Phenolic Leaf Compounds in Ash Trees (Fraxinus excelsior L.) in the Context of Ash Dieback
Abstract
1. Introduction
- To determine the variation in foliar phenolic content in mature ash trees (in response to ash dieback).
- To analyse how phenolic content varies with disease severity, year, and site.
- To evaluate the suitability of different monitoring methods (photometric determination of groups or HPLC for individual phenols) for assessing ADB.
2. Materials and Methods
2.1. Monitoring Sites and Scoring
2.2. Sampling and Sample Handling
2.3. Lyophilisation and Weighing
2.4. Methanolic Extraction of Lyophilised Samples for Photometric Analysis
2.5. Analysis of Phenols
2.5.1. Total Phenolic Content (Folin-Positive Compounds, TPH)
2.5.2. Vanillin-Positive Compounds (VAN)
2.5.3. Procyanidins (PC)
2.5.4. Ortho-Dihydroxyphenols (ODHP)
2.5.5. Individual Phenolic Compounds (HPLC Analysis)
2.6. Data Processing and Evaluation
2.6.1. Calculation of Phenolic Groups
2.6.2. Data Integration and Statistical Analyses
3. Results
3.1. General Overview of Phenolic Contents
3.2. Variability of Phenolic Groups
3.3. Correlation of Phenolic Groups and Individual Phenolic Compounds
3.4. Analysis of Main and Interaction Effects of Location, Year, and Damage Score
3.4.1. Overall Analysis
3.4.2. Comparison of Monitoring Plots
3.4.3. Comparison of Monitoring Years
4. Discussion
4.1. Correlation of Phenolic Groups with ADB Damage Scoring
4.2. Comparison of Analytical Methods
4.3. Differences in Crown Damage Classes
4.4. Influence of Site and Year
4.5. Further Possible Influencing Factors
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADB | ash dieback |
| ANOVA | analysis of variance |
| CDC | crown damage class |
| DM | dry mass |
| HPLC | high performance liquid chromatography |
| IBF | intensive monitoring plot (Intensivbeobachtungsfläche) |
| IQR | interquartile range |
| N | number |
| ODHP | ortho-dihydroxyphenols |
| PC | procyanidins |
| pH | potential of hydrogen |
| RF | response factor |
| TPH | total phenols (folin-positive compounds) |
| UV | ultraviolet |
| v/v | volume per volume |
| VAN | vanillin-positive compounds |
References
- Kostova, I.; Iossifova, T. Chemical Components of Fraxinus Species. Fitoterapia 2007, 78, 85–106. [Google Scholar] [CrossRef]
- Thomas, P.A. Biological Flora of the British Isles: Fraxinus Excelsior. J. Ecol. 2016, 104, 1158–1209. [Google Scholar] [CrossRef]
- Kiss, A.K.; Michalak, B.; Patyra, A.; Majdan, M. UHPLC-DAD-ESI-MS/MS and HPTLC Profiling of Ash Leaf Samples from Different Commercial and Natural Sources and Their in Vitro Effects on Mediators of Inflammation. Phytochem. Anal. 2020, 31, 57–67. [Google Scholar] [CrossRef]
- Nemesio-Gorriz, M.; Menezes, R.C.; Paetz, C.; Hammerbacher, A.; Steenackers, M.; Schamp, K.; Höfte, M.; Svatoš, A.; Gershenzon, J.; Douglas, G.C. Canditate Metabolites for Ash Dieback Tolerance in Fraxinus Excelsior. J. Exp. Bot. 2020, 71, 6074–6083. [Google Scholar] [CrossRef]
