Abstract
The Amazonian rainforest is a unique ecosystem that comprises habitat for thousands of animal species. Over the last decades, the ever-increasing human population has caused forest conversion to agricultural land with concomitant high biodiversity losses, mainly near a number of fast-growing cities in the Peruvian Amazon. In this research, we evaluated insect species richness and diversity in five ecosystems: natural forests, multistrata agroforests, cocoa agroforests, annual cropping monoculture and degraded grasslands. We determined the relationship between land use intensity and insect diversity changes. Collected insects were taxonomically determined to morphospecies and data evaluated using standardized biodiversity indices. The highest species richness and abundance were found in natural forests, followed by agroforestry systems. Conversely, monocultures and degraded grasslands were found to be biodiversity-poor ecosystems. Diversity indices were relatively high for all ecosystems assessed with decreasing values along the disturbance gradient. An increase in land use disturbance causes not only insect diversity decreases but also complete changes in species composition. As agroforests, especially those with cocoa, currently cover many hectares of tropical land and show a species composition similar to natural forest sites, we can consider them as biodiversity reservoirs for some of the rainforest insect species.
1. Introduction
The Amazon rainforest is considered a biodiversity hotspot [1]. Despite all efforts to conserve and protect this critically important and highly endangered ecosystem, the rainforests still continue to steadily disappear. Over the past 50 years, an estimated 32% of the Amazon rainforest has been converted to man-made systems and a further loss of 10%–15% has been projected by 2050 [2]. Conversion of natural ecosystems is a major cause of biodiversity loss and threatens ecosystem functions and livelihoods of indigenous people [3]. Diversified agricultural systems, such as agroforestry systems, are possibly able to conserve rainforest diversity.
During the last three decades, the Peruvian Amazon ecosystem has been profoundly modified. The extraction of natural resources drives much of the economy. Timber logging, petroleum and natural gas drilling, as well as intense fishing, are amongst the prevailing activities. Forests are the most visibly affected [4]. In Peru, there was an average deforestation rate of about 64,500 ha annually in the period 1999–2005 [5]. Other sources [5,6,7,8] further detail deforestation problems in the central part of the Peruvian Amazon, in the area surrounding the city of Pucallpa, where massive deforestation and change of land cover have been observed. Slash-and-burn farming is often blamed as the main cause. Slash-and-burn is a predominant agricultural system in tropical forests in general [9], and the Ucayali region, around Pucallpa city, in particular.
Land use here is quite heterogeneous, with some settlers developing small-scale cattle ranches, while others practice slash-and-burn agriculture (shifting cultivation) with substantial portions of land left fallow [10]. Small-scale farmers often burn down a large part of the forest, simply because they cannot manage the fire. According to Kleinman [11], slash-and-burn farmers transform nutrients stored in forests to soils by slashing, felling, and burning forests. Slash-and-burn is applied mostly in areas where loggers have previously removed the valuable timber trees, whereby the natural forest is then converted into residual or secondary forest. The remaining vegetation is further cut down, burnt and replaced by annual crops, such as rice (Oryza sativa), cassava (Manihot esculenta), plantain (Musa sp.) and/or maize (Zea mays), which are frequently planted for both subsistence and local markets. Farmers also have to combat noxious weeds, which often quickly infest degraded lands and pastures. The most problematic weeds are grasses, such as Imperata brasiliensis that often develop into green deserts that are periodically burnt down, further spiraling the degradation of the land.
A sustainable and suitable solution that slows down and even halts land degradation is agroforestry, which combines cultivation of trees and crops together. Individual components of this system have different ecological and economic functions. According to De Jong [12], there are various agroforestry systems being practiced in the Peruvian Amazon, near the Ucayali River. Mostly systems combining fruit trees with fast-growing trees earmarked for timber production are found. Guaba (Inga sp.) is used by many small-scale farmers as a pilot tree for new agroforestry plots, providing shade and improving the soil condition. It forms a good base for more economically-important species for wood and fruit production. In lowland terrain along the Ucayali River, use of Inga, fast-growing timber species bolaina (Guazuma crinita) and capirona (Calycophyllum spruceanum), in combination with plantains (Musa sp.), carambola (Averrhoa carambola) and various palm species is very common. Plant species composition depends on locality, economic factors and market opportunities for the farmer. In areas with higher rainfall and more fertile soil in the Ucayali region, the planting of cocoa (Theobroma cacao), combined with various fruit and timber trees to provide shade, is gaining popularity.
According to Puri and Panwar [13], agroforestry fields sited close to existing natural forests may benefit from increased biodiversity: for example, animals, birds and insects will be able to invade, and provide important ecosystems services such as pollination, pest control, seed dispersal, etc. If there is no such original primary or secondary forest vegetation in close proximity to an agroforestry plot, then the chance of being invaded by a wild species from outside decreases and takes a long time. Degraded lands and pastures in the Pucallpa zone are expanding, and thus the distances between new agroforestry plots and original vegetation are increasing. When the vegetation has already been destroyed or seriously damaged—for example, by unsustainable practices of overgrazing—agroforestry practices may help to restore some biodiversity, either by sheltering relics of the original flora and fauna and allowing them to proliferate, or by creating a new ecosystem, albeit with a different mix of species from that one which originally occupied the site. The most common colonizing species in agroforestry are birds, small vertebrates and invertebrates.
Invertebrates—mainly insects—are ideal indicators for biodiversity, because they are very closely related to the environment, vegetation cover, overall climate, and are sensitive to habitat disturbances. A fast reproductive cycle and a large number of ecological interactions, as well as easy and inexpensive collection, make them the best bio-indicators [14].
As Godfray et al. [15] state, understanding insect diversity in the humid tropics is one of the major challenges in modern ecology. Some information sources estimate that tropical forest in the Amazon basin forms a habitat for more than 30% of insect species globally; however, insects also play a significant role in agro-ecosystems found in the Amazon that have not yet been thoroughly studied. Crops grown in conventional monoculture systems often suffer from severe pest problems. This is usually attributed to the nature of the cropping system. Monoculture reduces a complex natural plant system to a single-species community. This can lead to decreased insect diversity and can promote rapid population growth of a single, or very few, insect species [16]. Agroforestry induces a different proportion of various insect species, even herbivorous ones, and this is supposed to lower the occurrence of pest species. Andow [17] conducted studies on 287 species of herbivorous insects. He reports a lower density of herbivore pests in polycultural systems than in monocultural cropping in approximately 52% of the species studied. Only 15% had higher densities. In monocultures in the Peruvian Amazon, there are mainly beetles (most commonly Chrysomelids), Hymenopterans represented by the family Vespidae, Homopterans, and others. Species diversity is normally low, but with a great number of individuals for each species. In agroforestry systems, another view on insect diversity, equilibrium and conservation could be offered.
The general objective of our study was to quantify the impact of landuse changes, with respect to diminishing biodiversity, along a disturbance gradient from a tropical forest to a degraded site. Specifically, we set out to evaluate the impact of agroforestry and agricultural practices on insect species composition and diversity in one of the most deforested regions of the Peruvian Amazon. We expected a decrease in insect diversity along the disturbance gradient.
