Rapid Shifts in Soil Nutrients and Decomposition Enzyme Activity in Early Succession Following Forest Fire
Abstract
1. Introduction
2. Methods
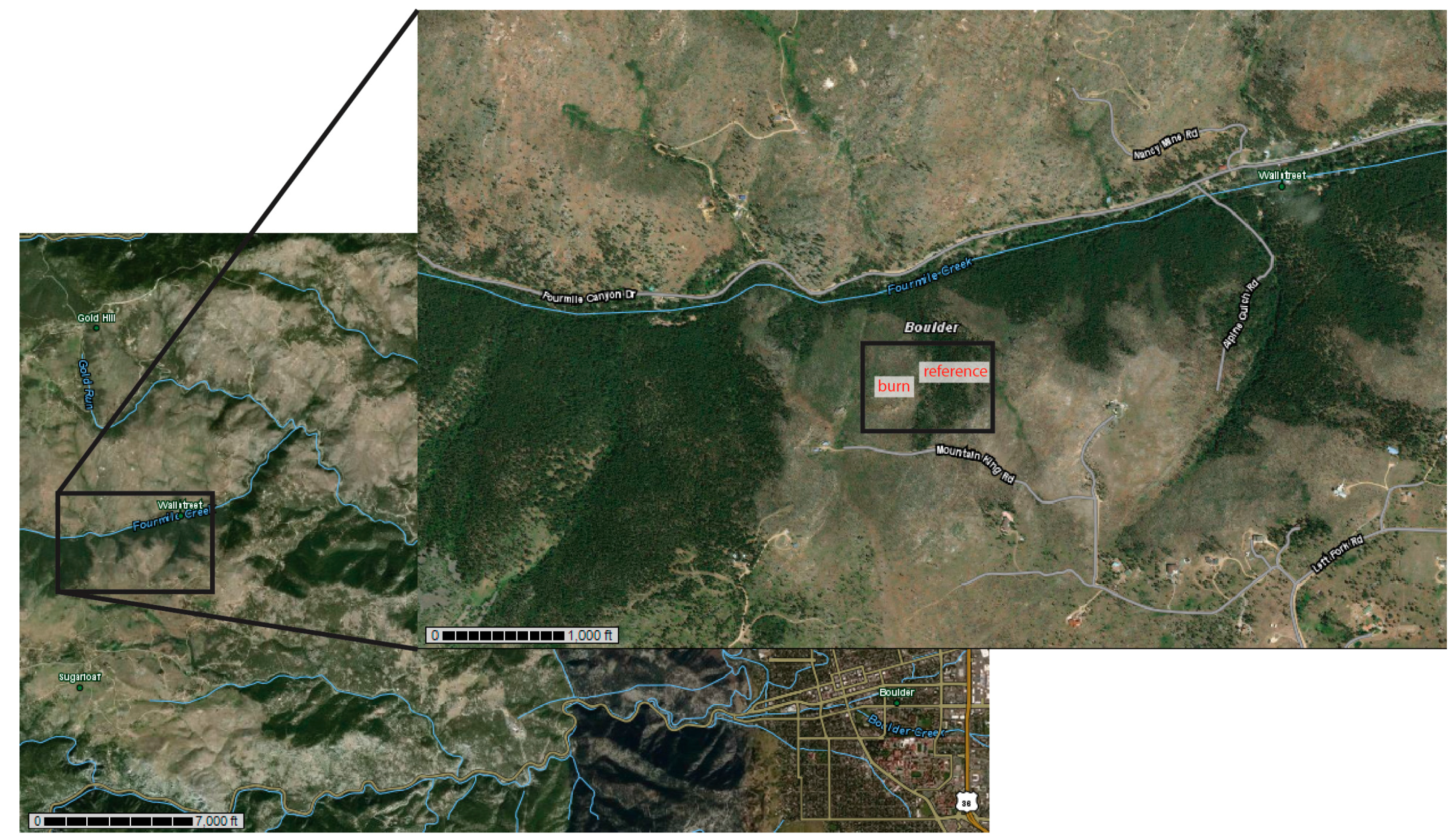
2.1. Site and Soil Properties
2.2. Enzyme Analysis
2.3. Statistical Analysis
2.4. Data Availability
3. Results
3.1. Extracellular Enzyme Activities
3.2. Soil Properties
3.3. Soil Properties and Extracellular Enzyme Activity
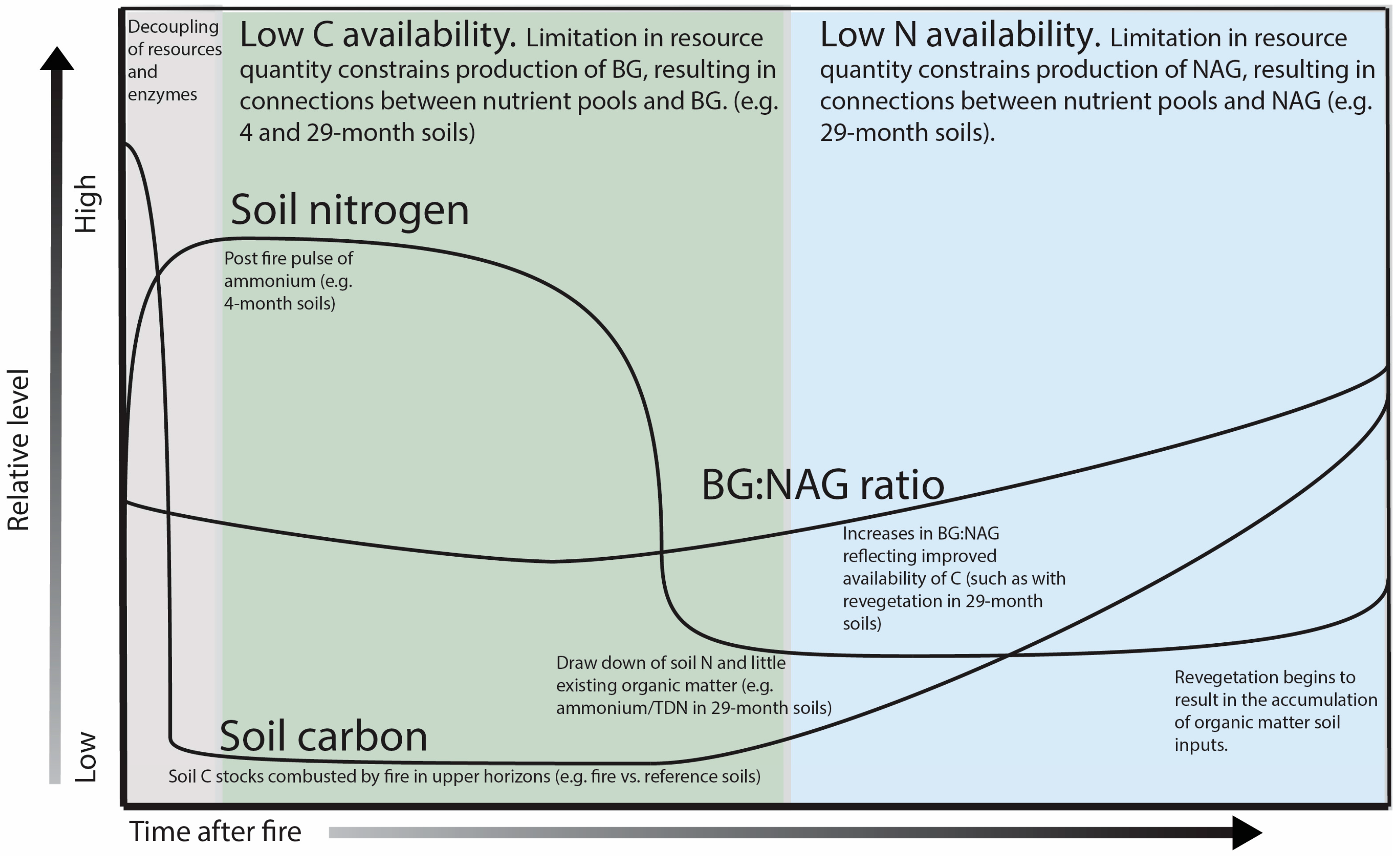
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Westerling, A.L. Warming and Earlier Spring Increase Western U.S. Forest Wildfire Activity. Science 2006, 313, 940–943. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.D.; Safford, H.D.; Crimmins, M.; Thode, A.E. Quantitative Evidence for Increasing Forest Fire Severity in the Sierra Nevada and Southern Cascade Mountains, California and Nevada, USA. Ecosystems 2009, 12, 16–32. [Google Scholar] [CrossRef]
- Rocca, M.E.; Brown, P.M.; MacDonald, L.H.; Carrico, C.M. Climate change impacts on fire regimes and key ecosystem services in Rocky Mountain forests. For. Ecol. Manag. 2014, 327, 290–305. [Google Scholar] [CrossRef]
- Graham, E.B.; Wieder, W.R.; Leff, J.W.; Weintraub, S.R.; Townsend, A.R.; Cleveland, C.C.; Philippot, L.; Nemergut, D.R. Do we need to understand microbial communities to predict ecosystem function? A comparison of statistical models of nitrogen cycling processes. Soil Biol. Biochem. 2014, 68, 279–282. [Google Scholar] [CrossRef]
- Graham, E.B.; Knelman, J.E.; Schindlbacher, A.; Siciliano, S.; Breulmann, M.; Yannarell, A.; Beman, J.M.; Abell, G.; Philippot, L.; Prosser, J.; et al. Microbes as Engines of Ecosystem Function: When Does Community Structure Enhance Predictions of Ecosystem Processes? Terr. Microbiol. 2016, 7, 214. [Google Scholar] [CrossRef] [PubMed]
