Trade-Offs between Drought Survival and Rooting Strategy of Two South American Mediterranean Tree Species: Implications for Dryland Forests Restoration
Abstract
:1. Introduction
2. Material and Methods
2.1. Plant Material
2.2. Site Description
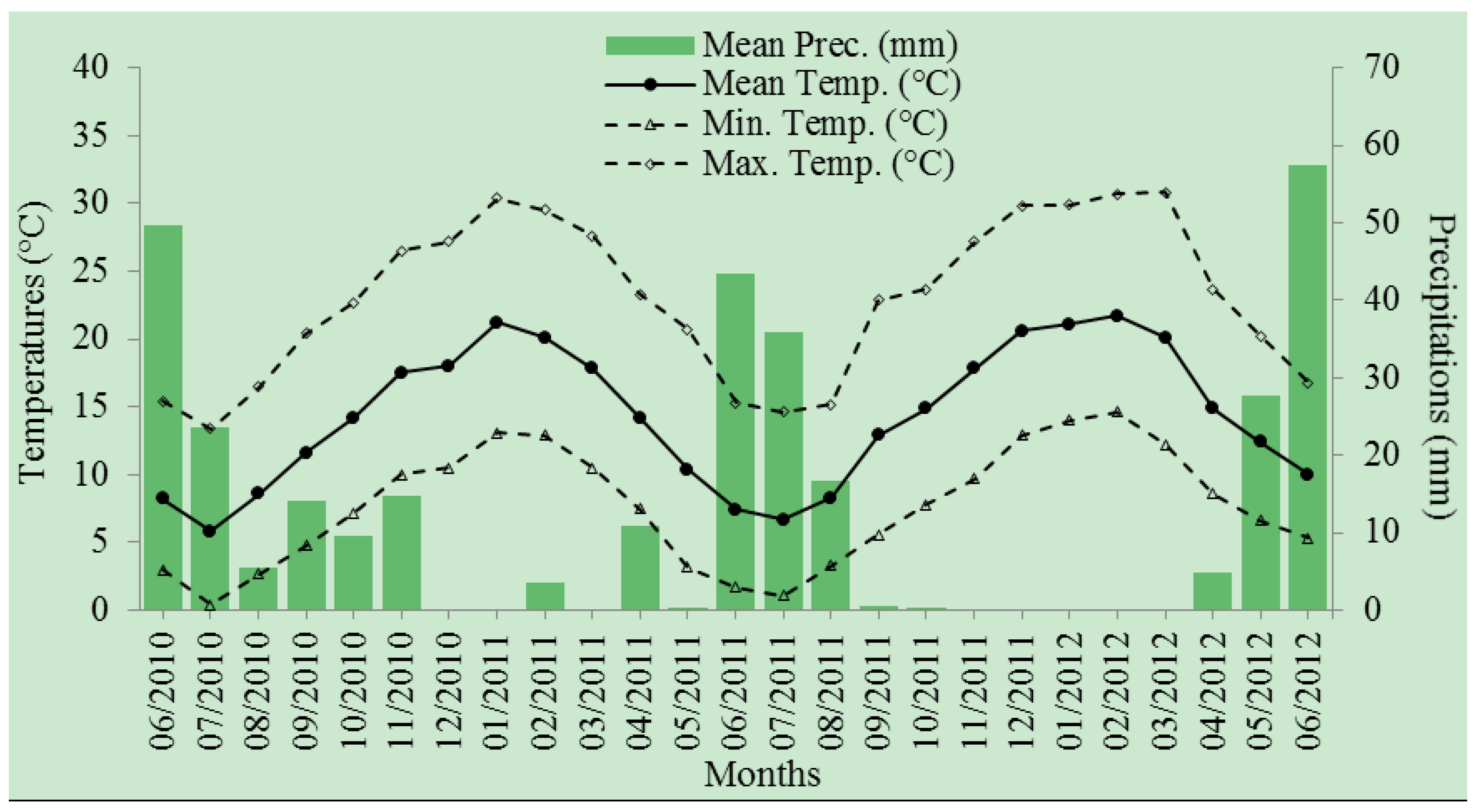
2.3. Experimental Design and Watering Treatments
2.4. Sampling and Measurements
2.5. Statistical Analysis
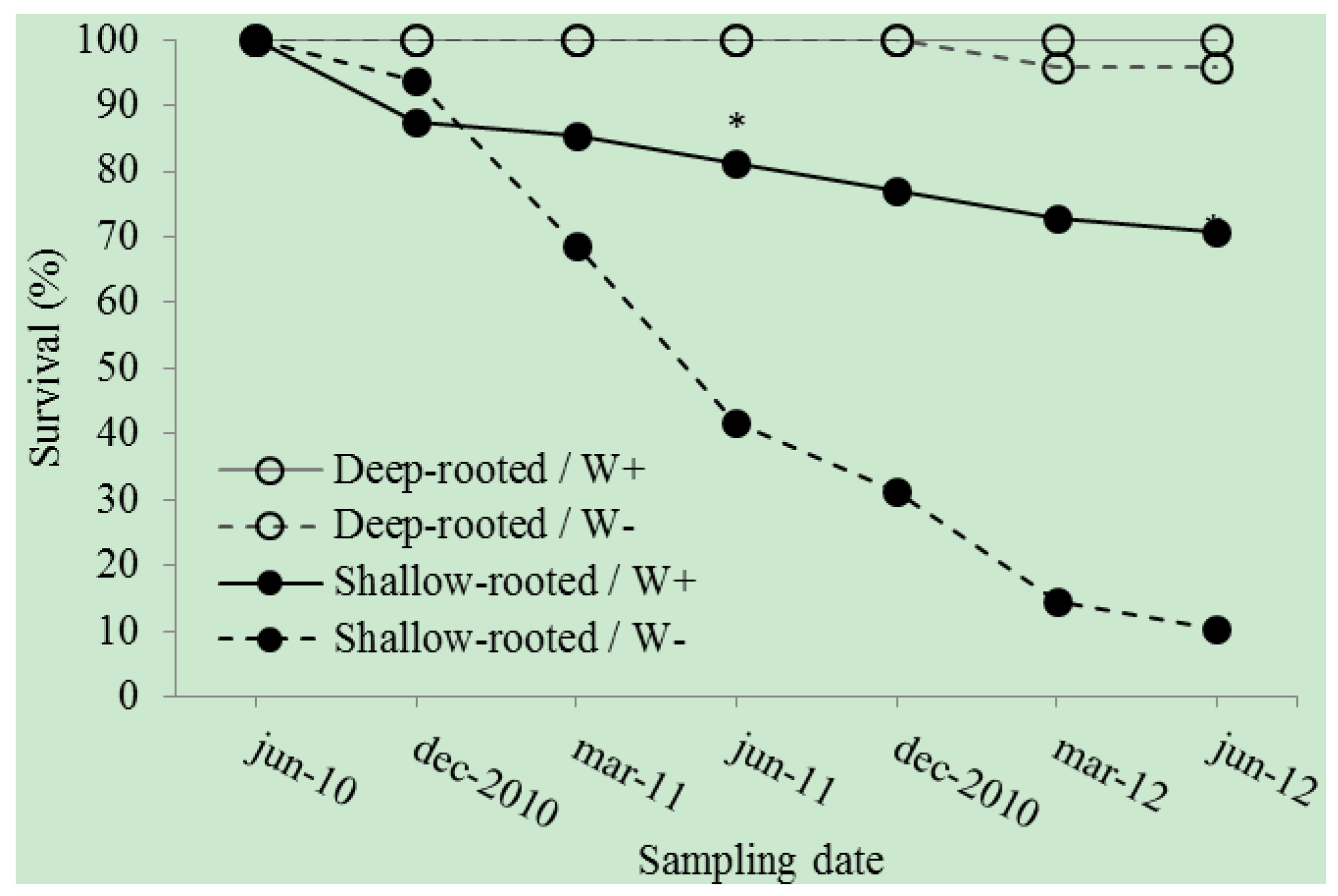
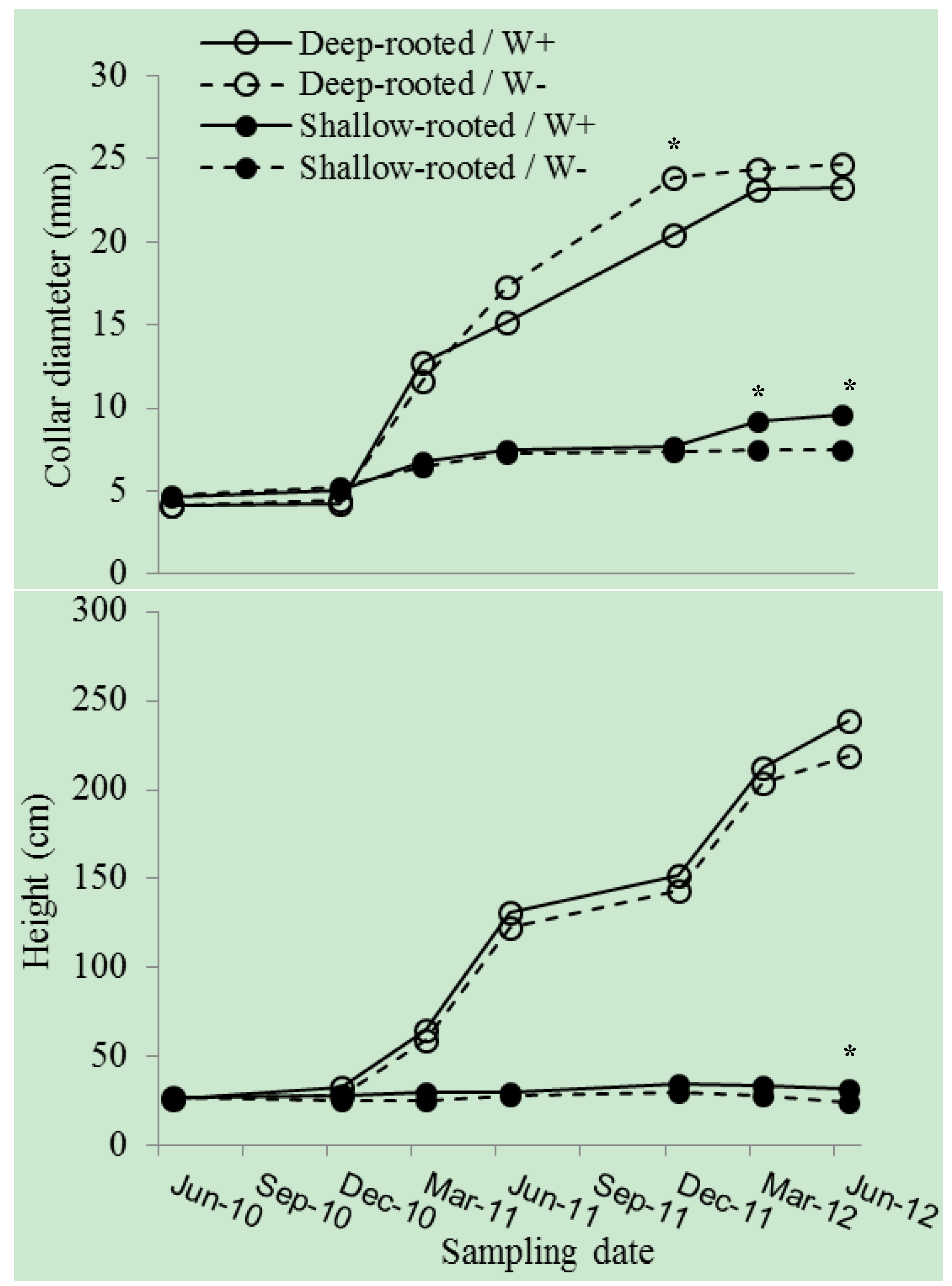
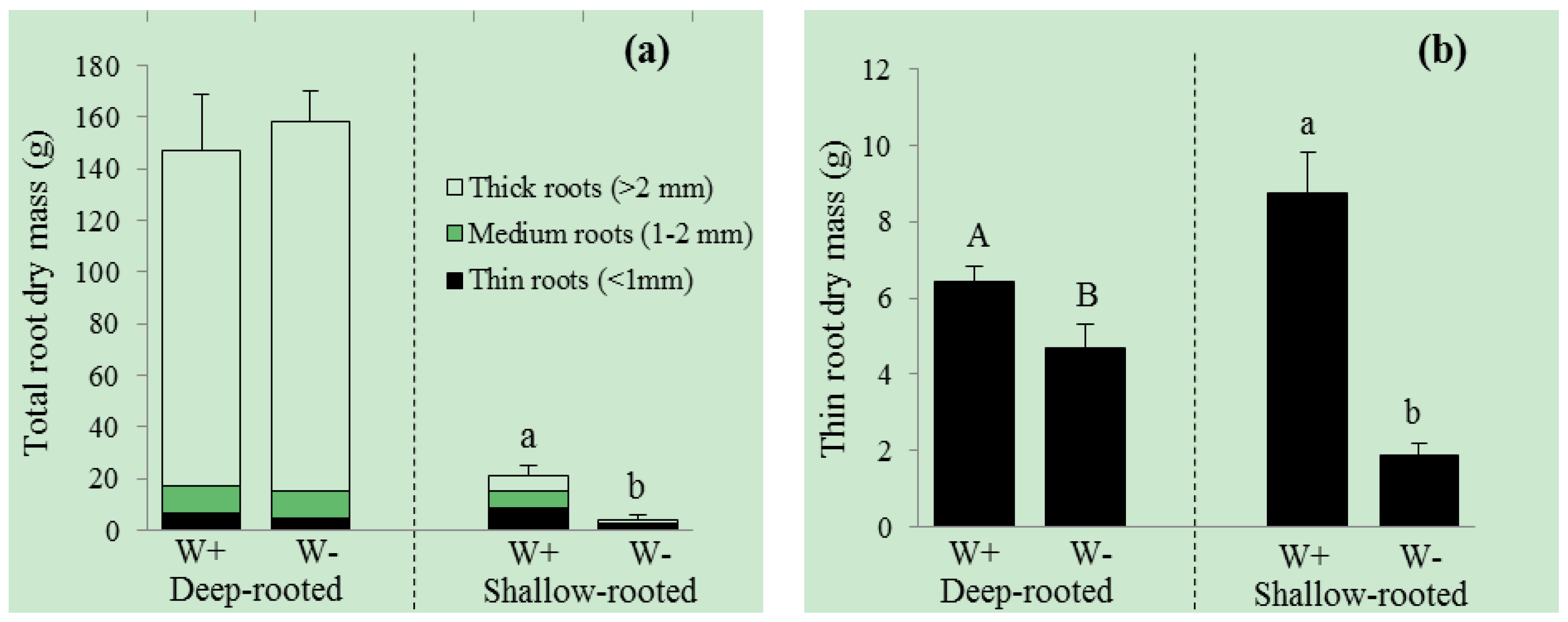
3. Results
| Variables | Deep-rooted | Shallow-rooted | ||||
|---|---|---|---|---|---|---|
| Mean ± SE | p-Value | Mean ± SE | p-Value | |||
| W+ | W− | W+ | W− | |||
| Shoot dry mass (g) | 1427.5 ± 134.1 | 858.6 ± 97.2 | 0.001 | 31.3 ± 5.5 | 4.7 ± 0.3 | <0.001 |
| Total root dry mass (g) | 147.2 ± 21.3 | 158.1 ± 12.0 | 0.055 | 21.4 ± 3.5 | 4.0 ± 2.2 | <0.001 |
| Total root length (m) | 23.4 ± 1.3 | 19.7 ± 0.5 | 0.006 | 23.9 ± 2.9 | 7.8 ± 0.9 | <0.001 |
| Root surface area (cm2) | 4241.4 ± 301.4 | 7462.2 ± 474.4 | <0.001 | 810.1 ± 102.2 | 240.0 ± 31.2 | <0.001 |
| Root volume (cm3) | 1054.7 ± 150.2 | 2343.7 ± 265.3 | <0.001 | 23.1 ± 3.1 | 6.2 ± 0.9 | 0.062 |
| Root diameter (mm) | 7.1 ± 0.7 | 12.1 ± 0.6 | <0.001 | 2.3 ± 0.3 | 1.8 ± 0.3 | 0.083 |
| Shoot/root ratio (g·g−1) | 9.7 ± 0.9 | 5.5 ± 0.3 | 0.042 | 1.9 ± 0.5 | 1.3 ± 0.3 | 0.357 |
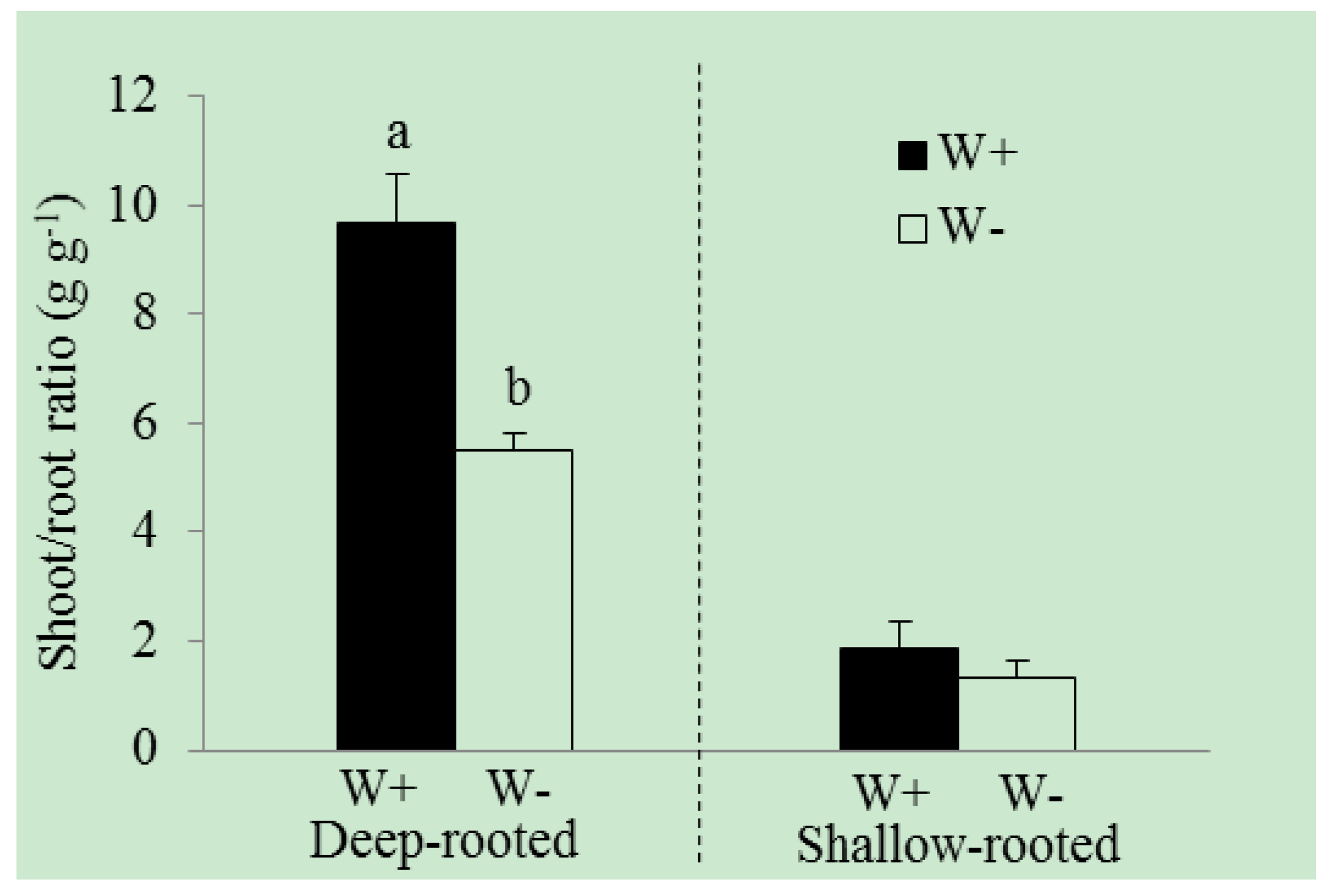
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflict of Interest
References
- Peñuelas, J. Severe drought effects on mediterranean woody. For. Sci. 2001, 47, 214–218. [Google Scholar]
- Vallejo, R.; Smanis, A.; Chirino, E.; Fuentes, D.; Valdecantos, A.; Vilagrosa, A. Perspectives in dryland restoration: Approaches for climate change adaptation. New For. 2012, 43, 561–579. [Google Scholar] [CrossRef]
- Zeppel, M.J.B.; Harrison, S.P.; Adams, H.D.; Kelley, D.I.; Li, G.; Tissue, D.T.; Dawson, T.E.; Fensham, R.; Belinda, E.; Palmer, A.; et al. Drought and resprouting plants. New Phytol. 2015, 206, 583–589. [Google Scholar] [CrossRef]
