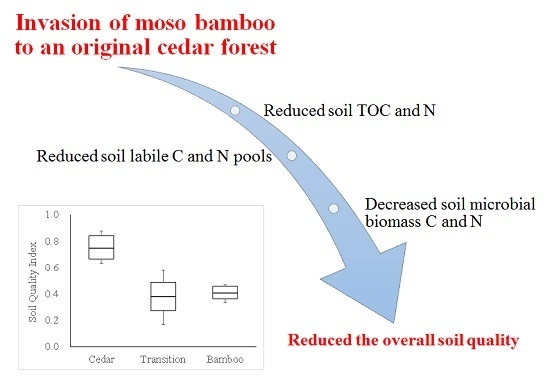
Changes in Soil Biochemical Properties in a Cedar Plantation Invaded by Moso Bamboo
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site and Soil Sampling
2.2. Laboratory Analysis
2.2.1. Soil General Property
2.2.2. Soil Extractable Nutrients
2.2.3. Total Mineralizable N
2.3. Statistical Analysis
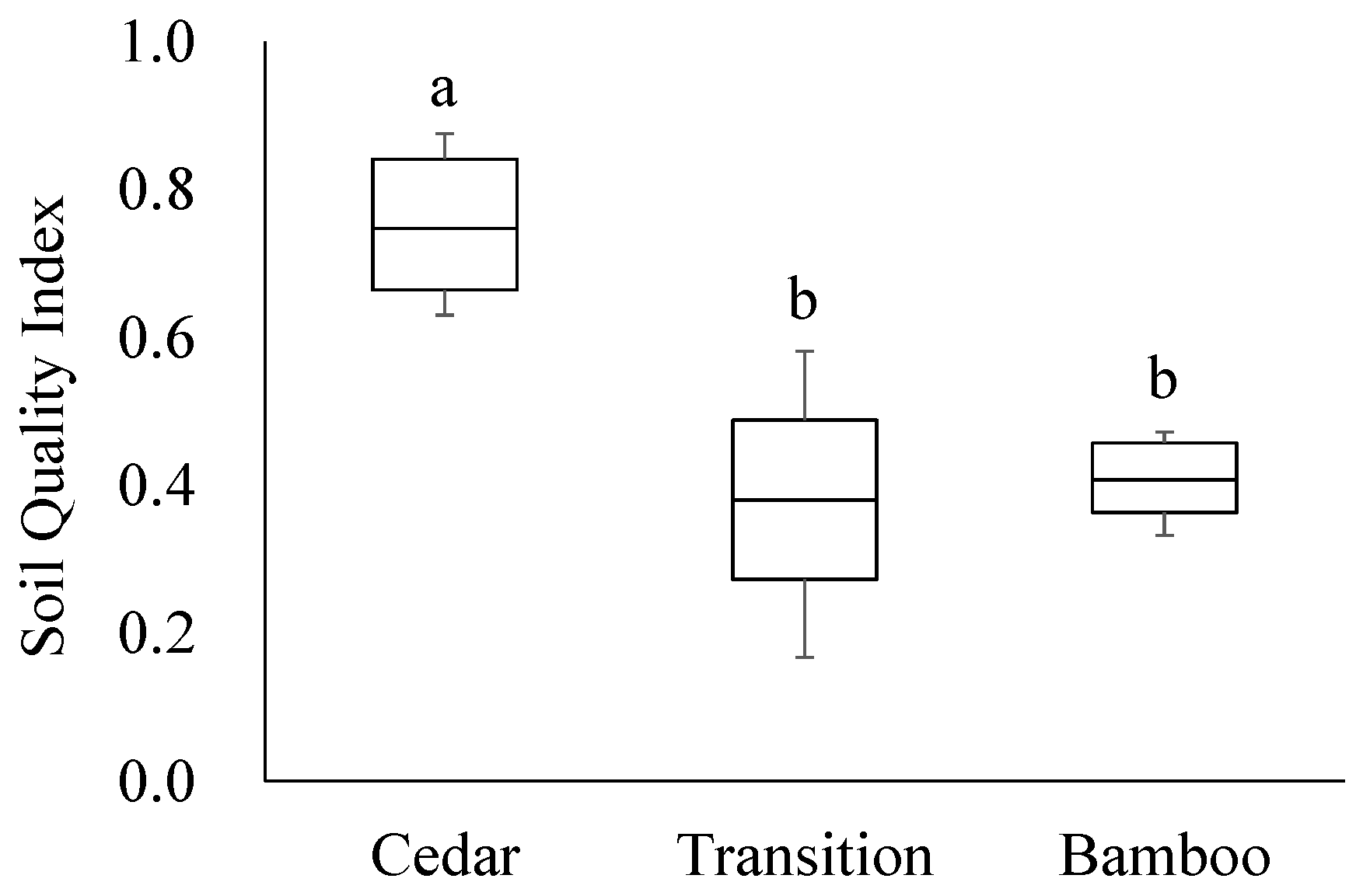
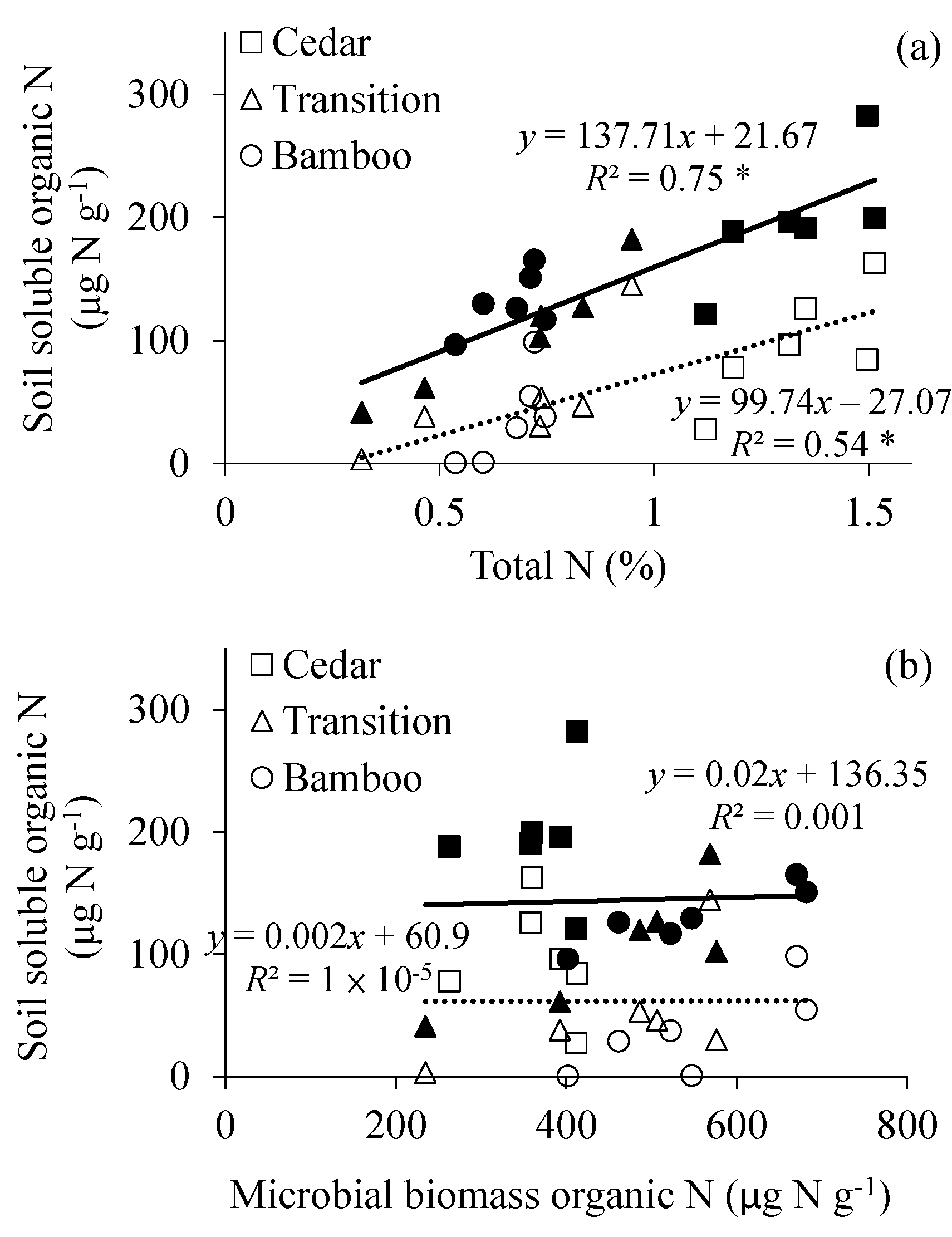
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tripathi, S.K.; Singh, K.P. Productivity and nutrient cycling in recently harvested and mature bamboo savannas in the dry tropics. J. Appl. Ecol. 1994, 31, 109–124. [Google Scholar] [CrossRef]
- Zhuang, S.Y.; Ji, H.B.; Zhang, H.X.; Sun, B. Carbon storage estimation of moso bamboo (phyllostachys pubescens) forest stands in Fujian, China. Trop. Ecol. 2015, 56, 383–391. [Google Scholar]
- Cheng, L.; Adhikari, S.; Wang, Z.H.; Ding, Y.L. Characterization of bamboo species at different ages and bio-oil production. J. Anal. Appl. Pyrolysis 2015, 116, 215–222. [Google Scholar] [CrossRef]
- Lobovikov, M.; OPaudel, S.; MPiazze, M.; Ren, H.; We, J. World Bamboo Resources: A Thematic Study Prepared in the Framework of the Global Forest Resources Assessment 2005; Food and Agriculture Organization of the United Nations: Rome, Italy, 2007. [Google Scholar]
- Hull, V.; Zhang, J.D.; Zhou, S.Q.; Huang, J.Y.; Vina, A.; Liu, W.; Tuanmu, M.N.; Li, R.G.; Liu, D.; Xu, W.H.; et al. Impact of livestock on giant pandas and their habitat. J. Nat. Conserv. 2014, 22, 256–264. [Google Scholar]
- SijiMol, K.; Dev, S.A.; Sreekumar, V.B. A review of the ecological functions of reed bamboo, genus ochlandra in the western ghats of India: Implications for sustainable conservation. Trop. Conserv. Sci. 2016, 9, 389–407. [Google Scholar]
- Xu, Y.; Wong, M.; Yang, J.; Ye, Z.; Jiang, P.; Zheng, S. Dynamics of carbon accumulation during the fast growth period of bamboo plant. Bot. Rev. 2011, 77, 287–295. [Google Scholar]