2. Experimental Section
2.1. Study Sites
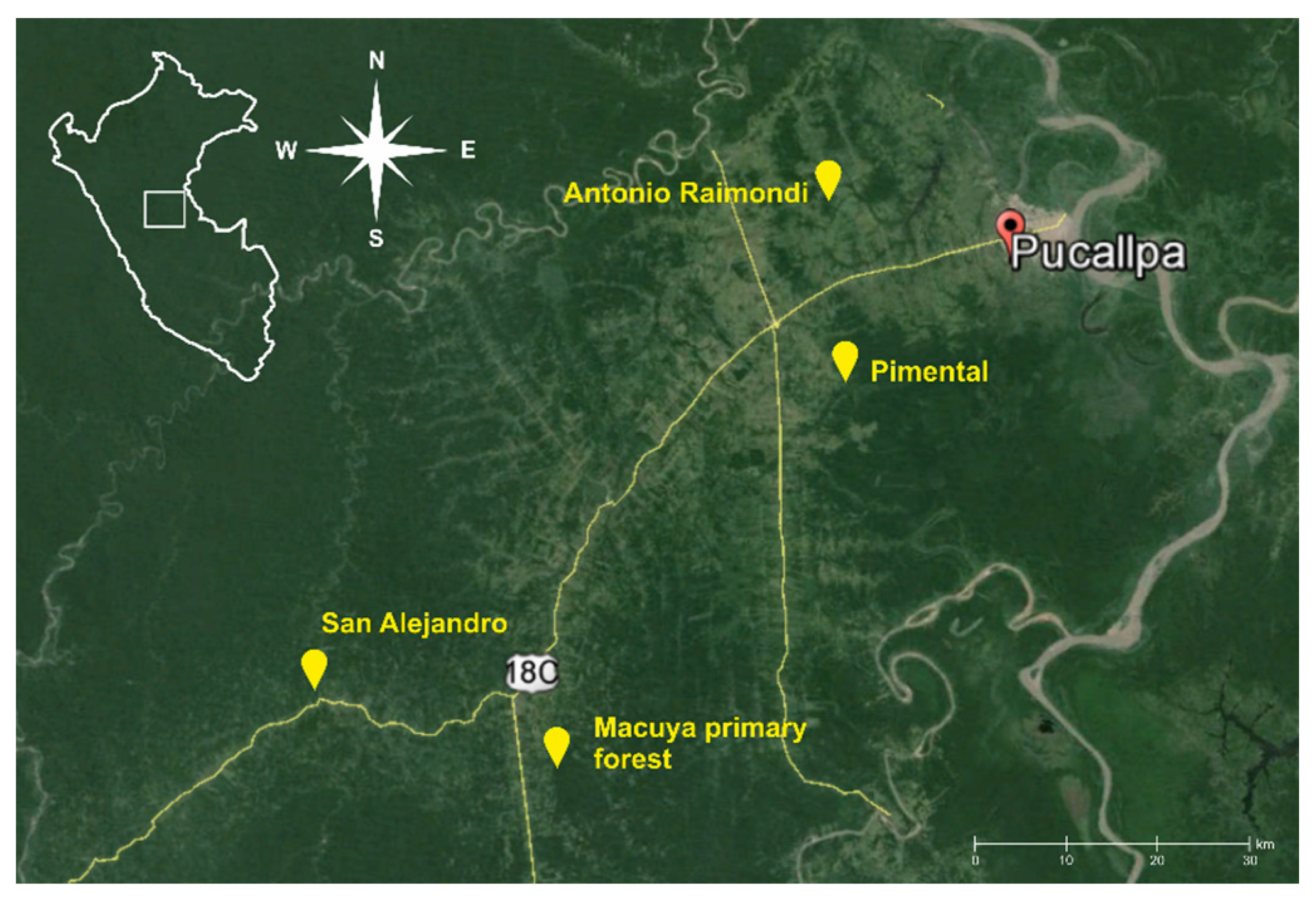
Several villages in close proximity to Pucallpa city, on the main road to Lima, were chosen as areas, with suitable study sites. The environs of Antonio Raimondi, Pimental and San Alejandro villages (Figure 1) were chosen, and we sampled following landuse systems: multistrata agroforests, cocoa agroforests, cassava monocultures and weedy grasslands on degraded soils (Table 1). As a sample of tropical forest vegetation, we chose the site of Macuya, a natural primary forest of the National University of Ucayali, located close to the Alexander von Humboldt village. The forest was selectively logged more than 20 years ago, displaying the last remnants of human activity. For the purpose of this study, we classified it as a partly logged, yet well-preserved natural forest.
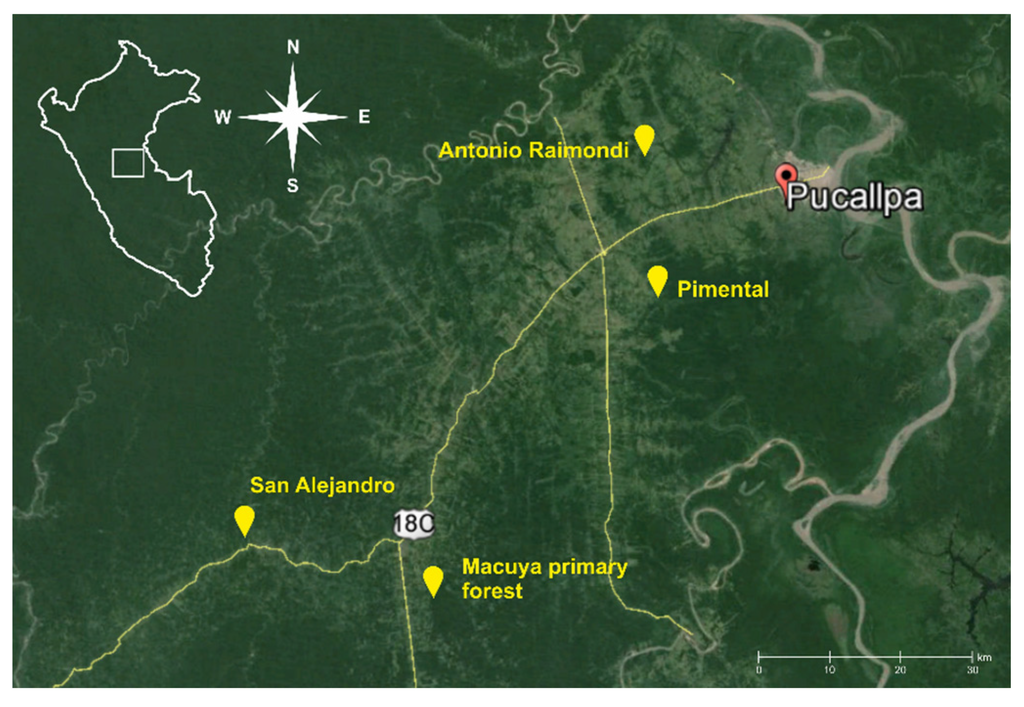
Figure 1.
