Abstract
There is limited information on how velvet leaf blueberry (Vaccinium myrtilloides Michx.) responds to fires and existing small forest gaps associated with narrow linear disturbances. We measured the effects of narrow forest linear gaps from seismic lines used for oil and gas exploration versus adjacent (control) forests across a fire severity (% tree mortality) gradient on the presence, abundance (cover), vigor (height), and berry production of Vaccinium myrtilloides in recently (five-year) burned jack pine (Pinus banksiana Lamb.) forests near Fort McMurray, Alberta. Presence was greatest in forests that experienced low to moderately-high fire severities with declines at high fire severity. Abundance did not differ among seismic lines or adjacent forest, nor did it differ along a fire severity gradient. In contrast, vigor and berry production were greater on seismic lines compared to adjacent forests with fire severity positively affecting berry production, but not plant vigor. After controlling for changes in plant cover and vigor, berry production still increased with fire severity and within seismic lines compared with adjacent forests. Our findings suggest that narrow gaps from linear disturbances and fire severity interact to affect the fecundity (berry production) and growth (height) of Vaccinium myrtilloides. This has important implications for assessing the ecological effects of fire on linear disturbances associated with energy exploration in the western boreal forest.
Keywords:
fire; severity; seismic line; disturbance; jack pine; production; vigor; abundance; presence; Ericaceae; velvet leaf blueberry; Vaccinium myrtilloides 1. Introduction
Fire is a common element in the boreal forest [1] with many of its dominant plants having adaptations that allow their long-term persistence, even under high fire frequencies [2,3,4]. For example, jack pine (Pinus banksiana Lamb.) is a common overstory species that dominates drier sites of the boreal forests of North America whose serotinous cones open and release seeds following fire [5,6]. Many shrub and herbaceous species are similarly adapted to disturbance, including fire, but instead through vegetative regeneration from underground rhizomes [7,8]. Fires also alter site conditions that favor understory plants by increasing light availability in the understory [9] and by reducing total plant cover, thereby reducing competition [10]. Fire severity also plays an important role in affecting the composition of vegetation both directly and indirectly through changes in below-ground processes [11]. For example, high severity fires in jack pine stands have lower species richness and cover, while lower severity fires have the highest species richness and cover, even when compared to unburned stands [12]. A common emphasis of forest fire studies is in understanding changes in tree composition and density [13,14,15,16]. Much less is known, however, about understory responses despite having the potential to influence the direction of post-fire succession [17].
Ericaceous plants are a common understory shrub of fire-prone forests that often respond positively to fire [18]. For instance, the germination rate of Erica umbellate L. increases following fire [19], while greater light availability post-fire increases the sexual reproduction and vigor of Gaultheria shallon Pursh [20]. Likewise, Kalamia augustifolia L. and Vaccinium species regenerate post-fire because their rhizomatic roots often escape the mortality of fires resulting in vegetative resprouting [7,21], although responses in these species may be negative when fire intensities are high due to extreme heat [19,21]. Although post-fire communities often exhibit lower levels of competition immediately following fire, dense tree regeneration can reduce understory cover, including ericaceous shrubs, due to direct competition for light and resources. For example, Vaccinium angustifolium Aiton and Vaccinium myrtilloides Michx. both responded negatively post-fire to competition with other rapidly colonizing species [22], including competition with tree species [23]. Thus, any positive responses following fire may be short lived once tree recruitment dominates a site.
Although fire is a common natural disturbance to boreal forests [24], anthropogenic disturbances have recently become more widespread [25]. In Alberta, Canada, common anthropogenic disturbances that create gaps in the forest canopy include forest harvest clear-cuts and seismic lines. Seismic lines are used to predict subsurface properties of the Earth, such as oil and gas reserves, and have been used in the exploration of oil and gas since 1924 [26]. Unlike clear-cuts, seismic lines are long, narrow, and linear forest gap features. As canopy gap size increases, the mean and variability of light levels within the gap also increases [27], affecting the amount of sunlight received by understory foliage and thus stimulating its growth [23]. For instance, Gaultheria procumbens responds positively to clear-cutting by stimulating below-ground vegetative growth and the release of new shoots from the existing network of rhizomes [8]. Open canopy gaps have also been shown to benefit Vaccinium myrtillus [28].
Although previous research has examined responses in Vaccinium myrtilloides to large forest openings from clear-cut forest harvesting [23], little is known about the effects of smaller forest canopy gaps, including the narrow linear seismic line disturbances, and how these small gaps interact with fires. Seismic line disturbances are individually narrow in their footprint (~3–12 m wide), but their densities are high (mean density of 1.77 km/km2) [29], making them the most abundant linear features in Alberta’s boreal forest. Many of these seismic lines are failing to recover decades after their initial disturbance [30,31], thus altering groundlayer vegetation composition [32]. Understanding specific responses to individual species, such as Vaccinium myrtilloides, is important for determining the overall effects of these linear disturbances on the boreal forest community and how that may change following natural disturbances of wildfires. The objectives of this study were to examine the responses of Vaccinium myrtilloides to small canopy gaps created by seismic lines (including variation in their width) and to test whether fire interacts with these disturbances along a fire severity gradient. Specifically, we examine changes in Vaccinium myrtilloides presence, abundance (cover), vigor (height), and berry production across a fire severity gradient five-years post-fire on narrow linear disturbances (corridors) used for seismic exploration and paired control sites in jack pine forests in northeast Alberta.
2. Materials and Methods
2.1. Study Area
The study area is located in the northeast part of Alberta, Canada, within an area known as the Richardson and McClelland Lake areas on the Athabasca Sand Plain approximately 115 km north of Fort McMurray. Elevation of the area is ~300 m above sea level and characterized by dry, sandy, gently sloping terrain dominated by jack pine (Pinus banksiana) forests that have among the highest observed fire frequencies in Canada’s boreal forest [24].