- Shiau, Y.J.; Wang, H.C.; Chen, T.H.; Jien, S.H.; Tian, G.L.; Chiu, C.Y. Improvement in the biochemical and chemical properties of badland soils by thorny bamboo. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Shinohara, Y.; Otsuki, K. Comparisons of soil-water content between a moso bamboo (phyllostachys pubescens) forest and an evergreen broadleaved forest in western Japan. Plant Species Biol. 2015, 30, 96–103. [Google Scholar]
- Yang, L.; Zhang, F.; Gao, Q.; Mao, R.; Liu, X. Impact of land-use types on soil nitrogen net mineralization in the sandstorm and water source area of Beijing, China. Catena 2010, 82, 15–22. [Google Scholar] [CrossRef]
- Li, R.; Werger, M.J.A.; During, H.J.; Zhong, Z.C. Carbon and nutrient dynamics in relation to growth rhythm in the giant bamboo phyllostachys pubescens. Plant Soil 1998, 201, 113–123. [Google Scholar]
- Chou, C.H.; Yang, C.M. Allelopathic research of subtropical vegetaions in Taiwan II. Comparative exclusion of understory by Phyllostachys edulis and Cryptomeria japonica. J. Chem. Ecol. 1982, 8, 1489–1507. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.; Zhou, G.; Wang, Y.; Liang, Q.; Chen, J.; Cheng, Y.; Shen, R. Plant species diversity and dynamics in forests invaded by moso bamboo (phyllostachys edulis) in Tianmu Mountain nature reserve. Biodivers. Sci. 2013, 21, 288–295. [Google Scholar]
- Zhang, C.; Xie, G.; Fan, S.; Zhen, L. Variation in vegetation structure and soil properties, and the relation between understory plants and environmental variables under different phyllostachys pubescens forests in southeastern China. Environ. Manag. 2010, 45, 779–792. [Google Scholar] [CrossRef] [PubMed]
- Umemura, M.; Takenaka, C. Retranslocation and localization of nutrient elements in various organs of moso bamboo (phyllostachys pubescens). Sci. Total Environ. 2014, 493, 845–853. [Google Scholar] [CrossRef] [PubMed]
- Chang, E.-H.; Chiu, C.-Y. Changes in soil microbial community structure and activity in a cedar plantation invaded by moso bamboo. Appl. Soil Ecol. 2015, 91, 1–7. [Google Scholar] [CrossRef]
- Lin, Y.-T.; Tang, S.-L.; Pai, C.-W.; Whitman, W.B.; Coleman, D.C.; Chiu, C.-Y. Changes in the soil bacterial communities in a cedar plantation invaded by moso bamboo. Microb. Ecol. 2013, 67, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-C.; Tian, G.; Chiu, C.-Y. Invasion of moso bamboo into a Japanese cedar plantation affects the chemical composition and humification of soil organic matter. Sci. Rep. 2016, 6, 32211. [Google Scholar] [CrossRef] [PubMed]
