Late-Successional and Old-Growth Forests in the Northeastern United States: Structure, Dynamics, and Prospects for Restoration
Abstract
:1. Introduction
- To quantify existing structure and structural change in a series of remeasured late-successional and old-growth forest plots from northern Maine, USA, with a focus on stocking levels, diameter distribution, and structural legacies including large live trees and standing and downed dead wood.
- To put these intensively studied plots in a regional context (encompassing northern New England and New York), by examining the abundance, development, and harvesting patterns of late-successional and old-growth plots from the U.S. Forest Service, Forest Inventory and Analysis program.
- To suggest both silvicultural strategies and policy directions to enhance the prospect for the restoration of late-successional and old-growth structures in forests of the northeastern United States.
2. Experimental Section
2.1. Intensive Plot Data and Analysis




2.2. Regional Analysis
3. Results and Discussion
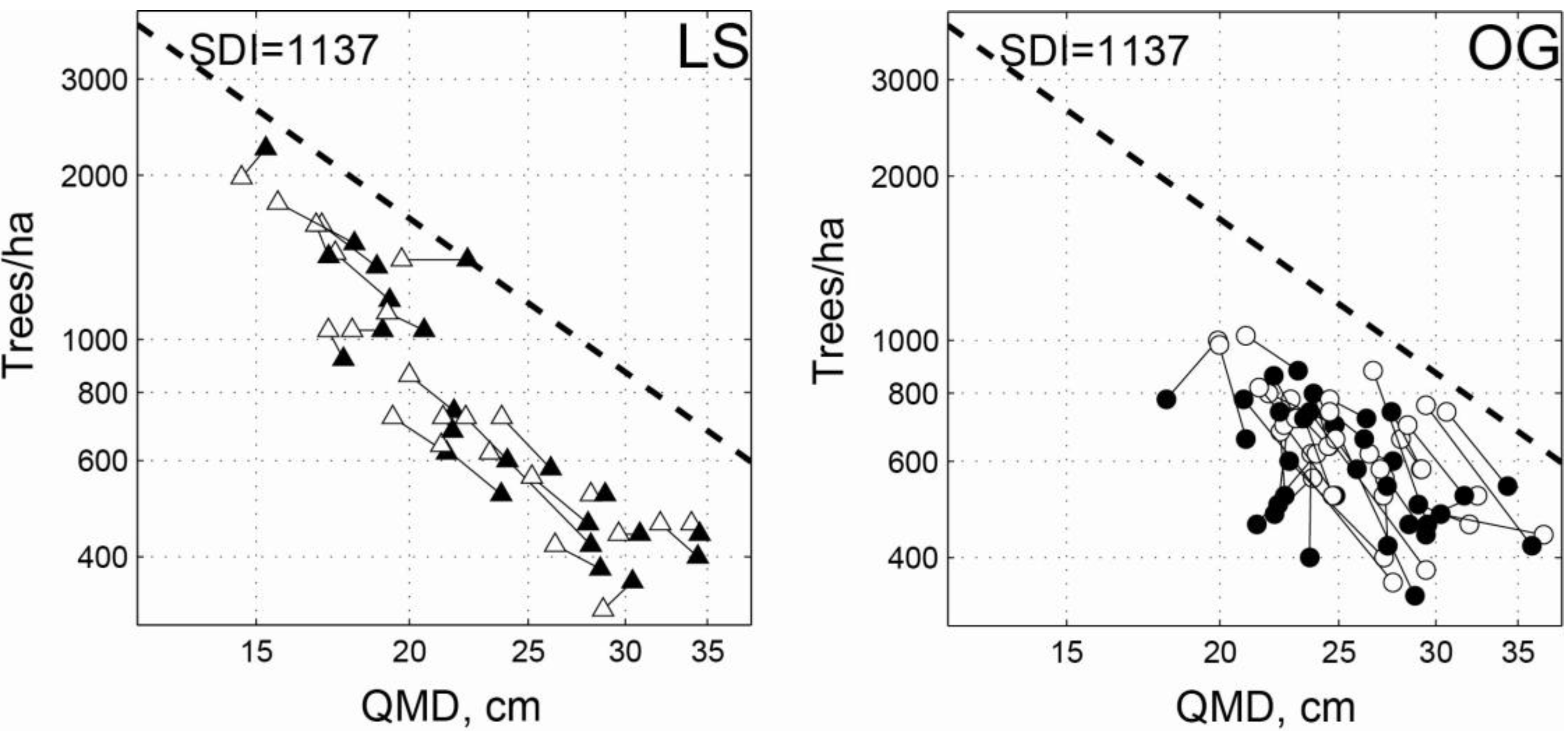
3.1. Intensive Study Plots
| Stand Type | Period | Basal Area, m2/ha | Trees/ha | QMD, cm | SWARD | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Min | Max | Mean | Min | Max | Mean | Min | Max | Mean | Min | Max | ||
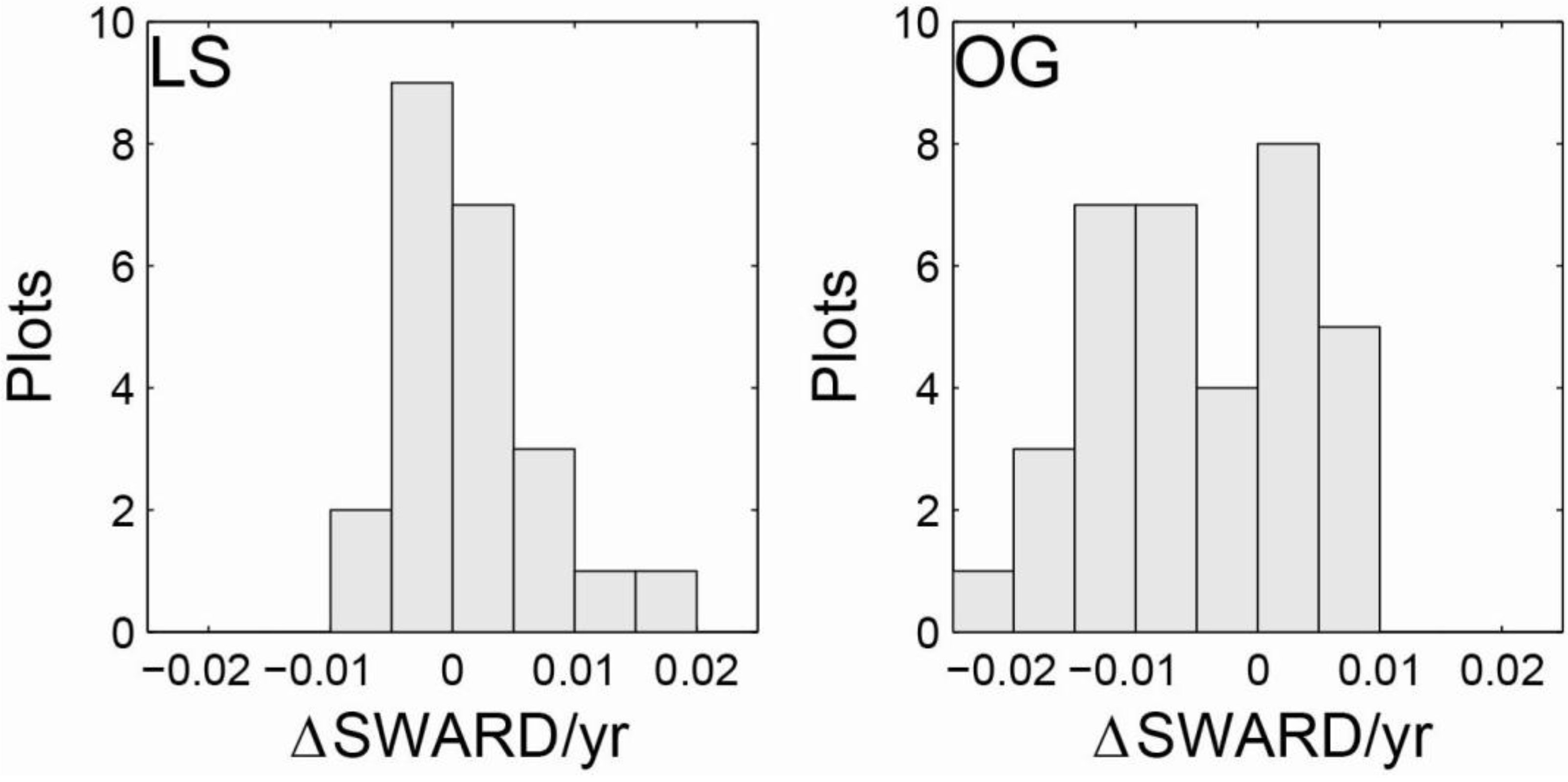
| LS | Initial | 30.2 | 20.8 | 42.7 | 923 | 320 | 1,980 | 22.2 | 14.6 | 34.0 | 0.706 | 0.437 | 0.947 |
| 2011 | 31.8 | 22.4 | 54.7 | 837 | 360 | 2,240 | 23.9 | 15.3 | 34.5 | 0.721 | 0.536 | 1.034 | |
| OG | Initial | 33.8 | 21.7 | 54.5 | 661 | 360 | 1,020 | 25.9 | 19.9 | 36.8 | 0.718 | 0.354 | 1.167 |
| 2011 | 30.6 | 16.6 | 49.9 | 596 | 340 | 880 | 25.8 | 18.1 | 35.9 | 0.635 | 0.405 | 1.026 | |
| Large Live Trees | Large Standing Dead | Large Downed Dead | |||||
|---|---|---|---|---|---|---|---|
| Stand Type | Period | Trees/ha | Basal Area | Trees/ha | Basal Area | Pieces/ha | Volume |
| LS | Initial | 29.6 ± 8.2 | 5.4 ± 1.6 | 1.7 ± 1.2 | 0.3 ± 0.2 | 7.3 ± 1.7 | 12.1 ± 3.3 |
| 2011 | 38.3 ± 9.1 | 6.8 ± 1.8 | 3.5 ± 2.0 | 0.6 ± 0.3 | 7.6 ± 1.6 | 11.5 ± 4.1 | |
| OG | Initial | 66.3 ± 6.1 | 13.7 ± 1.4 | 4.0 ± 1.6 | 0.8 ± 0.3 | - | - |
| 2011 | 72.6 ± 5.9 | 15.3 ± 1.3 | 2.3 ± 1.4 | 0.5 ± 0.3 | 10.7 ± 2.0 | 18.0 ± 3.4 | |
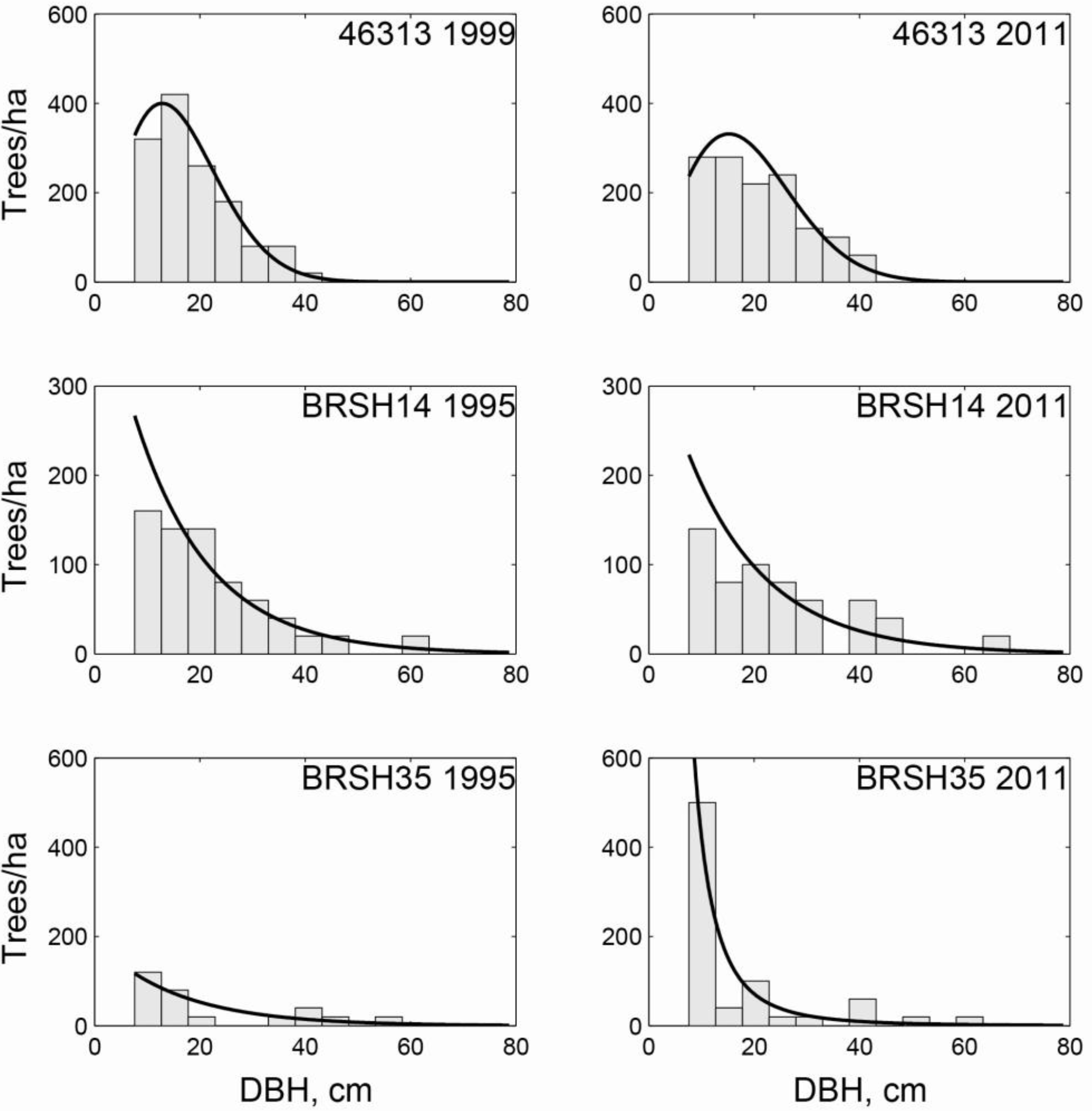
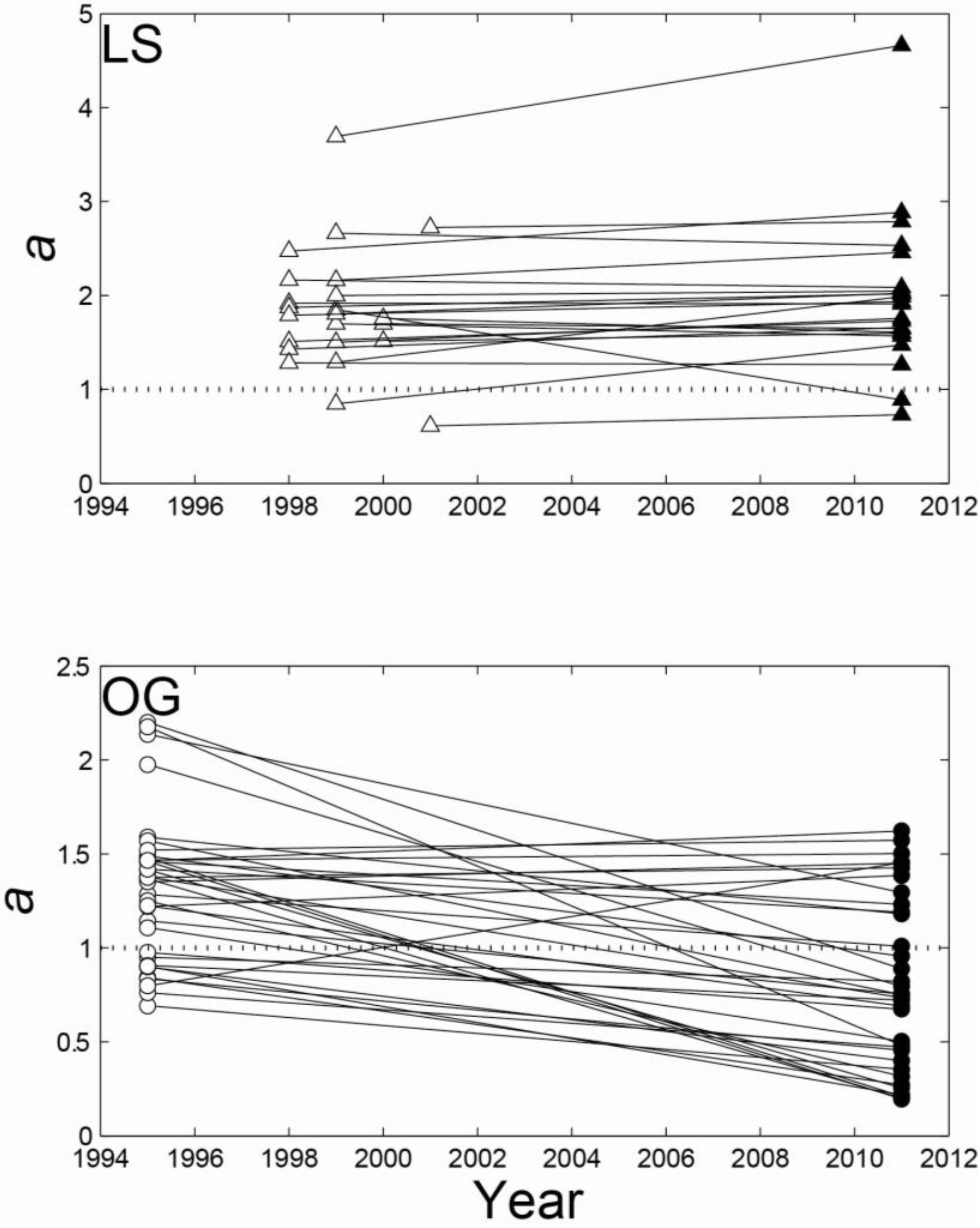
| LS | 2011 | OG | 2011 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Exp | Weib | GG | Total | Exp | Weib | GG | Total | ||||
| Exp | 0 | 1 | 3 | 4 | Exp | 13 | 10 | 1 | 24 | ||
| Initial | Weib | 1 | 11 | 1 | 13 | Initial | Weib | 1 | 1 | 2 | 4 |
| GG | 0 | 1 | 5 | 6 | GG | 3 | 3 | 1 | 7 | ||
| Total | 1 | 13 | 9 | 23 | Total | 17 | 14 | 4 | 35 |

3.2. Regional Analysis
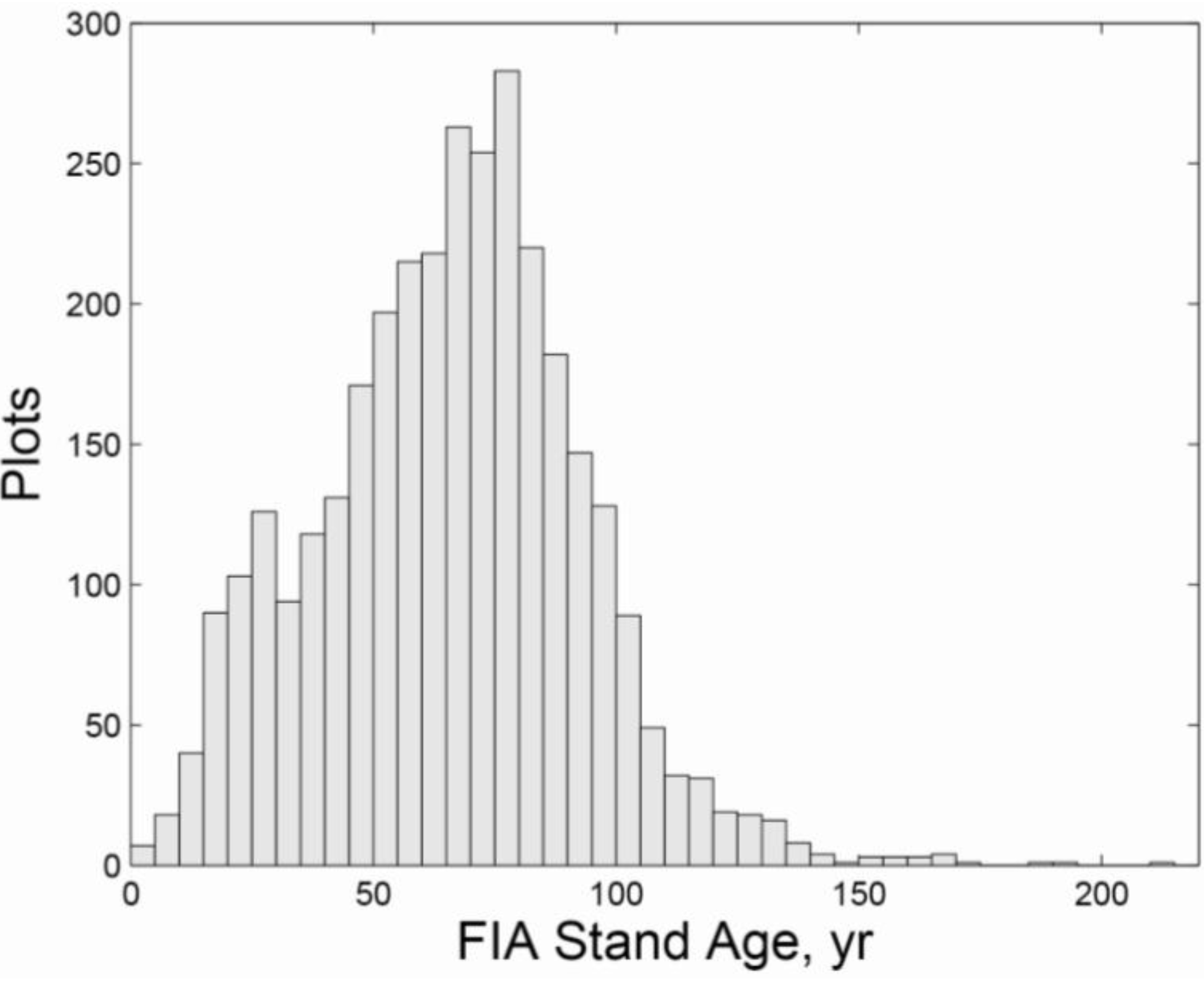
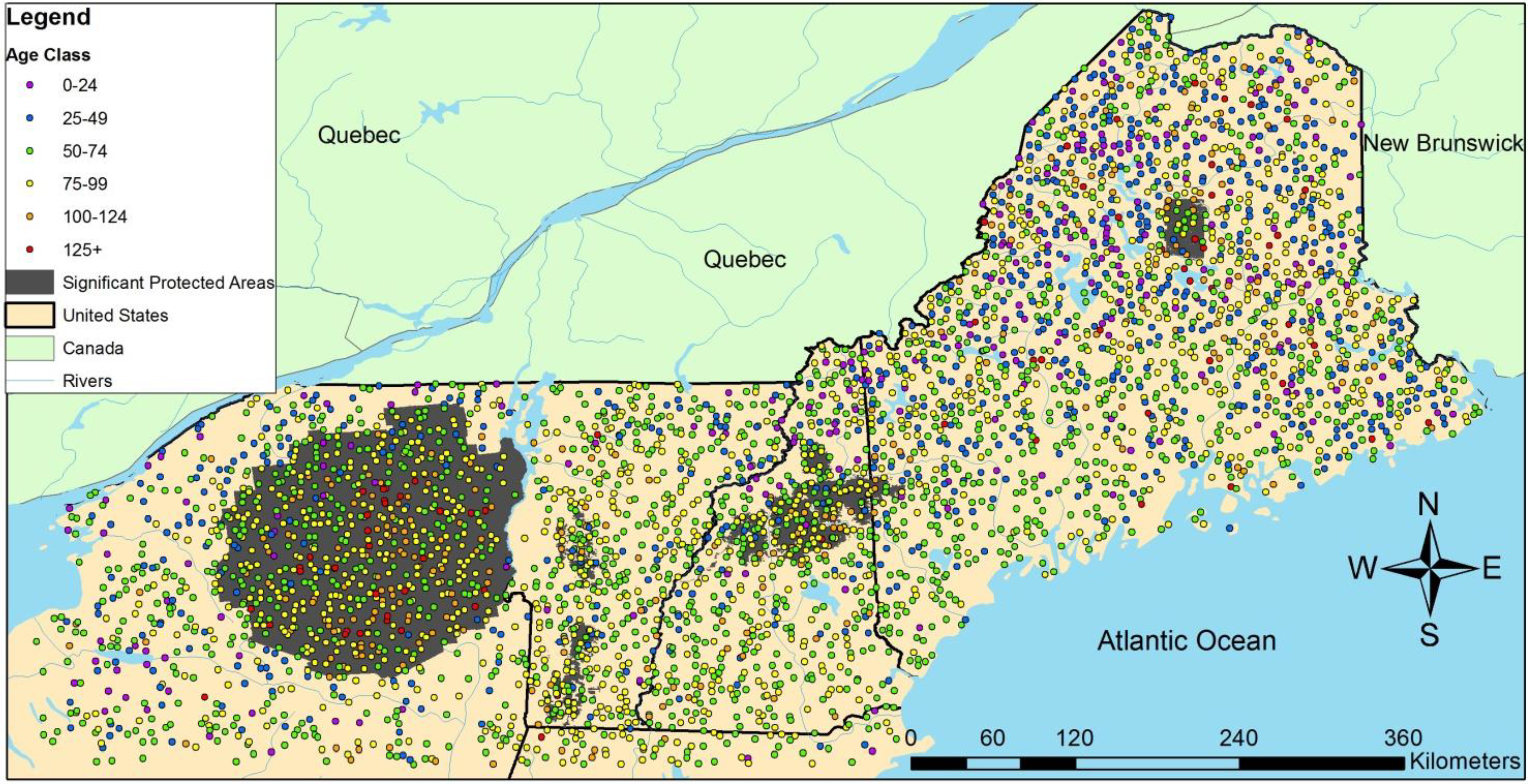
| Ownership | Protection | Harvested | Initial Age | n | QMD (cm) | Large Live Trees/ha | Large Standing Dead/ha | SWARD | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Initial | Change | Initial | Change | Initial | Change | Initial | Change | |||||