Location of selected study sites near Pucallpa city in the Peruvian Amazon.

Table 1.
Study sites characteristics, Peruvian Amazon.
San Alejandro is a small town, 118 km from Pucallpa (and situated along the Carretera Federico Basadre). The rural residents are dedicated to agriculture (annual crops, livestock, forestry and others). In the last decade, there has been an expansion of cocoa cultivation. For data collection, the village of Porvenir, 2 km from San Alejandro, was chosen. Terrain is diverse, dominated by steep hills with a mosaic of secondary forest vegetation and farm fields. The majority of cropping plots could be classified as agroforestry systems. Sampling was undertaken within two agroforestry plots with shaded cocoa trees.
Pimental is a rural village settlement 35 km from Pucallpa, and 6 km off the main road. Land use systems are based on shifting annual crop cultivation, pepper (Piper nigrum) plantations and various fruit tree agroforests. Data were collected within plots with multistrata agroforestry, cassava monoculture and degraded sites with weed vegetation.
Antonio Raymondi village is located 19 km from Pucallpa and 7 km from the main road to Lima, inhabited mainly by small-scale slash-and-burn farmers who cut and burned down large forest areas around the village; they continue to do so to establish new plots for cassava and other staple crops. Some of them apply a multistrata agroforestry system. The village surroundings are degraded lands, covered by the weed species (mainly Imperata brasiliensis). Data collection was carried out in two agroforestry plots, cassava monoculture and degraded, weed-infested land. Some of those grasslands had been burned up to three times a year.
2.2. Data Collection
Insects were collected from March to August 2009, and February to November 2010, covering both wet and dry seasons. Sampling was conducted on 12 plots (25 × 25 m), always between 6:30 a.m. and 10:30 a.m., using common entomologic methods. We established four sampling plots in each village. Because of the large distance between villages, each plot was sampled six times, with monthly repetition. Samples in the Macuya forest were taken only four times per sampling period because of logistic problems. On each plot, one Malaise trap, and nine pitfall traps in 5 × 5 m2 grids were installed; samples were collected after 24 h. As a fix solution for trapped insects, we used a solution of water, salt and soap-like detergent. We also used a sweeping net with a diameter of 40 cm in zigzag transects with a total length of 80 m (one transect per plot).
Each sample was first purified (small soil and plant remnants were taken out), separated into small plastic Ependorf’s test tubes or larger laboratory fix-boxes marked by the locality label, and fixed in 70% ethanol.
Samples were sorted into basic taxonomic orders, and determined into specific morphospecies (i.e., distinguished by its morphology from other species). Then the number of species and their relative abundance (number of individuals) were calculated. For biodiversity evaluation, orders of Coleoptera, Hymenoptera, Orthoptera, Homoptera and Dictyoptera (suborder Blattodea) were chosen. Other orders, such as Diptera, etc., were excluded: numerous samples were poorly identifiable under our conditions. These are stored in the depository of the Czech University of Life Sciences, Prague, and are being prepared for further investigation. All the samples were classified with the assistance of professional entomologists.
2.3. Data Evaluation
The goal was to evaluate insect community composition in each ecosystem. Firstly, we documented the insect species composition and their relative abundance in each habitat (landuse system). To account for differences in sample areas, the rarefaction method of Gotelli and Colwell [18] allowed us to construct the species accumulation curves for each habitat based on the number of sampled plots. To show how common or rare a species is in relation to other species in a given location or community, we constructed relative abundance model curves for each locality as a function of abundance and number of species per assessed ecosystem. The Jackknife formula was used to estimate species richness [19] and species heterogeneity was estimated using Shannon-Weiner (H base e logs) and Simpson (1-D) diversity indices [19,20,21] at the level of each sample plot and habitat. For similarity description, Sorensen’s index [22] was used. The formula comprises shared species between two habitats and allows us to address the question of how similar the two assemblages are.
To assess statistical differences among the above-mentioned indices and variables of the five land use systems, both parametrical One-way analysis of variance (ANOVA using Tukey’s SD test with Bonferroni correction) and non-parametrical Kruskal-Wallis (KW-ANOVA with Bonferroni correction) test were performed for the whole dataset using STATISTICA 9.0 software (StatSoft), as the data show moderate violation of assumptions (normality and heteroscedasticity). However, both approaches give the same results: there are statistically significant differences at least in between some of the groups (habitats). As parametric ANOVA is relatively robust against disruption of its usage assumptions, for post-hoc test (multiple comparisons) the Bonferroni test was chosen.
3. Results
3.1. Species Abundance and Composition
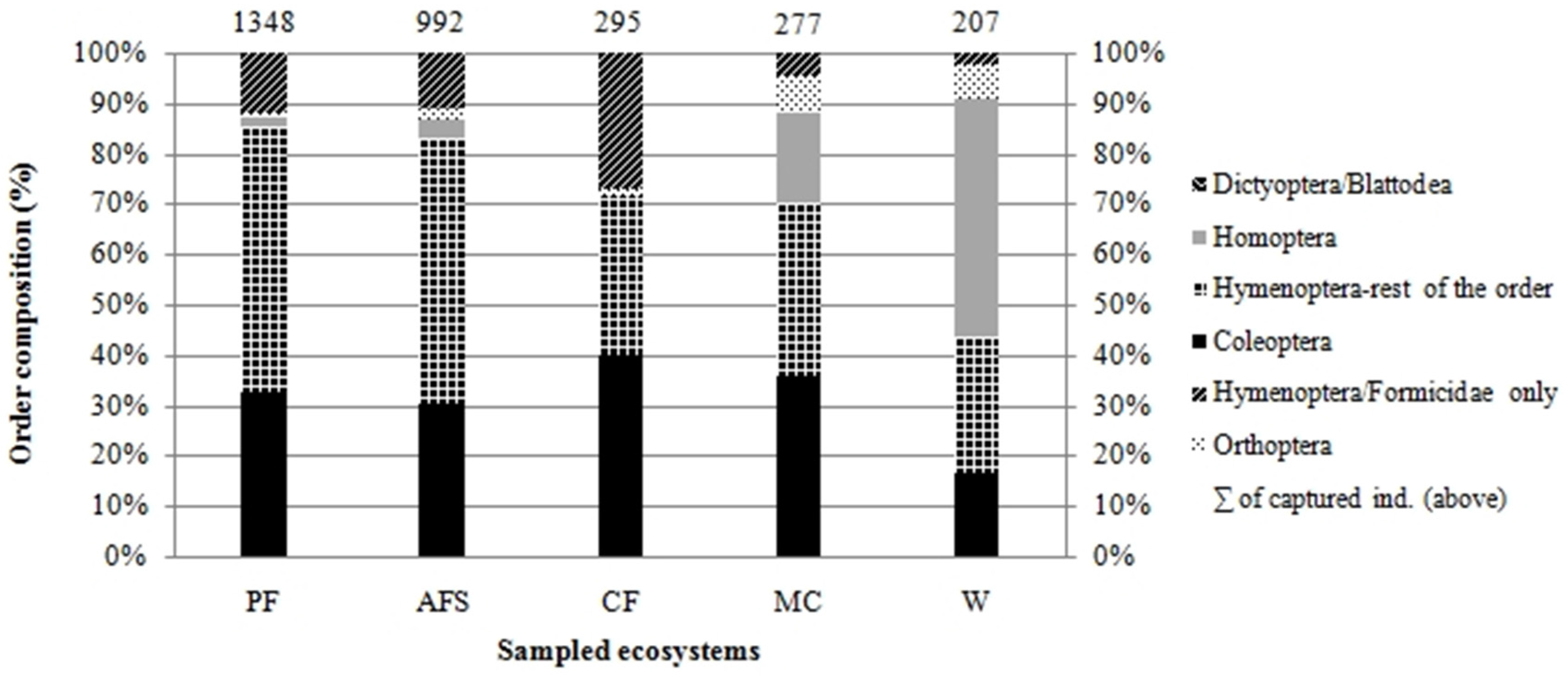
We collected insects from a total of 488 traps, from which we selected five orders of insects—Coleoptera, Hymenoptera, Orthoptera, Homoptera and Dictyoptera (suborder Blattodea). Altogether, 64 insect families, represented by 1164 morphospecies, were determined to the species level. In total, we analyzed 3119 individuals. Highest abundance was found in the natural forest, with its 1348 individuals in 758 morphospecies, followed by multistrata agroforests, represented by 992 individuals in 540 morphospecies. Results show a numerical similarity in abundance of cocoa agroforests to monocultures and weed vegetation; however, these localities vary in species composition (Figure 2).
