Low Nitrogen Retention in Soil and Litter under Conditions without Plants in a Subtropical Pine Plantation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description
| Soil or Litter | C (%) | N (%) | atom % 15N of Total N | NO3− (mg N·kg−1) | NH4− (mg·N·kg−1) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control N Addition | Control N Addition | Control N Addition | Control N Addition | Control N Addition | ||||||
| 0-5 cm soil | 1.83 (0.2) | 1.88 (0.2) | 0.12 (0.0) | 0.12 (0.0) | 0.37 (0.0) | 0.37 (0.0) | 13.7 (2.8) | 10.3 (2.1) | 16.0 (1.7) | 17.3 (4.3) |
| 5–10 cm soil | 0.82 (0.1) | 0.82 (0.1) | 0.07 (0.0) | 0.07 (0.0) | 0.37(0.0) | 0.37 (0.0) | 6.61 (0.8) | 7.05 (1.1) | 9.49 (0.6) | 13.6 (3.3) |
| 10–20 cm soil | 0.61 (0.1) | 0.60 (0.1) | 0.06 (0.0) | 0.06 (0.0) | 0.37 (0.0) | 0.37 (0.0) | 4.92 (0.5) | 4.92 (0.8) | 7.82 (0.4) | 7.78 (0.6) |
| 20–40 cm soil | 0.45 (0.1) | 0.44 (0.0) | 0.04 (0.0) | 0.04 (0.0) | 0.37 (0.0) | 0.37 (0.0) | 2.82 (0.4) | 3.50 (0.5) | 8.94 (2.7) | 7.00 (1.0) |
| P. elliottii litter | 51.2 (0.1) | 0.41 (0.0) | 0.36 (0.0) | 7.9 (2.9 ) | 18.5 (4.1) | |||||
| L. formosana litter | 44.8 (0.2) | 1.13 (0.0) | 0.36 (0.0) | 11.3 (4.6 ) | 21.2 (4.3) | |||||
2.2. Experimental Design
2.3. Sampling, Measurements and Calculations
2.4. Statistical Analyses
3. Results
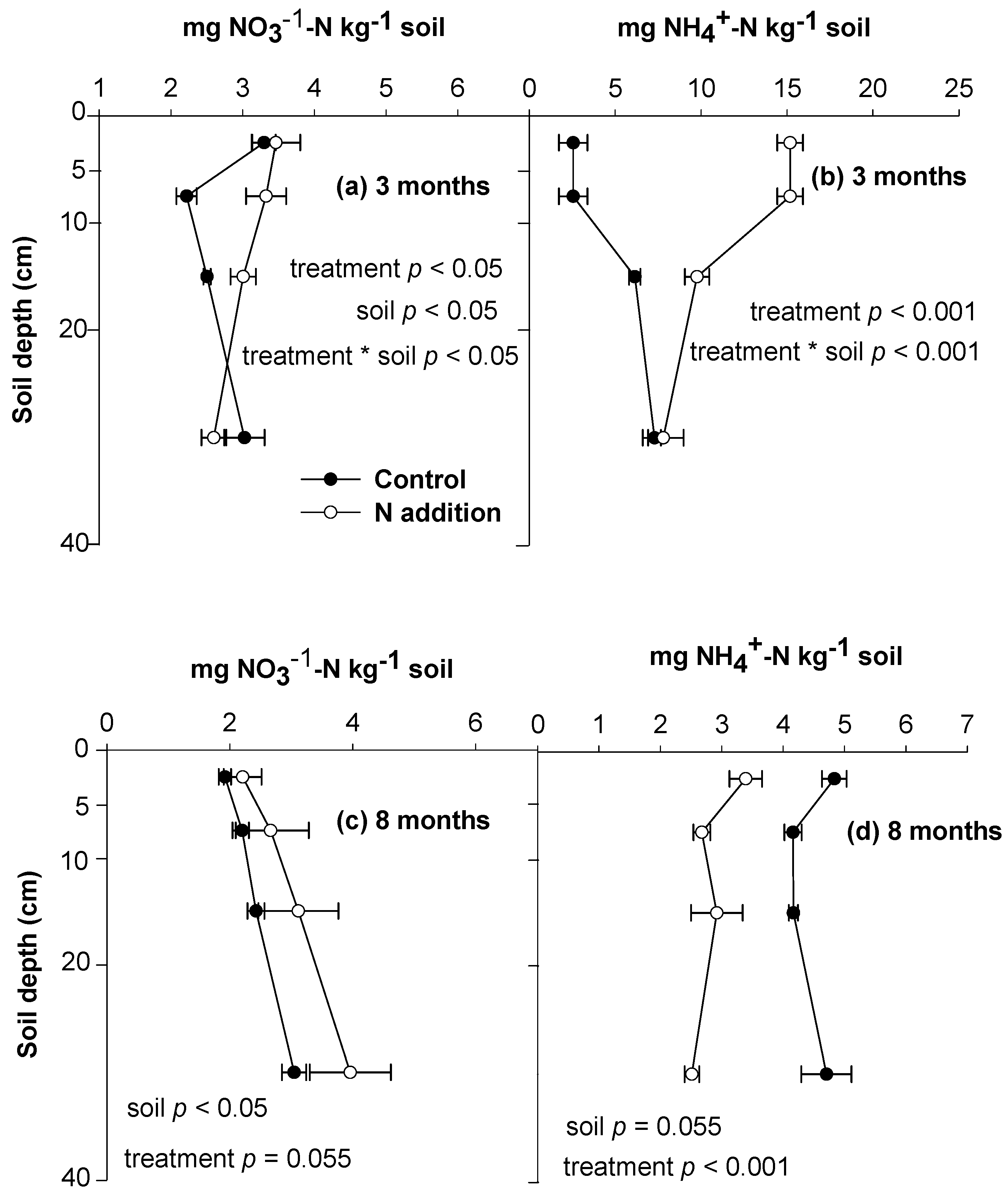
3.1. Soil N Retention and Behaviors of Soil Exogenous and Indigenous N
