Buying Time: Preliminary Assessment of Biocontrol in the Recovery of Native Forest Vegetation in the Aftermath of the Invasive Emerald Ash Borer
Abstract
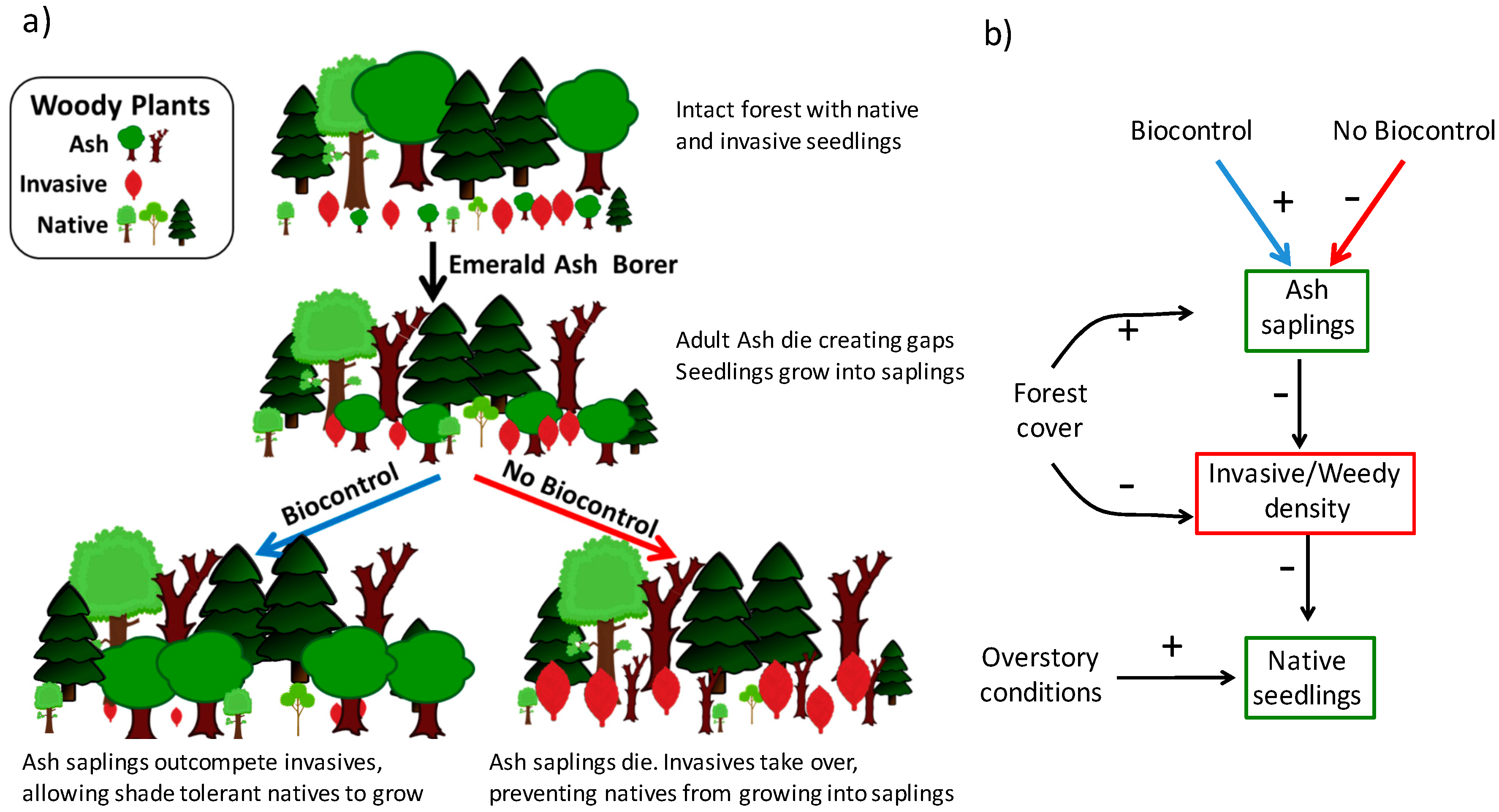
1. Introduction
2. Materials and Methods
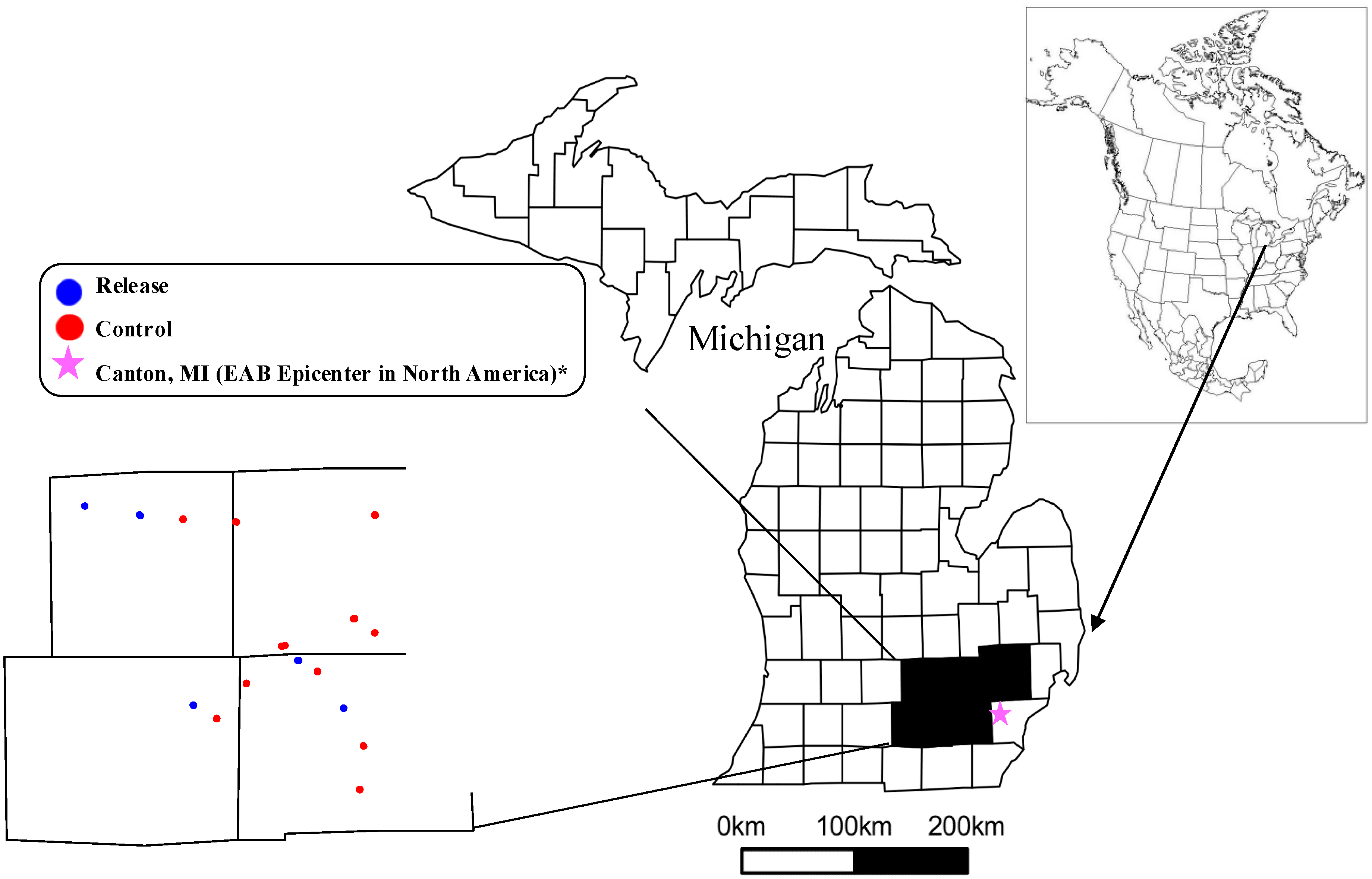
2.1. Study Sites
2.2. Biocontrol Releases
2.3. Vegetation Sampling
2.4. Environmental and Land Cover Data
2.5. Statistical Analysis
3. Results
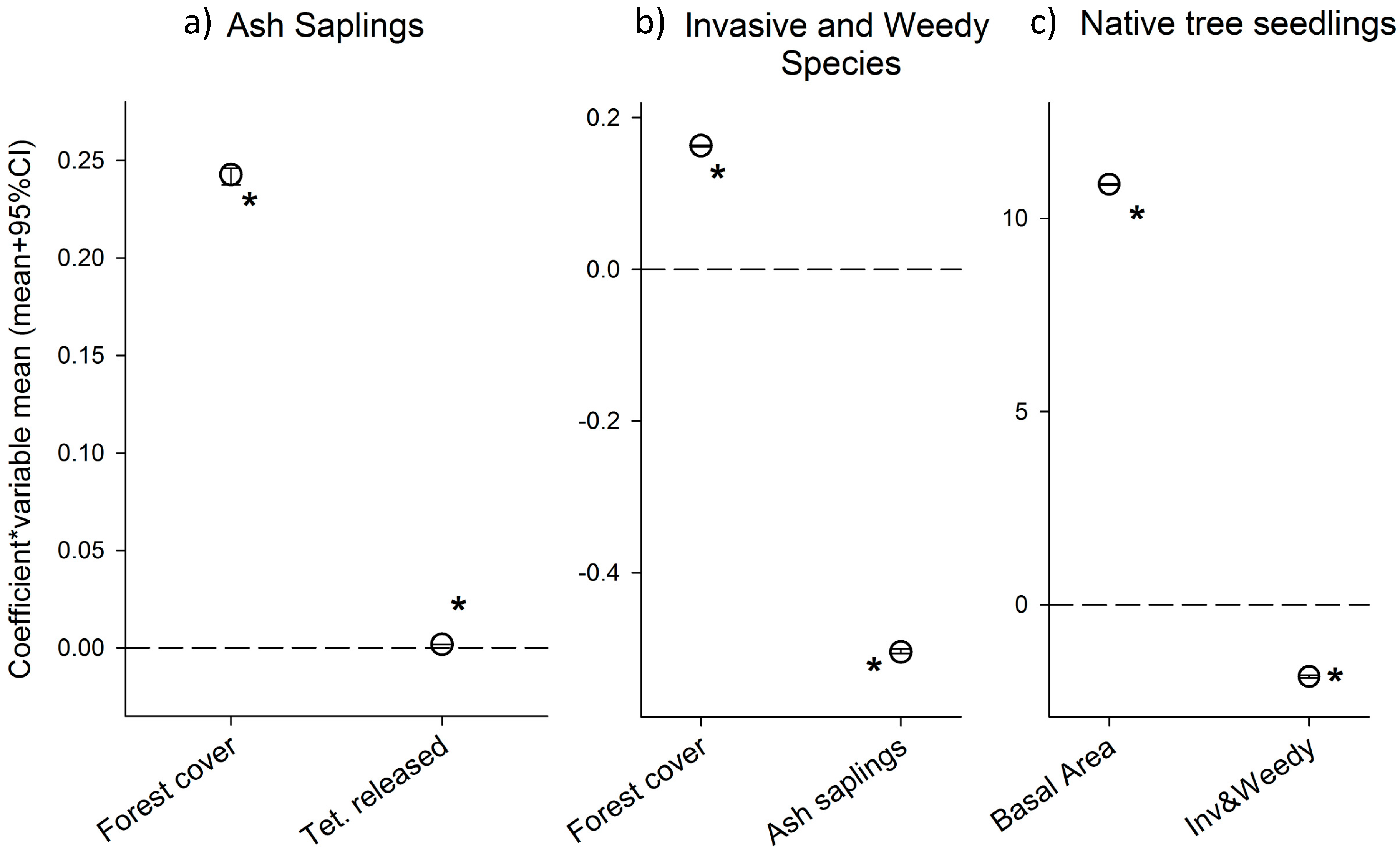
3.1. Results from the Ash Sapling Submodel
3.2. Results from the Invasive and Weedy Species Submodel
3.3 Results from the Native Seedlings Submodel
4. Discussion
4.1. Do Biocontrol Agents Affect Ash Sapling Density?
4.2. What Are the Variable Driving the Establishment of Invasive and Weedy Saplings in This System?
4.3. Is the Density of Invasive and Weedy Species Associated with Native Seedling Density?
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Liebhold, A.M.; Macdonald, W.L.; Bergdahl, D.; Mastro, V.C. Invasion by exotic pests: A threat to forest ecosystems. For. Sci. Monogr. 1995, 30, 1–49. [Google Scholar]
- Lovett, G.M.; Weiss, M.; Liebhold, A.M.; Holmes, T.P.; Leung, B.; Lambert, K.F.; Orwig, D.A.; Campbell, F.T.; Rosenthal, J.; Mccullough, D.G.; et al. Nonnative forest insects and pathogens in the United States: Impacts and policy options. Ecol. Appl. 2016, 26, 1437–1455. [Google Scholar] [CrossRef] [PubMed]