- Ghani, A.; Dexter, M.; Perrott, K.W. Hot-water extractable carbon in soils: A sensitive measurement for determining impacts of fertilisation, grazing and cultivation. Soil Biol. Biochem. 2003, 35, 1231–1243. [Google Scholar] [CrossRef]
- Huang, C.-Y.; Jien, S.-H.; Chen, T.-H.; Tian, G.; Chiu, C.-Y. Soluble organic c and n and their relationships with soil organic c and n and microbial characteristics in moso bamboo (phyllostachys edulis) plantations along an elevation gradient in central Taiwan. J. Soil Sedim. 2014, 14, 1061–1070. [Google Scholar] [CrossRef]
- Strosser, E. Methods for determination of labile soil organic matter: An overview. J. Agrobiol. 2010, 27, 49–60. [Google Scholar] [CrossRef]
- Ros, G.H.; Hoffland, E.; Temminghoff, E.J.M. Dynamics of dissolved and extractable organic nitrogen upon soil amendment with crop residues. Soil Biol. Biochem. 2010, 42, 2094–2101. [Google Scholar] [CrossRef]
- Soil Survey Staff. Keys to Soil Taxonomy, USDA-NRCS Agricultural Handbook No. 436, 11th ed.; U.S. Government Printing Office: Washington, DC, USA, 2010.
- Sollins, P.; Glassman, C.; Paul, E.A.; Swanston, C.; Lajtha, K.; Heil, J.W.; Elliott, E.T. Soil carbon and nitrogen: Pools and fractions. In Standard Soil Methods for Long-Term Ecological Research; Robertson, G.P., Coleman, D.C., Bledsoe, C.S., Sollins, P., Eds.; Oxford University Press: New York, NY, USA, 1999; pp. 89–105. [Google Scholar]
- Waring, S.A.; Bremner, J.M. Effect of soil mesh-size on estimation of mineralizable nitrogen in soils. Nature 1964, 202, 1141. [Google Scholar] [CrossRef]
- Lu, X.; Toda, H.; Ding, F.; Fang, S.; Yang, W.; Xu, H. Effect of vegetation types on chemical and biological properties of soils of karst ecosystems. Eur. J. Soil Biol. 2014, 61, 49–57. [Google Scholar] [CrossRef]
- Mukherjee, A.; Lal, R. Comparison of soil quality index using three methods. PLoS ONE 2014, 9, e105981. [Google Scholar] [CrossRef] [PubMed]