| Private | Unprotected | No | <100 | 2127 (1505) | 18.5 ± 0.1 | 0.20 ± 0.06 | 16.5 ± 0.5 | 3.1 ± 0.2 | 2.2 ± 0.2 | −0.27 ± 0.14 | 0.54 ± 0.01 | 0.061 ± 0.002 |
| 100+ | 139 (97) | 21.6 ± 0.4 | −0.21 ± 0.20 | 32.1 ± 2.8 | 5.0 ± 0.9 | 5.4 ± 1.1 | −1.06 ± 0.86 | 0.68 ± 0.02 | 0.037 ± 0.005 | |||
| Yes | <100 | 406 (285) | 19.6 ± 0.2 | −0.87 ± 0.23 | 20.4 ± 1.3 | −5.2 ± 0.9 | 2.2 ± 0.4 | −0.68 ± 0.29 | 0.63 ± 0.01 | −0.165 ± 0.010 | ||
| 100+ | 35 (23) | 22.7 ± 0.8 | −2.73 ± 1.00 | 31.0 ± 6.0 | −12.1 ± 3.4 | 5.2 ± 2.0 | −0.86 ± 1.64 | 0.63 ± 0.04 | −0.208 ± 0.032 | |||
| All | <100 | 2533 (1790) | 18.7 ± 0.1 | 0.03 ± 0.07 | 17.1 ± 0.5 | 1.7 ± 0.2 | 2.2 ± 0.2 | −0.34 ± 0.12 | 0.56 ± 0.01 | 0.025 ± 0.003 | ||
| 100+ | 174 (120) | 21.9 ± 0.4 | −0.71 ± 0.27 | 31.9 ± 2.5 | 1.6 ± 1.1 | 5.3 ± 0.9 | −1.02 ± 0.76 | 0.67 ± 0.02 | −0.012 ± 0.011 | |||
| All | 2707 (1910) | 18.9 ± 0.1 | −0.02 ± 0.06 | 18.0 ± 0.5 | 1.7 ± 0.2 | 2.4 ± 0.2 | −0.38 ± 0.13 | 0.56 ± 0.01 | 0.023 ± 0.003 | |||
| Protected | No (All) | <100 | 174 (103) | 20.7 ± 0.4 | 0.05 ± 0.17 | 33.3 ± 2.4 | 3.0 ± 0.8 | 7.7 ± 1.3 | −1.84 ± 0.72 | 0.71 ± 0.02 | 0.018 ± 0.006 | |
| 100+ | 60 (41) | 25.9 ± 0.7 | −0.63 ± 0.36 | 63.7 ± 4.8 | 3.8 ± 1.6 | 15.2 ± 2.5 | −0.38 ± 1.54 | 0.72 ± 0.02 | 0.029 ± 0.009 | |||
| All | 234 (144) | 22.1 ± 0.4 | −0.12 ± 0.16 | 41.1 ± 2.3 | 3.2 ± 0.7 | 9.8 ± 1.2 | −1.42 ± 0.68 | 0.71 ± 0.01 | 0.021 ± 0.005 | |||
| Public | Unprotected | No | <100 | 262 (187) | 19.1 ± 0.4 | 0.29 ± 0.21 | 20.8 ± 1.9 | 3.4 ± 0.6 | 2.7 ± 0.6 | 0.49 ± 0.38 | 0.58 ± 0.02 | 0.054 ± 0.005 |
| 100+ | 14 (9) | 22.5 ± 1.0 | 0.14 ± 0.66 | 44.6 ± 10.5 | 4.3 ± 2.9 | 1.7 ± 1.7 | 0.00 ± 0.00 | 0.74 ± 0.07 | 0.026 ± 0.016 | |||
| Yes | <100 | 40 (30) | 20.2 ± 0.8 | −0.56 ± 0.56 | 27.5 ± 4.3 | −9.3 ± 3.2 | 3.0 ± 1.1 | 2.56 ± 1.44 | 0.62 ± 0.03 | −0.105 ± 0.027 | ||
| 100+ | 2 (2) | 22.3 ± 0.2 | 0.73 ± 6.82 | 37.2 ± 7.4 | −19.2 ± 19.2 | 0.0 ± 0.0 | 0.00 ± 0.00 | 0.68 ± 0.08 | −0.533 ± 0.150 | |||
| All | <100 | 302 (217) | 19.3 ± 0.4 | 0.18 ± 0.20 | 21.7 ± 1.7 | 1.7 ± 0.7 | 2.7 ± 0.5 | 0.78 ± 0.39 | 0.58 ± 0.02 | 0.033 ± 0.006 | ||
| 100+ | 16 (11) | 22.4 ± 0.9 | 0.21 ± 0.85 | 43.7 ± 9.2 | 1.3 ± 3.7 | 1.4 ± 1.4 | 0.00 ± 0.00 | 0.73 ± 0.06 | −0.044 ± 0.052 | |||
| All | 318 (228) | 19.4 ± 0.4 | 0.18 ± 0.19 | 22.8 ± 1.7 | 1.7 ± 0.7 | 2.7 ± 0.5 | 0.74 ± 0.37 | 0.59 ± 0.02 | 0.029 ± 0.007 | |||
| Protected | No (All) | <100 | 19 (12) | 19.7 ± 1.0 | 0.26 ± 0.39 | 32.9 ± 7.6 | 4.0 ± 1.9 | 7.4 ± 5.0 | −1.99 ± 3.53 | 0.67 ± 0.06 | 0.036 ± 0.016 | |
| 100+ | 11 (8) | 23.2 ± 1.4 | 0.64 ± 0.82 | 54.1 ± 10.8 | 7.7 ± 4.6 | 14.9 ± 8.9 | 0.22 ± 2.55 | 0.71 ± 0.06 | 0.016 ± 0.022 | |||
| All | 30 (20) | 21.0 ± 0.9 | 0.40 ± 0.38 | 40.6 ± 6.4 | 5.4 ± 2.0 | 10.4 ± 4.6 | −1.11 ± 2.31 | 0.68 ± 0.04 | 0.029 ± 0.013 | |||
| All | All | All | <100 | 3028 (2122) | 18.9 ± 0.1 | 0.05 ± 0.06 | 18.6 ± 0.5 | 1.8 ± 0.2 | 2.5 ± 0.2 | −0.31 ± 0.12 | 0.57 ± 0.01 | 0.026 ± 0.003 |
