Soil Elements Influencing Community Structure in an Old-Growth Forest in Northeastern China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Data Acquisition
2.2. Relationship between Species Distribution and Soil Elements
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tuomisto, H.; Ruokolainen, K.; Aguilar, M.; Sarmiento, A. Floristic patterns along a 43-km long transect in an Amazonian rain forest. J. Ecol. 2003, 91, 743–756. [Google Scholar] [CrossRef]
- Thessler, S.; Ruokolainen, K.; Tuomisto, H.; Tomppo, E. Mapping gradual landscape-scale floristic changes in Amazonian primary rain forests by combining ordination and remote sensing. Glob. Ecol. Biogeogr. 2005, 14, 315–325. [Google Scholar] [CrossRef]
- Slik, J.W.; Raes, N.; Aiba, S.I.; Brearley, F.Q.; Cannon, C.H.; Meijaard, E.; Nagamasu, H.; Nilus, R.; Paoli, G.; Poulsen, A.D.; et al. Environmental correlates for tropical tree diversity and distribution patterns in Borneo. Divers. Distrib. 2009, 15, 523–532. [Google Scholar] [CrossRef]
- Clark, D.B.; Olivas, P.C.; Oberbauer, S.F.; Clark, D.A.; Ryan, M.G. First direct landscape-scale measurement of tropical rain forest leaf area index, a key driver of global primary productivity. Ecol. Lett. 2008, 11, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Gleason, S.M.; Read, J.; Ares, A.; Metcalfe, D.J. Species-soil associations, disturbance, and nutrient cycling in an Australian tropical rainforest. Oecologia 2010, 162, 1047–1058. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Mi, X.; Shao, H.; Ma, K. Strong plant-soil associations in a heterogeneous subtropical broad-leaved forest. Plant Soil 2011, 347, 211–220. [Google Scholar] [CrossRef]
- John, R.; Dalling, J.W.; Harms, K.E.; Yavitt, J.B.; Stallard, R.F.; Mirabello, M.; Hubbell, S.P.; Valencia, R.; Navarrete, H.; Vallejo, M.; et al. Soil nutrients influence spatial distributions of tropical tree species. Proc. Natl. Acad. Sci. USA 2007, 104, 864–869. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhao, Y.; Zhao, X.; Gadow, K.V. Species-Habitat Associations in a Northern Temperate Forest in China. Silva Fenn. 2012, 46, 501–519. [Google Scholar] [CrossRef]
- Hubbell, S.P.; Foster, R.B.; O’Brien, S.T.; Harms, K.E.; Condit, R.; Wechsler, B.; Wright, S.J.; De Lao, S.L. Light-gap disturbances, recruitment limitation, and tree diversity in a Neotropical forest. Science 1999, 283, 554–557. [Google Scholar] [CrossRef] [PubMed]