- Flower, C.E.; Gonzalez-Meler, M.A. Responses of Temperate Forest Productivity to Insect and Pathogen Disturbances. Annu. Rev. Plant Biol. 2015, 66, 547–569. [Google Scholar] [CrossRef] [PubMed]
- Aukema, J.E.; McCullough, D.G.; Von Holle, B.; Liebhold, A.M.; Britton, K.; Frankel, S.J. Historical Accumulation of Nonindigenous Forest Pests in the Continental United States. BioScience 2010, 60, 886–897. [Google Scholar] [CrossRef]
- Busby, P.E.; Canham, C.D. An exotic insect and pathogen disease complex reduces aboveground tree biomass in temperate forests of eastern North America. Can. J. For. Res. 2010, 41, 401–411. [Google Scholar] [CrossRef]
- Van de Gevel, S.L.; Hart, J.L.; Spond, M.D.; White, P.B.; Sutton, M.N.; Grissino-Mayer, H.D. American chestnut Castanea dentata to northern red oak Quercus rubra: Forest dynamics of an old-growth forest in the Blue Ridge Mountains; USA. Can. J. Bot. 2012, 90, 1263–1276. [Google Scholar] [CrossRef]
- Mosher, E.S.; Silander, J.A.; Latimer, A.M. The role of land-use history in major invasions by woody plant species in the northeastern North American landscape. Biol. Invasions 2009, 11, 2317–2328. [Google Scholar] [CrossRef]
- Lovett, G.M.; Canham, C.D.; Arthur, M.A.; Weathers, K.C.; Fitzhugh, R.D. 2006 Forest Ecosystem Responses to Exotic Pests and Pathogens in Eastern North America. BioScience 2006, 56, 395–405. [Google Scholar] [CrossRef]
