, G.M. Age-dependent tree-ring growth responses of subarctic white spruce to climate. Can. J. For. Res. 1994, 23, 120–132. [Google Scholar] [CrossRef]
- Carrer, M.; Urbinati, C. Age-dependent tree ring growth responses to climate of Larix decidua and Pinus cembra in the Italian Alps. Ecology 2004, 85, 730–740. [Google Scholar] [CrossRef]
- Rozas, V.; DeSoto, L.; Olano, J.M. Sex-specific, age-dependent sensitivity of tree-ring growth to climate in the dioecious tree Juniperus thurifera. New Phytol. 2009, 182, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liang, E.; Gričar, J.; Prislan, P.; Rossi, S.; Cufar, K. Age dependence of xylogenesis and its climatic sensitivity in Smith fir on the south-eastern Tibetan Plateau. Tree Physiol. 2013, 2013 33, 48–56. [Google Scholar] [CrossRef]
- Li, B.; Cheng, X.H.; Lv, L.X. Responses of radial growth to fire disturbance in alpine pine (Pinus densata) of different age classes in Nang County, Xizang, China. Chin. J. Plant Ecol. 2016, 40, 436–446. [Google Scholar]
- Yu, G.; Liu, Y.; Wang, X.; Ma, K. Age-dependent tree-ring growth responses to climate in Qilian juniper (Sabina przewalskii Kom.). Trees-Struct. Funct. 2008, 22, 197–204. [Google Scholar] [CrossRef]
- Linares, J.C.; Taïqui, L.; Sangüesa-Barreda, G.; Seco, J.I.; Camarero, J.J. Age-related drought sensitivity of Atlas cedar (Cedrus atlantica) in the Moroccan Middle Atlas forests. Dendrochronologia 2013, 31, 88–96. [Google Scholar] [CrossRef]
- Vautard, R.; Gobiet, A.; Sobolowski, S.; Kjellström, E.; Stegehuis, A.; Watkiss, P.; Mendlik, T.; Landgren, O.; Nikulin, G.; Teichmann, C.; et al. The European climate under a 2 °C global warming. Environ. Res. Lett. 2014, 9, 34006. [Google Scholar] [CrossRef]
- Pellizzari, E.; Camarero, J.J.; Gazol, A.; Granda, E.; Shetti, R.; Wilmking, M.; Moiseev, P.; Pividori, M.; Carrer, M. Diverging shrub and tree growth from the Polar to the Mediterranean biomes across the European continent. Glob. Chang. Biol. 2017, 23, 3169–3180. [Google Scholar] [CrossRef] [PubMed]
- Rolland, C.; Petitcolas, V.; Michalet, R. Change in radial tree growth for Picea abies, Larix decidua, Pinus cembra, and Pinus uncinata near the alpine timberline since 1750. Trees-Struct. Funct. 1998, 13, 40–53. [Google Scholar] [CrossRef]
- Salzer, M.W.; Hughes, M.K.; Bunn, A.G.; Kipfmueller, K.F. Recent unprecedented tree-ring growth in bristlecone pine at the highest elevations and possible causes. Proc. Natl. Acad. Sci. USA 2009, 106, 20348–20353. [Google Scholar] [CrossRef] [PubMed]
- Kullman, L. Tree line population monitoring of Pinus sylvestris in the Swedish Scandes, 1973–2005: Implications for tree line theory and climate change ecology. J. Ecol. 2007, 95, 41–52. [Google Scholar] [CrossRef]
- Slatyer, R.O.; Noble, I.R. Dynamic of Montane Treelines. In Landscape Boundaries: Consequences for Biotic Diversity and Ecological Flows; Hansen, A.J., di Castri, F., Eds.; Springer: New York, NY, USA, 1992; pp. 346–359. ISBN 978-1-4612-7677-7. [Google Scholar]
- Kirdyanov, A.; Hughes, M.; Vaganov, E.; Schweingruber, F.; Silkin, P. The importance of early summer temperature and date of snow melt for tree growth in the Siberian Subarctic. Trees-Struct. Funct. 2003, 17, 61–69. [Google Scholar] [CrossRef]
- Tardif, J.; Camarero, J.J.; Ribas, M.; Gutiérrez, E. Spatiotemporal variability in tree growth in the central Pyrenees: Climatic and site influences. Ecol. Monogr. 2003, 73, 241–257. [Google Scholar] [CrossRef]
- Körner, Ch. Alpine Treelines; Springer: Basel, Switzerland, 2012; p. 220. ISBN 978-3-0348-0395-3. [Google Scholar]
- Mencuccini, M.; Grace, J. Climate influence the leaf-area sapwood ratio in Scots pine. Tree Physiol. 1995, 15, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Deslauriers, A.; Anfodillo, T.; Carrer, M. Age-dependent xylogenesis in timberline conifers. New Phytol. 2008, 177, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Carrer, M.; von Arx, G.; Castagnieri, D.; Petit, G. Distilling environmental information from time series of conduit size: The standardization issue and its relation to allometric and hydraulic constraints. Tree Physiol. 2015, 35, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Konter, O.; Büntgen, U.; Carrer, M.; Esper, J. Testing for Climate Signal Age Effects at Two Treeline Sites in the European Alps and Tatra Mountains. In TRACE–Tree Rings in Archaeology, Climatology and Ecology; Wistuba, M., Cedro, A., Malik, I., Helle, G., Gärtner, H., Eds.; Scientific Technical Report STR; 17/04; GFZ German Research Centre for Geosciences: Potsdam, Germany, 2017; Volume 15. [Google Scholar]
- Ryan, M.G.; Yoder, B.J. Hydraulic limits to tree height and tree growth. Bioscience 1997, 47, 235–242. [Google Scholar] [CrossRef]
- Vaganov, E.A.; Hughes, M.K.; Shashkin, A.V. Growth Dynamics of Conifer Tree Rings; Springer: Berlin, Germany, 2006; p. 358. ISBN 978-3-540-26086-8. [Google Scholar]
- Vaganov, E.A.; Anchukaitis, K.J.; Evans, M.N. How well understood are the processes that create dendroclimatic records? A mechanistic model of the climatic control on conifer tree-ring growth dynamics. Dendroclimatology. In Developments in Paleoenvironmental Research; Hughes, M.K., Swetnam, T.W., Diaz, H.F., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 37–75. ISBN 978-1-4020-4010-8. [Google Scholar]