- Holopainen, J.K.; Virjamo, V.; Ghimire, R.P.; Blande, J.D.; Julkunen-Tiitto, R.; Kivimäenpää, M. Climate Change Effects on Secondary Compounds of Forest Trees in the Northern Hemisphere. Front. Plant Sci. 2018, 9, 1445. [Google Scholar] [CrossRef] [PubMed]
- Witzell, J.; Martín, J.A. Phenolic Metabolites in the Resistance of Northern Forest Trees to Pathogens—Past Experiences and Future Prospects. Can. J. For. Res. 2008, 38, 2711–2727. [Google Scholar] [CrossRef]
- Kumar, S.; Abedin, M.M.; Singh, A.K.; Das, S. Role of Phenolic Compounds in Plant-Defensive Mechanisms. In Plant Phenolics in Sustainable Agriculture; Lone, R., Shuab, R., Kamili, A.N., Eds.; Springer: Singapore, 2020; pp. 517–532. ISBN 9789811548895. [Google Scholar]
- Šamec, D.; Karalija, E.; Šola, I.; Vujčić Bok, V.; Salopek-Sondi, B. The Role of Polyphenols in Abiotic Stress Response: The Influence of Molecular Structure. Plants 2021, 10, 118. [Google Scholar] [CrossRef] [PubMed]
- Striganavičiūtė, G.; Žiauka, J.; Sirgedaitė-Šėžienė, V.; Vaitiekūnaitė, D. Priming of Resistance-Related Phenolics: A Study of Plant-Associated Bacteria and Hymenoscyphus Fraxineus. Microorganisms 2021, 9, 2504. [Google Scholar] [CrossRef]
- Nicholson, R.L.; Hammerschmidt, R. Phenolic Compounds and Their Role in Disease Resistance. Annu. Rev. Phytopathol. 1992, 30, 369–389. [Google Scholar] [CrossRef]
- Hulse, S.V.; Antonovics, J.; Hood, M.E.; Bruns, E.L. Specific Resistance Prevents the Evolution of General Resistance and Facilitates Disease Emergence. J. Evol. Biol. 2023, 36, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Moore, B.D.; Andrew, R.L.; Külheim, C.; Foley, W.J. Explaining Intraspecific Diversity in Plant Secondary Metabolites in an Ecological Context. New Phytol. 2014, 201, 733–750. [Google Scholar] [CrossRef] [PubMed]
- Husson, C.; Caël, O.; Grandjean, J.P.; Nageleisen, L.M.; Marçais, B. Occurrence of Hymenoscyphus Pseudoalbidus on Infected Ash Logs. Plant Pathol. 2012, 61, 889–895. [Google Scholar] [CrossRef]
- Gross, A.; Holdenrieder, O.; Pautasso, M.; Queloz, V.; Sieber, T.N. Hymenoscyphus Pseudoalbidus, the Causal Agent of European Ash Dieback. Mol. Plant Pathol. 2014, 15, 5–21. [Google Scholar] [CrossRef]
- Gross, A.; Holdenrieder, O. Pathogenicity of Hymenoscyphus Fraxineus and Hymenoscyphus Albidus towards Fraxinus Mandshurica Var. Japonica. For. Pathol. 2015, 45, 172–174. [Google Scholar] [CrossRef]
- Haňáčková, Z.; Koukol, O.; Čmoková, A.; Zahradník, D.; Havrdová, L. Direct Evidence of Hymenoscyphus Fraxineus Infection Pathway through the Petiole-Shoot Junction. For. Pathol. 2017, 47, e12370. [Google Scholar] [CrossRef]
- Thomas, H.E. Studies on Armillaria Mellea (Vahl) Quel., Infection, Parasitism, and Host Resistance. J. Agric. Res. 1934, 48, 187. [Google Scholar]
- Guillaumin, J.J.; Rykowski, K. Studies of the infection of walnut (Juglans regia) by Armillaria mellea under controlled experimental conditions. Folia For. Pol. A (Leśnictwo) 1980, 24, 191–213. [Google Scholar]