- Holden, S.R.; Rogers, B.M.; Treseder, K.K.; Randerson, J.T. Fire severity influences the response of soil microbes to a boreal forest fire. Environ. Res. Lett. 2016, 11, 035004. [Google Scholar] [CrossRef]
- Buchkowski, R.W.; Bradford, M.A.; Grandy, A.S.; Schmitz, O.J.; Wieder, W.R. Applying population and community ecology theory to advance understanding of belowground biogeochemistry. Ecol. Lett. 2017, 20, 231–245. [Google Scholar] [CrossRef] [PubMed]
- Fierer, N.; Jackson, R.B. The diversity and biogeography of soil bacterial communities. Proc. Natl. Acad. Sci. USA 2006, 103, 626–631. [Google Scholar] [CrossRef] [PubMed]
- Sinsabaugh, R.L.; Lauber, C.L.; Weintraub, M.N.; Ahmed, B.; Allison, S.D.; Crenshaw, C.; Contosta, A.R.; Cusack, D.; Frey, S.; Gallo, M.E.; et al. Stoichiometry of soil enzyme activity at global scale. Ecol. Lett. 2008, 11, 1252–1264. [Google Scholar] [CrossRef] [PubMed]
- Burns, R.G.; DeForest, J.L.; Marxsen, J.; Sinsabaugh, R.L.; Stromberger, M.E.; Wallenstein, M.D.; Weintraub, M.N.; Zoppini, A. Soil enzymes in a changing environment: Current knowledge and future directions. Soil Biol. Biochem. 2013, 58, 216–234. [Google Scholar] [CrossRef]
- Neary, D.G.; Klopatek, C.C.; DeBano, L.F.; Ffolliott, P.F. Fire effects on belowground sustainability: A review and synthesis. For. Ecol. Manag. 1999, 122, 51–71. [Google Scholar] [CrossRef]
- Certini, G. Effects of fire on properties of forest soils: A review. Oecologia 2005, 143, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Treseder, K.K.; Mack, M.C.; Cross, A. Relationships among fires, fungi, and soil dynamics in alaskan boreal forests. Ecol. Appl. 2004, 14, 1826–1838. [Google Scholar] [CrossRef]
- Holden, S.R.; Gutierrez, A.; Treseder, K.K. Changes in Soil Fungal Communities, Extracellular Enzyme Activities, and Litter Decomposition across a Fire Chronosequence in Alaskan Boreal Forests. Ecosystems 2012, 16, 34–46. [Google Scholar] [CrossRef]
