Site- and Species-Specific Influences on Sub-Alpine Conifer Growth in Mt. Rainier National Park, USA
Abstract
:1. Introduction
2. Materials and Methods
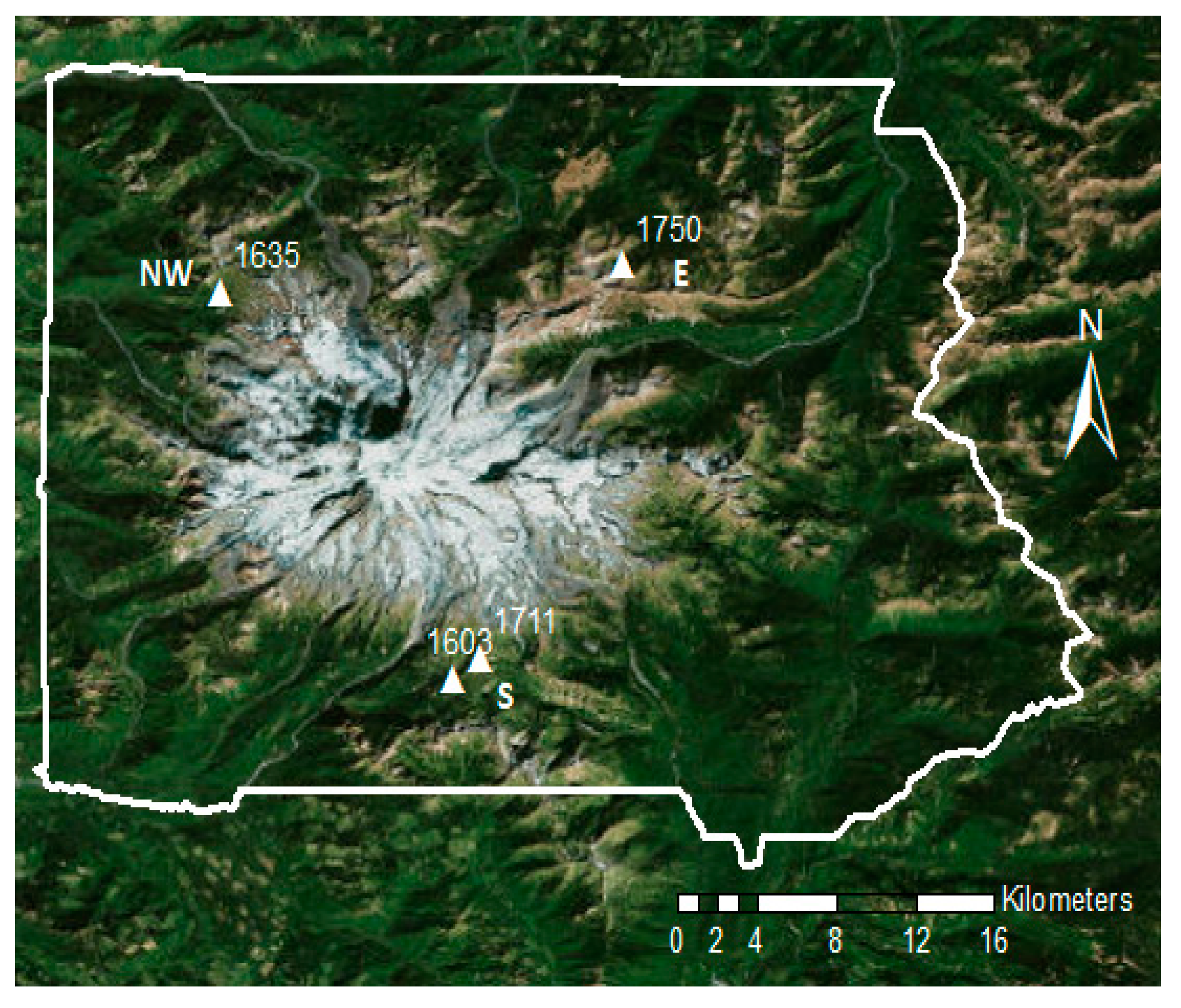
2.1. Study Site and Species
2.2. Tree Growth Data
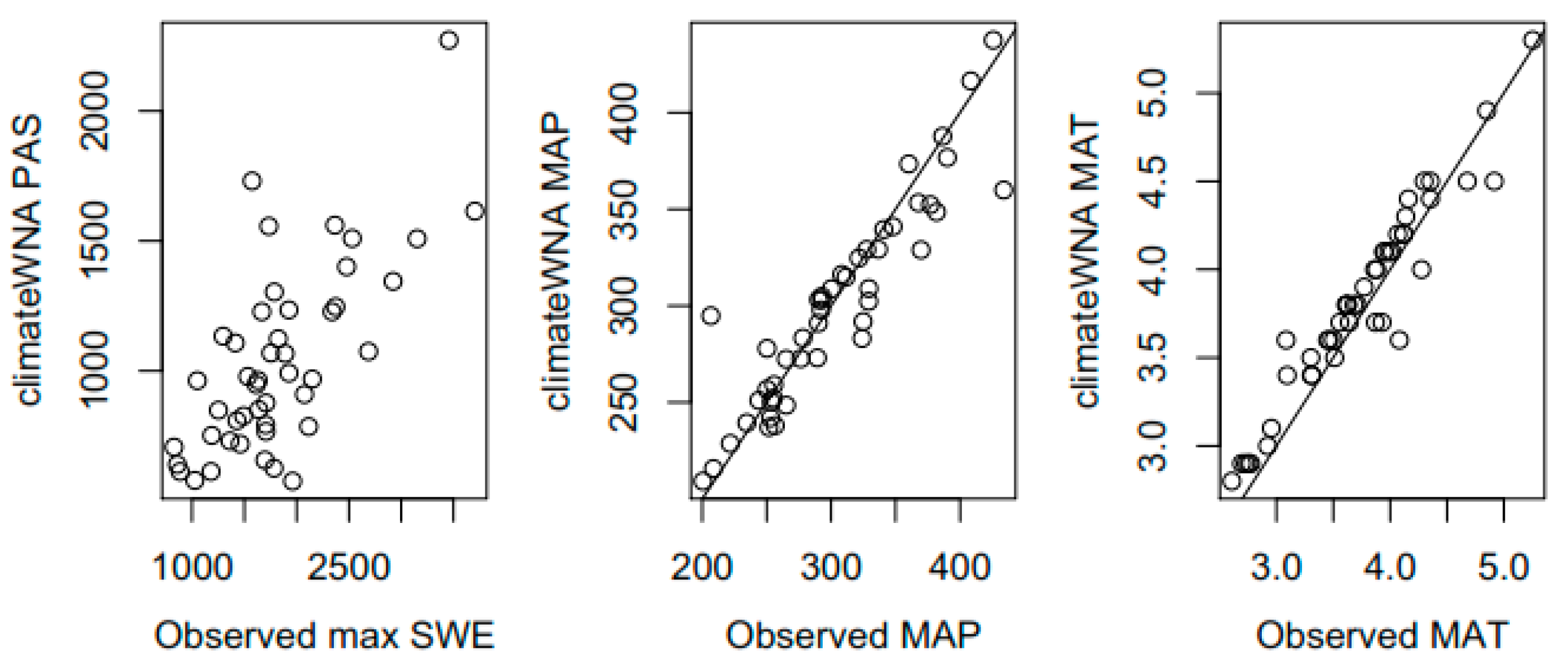
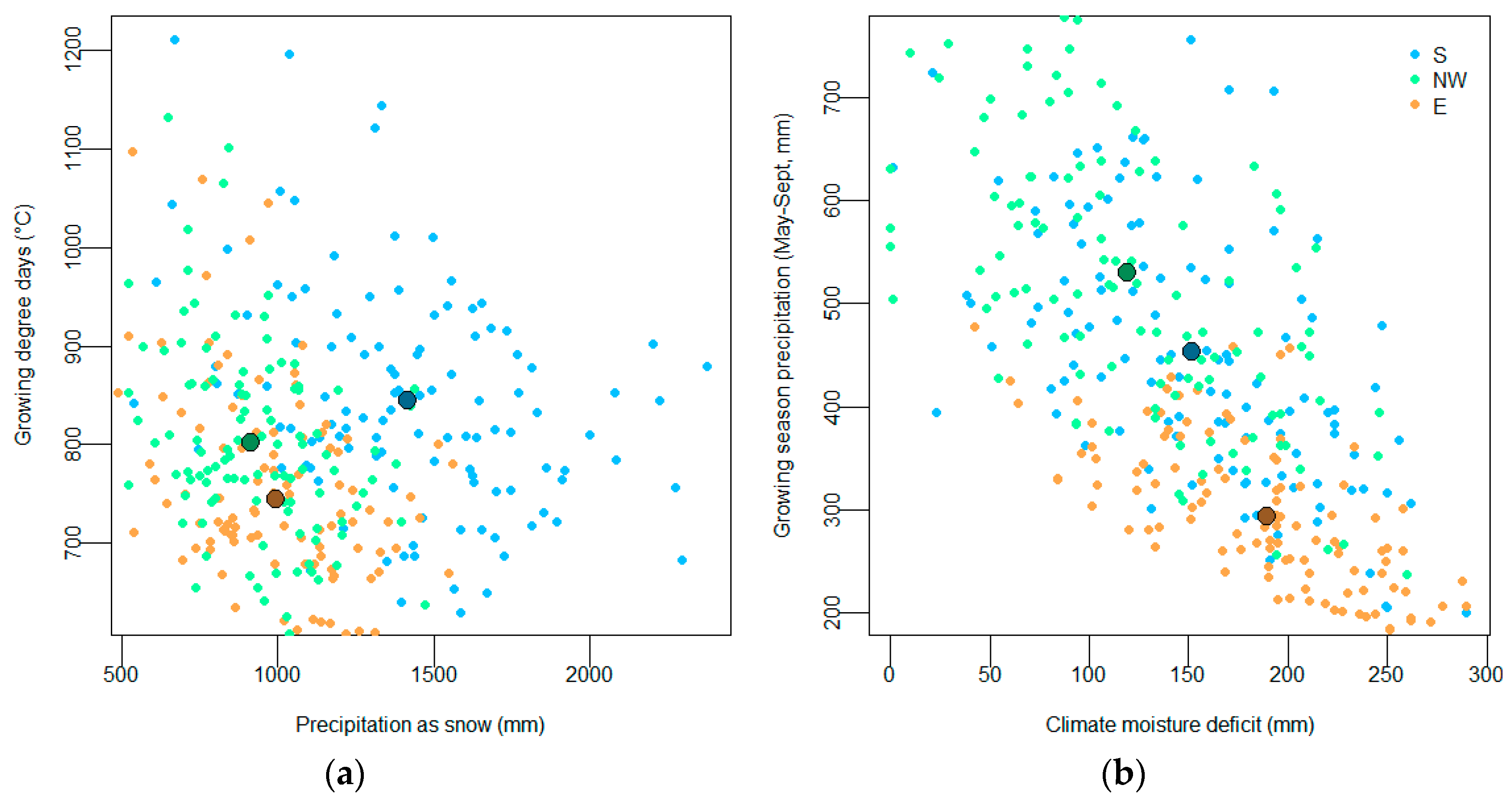
2.3. Climate Data
2.4. Climate Variable Selection, Model Selection and Statistical Analyses
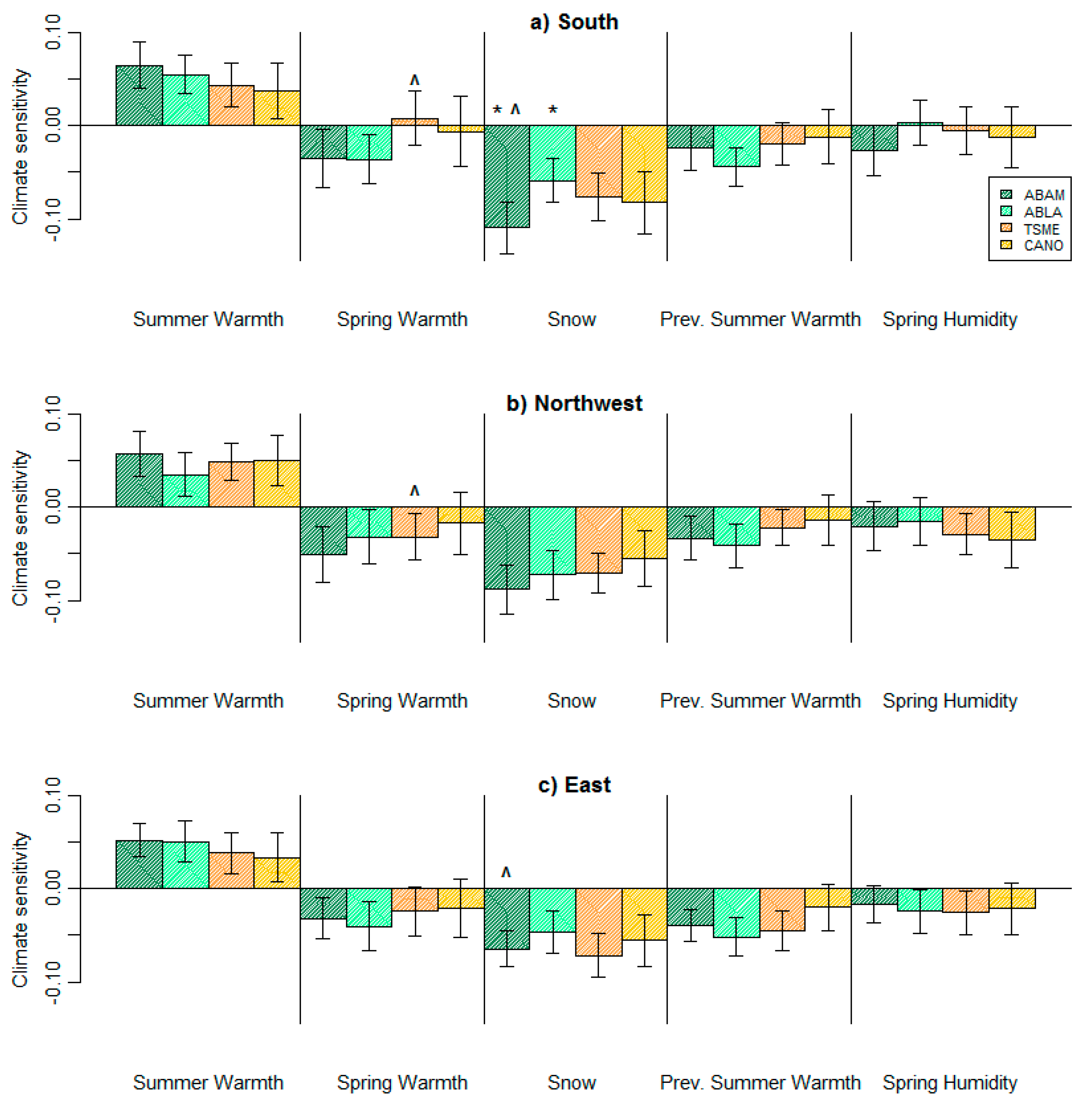
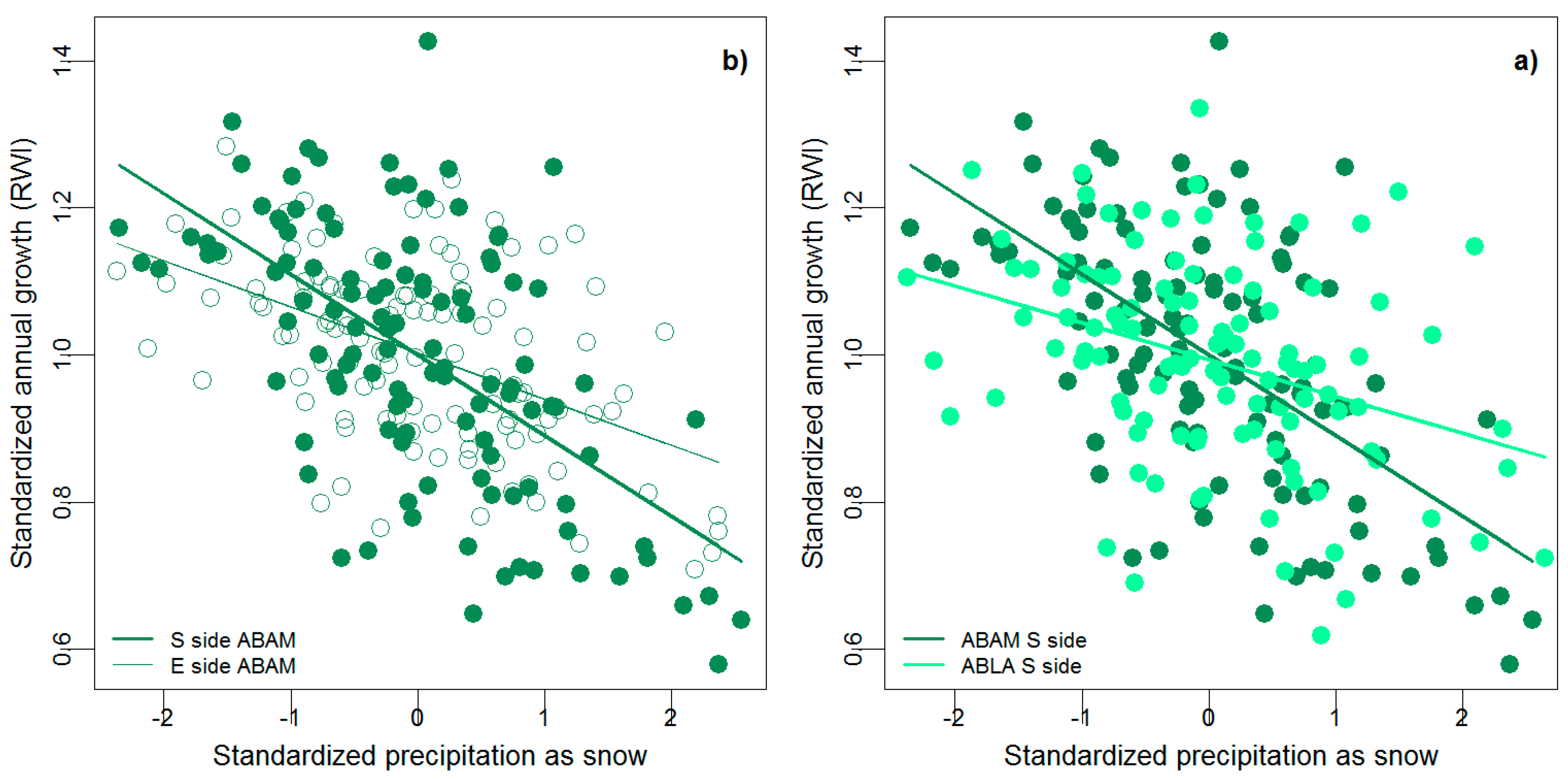
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| South | Northwest | East | |
|---|---|---|---|
| ABAM | 20 | 19 | 21 |
| ABLA | 10 | 19 | 20 |
| TSME | 20 | 19 | 19 |
| CANO | 19 | 20 | 19 |
| Species/Site | Comparison | F-Statistic | df | t-Value | p-Value |
|---|---|---|---|---|---|
| ABAM/SOUTH | siteEAST:PAS | 20.14 on 15 | 323 | 2.544 | 0.01143 |
| TSME/SOUTH | siteNORTHWEST:DD5_sp | 16.29 on 15 | 324 | −2.059 | 0.04031 |
| ABAM/SOUTH | speciesABLA:PAS | 14.67 on 20 | 428 | 2.542 | 0.01137 |
References
- Dirnböck, T.; Dullinger, S.; Grabherr, G. A regional impact assessment of climate and land-use change on alpine vegetation. J. Biogeogr. 2003, 30, 401–417. [Google Scholar] [CrossRef]
- Guisan, A.; Theurillat, J.-P. Assessing alpine plant vulnerability to climate change: A modeling perspective. Integr. Assess. 2000, 1, 307–320. [Google Scholar] [CrossRef]
- Rochefort, R.M.; Little, R.L.; Woodward, A.; Peterson, D.L. Changes in sub-alpine tree distribution in western North America: A review of climatic and other causal factors. Holocene 1994, 4, 89–100. [Google Scholar] [CrossRef]