- Rishbeth, J. Infection Cycle of Armillaria and Host Response. Eur. J. For. Pathol. 1985, 15, 332–341. [Google Scholar] [CrossRef]
- Meyn, R.; Langer, G.J.; Gross, A.; Langer, E.J. Fungal Colonization Patterns in Necrotic Rootstocks and Stem Bases of Dieback-Affected Fraxinus excelsior L. For. Pathol. 2019, 49, e12520. [Google Scholar] [CrossRef]
- Peters, S.; Gruschwitz, N.; Bien, S.; Fuchs, S.; Bubner, B.; Blunk, V.; Langer, G.J.; Langer, E.J. The Fungal Predominance in Stem Collar Necroses of Fraxinus Excelsior: A Study on Hymenoscyphus Fraxineus Multilocus Genotypes. J. Plant Dis. Prot. 2024, 13, 1341–1353. [Google Scholar] [CrossRef]
- Langer, G.J.; Fuchs, S.; Osewold, J.; Peters, S.; Schrewe, F.; Ridley, M.; Kätzel, R.; Bubner, B.; Grüner, J. FraxForFuture—Research on European Ash Dieback in Germany. J. Plant Dis. Prot. 2022, 129, 1285–1295. [Google Scholar] [CrossRef]
- Gross, A.; Hosoya, T.; Zhao, Y.J.; Baral, H.-O. Hymenoscyphus Linearis Sp Nov: Another Close Relative of the Ash Dieback Pathogen H. Fraxineus. Mycol. Prog. 2015, 14, 20. [Google Scholar] [CrossRef]
- Przybył, K. Fungi Associated with Necrotic Apical Parts of Fraxinus Excelsior Shoots. For. Pathol. 2002, 32, 387–394. [Google Scholar] [CrossRef]
- Kowalski, T. Chalara Fraxinea Sp. Nov. Associated with Dieback of Ash (Fraxinus Excelsior) in Poland. For. Pathol. 2006, 36, 264–270. [Google Scholar] [CrossRef]
- McKinney, L.V.; Nielsen, L.R.; Collinge, D.B.; Thomsen, I.M.; Hansen, J.K.; Kjær, E.D. The Ash Dieback Crisis: Genetic Variation in Resistance Can Prove a Long-term Solution. Plant Pathol. 2014, 63, 485–499. [Google Scholar] [CrossRef]
- Enderle, R.; Nakou, A.; Thomas, K.; Metzler, B. Susceptibility of Autochthonous German Fraxinus Excelsior Clones to Hymenoscyphus Pseudoalbidus Is Genetically Determined. Ann. For. Sci. 2015, 72, 183–193. [Google Scholar] [CrossRef]
- Enderle, R.; Stenlid, J.; Vasaitis, R. An Overview of Ash (Fraxinus Spp.) and the Ash Dieback Disease in Europe. CABI Rev. 2019, 14, 1–12. [Google Scholar] [CrossRef]
- Cleary, M.R.; Andersson, P.F.; Broberg, A.; Elfstrand, M.; Daniel, G.; Stenlid, J. Genotypes of Fraxinus Excelsior with Different Susceptibility to the Ash Dieback Pathogen Hymenoscyphus Pseudoalbidus and Their Response to the Phytotoxin Viridiol—A Metabolomic and Microscopic Study. Phytochemistry 2014, 102, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Sollars, E.S.A.; Harper, A.L.; Kelly, L.J.; Sambles, C.M.; Ramirez-Gonzalez, R.H.; Swarbreck, D.; Kaithakottil, G.; Cooper, E.D.; Uauy, C.; Havlickova, L.; et al. Genome Sequence and Genetic Diversity of European Ash Trees. Nature 2017, 541, 212–216. [Google Scholar] [CrossRef]
- Villari, C.; Dowkiw, A.; Enderle, R.; Ghasemkhani, M.; Kirisits, T.; Kjær, E.D.; Marčiulynienė, D.; McKinney, L.V.; Metzler, B.; Muñoz, F.; et al. Advanced Spectroscopy-Based Phenotyping Offers a Potential Solution to the Ash Dieback Epidemic. Sci. Rep. 2018, 8, 17448. [Google Scholar] [CrossRef] [PubMed]