- Nemergut, D.R.; Anderson, S.P.; Cleveland, C.C.; Martin, A.P.; Miller, A.E.; Seimon, A.; Schmidt, S.K. Microbial community succession in an unvegetated, recently deglaciated soil. Microb. Ecol. 2007, 53, 110–122. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, S.K.; Reed, S.C.; Nemergut, D.R.; Grandy, A.S.; Cleveland, C.C.; Weintraub, M.N.; Hill, A.W.; Costello, E.K.; Meyer, A.F.; Neff, J.C.; et al. The earliest stages of ecosystem succession in high-elevation (5000 m above sea level), recently deglaciated soils. Proc. R. Soc. B 2008, 275, 2793–2802. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.J.P.; Trahan, N.A.; Wilkes, P.; Quaife, T.; Stephens, B.B.; Elder, K.; Desai, A.R.; Negron, J.; Monson, R.K. Persistent reduced ecosystem respiration after insect disturbance in high elevation forests. Ecol. Lett. 2013, 16, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Knelman, J.E.; Graham, E.B.; Trahan, N.A.; Schmidt, S.K.; Nemergut, D.R. Fire severity shapes plant colonization effects on bacterial community structure, microbial biomass, and soil enzyme activity in secondary succession of a burned forest. Soil Biol. Biochem. 2015, 90, 161–168. [Google Scholar] [CrossRef]
- Almendros, G.; González-Vila, F.J. Fire-induced transformation of soil organic matter from an oak forest. An experimental approach to the effects of fire on humic substances. Soil Sci. 1990, 149, 158–168. [Google Scholar] [CrossRef]
- Boerner, R.E.J.; Giai, C.; Huang, J.; Miesel, J.R. Initial effects of fire and mechanical thinning on soil enzyme activity and nitrogen transformations in eight North American forest ecosystems. Soil Biol. Biochem. 2008, 40, 3076–3085. [Google Scholar] [CrossRef]