| 100+ | 261 (180) | 22.9 ± 0.3 | −0.58 ± 0.21 | 40.9 ± 2.3 | 2.3 ± 0.9 | 7.8 ± 1.0 | −0.76 ± 0.63 | 0.69 ± 0.01 | −0.003 ± 0.008 | |||
| All | 3289 (2302) | 19.2 ± 0.1 | −0.00 ± 0.06 | 20.4 ± 0.5 | 1.9 ± 0.2 | 2.9 ± 0.2 | −0.34 ± 0.12 | 0.58 ± 0.01 | 0.023 ± 0.002 | |||
3.3. Silvicultural Implications
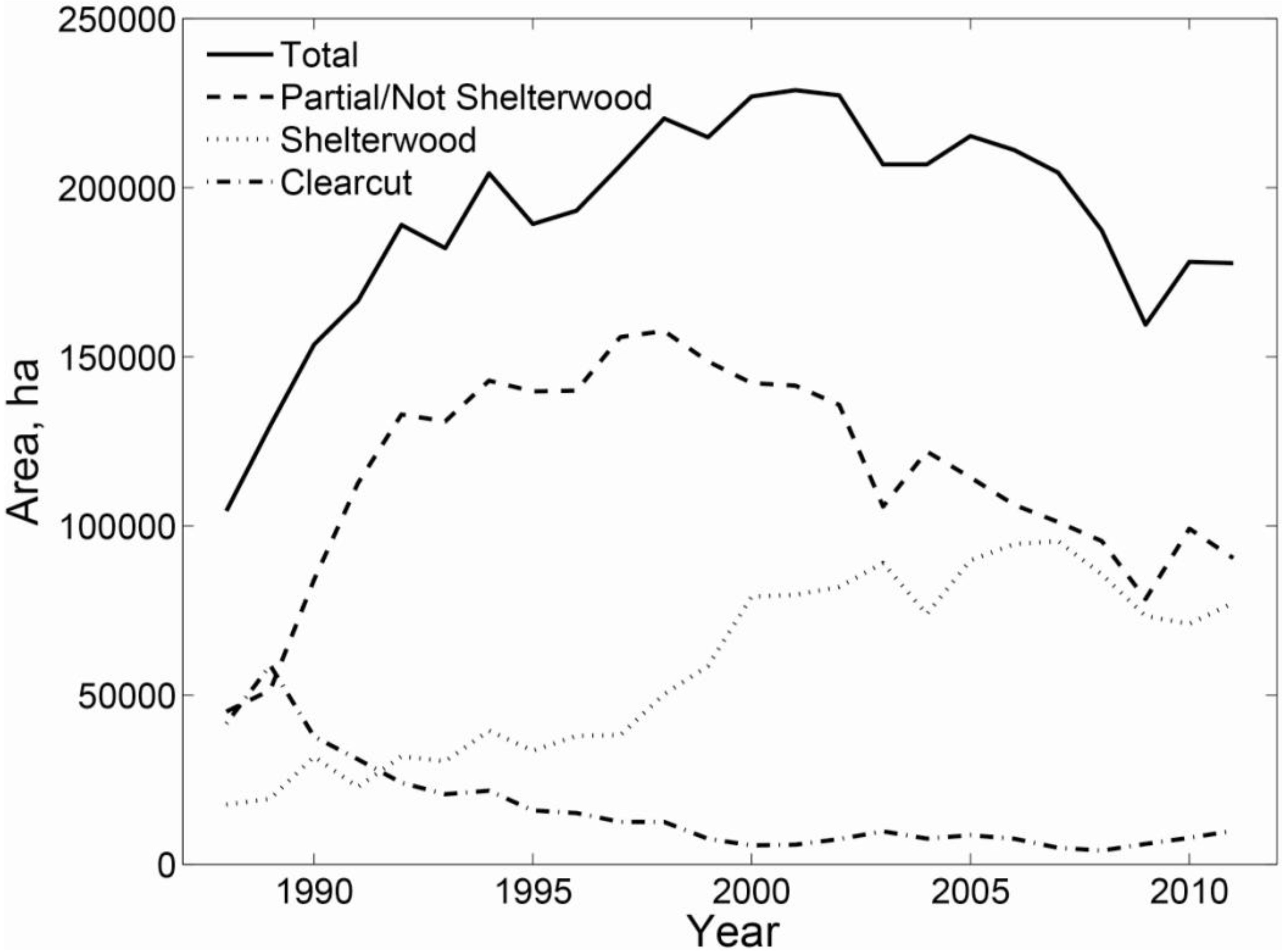
3.4. Policy Implications
| Certification System Standards | |||
|---|---|---|---|
| SFI (Sustainable Forestry Initiative 2010–2014 Standard) | ATFS (2010–2015 Standards of Sustainability for Forest Certification) | FSC (FSC-US Forest Management Standard v1.0, approved 8 July 2010) | |
| Does the standard require retention of forest structural attributes typical of LSOG forests (e.g., large live and dead standing trees)? | Yes | No | Yes |
| Does the standard require conservation of LS forest? | No | No | Yes |
| Criteria relevant to conservation of LS stands and forest structure | Objective 4: Conservation of Biological Diversity including Forests with Exceptional Conservation Value. Performance Indicator 4.1: Program Participants shall have programs to promote biological diversity at stand- and landscape-levels. Indicator 4: Development and implementation of criteria, as guided by regionally appropriate best scientific information, to retain stand-level wildlife habitat elements such as snags, stumps, mast trees, down woody debris, den trees and nest trees. Indicator 5: Program for assessment, conducted either individually or collaboratively, of forest cover types, age or size classes, and habitats at the individual ownership level and, where credible data are available, across the landscape, and take into account findings in planning and management activities. | None Applicable | Indicator 6.3.a.1: The forest owner or manager maintains, enhances, and/or restores under-represented successional stages in the FMU [Forest Management Unit] that would naturally occur on the types of sites found on the FMU. Where old growth of different community types that would naturally occur on the forest are under-represented in the landscape relative to natural conditions, a portion of the forest is managed to enhance and/or restore old growth characteristics. Indicator 6.3.f Management maintains, enhances, or restores habitat components and associated stand structures, in abundance and distribution that could be expected from naturally occurring processes. These components include: (a) large live trees, live trees with decay or declining health, snags, and well-distributed coarse down and dead woody material. Legacy trees where present are not harvested; and (b) vertical and horizontal complexity. Trees selected for retention are generally representative of the dominant species naturally found on the site. |
| Does the standard address old growth forest conservation? | Yes | Yes | Yes |
| Does the standard require protection of existing OG forest? | No | Yes | Yes |