- Sánchez-Salguero, R.; Camarero, J.J.; Gutiérrez, E.; González Rouco, F.; Gazol, A.; Sangüesa-Barreda, G.; Andreu-Hayles, L.; Linares, J.C.; Seftigen, K. Assessing forest vulnerability to climate warming using a process-based model of tree growth: Bad prospects for rear-edges. Glob. Chang. Biol. 2017, 23, 2705–2719. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Salguero, R.; Camarero, J.J.; Carrer, M.; Gutiérrez, E.; Alla, A.Q.; Andreu-Hayles, L.; Hevia, A.; Koutavas, A.; Martínez-Sancho, E.; Nola, P.; et al. Climate extremes and predicted warming threaten Mediterranean Holocene firs forests refugia. Proc. Natl. Acad. Sci. USA 2017, 114, E10142–E10150. [Google Scholar] [CrossRef] [PubMed]
- Tolwinski-Ward, S.E.; Anchukaitis, K.J.; Evans, M.N. Bayesian parameter estimation and interpretation for an intermediate model of tree-ring width. Clim. Past 2013, 9, 1481–1493. [Google Scholar] [CrossRef]
- Breitenmoser, P.; Bronnimann, S.; Frank, D. Forward modelling of tree-ring width and comparison with a global network of tree-ring chronologies. Clim. Past 2014, 10, 437–449. [Google Scholar] [CrossRef]
- Mina, M.; Martin-Benito, D.; Bugmann, H.; Cailleret, M. Forward modeling of tree-ring width improves simulation of forest growth responses to drought. Agric. For. Meteorol. 2016, 221, 13–33. [Google Scholar] [CrossRef]
- IPCC. Climate change 2013: The physical science basis. In Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013; 1535p. [Google Scholar]
- Batllori, E.; Gutiérrez, E. Regional tree line dynamics in response to global change in the Pyrenees. J. Ecol. 2008, 96, 1275–1288. [Google Scholar] [CrossRef]
- Ninot, J.M.; Carrillo, E.; Font, X.; Carreras, J.; Ferré, A.; Masalles, R.M.; Soriano, I.; Vigo, J. Altitude zonation in the Pyrenees. A geobotanic interpretation. Phytocoenologia 2007, 37, 371–398. [Google Scholar] [CrossRef]
- Camarero, J.J.; Gutiérrez, E. Structure and recent recruitment at alpine forest-pasture ecotones in the Spanish central Pyrenees. Ecoscience 1999, 6, 451–464. [Google Scholar] [CrossRef]
- Ceballos, L.; Ruiz de la Torre, J. Árboles y arbustos de la España Peninsular; Escuela Técnica Superior de Ingenieros de Montes: Madrid, Spain, 1979. ISBN 978847114 9688.
- Camarero, J.J.; Guerrero-Campo, J.; Gutiérrez, E. Tree-ring growth and structure of Pinus uncinata and Pinus sylvestris in the Central Spanish Pyrenees. Arct. Alp. Res. 1998, 30, 1–10. [Google Scholar] [CrossRef]
- López-Moreno, J.I.; García-Ruiz, J.M.; Beniston, M. Environmental Change and water management in the Pyrenees. Facts and future perspectives for Mediterranean mountains. Glob. Planet. Chang. 2008, 66, 300–312. [Google Scholar] [CrossRef]
- Balcells, E.; Gil Pelegrín, E. Consideraciones fenológicas de las biocenosis de altitud en el Parque Nacional de Ordesa y Monte Perdido, acompañadas y apoyadas mediante estudio preliminar de los datos meteorológicos obtenidos desde 1981 a 1989 en el observatorio de Góriz. Lucas Mallada 1992, 4, 71–162. [Google Scholar]
- Camarero, J.J. Growth and Regeneration Patterns and Processes in Pinus uncinata Ram. Treeline Ecotones in the Pyrenees and Anisolated Population in the Western Distribution Limit in Spain. Ph.D. Thesis, University of Barcelona, Barcelona, Spain, 1999. [Google Scholar]
- Shiyatov, S.G.; Terentev, M.M.; Fomin, V.V. Spatiotemporal dynamics of forest-tundra communities in the Polar Urals. Russ. J. Ecol. 2005, 36, 69–75. [Google Scholar] [CrossRef]
- Devi, N.; Hagedorn, F.; Moiseev, P.; Bugmann, H.; Shiyatov, S.; Mazepa, V.; Rigling, A. Expanding forests and changing growth forms of Siberian larch at the Polar Urals treeline during the 20th century. Glob. Chang. Biol. 2008, 14, 1581–1591. [Google Scholar] [CrossRef]
- Hagedorn, F.; Shiyatov, S.G.; Mazepa, V.S.; Devi, N.M.; Grigorèv, A.A.; Bartysh, A.A.; Fomin, V.V.; Kapralov, D.S.; Terentèv, M.; Bugman, H.; et al. Treeline advances along the Urals mountain range—Driven by improved winter conditions? Glob. Chang. Biol. 2014, 20, 3530–3543. [Google Scholar] [CrossRef] [PubMed]
- Holmes, R.L. Computer-assisted quality control in tree-ring dating and measurement. Tree-Ring Bull. 1983, 43, 69–78. [Google Scholar]
- Duncan, R.P. An evaluation of errors in tree age estimates based on increment cores in kahikatea (Dacrycarpus dacrydioides). N. Z. Nat. Sci. 1989, 16, 31–37. [Google Scholar]
- Cook, E.R. A Time-Series Analysis Approach to Tree Ring Standardization. Ph.D. Thesis, University of Arizona, Tucson, AZ, USA, 1985. [Google Scholar]
- Wigley, T.M.; Briffa, K.R.; Jones, P.D. On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J. Appl. Meteorol. Clim. 1984, 23, 201–213. [Google Scholar] [CrossRef]
- Quinn, G.P.; Keough, M.J. Experimental Design and Data Analysis for Biologists; Cambridge University Press: Cambridge, UK, 2002; p. 539. [Google Scholar]
- Harris, I.; Jones, P.D.; Osborn, T.J.; Lister, D.H. Updated high resolution grids of monthly climatic observations–the CRU TS3. 10 Dataset. Int. J. Clim. 2013, 34, 623–642. [Google Scholar] [CrossRef]
- Haylock, M.R.; Hofstra, N.; Klein Tank, A.M.G.; Klok, E.J.; Jones, P.D.; New, M. A European daily high-resolution gridded data set of surface temperature and precipitation for 1950–2006. J. Geophys. Res. Atmos. 2008, D20, 113. [Google Scholar] [CrossRef]
- Jones, P.D. The reliability of global and hemispheric surface temperature records. Adv. Atmos. Sci. 2016, 33, 269–282. [Google Scholar] [CrossRef]
- Moreno, A.; Hasenauer, H. Spatial downscaling of European climate data. Int. J. Climatol. 2016, 2016 36, 1444–1458. [Google Scholar] [CrossRef]
- Taylor, K.E.; Stouffer, R.J.; Meehl, G.A. An overview of CMIP5 and the experiment design. Bull. Am. Meteorol. Soc. 2012, 93, 485–498. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; R Development Core Team: Vienna, Austria, 2018. [Google Scholar]
- Barton, K. MUMIn: Multi-model inference. R. package version 1.40.4. 2018. Available online: https://cran.r-project.org/web/packages/MuMIn/index.html (accessed on 1 June 2018).