- Docherty, K.M.; Balser, T.C.; Bohannan, B.J.M.; Gutknecht, J.L.M. Soil microbial responses to fire and interacting global change factors in a California annual grassland. Biogeochemistry 2011, 109, 63–83. [Google Scholar] [CrossRef]
- Ferrenberg, S.; O’Neill, S.P.; Knelman, J.E.; Todd, B.; Duggan, S.; Bradley, D.; Robinson, T.; Schmidt, S.K.; Townsend, A.R.; Williams, M.W.; et al. Changes in assembly processes in soil bacterial communities following a wildfire disturbance. ISME J. 2013, 7, 1102–1111. [Google Scholar] [CrossRef] [PubMed]
- López-Poma, R.; Bautista, S. Plant regeneration functional groups modulate the response to fire of soil enzyme activities in a Mediterranean shrubland. Soil Biol. Biochem. 2014, 79, 5–13. [Google Scholar] [CrossRef]
- Nemergut, D.R.; Schmidt, S.K.; Fukami, T.; O’Neill, S.P.; Bilinski, T.M.; Stanish, L.F.; Knelman, J.E.; Darcy, J.L.; Lynch, R.C.; Wickey, P.; et al. Patterns and Processes of Microbial Community Assembly. Microbiol. Mol. Biol. Rev. 2013, 77, 342–356. [Google Scholar] [CrossRef] [PubMed]
- Knelman, J.E.; Nemergut, D.R. Changes in community assembly may shift the relationship between biodiversity and ecosystem function. Front. Microbiol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Miesel, J.R.; Hockaday, W.C.; Kolka, R.K.; Townsend, P.A. Soil organic matter composition and quality across fire severity gradients in coniferous and deciduous forests of the southern boreal region. J. Geophys. Res. Biogeosci. 2015, 120, 1124–1141. [Google Scholar] [CrossRef]
- Cline, L.C.; Zak, D.R. Soil Microbial Communities are Shaped by Plant-Driven Changes in Resource Availability During Secondary Succession. Ecology 2015, 96, 3374–3385. [Google Scholar] [CrossRef] [PubMed]
- Allison, S.D.; Gartner, T.B.; Holland, K.; Weintraub, M.N.; Sinsabaugh, R.L. Soil Enzymes: Linking Proteomics and Ecological Process. In Manual of Environmental Microbiology; ASM Press: Washington, DC, USA, 2007; pp. 704–711. [Google Scholar]