| Criteria relevant to conservation and protection of existing OG forest | Objective 4, Performance Indicator 4.1: Indicator 6: Support of and participation in plans or programs for the conservation of old-growth forests in the region of ownership. | Indicator 7.1.1: Forest owner must make a reasonable effort to locate and protect special sites appropriate for the size of the forest and the scale and intensity of forest management activities. [Special sites include “relic old growth”]. | Indicator 6.3.a.3: When they are present, management maintains the area, structure, composition, and processes of all Type 1 [three acres or more that have never been logged and that display old-growth characteristics] and Type 2 [20 acres that have been logged, but which retain significant old-growth structure and functions] old growth. Types 1 and 2 old growth are also protected and buffered as necessary with conservation zones, unless an alternative plan is developed that provides greater overall protection of old growth values. C6.4 Representative samples of existing ecosystems within the landscape shall be protected in their natural state and recorded on maps, appropriate to the scale and intensity of operations and the uniqueness of the affected resources. Intent: Representative Sample Areas (RSAs) are ecologically viable representative samples designated to serve one or more of three purposes: (1) To establish and/or maintain an ecological reference condition; or (2) To create or maintain an under-represented ecological condition (i.e., includes samples of successional phases, forest types, ecosystems, and/or ecological communities); or (3) To serve as a set of protected areas or refugia for species, communities and community types not captured in other Criteria of this Standard (e.g., to prevent common ecosystems or components from becoming rare). Indicator 9.1.a: The forest owner or manager identifies and maps the presence of High Conservation Value Forests (HCVF) within the FMU and, to the extent that data are available, adjacent to their FMU, in a manner consistent with the assessment process, definitions, data sources, and other guidance described in Appendix F. Given the relative rarity of old growth forests in the contiguous United States, these areas are normally designated as HCVF, and all old growth must be managed in conformance with Indicator 6.3.a.3 and requirements for legacy trees in Indicator 6.3.f. |
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Harmon, M.E.; Ferrell, W.K.; Franklin, J.F. Effects on carbon storage of conversion of old-growth forests to young forests. Science 1990, 247, 699–702. [Google Scholar]
- Luyssaert, S.; Schulze, E.; Börner, A. Old-growth forests as global carbon sinks. Nature 2008, 455, 213–215. [Google Scholar] [CrossRef]
- Whitman, A.A.; Hagan, J.M. An index to identify late-successional forest in temperate and boreal zones. For. Ecol. Manag. 2007, 246, 144–154. [Google Scholar] [CrossRef]