- Haugo, R.E.; Halpern, C.B.; Bakker, J.D. Landscape context and long-term tree influences shape the dynamics of forest-meadow ecotones in mountain ecosystems. Ecosphere 2011, 2. [Google Scholar] [CrossRef]
- Intergovernmental Panel on Climate Change (IPCC). Climate Change 2014: Synthesis Report; Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; in IPCC AR5 Synthesis Report Website; Core Writing Team, Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014; 151p. [Google Scholar]
- Krahulec, F.C.A.; Skálová, H.; Herben, T.; Hadincová, V.; Wildová, R.; Pecháčková, S. Vegetation changes following sheep grazing in abandoned mountain meadows. Appl. Veg. Sci. 2001, 4, 97–102. [Google Scholar] [CrossRef]
- Zhang, C.; Willis, C.G.; Klein, J.A.; Ma, Z.; Li, J.; Zhou, H.; Zhao, X. Recovery of plant species diversity during long-term experimental warming of a species-rich alpine meadow community on the Qinghai-Tibet plateau. Biol. Conserv. 2017, 213, 218–224. [Google Scholar] [CrossRef]
- Smithwick, E.A.H.; Harmon, M.E.; Remillard, S.M.; Acker, S.A.; Franklin, J.F. Potential upper bounds of carbon stores in forests of the Pacific Northwest. Ecol. Appl. 2002, 12, 1303–1317. [Google Scholar] [CrossRef]
- Wilson, A.; Bacher, K.; Breckheimer, I.; Lundquist, J.; Rochefort, R.; Theobald, E.; Whiteaker, L.; HilleRisLambers, J. Monitoring wildflower phenology using traditional science, citizen science, and crowd sourcing. Park Sci. 2017, 33, 17–26. Available online: https://www.nps.gov/ParkScience/articles/parkscience33_1_17-26_wilson_et_al_3850.htm (accessed on 18 September 2017).