In 2011, the Richardson fire burned 707,648 hectares of forest making it among Alberta’s largest recorded fire [33]. Fire severity (% tree mortality) was, however, highly variable with tree mortality ranging from 0 to 100 percent. Jack pine stands in the area varied in age, but mostly represented mature forests of jack pine that were often characterized as having semi-open woodland conditions where tree density was moderate to low. Common shrub and understory species included velvet leaf blueberry (Vaccinium myrtilloides), pin cherry (Prunus pensylvanica L.), saskatoon (Amelanchier alnifolia Nutt.), bearberry (Arctostaphylos uva-ursi L.), reindeer lichen (Cladina spp.), and rose (Rosa spp.). Some sites that were slightly more mesic contained a minor element of trembling aspen (Populus tremuloides Michx.) and green alder (Alnus crispa Aiton).
2.2. Experimental Design and Field Measures
2.2.1. Site Selection
Sample sites were selected (stratified) across a range of fire severity classes (Figure 1), which were defined by percent tree mortality (tree mortality rates of: 0–25%; 26%–50%; 51%–75%; 76%–100%) to ensure the representation of all levels of fire severity (≥8 replicate sites per strata with 2 paired plots per site). Fire severity was measured within stands adjacent to seismic lines based on percent tree mortality within the surrounding area. In total, 66 sites were sampled in the summer of 2016 (from 5 July to 15 August) representing conditions five-years post-fire. At each site, a pair of plots was established in areas with a similar fire severity and stand age with one plot positioned in the middle of the seismic line (treatment) and the other 25 m into the adjacent forest (control). The total number of plots equaled 132 (66 paired plots or sites). Direction from the seismic line for the control plot was randomly assigned to being left or right of the seismic line using a coin flip. To avoid edge effects, all plots were at least 30 m from any other forest types, forest edges, or another seismic line, and at least 90 m from clear-cuts, roads, or major trails. Each plot was represented as a 30-m long transect that followed the orientation of the seismic line and Vaccinium myrtilloides characteristics were measured along the transect. Additional forest measures were made in the adjacent forest stand (e.g., basal area, tree stem density, stand age), but were not further considered here since they had little additional effect on variation in Vaccinium myrtilloides measures.
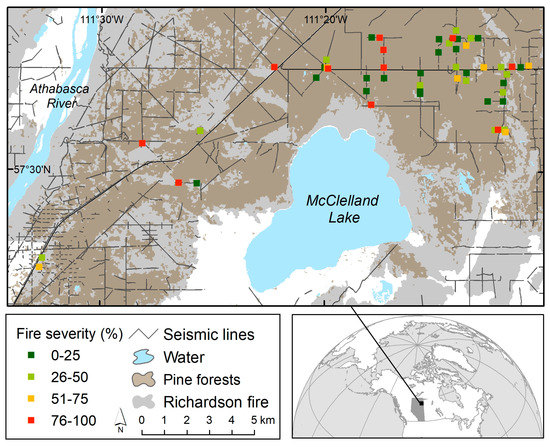
Figure 1.
Location of study site in northeast Alberta, Canada (inset map), with the main map depicting the location of the 2011 Richardson fire (gray), unburned areas (white), pine forests (dark brown where burned and in a few places light brown where unburned), seismic lines, and sample sites symbolized by four levels of fire severity. Each site represents two-paired plots (a seismic line and an adjacent forest) with sites restricted to areas with no forest harvesting (not shown, but extensive in the area). Note that nearly all pine forests were burned and that some sites were within ~200 m of each other (not shown in this map) when stand and fire severity levels differed or there were gaps in the seismic line disturbance.
2.2.2. Measured Responses in Vaccinium myrtilloides and Treatment Variables
Vaccinium myrtilloides presence, abundance (cover), vigor (maximum height), and berry production were measured along each 30-m transect in 1-m wide belts (i.e., a 30-m2 plot). Presence of Vaccinium myrtilloides within each 30-m2 plot was first recorded and if present the abundance (cover) of Vaccinium myrtilloides plants was measured within ten sequentially spaced sub-plots (quadrats), each having a 3 × 1 m size (3 m2). Within each quadrat, cover was estimated using 10 ordinal cover classes following the Carolina Vegetation Survey protocol that is similar to the Domin and Daubenmire techniques [34] with final cover estimates for each plot estimated as the average of midpoint cover values in each quadrat. Plant vigor was measured in each plot as the average maximum height of Vaccinium myrtilloides plants among the 10 sampled quadrats. Maximum height was used as a measure of vigor since it has been known to be an important predictor of fruit production [35], particularly after recent disturbances. Finally, berry production in Vaccinium myrtilloides was recorded after fruit set and before full ripening in each 3-m2 quadrat and summed to estimate total berry production on a per 30 m2 (1 × 30 m) basis.
Treatment variables used to describe responses in Vaccinium myrtilloides presence, abundance, vigor, and berry production included fire severity and location (seismic lines vs. adjacent forests). Fire severity was measured as either ordinal categories (0%–25%, 26%–50%, 51%–75%, or 76%–100%) for simple comparisons and graphs, or original continuous values for linear models. The seismic line and adjacent forest (location) factor was measured as a binary variable with seismic lines being coded as 1 and adjacent forest plots being coded as 0 (reference comparison).