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference; Springer: New York, NY, USA, 2002; p. 488. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; R Core Team. nlme: Linear and Nonlinear Mixed Effects Models, R package version 3.1-137; R Core Team: Vienna, Austria, 2018. [Google Scholar]
- Liu, B.; Liang, E.; Liu, K.; Camarero, J.J. Species- and elevation-dependent growth responses to climate warming of mountain forests in the Qinling Mountains, central China. Forests 2018, 9, 248. [Google Scholar] [CrossRef]
- Galván, D.; Camarero, J.J.; Gutiérrez, E. Seeing the trees for the forest: Drivers of individual growth responses to climate in Pinus uncinata mountain forests. J. Ecol. 2014, 102, 1244–1257. [Google Scholar] [CrossRef]
- González de Andrés, E.; Camarero, J.J.; Büntgen, U. Complex climate constraints of upper treeline formation in the Pyrenees. Trees-Struct. Funct. 2015, 29, 941–952. [Google Scholar] [CrossRef]
- Piper, F.; Viñegla, B.; Linares, J.C.; Camarero, J.J.; Cavieres, L.A.; Fajardo, A. Mediterranean and temperate treelines are controlled by different environmental drivers. J. Ecol. 2016, 104, 691–702. [Google Scholar] [CrossRef]
- Camarero, J.J.; Gazol, A.; Galván, J.D.; Sangüesa-Barreda, G.; Gutiérrez, E. Disparate effects of global-change drivers on mountain conifer forests: Warming-induced growth enhancement in young trees vs. CO2 fertilization in old trees from wet sites. Glob. Chang. Biol. 2015, 21, 738–749. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Campo, J.; Camarero, J.J.; Gutiérrez, E. Crecimiento estacional y caída de acículas en Pinus uncinata Ram. y Pinus sylvestris L. Investig. Agrar. Sist. Y Recur. For. 1998, 7, 155–172. [Google Scholar]
- Gutiérrez, E. Climate tree-growth relationships for Pinus uncinata Ram. in the Spanish pre-Pyrenees. Acta Oecol. 1991, 12, 213–225. [Google Scholar]
- Büntgen, U.; Frank, D.; Grudd, H.; Esper, J. Long-term summer temperature variations in the Pyrenees. Clim. Dyn. 2008, 31, 615–631. [Google Scholar] [CrossRef]
- Von Felten, S.; Hättenschwiler, S.; Saurer, M.; Siegwolf, R. Carbon allocation in shoots of alpine treeline conifers in a CO2 enriched environment. Trees Struct. Funct. 2007, 21, 283–294. [Google Scholar] [CrossRef]