- Camarero, J.; Franquesa, M.; Sangüesa-Barreda, G. Timing of drought triggers distinct growth responses in Holm Oak: Implications to predict warming-induced forest defoliation and growth decline. Forests 2015, 6, 1576–1597. [Google Scholar] [CrossRef]
- Frazer, J.; Davis, S. Differential survival of chaparral seedlings during the first summer drought after wildfire. Oecologia 1988, 76, 215–221. [Google Scholar] [CrossRef]
- Holmgren, M.; Segura, A.M.; Fuentes, E.R. Limiting mechanisms in the regeneration of the Chilean matorral Experiments on seedling establishment in burned and cleared mesic sites. Plant Ecol. 2000, 147, 49–57. [Google Scholar] [CrossRef]
- Gómez-Aparicio, L.; Pérez-Ramos, I.M.; Mendoza, I.; Matías, L.; Quero, J.L.; Castro, J.; Zamora, R.; Marañón, T. Oak seedling survival and growth along resource gradients in Mediterranean forests: Implications for regeneration in current and future environmental scenarios. Oikos 2008, 117, 1683–1699. [Google Scholar] [CrossRef]
- Becerra, P.I.; González-Rodríguez, V.; Smith-Ramírez, C.; Armesto, J.J. Spatio-temporal variation in the effect of herbaceous layer on woody seedling survival in a Chilean mediterranean ecosystem. J. Veg. Sci. 2011, 22, 847–855. [Google Scholar] [CrossRef]
- Benigno, S.M.; Dixon, K.W.; Stevens, J.C. Seedling mortality during biphasic drought in sandy Mediterranean soils. Funct. Plant Biol. 2014, 41, 1239–1248. [Google Scholar] [CrossRef]
- Morales, N.; Becerra, P.; Arellano, E.; Gilabert, H. Effect of large and small herbivores on seed and seedling survival of Beilschmiedia miersii in central Chile. Bosque (Valdivia) 2015, 36, 127–132. [Google Scholar]
- Lo Gullo, M.A.; Salleo, S. Different strategies of drought resistance in three Mediterranean sclerophyllous trees growing in the same environmental conditions. New Phytol. 1988, 108, 267–276. [Google Scholar] [CrossRef]
- Valladares, F.; Sánchez-Gómez, D. Ecophysiological traits associated with drought in Mediterranean tree seedlings: Individual responses versus interspecific trends in eleven species. Plant Biol. 2006, 8, 688–697. [Google Scholar] [CrossRef] [PubMed]
- West, A.G.; Dawson, T.E.; February, E.C.; Midgley, G.F.; Bond, W.J.; Aston, T.L. Diverse functional responses to drought in a Mediterranean-type shrubland in South Africa. New Phytol. 2012, 195, 396–407. [Google Scholar] [CrossRef] [PubMed]