2.3. Data Analysis
2.3.1. Responses in Vaccinium myrtilloides on Seismic Lines and Adjacent Forests
For each response measure of Vaccinium myrtilloides (presence, abundance, vigor, and berry production), we first summarized and graphed average values by treatment and fire severity class and then compared seismic line treatments to adjacent forests within each fire severity class using paired t-tests for continuous data (abundance, vigor, and berries) and a McNemar’s χ2 test for presence and absence data. Second, we used mixed effect regression models (logistic family for presence/absence and Gaussian family for all others) to examine responses across the full fire severity gradient (original linear values from 0 to 100% tree mortality), location (seismic line treatment; seismic line = 1; adjacent forests = 0), and their interaction. A random effect on site ID was used in all models to account for the paired nature of the sampling design (paired plots per site). All plots were used for presence data, while abundance, vigor, and berries were only assessed where plants were present at the site level. A log10 transformation with a constant of 1 was used for berry counts in all statistical analyses to normalize their distribution, while plant cover and the height of plants were kept in their original scale since their distribution was approximately normal. Responses to fire severity (tree mortality) were fit by both linear and quadratic terms to consider possible non-linear responses. Model selection followed the development of a global model that included all variables (fire severity, location treatment of seismic line, and their interaction) with the subsequent removal of non-significant (p < 0.1), non-treatment variables. Fire severity and location treatment were thus retained in all models regardless of their significance given that it was the main part of the study design. Model variance explained (R2) was used to assess the overall model fit.
2.3.2. Effects of Seismic Line Width (Forest Gap Size) on Vaccinium myrtilloides
As well as more generally comparing responses between seismic lines and adjacent forests across a fire severity gradient, we also assessed whether forest gap size, based on seismic line width (~4–10 m), affected responses in Vaccinium myrtilloides. We did this by comparing responses by seismic line width using simple linear regression including reporting on model fit assessed with adjusted R2 and model error assessed with Root Mean Square Error (RMSE). Here, only the seismic line treatments (plots) were included in the analyses since we were only comparing differences between seismic line disturbance widths. Collinearity between seismic line width and fire severity was low (r = 0.24) with a good representation of different line widths across the fire severity gradient. Scatter plots were used to help visualize univariate relationships. Models included all seismic line plots regardless of the presence of Vaccinium myrtilloides (n = 66), as well as only those plots where Vaccinium myrtilloides plants were present on seismic line plots (n = 41). The restriction to plots where plants were present allowed an evaluation of the changes in berry production due to forest gap size, while accounting for general changes in plant abundance (cover) and vigor (height) that would be expected to change under open forest gap conditions. Thus, we asked whether differences in gap size (seismic line width) increased the fecundity (berry production) of plants on a per capita basis through increases in resources (e.g., light) that would boost flowering and/or pollinator activity that could affect fruit set. All analyses were performed in STATA/SE 13.1 (STATA Corp., College Station, TX, USA).
3. Results
3.1. Presence
Of the 66 sites and 132 plots sampled, Vaccinium myrtilloides was present in 41 (62.1%) seismic line plots versus 43 (65.2%) adjacent forest plots (Figure 2) with no significant difference between treatments (p = 0.148; Table 1). Fire severity had, however, a significant, non-linear effect on presence (p < 0.001, Table 1) with increases occurring in both the seismic line and the adjacent forest stand up to a moderately-high fire severity (51%–75%; Figure 2). Thereafter, high fire severity (76%–100%) resulted in decreases in presence (Figure 2). The mixed effects linear model had a significant negative interaction between fire severity and seismic line treatment (p = 0.014; Table 1), demonstrating the reduced presence of Vaccinium myrtilloides on seismic lines compared to adjacent forests at moderate to high fire severity (Table 1; Figure 1). Overall model fit was high (R2 = 0.41; Table 1).
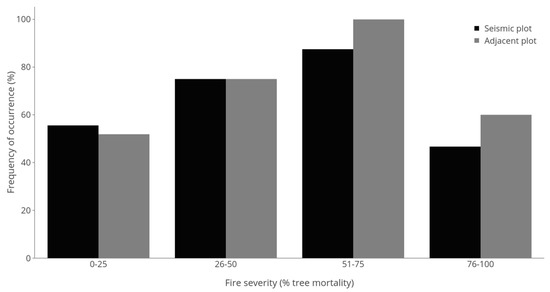
Figure 2.
Frequency (presence) of Vaccinium myrtilloides plants in 30-m2 plots on seismic lines versus adjacent forests across four fire severity classes. Comparisons between seismic and adjacent plots within individual fire severity classes using McNemar’s χ2 tests revealed no significant (p < 0.1) differences.

Table 1.
Mixed effect regression results relating the presence, abundance (cover), and vigor (maximum height) of Vaccinium myrtilloides plants as a function of location (seismic line versus adjacent forest), fire severity, and the interaction of fire severity (% tree mortality) and location. Model coefficients (Coef.), Standard Error (S.E.) of coefficients, and significance (p) reported. Main treatment variables were retained regardless of significance, while a non-linear effect for fire severity and interaction term between fire severity and seismic lines were included only if significant.
3.2. Abundance (Cover)
Abundance (cover) where Vaccinium myrtilloides was present within either plot at the site level did not significantly differ (p = 0.451; Table 1) between seismic lines (Mean cover = 2.0%, S.E. = 0.33) and adjacent forest stands (Mean cover = 1.7%, S.E. = 0.36). Likewise, there was no significant difference in cover across the fire severity gradient (p = 0.642; Table 1, Figure 3). A non-linear fire gradient response was not supported, nor was an interaction between location (seismic line) and fire severity. Model fit was low overall (R2 = 0.01; Table 1). This suggested no direct increase in abundance relative to differences in forest gap size (seismic line) or fire severity. Although variation was high and no significant responses were detected, there were weak trends towards greater cover in sites with moderate (26%–75%) fire severity, especially in adjacent control forests (Figure 3).