- Jiao, L.; Jiang, Y.; Zhang, W.T.; Wang, M.C.; Zhang, L.N.; Zhao, S.D. Divergent responses to climate factors in the radial growth of Larix sibirica in the eastern Tianshan Mountains, northwest China. Trees-Struct. Funct. 2015, 29, 1673–1686. [Google Scholar] [CrossRef]
- Frost, G.V.; Epstein, H.E. Tall shrub and tree expansion in Siberian tundra ecotones since the 1960s. Glob. Chang. Biol. 2014, 20, 1264–1277. [Google Scholar] [CrossRef] [PubMed]
- Vaganov, E.A.; Hughes, M.K.; Kirdyanov, A.V.; Schweingruber, F.H.; Silkin, P.P. Influence of snowfall and melt timing on tree growth in subarctic Eurasia. Nature 1999, 400, 149–151. [Google Scholar] [CrossRef]
- Moser, L.; Fonti, P.; Buentgen, U.; Franzen, J.; Esper, J.; Luterbacher, J.; Frank, D. Timing and duration of European larch growing season along altitudinal gradients in the Swiss Alps. Tree Physiol. 2010, 30, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Stine, A.R.; Huybers, P. Arctic tree rings as recorders of variations in light availability. Nat. Commun. 2014, 5, 3836. [Google Scholar] [CrossRef] [PubMed]
- Galván, J.D.; Camarero, J.J.; Sangüesa-Barreda, G.; Alla, A.Q.; Gutiérrez, E. Sapwood area drives growth in mountain conifer forests. J. Ecol. 2012, 100, 1233–1244. [Google Scholar]
- Mayr, S. Limits in water relations. In Trees at Their Upper Limit; Wieser, G., Tausz, M., Eds.; Springer: Berlin, Germany, 2007; pp. 145–162. [Google Scholar]
- Bond, B.J. Age-related changes in photosynthesis of woody plants. Trends Plant Sci. 2000, 5, 349–353. [Google Scholar] [CrossRef]
- Ryan, M.G.; Phillips, N.; Bond, B.J. The hydraulic limitation hypothesis revisited. Plant Cell Environ. 2006, 29, 367–381. [Google Scholar] [CrossRef] [PubMed]
- Domec, J.C.; Gartner, B.L. Age- and position-related changes in hydraulic versus mechanical dysfunction of xylem: Inferring the design criteria for Douglas-fir wood structure. Tree Physiol. 2002, 22, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Shiyatov, S.G.; Mazepa, V.S.; Vaganov, E.A.; Schweingruber, F.H. Summer Temperature Variations Reconstructed by Tree-Ring Data at the Polar Timberline in Siberia. In Tree Rings, Environment and Humanity, Proceedings of the International Conference, Tucson, Arizona; Dean, J.S., Meko, D.M., Swetnam, T.W., Eds.; Radiocarbon Department of Geosciences University of Arizona: Tucson, AZ, USA, 1996; pp. 249–258. [Google Scholar]
