Abstract
Early climate change ideas warned of widespread species extinctions. As scientists have probed more deeply into species responses, a more nuanced perspective emerged indicating that some species may persist in microrefugia (refugia), including in mountainous terrain. Refugia are habitats that buffer climate changes and allow species to persist in—and to potentially expand under—changing environmental conditions. While climate and species interactions in refugia have been noted as sources of uncertainty, land management practices and disturbances, such as wildland fire, should also be considered when assessing any given refugium. Our landscape scale study suggests that cold-air pools, an important type of small-scale refugia, have unique fire occurrence, frequency, and severity patterns in frequent-fire mixed conifer forests of California’s Sierra Nevada: cold-air pool refugia have less fire and if it occurs, it is lower severity. Therefore, individuals and small populations are less likely to be extirpated by fire. Active management, such as restoration and fuels treatments for climate change adaptation, may be required to maintain these distinctive and potentially important refugia.
1. Introduction
Early models of climate change impacts predicted widespread species extinctions as the rate of climate change outpaced the ability of plants and animals to migrate and track suitable climate [1,2,3]. Subsequent investigations suggest a more nuanced perspective, indicating that while species extinctions may still be dramatic, some species are likely to persist in microrefugia (referred to hereafter as refugia), including in mountainous terrain [4,5,6,7,8,9,10]. The heterogeneous conditions created by complex montane topography create local areas, refugia, where aspects of current climate may be maintained within the region for more than 100 years during climate change [6,11]. As the ice sheets retreated following the last ice age, these refugia are believed to have played an integral role in the rapid expansion of many species by providing source propagules for rapid species migration [8,9,10,12,13,14].
Many studies have sought to define refugia based on biological [7] or climatic [5] evidence. Keppel et al.’s (2011) biological definition of refugia is “habitats that components of biodiversity retreat to, persist in and can potentially expand from under changing environmental conditions.” Dobrowski et al., (2011) describe refugia as locations where extant climates (temperature and available water) are maintained during periods of climate change. Together they form a holistic definition, a habitat that buffers climate and allows species to persist in and to potentially expand from in response to changing environmental conditions. Refugia with relictual species, such as a disjunct southern population persisting from past large-scale populations during a past ice age, may or may not continue to function as refugia with climate change and changing disturbance regimes. Here, we focus on the importance of fire regimes as a component of potential refugia during periods of climate change. For fire sensitive species, landscape locations with reduced fire frequency or severity will serve as refugia with distinct disturbance regimes. Here we address the question of how some refugia in montane ecosystems may buffer both climate change and fire disturbance, with the potential to maintain species through episodes of climate change.
Refugia have attracted attention as important conservation areas [4,15], however the full range of conservation threats in these areas, including wildfire, have not been fully explored. Keppel and Wardell-Johnson (2012) highlight how refugia play a potentially important role in climate buffering and may offer in situ conservation benefits in the face of climate change and its biological effects [4]. We expand ‘climate change and its biological effects’ to explicitly include ecological processes such as fire. In addition, we consider the importance of land management practices because of how policies, such as fire suppression, have drastically changed fire frequency and severity patterns in many forests [16]. Climate, species interactions, ecological processes, and land management combine to create conservation challenges and opportunities for individual refugia (Figure 1).
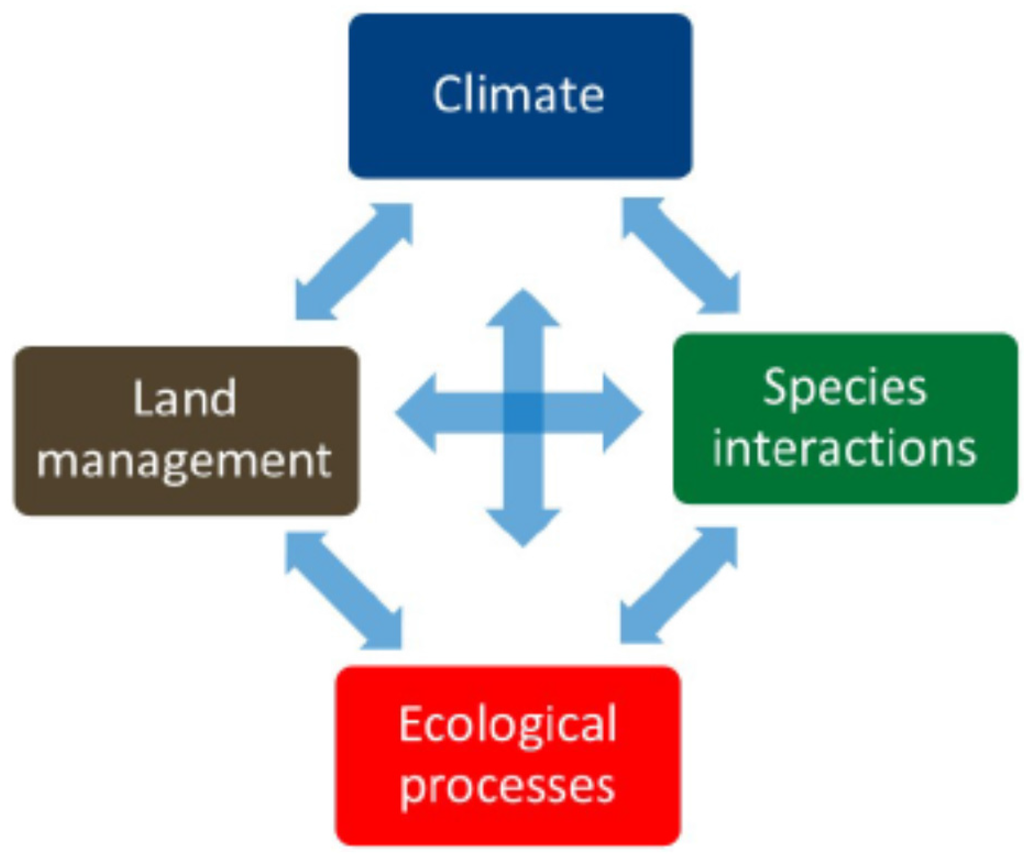
Figure 1.
Refugia ecology is complex and affected by climate, land management, species interactions, ecological processes, and their interactions.
Fire is one example of a changing ecological process. Fire frequency and severity are changing world-wide due to land management [16,17,18,19,20] and climate change [19,21,22,23]. Refugia populations are at a greater risk from changing fire patterns [21] due to their predisposition to local extinction because of their small, isolated nature, especially if the plants are fire avoiders [24]. Fire could alter species occurrence directly by killing vegetation or indirectly by altering vegetation’s moderating effect on climate [25,26] and the selection pressures on vegetation [27]. In more extreme cases fire can locally extirpate plants if fires are more frequent [28], less frequent [29], or more severe than historical fire regimes [30]. Some define refugia as areas with complete absence of fire [31]; however in arid regions with frequent fire, a reduction in frequency or severity may also create distinct conditions that serve as biological refugia for some taxa.
In this paper we focus on cold-air pool refugia (CAPs), a particular example of climatic refugia occurring in mountainous regions including our study region in the Sierra Nevada of California [5]. CAPs have lower temperatures and more frequent minimum temperatures, below any chosen threshold. And maximum temperatures are also lower, with fewer days exceeding certain thresholds. CAPS often have greater moisture availability than their surrounding landscape, due to reduced evaporative demand and water accumulation into low-lying areas [32,33]. While overall temperatures are warming, the weather patterns that produce cold-air pooling at a landscape scale are projected to increase, so the frequency and duration of cold-pools may increase in the Sierra Nevada with climate change [34,35]. Alternatively, if the weather patterns that produce cold-air pooling decrease, CAPs may warm at a greater rate than the landscape, yet still have lower average temperatures than the landscape. Overall CAPs have and will likely continue to have cooler and moister climates that their surrounding landscape although the magnitude of these differences is uncertain.