- Soja, A.J.; Tchebakova, N.M.; French, N.H.; Flannigan, M.D.; Shugart, H.H.; Stocks, B.J.; Sukhinin, A.I.; Parfenova, E.; Chapin, F.S.; Stackhouse, P.W. Climate-induced boreal forest change: Predictions versus current observations. Glob. Planet. Chang. 2007, 56, 274–296. [Google Scholar] [CrossRef]
- Sherriff, R.L.; Miller, A.E.; Muth, K.; Schriver, M.; Batzel, R. Spruce growth responses to warming vary by ecoregion and ecosystem type near the forest-tundra boundary in south-west Alaska. J. Biogeogr. 2017, 44, 1457–1468. [Google Scholar] [CrossRef]
- Dijak, W.D.; Hanberry, B.B.; Fraser, J.S.; He, H.S.; Wang, W.J.; Thompson, F.R. Revision and application of the LINKAGES model to simulate forest growth in central hardwood landscapes in response to climate change. Landsc. Ecol. 2016, 32, 1365–1384. [Google Scholar] [CrossRef]
- Saxe, H.; Cannell, M.G.R.; Johnsen, Ø.; Ryan, M.G.; Vourlitis, G. Tree and forest functioning in response to global warming. New Phytol. 2001, 149, 369–399. [Google Scholar] [CrossRef]
- Grace, J.; Berninger, F.; Nagy, L. Impacts of climate change on the Tree Line. Ann. Bot.-Lond. 2002, 90, 537–544. [Google Scholar] [CrossRef]
- Nicklen, E.F.; Roland, C.A.; Ruess, R.W.; Schmidt, J.H.; Lloyd, A.H. Local site conditions drive climate-growth responses of Picea mariana and Picea glauca in interior Alaska. Ecosphere 2016, 7, e01507. [Google Scholar] [CrossRef]
- Suarez, M.C.A.D.L.; Villalba, R.; Mundo, I.A.; Schroeder, N. Sensitivity of Nothofagus dombeyi tree growth to climate changes along a precipitation gradient in northern Patagonia, Argentina. Trees 2015, 29, 1053–1067. [Google Scholar] [CrossRef]