| Soil Layer | Total N (g·m−2) | Total NH4+ (g·m−2) | Total NO3−(g·m−2) | Atom% 15N of Total N | Atom% 15N of Total NH4+ | Atom% 15N of Total NO3− | Exogenous N (g·m−2) | Exogenous NH4+ (g·m−2) | Exogenous NO3− (g·m−2) | Exogenous Organic N (g·m−2) | Exogenous (NH4++NO3−)/Exogenous N (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0–5 cm | 61.4 (2.9) | 2.00 (0.2) | 0.22 (0.0) | 3.51 (0.3) | 60.1 (2.0) | 19.2 (2.6) | 1.96 (0.2) | 1.23 (0.13) | 0.04 (0.01) | 0.69 (0.1) | 64.8 (4.4) |
| 5–10 cm | 47.0 (2.2) | 1.76 (0.1) | 0.26 (0.0) | 1.91 (0.2) | 27.1 (3.6) | 15.7 (5.1) | 0.74 (0.2) | 0.23 (0.06) | 0.05 (0.02) | 0.46 (0.1) | 37.8 (3.4) |
| 10–20 cm | 62.7 (4.5) | 1.43 (0.1) | 0.67 (0.1) | 1.67 (0.2) | 11.9 (1.7) | 40.9 (3.2) | 0.78 (0.1) | 0.18 (0.04) | 0.28 (0.04) | 0.32 (0.0) | 59.0 (5.6) |
| 20–40 cm | 100.4 (4.0) | 1.85 (0.1) | 1.80 (0.1) | 1.81 (0.1) | 4.91 (0.8) | 62.0 (2.3) | 1.42 (0.1) | 0.09 (0.00) | 1.13 (0.11) | 0.20 (0.0) | 85.9 (2.3) |
| Whole 0–40 cm | 271 (11) | 7.04 (0.4) | 2.95 (0.2 ) | 4.90 (0.3) | 1.73 (0.13 ) | 1.5 ( 0.10) | 1.67 (0.2) | 65.9 (2.4) |
| Soil Layer | Total N (g·m−2) | Total NH4+ (g·m−2) | Total NO3− (g·m−2) | Atom% 15N of Total N | Atom% 15N of Total NH4+ | Atom% 15N of Total NO3− | Exogenous N (g·m−2) | Exogenous NH4+ (g·m−2) | Exogenous NO3− (g·m−2) | Exogenous Organic N (g·m−2) | Exogenous (NH4++NO3−)/Exogenous N (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0–5 cm | 56.7 (3.8) | 0.18 (0.0) | 0.012 (0.00) | 1.33 (0.0) | 1.05 (0.1) | 6.47 (1.0) | 0.54 (0.0) | 0.001 (0.00) | 0.008 (0.00) | 0.53 (0.0) | 1.6 (0.6) |