- Sidda, J.D.; Song, L.; Parker, J.L.; Studholme, D.J.; Sambles, C.; Grant, M. Diversity of Secoiridoid Glycosides in Leaves of UK and Danish Ash Provide New Insight for Ash Dieback Management. Sci. Rep. 2020, 10, 19566. [Google Scholar] [CrossRef]
- Grace, S.C. Phenolics as Antioxidants. In Antioxidants and Reactive Oxygen Species in Plants; Smirnoff, N., Ed.; Wiley: Hoboken, NJ, USA, 2005; pp. 141–168. ISBN 978-1-4051-2529-1. [Google Scholar]
- Tak, Y.; Kumar, M. Phenolics: A Key Defence Secondary Metabolite to Counter Biotic Stress. In Plant Phenolics in Sustainable Agriculture; Lone, R., Shuab, R., Kamili, A.N., Eds.; Springer: Singapore, 2020; pp. 309–329. ISBN 9789811548895. [Google Scholar]
- McPherson, B.A.; Mori, S.R.; Opiyo, S.O.; Conrad, A.O.; Wood, D.L.; Bonello, P. Association between Resistance to an Introduced Invasive Pathogen and Phenolic Compounds That May Serve as Biomarkers in Native Oaks. For. Ecol. Manag. 2014, 312, 154–160. [Google Scholar] [CrossRef]
- Peters, S.; Langer, G.; Kätzel, R. Bonitur Geschädigter Eschen Im Kontext Des Eschentriebsterbens. AFZ-DerWald 2021, 76, 28–31. [Google Scholar]
- Peters, S.; Langer, G.; Kätzel, R. Eschentriebsterben: Kriterien Zur Schadensbonitur an Eschen; Fachagentur Nachwachsende Rohstoffe eV (FNR): Gülzow-Prüzen, Germany, 2021.
- Cheynier, V.; Comte, G.; Davies, K.M.; Lattanzio, V.; Martens, S. Plant Phenolics: Recent Advances on Their Biosynthesis, Genetics, and Ecophysiology. Plant Physiol. Biochem. 2013, 72, 1–20. [Google Scholar] [CrossRef]
- Lunderstädt, J.; Ahlers, S. Zur Nahrungsqualität von Fichtennadeln Für Forstliche Schadinsekten. Z. Für Angew. Entomol. 1983, 95, 141–150. [Google Scholar] [CrossRef]
- Swain, T.; Hillis, W.E. The Phenolic Constituents of Prunus Domestica. I.—The Quantitative Analysis of Phenolic Constituents. J. Sci. Food Agric. 1959, 10, 63–68. [Google Scholar] [CrossRef]
- Broadhurst, R.B.; Jones, W.T. Analysis of Condensed Tannins Using Acidified Vanillin. J. Sci. Food Agric. 1978, 29, 788–794. [Google Scholar] [CrossRef]
- Stafford, H.A.; Cheng, T.-Y. The Procyanidins of Douglas Fir Seedlings, Callus and Cell Suspension Cultures Derived from Cotyledons. Phytochemistry 1980, 19, 131–135. [Google Scholar] [CrossRef]
- Arnow, L.E. Colorimetric Determination of the Components of 3, 4-Dihydroxyphenylalanine-Tyrosine Mixtures. J. Biol. Chem 1937, 118, 531–537. [Google Scholar] [CrossRef]
- Förster, N.; Dilling, S.; Ulrichs, C.; Huyskens-Keil, S. Nutritional Diversity in Leaves of Various Amaranth (Amaranthus spp.) Genotypes and Its Resilience to Drought Stress. J. Appl. Bot. Food Qual. 2023, 96, 1–10. [Google Scholar] [CrossRef]
- DWD Climate Data Center (CDC). Monatsmittel Der Stationsmessungen Der Lufttemperatur in 2 m Höhe in °C Für Deutschland, Version V21.3 2025. Available online: https://dwd-geoportal.de/products/OBS_DEU_P1M_T2M/ (accessed on 16 April 2025).