- Gedalof, Z.; Smith, D.J. Dendroclimatic response of mountain hemlock (Tsuga mertensiana) in Pacific North America. Can. J. For. Res. 2001, 31, 322–332. [Google Scholar] [CrossRef]
- Ettl, G.J.; Peterson, D.L. Growth response of subalpine fir (Abies lasiocarpa) to climate in the Olympic Mountains, Washington, USA. Glob. Chang. Biol. 1995, 1, 213–230. [Google Scholar] [CrossRef]
- Restaino, C.M.; Peterson, D.L.; Littell, J. Increased water deficit decreases Douglas fir growth throughout western US forests. Proc. Natl. Acad. Sci. USA 2016, 113, 9557–9562. [Google Scholar] [CrossRef] [PubMed]
- Littell, J.S.; Peterson, D.L.; Tjoelker, M. Douglas-fir growth in mountain ecosystems: Water limits tree growth from stand to region. Ecol. Monogr. 2008, 78, 349–368. [Google Scholar] [CrossRef]
- Wilson, R.J.S.; Hopfmueller, M. Dendrochronological investigations of Norway spruce along an elevational transect in the Bavarian Forest, Germany. Dendrochronologia 2001, 19, 67–79. [Google Scholar]
- Griesbauer, H.P.; Green, D.S. Regional and ecological patterns in interior Douglas-fir climate–growth relationships in British Columbia, Canada. Can. J. For. Res. 2010, 40, 308–321. [Google Scholar] [CrossRef]
- Griesbauer, H.P.; Green, D.S.; O’Neill, G.A. Using a spatiotemporal climate model to assess population-level Douglas-fir growth sensitivity to climate change across large climatic gradients in British Columbia, Canada. For. Ecol. Manag. 2011, 261, 589–600. [Google Scholar] [CrossRef]
- Griesbauer, H.P.; Green, D.S. Geographic and temporal patterns in white spruce climate–growth relationships in Yukon, Canada. For. Ecol. Manag. 2012, 267, 215–227. [Google Scholar] [CrossRef]