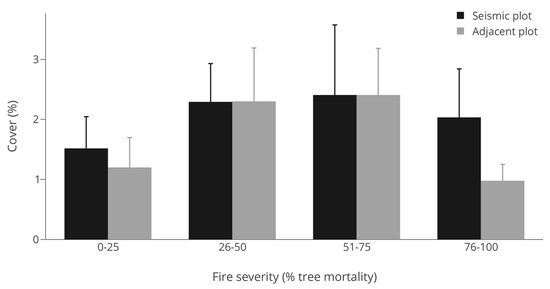
Figure 3.
Mean cover of Vaccinium myrtilloides when present for seismic lines and adjacent forests across a fire severity gradient. Comparisons between seismic and adjacent plots within fire severity classes were tested using paired t-tests revealing no significant (p < 0.1) differences.
3.3. Vigor (Maximum Height)
Seismic line disturbances increased plant vigor, when present in plots, as measured by the maximum height of plants when compared to adjacent forest stands (p = 0.001; Table 1). Maximum heights in seismic line forest gaps averaged 23.5 cm (S.E. = 1.40), while being 19.0 cm (S.E. = 0.82) in the adjacent forest. Plant vigor did not vary across fire severity classes (p = 0.832), nor was there a significant interaction between seismic lines and fire severity (Table 1), although vigor on seismic lines did decrease under the highest fire severity class with a t-test revealing significant location effects within individual fire severity classes up to 75% tree mortality (Figure 4). Model fit explaining plant vigor was low overall (R2 = 0.09; Table 1) demonstrating a high amount of unexplained variability.
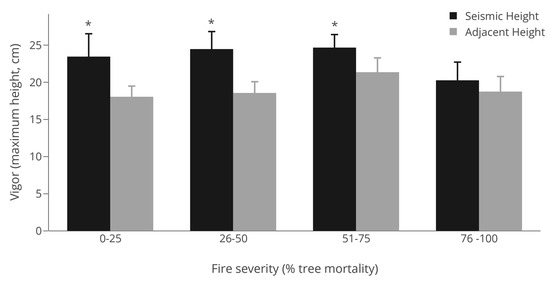
Figure 4.
Average maximum height of Vaccinium myrtilloides when present, across a fire severity gradient (four classes) for seismic lines (black) and adjacent forests (gray). Significance among location by fire severity class was assessed by paired t-tests (* = p < 0.1).
3.4. Berry Production Across All Sites
Average berry production for sites where Vaccinium myrtilloides was present in at least one of the paired plots was significantly higher (p < 0.001) in seismic lines (36.9 berries per 30 m2, S.E. = 8.5) compared with adjacent forest stands (11.4 berries per 30 m2, S.E. = 2.7; Table 2). Fire severity positively affected berry production (p = 0.009; Table 2, Figure 5), although there was a significant negative interaction between seismic line treatment and fire severity (p = 0.032; Table 2) demonstrating that increases in berry production across fire severity classes was primarily related to changes in adjacent forest stands with berry production being more constant in seismic lines (Figure 4). Indeed, when testing seismic lines versus adjacent forests for each fire severity class using paired t-tests, berry production was significantly higher on seismic lines in the low (0%–25%) and low-moderate (26%–50%) fire severity classes, but not the moderately high (51%–75%) to high (76%–100%) fire severity classes (Figure 4), where tree mortality was high and thus canopy cover low and light levels high. Model fit explaining berry production was moderately-low overall (R2 = 0.15; Table 2).

Table 2.
Mixed effect linear regression model testing differences in Vaccinium myrtilloides berry production in response to location (seismic line versus adjacent forest), fire severity, and the interaction of fire severity and location. Note that results are based on a log10 transformed count of berries.
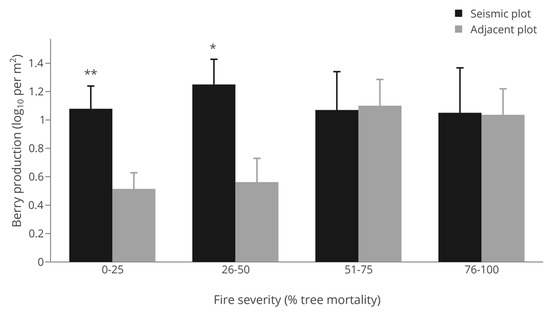
Figure 5.
Vaccinium myrtilloides berry production (log10 scale) by fire severity class and location (seismic line versus adjacent forest). Significance among location by fire severity class was assessed by paired t-tests (* = p < 0.1; ** = p < 0.01).
3.5. Responses in Berry Production When Plants Were Present
Further analysis of berry production was conducted using data only from those plots (transects) with Vaccinium myrtilloides plants present in order to assess changes in berry production after controlling for the effects of shrub cover and vigor. Not surprisingly, plots that contained taller Vaccinium myrtilloides plants (p < 0.001) and those with increased cover (p < 0.001) produced more berries (Table 3). More interesting was that berry production on seismic lines was still significantly greater than in the adjacent forest stand (p = 0.002) after accounting for differences in plant cover and vigor (Table 3). Likewise, fire severity also increased berry production (p < 0.001) after controlling for cover and vigor with a weak significant effect of the interaction of seismic line treatment and fire severity (p = 0.097), suggesting a diminishing effect on berry production on seismic lines that experienced higher fire severity (Table 3). Model fit explaining berry production where plants were present was high overall although much of this was likely due to the effects of changes in cover and vigor (R2 = 0.59; Table 3).

Table 3.
Mixed effect linear regression results relating the berry production of Vaccinium myrtilloides to location (seismic line versus adjacent forest), fire severity, interaction of fire severity and location, abundance (cover), and vigor.