- Liebhold, A.M.; Mccullough, D.G.; Blackburn, L.M.; Frankel, S.J.; Von Holle, B.; Aukema, J.E. A highly aggregated geographical distribution of forest pest invasions in the USA. Divers. Distrib. 2013, 19, 1208–1216. [Google Scholar] [CrossRef]
- Morin, R.S.; Liebhold, A.M.; Pugh, S.A.; Crocker, S.J. Regional assessment of emerald ash borer, Agrilus planipennis, impacts in forests of the Eastern United States. Biol. Invasions 2017, 19, 703–711. [Google Scholar] [CrossRef]
- Cappaert, D.L.; McCullough, D.G.; Poland, T.M.; Siegert, N.W. Emerald ash borer in North America: A research and regulatory challenge. Am. Entomol. 2005, 51, 152–165. [Google Scholar] [CrossRef]
- Rebek, E.J.; Herms, D.A.; Smitley, D.R. Interspecific Variation in Resistance to Emerald Ash Borer (Coleoptera: Buprestidae) Among North American and Asian Ash (Fraxinus spp.). Environ. Entomol. 2008, 37, 242–246. [Google Scholar] [CrossRef]
- Haack, R.A.; Jendek, E.; Liu, H.; Marchant, K.R.; Petrice, T.R.; Poland, T.M.; Ye, H. The emerald ash borer: A new exotic pest in North America. Newsl. Mich. Entomol. Soc. 2002, 47, 1–5. [Google Scholar]
- Poland, T.M.; McCullough, D.G. Emerald ash borer: Invasion of the urban forest and the threat to North America’s ash resource. J. For. 2006, 104, 118–124. [Google Scholar]
- USDA-APHIS. Emerald Ash Borer. Available online: https://www.aphis.usda.gov/plant_health/plant_pest_info/emerald_ash_b/downloads/MultiState.pdf (accessed on 10 July 2017).