- Friedrichs, D.A.; Trouet, V.; Büntgen, U.; Frank, D.C.; Esper, J.; Neuwirth, B.; Löffler, J. Species-specific climate sensitivity of tree growth in Central-West Germany. Trees 2009, 23, 729–739. [Google Scholar] [CrossRef]
- Brienen, R.J.W.; Zuidema, P.A. Relating tree growth to rainfall in Bolivian rain forests: A test for six species using tree ring analysis. Oecologia 2005, 146. [Google Scholar] [CrossRef] [PubMed]
- Gillner, S.; Bräuning, A.; Roloff, A. Dendrochronological analysis of urban trees: Climatic response and impact of drought on frequently used tree species. Trees 2014, 28, 1079–1093. [Google Scholar] [CrossRef]
- Kint, V.; Aertsen, W.; Campioli, M.; Vansteenkiste, D.; Delcloo, A.; Muys, B. Radial growth change of temperate tree species in response to altered regional climate and air quality in the period 1901–2008. Clim. Chang. 2012, 115, 343–363. [Google Scholar] [CrossRef]
- Nakawatase, J.M.; Peterson, D.L. Spatial variability in forest growth–climate relationships in the Olympic Mountains, Washington. Can. J. For. Res. 2006, 36, 77–91. [Google Scholar] [CrossRef]
- Deck, C.; Wiles, G.; Frederick, S.; Matsovsky, V.; Kuderina, T.; D’Arrigo, R.; Solomina, O.; Wiesenberg, N. Climate response of larch and birch forests across an elevational transect and hemisphere-wide comparisons, Kamchatka Peninsula, Russian Far East. Forests 2017, 8, 315. [Google Scholar] [CrossRef]
- Peterson, D.W.; Peterson, D.L. Effects of climate on radial growth of subalpine conifers in the North Cascade Mountains. Can. J. For. Res. 1994, 24, 1921–1932. [Google Scholar] [CrossRef]