3.6. Effect of Forest Gap Width from Seismic Lines
Vaccinium myrtilloides cover and vigor was positively and significantly (p = 0.003) related to seismic line forest gap width with model fit being higher for vigor (R2 = 0.19, RMSE = 2.02) than for cover (R2 = 0.12, RMSE = 8.09) (Table 4, Figure 6). Likewise, berry production was positively related to seismic line width (p <0.001, R2 = 0.16, RMSE = 0.71; Table 4, Figure 6), although this appeared to be due to seismic lines with larger widths having significantly higher cover and vigor since berry production on sites with plants present was related to plant cover (p = 0.004) and vigor (p = 0.018), but not line width (p = 0.298, R2 = 0.57, RMSE = 0.43; Table 5). Regardless, the presence of seismic lines still increased berry production above and beyond that expected by increases in cover and vigor (Table 3), but not due to changes in line width (gap size), suggesting an indirect effect of seismic line presence on berry production.

Table 4.
Linear regression results relating forest gap (seismic line) width to abundance and vigor of Vaccinium myrtilloides.
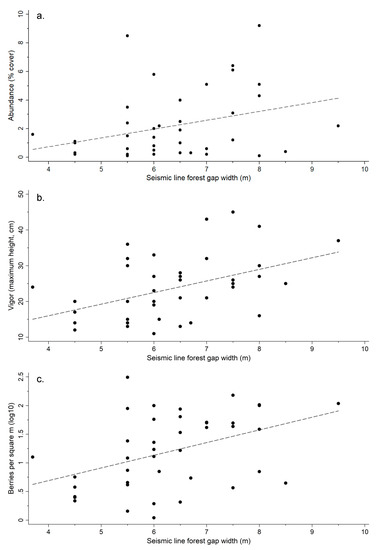
Figure 6.
Scatterplot of seismic line width and Vaccinium myrtilloides cover (a), vigor (b), and berry production (c) on seismic lines where plants were present. Dashed line represents the fit of a linear regression relationship with overall model fit as follows: abundance (cover) R2 = 0.12 (a), vigor (height) R2 = 0.19 (b), and berry production R2 = 0.16 (c).

Table 5.
Linear regression results relating forest gap (seismic line) width to berry production (log10 transformed) in Vaccinium myrtilloides.
4. Discussion
4.1. Responses to Fire
We found that the presence of Vaccinium myrtilloides in recently burned (five-years) jack pine forests depended on fire severity with low to moderately-high severity (0%–75% tree mortality) increasing the presence in both seismic line forest gaps and adjacent forest stands. These results support prior research where low to moderate severity fires triggered positive responses to ericaceous shrubs by increasing light availability from canopy openings [17], stimulating the germination rate [19], and decreasing initial competition among understory plants [10]. In contrast, fires of a higher severity (>76% tree mortality) decreased the presence of Vaccinium myrtilloides on seismic lines and adjacent forest stands. This suggests that high severity fires can cause direct lethal effects to ericaceous shrubs most likely by the elimination of rhizomes in the soil layer [21]. Interestingly, the cover and height of Vaccinium myrtilloides was not affected by fire severity suggesting that its main effect is related to initial mortality and potentially to fecundity (berry production). Indeed, we found fire severity to be positively related to berry production in Vaccinium myrtilloides. The reproduction of ericaceous and Vaccinium species is known to be stimulated post-fire by increases in available light resources [20]. Fire not only increases light availability, but also nutrients with nitrogen, phosphorus, potassium, calcium, and magnesium potentially increasing following fire which can further stimulate growth and reproduction [36,37].
Our results demonstrate important interactions between forest gaps (seismic lines) and fire severity. Berry production in low to low-moderate fire severity (0%–50% tree mortality) was significantly higher on seismic lines than adjacent forest stands. Under low fire severity, canopy in the adjacent forest remains largely intact in contrast to local open canopy gaps associated with seismic lines. Increased light availability on seismic lines should therefore increase in berry production. Likewise, light availability increases in forests exposed to higher severity fires so light should be similar here to seismic line forest gaps, thereby resulting in similar increases in berry production.
4.2. Response to Seismic Lines
Small forest canopy gaps associated with seismic line disturbances did not affect the presence or abundance (cover) of Vaccinium myrtilloides, but it did increase plant vigor (maximum height) and berry production after fire disturbance. Past studies examining the responses of ericaceous species to clear-cut forest harvesting have also suggested that plant vigor is coupled to increases in light resources [38], although it may also be partly related to releases of allelochemcials [39]. Small forest canopy gaps affected berry production in Vaccinium myrtilloides with seismic lines producing ~30% more berries than the adjacent forest stand. Because seismic lines did not affect the abundance (cover) of Vaccinium myrtilloides, increases in reproduction are associated with increases in resource availability (light and nutrients) and/or increases in insect pollination.
Research in forest clear-cuts demonstrated similar changes in the reproductive strategies of ericaceous species with reproduction being more strongly influenced by irradiance than shoot density [20]. Past research has suggested that only large plants were capable of sexual reproduction and therefore light indirectly influences sexual reproduction through effects on plant size [35]. However, we found that after accounting for abundance, vigor (height), and fire severity, berry production was still greater on seismic lines compared with adjacent forests. This may be due to the larger canopy opening of seismic lines allowing greater light availability to the understory and thus higher rates of photosynthesis that can support the development of more fruit structure [40].
The second possibility is that seismic lines are increasing abundance and behaviour of insect pollinators, thus affecting fruit set. Vaccinium species are known to be pollinated by bees [41] with fruit production, size, and seediness of Vaccinium species being significantly higher in plants exposed to natural pollination [42]. However, Vaccinium species have also been shown to have higher flowering densities in sites with an open canopy [42] and because we did not measure flower production we cannot distinguish between these factors. In fact, it is most likely to be a combination of greater flower production due to photosynthetic resources and increases in pollinator activity. If the effect is due to pollinators, then visitation rates by pollinators on seismic lines should be higher than in the adjacent stand. Future studies should examine these interactions by measuring flower production, pollinator visitation rates, and fruit set.