| 5–10 cm | 46.6 (3.6) | 0.19 (0.0) | 0.019 (0.00) | 0.68 (0.0) | 1.07 (0.2) | 8.24 (1.5) | 0.15 (0.0) | 0.001 (0.00) | 0.017 (0.01) | 0.13 (0.0) | 11.9 (3.8) |
| 10–20 cm | 69.6 (4.1) | 0.38 (0.0) | 0.045 (0.00) | 0.54 (0.0) | 0.76 (0.0) | 10.6 (2.0) | 0.12 (0.0) | 0.002 (0.00) | 0.049 (0.01) | 0.07 (0.0) | 41.8 (8.7) |
| 20–40 cm | 112.2 (9.8) | 1.21 (0.1) | 0.123 (0.03) | 0.63 (0.1) | 1.27 (0.3) | 16.8 (3.2) | 0.30 (0.1) | 0.006 (0.00) | 0.216 (0.05) | 0.07 (0.0) | 73.9 (4.3) |
| Whole 0–40 cm | 285 (17) | 1.96 (0.2) | 0.199 (0.03) | 1.11 (0.2) | 0.010 (0.00) | 0.288 (0.05) | 0.80 (0.0) | 26.9 (6.9) |
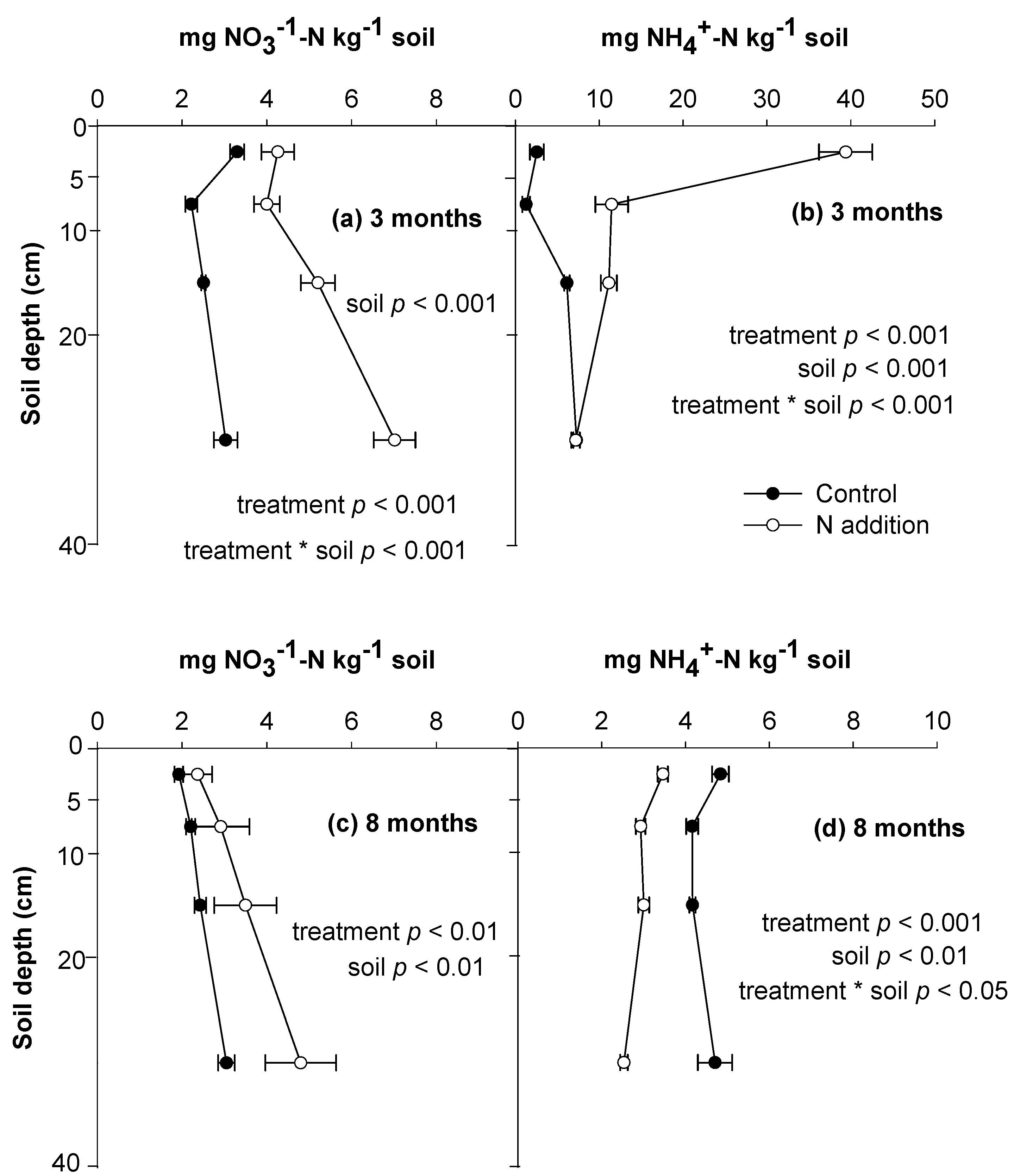

3.2. Litter N Retention and Behaviors of Litter Indigenous and Exogenous N
| Litter Species | Treatments | Initial Exogenous N (g·m−2) | Initial Indigenous N (g·m−2) | Total N Remaining | Atom% 15N of Total N Remaining | Exogenous N Remaining | Indigenous N Remaining | Exogenous N Immobilization | NH4+ (g·m−2) | NO3− (g·m−2) | Atom% 15N of NH4+ | Atom% 15N of NO3− | Exogenous NH4+ (g·m−2) | Exogenous NO3− (g·m−2) | Exogenous (NH4++NO3−)/exogenous N (%) | Indigenous NH4+ (g·m−2) | Indigenous NO3− (g·m−2) | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (g·m−2) (%) | (g·m−2) (%) | (g·m−2) (%) | (g·m−2) (%) | ||||||||||||||||||
| P. elliottii | Control | 0 | 1.84 | 1.85 (0.0) | 101 (1.9) a * | 0.36 (0.0) | 1.85 (0.0) a | 101 (1.9) a | 0.005 (0.00) | 0.001 (0.00) | 0.36 (0.0) | 0.36 (0.0) | 4.56 (0.78) a | 0.53 (0.09) a | |||||||