- Nakane, K. Soil carbon cycling in a Japanese cedar (cryptomeria-japonica) plantation. For. Ecol. Manag. 1995, 72, 185–197. [Google Scholar] [CrossRef]
- Fukushima, K.; Usui, N.; Ogawa, R.; Tokuchi, N. Impacts of moso bamboo (phyllostachys pubescens) invasion on dry matter and carbon and nitrogen stocks in a broad-leaved secondary forest located in kyoto, western Japan. Plant Species Biol. 2015, 30, 81–95. [Google Scholar] [CrossRef]
- Reeves, D.W. The role of soil organic matter in maintaining soil quality in continuous cropping systems. Soil Tillage Res. 1997, 43, 131–167. [Google Scholar] [CrossRef]
- Saleem, M. Microbiome Community Ecology: Fundamentals and Applications; Springer International Publishing: New York, NY, USA, 2015. [Google Scholar]
- Saleem, M.; Fetzer, I.; Harms, H.; Chatzinotas, A. Trophic complexity in aqueous systems: Bacterial species richness and protistan predation regulate dissolved organic carbon and dissolved total nitrogen removal. Proc. R. Soc. B Biol. Sci. 2016, 283, 20152724. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, M.; Sumikawa, Y.; Guan, G.; Marumoto, T. Relationship between microbial biomass and extractable organic carbon content in volcanic and non-volcanic ash soil. Appl. Soil Ecol. 2001, 17, 183–187. [Google Scholar] [CrossRef]
- Xu, J.M.; Cheng, H.H.; Koskinen, W.C.; Molina, J.A.E. Characterization of potentially bioreactive soil organic carbon and nitrogen by acid hydrolysis. Nutr. Cycl. Agroecosyst. 1997, 49, 267–271. [Google Scholar] [CrossRef]
- Wang, H.C.; Chou, C.Y.; Chiou, C.R.; Tian, G.L.; Chiu, C.Y. Humic acid composition and characteristics of soil organic matter in relation to the elevation gradient of moso bamboo plantations. PLoS ONE 2016, 11, e0162193. [Google Scholar] [CrossRef] [PubMed]

| Vegetation | SbOCKCl (μg/g soil) | NH4+KCl (μg/g soil) | NO3−KCl (μg/g soil) | SbONKCl (μg/g soil) | TDNKCl (μg/g soil) | TOC (%) | TN (%) | SbOCKCl/TOC (%) | SbONKCl/TN (%) |
|---|---|---|---|---|---|---|---|---|---|
| Cedar | 1315.3 a | 74.3 a | 58.1 a | 196.3 a | 328.7 a | 21.46 a | 1.33 a | 0.60 a | 1.46 b |