- Aguadé, D.; Poyatos, R.; Rosas, T.; Martínez-vilalta, J. Comparative drought responses of Quercus ilex L. and Pinus sylvestris L. in a montane forest undergoing a vegetation shift. Forests 2015, 6, 2505–2529. [Google Scholar] [CrossRef]
- Valladares, F.; Pearcy, R.W. Interactions between water stress, sun-shade acclimation, heat tolerance and photoinhibition in the sclerophyll Heteromeles arbutifolia. Plant Cell Environ. 1997, 20, 25–36. [Google Scholar] [CrossRef]
- Vilagrosa, A.; Bellot, J.; Vallejo, V.R.; Gil-Pelegrin, E. Cavitation, stomatal conductance, and leaf dieback in seedlings of two co-occurring Mediterranean shrubs during an intense drought. J. Exp. Bot. 2003, 54, 2015–2024. [Google Scholar] [CrossRef] [PubMed]
- Lloret, F.; Casanovas, C.; Peñuelas, J. Seedling survival of Mediterranean shrubland species in relation to root:shoot ratio, seed size and water and nitrogen use. Funct. Ecol. 1999, 13, 210–216. [Google Scholar] [CrossRef]
- Padilla, F.M.; Pugnaire, F.I. Rooting depth and soil moisture control Mediterranean woody seedling survival during drought. Funct. Ecol. 2007, 21, 489–495. [Google Scholar] [CrossRef]
- Markesteijn, L.; Poorter, L. Seedling root morphology and biomass allocation of 62 tropical tree species in relation to drought- and shade-tolerance. J. Ecol. 2009, 97, 311–325. [Google Scholar] [CrossRef]
- Paula, S.; Pausas, J.G. Root traits explain different foraging strategies between resprouting life histories. Oecologia 2011, 165, 321–331. [Google Scholar] [CrossRef] [PubMed]
- León, M.F.; Squeo, F.A.; Gutiérrez, J.R.; Holmgren, M. Rapid root extension during water pulses enhances establishment of shrub seedlings in the Atacama Desert. J. Veg. Sci. 2011, 22, 120–129. [Google Scholar] [CrossRef]
- Villar-Salvador, P.; Puértolas, J.; Cuesta, B.; Peñuelas, J.L.; Uscola, M.; Heredia-Guerrero, N.; Benayas, J.M.R. Increase in size and nitrogen concentration enhances seedling survival in Mediterranean plantations. Insights from an ecophysiological conceptual model of plant survival. New For. 2012, 43, 755–770. [Google Scholar] [CrossRef]