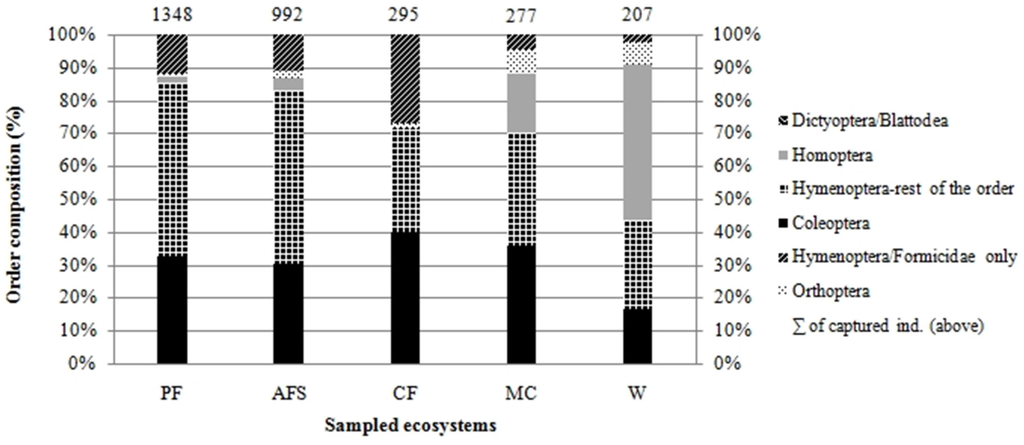
Figure 2.
Order-based relative insect composition of five habitats assessed in the Peruvian Amazon (MSF, natural forests; AFS, multistrata agroforests; CF, cocoa agroforests; MC, annual monoculture crops; W, weedy grassland).
Abundance models for each habitat indicate (Supplementary Materials S1) that in natural forest and agroforest habitats, a large number of species are represented by one or two individuals and only a few species are represented by a high number of individuals: this is an indicator of a high species diversity. This pattern occurs in natural non-disturbed ecosystems, or in ecosystems with only low disturbance. On the contrary, a low number of species and their higher abundance (low number of species with only several individuals), reflecting low diversity, can be observed in monoculture habitats and degraded lands infested by weedy grass.
In natural forests, the taxon with the highest number of individuals (relative abundance) was the ants (14% of species samples). The species with the most individuals trapped, was the stingless bee Trigona pallida with 41 individuals, and one morphospecies of Chrysomelidae beetles with 40 individuals (family Chrysomelidae forms 9% of samples of Macuya forest). In multistrata agroforests, the most abundant morphospecies were all of the Polistinae family (47 individuals), followed by a morphospecies of Cicadelinae (41) and Oxybellus sp., family Crabronidae (38 individuals). Overall, the dominant family was Vespidae, with a total number of 147 individuals. Cocoa agroforests were dominated by one morphospecies of the Dolichoderinae subfamily (Formicidae) with 59 individuals (20% of cocoa agroforest samples). The rest of the species in those localities were represented by an average of two individuals per morphospecies (Figure 3). In annual cropping, the most abundant was a morphospecies of digger wasp Liris (Crabronidae), with 30 individuals. Other species are represented by an average of three individuals. Degraded lands infested by weeds are dominated by a morphospecies of genus Liris (15 individuals) and a species of Agalinae subfamily (12 individuals). Annual crops and grasslands are characterized by low species richness and abundance (Figure 3). The most abundant species of all the study sites was Camponatus sp., an ant present in low numbers in all assessed forest and agroforest ecosystems, yet completely absent in annual cropping and degraded lands. The latter sites are dominated by a morphospecies of digger wasp Liris.