- Reed, W.J. The decision to conserve or harvest old-growth forest. Ecol. Econ. 1993, 8, 45–69. [Google Scholar]
- Freedman, B.; Zelazny, V.; Beaudette, D.; Fleming, T.; Fleming, S.; Forbes, G.; Gerrow, J.; Johnson, G.; Woodley, S. Biodiversity implications of changes in quantity of dead organic matter in managed forests. Environ. Rev. 1996, 4, 238–265. [Google Scholar] [CrossRef]
- Recher, H. Conservation and Management of Eucalypt Forest Vertebrates. In Conservation of Faunal Diversity in Forested Landscapes; DeGraaf, R.M., Miller, R.I., Eds.; Chapman and Hall: New York, NY, USA, 1996; pp. 339–388. [Google Scholar]
- Lindenmayer, D.B.; Franklin, J.F. Conserving Forest Biodiversity: A Comprehensive Multiscaled Approach; Island Press: Washington, DC, USA, 2002. [Google Scholar]
- Irland, L.C. The Northeast’s Changing Forests; Harvard University Press: Cambridge, MA, USA, 1999. [Google Scholar]
- Foster, D.R. Forests in Time: The Environmental Consequences of 1,000 Years of Change in New England; Yale University Press: New Haven, CT, USA, 2006. [Google Scholar]
- Pan, Y.; Chen, J.-M.; Birdsey, R.; McCullough, K.; He, L.; Deng, F. Age structure and disturbance legacy of North American forests. Biogeosciences 2011, 8, 715–732. [Google Scholar] [CrossRef]
- Lorimer, C.G. The Presettlement forest and natural disturbance cycle of northeastern maine. Ecology 1977, 58, 139–148. [Google Scholar] [CrossRef]
- Cogbill, C.V. Vegetation of the presettlement forests of northern New England and New York. Rhodora 2000, 102, 250–276. [Google Scholar]
- Lorimer, C.G. Historical and ecological roles of disturbance in eastern North American forests: 9,000 years of change. Wildl. Soc. Bull. 2001, 29, 425–439. [Google Scholar]
- Lorimer, C.G.; White, A.S. Scale and frequency of natural disturbances in the northeastern U.S.: Implications for early-successional forest habitats and regional age distributions. For. Ecol. Manag. 2003, 185, 41–64. [Google Scholar] [CrossRef]
- Davis, M.B. Eastern old-Growth Forests: Prospects for Rediscovery and Recovery; Island Press: Covelo, CA, USA, 1996. [Google Scholar]
- Keeton, W.S. Managing for late-successional/old-growth characteristics in northern hardwood-conifer forests. For. Ecol. Manag. 2006, 235, 129–142. [Google Scholar]
- North, M.P.; Keeton, W.S. Emulating Natural Disturbance Regimes: An Emerging Approach for Sustainable Forest Management. Chapter 17; In Patterns and Processes in Forest Landscapes; Lafortezza, R., Sanesi, G., Chen, J., Eds.; Springer Netherlands: Houten, The Netherlands, 2008; pp. 341–372. [Google Scholar]
- Bauhus, J.; Puettmann, K.; Messier, C. Silviculture for old-growth attributes. For. Ecol. Manag. 2009, 258, 525–537. [Google Scholar] [CrossRef]
- D’Amato, A.W.; Catanzaro, P.C. Restoring Old-Growth Characteristics. In Cooperative Extension Landowner Outreach Pamphlet; University of Massachusetts: Amherst, MA, USA, 2007; p. 18. [Google Scholar]