- Liu, H.; Bauer, L.S.; Gao, R.; Zhao, T.; Petrice, T.R.; Haack, R.A. Exploratory survey for the emerald ash borer; Agrilus planipennis Coleoptera: Buprestidae; and its natural enemies in China. Gt. Lakes Entomol. 2003, 36, 191–204. [Google Scholar]
- Eyles, A.; Jones, W.; Riedl, K.; Cipollini, D.; Schwartz, S.; Chan, K.; Herms, D.A.; Bonello, P. Comparative phloem chemistry of Manchurian Fraxinus mandshurica and two North American ash species Fraxinus americana and Fraxinus pennsylvanica. J. Chem. Ecol. 2007, 33, 1430–1448. [Google Scholar] [CrossRef] [PubMed]
- Villari, C.; Herms, D.A.; Whitehill, J.G.A.; Cipollini, D.; Bonello, P. Progress and gaps in understanding mechanisms of ash tree resistance to emerald ash borer; a model for wood-boring insects that kill angiosperms. New Phytol. 2016, 209, 63–79. [Google Scholar] [CrossRef] [PubMed]
- Anulewicz, A.C.; McCullough, D.G.; Cappaert, D.L.; Poland, T.M. Host range of the emerald ash borer Agrilus planipennis Fairmaire Coleoptera: Buprestidae in North America: Results of multiple-choice field experiments. Environ. Entomol. 2008, 37, 230–241. [Google Scholar] [CrossRef]
- Gandhi, K.J.K.; Herms, D.A. Potential biodiversity loss due to impending devastation of the North American genus Fraxinus by the exotic emerald ash borer. Biol. Invasions 2010, 12, 1839–1846. [Google Scholar] [CrossRef]
- Spei, B.A.; Kashian, D.M. Potential for persistence of blue ash in the presence of emerald ash borer in southeastern Michigan. For. Ecol. Manag. 2017, 392, 137–143. [Google Scholar] [CrossRef]
- Flower, C.E.; Knight, K.S.; Gonzalez-Meler, M.A. Impacts of the emerald ash borer Agrilus planipennis Fairmaire induced ash Fraxinus spp. mortality on forest carbon cycling and successional dynamics in the eastern United States. Biol. Invasions 2013, 15, 931–944. [Google Scholar] [CrossRef]
- Sadof, C.S.; Hughes, G.P.; Witte, A.R.; Peterson, D.J.; Ginzel, M.D. Tools for staging and managing emerald ash borer in the urban forest. Arboric. Urban For. 2007, 43, 15–26. [Google Scholar]
- Nowak, D.; Crane, D.; Stevens, J.; Walton, J. Potential Damage from Emerald Ash Borer; United States Department of Agriculture; Forest Service; Northern Research Station: Syracuse, NY, USA, 2003; pp. 1–5. Available online: https://www.nrs.fs.fed.us/disturbance/invasive_species/eab/local-resources/downloads/EAB_potential.pdf (accessed on 26 September 2017).
- Federal Register 2003. Emerald Ash Borer; Quarantine and Regulations. 7 CFR Part 301 [docket Number 02-125-1]. Available online: https://www.federalregister.gov/articles/2003/10/14/03-25881/emerald-ash-borer-quarantine-and-regulations (accessed on 10 May 2017).