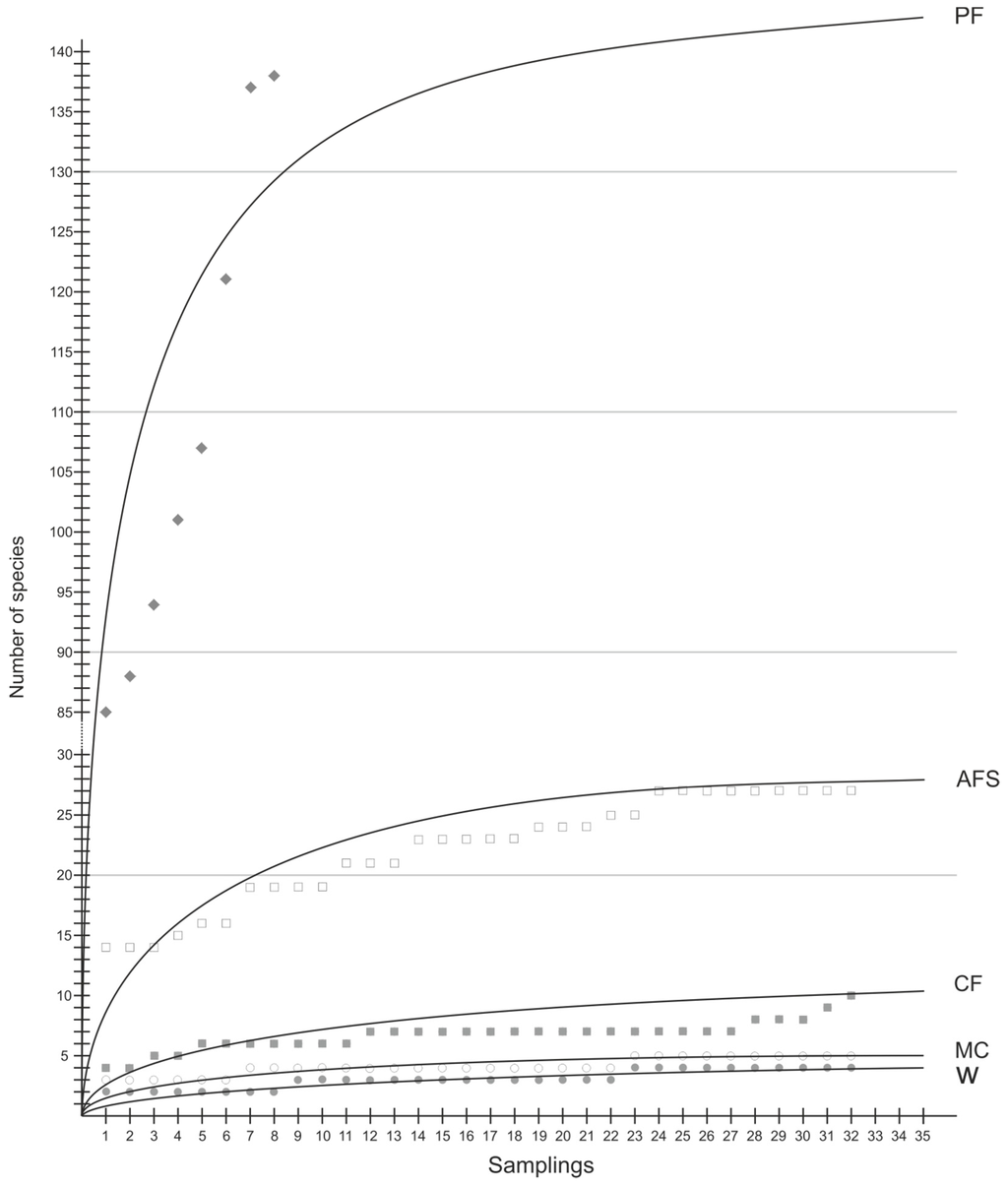
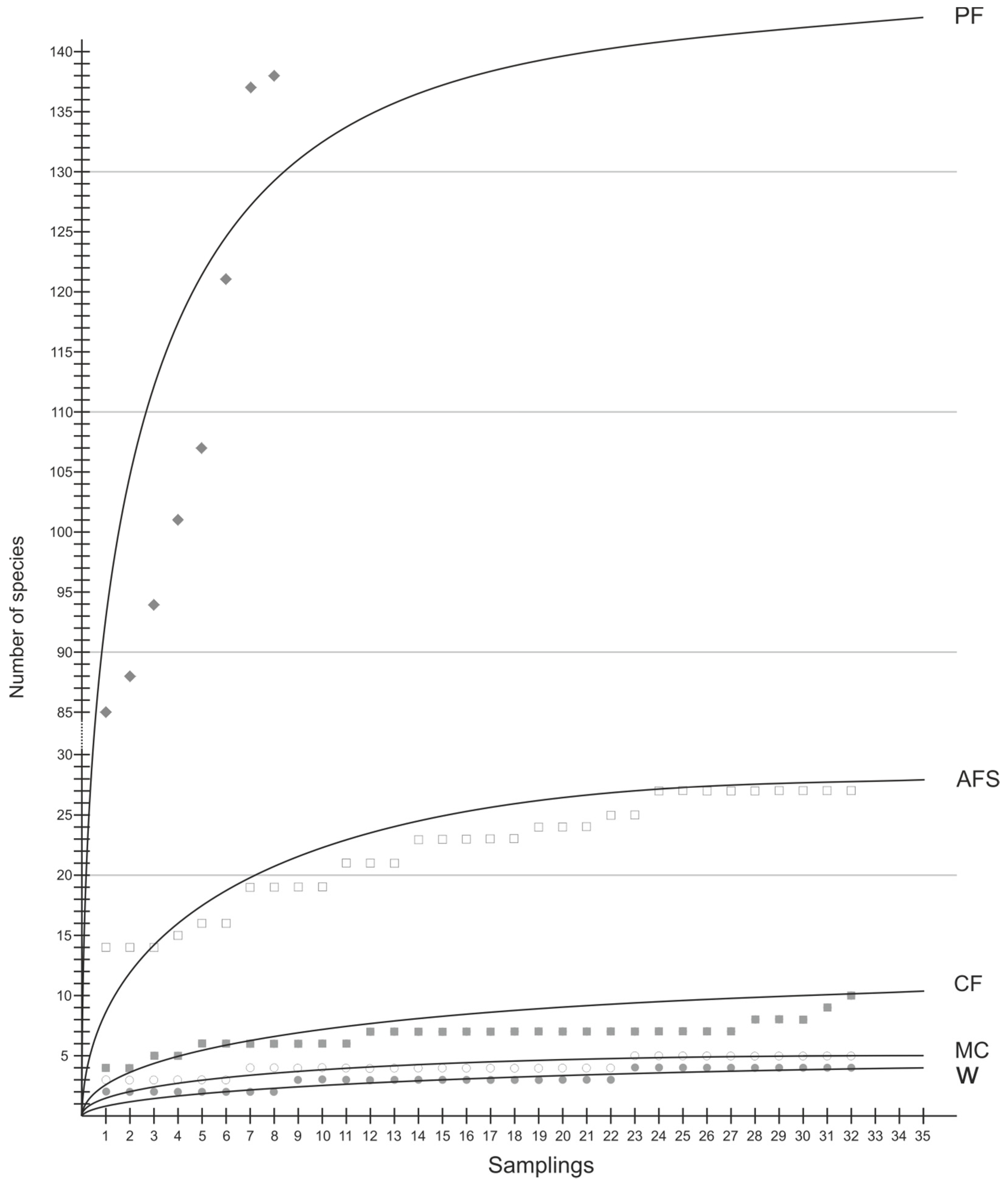
Figure 3.
Insect species richness accumulation curves based on number of samples (PF, natural forests; AFS, multistrata agroforests; CF, cocoa agroforests; MC, annual monoculture crops; W, weedy grassland).
3.2. Species Richness and Diversity
To standardize different sample sizes from each of the five habitats, we compared them according to their species accumulation curves (Figure 3). The curve for natural forests based on the number of samples is far from asymptotic, indicating that the area sampled was too small to estimate the total number of species in this land use type. However, with an increased sampling size, insect species richness is likely to be significantly higher in natural forest compared to that of other habitats. It is followed by both agroforestry systems (higher in multistrata agroforests than in cocoa agroforests) and species richness is low but comparable between annual cropping, and weedy grasslands.
To compare relative species richness and diversity among the five habitats (landuse systems), we evaluated the samples in two ways: (i) in total per habitat; and (ii) statistical differences of means per sample and plot (Table 2). In total, all the indices decrease along the human disturbance gradient from undisturbed natural forest to severally disturbed degraded sites covered with weed vegetation (Table 2—total). A comparison of agricultural land use systems indicated, the species richness in cocoa agroforests was more than two times lower than in multistrata agroforests. In annual crop monoculture the species richness was up to six times lower than in multistrata agroforestry and two times lower than in cocoa agroforests.