| N addition | 7.00 | 1.84 | 3.02 (0.1) | 34.2 (1.7) b | 54.9 (2.2) | 1.67 (0.1) | 23.0 (1.5) | 1.35 (0.0) b | 73.4 (3.0) b | 0.95 (0.1) | 13.6 (1.6) | 0.59 (0.05) | 0.24 (0.04) | 84.3 (1.6) | 92.4 (1.0) | 0.496 (0.04) | 0.226 (0.04) | 43.2 (2.7) | 585 (55) b | 242 (43) b | |
| L. formosana | Control | 0 | 5.08 | 5.33 (0.1) | 105 (2.1) a | 0.36 (0.0) | 5.33 (0.1) a | 105 (2.1) a | 0.010 (0.00) | 0.001 (0.00) | 0.36 (0.0) | 0.36 (0.0) | 10.38 (1.21) a | 1.30 (0.13) a | |||||||
| N addition | 7.00 | 5.08 | 5.36 (0.3) | 44.4 (2.5) b | 28.3 (2.2) | 1.50 (0.1) | 20.7 (1.1) | 3.86 (0.2) b | 76.0 (5.1) b | 0.69 (0.12) | 9.8 (1.7) | 0.48 (0.08) | 0.48 (0.10) | 70.3 (1.8) | 96.8 (0.6) | 0.340 (0.06) | 0.472 (0.11) | 54.1 (8.1) | 478 (77) b | 481 (111) b | |
4. Discussion
4.1. Low N Retention in the Soil Profile and Litter
4.2. N Addition Altered the Behaviors of Indigenous N in Soil and Litter
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Galloway, J.N.; Townsend, A.R.; Erisman, J.W.; Bekunda, M.; Cai, Z.C.; Freney, J.R.; Martinelli, L.A.; Seitzinger, S.P.; Sutton, M.A. Transformation of the nitrogen cycle: Recent trends, questions, and potential solutions. Science 2008, 320, 889–892. [Google Scholar] [CrossRef] [PubMed]
- Gruber, N.; Galloway, J.N. An earth-system perspective of the global nitrogen cycle. Nature 2008, 451, 293–296. [Google Scholar] [CrossRef] [PubMed]
- National Atmospheric Deposition Program. National Atmospheric Deposition Program Annual Data Summary: Precipitation Chemistry in the United States; NADP Program Office: Champaign, IL, USA, 2000; Available online: http://nadp.sws.uiuc.edu/lib/data/2000as.pdf (accessed on 1 July 2015).