- Levine, J.M.; Murrell, D.J. The community-level consequences of seed dispersal patterns. Annu. Rev. Ecol. Evol. Syst. 2003, 34, 549–574. [Google Scholar] [CrossRef]
- Beckage, B.; Clark, J.S. Seedling survival and growth of three forest tree species: The role of spatial heterogeneity. Ecology 2003, 84, 1849–1861. [Google Scholar] [CrossRef]
- Lebrija-Trejos, E.; Pérez-García, E.A.; Meave, J.A.; Bongers, F.; Poorter, L. Functional traits and environmental filtering drive community assembly in a species-rich tropical system. Ecology 2010, 91, 386–398. [Google Scholar] [CrossRef] [PubMed]
- Harper, J.L. Population Biology of Plants; Academic Press: London, UK, 1977. [Google Scholar]
- Tilman, D.; Pacala, S. The maintenance of species richness in plant communities. In Species Diversity in Ecological Communities; Ricklefs, R.E., Ed.; University of Chicago Press: Chicago, IL, USA, 1993; pp. 13–25. [Google Scholar]
- Webb, C.O.; Peart, D.R. Habitat associations of trees and seedlings in a Bornean rain forest. J. Ecol. 2000, 88, 464–478. [Google Scholar] [CrossRef]
- Webb, C.O. Exploring the phylogenetic structure of ecological communities: An example for rain forest trees. J. Am. Nat. 2000, 156, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.B. Patterns in the balance of nature and related problems of quantitative ecology. Academic Press: New York, NY, USA, 1964. [Google Scholar]
- Bartels, S.F.; Chen, H.Y. Is understory plant species diversity driven by resource quantity or resource heterogeneity? Ecology 2010, 91, 1931–1938. [Google Scholar] [CrossRef] [PubMed]
- Baldeck, C.A.; Harms, K.E.; Yavitt, J.B.; John, R.; Turner, B.L.; Valencia, R.; Navarrete, H.; Davies, S.J.; Chuyong, G.B.; Kenfack, D.; et al. Soil resources and topography shape local tree community structure in tropical forests. Proc. R. Soc. Lond. B Biol. 2013, 280, 20122532. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.S.; McKenna, J.J.; Ashton, P.M.S.; Gregoire, T.G. Habitat characterizations underestimate the role of edaphic factors controlling the distribution of Entandrophragma. Ecology 2004, 85, 2171–2183. [Google Scholar] [CrossRef]
- Bonifacio, E.; Caimi, A.; Falsone, G.; Trofimov, S.Y.; Zanini, E.; Godbold, D.L. Soil properties under Norway spruce differ in spruce dominated and mixed broadleaf forests of the Southern Taiga. Plant Soil 2008, 308, 149–159. [Google Scholar] [CrossRef]