- Herms, D.A.; McCullough, D.G. Emerald ash borer invasion of North America: History, biology, ecology, impacts, and management. Annu. Rev. Entomol. 2014, 59, 13–30. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.B.; Duan, J.J.; Fuester, R.W.; Hoddle, M.; Van Driesche, R.G. Parasitoid Guilds of Agrilus Woodborers Coleoptera: Buprestidae: Their Diversity and Potential for Use in Biological Control. Psyche 2012, 2012, 1–10. [Google Scholar] [CrossRef]
- Bauer, L.S.; Duan, J.J.; Gould, J.R. Emerald ash borer Agrilus planipennis Fairmaire Coleoptera: Buprestidae. In The Use of Classical Biological Control to Preserve Forests in North America; van Driesche, R., Reardon, R., Eds.; United States Department of Agriculture; Forest Service; Forest Health and Technology Enterprise Team; FHTET-2013-2: Morgantown, WV, USA, 2014; pp. 189–209. [Google Scholar]
- Duan, J.J.; Yurchenko, G.; Fuester, R. Occurrence of emerald ash borer Coleoptera: Buprestidae and biotic factors affecting its immature stages in the Russian Far East. Environ. Entomol. 2012, 41, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Y.; Jennings, D.J.; Duan, J.J. Trade-offs in parasitism efficiency and brood size mediate parasitoid coexistence; with implications for biological control of the invasive emerald ash borer. J. Appl. Ecol. 2015, 52, 1255–1263. [Google Scholar] [CrossRef]
- Bauer, L.S.; Duan, J.J.; Gould, J.R.; Van Driesche, R.G. Progress in the classical biological control of Agrilus planipennis Fairmaire Coleoptera: Buprestidae in North America. Can. Entomol. 2015, 147, 300–317. [Google Scholar] [CrossRef]
- Federal Register. Availability of an Environmental Assessment for the Proposed Release of Three Parasitoids for the Biological Control of the Emerald Ash Borer Agrilus planipennis in the Continental United States. Fed. Regist. 2007, 72, 28947–28948, [docket number APHIS-2007-006]. Available online: http://www.regulations.gov/#!documentDetail; D=APHIS-2007-0060-0043 (accessed on 10 May 2017).
- Bauer, L.; Jennings, D.; Duan, J.; van Driesche, R.; Gould, J.; Kashian, D.; Miller, D.; Petrice, T.; Morris, E.; Poland, T. Recent Progress in Biological Control of Emerald Ash Borer. In Proceedings of the 27th USDA Interagency Research Forum on Invasive Species, Annapolis, MD, USA, 12–15 January 2016; pp. 22–25. [Google Scholar]
- Liu, H.; Bauer, L.S.; Miller, D.L.; Zhao, T.; Gao, R.; Song, L.; Luan, Q.; Jin, R.; Gao, C. Seasonal abundance of Agrilus planipennis Coleoptera: Buprestidae and its natural enemies Oobius agrili Hymenoptera: Encyrtidae and Tetrastichus planipennisi Hymenoptera: Eulophidae in China. Biol. Control 2007, 42, 61–71. [Google Scholar] [CrossRef]
- Knight, K.S.; Brown, J.P.; Long, R.P. Factors affecting the survival of ash Fraxinus spp. trees infested by emerald ash borer Agrilus planipennis. Biol. Invasions 2013, 15, 371–383. [Google Scholar] [CrossRef]
- Kashian, D.M. Sprouting and seed production may promote persistence of green ash in the presence of the emerald ash borer. Ecosphere 2016, 7, 1–15. [Google Scholar] [CrossRef]
- Duan, J.J.; Bauer, L.S.; Van Driesche, R.G. Emerald ash borer biocontrol in ash saplings: The potential for early stage recovery of North American ash trees. For. Ecol. Manag. 2017, 394, 64–72. [Google Scholar] [CrossRef]
- Schlesinger, R.C. Fraxinus americana L.: White ash. In Silvics of North America: Hardwoods; Agriculture Handbook 654; Burns, R.M., Honkala, B.H., Eds.; USDA Forest Service: Washington, DC, USA, 1990; pp. 333–338. [Google Scholar]
- Kashian, D.M.; Witter, J.A. Assessing the potential for ash canopy tree replacement via current regeneration following emerald ash borer-caused mortality on southeastern Michigan landscapes. For. Ecol. Manag. 2011, 261, 480–488. [Google Scholar] [CrossRef]
- Abell, K.J.; Duan, J.J.; Bauer, L.; Lelito, J.P.; Van Driesche, R.G. The effect of bark thickness on host partitioning between Tetrastichus planipennisi Hymen: Eulophidae and Atanycolus spp. Hymen: Braconidae; two parasitoids of emerald ash borer Coleop: Buprestidae. Biol. Control 2012, 63, 320–325. [Google Scholar] [CrossRef]
- Duan, J.J.; Oppel, C.B. Critical rearing parameters of Tetrastichus planipennisi Hymenoptera: Eulophidae as affected by host-plant substrate and host-parasitoid group structure. J. Econ. Entomol. 2012, 105, 792–801. [Google Scholar] [CrossRef] [PubMed]