4.3. Management Implications: Fire and Seismic Lines
Jack pine forests have evolved with fire as evidence of their aerial seedbanks of serotinous cones that require heat for the opening and release of seeds [13]. Post-disturbance boreal forest stands are often dominated by an understory of ericaceous species [4]. This flush in ericaceous productivity is an important phase of forest succession, providing a valuable habitat for wildlife. For instance, increasing the foraging opportunities of black bears [43] that are known to prefer seismic lines over forest stands, particularly in mid-to-late-summer due to increased berry production [44]. Indigenous peoples also benefit from post-fire resources with fire historically used to maintain a mosaic of successional stages that increase the diversity and abundance of forest resources, including berries from Vaccinium species [45]. Indeed, the gathering of Vaccinium berries is one of the most important aspects of indigenous traditions [46]. Therefore, management should consider the natural role that fire plays in jack pine forests and how changes in fire regimes may affect cultural values.
A number of considerations must be taken into account when considering seismic lines. These disturbances are known to affect wildlife by changing their behaviour and predator-prey relationships. In particular, woodland caribou in Northeastern Alberta are listed as threatened under the Federal Species at Risk Act [47] with seismic line restoration being a top priority [48] since these linear disturbances affect hunting and predation efficiency on caribou [49,50]. Black bear distribution in the forest is also affected by seismic line disturbances [44], possibly increasing opportunities for the human harvesting of black bear, as well as the predation of caribou calves by bears in recent disturbances [51]. Seismic lines also displace songbirds, although disturbances of smaller widths (canopy gaps) have less effect [52]. Regardless, there is general support for directly or indirectly promoting tree growth on seismic lines that fail to recover trees. Although the management of seismic lines is not directed towards Vaccinium myrtilloides, we expect such restoration actions that promote tree growth on seismic lines to reduce vigor and berry production similar to that of adjacent forests. Secondly, natural fires on these lines can interact to promote higher berry production. This may further affect wildlife, such as black bears, and the value of sites for harvesting by indigenous peoples.
5. Conclusions
Our study demonstrates that vigor and berry production in Vaccinium myrtilloides increases in narrow forest gaps associated with linear (seismic line) disturbances compared to adjacent forest stands after forest fires. We also found that as canopy gap (seismic line) width increases from 4 to 10 m, the cover, vigor, and berry production of Vaccinium myrtilloides also increases. This suggests that plants respond to changes in light and pollinators even within these small (4–10 m) forest gaps. Future research should manipulate these factors experimentally to further understand the mechanisms involved. Relative to wildfire, Vaccinium myrtilloides responded positively, except for high severity fires where declines were apparent. The management of seismic lines should consider the risks and benefits associated with restoration actions and wildfires on the habitat for Vaccinium myrtilloides if attempting to manage this species or species that utilize it, including wildlife (e.g., black bears) and indigenous peoples.
Acknowledgments
This work was conducted as part of the Boreal Ecosystem and Recovery Assessment (BERA) project with research and publication fees supported by grants from industry partners Cenovus Energy, Conoco Phillips Canada, and Alberta-Pacific Forest Industries, as well as matching funding from the Natural Sciences and Engineering Research Council of Canada (NSERC). Additional support was provided by the Alberta Biodiversity Conservation Chairs program funded by Canada’s Oil Sands Innovation Alliance (COSIA), NSERC, Alberta Innovates, and Alberta Agriculture and Forestry.
Author Contributions
S.E.N., A.T.F., and C.A.D. conceived and designed the experiments; A.T.F. and C.A.D. collected data; S.E.N. and C.A.D. analyzed the data; C.A.D. and S.E.N. wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest. The funding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
References
- Bradbury, S.M. Response of the Post-Fire Bryophyte Community to Salvage Logging in Boreal Mixedwood Forests of Northeastern Alberta, Canada. For. Ecol. Manag. 2006, 234, 313–322. [Google Scholar] [CrossRef]
- Kalamees, R.; Püssa, K.; Tamm, S.; Zobel, K. Adaptation to Boreal Forest Wildfire in Herbs: Responses to Post-Fire Environmental Cues in Two Pulsatilla Species. Acta Oecol. 2012, 38, 1–7. [Google Scholar] [CrossRef]
- Wein, R.W.; MacLean, D.A. The Role of Fire in Northern Circumpolar Ecosystems; Published on behalf of the Scientific Committee on Problems of the Environment of the International Council of Scientific Unions; Wiley: Chichester, UK, 1983. [Google Scholar]
- Mallik, A.U. Conifer Regeneration Problems in Boreal and Temperate Forests with Ericaceous Understory: Role of Disturbance, Seedbed Limitation, and Keytsone Species Change. Crit. Rev. Plant Sci. 2003, 22, 341–366. [Google Scholar] [CrossRef]
- Beaufait, W. Some Effects of High Temperatures on the Cones and Seeds of Jack Pine. For. Sci. 1960, 6, 194–198. [Google Scholar]
- Radeloff, V.C.; Mladenoff, D.J.; Guries, R.P.; Boyce, M.S. Spatial Patterns of Cone Serotiny in Pinus Banksiana in Relation to Fire Disturbance. For. Ecol. Manag. 2004, 189, 133–141. [Google Scholar] [CrossRef]