- Institute of Soil Science, CAS. Chinese Soil Taxonomy; Science Press: Beijing, China, 2001. [Google Scholar]
- Zhang, C.; Wei, Y.; Zhao, X.; von Gadow, K. Spatial characteristics of tree diameter distributions in a temperate old-growth forest. PLoS ONE 2013, 8, e58983. [Google Scholar] [CrossRef] [PubMed]
- China Soil Council. Soil Agricultural Chemical Analysis Procedure; Chinese Agricultural Science Press: Beijing, China, 1999. [Google Scholar]
- Harms, K.E.; Condit, R.; Hubbell, S.P.; Foster, R.B. Habitat associations of trees and shrubs in a 50-ha Neotropical forest plot. J. Ecol. 2001, 89, 947–959. [Google Scholar] [CrossRef]
- Waagepetersen, R.P. An estimating function approach to inference for inhomogeneous Neyman-Scott processes. Biometrics 2007, 63, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Shen, G.; Yu, M.; Hu, X.S.; Mi, X.; Ren, H.; Sun, I.F.; Ma, K. Species-area relationships explained by the joint effects of dispersal limitation and habitat heterogeneity. Ecology 2009, 90, 3033–3041. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Chang, L.W.; Yang, K.C.; Wang, H.H.; Sun, I.F. Point patterns of tree distribution determined by habitat heterogeneity and dispersal limitation. Oecologia 2011, 165, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Kohn, A.J. Microhabitats, abundance and food of Conus on atoll reefs in the Maldive and Chagos Islands. Ecology 1968, 49, 1046–1062. [Google Scholar] [CrossRef]
- Levins, R. Evolution in Changing Environments: Some Theoretical Explorations (No. 2); Princeton University Press: Princeton, NJ, USA, 1968. [Google Scholar]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2012. [Google Scholar]
- Li, B.; Hao, Z.; Bin, Y.; Zhang, J.; Wang, M. Seed rain dynamics reveals strong dispersal limitation, different reproductive strategies and responses to climate in a temperate forest in northeast China. J. Veg. Sci. 2012, 23, 271–279. [Google Scholar] [CrossRef]
- Wang, X.; Wiegand, T.; Hao, Z.; Li, B.; Ye, J.; Lin, F. Species associations in an old-growth temperate forest in north-eastern China. J. Ecol. 2010, 98, 674–686. [Google Scholar] [CrossRef]
- Read, J. Soil and rainforest composition in Tasmania: Correlations of soil characteristics with canopy composition and growth rates in Nothofagus cunninghamii associations. Aust. J. Bot. 2001, 49, 121–135. [Google Scholar] [CrossRef]