- Padilla, F.M.; Miranda, J.D.D.; Pugnaire, F.I. Early root growth plasticity in seedlings of three Mediterranean woody species. Plant Soil 2007, 296, 103–113. [Google Scholar] [CrossRef]
- Padilla, F.M.; Miranda, J.D.; Jorquera, M.J.; Pugnaire, F.I. Variability in amount and frequency of water supply affects roots but not growth of arid shrubs. Plant Ecol. 2009, 204, 261–270. [Google Scholar] [CrossRef]
- Comas, L.H.; Becker, S.R.; Cruz, V.M.V; Byrne, P.F.; Dierig, D. Root traits contributing to plant productivity under drought. Front. Plant Sci. 2013, 4, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Olmo, M.; Lopez-Iglesias, B.; Villar, R. Drought changes the structure and elemental composition of very fine roots in seedlings of ten woody tree species. Implications for a drier climate. Plant Soil 2014, 384, 113–129. [Google Scholar] [CrossRef]
- Giliberto, J.; Estay, H. Seasonal water stress in some chilean matorral shrubs. Bot. Gaz. 1978, 139, 236–240. [Google Scholar] [CrossRef]
- Peñuelas, J.; Filella, I. Deuterium labelling of roots provides evidence of deep water access and hydraulic lift by Pinus nigra in a Mediterranean forest of NE Spain. Environ. Exp. Bot. 2003, 49, 201–208. [Google Scholar] [CrossRef]
- Armas, C.; Padilla, F.M.; Pugnaire, F.I.; Jackson, R.B. Hydraulic lift and tolerance to salinity of semiarid species: Consequences for species interactions. Oecologia 2010, 162, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Torres, R.; Squeo, F.; Jorquera, C.; Aguirre, E.; Ehleringer, J.R. Evaluación de la capacidad estacional de utilizar eventos de precipitación en tres especies de arbustos nativos de Chile con distintos sistemas radiculares. Rev. Chil. Hist. Nat. 2002, 75, 737–749. [Google Scholar] [CrossRef]
- Lynch, J.P. Steep, cheap and deep: An ideotype to optimize water and N acquisition by maize root systems. Ann. Bot. 2013, 112, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Canadell, J.; Zedler, P. Underground structures of woody plants in Mediterranean ecosystems of Australia, California, and Chile. In Ecology and Biogeography of Mediterranean Ecosystems in Chile, California, and Australia; Arroyo, M., Zedler, P., Fox, M., Eds.; Springer: Berlin, Germany, 1995; pp. 177–210. [Google Scholar]