- Liang, E.; Shao, X.; Eckstein, D.; Huang, L.; Liu, X. Topography- and species-dependent growth responses of Sabina przewalskii and Picea crassifolia to climate on the northeast Tibetan Plateau. For. Ecol. Manag. 2006, 236, 268–277. [Google Scholar] [CrossRef]
- Daly, C.; Halbleib, M.; Smith, J.I.; Gibson, W.P.; Doggett, M.K.; Taylor, G.H.; Curtis, J.; Pasteris, P.P. Physiographically sensitive mapping of climatological temperature and precipitation across the conterminous United States. Int. J. Climatol. 2008, 28, 2031–2064. [Google Scholar] [CrossRef]
- Wang, T.; Hamann, A.; Spittlehouse, D.L.; Murdock, T.Q. ClimateWNA—High-Resolution Spatial Climate Data for Western North America. J. Appl. Meteorol. Clim. 2012, 51, 16–29. [Google Scholar] [CrossRef]
- Kroiss, S.J.; Hillerislambers, J. Recruitment limitation of long-lived conifers: Implications for climate change responses. Ecology 2015, 96, 1286–1297. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, A.K.; Ford, K.R.; Hillerislambers, J. Climate determines upper, but not lower, altitudinal range limits of Pacific Northwest conifers. Ecology 2011, 92, 1323–1331. [Google Scholar] [CrossRef] [PubMed]
- Hillerislambers, J.; Anderegg, L.D.L.; Breckheimer, I.; Burns, K.M.; Ettinger, A.K.; Franklin, J.F.; Freund, J.A.; Ford, K.R.; Krolss, S.J. Implications of climate change for turnover in forest composition. Northwest Sci. 2015, 89, 201–218. [Google Scholar] [CrossRef]
- Franklin, J.F.; Dyrness, C.T. Natural Vegetation of Oregon and Washington; Report PNW-8; USDA Forest Service, Pacific Northwest Forest and Range Experiment Station: Portland, OR, USA, 1973; 417p.