- DWD Climate Data Center (CDC). Rasterdaten Der Monatssumme Der Niederschlagshöhe Für Deutschland, Version V19.3 2025. Available online: https://dwd-geoportal.de/products/GRD_DEU_P1M_RR/ (accessed on 16 April 2025).
- Carnat, A.U.; Lamaison, J.L.; Duband, F. Contents of the Main Components in the Ash Leaves, Fraxinus excelsior L. Plantes Med. Phytother. 1990, 24, 145–151. [Google Scholar]
- Sticher, O. Phenolische Verbindungen. In Pharmakognosie–Phytopharmazie; Hänsel, R., Sticher, O., Eds.; Springer Medizin: Heidelberg, Germany, 2010; pp. 1053–1206. ISBN 978-3-642-00962-4. [Google Scholar]
- Fuchs, S.; Häuser, H.; Peters, S.; Knauf, L.; Rentschler, F.; Kahlenberg, G.; Kätzel, R.; Evers, J.; Paar, U.; Langer, G.J. Ash Dieback Assessments on Intensive Monitoring Plots in Germany: Influence of Stand, Site and Time on Disease Progression. J. Plant Dis. Prot. 2024, 131, 1355–1372. [Google Scholar] [CrossRef]
- Babenko, L.M.; Smirnov, O.E.; Romanenko, K.O.; Trunova, O.K.; Kosakivska, I.V. Phenolic Compounds in Plants: Biogenesis and Functions. Ukr. Biochem. J. 2019, 91, 5–18. [Google Scholar] [CrossRef]
- Hietala, A.M.; Timmermann, V.; BØrja, I.; Solheim, H. The Invasive Ash Dieback Pathogen Hymenoscyphus Pseudoalbidus Exerts Maximal Infection Pressure Prior to the Onset of Host Leaf Senescence. Fungal Ecol. 2013, 6, 302–308. [Google Scholar] [CrossRef]
- Garcia-Molina, A.; Pastor, V. Systemic Analysis of Metabolome Reconfiguration in Arabidopsis after Abiotic Stressors Uncovers Metabolites That Modulate Defense against Pathogens. Plant Commun. 2024, 5, 100645. [Google Scholar] [CrossRef] [PubMed]
- Barthod, S.; Cerovic, Z.; Epron, D. Can Dual Chlorophyll Fluorescence Excitation Be Used to Assess the Variation in the Content of UV-Absorbing Phenolic Compounds in Leaves of Temperate Tree Species along a Light Gradient? J. Exp. Bot. 2007, 58, 1753–1760. [Google Scholar] [CrossRef]
- Jaakola, L.; Hohtola, A. Effect of Latitude on Flavonoid Biosynthesis in Plants. Plant Cell Environ. 2010, 33, 1239–1247. [Google Scholar] [CrossRef]
- Yang, B.; Zheng, J.; Laaksonen, O.; Tahvonen, R.; Kallio, H. Effects of Latitude and Weather Conditions on Phenolic Compounds in Currant (Ribes spp.) Cultivars. J. Agric. Food Chem. 2013, 61, 3517–3532. [Google Scholar] [CrossRef]
- Vagiri, M.; Ekholm, A.; Öberg, E.; Johansson, E.; Andersson, S.C.; Rumpunen, K. Phenols and Ascorbic Acid in Black Currants (Ribes nigrum L.): Variation Due to Genotype, Location, and Year. J. Agric. Food Chem. 2013, 61, 9298–9306. [Google Scholar] [CrossRef]
- Marin, A.; Ferreres, F.; Barberá, G.G.; Gil, M.I. Weather Variability Influences Color and Phenolic Content of Pigmented Baby Leaf Lettuces throughout the Season. J. Agric. Food Chem. 2015, 63, 1673–1681. [Google Scholar] [CrossRef]
- Fachagentur Nachwachsende Rohstoffe e. V. (FNR). Schlussbericht zum Verbundvorhaben FraxMon; FNR: Gülzow, Germany, 2024; p. 66. Available online: https://www.fnr.de/fileadmin/projektdatenbank/2219WK20B4.pdf (accessed on 3 April 2025).