- Mallik, A.U. Ecology of a Forest Weed of Newfoundland: Vegetative Regeneration Strategy of Kalmia angustifolia. Can. J. Bot. 1993, 71, 161–166. [Google Scholar] [CrossRef]
- Moola, F.M.; Vasseur, L. The Importance of Clonal Growth to the Recovery of Gaultheria procumbens L. (Ericaceae) after Forest Disturbance. For. Ecol. Recent Adv. Plant Ecol. 2009, 319–337. [Google Scholar] [CrossRef]
- Ois Hébert, F.; Thiffault, N.; Ruel, J.-C.; Munson, A.D. Ericaceous Shrubs Affect Black Spruce Physiology Independently from Inherent Site Fertility. For. Ecol. Manag. 2010, 260, 219–228. [Google Scholar] [CrossRef]
- Vila, M.; Terradas, J. Effects of Competition and Disturbance on the Resprouting Performance of the Mediterranean Shrub Erica Multiflora L. (Ericaceae). Am. J. Bot. 1995, 82, 1241. [Google Scholar] [CrossRef]
- Neary, D.G.; Klopatek, C.C.; DeBano, L.F.; Ffolliott, P.F. Fire Effects on Belowground Sustainability: A Review and Synthesis. For. Ecol. Manag. 1999, 122, 51–71. [Google Scholar] [CrossRef]
- Pinno, B.D.; Errington, R.C. Burn Severity Dominates Understory Plant Community Response to Fire in Xeric Jack Pine Forests. Forests 2016, 7. [Google Scholar] [CrossRef]
- Alexander, M.E.; Cruz, M.G. Modelling the Effects of Surface and Crown Fire Behaviour on Serotinous Cone Opening in Jack Pine and Lodgepole Pine Forests. Int. J. Wildland Fire 2012, 21, 709–721. [Google Scholar] [CrossRef]
- Ilisson, T.; Chen, H.Y.H. The Direct Regeneration Hypothesis in Northern Forests. J. Veg. Sci. 2009, 20, 735–744. [Google Scholar] [CrossRef]
- Chiang, J.-M.; McEwan, R.W.; Yaussy, D.A.; Brown, K.J. The Effects of Prescribed Fire and Silvicultural Thinning on the Aboveground Carbon Stocks and Net Primary Production of Overstory Trees in an Oak-Hickory Ecosystem in Southern Ohio. For. Ecol. Manag. 2008, 255, 1584–1594. [Google Scholar] [CrossRef]
- Johnstone, J.F.; Chapin, F.S., III; Foote, J.; Kemmett, S.; Price, K.; Viereck, L. Decadal Observations of Tree Regeneration Following Fire in Boreal Forests. Can. J. For. Res. 2004, 34, 267–273. [Google Scholar] [CrossRef]
- Mallik, A.U. Conversion of Temperate Forests into Heaths: Role of Ecosystem Disturbance and Ericaceous Plants. Environ. Manag. 1995, 19, 675–684. [Google Scholar] [CrossRef]
- Feurdean, A.; Florescu, G.; Vannière, B.; Tanţău, I.; O‘Hara, R.B.; Pfeiffer, M.; Hutchinson, S.M.; Gałka, M.; Moskal-del Hoyo, M.; Hickler, T. Fire Has Been an Important Driver of Forest Dynamics in the Carpathian Mountains during the Holocene. For. Ecol. Manag. 2017, 389, 15–26. [Google Scholar] [CrossRef]
- Vizcaíno, E.A.D.; Rodríguez, A.I.; Fernández, M. Interannual Variability in Fire-Induced Germination Responses of the Characteristic Ericaceae of the NW Iberian Peninsula. For. Ecol. Manag. 2006, 234, S179. [Google Scholar] [CrossRef]
- Bunnell, F.L. Reproduction of Salal (Gaultheria Shallon) under Forest Canopy. Can. J. For. Res. 1990, 20, 91–100. [Google Scholar] [CrossRef]
- Schimmel, J.; Granström, A. Fire Severity and Vegetation Response in the Boreal Swedish Forest. Ecology 1996, 77, 1436–1450. [Google Scholar] [CrossRef]
- Hall, I.V. Floristic Changes Following the Cutting and Burning of a Woodlot for Blueberry Production. Can. J. Agric. Sci. 1955, 35, 143–152. [Google Scholar]
- Moola, F.M.; Mallik, A.U. Morphological Plasticity and Regeneration Strategies of Velvet Leaf Blueberry (Vaccinium myrtiltoides Michx.) Following Canopy Disturbance in Boreal Mixedwood Forests. For. Ecol. Manag. 1998, 111, 35–50. [Google Scholar] [CrossRef]
- Stocks, B.J.; Mason, J.A.; Todd, J.B.; Bosch, E.M.; Wotton, B.M.; Amiro, B.D.; Flannigan, M.D.; Hirsch, K.G.; Logan, K.A.; Martell, D.L.; et al. Large Forest Fires in Canada, 1959–1997. J. Geophys. Res. 2002, 108, 8149. [Google Scholar] [CrossRef]
- Bayne, E.M.; Habib, L.; Boutin, S. Impacts of Chronic Anthropogenic Noise from Energy-Sector Activity on Abundance of Songbirds in the Boreal Forest. Conserv. Biol. 2008, 22, 1186–1193. [Google Scholar] [CrossRef] [PubMed]
- Sheriff, R.E.; Geldart, L.P. Background Mathematics. In Exploration Seismology; Cambridge University Press: Cambridge, UK, 1995; pp. 517–568. [Google Scholar]
- Canham, C.D.; Denslow, J.S.; Platt, W.J.; Runkle, J.; Spies, T.A.; White, P.S. Light Regimes beneath Closed Canopies and Tree-Fall Gaps in Temperate and Tropical Forests. Can. J. For. Res. 1990, 20, 620. [Google Scholar] [CrossRef]
- Montané, F.; Guixé, D.; Camprodon, J. Canopy Cover and Understory Composition Determine Abundance of Vaccinium myrtillus L., a Key Plant for Capercaillie (Tetrao urogallus), in Subalpine Forests in the Pyrenees. Plant Ecol. Divers. 2016, 9, 187–198. [Google Scholar] [CrossRef]
- Arienti, M.C.; Cumming, S.G.; Krawchuk, M.A.; Boutin, S. Road Network Density Correlated with Increased Lightning Fire Incidence in the Canadian Western Boreal Forest. Int. J. Wildland Fire 2009, 18, 970–982. [Google Scholar] [CrossRef]
- Lee, P.; Boutin, S. Persistence and Developmental Transition of Wide Seismic Lines in the Western Boreal Plains of Canada. J. Environ. Manag. 2006, 78, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Van Rensen, C.K.; Nielsen, S.E.; White, B.; Vinge, T.; Lieffers, V.J. Natural Regeneration of Forest Vegetation on Legacy Seismic Lines in Boreal Habitats in Alberta’S Oil Sands Region. Biol. Conserv. 2015, 184, 127–135. [Google Scholar] [CrossRef]
- Dabros, A.; Hammond, H.E.J.; Pinzon, J.; Pinno, B.; Langor, D. Edge Influence of Low-Impact Seismic Lines for Oil Exploration on Upland Forest Vegetation in Northern Alberta (Canada). For. Ecol. Manag. 2017, 400, 278–288. [Google Scholar] [CrossRef]
- Historical Wildfire Database 1999–2014. AAF—Agriculture and Forestry. Available online: http://wildfire.alberta.ca/resources/historical-data/historical-wildfire-database.aspx (accessed on 14 July 2017).