Plants respond to environmental conditions within CAPs and these sites often have plants characteristically found at higher elevations or latitudes. Similar climatic refugia forests are known as frost hollows in Quebec’s boreal temperate forests [36] and cove forests in southern Appalachia especially in Great Smoky Mountain National Park [37]. Another well-known example is subalpine fir (Abies lasiocarpa), which descend more than 300 m into river valleys with cold-air pools in Idaho [38,39]. In a more visually extreme case, the subalpine tree line is inverted due to cold-air pooling where mixed conifer forests grow above subalpine treeless regions on Mt. Hotham in Australia [40]. In the White Mountains of California, bristlecone pine are expanding downhill into local cold-air pools as temperatures warm [41]. Lesser known examples in the CAPs of Yosemite National Park in California include the Merced Grove, with disjunct distributions of species more characteristic of the Pacific Northwest, such as mountain lady's slipper (Cypripedium montanum) and Old-man’s-beard Lichen (Alectoria sarmentosa), growing together with Sierran species. These areas are dominated by ponderosa pine (Pinus ponderosa) intermingled with sugar pine (Pinus lambertiana), white fir (Abies concolor), and mountain hemlock (Tsuga mertensiana) with western hazelnut (Corylus cornuta) and mountain dogwood (Cornus nuttallii) in the understory [42] (Figure 2). As the climate changes and species ranges shift, we may see different species become restricted to CAPs, and the fate of the examples noted here are unknown.

Figure 2.
Refugia where species retreated to after the last ice age, such as regions of Yosemite National Park’s Merced Giant Sequoia Grove, have cooler climates with disjunct species characteristic of the Pacific Northwest such as Alectoria sarmentosa (Witch’s hair lichen) pictured here. Yosemite National Park has used prescribed fires to protect areas from large, nearly unstoppable fires such as the catastrophic Rim Fire in 2013. Photo by Martin Hutten.
Fire refugia and climate change refugia are similar, but they are also distinct. These refugia share some topographical similarities, and are both often associated with cooler, wetter places on the landscape [5,43,44]. Fire refugia are areas where fire has been excluded [45] whereas climate change refugia are areas that buffer climate change and the fire regime may be dramatically or subtlety different than the surrounding landscape [5]. Both likely have distinct fire regimes from their surrounding areas, although the magnitude may be different.
CAPs have unique climates and plants and they may serve as in situ conservation opportunities in the face of climate change. However, little is known about CAPs’ fire ecology and risk. Therefore, we investigate the fire ecology of CAPs. Specifically we ask:
- Do cold-air pools have similar fire occurrence as their surrounding landscape?
- Do cold-air pools have similar fire frequency as their surrounding landscape?
- If fires occur, do cold-air pools have similar fire severity patterns as their surrounding landscape?
2. Materials and Methods
2.1. Study Site
The study was conducted in mixed conifer forests in central California’s Sierra Nevada Range within Yosemite National Park (37.8499° N, 119.5677° W), approximately 240 km inland from the Pacific Ocean. Historical fires were frequent and burnt every 6 to 15 years [46,47]. Fire suppression occurred from the late 1800s to the mid-1900s. Beginning in the 1970s, fire patterns were allowed to return to historical patterns, as lightning strike fires were allowed to burn freely through nearly one-third of the study area and prescribed fires occurred over a large area as well [23,46,47]. A large fire deficit persists for more than two-thirds of our study area. Vegetation is mixed conifer forest co-dominated by fire resilient species including ponderosa pine (Pinus ponderosa), Jeffrey pine (Pinus jeffreyi), white fir (Abies concolor), and red fir (Abies magnifica), and scattered meadows and shrublands. The study region is from 1000 to 2600 m elevation and has hot, dry summers followed by cold, snowy winters.
2.2. Study Design
Spatial data was prepared in ArcGIS 10.2 (ESRI, Redlands CA, USA) and projected using bilinear interpolation into NAD 83 UTM 11N. Relevant spatial data was compiled from private and public sources, including predicted location of CAPs (based on methodology of [32,48]), fire history polygons from 1930 until 2012 [49], and Relative differenced Normalized Burn Ratio (RdNBR) fire severity categories based on changes in tree cover from 1984 to 2012 [50,51] (Figure 3). Predicted CAP values included absent (no cold-air pooling occurs), marginal (areas with no clear signal for cold-air pooling due to topography and weather patterns and present (areas with potential for cold-air pooling to occur). The predicted CAPs are areas with potential to pool cold-air and there is variation in the frequency and duration of cold-air pooling. CAPs include areas with short and infrequent cold-air pooling that have similar climate and fire history to the surrounding area that are false-positives for climate change refugia. Conversely, areas predicted to lack CAPs may have CAPs present and thus represent false-negatives from the modeling procedure. Fire severity classes correlate to the change in canopy cover and tree basal area whereby very low and low severity fires have between 0 and 25% change, moderate severity fires have between 26 and 75% change, and high severity fires have more than 76% change in canopy cover and tree basal area [52].
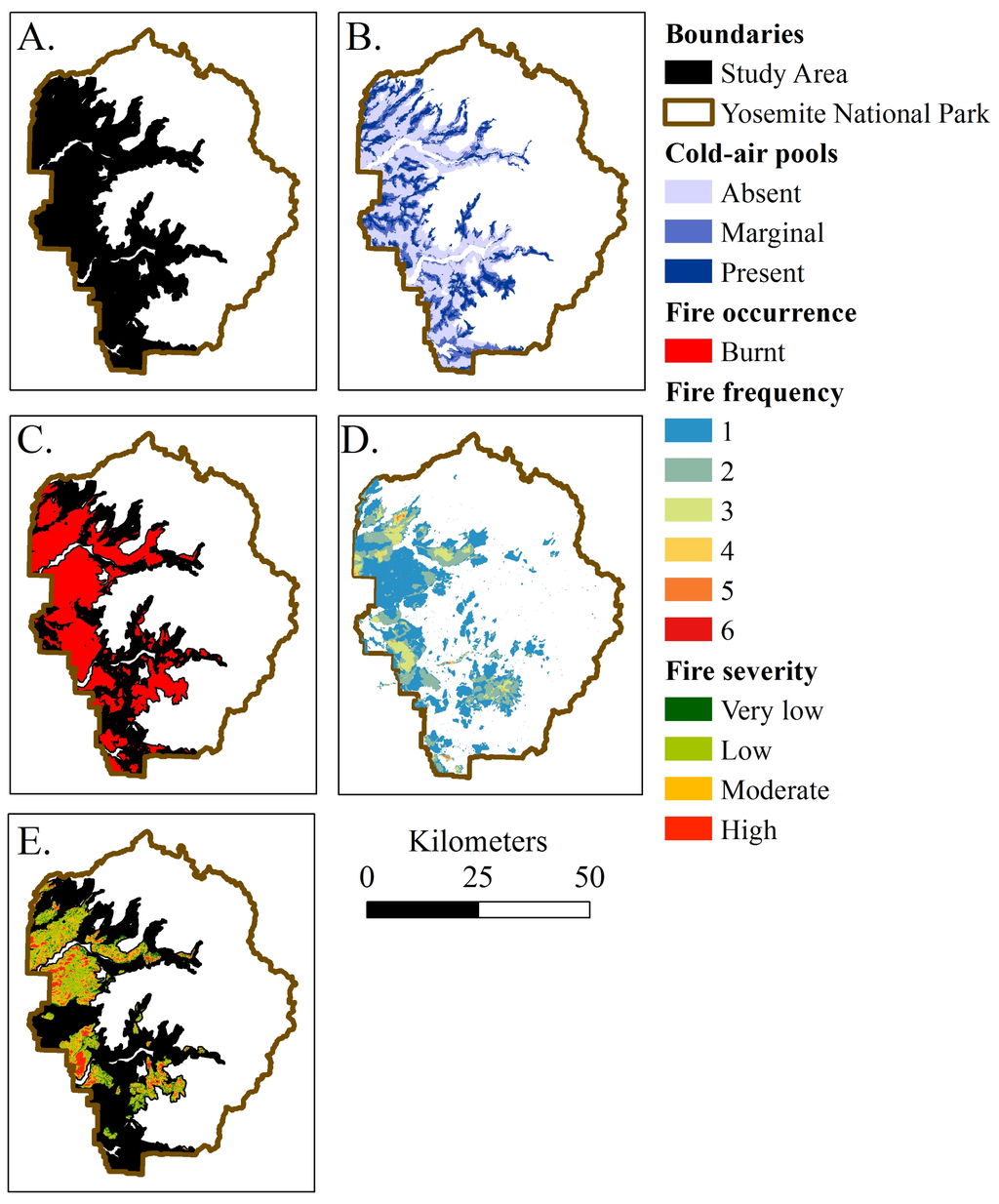
Figure 3.