- Burrascano, S.; Keeton, W.S.; Sabatini, F.M.; Blasi, C. Commonality and variability in the structural attributes of moist temperate old-growth forests: A global review. For. Ecol. Manag. 2013, 291, 458–479. [Google Scholar]
- Gore, J.A.; Patterson, W.A., III. Mass of downed wood in northern hardwood forests in New Hampshire: Potential effects of forest management. Can. J. For. Res. 1986, 16, 335–339. [Google Scholar] [CrossRef]
- Leak, W.B. Characteristics of Five Climax Stands in New Hampshire; Research Note NE-336; USDA Forest Service, Northeastern Forest Experiment Station: Broomall, PA, USA, 1987.
- Odum, E.P. The strategy of ecosystem development. Science 1969, 164, 262–270. [Google Scholar]
- Jarvis, P.G. Atmospheric carbon dioxide and forests. Philos. Trans. R. Soc. Biol. Sci. 1989, 324, 369–392. [Google Scholar]
- Keith, H.; Mackey, B.G.; Lindenmayer, D.B. Re-evaluation of forest biomass carbon stocks and lessons from the world’s most carbon-dense forests. Proc. Natl. Acad. Sci. USA 2009, 106, 11635–11640. [Google Scholar]
- Keeton, W.; Whitman, A.; McGee, G.G.; Goodale, C.L. Late-successional biomass development in northern hardwood-conifer forests of the northeastern United States. For. Sci. 2011, 57, 489–505. [Google Scholar]
- Galik, C.S.; Jackson, R.B. Risks to forest carbon offset projects in a changing climate. For. Ecol. Manag. 2009, 257, 2209–2216. [Google Scholar] [CrossRef]
- Hurteau, M.D.; Hungate, B.A.; Koch, G.W. Accounting for risk in valuing forest carbon offsets. Carbon Balanc. Manag. 2009, 4, 1–5. [Google Scholar] [CrossRef]
- Tyrrell, L.E.; Crow, T.R. Structural characteristics of old-growth hemlock–hardwood forests in relation to age. Ecology 1994, 75, 370–386. [Google Scholar] [CrossRef]
- McGee, G.G.; Leopold, D.J.; Nyland, R.D. Structural characteristics of old-growth, maturing, and partially cut northern hardwood forests. Ecol. Appl. 1999, 9, 1316–1329. [Google Scholar] [CrossRef]
- Henry, J.D.; Swan, J.M.A. Reconstructing forest history from live and dead plant material--an approach to the study of forest succession in southwest New Hampshire. Ecology 1974, 55, 772–783. [Google Scholar] [CrossRef]
- Ziegler, S.S. Composition, structure, and disturbance history of old-growth and second-growth forests in Adirondack Park, New York. Phys. Geogr. 2004, 25, 152–169. [Google Scholar] [CrossRef]
- D’Amato, A.W.; Orwig, D.A.; Foster, D.R. The influence of successional processes and disturbance on the structure of Tsuga canadensis forests. Ecol. Appl. 2004, 18, 1182–1199. [Google Scholar]
- Foster, D.R. Disturbance history, community organization and vegetation dynamics of the old-growth Pisgah Forest, southwestern New Hampshire, USA. J. Ecol. 1988, 76, 105–134. [Google Scholar] [CrossRef]
- Filip, S.M.; Marquis, D.A.; Leak, W.B. Development of Old-Growth Northern Hardwoods on Bartlett Experimental Forest—A 22-Year Record; U.S. Forest Service, Northeastern Forest Experiment Station Paper 135; U.S. Forest Service: Washington, DC, USA, 1960.