- Phillips, N.G.; Buckley, T.N.; Tissue, D.T. Capacity of Old Trees to Respond to Environmental Change. J. Integr. Plant Biol. 2008, 50, 1355–1364. [Google Scholar] [CrossRef] [PubMed]
- Hunt, E.R.; Lavigne, M.B.; Franklin, S.E. Factors controlling the decline of net primary production with stand age for balsam fir in Newfoundland assessed using an ecosystem simulation model. Ecol. Mod. 1999, 122, 151–164. [Google Scholar] [CrossRef]
- Ruosch, M.; Spahni, R.; Joos, F.; Henne, P.D.; van der Knaap, W.O.; Tinner, W. Past and future evolution of Abies alba forests in Europe—Comparison of a dynamic vegetation model with Palaeo data and observations. Glob. Chang. Biol. 2016, 22, 727–740. [Google Scholar] [CrossRef] [PubMed]
- Lavergne, A.; Daux, V.; Villalba, R.; Barichivich, J. Temporal changes in climatic limitation of tree-growth at upper treeline forests: Contrasted responses along the west to-east humidity gradient in Northern Patagonia. Dendrochronologia 2015, 36, 49–59. [Google Scholar] [CrossRef]
- Yang, B.; He, M.; Shishov, V.; Tychkov, I.; Vaganov, E.H.; Rossi, S.; Ljungqvist, F.C.; Bräuning, A.; Grießinger, J. New perspective on spring vegetation phenology. Proc. Natl. Acad. Sci. USA 2017, 114, 6966–6971. [Google Scholar] [CrossRef] [PubMed]
- Badeck, F.; Bondeau, A.; Böttcher, K.; Doktor, D.; Lucht, W.; Schaber, J.; Sitch, S. Responses of spring phenology to climate change. New Phytol. 2004, 162, 295–309. [Google Scholar] [CrossRef]
- Shiyatov, S.G.; Terentév, M.M.; Fomin, V.V.; Zimmermann, N.E. Altitudinal and horizontal shifts of the upper boundaries of open and closed forests in the Polar Urals in the 20th century. Russ. J. Ecol. 2007, 38, 223–227. [Google Scholar] [CrossRef]
- Vitasse, Y.; Franc, C.; Delpierre, N.; Dufrêne, E.; Kremer, A.; Chuine, I.; Delzon, S. Assessing the effects of climate change on the phenology of European temperate trees. Agric. For. Meteorol. 2011, 151, 969–980. [Google Scholar] [CrossRef]
- Sangüesa-Barreda, G.; Camarero, J.J.; Esper, J.; Galván, D.; Büntgen, U. A millennium-long perspective on high-elevation pine recruitment in the Spanish central Pyrenees. Can. J. For. Res. 2018, 48, 1108–1113. [Google Scholar]