- Schlesinger, W.H. On the fate of anthropogenic nitrogen. Proc. Natl. Acad. Sci. USA 2009, 106, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Duan, L.; Mo, J.; Du, E.; Shen, J.; Lu, X.; Zhang, Y.; Zhou, X.; He, C.; Zhang, F. Nitrogen deposition and its ecological impact in China: An overview. Environ. Poll. 2011, 159, 2251–2264. [Google Scholar] [CrossRef] [PubMed]
- Vitousek, P.M.; Howarth, R.W. Nitrogen limitation on land and in the sea: How can it occur? Biogeochemistry 1991, 13, 87–115. [Google Scholar] [CrossRef]
- LeBauer, D.S.; Treseder, K.K. Nitrogen limitation of net primary productivity in terrestrial ecosystems is globally distributed. Ecology 2008, 89, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Wanek, W.; Zhou, C.; Richter, A.; Song, M.; Cao, G.; Ouyang, H.; Kuzyakov, Y. Nutrient limitation of alpine plants: Implications from leaf N: P stoichiometry and leaf δ15N. J. Plant Nutr. Soil Sci. 2014, 177, 378–387. [Google Scholar] [CrossRef]
- Xia, J.Y.; Wan, S.Q. Global response patterns of terrestrial plant species to nitrogen addition. N. Phytol. 2008, 179, 428–439. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.Q.; Canham, C.D.; Weathers, K.C.; Goodale, C.L. Increased tree carbon storage in response tonitrogen deposition in the US. Nat. Geosci. 2010, 3, 13–17. [Google Scholar] [CrossRef]
- Aber, J.D.; McDowell, W.H.; Nadelhoffer, K.J. Nitrogen saturation in temperateforest ecosystems—Hypothesis revisited. BioScience 1998, 48, 921–934. [Google Scholar] [CrossRef]
- Gundersen, P.; Emmett, B.A.; Kjønaas, O.J.; Koopmans, C.J.; Tietema, A. Impact of nitrogen deposition on nitrogen cycling in forests: A synthesis of NITREX data. For. Ecol. Manag. 1998, 101, 37–55. [Google Scholar] [CrossRef]
- Fang, Y.; Yoh, M.; Koba, K.; Zhu, W.; Takebayashi, Y.; Xiao, Y.; Lei, C.; Mo, J.; Zhang, W.; Lu, X. Nitrogen deposition and forest nitrogen cycling along anurban-rural transect in southern China. Glob. Chang. Biol. 2011, 17, 872–885. [Google Scholar] [CrossRef]
- Nadelhoffer, K.J.; Downs, M.; Fry, B.; Aber, J.D.; Magill, A.H.; Melillo, J.M. The fate of 15N-labeled nitrate additions to a northern hardwood forest in eastern Maine, USA. Oecologia 1995, 103, 292–301. [Google Scholar] [CrossRef]
- Zak, D.R.; Pregitzer, K.S.; Holmes, W.E.; Burton, A.J.; Zogg, G.P. Anthropogenic N deposition and the fate of 15NO3—In a northern hardwood ecosystem. Biogeochemistry 2004, 69, 143–157. [Google Scholar] [CrossRef]
- Templer, P.H.; Mack, M.C.; Chapin, F.S.; Christenson, L.M; Compton, J.E.; Crook, H.D.; Currie, W.S.; Curtis, C.J.; Dail, D.B.; D’Antonio, C.M.; et al. Sinks for nitrogen inputs in terrestrial ecosystems: A meta-analysis of 15N tracer field studies. Ecology 2012, 93, 1816–1829. [Google Scholar] [CrossRef] [PubMed]
- Nadelhoffer, K.J.; Emmett, B.A.; Gundersen, P.; Kjønaas, O.J.; Koopmans, C.J.; Schleppi, P.; Tietema, A.; Wright, R.F. Nitrogen deposition makes a minor contribution to carbon sequestration in temperate forests. Nature 1999, 398, 145–148. [Google Scholar] [CrossRef]