Table 2.
Insect diversity indices of five habitats in the Peruvian Amazon (sp., species; ind., individuals; sam., sample; Singletons, species with only one individual in samples; Unique sp., species that occur in only one sample, SD, standard deviation). Letters a–d following biodiversity indices values indicate groups significantly differing one from another using Bonferroni post-hoc tests (α = 0.05). For detailed statistical results and post-hoc p-values, see Supplementary Material S1.
Statistical evaluation of means per sample and plot indicated significant differences among habitats (Table 2—Means per sample). The results corresponded with the graphic interpretation of data (boxplots and detailed statistical evaluation are presented in Supplementary Materials S2). Both observed species richness and Jackknife estimate of species richness, were highest in natural forest and the values significantly decreased with increasing intensity of land use and/or conversion from multistrata agroforestry towards degraded lands. However, we did not found significant differences among cocoa agroforests, annual cropping and degraded sites with weed vegetation. For species diversity evaluation, the Shannon-Weiner and Simpson’s diversity indices were compared. These results also confirm significant decrease in diversity along the disturbance gradient (from natural forests till degraded sites). Shannon-Weiner diversity index reached highest values in natural forests followed by both agroforestry systems (with no significant differences between them) and lowest values (not significantly different) were found in annual cropping and weedy grasslands. Using Simpson’s diversity index was highest in natural forests but not significantly different of multistrata agroforests, followed by cocoa agroforests, annual cropping and weedy grasslands (with significant differences among them). The differences between both indices could be explain by the fact that Simpson’s index is more weighted towards dominant species compared to Shannon index and those species are more common in disturbed habitats.
3.3. Similarity among the Habitats
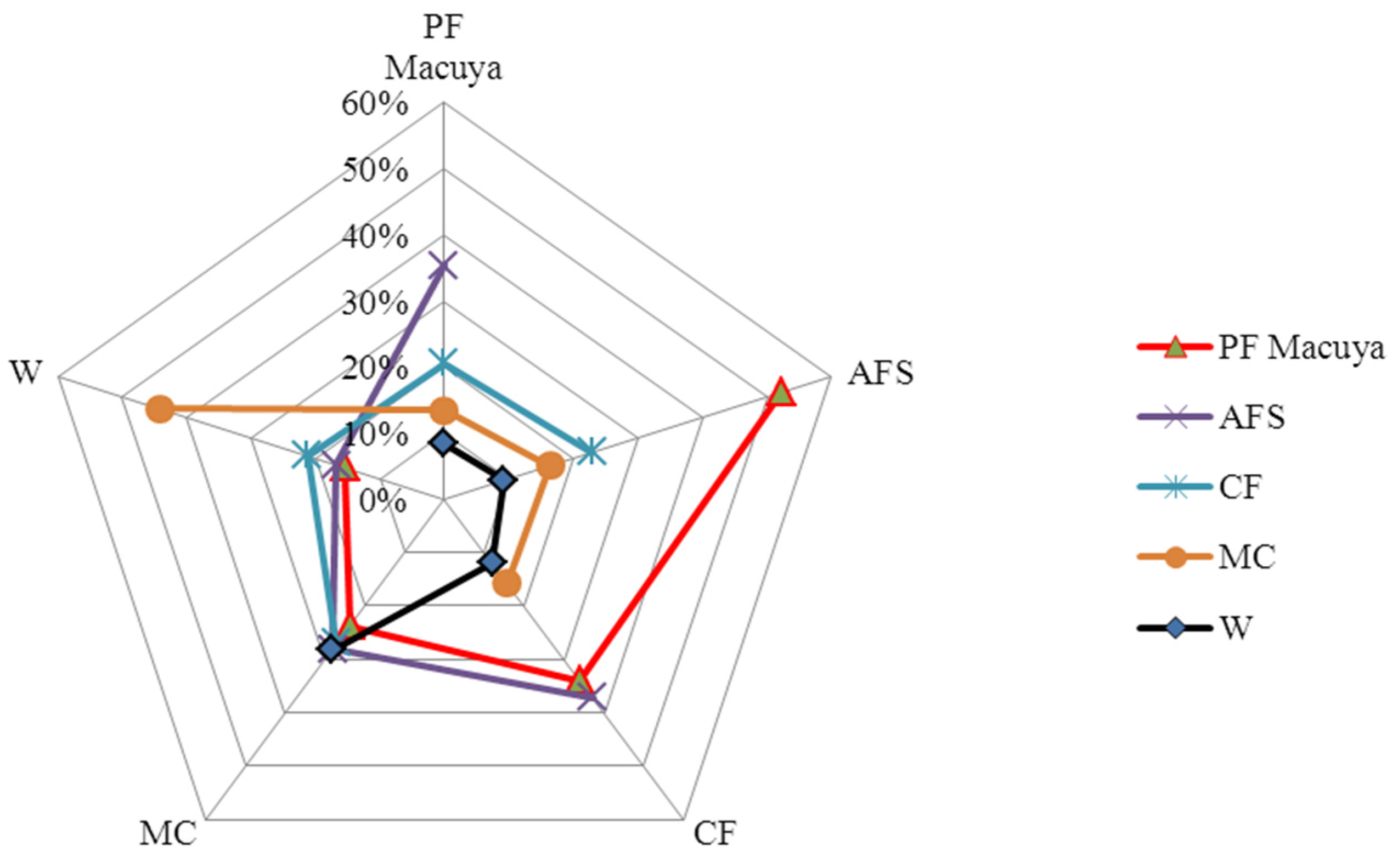
According to Sørensen’s index (Table 3), the multistrata agroforestry is highly similar to natural forest (52%), whereby both types share 158 species. Cocoa agroforest shared 34% of species with natural forest and 37% with multistrata agroforests. Monoculture and weedy grasslands shared 30 species and were highly similar (44%). We found only three species shared by all habitats.

Table 3.
Similarity between habitats expressed as Sørensen index (in %) and number of shared morphospecies (no. of species) in the Peruvian Amazon. (PF: natural forests; AFS: multistrata agroforests; CF: cocoa agroforests; MC: annual monoculture crops; W: weedy grasslands).
The similarity relationship is visible on the radar chart graph (spider net—Figure 4), where the majority of ecosystems have their maximum values on the right hand side of the graph (Figure 4), whereas curves of monocultures and weedy grasslands clustered in the left hand side. A “diagonal” difference in species composition can be observed.