- Lusk, C.H.; Matus, F. Juvenile tree growth rates and species sorting on fine-scale soil fertility gradients in a Chilean temperate rain forest. J. Biogeogr. 2000, 27, 1011–1020. [Google Scholar] [CrossRef]
- Baltzer, J.L.; Thomas, S.C.; Nilus, R.; Burslem, D.F.R. Edaphic specialization in tropical trees: Physiological correlates and responses to reciprocal transplantation. Ecology 2005, 86, 3063–3077. [Google Scholar] [CrossRef]
- Pyke, C.R.; Condit, R.; Aguilar, S.; Lao, S. Floristic composition across a climatic gradient in a Neotropical lowland forest. J. Veg. Sci. 2001, 12, 553–566. [Google Scholar] [CrossRef]
- Paoli, G.D.; Curran, L.M.; Zak, D.R. Soil nutrients and beta diversity in the Bornean Dipterocarpaceae: Evidence for niche partitioning by tropical rain forest trees. J. Ecol. 2006, 94, 157–170. [Google Scholar] [CrossRef]
- Reich, P.B.; Hobbie, S.E.; Lee, T.; Ellsworth, D.S.; West, J.B.; Tilman, D.; Knops, J.M.; Naeem, S.; Trost, J. Nitrogen limitation constrains sustainability of ecosystem response to CO2. Nature 2006, 440, 922–925. [Google Scholar] [CrossRef] [PubMed]
- Thuiller, W. On the importance of edaphic variables to predict plant species distributions-limits and prospects. J. Veg. Sci. 2013, 24, 591–592. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Lelandais, M.; Kunert, K.J. Photooxidative stress in plant. Physiol. Plantarum 1994, 92, 708–719. [Google Scholar] [CrossRef]
- Broadley, M.R.; White, P.J.; Hammond, J.P.; Zelko, I.; Lux, A. Zinc in plants. New Phytol. 2007, 173, 677–702. [Google Scholar] [CrossRef] [PubMed]
- Marschner, H.; Rimmington, G. Mineral nutrition of higher plants. Plant Cell Environ. 1988, 11, 147–148. [Google Scholar]
- Chaoui, A.; Mazhoudi, S.; Ghorbal, M.H.; El Ferjani, E. Cadmium and zinc induction of lipid peroxidation and effects on antioxidant enzyme activities in bean (Phaseolus vulgaris L.). Plant Sci. 1997, 127, 139–147. [Google Scholar] [CrossRef]
- Gallego, S.M.; Benavides, M.P.; Tomaro, M.L. Effect of heavy metal ion excess on sunflower leaves: Evidence for involvement of oxidative stress. Plant Sci. 1996, 121, 125–159. [Google Scholar] [CrossRef]
- Shah, K.; Dubey, R.S. Cadmium elevates level of protein, amino acids and alters activity of proteolytic enzymes in germinating rice seeds. Acta Physiol. Plant. 1998, 20, 189–196. [Google Scholar] [CrossRef]
- Gupta, S.; Srivastava, S.; Saradhi, P.P. Chromium increases photosystem 2 activity in Brassica juncea. J. Biol. Plantarum 2009, 53, 100–104. [Google Scholar] [CrossRef]