- Corre, M.D.; Brumme, R.; Veldkamp, E.; Beese, F.O. Changes in nitrogen cycling and retention processes insoils under spruce forests along a nitrogen enrichmentgradient in Germany. Glob. Chang. Biol. 2007, 13, 1509–1527. [Google Scholar] [CrossRef]
- Bryan Dail, D.; Davidson, E.A.; Chorover, J. Rapid abiotictransformation of nitrate in an acid forest soil. Biogeochemistry 2001, 54, 131–146. [Google Scholar] [CrossRef]
- Davidson, E.A.; Chorover, J.; Dail, D.B. A mechanism of abiotic immobilization of nitrate in forest ecosystems: The ferrous wheel hypothesis. Glob. Chang. Biol. 2003, 9, 228–236. [Google Scholar] [CrossRef]
- Zogg, G.P.; Zak, D.R.; Pregitzer, K.S.; Burton, A.J. Microbial immobilization and the retention of anthropogenic nitrate in a northern hardwood forest. Ecology 2000, 81, 1858–1866. [Google Scholar] [CrossRef]
- Morier, I.; Schleppi, P.; Siegwolf, R.; Knicker, H.; Guenat, C. 15N immobilization in forest soil: A sterilization experiment coupled with 15CPMAS NMR spectroscopy. Eur. J. Soil Sci. 2008, 59, 467–475. [Google Scholar] [CrossRef]
- Lewis, D.B.; Castellano, M.J.; Kaye, J.P. Forest succession, soil carbon accumulation, and rapid nitrogen storage in poorly remineralized soil organic matter. Ecology 2014, 95, 3687–2693. [Google Scholar] [CrossRef]
- Nadelhoffer, K.J.; Colman, B.P.; Currie, W.S.; Magill, A.; Aber, J.D. Decadal-scale fates of 15N tracers added to oak and pine stands under ambient and elevated N inputs at the Harvard Forest (USA). For. Ecol. Manag. 2004, 196, 89–107. [Google Scholar] [CrossRef]
- Corre, M.D.; Beese, F.O.; Brumme, R. Soil nitrogen cycle in high nitrogen deposition forest: Changes under nitrogen saturation and liming. Ecol. Appl. 2003, 13, 287–298. [Google Scholar] [CrossRef]
- Vourlitis, G.L.; Zorba, G. Nitrogen and carbon mineralization of semi-arid shrublandsoil exposed to long-term atmospheric nitrogen deposition. Biol. Fertil. Soils 2007, 43, 611–615. [Google Scholar] [CrossRef]
- Lu, M.; Yang, Y.; Luo, Y.; Fang, C.; Zhou, X.; Chen, J.; Yang, X.; Li, B. Responses of ecosystem nitrogen cycle to nitrogen addition: A meta-analysis. N. Phytol. 2011, 189, 1040–1050. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Gunderse, P.; Mo, J.; Zhu, W. Input and output of dissolved organic and inorganic nitrogenin subtropical forests of South China under high air pollution. Biogeosciences 2008, 5, 339–352. [Google Scholar] [CrossRef]
- Jenkinson, D.S.; Fox, R.H.; Rayner, J.H. Interactions between fertilizernitrogen and soil nitrogen: The so-called “priming” effect. J. Soil Sci. 1985, 36, 425–444. [Google Scholar] [CrossRef]
- Woods, L.E.; Cole, L.K.; Porter, L.K.; Coleman, D.C. Transformations of added and indigenousnitrogen in gnotobiotic soil: A comment on the priming effect. Soil Biol. Biochem. 1987, 19, 673–678. [Google Scholar] [CrossRef]
- Azam, F. Added nitrogen interaction in the soil-plant system—A review. Pak. J. Agron. 2002, 1, 54–59. [Google Scholar]
- Wang, Y.; Wang, H.; Ma, Z.; Dai, X.; Wen, X.; Liu, Y.; Wang, Z.L. The litter layer acts as a moisture-induced bidirectional buffer for atmospheric methane uptake by soil of a subtropical pine plantation. Soil Biol. Biochem. 2013, 66, 45–50. [Google Scholar] [CrossRef]