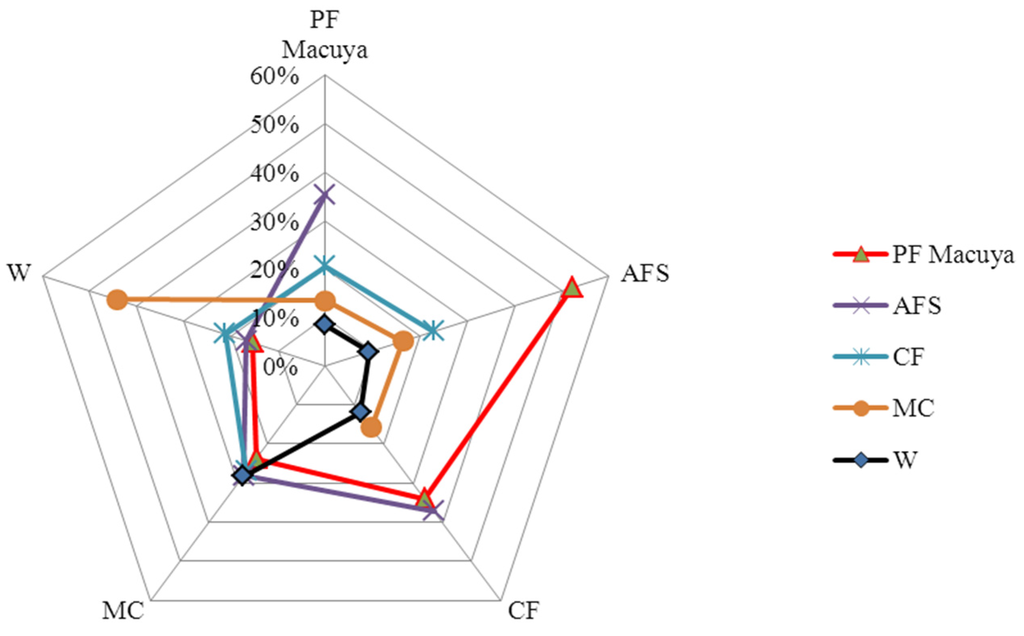
Figure 4.
A radar chart (spider net) graph of habitat similarities based on Sørensen index. (PF: natural forests; AFS: multistrata agroforests; CF: cocoa agroforests; MC: annual monoculture cropping; W: weedy grasslands).
4. Discussion
According to our results, the values of species richness and diversity decreased consistently along the disturbance gradient from natural forest towards agroforestry, monoculture crops and degraded lands. This can be explained by the structure of the vegetation cover that is most complex in natural forest. Multistrata agroforests, as the name implies, are characterized by a high diversity of trees, shrubs and annual plants in the undergrowth. They form more complex and suitable environment for various insect species than cocoa agroforests or mono-cropping fields. Cocoa agroforests are mainly cocoa trees shaded by fast-growing timber trees with almost no undergrowth herbaceous plants in them.
Our results are different from those obtained by Bos et al. [23], who observed that, compared to other forms of agricultural land use such as annual mono-crops and oil palm plantations, the insect species richness of cocoa agroforests was high and comparable with that of natural forests. Other authors also found cocoa agroforests to be very rich and good biodiversity reservoirs. For example, Rice and Greenberg [24] noted that cocoa plantations can resemble forests in terms of tree cover. Low insect species diversity in this type of vegetation in our study could be caused by specific microclimatic conditions within cocoa plantations, where the undergrowth was nearly missing and herbaceous plant composition was very simple. According to our results, cocoa agroforests are not as high in insect species richness and diversity as multistrata agroforests; however, as cocoa agroforestry systems cover eight million hectares of land worldwide, they have received increased attention for their potential to harbor some of the natural forest biodiversity [25]. According to Bos et al. [23], several management practices have an impact on insect richness directly by affecting resources, or indirectly, through creating microclimatic changes and changes in species assemblages. Although there were various factors potentially influencing our results such as proximity of natural water sources in assessed localities and distance from natural forests, we can clearly see that the land use intensity, and ecosystem modification, which is highest in the annual cropping monoculture, highly affects the insect biodiversity and causes visible biodiversity losses and changes in species composition.
Our results on habitat similarity and species composition show a high similarity between agroforestry systems (multistrata and cocoa agroforests) and natural forest. This is supported by Bhagwat [26] who confirmed that heterogeneous agroforestry systems, in which tall trees are maintained and planted for shade (agroforestry systems), form a good refuge for tropical biodiversity. Our results are also consistent with those from other studies [23,27], where the authors claim that, although cocoa agroforests can easily be as rich in insect species as nearby natural forest sites, species assemblages have been found to differ between natural forests and cocoa agroforests, and between differently shaded cocoa agroforests [23].
According to various authors [23,27,28], ants are a rich and ecologically important group of insects that are directly affected by changes in management, for example, by altered resource and nest site availability, but also indirectly, through a complex interplay between microclimate and subsequent changes in species composition. Delabie et al. [27] found that a Brazilian cocoa agroforest with a species-poor stand of shade trees harbored a high proportion of forest ants, as compared to other tropical agroecosystems and urban habitats. Similarly, Bos et al. [23] found that about half the ant species in Indonesian cocoa agroforests also occurred in nearby forest sites, but species richness decreased with decreasing shade cover [23].
5. Conclusions
Our study presents a detailed analysis of insect biodiversity in several landuse systems in the Ucayali region, Peruvian Amazon. For insect biodiversity, we assessed the natural forest as the most species-rich and diverse, followed by various agroforestry systems. Our results suggest that insect biodiversity may well relate to vegetation structure and complexity. The habitats with least structure, such as annual monocropping and degraded grasslands, showed poor species richness and diversity.
We also found that agroforestry systems possess species composition more similar to natural forest, compared to annual cropping or degraded grasslands. Although species diversity in the latter systems was still relatively high, species composition differed significantly from that in natural forests. These altered and disturbed habitats contained more phytophagous species and a low proportion of predatory species. Higher intensity of land use causes not only a biodiversity decrease, but also a complete change of species composition. As cocoa agroforests nowadays cover large areas of tropical land, there is general recognition that they are biodiversity reservoirs for rainforest species.
Natural forests, as well as all natural vegetation types in the tropics, are worthy of protecting because of their high species richness and ecological potential. The governments and citizens of all tropical countries (not only in Amazon) are responsible for developing agricultural practices that prevent complete ecosystem conversion and loss of the unique biodiversity. We believe that various agroforestry systems, such as multistrata and cocoa agroforests, are able to conserve some of the original biodiversity of original forests, and can form a suitable reservoir for some insect species that occur in primary tropical forests.
Supplementary Files
Supplementary File 1Acknowledgments
We would like to thank the professional entomologists Jakub Straka, Štěpán Kubík, Miroslav Barták, Petr Šípek, Petr Janšta, Jakub Straka, and Martin Fikáček as well as the National Museum in Prague. This research was made possible with the financial and scientific support of the Czech Development Cooperation Program (23/Mze/B/07-10), CIGA (Project No. 20145005) and IGA (Project No. 20165016).