- Stokes, M.A.; Smiley, T.L. An Introduction to Tree-Ring Dating; University of Chicago Press: Chicago, IL, USA, 1968. [Google Scholar]
- Regent Instruments Inc. WinDendro; Version 2009b; Regent Instruments Inc.: Quebec, QC, Canada, 2009. [Google Scholar]
- Holmes, R.L. Computer-assisted quality control in tree-ring dating and measurement. Tree-Ring Bull. 1983, 43, 69–78. [Google Scholar]
- Grissino-Mayer, H. Evaluating crossdating accuracy: A manual and tutorial for the computer program COFECHA. Tree-Ring Res. 2001, 57, 205–221. [Google Scholar]
- Bunn, A.G. Statistical and visual crossdating in R using the dplR library. Dendrochronologia 2010, 28, 251–258. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016; Available online: https://www.R-project.org/ (accessed on 25 September 2017).
- Hamann, A.; Wang, T. Potential effects of climate change on ecosystem and tree species distribution in British Columbia. Ecology 2006, 87, 2773–2786. [Google Scholar] [CrossRef]
- DeLong, C.; Griesbauer, H.; Mackenzie, W.; Foord, V. Corroboration of biogeoclimatic ecosystem classification climate zonation by spatially modelled climate data. BC J. Ecosyst. Manag. 2010, 10, 49–64. [Google Scholar]
- Daly, C.; Gibson, W.; Taylor, G.; Johnson, G.; Pasteris, P. A knowledge-based approach to the statistical mapping of climate. Clim. Res. 2002, 22, 99–113. [Google Scholar] [CrossRef]
- Wang, T.; Hamann, A.; Spittlehouse, D.; Carroll, C. Locally downscaled and spatially customizable climate data for historical and future periods for North America. PLoS ONE 2016, 11, e0156720. [Google Scholar] [CrossRef] [PubMed]
- GPS Geoplaner Online. Available online: https://geoplaner.com (accessed on 12 March 2017).
- Zang, C.; Biondi, F. Treeclim: An R package for the numerical calibration of proxy-climate relationships. Ecography 2015, 38, 431–436. [Google Scholar] [CrossRef]
- Hargreaves, G.H.; Samani, Z.A. Reference crop evapotranspiration from temperature. Appl. Eng. Agric. 1985, 1, 96–99. [Google Scholar] [CrossRef]
- Bartoń, K. MuMIn: Multi-Model Inference, R Package Version 1.15.6; 2016. Available online: https://CRAN.R-project.org/package=MuMIn (accessed on 25 September 2017).
- Babst, F.; Poulter, B.; Trouet, V.; Tan, K.; Neuwirth, B.; Wilson, R.; Carrer, M.; Grabner, M.; Tegel, W.; Levanic, T.; et al. Site- and species-specific responses of forest growth to climate across the European continent. Glob. Ecol. Biogeogr. 2013, 22, 706–717. [Google Scholar] [CrossRef]
- Filip, G.M.; Schmitt, C.L. Rx for Abies: Silvicultural Options for Diseased Firs in Oregon and Washington; Gen. Tech. Rep. NW-GTR-252; U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station: Portland, OR, USA, 1990; 34p.