(A) The study area included mixed conifer forests of Yosemite National Park from 1000 to 3600 m in elevation, which encompassed about 170,000 ha; (B) Predicted cold-air pools (CAPs) followed drainages and had a semi-regular pattern throughout the study area. The study area was dominated by areas without CAPs (100,000 ha) followed by areas with marginal CAPs (40,000 ha), and lastly CAPs (30,000 ha); (C) Fires have occurred through about 60% study area from 1930 to 2012; (D) Areas within the study area have burnt up to 6 times; (E) Fire severity distribution from 1984 to 2012 was dominated by low severity followed by moderate severity fires. Fire severity area included: very low severity 16,000 ha, low severity 44,000 ha, moderate severity 28,000 ha, and high severity 12,000 ha.
Spatial data was clipped with a USGS 10 meter Digital Elevation Model to restrict it to the mixed conifer zone [53]. Spatial autocorrelation of cold-air pools, fire frequency, and fire severity were tested with ArcGIS’s Spatial Autocorrelation Global Moran’s I test from 100–300 m and statistical significance was assessed with the z-score. The data were not autocorrelated at 100 m, but they were autocorrelated for other distances tested (Figure A1, Figure A2 and Figure A3). Therefore, a 100 m point grid was created in ArcGIS with the Create Fishnet Geoprocessing Tool. Equal sample sizes from all levels of CAPs (absent, marginal, present) were randomly subsampled without replacement for both analyses, including fire occurrence, and if fire occurs, its severity (using the sample function in R 3.1.2 [54]).
2.3. Analysis
All analyses were conducted in R 3.1.2 [54]. We constructed statistical models to test if cold-air pools are related to fire Equation (1).
We used linear models to test the significance of fire occurrence and frequency. We used binomial distribution for fire occurrence and a Gaussian distribution for fire severity, based on the four ordered fire severity classes. All model dispersion and residuals were reviewed. We used chi-squared test to test the significance of fire severity.
3. Results
CAPs were significantly less likely to have experienced a fire than their surrounding landscape (Figure 4A) (P < 0.0001). Areas without CAPs burnt 60% of the time on average,, marginal CAPs burnt 52% of the time on average, and CAPs burnt 44% of the time on average. CAPs also had significantly fewer fires than their surrounding landscape as well (Figure 4B) (P < 0.0001). Areas without CAPS had a fire frequency of 0.95 on average, areas with marginal CAPs had a fire frequency of 0.78 on average, and areas with CAPs had a fire frequency of 0.60 on average.
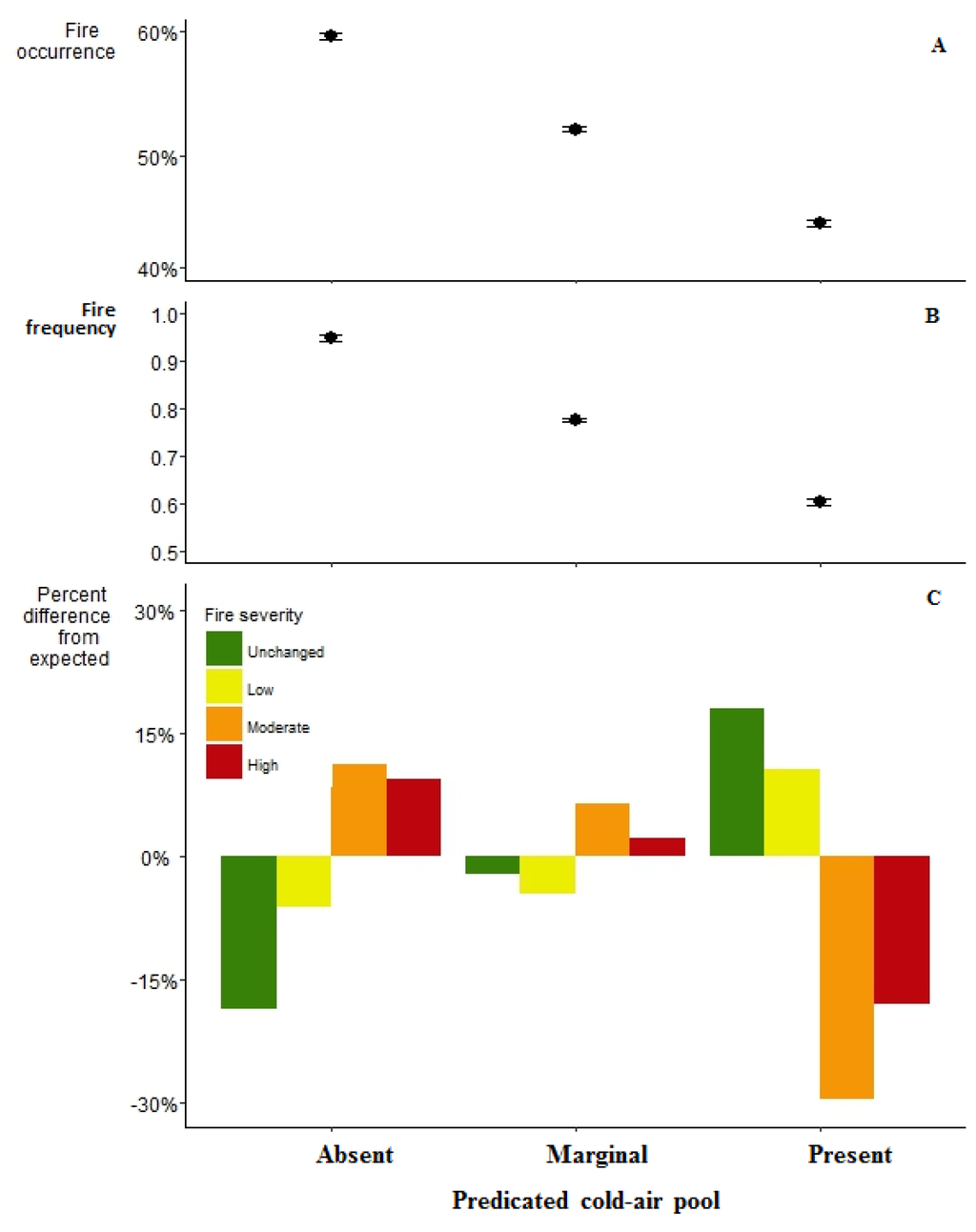
Figure 4.
(A) Regression estimates for area burnt grouped by their predicted CAP level where dots represent the regression coefficients and whiskers are the 95% confidence intervals; (B) Regression estimates for number of times burnt grouped by their predicted CAP level where dots represent the regression coefficients and whiskers are the 95% confidence intervals; (C) Chi-square estimates for the percent differences in the actual fire severity from expected by their predicted CAP level.
Additionally, if CAPs burned, they had significantly less severe fires than their surrounding landscapes (P < 0.0001). Areas with CAPs had 16% more very low severity fire, 10% more low severity fire, 29% less moderate severity fire, and 16% less high severity fire than expected. Areas with marginal CAPs had 3% less very low severity fire, 4% less low severity fire, 7% more moderate severity fire, and 3% more high severity fire than expected. Areas without CAPs had 16% less very low severity fire, 6% less low severity fire, 11% more moderate severity fire, and 9% more high severity fire.