- Woods, K.D. Dynamics in late-successional hemlock-hardwood forests over three decades. Ecology 2000, 81, 110–126. [Google Scholar]
- Woods, K.D. Long-term change and spatial pattern in a late-successional hemlock–northern hardwood forest. J. Ecol. 2000, 88, 267–282. [Google Scholar] [CrossRef]
- Lorimer, C.G.; Dahir, S.E.; Nordheim, E.V. Tree mortality rates and longevity in mature and old-growth hemlock-hardwood forests. J. Ecol. 2001, 89, 960–971. [Google Scholar] [CrossRef]
- Busing, R.T. Tree mortality, canopy turnover, and woody detritus in old cove forests of the southern Appalachians. Ecology 2005, 86, 73–84. [Google Scholar] [CrossRef]
- Hagan, J.M.; Grove, S.L. Coarse woody debris: Humans and nature competing for trees. J. For. 1999, 97, 6–11. [Google Scholar]
- Gunn, J.S.; Hagan, J.M. Woodpecker abundance and tree use in uneven-aged managed, and unmanaged, forest in northern Maine. For. Ecol. Manag. 2000, 126, 1–12. [Google Scholar] [CrossRef]
- Gunn, J.S.; Ducey, M.J.; Whitman, A.A. Late-successional and old-growth forest carbon temporal dynamics in the northern forest (northeastern USA). For. Ecol. Manag. 2013, in press. [Google Scholar]
- Whitman, A. Manomet Center for Conservation Sciences: Brunswick, ME, USA, Unpublished Data. 2013.
- Fraver, S.; White, A.S.; Seymour, R.S. Natural disturbance in an old-growth landscape of northern Maine, USA. J. Ecol. 2009, 97, 289–298. [Google Scholar] [CrossRef]
- Tyrrell, L.E.; Nowacki, G.J.; Crow, T.R.; Buckley, D.S.; Nauertz, E.A.; Niese, J.N.; Rollinger, J.L.; Zasada, J.C. Information about Old Growth for Selected Forest Type Groups in the Eastern United States; General Technical Report NC-197; USDA Forest Service, North Central Forest Experiment Station: St. Paul, MN, USA, 1998.
- Gove, J.H.; van Deusen, P.C. On fixed-area plot sampling for downed coarse woody debris. Forestry 2011, 84, 109–117. [Google Scholar] [CrossRef]
- Reineke, L.H. Perfecting a stand-density index for even-aged forests. J. Agric. Res. 1933, 46, 627–638. [Google Scholar]
- Ducey, M.J.; Knapp, R.A. A stand density index for complex mixed species forests in the northeastern United States. For. Ecol. Manag. 2010, 260, 1613–1622. [Google Scholar] [CrossRef]
- Gingrich, S.F. Measuring and evaluating stocking and stand density in upland hardwood forests in the Central States. For. Sci. 1967, 13, 38–53. [Google Scholar]
- Meyer, H.A.; Stevenson, D.D. The structure and growth of virgin beech-birch-maple-hemlock forests in northern Pennsylvania. J. Agric. Res. 1943, 67, 465–484. [Google Scholar]
- Meyer, H.A. Structure, growth, and drain in uneven-aged forests. J. For. 1952, 50, 85–92. [Google Scholar]
- De Liocourt, F. De l’amanagement des sapinieres. In Bulletin Trimestriel; Société forestière de Franche-Comté et Belfort, Ed.; Impr. et litographie de Paul Jacquin: Besancon, France, 1898. (in French) [Google Scholar]
- Leak, W.B. The J-shaped probability distribution. For. Sci. 1965, 11, 405–409. [Google Scholar]
- Leak, W.B. An expression of diameter distribution for unbalanced, uneven-aged stands and forests. For. Sci. 1964, 10, 39–50. [Google Scholar]
- Goff, F.G.; West, D. Canopy-understory interaction effects on forest population structure. For. Sci. 1975, 21, 98–108. [Google Scholar]
- Bailey, R.L.; Dell, T.R. Quantifying diameter distributions with the Weibull function. For. Sci. 1973, 19, 97–104. [Google Scholar]
- Stacy, E.W. A generalization of the gamma distribution. Ann. Math. Stat. 1962, 33, 1187–1192. [Google Scholar] [CrossRef]
- McDonald, J.B. Some generalized functions for the size distribution of income. Econometrica 1984, 52, 647–664. [Google Scholar] [CrossRef]
- Akaike, H. A new look at the statistical model identification. IEEE Trans. Autom. Control 1974, 19, 716–723. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, 2nd ed.; Springer-Verlag: New York, NY, USA, 2002. [Google Scholar]
- The Enhanced Forest Inventory and Analysis Program—National Sampling Design and Estimation Procedures; General Technical Report SRS-80; Bechtold, W.A.; Patterson, P.L. (Eds.) USDA Forest Service, Southern Research Station: Asheville, NC, USA, 2005.
- R Core Team. R: A Language and Environment for Statistical Computing, R Foundation for Statistical Computing: Vienna, Austria, 2012.
- Beaudet, M.; Brisson, J.; Gravel, D.; Messier, C. Effect of a major canopy disturbance on the coexistence of Acer saccharum and Fagus grandifolia in the understorey of an old-growth forest. J. Ecol. 2007, 95, 458–467. [Google Scholar] [CrossRef]
- Keeton, W.S.; Kraft, C.E.; Warren, D.R. Mature and old-growth riparian forests: Structure, dynamics, and effects on Adirondack stream habitats. Ecol. Appl. 2007, 17, 852–868. [Google Scholar] [CrossRef]
- Hoover, C.M.; Leak, W.B.; Keel, B.G. Benchmark carbon stocks from old-growth forests in northern New England, USA. For. Ecol. Manag. 2012, 266, 108–114. [Google Scholar] [CrossRef]
- McGarrigle, E.; Kershaw, J.A.; Ducey, M.J.; Lavigne, M.B. A new approach to modeling stand-level dynamics based on informed random walks: Influence of bandwidth and sample size. Forestry 2013, 86, 377–389. [Google Scholar] [CrossRef]
- Oliver, C.D.; Larson, B.C. Forest Stand Dynamics; Wiley: New York, NY, USA, 1996. [Google Scholar]
- Franklin, J.F.; Spies, T.A.; van Pelt, R.; Carey, A.; Thornburgh, D.; Berg, D.R.; Lindenmayer, D.; Harmon, M.; Keeton, W.S.; Shaw, D.C.; et al. Disturbances and the structural development of natural forest ecosystems with some implications for silviculture. For. Ecol. Manag. 2002, 155, 399–423. [Google Scholar] [CrossRef]
- Morin, R.S.; Liebhold, A.M.; Tobin, P.C.; Gottschalk, K.W.; Luzader, E. Spread of beech bark disease in the eastern United States and its relationship to regional forest composition. Can. J. For. Res. 2007, 37, 726–736. [Google Scholar] [CrossRef]
- Kasson, M.T.; Livingston, W.H. Relationships among beech bark disease, climate, radial growth response and mortality of American beech in northern Maine, USA. For. Pathol. 2012, 42, 199–212. [Google Scholar] [CrossRef]
- McCaskill, G.L.; McWilliams, W.H.; Barnett, C.J.; Butler, B.J.; Hatfield, M.A.; Kurtz, C.M.; Morin, R.S.; Moser, W.K.; Perry, C.H.; Woodall, C.W. Maine’s Forests 2008; Resource Bulletin No. NRS-48; USDA Forest Service, Northern Research Station: Newtown Square, PA, USA, 2011.
