Consequences of Shifts in Abundance and Distribution of American Chestnut for Restoration of a Foundation Forest Tree
Abstract
:1. Introduction
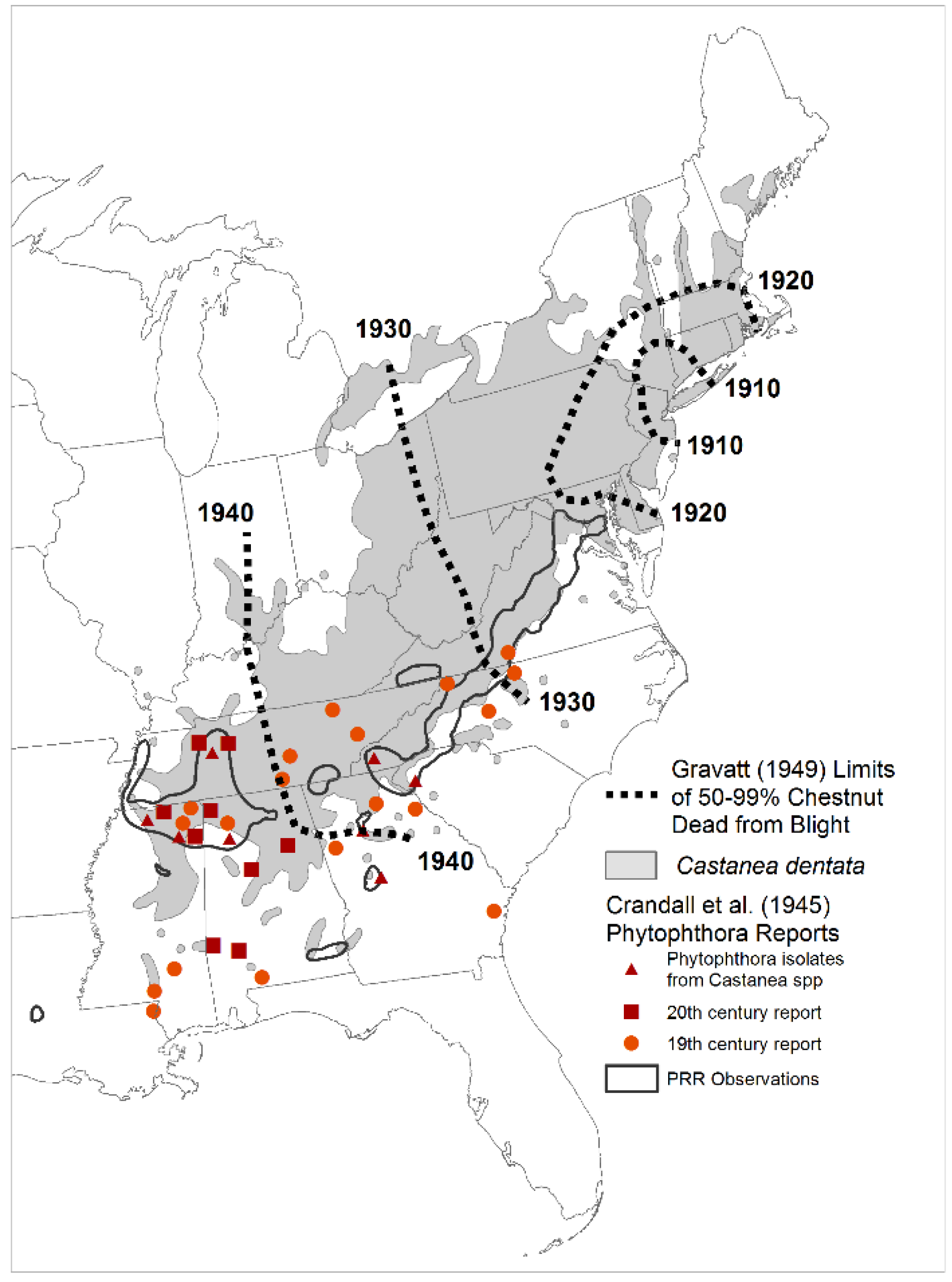
2. Methods

| State | Evaluation Group |
|---|---|
| Alabama | 2006–2012 |
| Arkansas | 2006–2012 |
| Connecticut | 2007–2011 |
| Delaware | 2007–2011 |
| Florida | 2002–2010 |
| Georgia | 2005–2011 |
| Illinois | 2007–2011 |
| Indiana | 2007–2011 |
| Kentucky | 2005–2011 |
| Louisiana | 2001–2009 |
| Maine | 2007–2011 |
| Maryland | 2007–2011 |
| Massachusetts | 2007–2011 |
| Michigan | 2007–2011 |
| Minnesota | 2009–2012 |
| Mississippi | 2006–2012 |
| Missouri | 2007–2011 |
| New Hampshire | 2007–2011 |
| New Jersey | 2007–2011 |
| New York | 2007–2011 |
| North Carolina | 2003–2010 |
| Ohio | 2007–2011 |
| Pennsylvania | 2007–2011 |
| Rhode Island | 2007–2011 |
| South Carolina | 2007–2011 |
| Tennessee | 2005–2011 |
| Vermont | 2007–2011 |
| Virginia | 2008–2011 |
| West Virginia | 2007–2011 |
| Wisconsin | 2008–2012 |
3. Results
4. Discussion
4.1. Density and Distribution of Castanea Dentata
4.2. Climate
4.3. Genetic Diversity
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jackson, S.T.; Hobbs, R.J. Ecological restoration in the light of ecological history. Science 2009, 325, 567–569. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.F.; Oliet, J.A.; Aronson, J.; Bolte, A.; Bullock, J.M.; Donoso, P.J.; Landhäusser, S.M.; Madsen, P.; Peng, S.; Rey-Benayas, J.M.; et al. Restoring forests: What constitutes success in the twenty-first century? New For. 2015, 46, 601–614. [Google Scholar] [CrossRef]
- Ellison, A.M.; Bank, M.S.; Clinton, B.D.; Colburn, E.A.; Elliot, K.; Ford, C.R.; Foster, B.D.; Kloeppel, B.D.; Knoepp, J.D.; Lovett, G.M.; et al. Loss of foundation species: Consequences for the structure and dynamics of forested ecosystems. Front. Ecol. Environ. 2005, 9, 479–486. [Google Scholar] [CrossRef]