- Suseela, M.R.; Sinha, S.; Singh, S.; Saxena, R. Accumulation of chromium and scanning electron microscopic studies in Scirpus lacustris L. treated with metal and tannery effluent. Bull. Environ. Contam. Toxicol. 2002, 68, 540–548. [Google Scholar] [CrossRef]
- Pandey, V.; Dixit, V.; Shyam, R. Antioxidative responses in relation to growth of mustard (Brassica juincea cv. Pusa Jaikisan) plants exposed to hexavalent chromium. Chemosphere 2005, 61, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Shanker, A.K.; Djanaguiraman, M.; Sudhagar, R.; Chandrashekar, C.N.; Pathmanabhan, G. Differential antioxidative response of ascorbate glutathione pathway enzymes and metabolites to chromium speciation stress in green gram (Vigna radiata (L.) R. Wilczek. cv. CO 4) roots. Plant Sci. 2004, 166, 1035–1043. [Google Scholar] [CrossRef]
- Spencer, D.; Possingham, J.V. The effect of Mn deficiency on photophosphorylation and the oxygen—Evolving system in spinach chloroplasts. Biochim. Biophys. 1961, 52, 379–381. [Google Scholar] [CrossRef]
- Gerretsen, F.C. Manganese in relation to photosynthesis. II. Redox potentials of illuminated crude chloroplast suspensions. Plant Soil 1950, 11, 159–193. [Google Scholar] [CrossRef]
- Homann, P.H. Studies on the manganese of chloroplast suspensions. Plant Physiol. 1967, 42, 997–1007. [Google Scholar] [CrossRef] [PubMed]
- Yachandra, V.K.; Sauer, K.; Klein, M.P. Manganese cluster in photosynthesis: Where plants oxidize water to dioxygen. Chem. Rev. 1966, 96, 2927–2950. [Google Scholar] [CrossRef]
- Bottrill, D.E.; Possingham, J.V.; Kriedemann, P.E. The effect of nutrient deficiencies on phosynthesis and respiration in spinach. Plant Soil 1970, 32, 424–438. [Google Scholar] [CrossRef]
- Rowe, N.; Speck, T. Plant growth forms: An ecological and evolutionary perspective. New Phytol. 2005, 166, 61–72. [Google Scholar] [CrossRef] [PubMed]
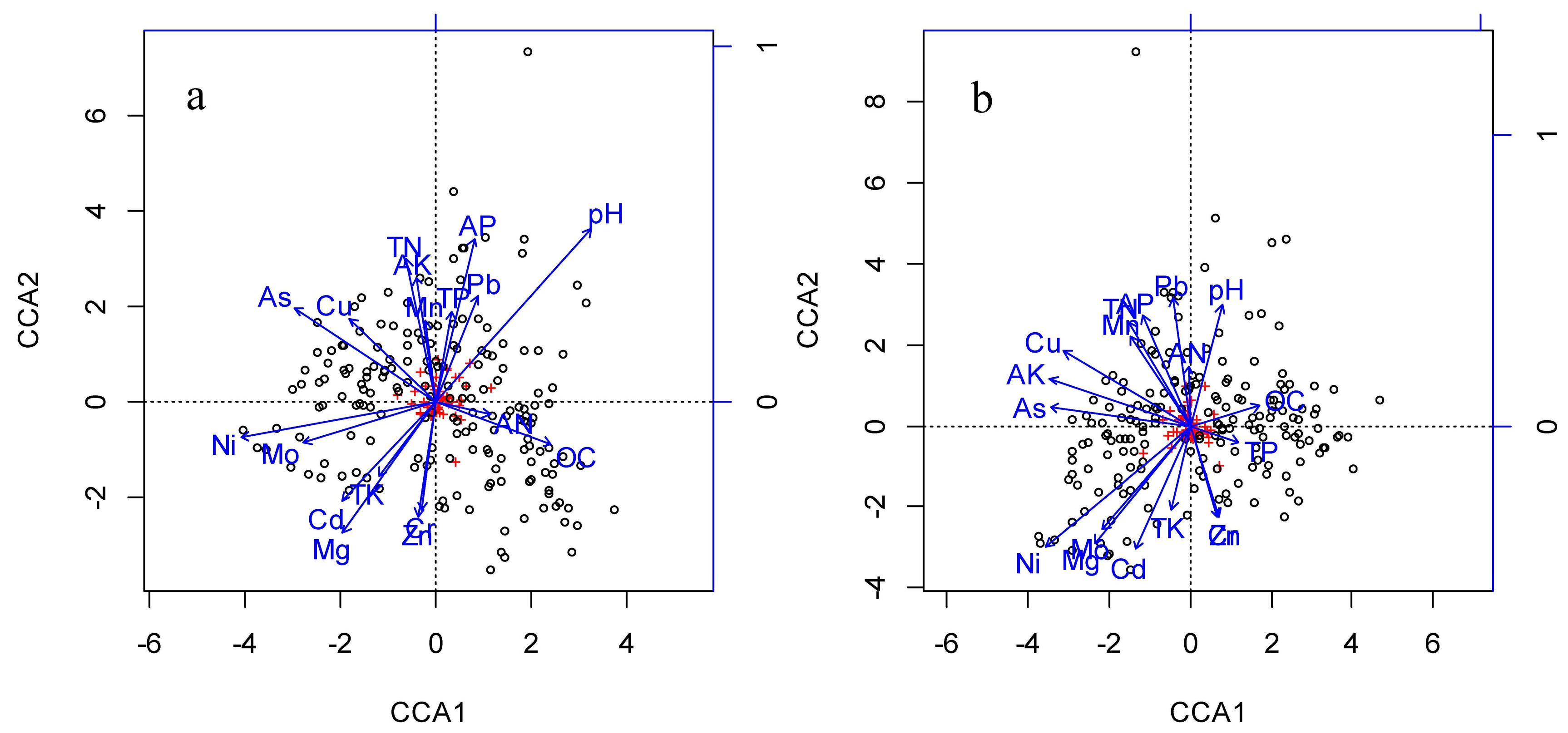
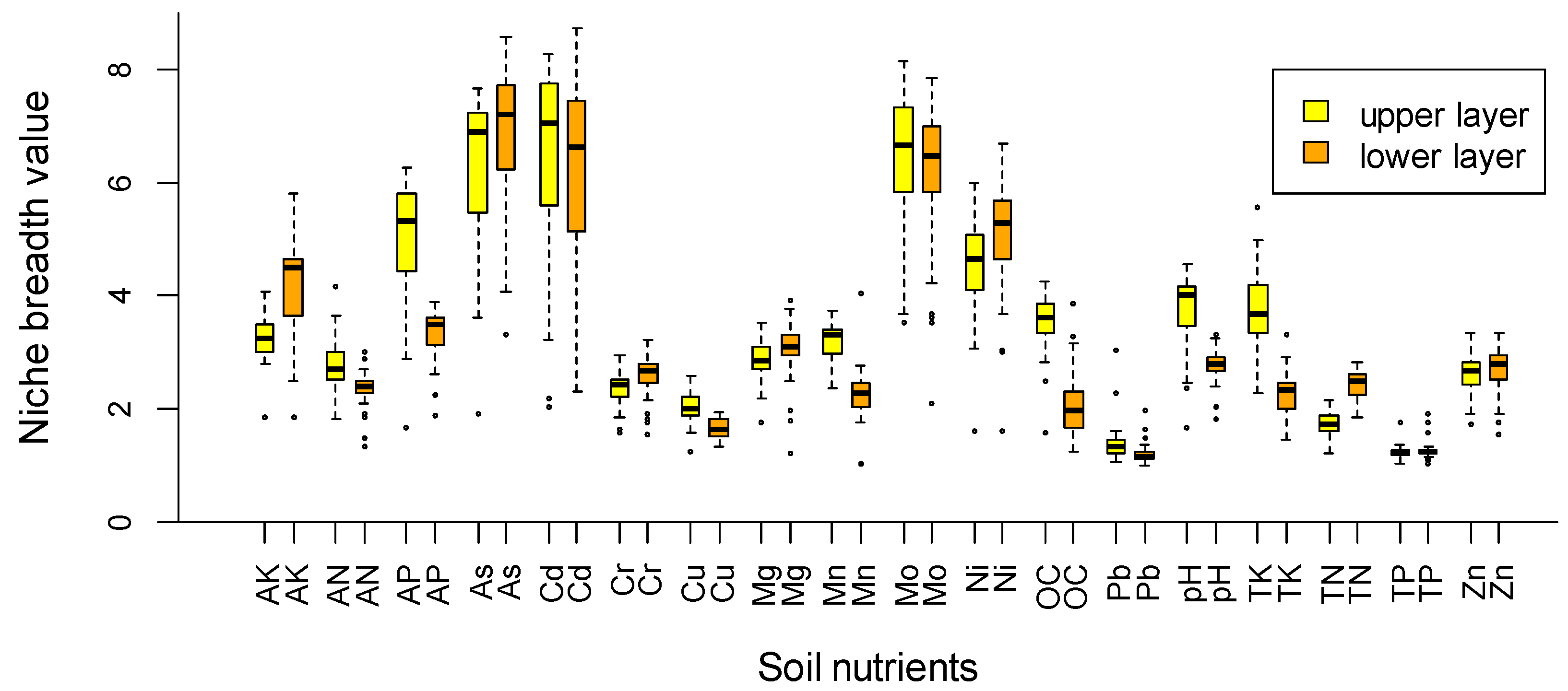
| Soil Variables | Upper Soil Layer | Lower Soil Layer | ||
|---|---|---|---|---|
| CCA1 | CCA2 | CCA1 | CCA2 | |
| available nitrogen | −0.12 | −0.13 | −0.11 | −0.14 |
| total nitrogen | −0.91 *** | 0.07 | −0.91 *** | 0.27 *** |
| available potassium | 0.13 | 0.05 | 0.03 | −0.05 |
| total potassium | −0.02 | −0.41 *** | −0.05 | −0.36 *** |
| available phosphorus | −0.05 | −0.36 *** | −0.22 ** | −0.44 *** |