4. Discussion
CAP refugia had significantly different fire patterns from the surrounding landscape likely due to a combination of their vegetation, topography, or microclimate. Surprisingly, these trends persisted despite our landscape-scale study using predicted CAPs which may have reduced the strength of our results. The fire regime differences between CAP refugia and their surrounding landscape are likely a conservative estimate because prediction of a CAP does not include its frequency, duration or if biological species respond to its effects. The actual magnitude of the effects might be greater if we had directly field mapped CAPs.
Refugia in rugged terrain separated by fire barriers such as rock or water are more likely to have decreased fire occurrence than their surrounding terrain through these bottom-up controls [55]. Other refugia may lack a physical barrier to fire, but their distinct microclimates can influence fire behavior [44,45]. CAPs may have direct effects on fire behavior (temperature, wind, and fuel moisture) as well as indirect effects mediated through vegetation and fuel characteristics (amount and size distribution of fuel, fuel continuity, fuel moisture, forest structure, and relative humidity) [56]. These effects are realized as reductions in fire energy (commonly called intensity) and changes in dominant vegetation (commonly called severity), and in some cases even reducing fire extent if the fire self-extinguishes (Figure 5).
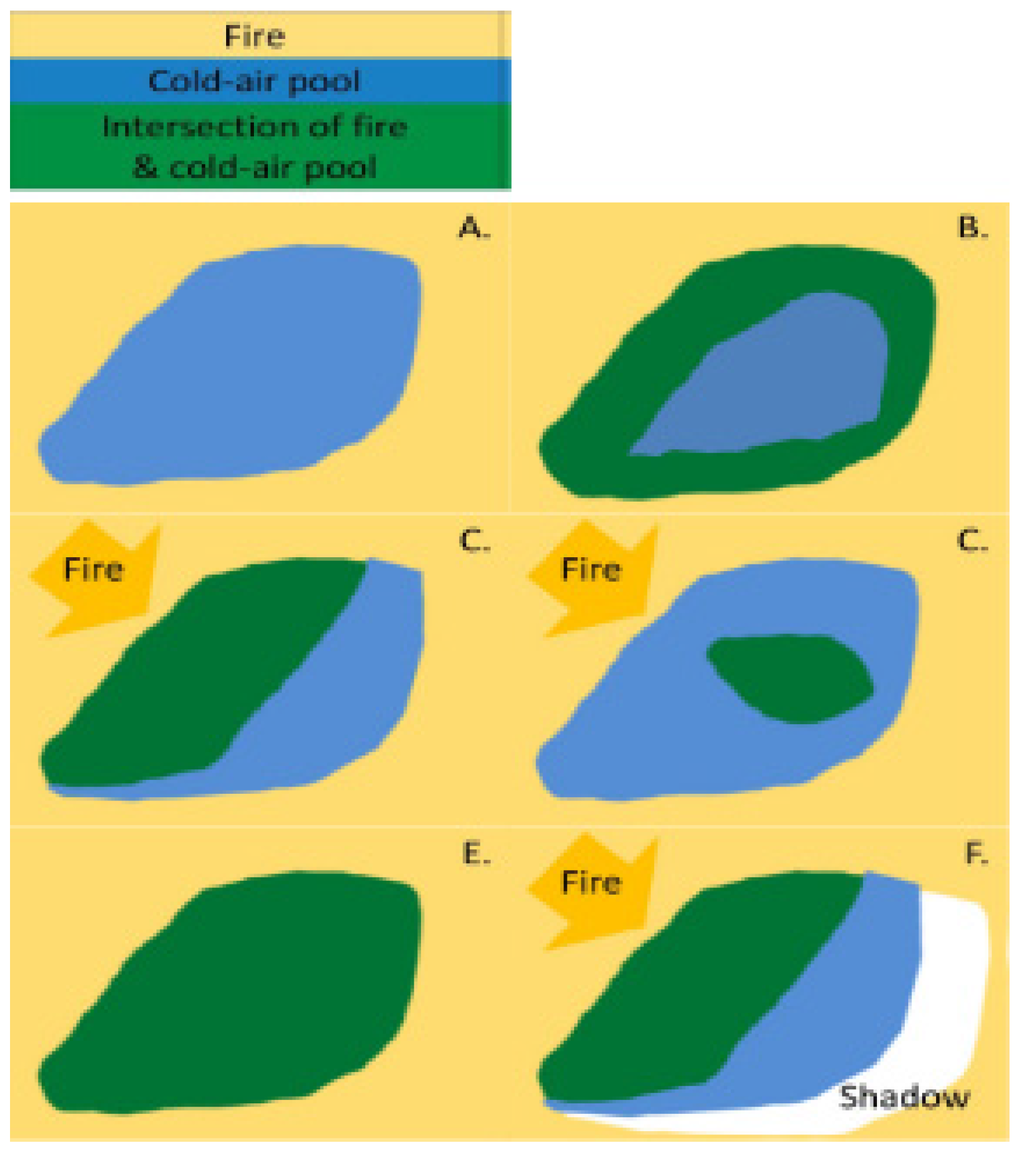
Figure 5.
The interaction of fire and CAPs may be dependent upon fire behavior including the fire’s direction, magnitude, and intensity. The yellow background indicates where fire occurred and the golden arrows indicate the fire’s direction of movement. (A) Fires which move slowly (low magnitude) and release little energy (low intensity) may respond quickly to a refugium’s microenvironment and not penetrate the CAP, whereas (B) fires with high magnitude and intensity may respond slowly to a CAP (burn a buffer around the perimeter); (C) and/or a larger region near the flame front; (D) create a spot fire ignites within the CAP which grows especially during dry windy conditions; or (E) can burn the entire CAP. (F) There also may be a CAP fire shadow where a reduction in fire extent or severity occurs because of its proximity to a CAP and the subsequent reduction in fire presence or severity.
The interaction between CAPs and fire may be influenced by diurnal patterns because drivers of fire behavior vary diurnally including temperature, wind, and humidity. CAPs may be cooler in the evening and morning but reach similar maximum daytime temperatures as surrounding areas, but fire effects may be lessened by higher humidity and residual fuel moisture [32,56,57]. Overall, our analysis demonstrates that the unique climate and topographic placement of CAPs reduce fire frequency and intensity and thus, quite possibly, the impact of fire on these ecosystems.
The interaction between CAPs and fire may be moderated by effects of season and weather on fire behavior. Historically fuel moisture would have been lower in fall due to seasonal drought and thus there may have been an increase in fire occurrence and severity during this time. Climate change may magnify this trend because extreme fire weather (hot, dry and windy weather) is becoming more common, which allows fires to grow rapidly and reach unprecedented fire intensity and size [58]. As fire behavior becomes more extreme, bottom-up controls such as microclimate, topography, and fire barriers are weaker [55]. If fire becomes driven by extreme events such as drought, high winds, or in extreme cases where the combination of weather, topography and fuels catalyze fire-generated weather phenomena such as plume-dominated fires, then historic fire barriers of refugia may no longer function [59]. Large, severe fire was evident in the 2013 Rim Fire, which burned over 100,000 ha, impacting a noticeable portion of our study area [60]. Refugia that historically have had physical and climatic barriers to fire may be more susceptible to high-severity mega-fires becaue they produce effects well outside of desired ranges [20].
Refugia are commonly embedded within riparian areas in our study region. Other studies focused on riparian forest fire ecology suggest that these areas did burn historically, that these areas have demonstrated the greatest rate of fuel accumulation during fire suppression, and are at greatest risk for uncharacteristic high severity fire now due to fuel accumulations [61,62]. While historic fires only influenced aquatic communities in the short-term if at all [63], uncharacteristic large, high severity fires may have a larger influence. Uncharacteristic high severity fires reduce canopy cover, change the peak water temperatures in creeks, and may sometimes negatively influence sensitive aquatic species [64,65].