- Camarero, J.J.; García-Ruiz, J.M.; Sangüesa-Barreda, G.; Galván, J.D.; Alla, A.Q.; Sanjuán, Y.; Beguería, S.; Gutiérrez, E. Recent and intense dynamics in a formerly static treeline. Arct. Ant. Alp. Res. 2015, 47, 773–783. [Google Scholar] [CrossRef]
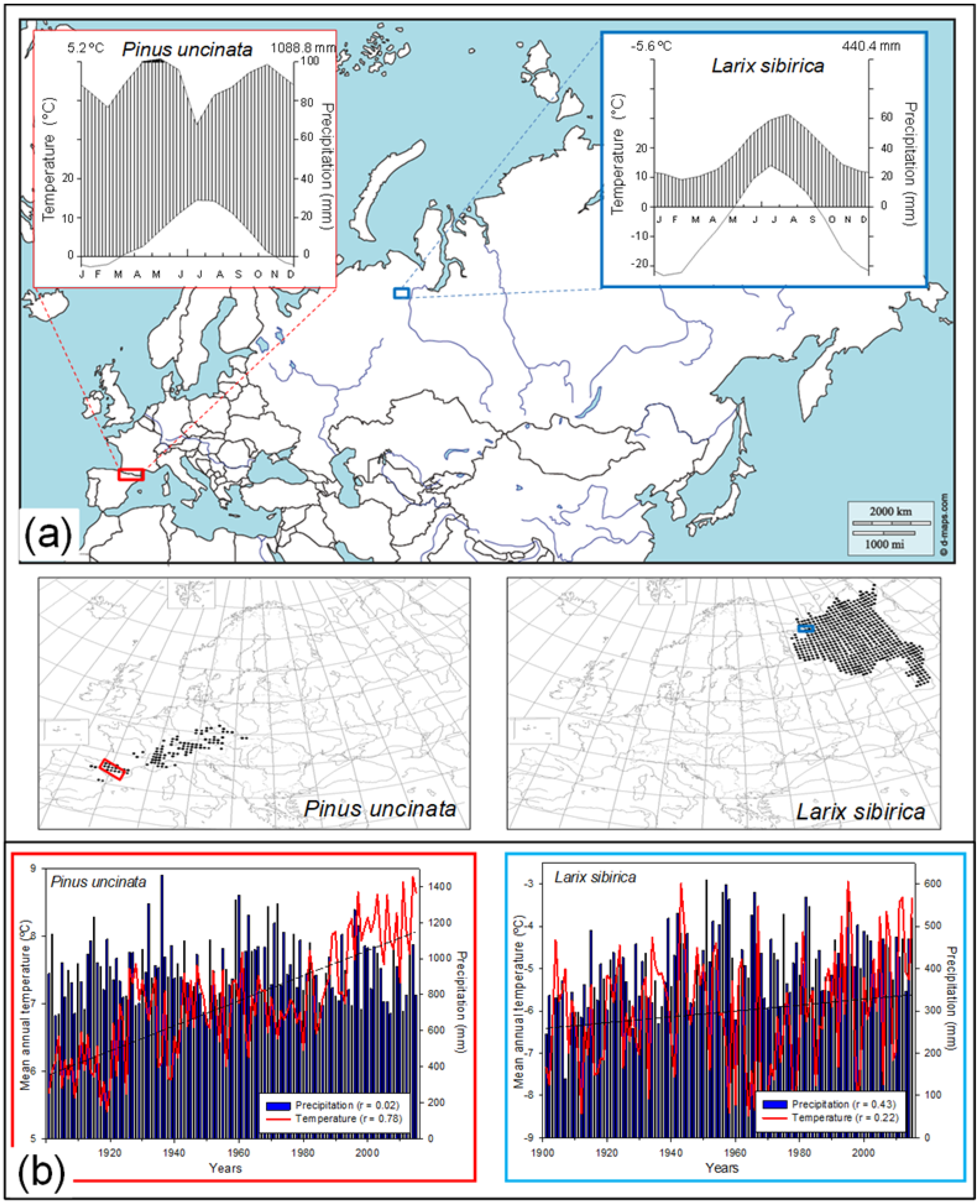
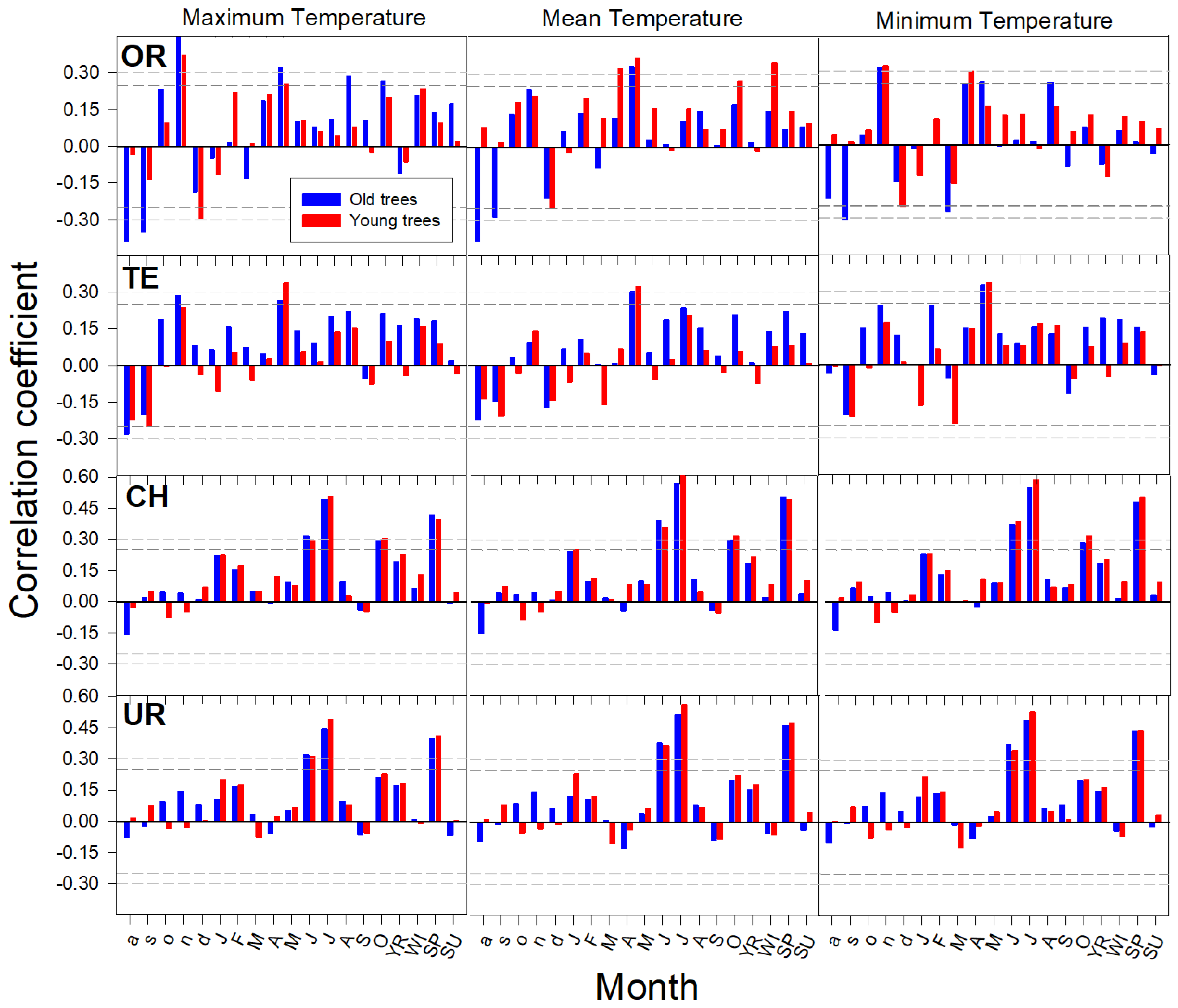
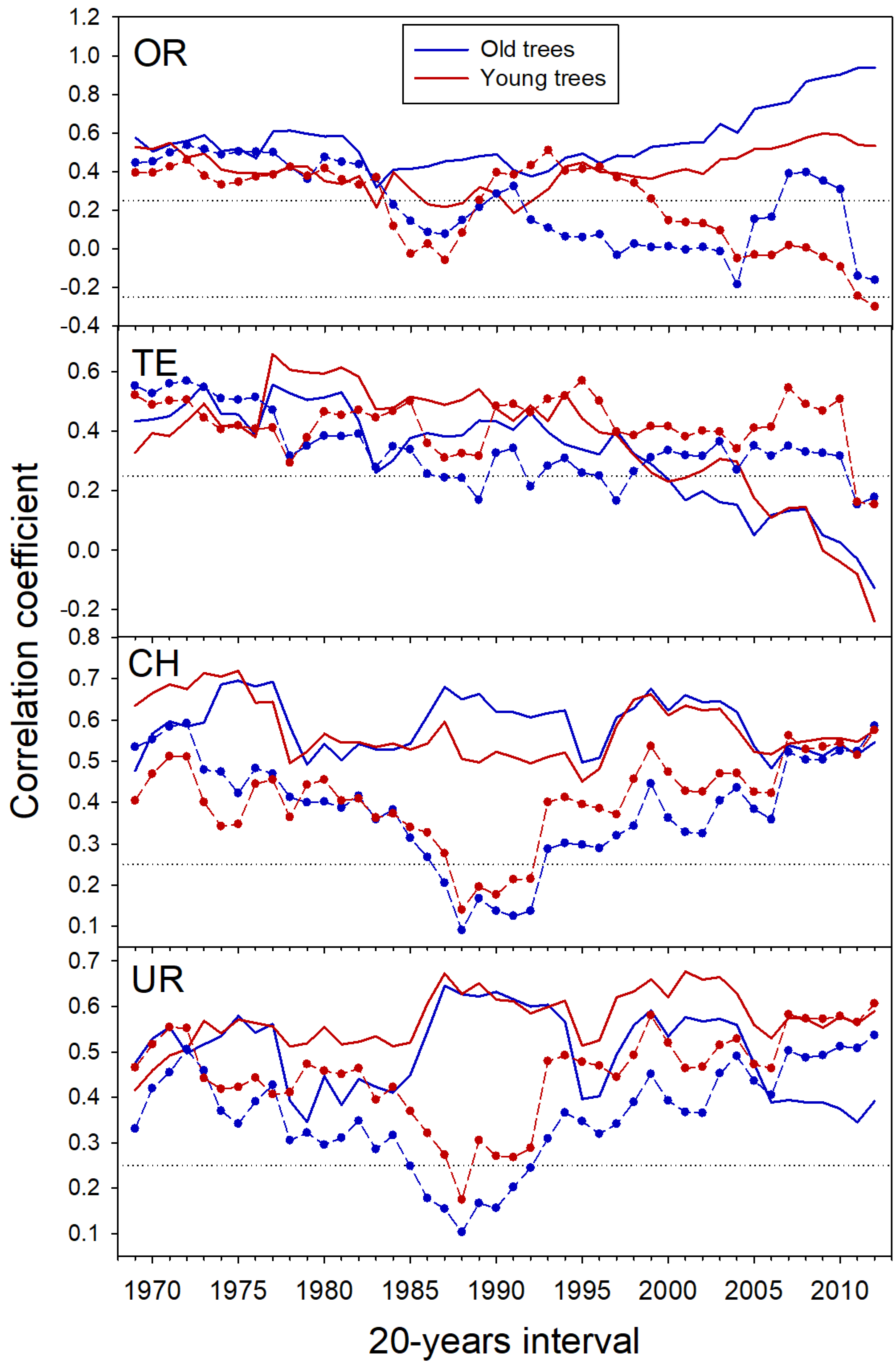
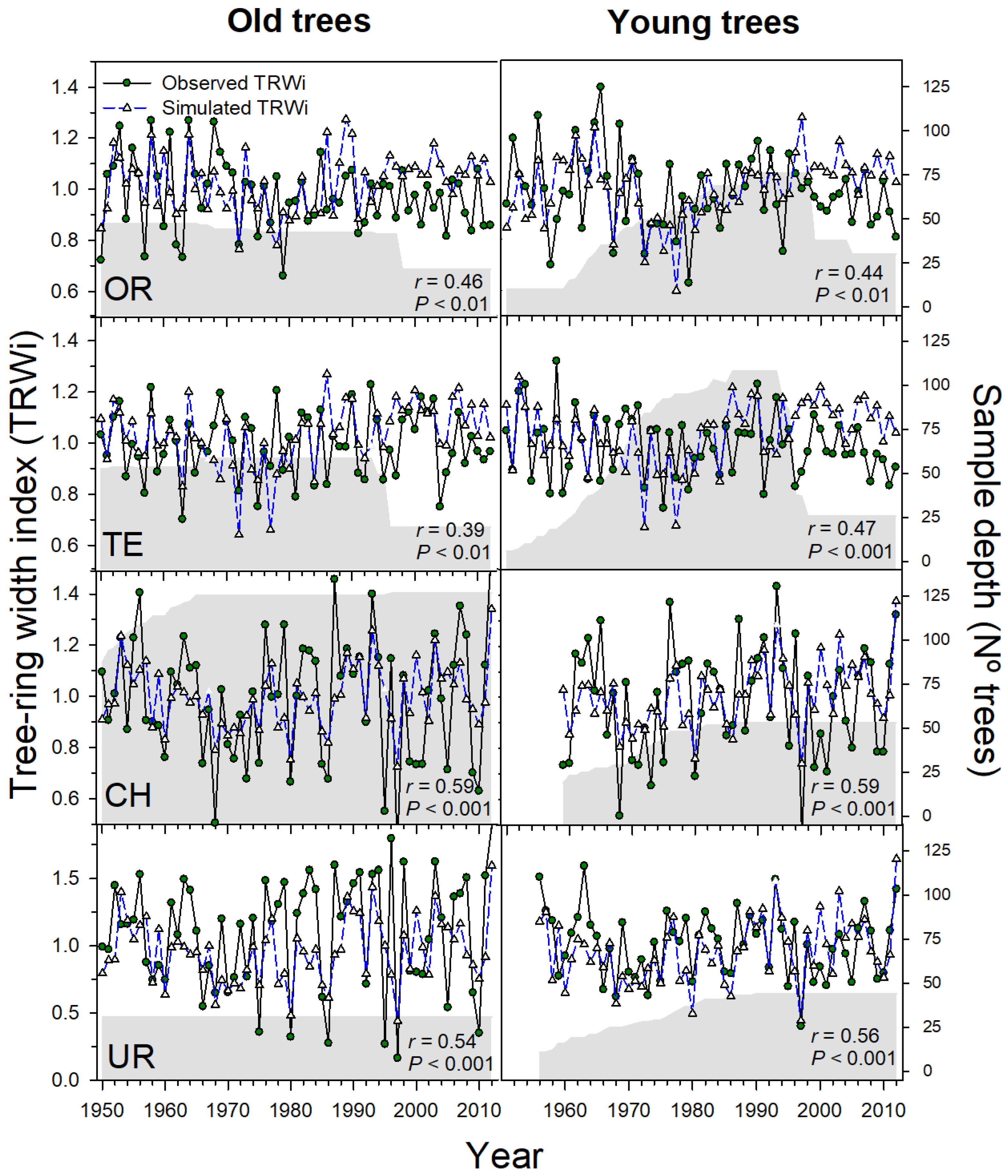
| Range | Site (Code) | Tree Species | Longitude (W/E) | Latitude (N) | Treeline‒Forest Limit Elevations (m a.s.l.) | Aspect | Slope (°) |
|---|---|---|---|---|---|---|---|
| Spanish Pyrenees | Ordesa (OR) | Mountain pine (Pinus uncinata) | 0°02′ W | 42°37′ | 2130–2100 | S | 20 |
| Tessó (TE) | 1° 03′ E | 42°36′ | 2360–2330 | NE | 35 | ||
| Russian Polar Urals | Urals treeline (CH) | Larch (Larix sibirica) | 66°40′ E | 66°32′ | 450–320 | S-SW | 20 |
| Urals treeline ecotone (UR) | 66°38′ E | 66°30′ | 220–320 | S-SW | 15 |
| Variables | Ordesa (OR) | Tessó (TE) | Urals Treeline (CH) | Urals Treeline Ecotone (UR) | ||||
|---|---|---|---|---|---|---|---|---|
| Old | Young | Old | Young | Old | Young | Old | Young | |
| N° Trees | 48 | 76 | 59 | 108 | 135 | 53 | 31 | 44 |
| Diameter at breast height (cm) | 26.5 ± 0.9a | 10.5 ± 0.6b | 31.3 ± 1.9a | 9.2 ± 0.8b | 20.7 ± 0.9a | 9.8 ± 0.5b | 19.1 ± 0.7a | 9.5 ± 0.5b |
| Tree-ring width ± SD (mm) 1 | 1.11± 0.62b | 1.58 ± 0.89a | 1.52 ± 0.73b | 1.65 ± 0.81a | 0.88 ± 0.42b | 1.17 ± 0.53a | 0.43 ± 0.22b | 1.02 ± 0.49a |
| Age ± SD (years) | 108 ± 9a | 33 ± 2b | 115 ± 7a | 32 ± 1b | 97 ± 4a | 41 ± 3b | 120 ± 15a | 42 ± 2b |
| AC 2 | 0.76 ± 0.01a | 0.57 ± 0.03b | 0.79 ± 0.01 | 0.63 ± 0.02 | 0.54 ± 0.01 | 0.56 ± 0.02 | 0.64 ± 0.03 | 0.52 ± 0.02 |
| MS | 0.24 ± 0.02b | 0.28 ± 0.01a | 0.20 ± 0.02 | 0.24 ± 0.01 | 0.36 ± 0.01a | 0.29 ± 0.01b | 0.45 ± 0.01a | 0.32 ± 0.01b |
| rbt | 0.58 ± 0.02a | 0.42 ± 0.02b | 0.47 ± 0.01 | 0.46 ± 0.01 | 0.71 ± 0.01 | 0.67 ± 0.01 | 0.81 ± 0.01a | 0.68 ± 0.01b |
| PC1 (%) | 36.2 | 30.7 | 35.1 | 31.8 | 54.9 | 57.1 | 67.7 | 56.3 |
| EPS | 0.89 | 0.90 | 0.89 | 0.91 | 0.94 | 0.95 | 0.93 | 0.94 |