| total phosphorus | −0.05 | 0.12 | 0.00 | −0.01 |
| organic carbon | 0.08 | −0.51 *** | −0.07 | −0.39 *** |
| Cu | 0.07 | −0.26 *** | 0.08 | 0.10 |
| Ni | 0.15 * | 0.19 * | 0.05 | 0.18 * |
| Cd | −0.16 * | −0.23 ** | −0.19 * | −0.41 *** |
| As | −0.34 *** | 0.02 | −0.30 *** | 0.03 |
| Pb | −0.10 | 0.17 * | −0.07 | 0.15 * |
| Zn | −0.21 ** | −0.31 *** | −0.21 ** | −0.29 *** |
| Mo | 0.05 | −0.26 *** | 0.03 | −0.35 *** |
| Cr | 0.03 | 0.33 *** | 0.05 | 0.28 *** |
| Mn | −0.16 | −0.01 | −0.09 | −0.03 |
| Mg | 0.02 | 0.31 *** | 0.04 | 0.29 *** |
| pH | −0.04 | −0.23 ** | −0.04 | −0.36 *** |
| Soil Variable | Upper Soil Layer | Lower Soil Layer | ||||||
|---|---|---|---|---|---|---|---|---|
| PC1 | PC2 | PC3 | PC4 | PC1 | PC2 | PC3 | PC4 | |
| available nitrogen | −0.387 | −0.318 | −0.445 | −0.122 | 0.111 | |||
| total nitrogen | −0.130 | −0.370 | −0.219 | −0.419 | 0.164 | |||
| available potassium | 0.191 | −0.306 | −0.369 | 0.140 | 0.195 | |||
| total potassium | −0.229 | 0.296 | −0.156 | 0.260 | ||||
| available phosphorus | −0.379 | −0.119 | −0.190 | 0.145 | −0.160 | |||
| total phosphorus | 0.142 | −0.152 | ||||||
| organic carbon | 0.318 | 0.355 | −0.203 | 0.202 | −0.267 | −0.308 | −0.238 | |
| Cu | −0.252 | −0.345 | 0.422 | −0.103 | −0.222 | 0.442 | −0.356 | |
| Ni | −0.184 | 0.248 | 0.315 | −0.259 | 0.153 | −0.264 | ||
| Cd | −0.389 | −0.134 | −0.150 | −0.415 | 0.161 | 0.210 | ||
| As | −0.270 | −0.378 | −0.154 | −0.293 | −0.165 | 0.318 | 0.159 | |
| Pb | 0.113 | −0.232 | 0.493 | −0.103 | 0.393 | −0.494 | ||
| Zn | −0.318 | 0.401 | −0.234 | −0.141 | −0.348 | −0.379 | −0.25 | |
| Mo | −0.421 | −0.216 | −0.430 | 0.127 | 0.317 | |||
| Cr | −0.311 | 0.401 | −0.238 | −0.157 | −0.334 | −0.102 | −0.380 | −0.255 |
| Mn | −0.158 | −0.224 | 0.317 | −0.196 | −0.296 | −0.174 | ||
| Mg | −0.292 | 0.176 | 0.306 | −0.372 | ||||
| pH | 0.196 | −0.318 | −0.177 | 0.149 | −0.384 | |||
| Variance (%) | 21.9 | 11.3 | 10.9 | 8.9 | 20.3 | 14.1 | 10.7 | 9.1 |
| Species (n ≥ 5) | 25 | 25 | 16 | 23 | 24 | 20 | 19 | 17 |
| Species (n ≥ 30) | 24 | 23 | 14 | 20 | 23 | 20 | 17 | 16 |
| Tree Species | HomP | HomT | ||||||
|---|---|---|---|---|---|---|---|---|
| PC1 | PC2 | PC3 | PC4 | PC1 | PC2 | PC3 | PC4 | |
| Betula platyphylla | − | − | − | |||||
| Acer mandshuricum | + | − | − | − | ||||
| Padus racemosa | − | − | − | |||||
| Abies nephrolepis | + | + | + | |||||
| Ulmus davidiana var. japonica | + | − | + | |||||