- Costilow, K.C.; Knight, K.S.; Flower, C.E. Disturbance severity and canopy position control the radial growth response of maple trees (Acer spp.) in forests of northwest Ohio impacted by emerald ash borer (Agrilus planipennis). Ann. For. Sci. 2017, 74, 1–10. [Google Scholar] [CrossRef]
- Rejmanek, M. Invasibility of Plant Communities. In Biological Invasions: A Global Perspective; Drake, J.A., Mooney, H., di Castri, F., Groves, R., Kruger, F., Rejmánek, M., Williamson, M., Eds.; Wiley: Chichester, UK, 1989; pp. 369–388. [Google Scholar]
- Baraloto, C.; Goldberg, D.E.; Bonal, D. Performance Trade-offs among Tropical Tree Seedlings in Contrasting Microhabitats. Ecology 2005, 86, 2461–2472. [Google Scholar] [CrossRef]
- Muller-Landau, H.C.; Wright, S.J.; Calderón, O.; Condit, R.; Hubbell, S.P. Interspecific variation in primary seed dispersal in a tropical forest. J. Ecol. 2008, 96, 653–667. [Google Scholar] [CrossRef]
- Zhao, F.; Qi, L.; Fang, L.; Yang, J. Influencing factors of seed long-distance dispersal on a fragmented forest landscape on Changbai Mountains; China. Chin. Geogr. Sci. 2016, 26, 68–77. [Google Scholar] [CrossRef]
- Greene, D.F.; Johnson, E.A. Modelling the temporal variation in the seed production of North American trees. Can. J. For. Res. 2004, 34, 65–75. [Google Scholar] [CrossRef]
- Jasper, J.M.; Bleher, B.; Bohing-Gaese, K.; Chira, R.; Farwig, N. Fragmentation and local disturbance of forests reduce frugivore diversity and fruit removal in Ficus thonningii trees. Basic Appl. Ecol. 2008, 9, 663–672. [Google Scholar]
- Lundgren, M.R.; Small, C.J.; Dreyer, G.D. Influence of Land Use and Site Characteristics on Invasive Plant Abundance in the Quinebaug Highlands of Southern New England. Northeast. Nat. 2004, 11, 313–332. [Google Scholar] [CrossRef]
- With, K.A. The landscape ecology of invasive species. Conserv. Biol. 2002, 16, 1192–1203. [Google Scholar] [CrossRef]
- González-Moreno, P.; Diez, J.M.; Ibáñez, I.; Font, X.; Vilà, M. Plant invasions are context-dependent: Multiscale effects of climate; human activity and habitat. Divers. Distrib. 2014, 20, 720–731. [Google Scholar] [CrossRef]
- Stachowicz, J.J.; Tilman, D. Species Invasion and the Relationships between Species Diversity, Community Saturation and Ecosystem Functioning. In Species Invasions: Insights into Ecology, Evolution, and Biogeography; Sax, D.F., Ed.; Sinauer Associates Inc.: Massachusetts, MA, USA, 2005; pp. 41–64. [Google Scholar]
- Huston, M.A. Management strategies for plant invasions: Manipulating productivity; disturbance; and competition. Divers. Distrib. 2004, 10, 167–178. [Google Scholar] [CrossRef]
- Pavlovic, N.B.; Leicht-Young, S.A. Are temperate mature forests buffered from invasive lianas? J. Torrey Bot. Soc. 2011, 138, 85–92. [Google Scholar] [CrossRef]
- Simberloff, D.; Souza, L.; Nuñez, M.A.; Barrios-Garcia, M.N.; Bunn, W. The natives are restless, but not often and mostly when disturbed. Ecology 2012, 93, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Von Holle, B.; Delcourt, H.R.; Simberloff, D. Theimportance of biological inertia in pant community resistance to invasion. J. Veg. Sci. 2003, 14, 425–432. [Google Scholar] [CrossRef]
- Brym, Z.T.; Allen, D.; Ibáñez, I. Community control on growth and survival of an exotic shrub. Biol. Invasions 2014, 16, 2529–2541. [Google Scholar] [CrossRef]
- Davis, J.C.; Shannon, J.P.; Bolton, N.W.; Kolka, R.K.; Pypker, T.G. Vegetation responses to simulated emerald ash borer infestation in Fraxinus nigra dominated wetlands of Upper Michigan; USA. Can. J. For. Res. 2017, 47, 319–330. [Google Scholar] [CrossRef]
- Hausman, C.E.; Jaeger, J.F.; Rocha, O.J. Impacts of the emerald ash borer EAB eradication and tree mortality: Potential for a secondary spread of invasive plant species. Biol. Invasions 2010, 12, 2013–2023. [Google Scholar] [CrossRef]
- Siegert, N.W.; McCullough, D.G.; Liebhold, A.M.; Telewski, F.W. Dendrochronological reconstruction of the epicentre and early spread of emerald ash borer in North America. Divers. Distrib. 2014, 20, 847–858. [Google Scholar] [CrossRef]
- Mapbiocontrol Agent Release Tracking and Data Management for Federal; State; and Researchers Releasing Three Biocontrol Agents Released Against Emerald Ash Borer. 2017. Available online: www.mapbiocontrol.org (accessed on 14 August 2014).