- Sinsabaugh, R.L.; Hill, B.H.; Follstad Shah, J.J. Ecoenzymatic stoichiometry of microbial organic nutrient acquisition in soil and sediment. Nature 2009, 462, 795–798. [Google Scholar] [CrossRef] [PubMed]
- Gartner, T.B.; Treseder, K.K.; Malcolm, G.M.; Sinsabaugh, R.L. Extracellular enzyme activity in the mycorrhizospheres of a boreal fire chronosequence. Pedobiologia 2012, 55, 121–127. [Google Scholar] [CrossRef]
- Graham, R.; Finney, M.; McHugh, C.; Cohen, J.; Calkin, D.; Stratton, R.; Bradshaw, L.; Nikolov, N. Fourmile Canyon Fire Findings. Gen. Tech. Rep. 2012. [Google Scholar] [CrossRef]
- Veblen, T.T.; Kitzberger, T.; Donnegan, J. Climatic and human influences on fire regimes in ponderosa pine forests in the colorado front range. Ecol. Appl. 2000, 10, 1178–1195. [Google Scholar] [CrossRef]
- Matejovic, I. Determination of carbon and nitrogen in samples of various soils by the dry combustion. Commun. Soil Sci. Plant Anal. 1997, 28, 1499–1511. [Google Scholar] [CrossRef]
- Sinsabaugh, R.; Carreiro, M.; Repert, D. Allocation of extracellular enzymatic activity in relation to litter composition, N deposition, and mass loss. Biogeochemistry 2002, 60, 1–24. [Google Scholar] [CrossRef]
- Weintraub, S.R.; Wieder, W.R.; Cleveland, C.C.; Townsend, A.R. Organic matter inputs shift soil enzyme activity and allocation patterns in a wet tropical forest. Biogeochemistry 2012, 114, 313–326. [Google Scholar] [CrossRef]
- Bueno de Mesquita, C.P.; Knelman, J.E.; King, A.J.; Farrer, E.C.; Porazinska, D.L.; Schmidt, S.K.; Suding, K.N. Plant colonization of moss-dominated soils in the alpine: Microbial and biogeochemical implications. Soil Biol. Biochem. 2017, 111, 135–142. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Figshare. Available online: https://doi.org/10.6084/m9.figshare.1556158.v1 (accessed on 14 September 2017).