| Ulmus macrocarpa | + | − | − | |||||
| Betula costata | − | − | ||||||
| Betula dahurica | + | |||||||
| Pinus koraiensis | − | + | − | |||||
| Juglans mandshurica | + | − | − | |||||
| Sorbus pohuashanensis | + | + | ||||||
| Phellodendron amurense | − | − | − | |||||
| Tilia mandschurica | + | + | + | |||||
| Ulmus laciniata | + | − | − | + | − | |||
| Quercus mongolica | + | − | + | |||||
| Carpinus cordata | + | + | − | |||||
| Acer tegmentosum | − | + | + | − | − | + | ||
| Acer mono | + | − | − | |||||
| Abies holophylla | + | − | ||||||
| Fraxinus mandshurica | + | − | + | − | ||||
| Sorbus alnifolia | − | − | ||||||
| Populus koreana | − | |||||||
| Tilia amurensis | + | + | ||||||
| Syringa reticulata var. amurensis | + | − | − | + | + | + | ||
| Acer barbinerve | − | + | + | |||||
| Tilia mandschurica | + | + | ||||||
| Acer ukurunduense | − | + | + | + | + | |||
| Lonicera maackii | + | |||||||
| Euonymus macropterus | + | |||||||
| Eleutherococcus senticosus | + | − | + | − | ||||
| Euonymus pauciflorus | − | + | + | |||||
| Corylus mandshurica | − | − | − | + | ||||
| Rhamnus davurica | − | |||||||
| Significant negative correlation | 10 | 11 | 11 | 9 | 2 | 2 | 2 | 1 |
| Significant positive correlation | 14 | 12 | 3 | 11 | 3 | 2 | 2 | 2 |
| Non-significant correlation | 9 | 10 | 19 | 13 | 28 | 29 | 29 | 30 |
| Plant Species | HomP | HomT | ||||||
|---|---|---|---|---|---|---|---|---|
| PC1 | PC2 | PC3 | PC4 | PC1 | PC2 | PC3 | PC4 | |
| Betula platyphylla | + | − | + | |||||
| Acer mandshuricum | + | − | + | − | + | |||
| Padus racemosa | − | − | ||||||
| Abies nephrolepis | + | − | ||||||
| Ulmus davidiana var. japonica | + | − | + | − | ||||
| Ulmus macrocarpa | + | − | ||||||
| Betula costata | − | + | − | + | ||||
| Betula dahurica | + | |||||||
| Pinus koraiensis | − | + | + | |||||
| Juglans mandshurica | + | |||||||
| Sorbus pohuashanensis | ||||||||
| Phellodendron amurense | − | + | + | |||||
| Tilia mandschurica | + | + | − | − | ||||
| Ulmus laciniata | + | + | + | |||||
| Quercus mongolica | + | |||||||
| Carpinus cordata | + | + | − | − | − | |||
| Acer tegmentosum | − | + | − | + | + | + | ||
| Acer mono | + | |||||||
| Abies holophylla | − | + | − | + | ||||
| Fraxinus mandshurica | + | − | + | + | ||||
| Sorbus alnifolia | + | + | ||||||
| Populus koreana | + | + | + | |||||
| Tilia amurensis | ||||||||