- Johnson, D.M.; Brodersen, C.R.; Reed, M.; Domec, J.-C.; Jackson, R.B. Contrasting hydraulic architecture and function in deep and shallow roots of tree species from a semi-arid habitat. Ann. Bot. 2014, 113, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Poorter, H.; Ryser, P. The limits to leaf and root plasticity: What is so special about specific root length? New Phytol. 2015, 206, 1188–1190. [Google Scholar] [CrossRef] [PubMed]
- Villar-Salvador, P.; Planelles, R.; Oliet, J.; Peñuelas-Rubira, J.L.; Jacobs, D.F.; González, M. Drought tolerance and transplanting performance of holm oak (Quercus ilex) seedlings after drought hardening in the nursery. Tree Physiol. 2004, 24, 1147–1155. [Google Scholar] [CrossRef] [PubMed]
- Chirino, E.; Vilagrosa, A.; Hernández, E.; Matos, A.; Vallejo, R. Effects of a deep container on morpho-functional characteristics and root colonization in Quercus suber L. seedlings for reforestation in Mediterranean climate. For. Ecol. Manag. 2008, 256, 779–785. [Google Scholar] [CrossRef]
- Jacobs, D.F.; Salifu, K.F.; Davis, A.S. Drought susceptibility and recovery of transplanted Quercus rubra seedlings in relation to root system morphology. Ann. For. Sci. 2009, 66, 1–12. [Google Scholar] [CrossRef]
- Andivia, E.; Fernández, M.; Vázquez-piqué, J. Assessing the effect of late-season fertilization on Holm oak plant quality: Insights from morpho-nutritional characterizations and water relations parameters. New For. 2014, 45, 149–163. [Google Scholar] [CrossRef]
- Clark, L.J.; Whalley, W.R.; Barraclough, P.B. How do roots penetrate strong soil? Plant Soil 2003, 255, 93–104. [Google Scholar] [CrossRef]
- Whitmore, A.P.; Whalley, W.R. Physical effects of soil drying on roots and crop growth. J. Exp. Bot. 2009, 60, 2845–2857. [Google Scholar] [CrossRef] [PubMed]
- Gaul, D.; Hertel, D.; Borken, W.; Matzner, E.; Leuschner, C. Effects of experimental drought on the fine root system of mature Norway spruce. For. Ecol. Manag. 2008, 256, 1151–1159. [Google Scholar] [CrossRef]