- Klinka, K.; Krajina, V.J.; Ceska, A.; Scagel, A.M. Indicator Plants of Coastal British Columbia; University of British Columbia Press: Vancouver, BC, Canada, 1989; 288p. [Google Scholar]
- Dorangeville, L.; Duchesne, L.; Houle, D.; Kneeshaw, D.; Cote, B.; Pederson, N. Northeastern North America as a potential refugium for boreal forests in a warming climate. Science 2016, 352, 1452–1455. [Google Scholar] [CrossRef] [PubMed]
- Leonelli, G.; Pelfini, M.; Battipaglia, G.; Cherubini, P. Site-aspect influence on climate sensitivity over time of a high-altitude Pinus cembra tree-ring network. Clim. Chang. 2009, 96, 185–201. [Google Scholar] [CrossRef]
- Stage, A.R.; Salas, C. Composition and productivity. For. Sci. 2007, 53, 486–492. [Google Scholar]
- Primicia, I.; Camarero, J.J.; Janda, P.; Čada, V.; Morrissey, R.C.; Trotsiuk, V.; Bače, R.; Teodosiu, M.; Svoboda, M. Age, competition, disturbance and elevation effects on tree and stand growth response of primary Picea abies forest to climate. For. Ecol. Manag. 2015, 354, 77–86. [Google Scholar] [CrossRef]
- Raney, P.A.; Leopold, D.J.; Dovciak, M.; Beier, C.M. Hydrologic position mediates sensitivity of tree growth to climate: Groundwater subsidies provide a thermal buffer effect in wetlands. For. Ecol. Manag. 2016, 379, 70–80. [Google Scholar] [CrossRef]
- Sakai, A.; Matsui, K.; Kabeya, D.; Sakai, S. Altitudinal variation in lifetime growth trajectory and reproductive schedule of a sub-alpine conifer, Abies mariesii. Evol. Ecol. Res. 2003, 5, 671–689. [Google Scholar]
- Briffa, K.R.; Schweingruber, F.H.; Jones, P.D.; Osborn, T.J.; Shiyatov, S.G.; Vaganov, E.A. Reduced sensitivity of recent tree-growth to temperature at high northern latitudes. Nature 1998, 391, 678–682. [Google Scholar] [CrossRef]
- Carrer, M.; Urbinati, C. Long-term change in the sensitivity of tree-ring growth to climate forcing in Larix decidua. New Phytol. 2006, 170, 861–872. [Google Scholar] [CrossRef] [PubMed]
- Marcinkowski, K.; Peterson, D.L.; Ettl, G.J. Nonstationary temporal response of mountain hemlock growth to climatic variability in the North Cascade Range, Washington, USA. Can. J. For. Res. 2015, 45, 676–688. [Google Scholar] [CrossRef]

| Species/Site | GDD_sm | GDD_sp | PAS | Prev_GDD_sm | RH_sp |
|---|---|---|---|---|---|
| ABAM/S | 1.00 | 0.74 | 1.00 | 0.83 | 0.65 |
| ABAM/NW | 1.00 | 1.00 | 1.00 | 1.00 | 0.51 |
| ABAM/E | 1.00 | 1.00 | 1.00 | 1.00 | 0.56 |
| ABLA/S | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| ABLA/NW | 0.97 | 0.65 | 1.00 | 1.00 | 0.33 |
| ABLA/E | 1.00 | 1.00 | 1.00 | 1.00 | 0.72 |
| TSME/S | 1.00 | 0.23 | 1.00 | 0.58 | 0.24 |
| TSME/NW | 1.00 | 0.92 | 1.00 | 0.82 | 0.94 |
| TSME/E | 1.00 | 0.47 | 1.00 | 1.00 | 0.73 |
| CANO/S | 0.89 | 0.21 | 1.00 | 0.31 | 0.29 |
| CANO/NW | 1.00 | 0.36 | 1.00 | 0.50 | 0.83 |
| CANO/E | 0.86 | 0.33 | 1.00 | 0.62 | 0.43 |
| Average | 0.98 | 0.66 | 1.00 | 0.81 | 0.60 |
| Model | df | ΔAIC | R2 |
|---|---|---|---|
| rwi~1 | 2 | 572.80 | 0 |
| rwi~climate | 7 | 0 | 0.349 |
| rwi~climate*site | 17 | −2.42 | 0.360 |
| ◊ rwi~climate*species | 22 | −8.20 | 0.367 |
| rwi~climate*species*site | 62 | 31.90 | 0.386 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Legendre-Fixx, M.; Anderegg, L.D.L.; Ettinger, A.K.; HilleRisLambers, J. Site- and Species-Specific Influences on Sub-Alpine Conifer Growth in Mt. Rainier National Park, USA. Forests 2018, 9, 1. https://doi.org/10.3390/f9010001
Legendre-Fixx M, Anderegg LDL, Ettinger AK, HilleRisLambers J. Site- and Species-Specific Influences on Sub-Alpine Conifer Growth in Mt. Rainier National Park, USA. Forests. 2018; 9(1):1. https://doi.org/10.3390/f9010001
Chicago/Turabian StyleLegendre-Fixx, Myesa, Leander D. L. Anderegg, Ailene K. Ettinger, and Janneke HilleRisLambers. 2018. "Site- and Species-Specific Influences on Sub-Alpine Conifer Growth in Mt. Rainier National Park, USA" Forests 9, no. 1: 1. https://doi.org/10.3390/f9010001
APA StyleLegendre-Fixx, M., Anderegg, L. D. L., Ettinger, A. K., & HilleRisLambers, J. (2018). Site- and Species-Specific Influences on Sub-Alpine Conifer Growth in Mt. Rainier National Park, USA. Forests, 9(1), 1. https://doi.org/10.3390/f9010001