Implications for Management
More than 100 years of fire suppression in the Sierra Nevada have fostered dense tree stands with ladder fuels including small trees reaching from the forest floor to its’ canopy. Today’s mixed conifer forests are more susceptible to high severity fire than ever before [66]. There is a consensus that we need to reduce tree density and fuels to make forests resilient to climate-change induced disturbances [67,68,69]. While prescribed fire and mechanical fuel reduction treatments may have short-term effects on the inhabitants of refugia, the long-term lack of fire exacerbates climate change’s increasing disturbance threats to biodiversity, including increased fire frequency and severity [22,70].
Refugia, especially in arid regions like Yosemite National Park have greater moisture, fuel production, and historically lower fire occurrence than the surrounding landscape [5,32,62]. Refugia fire occurrence will likely increase relative to historical levels due to the combination of fire suppression and climate change induced extreme fires or mega-fires [20,71]. Even as fire risk/frequency increases, refugia are likely to maintain distinct fire regimes relative to surrounding areas. Refugia commonly occur in riparian areas, which were heavily altered by fire suppression resulting in extraordinary amounts of fuel (more than five times greater than historic levels), leaving them uncharacteristically susceptible to high severity fire that might be quite detrimental to biodiversity [62]. As climate becomes drier, these fuels have reduced moisture and are therefore available to burn for a larger portion of the year [72]. These additional fuels can contribute to more severe fires.
High-severity fire risk could be mitigated with prescribed fire, mechanical treatments, or managed wildfire within the CAP and at the landscape level [73]. Fuel reduction could have unintended consequences, since trees moderate climate. Trees filter solar radiation, providing a buffer between CAPs and the atmosphere; removing trees can increase maximum surface temperatures 10 to 40 °C [26]. Oddly, fire suppression which increases tree density may have also made CAPs climates cooler. To reduce the potential to increase temperatures dominant trees should be preserved in fuel treatments [74]. Nonetheless, managers must understand the trade-offs for refugia management and thresholds between reducing fire risk and altering the very environment that they wish to protect.
Stand-replacing fires surrounding CAPs are also of concern because large-scale high severity fires can kill many trees within the CAP air shed and in doing so may also change the local climate. Killing trees that moderate the temperature and soil moisture in the greater air shed may reduce the cold-air source and thus the frequency and duration of cold-air pooling. Trees likely have strong biological feedback on climate in gentle sloping environments because there are not strong topographical drivers of cold-air movement. Steeper slopes have faster cold-air run-off and trees likely play a smaller role here. Fuel and restoration treatments could be completed in areas surrounding CAPs to protect cold-air sources from severe fire. As before, caution is needed when applying fuel treatments because trees moderate temperature and minimizing these impacts will be a higher priority with climate change.
5. Conclusions
Conservation planners are advised that their “highest priority (is) to reduce negative edge effects and improve in situ management of existing habitat patches” [75]. Refugia do exactly this, allowing in situ management of habitat patches [75]. Refugia are complex habitats influenced by species interactions, climate, and fire that interact with one another. Therefore, protecting the land associated with refugia is not sufficient to protect the biological and physical properties of refugia; additional management actions are necessary. Many of these actions, like strategic fuel treatments or managed wildfire, are already recommended for both forest restoration and climate change adaptation [70,73]. Refugia are also susceptible to disturbances, therefore redundancy on the landscape is necessary to utilize this conservation strategy. Actions to manage refugia are similar to manager’s current tool kits for conservation, but the need for these actions in refugia may be an additional incentive to complete them. Managers will be asked to make decisions about refugia without understanding their full ecological complexity and they must understand that refugia are not static.
Acknowledgments
The George Melendez Wright Youth Climate Change Initiative supported Wilkin for this project that catalyzed National Park Service collaborations that enriched the final product. Special thanks to Yosemite National Park employees (Alison Colwell, Martin Hutten, Kent Van Wagtendonk, Mitzi Thornley, and Linda Mazzu) and Devils Postpile National Monument employees (Monica Buhler and Deanna Dulen) for their support with project development, grant and manuscript reviews, and their insights into managing climate change refugia. This paper would not have been possible without the Climate Refugia Workshop in Eugene, Oregon in August of 2012 sponsored by the Ecological Society of America. K. W. is grateful to those who coordinated this meeting (Dan Gavin and Erin Herring) and the participants who inspired analysis of refugia management for the future with climate change, especially Zack Holden and Arndt Hampe. Special thanks to Jessica Lundquist for fostering a sound understanding of cold-air pools and to Axel Kuhn for producing and sharing a Sierra Nevada data layer based on Lundquist (2008).
Author Contributions
Kate Wilkin, David Ackerly and Scott Stephens conceived and designed the analysis; Kate Wilkin performed the experiments; Kate Wilkin analyzed the data; Kate Wilkin wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
Abbreviations
The following abbreviations are used in this manuscript:
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| CAP | Cold-air pool |
| RdNBR | Relative differenced Normalized Burn Ratio |
| Refugia | microrefugia |
Appendix A. Spatial autocorrelation
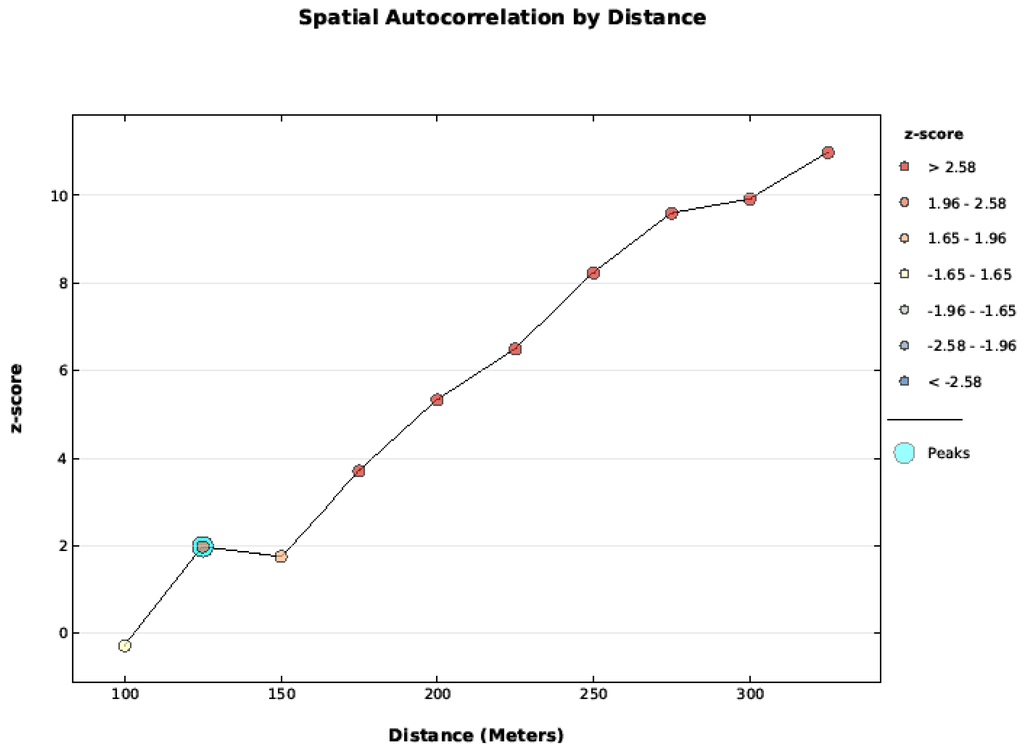
Figure A1.
Cold-air pool data was tested with the Global Moran’s I applied to Incremental Spatial Autocorrelation starting at 100 m and increasing at 50 m intervals. The data was not spatially auto correlated at 100 m, but became correlated by 125 m and beyond.