- Bengough, G.; McKenzie, B.M.; Hallett, P.D.; Valentine, T. Root elongation, water stress, and mechanical impedance: A review of limiting stresses and beneficial root tip traits. J. Exp. Bot. 2011, 62, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Stocker, T.; Qin, D.; Plattner, G.; Tignor, M.; Simon, A.; Boschung, J.; Navels, A.; Xia, Y.; Bex, V.; Migley, P. The Physical Science Basis. In Working Group I Contribution to the Fifth Assessment Panel on Climate Change. Summary for Policymarkes; IPCC: Geneva, Switzerland, 2013; pp. 33–115. [Google Scholar]
- Padilla, F.; Miranda, J.D.D.; Armas, C.; Pugnaire, F. Effects of changes in rainfall amount and pattern on root dynamics in an arid shrubland. J. Arid Environ. 2015, 114, 49–53. [Google Scholar] [CrossRef]
- Hertel, D.; Strecker, T.; Müller-Haubold, H.; Leuschner, C. Fine root biomass and dynamics in beech forests across a precipitation gradient—Is optimal resource partitioning theory applicable to water-limited mature trees? J. Ecol. 2013, 101, 1183–1200. [Google Scholar] [CrossRef]
- Brunner, I.; Pannatier, E.G.; Frey, B.; Rigling, A.; Landolt, W.; Zimmermann, S.; Dobbertin, M. Morphological and physiological responses of Scots pine fine roots to water supply in a dry climatic region in Switzerland. Tree Physiol. 2009, 29, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Benayas, J.M.R.; Camacho-Cruz, A. Performance of Quercus ilex saplings planted in abandoned Mediterranean cropland after long-term interruption of their management. For. Ecol. Manag. 2004, 194, 223–233. [Google Scholar] [CrossRef]
- Valdecantos, A.; Fuentes, D.; Smanis, A.; Llovet, J.; Morcillo, L.; Bautista, S. Effectiveness of low-cost planting techniques for improving water availability to Olea europaea seedlings in degraded drylands. Restor. Ecol. 2014, 22, 327–335. [Google Scholar] [CrossRef]
- Holmgren, M.; López, B.; Gutiérrez, J. Herbivory and plant growth rate determine the success of El Nino Southern Oscillation-driven tree establishment in semiarid South America. Glob. Chang. Biol. 2006, 12, 2263–2271. [Google Scholar] [CrossRef]