| Ordesa (OR) | Tessó (TE) | Urals Treeline (CH) | Urals Treeline Ecotone (UR) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Period | IPCC Scenario | Old | Young | Old | Young | Old | Young | Old | Young |
| 1950–1975 | Observed | 0.44 | 0.61 | 0.36 | 0.28 | 0.55 | 0.54 | 0.43 | 0.56 |
| 1975–2012 | 0.39 | 0.38 | 0.35 | 0.38 | 0.60 | 0.58 | 0.57 | 0.62 | |
| 1950–2012 | 0.46 | 0.44 | 0.39 | 0.47 | 0.59 | 0.59 | 0.54 | 0.56 | |
| 2013–2050 | RCP 2.6 | 0.79 | 0.46 | 0.50 | 0.48 | 0.84 | 0.80 | 0.83 | 0.69 |
| RCP 8.5 | 0.76 | 0.39 | 0.46 | 0.42 | 0.95 | 0.75 | 0.91 | 0.77 | |
| 2050–2100 | RCP 2.6 | 0.44 | 0.32 | 0.30 | 0.29 | 0.75 | 0.58 | 0.80 | 0.60 |
| RCP 8.5 | 0.91 | 0.86 | 0.50 | 0.48 | 0.98 | 0.89 | 0.95 | 0.93 | |
| Site (Code) | Group | R2adj | F–Value | p-Value | Equations |
|---|---|---|---|---|---|
| Ordesa (OR) | Old | 0.69 | 8.65 | <0.0001 | 1.01 + 0.05tx5 + 0.07tx7 + 0.07tx9 + 0.09tx12p + 0.04tm − 0.07Pwi + 0.03Psp |
| Young | 0.45 | 5.11 | 0.0002 | 0.98 + 0.04tx2 + 0.03tx5 − 0.03tm3 + 0.03tm4 − 0.04tmwi + 0.02P2 | |
| Tessó (TE) | Old | 0.47 | 5.21 | <0.0001 | 0.99 − 0.06tx2 + 0.06tx9p − 0.03tx11p + 0.09ti2 + 0.07ti5 − 0.04ti9 − 0.04P1 |
| Young | 0.33 | 5.46 | 0.0008 | 0.99 + 0.06tx5 − 0.04ti3 − 0.02tm6 − 0.03P1 | |
| Urals treeline (CH) | Old | 0.55 | 9.96 | <0.0001 | 0.99 − 0.18tx6 − 0.11tsux + 0.05ti12p − 0.05tm4 + 0.33tm6 + 0.15tm7 |
| Young | 0.54 | 11.02 | <0.0001 | 0.98 − 0.11txsu + 0.12ti6 + 0.16tm7 − 0.04p3 + 0.04p7 | |
| Urals treeline ecotone (UR) | Old | 0.57 | 9.56 | <0.0001 | 1.09 + 0.35tx8 − 0.16ti8-0.11tm4 + 0.60tm6 + 0.42tm7 + 0.11tm12p |
| Young | 0.53 | 8.91 | <0.0001 | 1.01 + 0.16tx7 + 0.06tisp + 0.13tm6 + 0.30tm7 − 0.06P3 |
| IPCC Emission Scenarios | |||||||
|---|---|---|---|---|---|---|---|
| Observed | RCP 2.6 | RCP 8.5 | |||||
| Site | Age Group 1 | Parameter (unit) | 1950–2012 | 2013–2050 | 2050–2100 | 2013–2050 | 2050–2100 |
| Ordesa (OR) | O/Y | T1 (°C) | 3.17/2.51 | 5.38/4.71 | 6.34/5.61 | 5.65/4.83 | 5.17/7.23 |
| O/Y | T2 (°C) | 11.92/12.03 | 20.78/20.73 | 17.30/18.55 | 14.20/21.52 | 12.62/22.45 | |
| O/Y | M1 (v/v) | 0.080/0.068 | 0.037/0.018 | 0.014/0.013 | 0.031/0.037 | 0.027/0.011 | |
| O/Y | M2 (v/v) | 0.447/0.472 | 0.294/0.339 | 0.317/0.270 | 0.232/0.140 | 0.218/0.332 | |
| Tessó (TE) | O/Y | T1 (°C) | 6.83/7.69 | 8.62/8.63 | 6.69/8.82 | 5.97/4.79 | 7.73/3.98 |