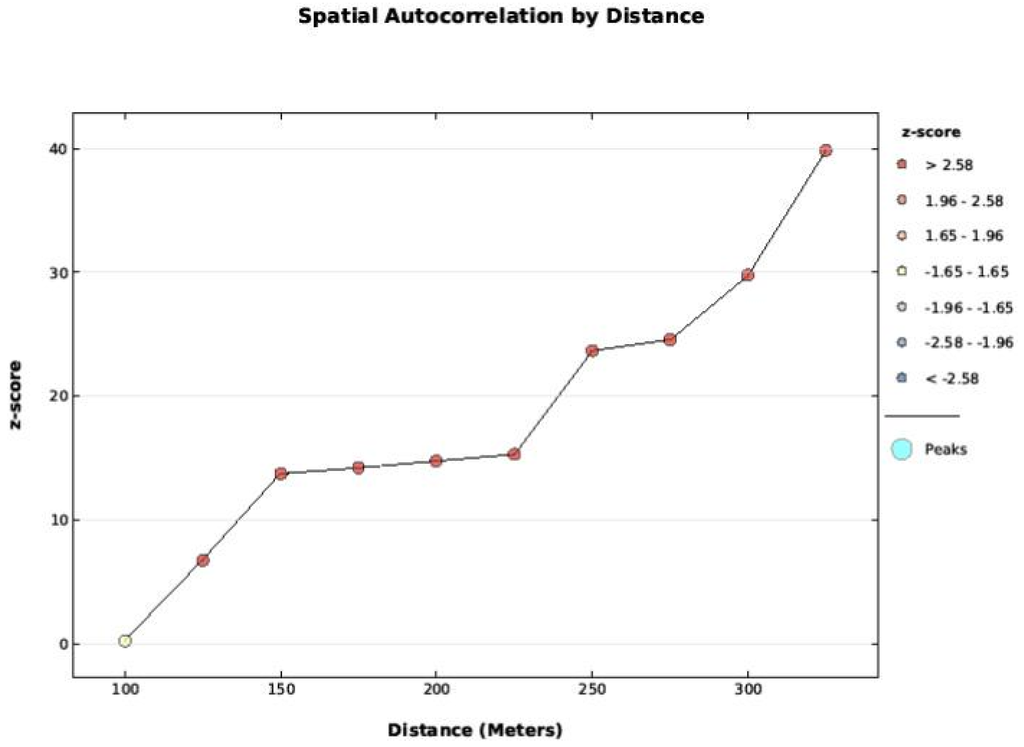
Figure A2.
Fire frequency data was tested with the Global Moran’s I applied to Incremental Spatial Autocorrelation starting at 100 m and increasing at 50 m intervals. The data was not spatially auto correlated at 100 m, but become increasingly correlated as distance increased.
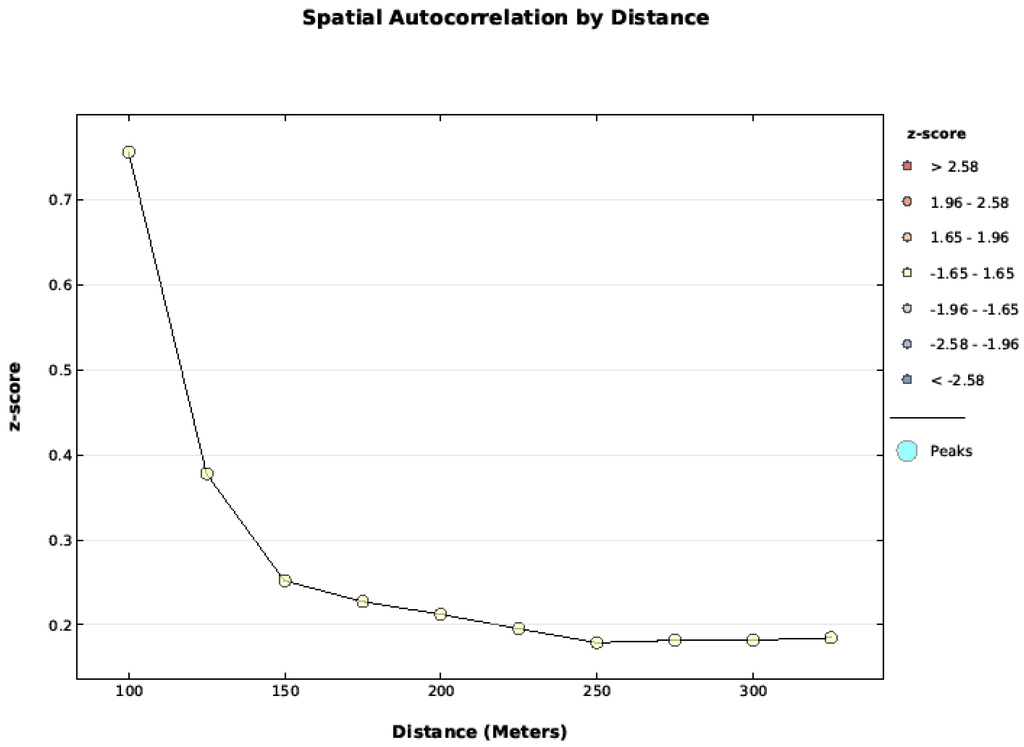
Figure A3.
Fire severity data was tested with the Global Moran's I applied to Incremental Spatial Autocorrelation starting at 100 m and increasing at 50 m intervals. The data was not spatially auto correlated between 100 and 300 m.
References
- Walther, G.R.; Post, E.; Convey, P.; Menzel, A.; Parmesank, C.; Beebee, T.J.C.; Fromentin, J.; Hoegh-GuldbergI, O.; Bairlein, F. Ecological responses to recent climate change. Nature 2002, 416, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Root, T.L.; Price, J.T.; Hall, K.R.; Schneider, S.H.; Rosenzweig, C.; Pounds, J.A. Fingerprints of global warming on wild animals and plants. Nature 2003, 421, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.D.; Cameron, A.; Green, R.E.; Bakkenes, M.; Beaumont, L.J.; Collingham, Y.C.; Erasmus, B.F.; De Siqueira, M.F.; Grainger, A.; Hannah, L. Extinction risk from climate change. Nature 2004, 427, 145–148. [Google Scholar] [CrossRef] [PubMed]
- Keppel, G.; Wardell-johnson, G.W. Refugia: Keys to climate change management. Glob. Chan. Biol. 2012, 2389–2391. [Google Scholar] [CrossRef]
- Dobrowski, S.Z. A climatic basis for microrefugia: The influence of terrain on climate. Glob. Chan. Biol. 2011, 17, 1022–1035. [Google Scholar] [CrossRef]
- Loarie, S.R.; Duffy, P.B.; Hamilton, H.; Asner, G.P.; Field, C.B.; Ackerly, D.D. The velocity of climate change. Nature 2009, 462, 1052–1055. [Google Scholar] [CrossRef] [PubMed]
- Keppel, G.; van Niel, K.P.; Wardell-Johnson, G.W.; Yates, C.J.; Byrne, M.; Mucina, L.; Schut, A.G.T.; Hopper, S.D.; Franklin, S.E. Refugia: Identifying and understanding safe havens for biodiversity under climate change. Glob. Ecol. Biogeogr. 2011, 393–404. [Google Scholar] [CrossRef]
- Taberlet, P.; Cheddadi, R. Quaternary refugia and persistence of biodiversity. Science 2002, 297, 2009–2010. [Google Scholar] [CrossRef] [PubMed]
- Petit, R.J.; Aguinagalde, I.; de Beaulieu, J.-L.; Bittkau, C.; Brewer, S.; Cheddadi, R.; Ennos, R.; Fineschi, S.; Grivet, D.; Lascoux, M.; et al. Glacial refugia: Hotspots but not melting pots of genetic diversity. Science 2003, 300, 1563–1565. [Google Scholar] [CrossRef] [PubMed]
- Petit, R.J.; Csaikl, U.M.; Bordács, S.; Burg, K.; Coart, E.; Cottrell, J.; van Dam, B.; Deans, J.D.; Dumolin-Lapègue, S.; Fineschi, S.; et al. Chloroplast DNA variation in european white oaks: Phylogeography and patterns of diversity based on data from over 2600 populations. For. Ecol. Manag. 2002, 156, 5–26. [Google Scholar] [CrossRef]
- Dobrowski, S.Z.; Abatzoglou, J.; Swanson, A.K.; Greenberg, J.A.; Mynsberge, A.R.; Holden, Z.A.; Schwartz, M.K. The climate velocity of the contiguous united states during the 20th century. Glob. Chan. Biol. 2012, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Taberlet, P.; Fumagalli, L.; Wust-Saucy, A.-G.; Cosson, J.-F. Comparative phylogeography and postglacial colonization routes in europe. Mol. Ecol. 1998, 7, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Willis, K.J.; Whittaker, R.J. The refugial debate. Science 2000, 287, 1406–1407. [Google Scholar] [CrossRef] [PubMed]
- Pearson, R.G. Climate change and the migration capacity of species. Trends Ecol. Evol. 2006, 21, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Groves, C.; Game, E.; Anderson, M.; Cross, M.; Enquist, C.; Ferdaña, Z.; Girvetz, E.; Gondor, A.; Hall, K.; Higgins, J.; et al. Incorporating climate change into systematic conservation planning. Biodivers. Conserv. 2012, 21, 1651–1671. [Google Scholar] [CrossRef]
- Miller, J.D.; Collins, B.M.; Lutz, J.A.; Stephens, S.L.; van Wagtendonk, J.W.; Yasuda, D.A. Differences in wildfires among ecoregions and land management agencies in the Sierra Nevada region, California, USA. Ecosphere 2012, 3, 1–20. [Google Scholar] [CrossRef]
- Anderson, K. Tending the wild: Native American Knowledge and the Management of California’s Natural Resources; University of California Press: Berkeley, CA, USA, 2005; p. 558. [Google Scholar]
- Falk, D.A.; Heyerdahl, E.K.; Brown, P.M.; Farris, C.; Fulé, P.Z.; McKenzie, D.; Swetnam, T.W.; Taylor, A.H.; van Horne, M.L. Multi-scale controls of historical forest-fire regimes: New insights from fire-scar networks. Front. Ecol. Environ. 2011, 9, 446–454. [Google Scholar] [CrossRef]
- Trouet, V.; Taylor, A.H.; Wahl, E.R.; Skinner, C.N.; Stephens, S.L. Fire-climate interactions in the american west since 1400 ce. Geophys. Res. Lett. 2010, 37, L04702. [Google Scholar] [CrossRef]
- Stephens, S.L.; Burrows, N.; Buyantuyev, A.; Gray, R.W.; Keane, R.E.; Kubian, R.; Liu, S.; Seijo, F.; Shu, L.; Tolhurst, K.G. Temperate and boreal forest mega-fires: Characteristics and challenges. Front. Ecol. Environ. 2014, 12, 115–122. [Google Scholar] [CrossRef]
- Moritz, M.A.; Parisien, M.-A.; Batllori, E.; Krawchuk, M.A.; van Dorn, J.; Ganz, D.J.; Hayhoe, K. Climate change and disruptions to global fire activity. Ecosphere 2012, 3, 1–22. [Google Scholar] [CrossRef]