Author Contributions
Jitka Perry, was in charge of the whole study (methodology, data collection and evaluation, and writing the manuscript); Bohdan Lojka, the corresponding author, supervised the entire research project, writing of the article, and reviewed and commented on successive drafts of the paper; Lourdes G. Quinones Ruiz supervised data collection and reviewed and commented on successive drafts of the paper; Patrick Van Damme participated in the article writing and reviewed and commented on successive drafts of the paper; Jakub Houška has worked on statistical evaluation of the results and helped with the final editing of the manuscript; and Eloy Fernandez Cusimamani performed data analysis and the interpretation of the results.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; da Fonesca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Millenium Ecosystem Assessment. Ecosystems and Human Well-Being: Biodiversity Synthesis; World Resources Institute: Washington, DC, USA, 2005. [Google Scholar]
- Hoekstra, J.M.; Boucher, T.M.; Ricketts, T.H.; Roberts, C. Confronting a biome crisis: Global disparities of habitat loss and protection. Ecol. Lett. 2005, 8, 23–29. [Google Scholar] [CrossRef]
- Clay, J.W.; Clement, C.R. Selected Species and Strategies to Enhance Income Generation from Amazonian Forests. FAO: Misc/93/6 Working Paper: Food and Agriculture Organization of the United Nations. Available online: http://www.fao.org/3/a-v0784e.pdf (accessed on 28 August 2015).
- Oliviera, P.J.C.; Asner, G.P.; Knapp, D.E.; Almeyda, A.; Galván-Gildemeister, R.; Keene, S.; Raybin, R.F.; Smith, R.C. Land use Allocation Protects the Peruvian Amazon. Science 2007, 317, 1233–1237. [Google Scholar] [CrossRef] [PubMed]
- Hyman, G.; Fujisaka, S. Final Report Desakota, Part II G4. Case Study: Central Peruvian Amazon—A test Case for Desakota Development in the Amazon. International Centre for Tropical Agriculture (CIAT); p. 9. Available online: http://www.dfid.gov.uk/r4d/Output/179310/Default.aspx (accessed on 13 September 2008).
- Hyman, G.; Puig, J.; Bolanos, S. Multi-source remote sensing and GIS for exploring deforestation patterns and processes in the Central Peruvian Amazon. In Proceedings of the 29th International Symposium on Remote Sensing of Environment, Buenos Aires, Argentina, 8–12 April 2002.
- Swallow, B.; van Noordwijk, M.; Dewi, S.; Murdiyarso, D.; White, D.; Gockowski, J.; Hyman, G.; Budidarsono, S.; Robiglio, V.; Meadu, V.; et al. Opportunities for Avoided Deforestation with Sustainable Benefits. Available online: http://www.asb.cgiar.org/PDFwebdocs/Report-on-Opportunitiesfor-Avoided-Deforestation-Sustainable-Benefits-web-low.pdf (accessed on 6 October 2007).
- Takasaki, Y. Economic Model of Shifting Cultivation: A Review; Tsukuba Economics Working Papers No. 2011-006; University of Tsukuba: Tsukuba, Japan; p. 18.
- Fujisaka, S.; White, D. Pasture or permanent crops after slash-and-burn cultivation? Land-use choice in three Amazon colonies. Agrofor. Syst. 1998, 42, 45–59. [Google Scholar] [CrossRef]
- Kleinman, P.J.A.; Pimentel, D.; Bryant, R.B. The ecological sustainability of slash-and-burn agriculture. Agric. Ecosyst. Environ. 1995, 52, 235–249. [Google Scholar] [CrossRef]
- De Jong, W.; Freitas, L.; Baluarte, J.; van de Kop, P.; Salazar, A.; Inga, E.; Melendez, W.; Germaná, C. Secondary forest dynamics in the Amazon floodplain in Peru. For. Ecol. Manag. 2001, 150, 135–146. [Google Scholar] [CrossRef]
- Puri, S.; Panwar, P. Agroforestry: System and Practices; New India Publishing Agency: New Delhi, India, 2007; p. 657. [Google Scholar]
- Brown, K.S., Jr.; Hutchings, R.W. Chapter 7. Disturbance, Fragmentation, and the Dynamics of Diversity in Amazonian Forest Butterflies. In Tropical Forest Remnants—Ecology, Management and Conservation of Fragmented Communities; Laurance, W.F., Bierregaard, R.O., Jr., Eds.; The University of Chicago Press: Chicago, UK, 1997; p. 616. [Google Scholar]
- Godfray, H.C.J.; Lewis, O.T.; Memmott, J. Studying insect diversity in the tropics. Philos. Trans. R. Soc. Lond. 1999, 354, 1811–1824. [Google Scholar] [CrossRef] [PubMed]
- Stamps, W.; Linit, M. Plant diversity and arthropod communities: Implications for temperate agroforestry. Agrofor. Syst. 1997, 39, 73–89. [Google Scholar] [CrossRef]
- Andow, D.A. Vegetational diversity and arthropod population response. Annu. Rev. Entomol. 1991, 36, 561–586. [Google Scholar] [CrossRef]
- Gotelli, N.J.; Colwell, R.K. Quantifying biodiversity: Procedures and pitfalls in the measurement and comparison of species richness. Ecol. Lett. 2001, 4, 379–391. [Google Scholar] [CrossRef]
- Krebs, C.J. Ecological Methodology, 2nd ed.; Addison-Wesley Educational Publishers, Inc.: Boston, MA, USA, 1999. [Google Scholar]
- Clarke, K.R.; Warwick, R.M. Changes in Marine Communities: An Approach to Statistical Analysis and Interpretation, 3rd ed.; PRIMER-E: Plymouth, UK, 2001; p. 260. [Google Scholar]
- Khan, A. Methodology for Assessing Biodiversity. Centre of Advanced Study in Marine Biology, Annamalai University. Available online: http://www.slideshare.net/MMASSY/methodology-forassessmentbiodiversity (accessed on 28 August 2015).
- Sörensen, T.A. A method of establishing groups of equal amplitude in plant sociology based on similarity of species content, and its aplication to analyses of the vegetation on Danish commons. K. Dan. Vidensk. Selsk. 1948, 5, 1–34. [Google Scholar]
- Bos, M.M.; Steffan-Dewenter, I.; Tscharntke, T. The contribution of cacao agroforests to the conservation of lower canopy ant and beetle diversity in Indonesia. Biodivers. Conserv. 2007, 16, 2429–2444. [Google Scholar] [CrossRef]
- Rice, R.A.; Greenberg, R. Cacao cultivation and the conservation of biological diversity. Ambio 2000, 29, 167–172. [Google Scholar] [CrossRef]
- Urquhart DH. Cocoa; Longmans Green and Co: London, UK, 1961. [Google Scholar]
- Bhagwat, S.A.; Willis, K.J.; Birks, H.J.B.; Whittaker, R.J. Agroforestry: A refuge for tropical biodiversity? Trends Ecol. Evol. 2008, 23, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Delabie, J.H.C.; Jahyny, B.; Cardoso do Nascimento, I.; Mariano, C.S.F. Contribution of cocoa plantations to the conservation of native ants (Insecta: Hymenoptera: Formicidae) with a special emphasis on the Atlantic Forest fauna of southern Bahia, Brazil. Biodivers. Conserv. 2007, 16, 2359–2384. [Google Scholar] [CrossRef]
- Ambrecht, I.; Perfecto, I.; Vandermeer, J. Enigmatic biodiversity correlations: Ant diversity responds to diverse resources. Science 2004, 304, 284–286. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).