- Duan, J.J.; Bauer, L.S.; Abell, K.J.; Lelito, J.P.; Van Driesche, R.G. Establishment and abundance of Tetrastichus planipennisi (Hymenoptera: Eulophidae) in Michigan: Potential for success in classical biocontrol of the invasive emerald ash borer (Coleoptera: Buprestidae) establishment and abundance of Tetrastichus planipennisi. J. Econ. Entomol. 2013, 106, 1145–1154. [Google Scholar] [PubMed]
- Abell, K.J.; Bauer, L.S.; Duan, J.J.; Van Driesche, R.G. Long-term monitoring of the introduced emerald ash borer Coleoptera: Buprestidae egg parasitoid; Oobius agrili Hymenoptera: Encyrtidae, in Michigan, USA and evaluation of a newly developed monitoring technique. Biol. Control 2014, 79, 36–42. [Google Scholar] [CrossRef]
- Michigan Geographic Data Library. 2014. Available online: http://www.mcgi.state.mi.us/mgdl/?rel=ext&action=sext (accessed on 20 May 2014).
- Clark, J.S. Why environmental scientists are becoming Bayesians. Ecol. Lett. 2005, 8, 2–14. [Google Scholar] [CrossRef]
- Spiegelhalter, D.J.; Best, N.G.; Carlin, B.P.; van Der Linde, A. Bayesian Measures of Model Complexity and Fit. J. R. Stat. Soc. 2002, 64, 583–639. [Google Scholar] [CrossRef]
- Chytrý, M.; Jarošík, V.; Pyšek, P.; Hájek, O.; Knollová, I.; Tichy, L.; Danihelka, J. 2008 Separating habitat invasibility by alien plants from the actual level of invasion. Ecology 2008, 89, 1541–1553. [Google Scholar] [CrossRef] [PubMed]
- González-Moreno, P.; Pino, J.; Carreras, D.; Basnou, C.; Fernández-Rebollar, I.; Vilà, M. Quantifying the landscape influence on plant invasions in Mediterranean coastal habitats. Landsc. Ecol. 2003, 28, 891–903. [Google Scholar] [CrossRef]
- Ilisson, T.; Chen, H.Y.H. The direct regeneration hypothesis in northern forests. J. Veg. Sci. 2009, 20, 735–744. [Google Scholar] [CrossRef]
- Gelman, A.; Hill, J. Data Analysis Using Regression and Multilevel/Hierarchical Models; Cambridge University Press: New York, NY, USA, 2007. [Google Scholar]
- Thomas, A.; O’Hara, B.; Ligges, U.; Sturtz, S. Making BUGS open. R News 2006, 6, 12–17. [Google Scholar]
- Meyer, J.Y.; Fourdrigniez, M. Conservation benefits of biological control: The recovery of a threatened plant subsequent to the introduction of a pathogen to contain an invasive tree species. Biol. Conserv. 2011, 144, 106–113. [Google Scholar] [CrossRef]
- Denslow, J.S.; D’Antonio, C.M.D. After biocontrol: Assessing indirect effects of insect releases. Biol. Control 2005, 35, 307–318. [Google Scholar] [CrossRef]
- Barton, J.; Fowler, S.V.; Gianotti, A.F.; Winks, C.J.; Beurs, M.D.; Arnold, G.C.; Forrester, G. Successful biological control of mist flower Ageratina riparia in New Zealand: Agent establishment; impact and benefits to the native flora. Biol. Control 2007, 40, 370–385. [Google Scholar] [CrossRef]
- Duan, J.J.; Bauer, L.S.; Abell, K.J.; Ulyshen, M.D.; Van Driesche, R.G. Population dynamics of an invasive forest insect and associated natural enemies in the aftermath of invasion: Implications for biological control. J. Appl. Ecol. 2015, 52, 1246–1254. [Google Scholar] [CrossRef]
- Aubin, I.; Cardou, F.; Ryall, K.; Kreutzweiser, D.; Scarr, T. Ash regeneration capacity after emerald ash borer (EAB) outbreaks: Some early results. For. Chron. 2015, 91, 291–298. [Google Scholar] [CrossRef]
- Wang, X.; Yang, Z.; Wu, H.; Liu, S.; Wang, H.; Bai, L. Parasitism and reproductive biology of Spathius agrili Yang Hymenoptera: Braconidae. Acta Entomol. Sin. 2007, 50, 920–926. [Google Scholar]
- Duan, J.J.; Bauer, L.S.; Hansen, J.A.; Abell, K.J.; Van Driesche, R.G. An improved method for monitoring parasitism and establishment of Oobius agrili (Hymenoptera: Encyrtidae), an egg parasitoid introduced for biological control of the emerald ash borer (Coleoptera: Buprestidae) in North America. Biol. Control 2012, 60, 255–261. [Google Scholar] [CrossRef]
- Martin, P.H.; Canham, C.D.; Marks, P.L. Why forests appear resistant to exotic plant invasions: Intentional introductions, stand dynamics, and the role of shade tolerance. Front. Ecol. Envirion. 2009, 7, 142–179. [Google Scholar] [CrossRef]
- Yates, E.D.; Levia, D.F.; Williams, C.L. Recruitment of three non-native invasive plants into a fragmented forest in southern Illinois. For. Ecol. Manag. 2004, 190, 119–130. [Google Scholar] [CrossRef]