- Westerling, A.; Bryant, B. Climate change and wildfire in california. Clim. Chan. 2008, 87, 231–249. [Google Scholar] [CrossRef]
- Swetnam, T.W. Fire history and climate change in giant sequoia groves. Science 1993, 262, 885–889. [Google Scholar] [CrossRef] [PubMed]
- Rowe, J.S. Concepts of fire effects on plant individuals and species. The Role of Fire in Northern Circumpolar Ecosystems 1983, 18. [Google Scholar]
- Rambo, T.R.; North, M.P. Canopy microclimate response to pattern and density of thinning in a sierra nevada forest. For. Ecol. Manag. 2009, 257, 435–442. [Google Scholar] [CrossRef]
- Ford, K.R.; Ettinger, A.K.; Lundquist, J.D.; Raleigh, M.S.; Lambers, J.H.R. Spatial Heterogeneity in Ecologically Important Climate Variables at Coarse and Fine Scales in a High-Snow Mountain Landscape. PLoS ONE 2013, 8, e65008. [Google Scholar] [CrossRef] [PubMed]
- Bond, W.J.; Keeley, J.E. Fire as a global ‘herbivore’: The ecology and evolution of flammable ecosystems. Trends Ecol. Evol. 2005, 20, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Keeley, J.E.; Ne’eman, G.; Fotheringham, C. Immaturity risk in a fire-dependent pine. J. Mediterr. Ecol. 1999, 1, 41–48. [Google Scholar]
- Menges, E.S.; Hawkes, C.V. Interactive effects of fire and microhabitat on plants of florida scrub. Ecol. Appl. 1998, 8, 935–946. [Google Scholar] [CrossRef]
- Collins, B.M.; Roller, G.B. Early forest dynamics in stand-replacing fire patches in the northern sierra nevada, california, USA. Landsc. Ecol. 2013, 28, 1801–1813. [Google Scholar] [CrossRef]
- Nordén, B.; Dahlberg, A.; Brandrud, T.E.; Fritz, Ö.; Ejrnaes, R.; Ovaskainen, O. Effects of ecological continuity on species richness and composition in forests and woodlands: A review. Ecoscience 2014, 21, 34–45. [Google Scholar] [CrossRef]
- Lundquist, J.D.; Pepin, N.; Rochford, C. Automated algorithm for mapping regions of cold-air pooling in complex terrain. J. Geophys. Res. 2008, 113, D22. [Google Scholar] [CrossRef]
- Dobrowski, S.Z.; Abatzoglou, J.T.; Greenberg, J.A.; Schladow, S. How much influence does landscape-scale physiography have on air temperature in a mountain environment? Agric. For. Meteorol. 2009, 149, 1751–1758. [Google Scholar] [CrossRef]
- Daly, C.; Conklin, D.R.; Unsworth, M.H. Local atmospheric decoupling in complex topography alters climate change impacts. Int. J. Clim. 2010, 30, 1857–1864. [Google Scholar] [CrossRef]
- Pepin, N.C.; Daly, C.; Lundquist, J. The influence of surface versus free-air decoupling on temperature trend patterns in the western United States. J. Geophys. Res. 2011, 116, 16. [Google Scholar] [CrossRef]
- Dy, G.; Payette, S. Frost hollows of the boreal forest as extreme environments for black spruce tree growth. Can. J. For. Res. 2007, 37, 492–504. [Google Scholar] [CrossRef]
- Shanks, R.E. Climates of the great smoky mountains. Ecology 1954, 35, 354–361. [Google Scholar] [CrossRef]
- Axelrod, D.I. The Eocene Thunder Mountain flora of Central Idaho; University of California Press: Berkeley, CA, USA, 1998; Vol. 142. [Google Scholar]
- Daubenmire, R. Mountain topography and vegetation patterns. Northwest Sci. 1980, 54, 146–152. [Google Scholar]
- Wearne, L.J.; Morgan, J.W. Floristic composition and variability of subalpine grasslands in the mt hotham region, north-eastern victoria. Aust. J. Bot. 2001, 49, 721–734. [Google Scholar] [CrossRef]
- Millar, C.I.; Westfall, R.D.; Delany, D.L.; Flint, A.L.; Flint, L.E. Recruitment patterns and growth of high-elevation pines in response to climatic variability (1883–2013), in the western great basin, USA. Can. J. For. Res. 2015, 45, 1299–1312. [Google Scholar] [CrossRef]
- Colwell, A. Yosemite National Park Special Status Plants (Dataset); Yosemite National Park: Berkeley, CA, USA, 2012. [Google Scholar]
- Eberhart, K.E.; Woodard, P.M. Distribution of residual vegetation associated with large fires in alberta. Can. J. For. Res. 1987, 17, 1207–1212. [Google Scholar] [CrossRef]
- Camp, A.; Oliver, C.; Hessburg, P.; Everett, R. Predicting late-successional fire refugia pre-dating european settlement in the wenatchee mountains. For. Ecol. Manag. 1997, 95, 63–77. [Google Scholar] [CrossRef]
- Ouarmim, S.; Asselin, H.; Hély, C.; Bergeron, Y.; Ali, A.A. Long-term dynamics of fire refuges in boreal mixedwood forests. J. Quat. Sci. 2014, 29, 123–129. [Google Scholar] [CrossRef]
- Scholl, A.E.; Taylor, A.H. Fire regimes, forest change, and self-organization in an old-growth mixed-conifer forest, yosemite national park, USA. Ecol. Appl. 2010, 20, 362–380. [Google Scholar] [CrossRef] [PubMed]
- Collins, B.M.; Stephens, S.L. Managing natural wildfires in sierra nevada wilderness areas. Front. Ecol. Environ. 2007, 5, 523–527. [Google Scholar] [CrossRef]
- Lundquist, J.D.; Cayan, D.R. Surface temperature patterns in complex terrain: Daily variations and long-term change in the central Sierra Nevada, California. J. Geophys. Res. 2007, 112, D11124. [Google Scholar] [CrossRef]
- Yosemite National Park. Yosemite National Park Fire History Polygons from 1930 to 2011 (Dataset); Yosemite National Park: Berkeley, CA, USA, 2012. [Google Scholar]
- Miller, J.D. Yosemite National Park Wildfire Fire Severity from 1984 to 2010; USDA: McClellan, CA, USA, 2012.