| Transition | 455.9 b | 42.6 b | 48.2 a | 105.6 b | 196.4 b | 7.54 b | 0.67 b | 0.58 a | 1.51 b |
| Bamboo | 552.2 b | 37.4 b | 39.8 a | 130.8 ab | 208.1 b | 8.00 b | 0.67 b | 0.69 a | 1.96 a |
| Vegetation | SbOCHW (μg/g soil) | NH4+HW (μg/g soil) | NO3−HW (μg/g soil) | SbONHW (μg/g soil) | TDNHW (μg/g soil) | Mineralizable N (μg N/g soil/d) | SbOCHW/TOC (%) | SbONHW/TN (%) |
|---|---|---|---|---|---|---|---|---|
| Cedar | 7180.2 a | 118.3 a | 32.1 a | 95.7 a | 246.2 a | 160.0 b | 3.28 a | 0.70 a |
| Transition | 1838.3 b | 60.8 b | 37.4 a | 52.6 a | 150.8 b | 187.8 ab | 2.45 b | 0.69 a |
| Bamboo | 2081.9 b | 74.6 b | 29.0 a | 36.8 a | 139.9 b | 218.4 a | 2.58 b | 0.51 a |
| Principal Components | PC-1 | PC-2 | PC-3 |
|---|---|---|---|
| Eigenvalue | 10.33 | 2.63 | 1.60 |
| Eigenvactors: | |||
| NH4+KCl | 0.252 | −0.122 | −0.194 |
| NO3−KCl | 0.126 | 0.415 | −0.477 *,† |
| SbONKCl | 0.269 | 0.191 | 0.189 |
| SbOCKCl | 0.297 † | 0.024 | 0.161 |
| NH4+HW | 0.292 † | 0.030 | 0.030 |
| NO3−HW | 0.048 | 0.476 † | −0.466 † |
| SbONHW | 0.236 | 0.168 | 0.221 |
| SbOCHW | 0.300 † | −0.062 | 0.124 |
| Total mineralizable N | −0.084 | 0.415 | 0.474 † |
| Cmic 1 | 0.241 | 0.227 | −0.125 |
| Nmic 1 | −0.115 | 0.491 *,† | 0.343 |
| TOC | 0.308 *,† | −0.025 | 0.027 |
| TN | 0.307 † | 0.034 | 0.038 |
| AHPI-C 2 | 0.291 † | −0.022 | 0.039 |
| AHPII-C 2 | 0.286 † | −0.168 | 0.142 |
| RP-C 2 | 0.306 † | −0.145 | −0.091 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shiau, Y.-J.; Chiu, C.-Y. Changes in Soil Biochemical Properties in a Cedar Plantation Invaded by Moso Bamboo. Forests 2017, 8, 222. https://doi.org/10.3390/f8070222
Shiau Y-J, Chiu C-Y. Changes in Soil Biochemical Properties in a Cedar Plantation Invaded by Moso Bamboo. Forests. 2017; 8(7):222. https://doi.org/10.3390/f8070222
Chicago/Turabian StyleShiau, Yo-Jin, and Chih-Yu Chiu. 2017. "Changes in Soil Biochemical Properties in a Cedar Plantation Invaded by Moso Bamboo" Forests 8, no. 7: 222. https://doi.org/10.3390/f8070222
APA StyleShiau, Y.-J., & Chiu, C.-Y. (2017). Changes in Soil Biochemical Properties in a Cedar Plantation Invaded by Moso Bamboo. Forests, 8(7), 222. https://doi.org/10.3390/f8070222