- Dalgleish, H.J.; Swihart, R.K. American chestnut past and future: Implications of restoration for resource pulses and consumer populations of eastern U.S. forests. Restor. Ecol. 2012, 20, 490–497. [Google Scholar] [CrossRef]
- Anagnostakis, S.L. Chestnut blight: The classical problem of an introduced pathogen. Mycologia 1987, 79, 23–37. [Google Scholar] [CrossRef]
- Beattie, R.K.; Diller, J.D. Fifty years of chestnut blight in America. J. For. 1954, 52, 323–329. [Google Scholar]
- Paillet, F.L. Chestnut: History and ecology of a transformed species. J. Biogeogr. 2002, 29, 1517–1530. [Google Scholar] [CrossRef]
- Jacobs, D.F.; Dalgleish, H.J.; Nelson, C.D. A conceputal framework for restoration of threatened plants: The effective model of American chestnut (Castanea dentata) restoration. New Phytol. 2013, 197, 378–393. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.F. Toward development of silvical strategies for forest restoration of American chestnut (Castanea dentata) using blight-resistant hybrids. Biol. Conserv. 2007, 137, 497–506. [Google Scholar] [CrossRef]
- McWilliams, W.H.; Lister, T.W.; LaPoint, E.B.; Rose, A.K.; Vissage, J.S. Current status of Chestnut in Eastern U.S. Forests. In Proceedings of Restoration of American Chestnut to Forest Lands, Asheville, NC, USA, 4–6 May 2004; Steiner, K.C., Carlson, J.E., Eds.; Department of the Interior, National Park Service: Washington, DC, USA, 2006; pp. 31–39. [Google Scholar]
- The Forest Inventory and Analysis Database: Database Description and User’s Manual; Forest Inventory and Analysis Program, Version 4.0 for Phase 2, revision 3 DRAFT; Department of Agriculture, Forest Service: Asheville, NC, USA, February 2010.