- Canham, C.D.; Rogers, N.; Buchholz, T. Regional variation in forest harvest regimes in the northeastern United States. Ecol. Appl. 2013, 23, 515–522. [Google Scholar] [CrossRef]
- Zheng, D.L.; Heath, L.S.; Ducey, M.J.; Butler, B. Relationships between ownership, forest aboveground biomass distributions and landscape dynamics in the New England states of USA. Environ. Manag. 2010, 45, 377–386. [Google Scholar] [CrossRef]
- Benjamin, J.; Lilieholm, R.J.; Damery, D. Challenges and opportunities for the northeastern forest bioindustry. J. For. 2009, 107, 125–131. [Google Scholar]
- Franklin, J.F. Toward a new forestry. Am. For. 1989, 95, 37–44. [Google Scholar]
- Orwig, D.A.; Cogbill, C.V.; Foster, D.R.; O’Keefe, J.F. Variations in old-growth structure and definitions: Forest dynamics on Wachusett Mountain, Massachusetts. Ecol. Appl. 2001, 11, 437–452. [Google Scholar] [CrossRef]
- Leak, W.B.; Solomon, D.S.; de Bald, P.S. Silvicultural Guide to Northern Hardwood Types in the Northeast, Revised; Research Paper NE-603; USDA Forest Service, Northeastern Forest Research Station: Broomall, PA, USA, 1987.
- Frank, R.M.; Bjorkbom, J.C. A Silvicultural Guide for Spruce-Fir in the Northeast; General Technical Report NE-6; USDA Forest Service, Northeastern Forest Research Station: Upper Darby, PA, USA, 1973.
- Gronewald, C.A.; D’Amato, A.W.; Palik, B.J. The influence of cutting cycle and stocking level on the structure and composition of managed old-growth northern hardwoods. For. Ecol. Manag. 2010, 259, 1151–1160. [Google Scholar] [CrossRef]
- Smith, D.M.; Larson, B.C.; Kelty, M.J.; Ashton, P.M.S. The Practice of Silviculture: Applied Forest Ecology, 9th ed.; John Wiley and Sons: New York, NY, USA, 2007. [Google Scholar]
- Smith, D.M. Concluding Remarks. In The Ecology and Silviculture of Mixed-Species Forests; Kelty, M.J., Oliver, C.D., Larson, B.C., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1992; pp. 281–287. [Google Scholar]
- O’Hara, K.L.; Gersonde, R.F. Stocking control concepts in uneven-aged silviculture. Forestry 2004, 77, 131–143. [Google Scholar] [CrossRef]
- Ducey, M.J. The Reverse-J and Beyond: Developing Practical, Effective Marking Guides. Proceedings of Implementing Uneven-Aged Management in New England: Is It Practical? Fox Research and Demonstration Forest; Caroline, A., Ed.; U.N.H. Cooperative Extension: Durham, NH, USA, 2006. Available online: http://extension.unh. edu/resources/files/Resource002220_Rep3279.pdf (accessed on 1 October 2013).
- Goodburn, J.M.; Lorimer, C.G. Population structure in old-growth and managed northern hardwoods: An examination of the balanced diameter distribution concept. For. Ecol. Manag. 1999, 118, 11–29. [Google Scholar] [CrossRef]
- Rouvinen, S.; Kuuluvainen, T. Tree diameter distributions in natural and managed old Pinus sylvestris-dominated forests. For. Ecol. Manag. 1995, 208, 45–61. [Google Scholar] [CrossRef]
- Schütz, J.P. Modelling the demographic sustainability of pure beech plenter forests in Eastern Germany. Ann. For. Sci. 2006, 63, 93–100. [Google Scholar] [CrossRef]
- Gove, J.H.; Ducey, M.J.; Leak, W.B.; Zhang, L. Rotated sigmoid structures in managed uneven-aged northern hardwood stands: A look at the Burr Type III distribution. Forestry 2008, 81, 161–176. [Google Scholar] [CrossRef]
- Allesandrini, A.; Biondi, F.; di Fillippo, A.; Ziaco, E.; Piovesan, G. Tree size distribution at increase spatial scales converges to the rotated sigmoid curve in two old-growth beech stands of the Italian Apennines. For. Ecol. Manag. 2011, 262, 1950–1962. [Google Scholar] [CrossRef]
- Singer, M.T.; Lorimer, C.G. Crown release as a potential old-growth restoration approach in northern hardwoods. Can. J. For. Res. 1997, 27, 1222–1232. [Google Scholar] [CrossRef]
- Ma, Z.; Kittredge, D.B. How family forest owners consider timber harvesting, land sale, and conservation easement decisions: Insights from Massachusetts, USA. Int. J. For. Res. 2011, 2011, 1–13. [Google Scholar]
- Majumdar, I.; Laband, D.; Teeter, L.; Butler, B.J. Motivations and land-use intentions of nonindustrial private forest landowners: Comparing inheritors to noninheritors. For Sci. 2009, 55, 423–432. [Google Scholar]
- Maine Forest Practices Act, 29 July 2004. Available online: http://www.maine.gov/doc/mfs/pubs/htm/fpa_04.html (accessed on 26 June 2013).
- State of Maine, Department of Conservation, Maine Forest Service Silvicultural Activities Reports 1996–2011. Available online: http://www.maine.gov/doc/mfs/pubs.htm (accessed on 26 June 2013).
- State of Maine. An Act to Amend the Maine Tree Growth Tax Law and the Open Space Tax Law. Public Law Chapter 618, H.P. 844-L.D. 1138. 2012. Available online: http://www. mainelegislature.org/legis/bills/getPDF.asp?paper=HP0844&item=4&snum=125 (accessed on 29 June 2013).
- Sustainable Forestry Initiative 2010–2014 Standard. Available online: http://www.sfiprogram.org/files/pdf/section2sfirequirements2010-2014pdf/ (accessed on 26 June 2013).
- American Forest Foundation 2010–2015 Standards of Sustainability for Forest Certification. Available online: http://www.treefarmsystem.org/certification-american-tree-farm-standards (accessed on 26 June 2013).
- FSC-US Forest Management Standard, Ver. 1.0. Available online: http://us.fsc.org/download.fsc-us-forest-management-standard-v1-0.95.pdf (accessed on 27 June 2013).
- Gunn, J.S.; Saah, D.S.; Fernholz, K.; Ganz, D.J. Carbon credit eligibility under area regulation of harvest levels in Northern Minnesota. For. Sci. 2011, 57, 470–478. [Google Scholar]
- Nunery, J.S.; Keeton, W.S. Forest carbon storage in the northeastern United States: Net effects of harvesting frequency, post-harvest retention, and wood products. For. Ecol. Manag. 2010, 259, 1363–1375. [Google Scholar] [CrossRef]
- Climate Action Reserve Forest Project Protocol Version 3.2. Available online: http://www.climateactionreserve.org/how/protocols/forest/dev/version-3-2/ (accessed on 27 June 2013).
- American Carbon Registry Forest Carbon Project Standard v2.1. Available online: http://americancarbonregistry.org/carbon-accounting/carbon-accounting/forest-carbon-project-standard-v2.0 (accessed on 27 June 2013).
- Fishburn, I.S.; Kareiva, P.; Gaston, K.J.; Armsworth, P.R. The growth of easements as a conservation tool. PLoS One 2009, 4, e4996. [Google Scholar]
- Rissman, A.R.; Lozier, L.; Comendant, T.; Kareiva, P.; Kiesecker, J.M.; Shaw, M.R.; Merenlender, A.M. Conservation easements: Biodiversity protection and private use. Conserv. Biol. 2007, 21, 709–718. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ducey, M.J.; Gunn, J.S.; Whitman, A.A. Late-Successional and Old-Growth Forests in the Northeastern United States: Structure, Dynamics, and Prospects for Restoration. Forests 2013, 4, 1055-1086. https://doi.org/10.3390/f4041055
Ducey MJ, Gunn JS, Whitman AA. Late-Successional and Old-Growth Forests in the Northeastern United States: Structure, Dynamics, and Prospects for Restoration. Forests. 2013; 4(4):1055-1086. https://doi.org/10.3390/f4041055
Chicago/Turabian StyleDucey, Mark J., John S. Gunn, and Andrew A. Whitman. 2013. "Late-Successional and Old-Growth Forests in the Northeastern United States: Structure, Dynamics, and Prospects for Restoration" Forests 4, no. 4: 1055-1086. https://doi.org/10.3390/f4041055
APA StyleDucey, M. J., Gunn, J. S., & Whitman, A. A. (2013). Late-Successional and Old-Growth Forests in the Northeastern United States: Structure, Dynamics, and Prospects for Restoration. Forests, 4(4), 1055-1086. https://doi.org/10.3390/f4041055