| O/Y | T2 (°C) | 10.52/10.89 | 15.88/18.25 | 14.87/16.62 | 14.09/17.05 | 22.03/21.71 | |
| O/Y | M1 (v/v) | 0.011/0.014 | 0.008/0213 | 0.024/0.029 | 0.023/0.056 | 0.087/0.035 | |
| O/Y | M2 (v/v) | 0.272/0.436 | 0.213/0.346 | 0.256/0.139 | 0.247/0.431 | 0.204/0.133 | |
| Urals treeline (CH) | O/Y | T1 (°C) | 8.76/8.45 | 8.30/8.93 | 8.00/7.84 | 8.58/7.35 | 5.01/6.44 |
| O/Y | T2 (°C) | 19.18/18.86 | 20.14/10.67 | 17.21/11.26 | 18.89/14.33 | 18.00/19.55 | |
| O/Y | M1 (v/v) | 0.030/0.071 | 0.060/0.058 | 0.073/0.073 | 0.037/0.084 | 0.002/0.040 | |
| O/Y | M2 (v/v) | 0.255/0.266 | 0.224/0.292 | 0.276/0.241 | 0.167/0.335 | 0.278/0.320 | |
| Urals treeline ecotone (UR) | O/Y | T1 (°C) | 8.63/8.77 | 6.48/4.11 | 6.15/6.64 | 8.47/7.79 | 7.45/4.70 |
| O/Y | T2 (°C) | 18.31/18.20 | 11.06/11.61 | 14.90/14.61 | 15.09/17.33 | 12.17/11.00 | |
| O/Y | M1 (v/v) | 0.072/0.068 | 0.025/0.012 | 0.043/0.007 | 0.069/0.087 | 0.009/0.025 | |
| O/Y | M2 (v/v) | 0.198/0.2374 | 0.267/0.350 | 0.209/0.273 | 0.274/0.289 | 0.225/0.251 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanchez-Salguero, R.; Camarero, J.J.; Gutiérrez, E.; Gazol, A.; Sangüesa-Barreda, G.; Moiseev, P.; Linares, J.C. Climate Warming Alters Age-Dependent Growth Sensitivity to Temperature in Eurasian Alpine Treelines. Forests 2018, 9, 688. https://doi.org/10.3390/f9110688
Sanchez-Salguero R, Camarero JJ, Gutiérrez E, Gazol A, Sangüesa-Barreda G, Moiseev P, Linares JC. Climate Warming Alters Age-Dependent Growth Sensitivity to Temperature in Eurasian Alpine Treelines. Forests. 2018; 9(11):688. https://doi.org/10.3390/f9110688
Chicago/Turabian StyleSanchez-Salguero, Raúl, J. Julio Camarero, Emilia Gutiérrez, Antonio Gazol, Gabriel Sangüesa-Barreda, Pavel Moiseev, and Juan C. Linares. 2018. "Climate Warming Alters Age-Dependent Growth Sensitivity to Temperature in Eurasian Alpine Treelines" Forests 9, no. 11: 688. https://doi.org/10.3390/f9110688
APA StyleSanchez-Salguero, R., Camarero, J. J., Gutiérrez, E., Gazol, A., Sangüesa-Barreda, G., Moiseev, P., & Linares, J. C. (2018). Climate Warming Alters Age-Dependent Growth Sensitivity to Temperature in Eurasian Alpine Treelines. Forests, 9(11), 688. https://doi.org/10.3390/f9110688