- Kartesz, J.T. The Biota of North America Program (BONAP); North American Plant Atlas: Chapel Hill, NC, USA, 2013. [Google Scholar]
- Bechtold, W.A.; Patterson, P.L. The Enhanced Forest Inventory and Analysis Program—National Sampling Design and Estimation Procedures; Gen. Tech. Rep. SRS-80; Department of Agriculture, Forest Service, Southern Research Station: Asheville, NC, USA, 2005; p. 85.
- USDA Forest Service. Bailey’s Ecoregions and Subregions of the United States, Puerto Rico, and the US Virgin Islands; National Atlas of the United States: Reston, VA, USA, June 2004.
- Little, E.L., Jr. Atlas of United States Trees; US Department of Agriculture: Washington, DC, USA, 1977.
- Gravatt, F. Chestnut blight in Asia and North America. Unasylva 1949, 3, 2–7. [Google Scholar]
- Wang, G.G.; Bauerle, W.L.; Mudder, B.T. Effects of light acclimation on the photosynthesis, growth, and biomass allocation in American chestnut (Castanea dentata) seedlings. For. Ecol. Manag. 2006, 226, 173–180. [Google Scholar] [CrossRef]
- Joesting, H.M.; McCarthy, B.C.; Brown, K.J. Determining the shade tolerance of American chestnut using morphological and physiological leaf parameters. For. Ecol. Manag. 2009, 257, 280–286. [Google Scholar] [CrossRef]
- Davelos, A.L.; Jarosz, A.M. Demography of American chestnut populations: Effects of a pathogen and a hytperparasite. J. Ecol. 2004, 92, 675–685. [Google Scholar] [CrossRef]
- Russell, E.W.B. Pre-blight distribution of Castanea dentata (Marsh.) Borkh. Bull. Torrey Bot. Club 1987, 114, 183–190. [Google Scholar] [CrossRef]
- Anagnostakis, S. The effect of multiple importations of pests and pathogens on a native tree. Biol. Invasions 2001, 3, 245–254. [Google Scholar] [CrossRef]
- Crandall, B.S.; Gravatt, G.F.; Ryan, M.M. Root diseases of Castanea species and some coniferous and broadleaf nursery stocks, caused by Phytophthora cinnamomi. Phytopathology 1945, 35, 162–180. [Google Scholar]
- Brewer, L.G. Ecology of survival and recovery from blight in American chestnut trees (Castanea dentata (Marsh.) Borkh.) in Michigan. Bull. Torrey Bot. Club 1995, 122, 40–57. [Google Scholar] [CrossRef]
- Paillet, F.L.; Rutter, P.A. Replacement of native oak and hickory tre species by the introduced American chestnut (Castanea dentata) in southwestern Wisconsin. Can. J. Bot. 1989, 67, 3457–3469. [Google Scholar] [CrossRef]
- Woodall, C.W.; Ostwald, C.M.; Westfall, J.A.; Parry, C.H.; Nelson, M.D.; Finley, A.O. An indicator of tree migration in forests of the eastern United States. For. Ecol. Manag. 2009, 257, 1434–1444. [Google Scholar] [CrossRef]
- Zhu, K.; Woodall, C.W.; Clark, J.S. Failure to migrate: Lack of tree range expansion in response to climate change. Glob. Chang. Biol. 2012, 18, 1042–1052. [Google Scholar] [CrossRef]
- Nelson, C.D.; Powell, W.A.; Maynard, C.A.; Baier, K.M.; Newhouse, A.; Merkle, S.A.; Nairn, C.J.; Kong, L.; Carlson, J.E.; Addo, A.G. The Forest Health Initiative, American chestnut (Castanea dentata) as a model for forest tree restoration. Biol. Res. Program Acta Hort 2013, 1019, 179–189. [Google Scholar]
- Fei, S.; Liang, L.; Paillet, F.L.; Steiner, K.C.; Fang, J.; Shen, Z.; Wang, Z.; Hebard, F.V. Modelling chestnut biogeography for American chestnut restoration. Divers. Distrib. 2012, 18, 754–768. [Google Scholar] [CrossRef]
- Wang, G.G.; Knapp, B.O.; Clark, S.L.; Mudder, B.T. The silvics of Castanea dentata (Marsh.) Borkh, American Chestnut, Fagaceae (Beech Family); General Technical Report SRS-173; USDA Forest Service: Asheville, NC, USA, 2013.