- Flory, S.L.; Clay, K. Effects of roads and forest successional age on experimental plant invasions. Biol. Conserv. 2009, 142, 2531–2537. [Google Scholar] [CrossRef]
- Kuuluvainen, T. Gap disturbance, ground microtopography; and the regeneration dynamics of boreal coniferous forests in Finland: A review. Ann. Zool. Fenn. 1994, 31, 35–51. [Google Scholar]
- Kueffer, C.; Schumacher, E.; Dietz, H.; Fleischmann, K.; Edwards, P.J. Managing successional trajectories in alien-dominated; novel ecosystems by facilitating seedling regeneration: A case study. Biol. Conserv. 2010, 143, 1792–1802. [Google Scholar] [CrossRef]
- Thompson, J.R.; Carpenter, D.N.; Cogbill, C.V.; Foster, D.R. Four centuries of change in northeastern United States forests. PLoS ONE 2013, 8, e72540. [Google Scholar] [CrossRef] [PubMed]
- Vilà, M.; Ibáñez, I. Plant invasions in the landscape. Landsc. Ecol. 2011, 26, 461–472. [Google Scholar] [CrossRef]
- Dickmann, D.L.; Leefers, L.A. The Forests of Michigan; University of Michigan Press: Ann Arbor, MI, USA, 2003. [Google Scholar]
- Hutchinson, T.F.; Vankat, J.L. Society for Conservation Biology Invasibility and Effects of Amur Honeysuckle in Southwestern Ohio Forests. Conserv. Biol. 1997, 11, 1117–1124. [Google Scholar]
- Ohlemüller, R.; Walker, S.; Wilson, J.B.; Memmott, J. Local vs. regional factors as determinants of the invasibility of indigenous forest fragments by alien plant species. Oikos 2006, 112, 493–501. [Google Scholar] [CrossRef]
- Lookwood, J.L.; Hoopes, M.F.; Marchetti, M.P. Invasion Ecology; Wiley-Blackwell: Oxford, UK, 2007. [Google Scholar]
- Ruckli, R.; Rusterholz, H.P.; Baur, B. Invasion of an annual exotic plant into deciduous forests suppresses arbuscular mycorrhiza symbiosis and reduces performance of sycamore maple saplings. For. Ecol. Manag. 2014, 318, 285–293. [Google Scholar] [CrossRef]
- Levine, J.M.; Vilà, M.; D’Antonio, C.M.; Dukes, J.S.; Grigulis, K.; Lavorel, S. Mechanisms underlying the impacts of exotic plant invasions. Proc. R. Soc. Lond. Biol. Sci. 2003, 270, 775–781. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, D.M.; Hufbauer, R.A. Increased plant size in exotic populations: A common-garden test with 14 invasive species. Ecology 2007, 88, 2758–2765. [Google Scholar] [CrossRef] [PubMed]
- Yih, K.; Boucher, D.H.; Vandermeer, J.H.; Zamora, N. Recovery of the rain forest of southeastern Nicaragua after destruction by Hurricane Joan. Biotropica 1991, 23, 106–113. [Google Scholar] [CrossRef]
| Parameter | Mean ± SD | 95% CI | ||
|---|---|---|---|---|
| Ash saplings submodel: | ||||
| α1 | intercept | −0.382 ± 0.002 | −0.385 | −0.377 |
| α2 | forest cover | 0.406 ± 0.003 | 0.398 | 0.412 |
| α3 | number of T. planipennesis released | 0.001736 ± 0.0000019 | 0.001734 | 0.001737 |
| Invasive and weedy species submodel: | ||||
| β1 | intercept | 0.8999 ± 0.00015 | 0.8996 | 0.9002 |
| β2 | ash saplings | −1.506 ± 0.005 | −1.512 | −1.492 |
| β3 | forest cover | 0.2737 ± 0.0015 | 0.2715 | 0.2753 |
| Native seedlings submodel: | ||||
| γ1 | intercept | 1.97 ± 0.19 | −2.31 | −1.64 |
| γ2 | basal area | 3830 ± 3.62 | 3822 | 3835 |
| γ3 | invasive and weedy saplings | −4.95 ± 0.05 | −5.04 | −4.86 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Margulies, E.; Bauer, L.; Ibáñez, I. Buying Time: Preliminary Assessment of Biocontrol in the Recovery of Native Forest Vegetation in the Aftermath of the Invasive Emerald Ash Borer. Forests 2017, 8, 369. https://doi.org/10.3390/f8100369
Margulies E, Bauer L, Ibáñez I. Buying Time: Preliminary Assessment of Biocontrol in the Recovery of Native Forest Vegetation in the Aftermath of the Invasive Emerald Ash Borer. Forests. 2017; 8(10):369. https://doi.org/10.3390/f8100369
Chicago/Turabian StyleMargulies, Elan, Leah Bauer, and Inés Ibáñez. 2017. "Buying Time: Preliminary Assessment of Biocontrol in the Recovery of Native Forest Vegetation in the Aftermath of the Invasive Emerald Ash Borer" Forests 8, no. 10: 369. https://doi.org/10.3390/f8100369
APA StyleMargulies, E., Bauer, L., & Ibáñez, I. (2017). Buying Time: Preliminary Assessment of Biocontrol in the Recovery of Native Forest Vegetation in the Aftermath of the Invasive Emerald Ash Borer. Forests, 8(10), 369. https://doi.org/10.3390/f8100369