- Allison, S.D.; Gartner, T.B.; Mack, M.C.; McGuire, K.; Treseder, K. Nitrogen alters carbon dynamics during early succession in boreal forest. Soil Biol. Biochem. 2010, 42, 1157–1164. [Google Scholar] [CrossRef]
- Dooley, S.R.; Treseder, K.K. The effect of fire on microbial biomass: A meta-analysis of field studies. Biogeochemistry 2012, 109, 49–61. [Google Scholar] [CrossRef]
- Göransson, H.; Olde Venterink, H.; Bååth, E. Soil bacterial growth and nutrient limitation along a chronosequence from a glacier forefield. Soil Biol. Biochem. 2011, 43, 1333–1340. [Google Scholar] [CrossRef]
- Yoshitake, S.; Uchida, M.; Koizumi, H.; Nakatsubo, T. Carbon and nitrogen limitation of soil microbial respiration in a High Arctic successional glacier foreland near Ny-Ålesund, Svalbard. Polar Res. 2007, 26, 22–30. [Google Scholar] [CrossRef]
- Knelman, J.E.; Schmidt, S.K.; Lynch, R.C.; Darcy, J.L.; Castle, S.C.; Cleveland, C.C.; Nemergut, D.R. Nutrient Addition Dramatically Accelerates Microbial Community Succession. PLoS ONE 2014, 9, e102609. [Google Scholar] [CrossRef] [PubMed]
- Chapin, F.S.; Walker, L.R.; Fastie, C.L.; Sharman, L.C. Mechanisms of primary succession following deglaciation at Glacier Bay, Alaska. Ecol. Monogr. 1994, 64, 149–175. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Farrington, H. Nutrient limitation and soil development: Experimental test of a biogeochemical theory. Biogeochemistry 1997, 37, 63–75. [Google Scholar] [CrossRef]
- Neff, J.C.; Harden, J.W.; Gleixner, G. Fire effects on soil organic matter content, composition, and nutrients in boreal interior Alaska. Can. J. For. Res. 2005, 35, 2178–2187. [Google Scholar] [CrossRef]
- Knelman, J.E.; Legg, T.M.; O’Neill, S.P.; Washenberger, C.L.; González, A.; Cleveland, C.C.; Nemergut, D.R. Bacterial community structure and function change in association with colonizer plants during early primary succession in a glacier forefield. Soil Biol. Biochem. 2012, 46, 172–180. [Google Scholar] [CrossRef]
- Yuan, X.; Knelman, J.E.; Gasarch, E.; Wang, D.; Nemergut, D.R.; Seastedt, T.R. Plant community and soil chemistry responses to long-term nitrogen inputs drive changes in alpine bacterial communities. Ecology 2016, 97, 1543–1554. [Google Scholar] [CrossRef] [PubMed]
- Bardgett, R.D.; Bowman, W.D.; Kaufmann, R.; Schmidt, S.K. A temporal approach to linking aboveground and belowground ecology. Trends Ecol. Evol. 2005, 20, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Nemergut, D.R.; Knelman, J.E.; Ferrenberg, S.; Bilinski, T.; Melbourne, B.; Jiang, L.; Violle, C.; Darcy, J.L.; Prest, T.; Schmidt, S.K.; et al. Decreases in average bacterial community rRNA operon copy number during succession. ISME J. 2015, 10, 1147. [Google Scholar] [CrossRef] [PubMed]
- Castle, S.C.; Nemergut, D.R.; Grandy, A.S.; Leff, J.W.; Graham, E.B.; Hood, E.; Schmidt, S.K.; Wickings, K.; Cleveland, C.C. Biogeochemical drivers of microbial community convergence across actively retreating glaciers. Soil Biol. Biochem. 2016, 101, 74–84. [Google Scholar] [CrossRef]
| Category | pH | % Moisture | % N | % C | NH4 (mg/kg Soil) | NPOC (mg/g Soil) | TDN (mg/g Soil) | BG (nmol Activity/h/g Soil) | NAG (nmol Activity/h/g Soil) | BG:NAG Ratio |