- Huang, K.; Wang, S.; Zhou, L.; Wang, H.; Liu, Y.; Yang, F. Effects of drought and ice rain onpotential productivity of a subtropicalconiferous plantation from 2003 to 2010 based on eddy covariance flux observation. Environ. Res. Lett. 2013, 8, 035021. [Google Scholar] [CrossRef]
- Wen, X.F.; Wang, H.M.; Yu, G.R.; Sun, X.M. Ecosystem carbon exchange of a subtropical evergreen coniferous plantation subjected to seasonal drought, 2003–2007. Biogeosciences 2010, 7, 357–369. [Google Scholar] [CrossRef]
- Lü, C.; Tian, H. Spatial and temporal patterns of nitrogen deposition in China: Synthesis of observational data. J. Geophys. Res. 2007, 112, D22S05. [Google Scholar] [CrossRef]
- Du, E.; Liu, X. High rates of wet nitrogen deposition in China: Asynthesis. In Nitrogen Deposition, Critical Loads and Biodiversity; Sutton, M.A., Mason, K.E., et al., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 49–56. [Google Scholar]
- Stevens, R.J.; Laughlin, R.J. Determining nitrogen-15 in nitrite ornitrate by producing nitrous oxide. Soil Sci. Soc. Am. J. 1994, 58, 1108–1116. [Google Scholar] [CrossRef]
- Laughlin, R.J.; Stevens, R.J.; Zhuo, S. Determining nitrogen-15 inammonium by producing nitrous oxide. Soil Sci. Soc. Am. J. 1997, 61, 462–465. [Google Scholar] [CrossRef]
- Micks, P.; Downs, M.R.; Magill, A.H.; Nadelhoffer, K.J.; Aber, J.D. Decomposing litter as a sink for 15N-enriched additions to an oak forest and a red pine plantation. For. Ecol. Manag. 2004, 196, 71–87. [Google Scholar] [CrossRef]
- Heilman, P.E.; Dao, T.; Cheng, H.H.; Webster, S.R.; Christensen, L. Comparison of fall and spring applications of 15N-labeled urea to Douglas-fir: II. Fertilizer nitrogen recovery in trees and soil after 2 years. Soil Sci. Soc. Am. J. 1982, 46, 1300–1304. [Google Scholar] [CrossRef]
- Nason, G.E. Dynamics of Fertilizer and Native Nitrogen in a Douglas-fir Ecosystem. Ph.D. Thesis, University of Alberta, AB, Canada, 1989. [Google Scholar]
- Tietema, A.; Emmett, B.A.; Gundersen, P.; Kjønaas, O.J.; Koopmans, C.J. The fate of 15N-labelled nitrogen deposition in coniferous forest ecosystems. For. Ecol. Manag. 1998, 101, 19–27. [Google Scholar] [CrossRef]
- Preston, C.M.; Marshall, V.G.; McCullough, K.; Mead, D.J. Fate of 15N-labelled fertilizer applied on snow at two forest sites in British Columbia. Can. J. For. Res. 1990, 20, 1583–1592. [Google Scholar] [CrossRef]
- Mead, D.J.; Chang, S.X.; Preston, C.M. Recovery of 15N-urea 10 years after application to a Douglas-fir pole standin coastal British Columbia. For. Ecol. Manag. 2008, 256, 694–701. [Google Scholar] [CrossRef]
- Perakis, S.S.; Hedin, L.O. Fluxes and fates of nitrogen in soil of an unpolluted old-growth temperate forest, Southern Chile. Ecology 2001, 82, 2245–2260. [Google Scholar] [CrossRef]
- Kuzyakov, Y.; Xu, X. Competition between roots and microorganisms for nitrogen: Mechanisms and ecological relevance. N. Phytol. 2013, 198, 656–669. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Xu, Z.; Blumfield, T.J.; Chen, C.; Bubb, K. Soil nitrogen mineralization and fate of (15NH4)2SO4 in field-incubated soil in a hardwood plantationof subtropical Australia: The effect of mulching. J. Soils Sediments 2008, 8, 389–397. [Google Scholar] [CrossRef]