- Miller, J.D.; Thode, A.E. Quantifying burn severity in a heterogeneous landscape with a relative version of the delta normalized burn ratio (dnbr). Remote. Sens. Environ. 2007, 109, 66–80. [Google Scholar] [CrossRef]
- Miller, J.D.; Knapp, E.E.; Key, C.H.; Skinner, C.N.; Isbell, C.J.; Creasy, R.M.; Sherlock, J.W. Calibration and validation of the relative differenced normalized burn ratio (rdnbr) to three measures of fire severity in the sierra nevada and klamath mountains, California, USA. Remote. Sens. Environ. 2009, 13, 645–656. [Google Scholar] [CrossRef]
- Gesch, D.B. The National Elevation Dataset. American Society for Photogrammetry and Remote Sensing; American Society for Photogrammetry and Remote Sensing: Bethesda, MD, USA, 2007. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2008. [Google Scholar]
- Heyerdahl, E.K.; Brubaker, L.B.; Agee, J.K. Spatial controls of historical fire regimes: A multiscale example from the interior west, USA. Ecology 2001, 82, 660–678. [Google Scholar]
- Brown, J.K. Handbook for inventorying downed woody material; USDA, Ed.; Intermountain Forest and Range Experiment Station: Ogden, UT, USA, 1974. [Google Scholar]
- Fosberg, M.A.; Rothermel, R.C.; Andrews, P.L. Moisture content calculations for 1000-hour timelag fuels. For. Sci. 1981, 27, 19–26. [Google Scholar]
- Collins, B.M. Fire weather and large fire potential in the northern sierra nevada. Agric. For. Meteorol. 2014, 189–190, 30–35. [Google Scholar] [CrossRef]
- Turner, M.G.; Romme, W.H.; Gardner, R.H. Prefire heterogeneity, fire severity, and early postfire plant reestablishment in subalpine forests of yellowstone national park, wyoming. Int. J. Wildland Fire 1999, 9, 21–36. [Google Scholar] [CrossRef]
- Lydersen, J.M.; North, M.P.; Collins, B.M. Severity of an uncharacteristically large wildfire, the rim fire, in forests with relatively restored frequent fire regimes. For. Ecol. Manag. 2014, 328, 326–334. [Google Scholar] [CrossRef]
- Van de Water, K.; North, M. Fire history of coniferous riparian forests in the sierra nevada. For. Ecol. Manag. 2010, 260, 384–395. [Google Scholar] [CrossRef]
- Van de Water, K.; North, M. Stand structure, fuel loads, and fire behavior in riparian and upland forests, sierra nevada mountains, USA; a comparison of current and reconstructed conditions. For. Ecol. Manag. 2011, 262, 215–228. [Google Scholar] [CrossRef]
- Beche, L.A.; Stephens, S.L.; Resh, V.H. Effects of prescribed fire on a sierra nevada (california, USA) stream and its riparian zone. For. Ecol. Manag. 2005, 218, 37–59. [Google Scholar] [CrossRef]
- Rieman, B.; Clayton, J. Wildfire and native fish: Issues of forest health and conservation of sensitive species. Fisheries 1997, 22, 6–15. [Google Scholar] [CrossRef]
- Hitt, N.P. Immediate effects of wildfire on stream temperature. J. Freshw. Ecol. 2003, 18, 171–173. [Google Scholar] [CrossRef]
- Stephens, S.; Agee, J.K.; Fulé, P.; North, M.; Romme, W.; Swetnam, T.; Turner, M.G. Managing forests and fire in changing climates. Science 2013, 342, 41–42. [Google Scholar] [CrossRef] [PubMed]
- Millar, C.I.; Stephenson, N.L.; Stephens, S.L. Climate change and forests of the future: Managing in the face of uncertainty. Ecol. Appl. 2007, 17, 2145–2151. [Google Scholar] [CrossRef] [PubMed]
- Collins, B.M.; Miller, J.D.; Thode, A.E.; Kelly, M.; van Wagtendonk, J.W.; Stephens, S. Interactions among wildland fires in a long-established sierra nevada natural fire area. Ecosystems 2009, 12, 114–128. [Google Scholar] [CrossRef]
- Schwilk, D.W.; Keeley, J.E.; Knapp, E.E.; McIver, J.; Bailey, J.D.; Fettig, C.J.; Fiedler, C.E.; Harrod, R.J.; Moghaddas, J.J.; Outcalt, K.W.; et al. The national fire and fire surrogate study: Effects of fuel reduction methods on forest vegetation structure and fuels. Ecol. Appl. 2009, 19, 285–304. [Google Scholar] [CrossRef] [PubMed]
- Westerling, A.L.; Gershunov, A.; Brown, T.J.; Cayan, D.R.; Dettinger, M.D. Climate and wildfire in the western united states. Bull. Am. Meteorol. Soc. 2003, 84, 595–604. [Google Scholar] [CrossRef]
- Williams, J. Exploring the onset of high-impact mega-fires through a forest land management prism. For. Ecol. Manag. 2013, 294, 4–10. [Google Scholar] [CrossRef]
- Batllori, E.; Parisien, M.; Krawchuk, M.A.; Moritz, M. Climate change-induced shifts in fire for mediterranean ecosystems. Glob. Ecol. Biogeogr. 2013, 22, 1118–1129. [Google Scholar] [CrossRef]
- Stephens, S.L.; Millar, C.I.; Collins, B.M. Operational approaches to managing forests of the future in Mediterranean regions within a context of changing climates. Environ. Res. Lett. 2010, 5, 024003. [Google Scholar] [CrossRef]
- Agee, J.K.; Skinner, C.N. Basic principles of forest fuel reduction treatments. For. Ecol. Manag. 2005, 211, 83–96. [Google Scholar] [CrossRef]
- Oliver, T.H.; Smithers, R.J.; Bailey, S.; Walmsley, C.A.; Watts, K. A decision framework for considering climate change adaptation in biodiversity conservation planning. J. Appl. Ecol. 2012, 49, 1247–1255. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).