|---|---|---|---|---|---|---|---|---|---|---|
| BURN | ||||||||||
| 1-month | 7.21 (0.30) AB | 9.93 (4.55) BA | 0.14 (0.04) | 2.43 (0.95) | N/A | N/A | N/A | 53.94 (12.02) C | 78.53 (31.46) AB | 0.74 (0.23) B |
| 4-months | 8.08 (0.41) A | 8.55 (2.36) ABC | 0.11 (0.04) | 2.33 (1.07) | 48.94 (14.95) A | 0.30 (0.16) | 0.06 (0.02) A | 76.38 (27.67) BC | 107.54 (43.07) A | 0.75 (0.18) B |
| 29-months | 7.00 (0.28) B | 2.16 (0.80) C | 0.12 (0.03) | 2.48 (0.66) | 2.30 (1.33) B | 0.31 (0.30) | 0.02 (0.02) B | 127.00 (27.57) A | 44.41 (14.61) B | 3.01 (0.71) A |
| REFERENCE | ||||||||||
| 1-month | 6.44 (0.67) B | 19.74 (10.17) A | 0.27 (0.08) | 6.21 (2.01) | N/A | N/A | N/A | N/A | N/A | N/A |
| 4-months | 7.08 (0.46) A | 6.35 (3.50) C | 0.24 (0.17) | 6.59 (5.08) | 2.39 (1.40) | 0.26 (0.18) | 0.02 (0.01) | 147.83 (54.97) B | 136.29 (63.12) | 1.17 (0.38) |
| 29-months | 6.63 (0.34) AB | 8.40 (6.12) BC | 0.24 (0.08) | 5.20 (2.19) | 1.24 (0.60) | 0.09 (0.04) | 0.01 (0.004) | 254.41 (86.14) A | 251.77 (116.38) | 1.13 (0.38) |
| BURNED PLOTS | REFERENCE PLOTS | ||||
|---|---|---|---|---|---|
| Time | Factors | BG | NAG | BG | NAG |
| 1-month post-fire October | pH | NS | NS | N/A | N/A |
| moisture | NS | NS | N/A | N/A | |
| C | NS | NS | N/A | N/A | |
| N | NS | NS | N/A | N/A | |
| C:N | NS | NS | N/A | N/A | |
| 4-months post-fire January | pH | NS | NS | NS | NS |
| moisture | NS | NS | 0.9 | 0.76 | |
| C | 0.83 | NS | 0.81 | 0.79 | |
| N | 0.9 | NS | 0.78 | 0.72 | |
| C:N | NS | NS | 0.77 | NS | |
| 29-months post-fire June | pH | NS | NS | NS | NS |
| moisture | NS | NS | NS | NS | |
| C | 0.69 | NS | NS | NS | |
| N | NS | NS | NS | NS | |
| C:N | NS | 0.69 | NS | NS | |
| 33-months post-fire October | pH | NS | NS | NS | NS |
| moisture | 0.64 | NS | NS | NS | |
| C | NS | NS | 0.84 | 0.84 | |
| N | NS | 0.66 | 0.96 | 0.86 | |
| C:N | NS | 0.74 | NS | NS | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knelman, J.E.; Graham, E.B.; Ferrenberg, S.; Lecoeuvre, A.; Labrado, A.; Darcy, J.L.; Nemergut, D.R.; Schmidt, S.K. Rapid Shifts in Soil Nutrients and Decomposition Enzyme Activity in Early Succession Following Forest Fire. Forests 2017, 8, 347. https://doi.org/10.3390/f8090347
Knelman JE, Graham EB, Ferrenberg S, Lecoeuvre A, Labrado A, Darcy JL, Nemergut DR, Schmidt SK. Rapid Shifts in Soil Nutrients and Decomposition Enzyme Activity in Early Succession Following Forest Fire. Forests. 2017; 8(9):347. https://doi.org/10.3390/f8090347
Chicago/Turabian StyleKnelman, Joseph E., Emily B. Graham, Scott Ferrenberg, Aurélien Lecoeuvre, Amanda Labrado, John L. Darcy, Diana R. Nemergut, and Steven K. Schmidt. 2017. "Rapid Shifts in Soil Nutrients and Decomposition Enzyme Activity in Early Succession Following Forest Fire" Forests 8, no. 9: 347. https://doi.org/10.3390/f8090347
APA StyleKnelman, J. E., Graham, E. B., Ferrenberg, S., Lecoeuvre, A., Labrado, A., Darcy, J. L., Nemergut, D. R., & Schmidt, S. K. (2017). Rapid Shifts in Soil Nutrients and Decomposition Enzyme Activity in Early Succession Following Forest Fire. Forests, 8(9), 347. https://doi.org/10.3390/f8090347