- Peet, R.K.; Wentworth, T.R.; White, P.S. A Flexible, Multipurpose Method for Recording Vegetation Composition and Structure. Castanea 1998, 63, 262–274. [Google Scholar]
- Pitelka, L.F.; Stanton, D.S.; Peckenham, M.O. Effects of Light and Density on Resource Allocation in a Forest Herb, Aster Acuminatus (Compositae). Am. J. Bot. 1980, 67, 942. [Google Scholar] [CrossRef]
- Christensen, N.L. Fire and Soil-Plant Nutrient Relations in a Pine-Wiregrass Savanna on the Coastal Plain of North Carolina. Oecologia 1977, 31, 27–44. [Google Scholar] [CrossRef] [PubMed]
- Simms, E.L. The Effect of Nitrogen and Phosphorus Addition on the Growth, Reproduction, and Nutrient Dynamics of Two Ericaceous Shrubs. Oecologia 1987, 71, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Faison, E.K.; Del Tredici, P.; Foster, D.R. To Sprout or Not to Sprout: Multiple Factors Determine the Vigor of Kalmia Latifolia (Ericaceae) in Southwestern Connecticut. Rhodora 2014, 116, 148–162. [Google Scholar] [CrossRef]
- Ballester, A.; Vieitez, A.M.; Vieitez, E. Allelopathic Potential ofErica Vagans, Calluna Vulgaris, and Daboecia Cantabrica. J. Chem. Ecol. 1982, 8, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Watson, M.A.; Casper, B.B. Morphogenetic Constraints on Patterns of Carbon Distribution in Plants. Annu. Rev. Ecol. Syst. 1984, 15, 233–258. [Google Scholar] [CrossRef]
- Reader, R.J. Bog Ericad Flowers: Self-Compatibility and Relative Attractiveness to Bees. Can. J. Bot. 1977, 55, 2279–2287. [Google Scholar] [CrossRef]
- Usui, M.; Kevan, P.G.; Obbard, M. Pollination and Breeding System of Lowbush Blueberries, Vaccinium angustifolium Ait. and V. myrtilloides Michx. (Ericacaeae), in the Boreal Forest. Can. Field Nat. 2005, 119, 48. [Google Scholar] [CrossRef]
- Brodeur, V.; Ouellet, J.-P.; Courtois, R.; Fortin, D. Habitat Selection by Black Bears in an Intensively Logged Boreal Forest. Can. J. Zool. 2008, 86, 1307–1316. [Google Scholar] [CrossRef]
- Tigner, J.; Bayne, E.M.; Boutin, S. Black Bear Use of Seismic Lines in Northern Canada. J. Wildl. Manag. 2014, 78, 282–292. [Google Scholar] [CrossRef]
- Turner, N.J.; Cocksedge, W. Aboriginal Use of Non-Timber Forest Products in Northw Estern North America. J. Sustain. For. 2001, 13, 31–58. [Google Scholar] [CrossRef]
- Gottesfeld, L.M.J. Aboriginal Burning for Vegetation Management in Northwest British Columbia. Hum. Ecol. 1994, 22, 171–188. [Google Scholar] [CrossRef]
- Environment Canada. Recovery Strategy for the Woodland Caribou (Rangifer tarandus caribou), Boreal Population, in Canada; Species at Risk Act Recovery Strategy Series; Environment Canada: Ottawa, ON, Canada, 2012. [Google Scholar]
- Hebblewhite, M. Billion Dollar Boreal Woodland Caribou and the Biodiversity Impacts of the Global Oil and Gas Industry. Biol. Conserv. 2016, 206, 102–111. [Google Scholar] [CrossRef]
- James, A.R.C.; Stuart-Smith, A.K. Distribution of Caribou and Wolves in Relation to Linear Corridors. J. Wildl. Manag. 2000, 64, 154. [Google Scholar] [CrossRef]
- Edmonds, E.J. Population Status, Distribution, and Movements of Woodland Caribou in West Central Alberta. Can. J. Zool. 1988, 66, 817–826. [Google Scholar] [CrossRef]
- Pinard, V.; Dussault, C.; Ouellet, J.-P.; Fortin, D.; Courtois, R. Calving Rate, Calf Survival Rate, and Habitat Selection of Forest-Dwelling Caribou in a Highly Managed Landscape. J. Wildl. Manag. 2012, 76, 189–199. [Google Scholar] [CrossRef]
- Machtans, C.S. Songbird Response to Seismic Lines in the Western Boreal Forest: A Manipulative Experiment. Can. J. Zool. 2006, 84, 1421–1430. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).