- Donoso, C. Reseña ecológica de los bosques Mediterráneos de Chile. Bosque (Valdivia) 1982, 4, 117–146. [Google Scholar]
- Becerra, P.I.; Cruz, G.; Ríos, S.; Castelli, G. Importance of irrigation and plant size in the establishment success of different native species in a degraded ecosystem of central Chile. Bosque (Valdivia) 2013, 34, 23–24. [Google Scholar] [CrossRef]
- Hoffmann, A.; Kummerow, J. Root Studies in the Chilean Matorral. Oecologia 1978, 69, 57–69. [Google Scholar] [CrossRef]
- Pérez-Harguindeguy, N.; Garnier, E.; Lavorel, S.; Poorter, H.; Jaureguiberry, P.; Cornwell, W.K.; Craine, J.M.; Gurvich, D.E.; Urcelay, C.; Veneklaas, E.J.; et al. New handbook for standardised measurement of plant functional traits worldwide. Aust. J. Bot. 2013, 61, 167–234. [Google Scholar] [CrossRef]
- Di Castri, F.; Hajek, E. Bioclimatología de Chile; Di Castri, F., Hajek, E., Eds.; Vicerrectoría UC: Santiago, Chile, 1976. [Google Scholar]
- Valenzuela, L. Evaluación de un ensayo de riego y fertilización de quillay (Quillaja saponaria Mol.) en la comuna de San Pedro, provincia de Melipilla, Región Metropolitana. Undergraduate’ Thesis, Universidad de Chile, Santiago, Chile, 2007; p. 66. [Google Scholar]
- Efron, B. Bootstrap methods: Another look at the Jackknife. Ann. Stat. 1979, 7, 1–26. [Google Scholar] [CrossRef]
- Ostonen, I.; Püttsepp, Ü.; Biel, C.; Alberton, O.; Bakker, M.; Lohmus, K.; Majdi, H.; Metcalfe, D.; Olsthoorn, A.; Pronk, A.; et al. Specific root length as an indicator of environmental change. Plant Biosyst. 2007, 141, 426–442. [Google Scholar] [CrossRef]
- Donoso, S.; Peña, K.; Pacheco, C.; Luna, G.; Aguirre, A. Physiological and growth response in Quillaja saponaria and Cryptocarya alba plants under restricted water conditions. Bosque (Valdivia) 2011, 32, 187–195. [Google Scholar] [CrossRef]
- Zida, D.; Tigabu, M.; Sawadogo, L.; Odén, P.C. Initial seedling morphological characteristics and field performance of two Sudanian savanna species in relation to nursery production period and watering regimes. For. Ecol. Manag. 2008, 255, 2151–2162. [Google Scholar] [CrossRef]