- Guerney, K.M.; Schaberg, P.G.; Hawley, G.J.; Shane, J.B. Inadequate cold tolerance as a possible limitation to American chestnut restoration in the northeastern United States. Restor. Ecol. 2011, 19, 55–63. [Google Scholar] [CrossRef]
- Rhoades, C.C.; Brosi, S.L.; Dattilo, A.J.; Vincelli, P. Effect of soil compaction and moisture on incidence of phytophthora root rot on American chestnut (Castanea dentata) seedlings. For. Ecol. Manag. 2003, 184, 47–54. [Google Scholar] [CrossRef]
- Huang, H.W.; Dane, F.; Kubisiak, T.L. Allozyme and RAPD analysis of the genetic diversity and geographic variation in wild populations of the American chestnut (Fagaceae). Am. J. Bot. 1998, 85, 1013–1021. [Google Scholar] [CrossRef] [PubMed]
- Kubisiak, T.L.; Roberds, J.H. Genetic Structure of American Chestnut Populations Based on Neutral DNA Markers. In Proceedings of Restoration of American Chestnut to Forest Lands, Asheville, NC, USA, 4–6 May 2004; Steiner, K.C., Carlson, J.E., Eds.; National Park Service: Asheville, NC, USA, 2006; pp. 109–122. [Google Scholar]
- Dane, F. Comparative phylogeography of Castanea species. Acta Hort 2009, 844, 211–222. [Google Scholar] [CrossRef]
- Li, X.; Dane, F. Comparative chloroplast and nuclear DNA analysis of Castanea species in the southern region of the USA. Tree Genet. Genomes 2013, 9, 107–116. [Google Scholar] [CrossRef]
- Shaw, J.; Craddock, J.H.; Binkley, M.A. Phylogeny and phylogeography of North American Castanea Mill. (Fagaceae) using cpDNA suggests gene sharing in the southern Appalachians (Castanea Mill., Fagaceae). Castanea 2012, 77, 186–211. [Google Scholar] [CrossRef]
- Dane, F.; Lang, P.; Huang, H.; Fu, Y. Intercontinental genetic divergence of Castanea species in eastern Asia and eastern North America. Heredity 2003, 91, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Casasoli, M.; Pot, D.; Plomion, C.; Monteverdi, M.C.; Barreneche, T.; Lauteri, M.; Villani, F. Identificaiton of QTLs affecting adaptive traits in Castanea sativa Mill. Plant Cell Environ. 2004, 27, 1088–1101. [Google Scholar] [CrossRef]
- Soto-Miguez, B.; Fernandez-Lopez, J. Variation in adaptive traits among and within Spanish and European popualtions of Castanea sativa: Selection of trees for timber production. New For. 2015, 46, 23–50. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dalgleish, H.J.; Nelson, C.D.; Scrivani, J.A.; Jacobs, D.F. Consequences of Shifts in Abundance and Distribution of American Chestnut for Restoration of a Foundation Forest Tree. Forests 2016, 7, 4. https://doi.org/10.3390/f7010004
Dalgleish HJ, Nelson CD, Scrivani JA, Jacobs DF. Consequences of Shifts in Abundance and Distribution of American Chestnut for Restoration of a Foundation Forest Tree. Forests. 2016; 7(1):4. https://doi.org/10.3390/f7010004
Chicago/Turabian StyleDalgleish, Harmony J., C. Dana Nelson, John A. Scrivani, and Douglass F. Jacobs. 2016. "Consequences of Shifts in Abundance and Distribution of American Chestnut for Restoration of a Foundation Forest Tree" Forests 7, no. 1: 4. https://doi.org/10.3390/f7010004
APA StyleDalgleish, H. J., Nelson, C. D., Scrivani, J. A., & Jacobs, D. F. (2016). Consequences of Shifts in Abundance and Distribution of American Chestnut for Restoration of a Foundation Forest Tree. Forests, 7(1), 4. https://doi.org/10.3390/f7010004