| Syringa reticulata var. amurensis | + | − | + | − | − | |||
| Acer barbinerve | − | + | + | |||||
| Tilia mandschurica | − | − | ||||||
| Acer ukurunduense | − | + | − | |||||
| Lonicera maackii | + | − | ||||||
| Euonymus macropterus | ||||||||
| Eleutherococcus senticosus | ||||||||
| Euonymus pauciflorus | − | + | − | + | − | |||
| Corylus mandshurica | − | − | + | + | ||||
| Rhamnus davurica | − | − | ||||||
| Significant negative correlation | 10 | 10 | 6 | 9 | 1 | 1 | 1 | 1 |
| Significant positive correlation | 13 | 10 | 11 | 7 | 1 | 5 | 2 | 1 |
| Non-significant correlation | 10 | 13 | 16 | 17 | 31 | 27 | 30 | 31 |
| Soil Layer | Canopy Species | Subcanopy Species | Shrub Species |
|---|---|---|---|
| 23 Species | 5 Species | 5 Species | |
| Upper | |||
| Number of species related to PC1 | 5 (3+, 2−) | 0 (0+, 0−) | 0 (0+, 0−) |
| Number of species related to PC2 | 3 (1+, 2−) | 1 (1+, 0−) | 0 (0+, 0−) |
| Number of species related to PC3 | 2 (0+, 2−) | 1 (1+, 0−) | 1 (1+, 0−) |
| Number of species related to PC4 | 3 (3+, 0−) | 1 (1+, 0−) | 0 (0+, 0−) |
| Number of species unrelated to PCs | 14 | 3 | 4 |
| Lower | |||
| Number of species related to PC1 | 2 (1+, 1−) | 0 (0+, 0−) | 0 (0+, 0−) |
| Number of species related to PC2 | 4 (4+, 0−) | 1 (0+, 1−) | 1 (1+, 0−) |
| Number of species related to PC3 | 3 (2+, 1−) | 0 (0+, 0−) | 0 (0+, 0−) |
| Number of species related to PC4 | 1 (1+, 0−) | 0 (0+, 0−) | 1 (0+, 1−) |
| Number of species unrelated to PCs | 15 | 4 | 4 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, W.; Hao, M.; Wang, J.; Zhang, C.; Zhao, X.; Gadow, K.V. Soil Elements Influencing Community Structure in an Old-Growth Forest in Northeastern China. Forests 2016, 7, 159. https://doi.org/10.3390/f7080159
Xu W, Hao M, Wang J, Zhang C, Zhao X, Gadow KV. Soil Elements Influencing Community Structure in an Old-Growth Forest in Northeastern China. Forests. 2016; 7(8):159. https://doi.org/10.3390/f7080159
Chicago/Turabian StyleXu, Wei, Minhui Hao, Juan Wang, Chunyu Zhang, Xiuhai Zhao, and Klaus Von Gadow. 2016. "Soil Elements Influencing Community Structure in an Old-Growth Forest in Northeastern China" Forests 7, no. 8: 159. https://doi.org/10.3390/f7080159
APA StyleXu, W., Hao, M., Wang, J., Zhang, C., Zhao, X., & Gadow, K. V. (2016). Soil Elements Influencing Community Structure in an Old-Growth Forest in Northeastern China. Forests, 7(8), 159. https://doi.org/10.3390/f7080159