- Bloom, A.J.; Chapin, F.S.; Mooney, H. Resource limitation in plants—An economic analogy. Ann. Rev. Ecol. Syst. 1985, 16, 363–392. [Google Scholar] [CrossRef]
- Hernández, E.; Vilagrosa, A.; Luis, V.C.; Llorca, M.; Chirino, E.; Vallejo, V.R. Root hydraulic conductance, gas exchange and leaf water potential in seedlings of Pistacia lentiscus L. and Quercus suber L. grown under different fertilization and light regimes. Environ. Exp. Bot. 2009, 67, 269–276. [Google Scholar] [CrossRef]
- Jin, K.; Shen, J.; Ashton, R.W.; White, R.P.; Dodd, I.C.; Parry, M.A.J.; Whalley, W.R. Wheat root growth responses to horizontal stratification of fertiliser in a water-limited environment. Plant Soil 2015, 386, 77–88. [Google Scholar] [CrossRef]
- Meier, I.C.; Leuschner, C. Genotypic variation and phenotypic plasticity in the drought response of fine roots of European beech. Tree Physiol. 2008, 28, 297–309. [Google Scholar] [CrossRef]
- Lo Gullo, M.; Salleo, S.; Rosso, R.; Trifilò, P. Drought resistance of 2-year-old saplings of Mediterranean forest trees in the field: Relations between water relations, hydraulics and productivity. Plant Soil 2003, 250, 259–272. [Google Scholar] [CrossRef]
- Cowling, R.M.; Rundel, P.W.; Lamont, B.B.; Arroyo, M.K.; Arianoutsou, M. Plant diversity in Mediterranean-climate regions. Trends Ecol. Evol. 1996, 11, 362–366. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ovalle, J.F.; Arellano, E.C.; Ginocchio, R. Trade-Offs between Drought Survival and Rooting Strategy of Two South American Mediterranean Tree Species: Implications for Dryland Forests Restoration. Forests 2015, 6, 3733-3747. https://doi.org/10.3390/f6103733
Ovalle JF, Arellano EC, Ginocchio R. Trade-Offs between Drought Survival and Rooting Strategy of Two South American Mediterranean Tree Species: Implications for Dryland Forests Restoration. Forests. 2015; 6(10):3733-3747. https://doi.org/10.3390/f6103733
Chicago/Turabian StyleOvalle, Juan F., Eduardo C. Arellano, and Rosanna Ginocchio. 2015. "Trade-Offs between Drought Survival and Rooting Strategy of Two South American Mediterranean Tree Species: Implications for Dryland Forests Restoration" Forests 6, no. 10: 3733-3747. https://doi.org/10.3390/f6103733