- Pan, K.; Xu, Z.; Blumfield, T.J.; Tutua, S.; Lu, M. Application of (15NH4)2SO4 to study N dynamics in hooppine plantation and adjacent native forest of subtropicalAustralia: The effects of injection depth and litter addition. J. Soils Sediments 2009, 9, 515–525. [Google Scholar] [CrossRef]
- Buchmann, N.; Gebauer, G.; Schulze, E.D. Partitioning of 15N-labeled ammonium and nitrate among soil, litter, below- and above-ground biomass of trees and understory in a 15-year-old Piceaabies plantation. Biogeochemistry 1996, 33, 1–23. [Google Scholar] [CrossRef]
- Sheng, W.P.; Yu, G.R.; Fang, H.J.; Jiang, C.M.; Yan, J.H.; Zhou, M. Sinks for inorganic nitrogen deposition in forest ecosystems with low and high nitrogen deposition in China. PLoS ONE 2014, 9, e89322. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cheng, S.; Fang, H.; Yu, G.; Xu, M.; Dang, X.; Li, L.; Wang, L. Simulated nitrogen deposition reduces CH4 uptake and increases N2O emission from a subtropical plantation forest soil in southern China. PLoS ONE 2014, 9, e93571. [Google Scholar] [CrossRef] [PubMed]
- Matson, P.A.; McDowell, W.H.; Townsend, A.R.; Vitousek, P.M. The globalization of N deposition: Ecosystem consequences in tropical environments. Biogeochemistry 1999, 46, 67–83. [Google Scholar] [CrossRef]
- Azam, F.; Asharf, M.; Lodhi, A.; Sajjad, M.I. Fate and interaction with native soil N of ammonium N applied to wetland rice (Oryza sativa L.) grown under saline and non-saline conditions. Biol. Fertil. Soils 1992, 13, 102–107. [Google Scholar] [CrossRef]
- Lin, S.; Dittert, K.; Wu, W.L.; Sattelmacher, B. Added nitrogen interaction as affected by soil nitrogen pool size and fertilization-significance of displacement of fixed ammonium. J. Plant Nutr. Soil Sci. 2004, 167, 138–146. [Google Scholar] [CrossRef]
- Ochoa-Hueso, R.; Maestre, F.T.; de los Ríos, A.; Valea, S.; Theobald, M.R.; Vivanco, M.G.; Manrique, E.; Bowker, M.A. Nitrogen deposition alters nitrogen cycling and reduces soil carboncontent in low-productivity semiarid Mediterranean ecosystems. Environ. Poll. 2013, 179, 185–193. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiong, Y.; Xu, X.; Zeng, H.; Wang, H.; Chen, F.; Guo, D. Low Nitrogen Retention in Soil and Litter under Conditions without Plants in a Subtropical Pine Plantation. Forests 2015, 6, 2387-2404. https://doi.org/10.3390/f6072387
Xiong Y, Xu X, Zeng H, Wang H, Chen F, Guo D. Low Nitrogen Retention in Soil and Litter under Conditions without Plants in a Subtropical Pine Plantation. Forests. 2015; 6(7):2387-2404. https://doi.org/10.3390/f6072387
Chicago/Turabian StyleXiong, Yanmei, Xingliang Xu, Hui Zeng, Huimin Wang, Fusheng Chen, and Dali Guo. 2015. "Low Nitrogen Retention in Soil and Litter under Conditions without Plants in a Subtropical Pine Plantation" Forests 6, no. 7: 2387-2404. https://doi.org/10.3390/f6072387
APA StyleXiong, Y., Xu, X., Zeng, H., Wang, H., Chen, F., & Guo, D. (2015). Low Nitrogen Retention in Soil and Litter under Conditions without Plants in a Subtropical Pine Plantation. Forests, 6(7), 2387-2404. https://doi.org/10.3390/f6072387