- Burghard, V.; Wende, S.; Schneck, V.; Ulrich, A. Inoculation of Fraxinus Excelsior Seedlings with Bacterial Strains to Enhance the Tolerance against Ash Dieback. J. Plant Dis. Prot. 2024, 132, 9. [Google Scholar] [CrossRef]
- Reddivari, L.; Hale, A.L.; Miller, J.C. Genotype, Location, and Year Influence Antioxidant Activity, Carotenoid Content, Phenolic Content, and Composition in Specialty Potatoes. J. Agric. Food Chem. 2007, 55, 8073–8079. [Google Scholar] [CrossRef]
- Brandolini, A.; Castoldi, P.; Plizzari, L.; Hidalgo, A. Phenolic Acids Composition, Total Polyphenols Content and Antioxidant Activity of Triticum Monococcum, Triticum Turgidum and Triticum Aestivum: A Two-Years Evaluation. J. Cereal Sci. 2013, 58, 123–131. [Google Scholar] [CrossRef]
- Budde, K. (University of Göttingen, Göttingen, Germany). Personal communication, 2023.
- Sambles, C.M.; Salmon, D.L.; Florance, H.; Howard, T.P.; Smirnoff, N.; Nielsen, L.R.; McKinney, L.V.; Kjær, E.D.; Buggs, R.J.A.; Studholme, D.J.; et al. Ash Leaf Metabolomes Reveal Differences between Trees Tolerant and Susceptible to Ash Dieback Disease. Sci. Data 2017, 4, 170190. [Google Scholar] [CrossRef]
- Erb, M.; Kliebenstein, D.J. Plant Secondary Metabolites as Defenses, Regulators, and Primary Metabolites: The Blurred Functional Trichotomy. Plant Physiol. 2020, 184, 39–52. [Google Scholar] [CrossRef]
- Havrdová, L.; Novotná, K.; Zahradník, D.; Buriánek, V.; Pešková, V.; Šrůtka, P.; Černý, K. Differences in Susceptibility to Ash Dieback in Czech Provenances of Fraxinus Excelsior. For. Pathol. 2016, 46, 281–288. [Google Scholar] [CrossRef]
- Muñoz, F.; Marçais, B.; Dufour, J.; Dowkiw, A. Rising Out of the Ashes: Additive Genetic Variation for Crown and Collar Resistance to Hymenoscyphus fraxineus in Fraxinus excelsior. Phytopathology 2016, 106, 1535–1543. [Google Scholar] [CrossRef]
- Gossner, M.M.; Perret-Gentil, A.; Britt, E.; Queloz, V.; Glauser, G.; Ladd, T.; Roe, A.D.; Cleary, M.; Liziniewicz, M.; Nielsen, L.R.; et al. A Glimmer of Hope—Ash Genotypes with Increased Resistance to Ash Dieback Pathogen Show Cross-resistance to Emerald Ash Borer. New Phytol. 2023, 240, 1219–1232. [Google Scholar] [CrossRef]
- Núñez-Farfán, J.; Fornoni, J.; Valverde, P.L. The Evolution of Resistance and Tolerance to Herbivores. Annu. Rev. Ecol. Evol. Syst. 2007, 38, 541–566. [Google Scholar] [CrossRef]
- Landolt, J.; Gross, A.; Holdenrieder, O.; Pautasso, M. Ash Dieback Due to Hymenoscyphus Fraxineus: What Can Be Learnt from Evolutionary Ecology? Plant Pathol. 2016, 65, 1056–1070. [Google Scholar] [CrossRef]
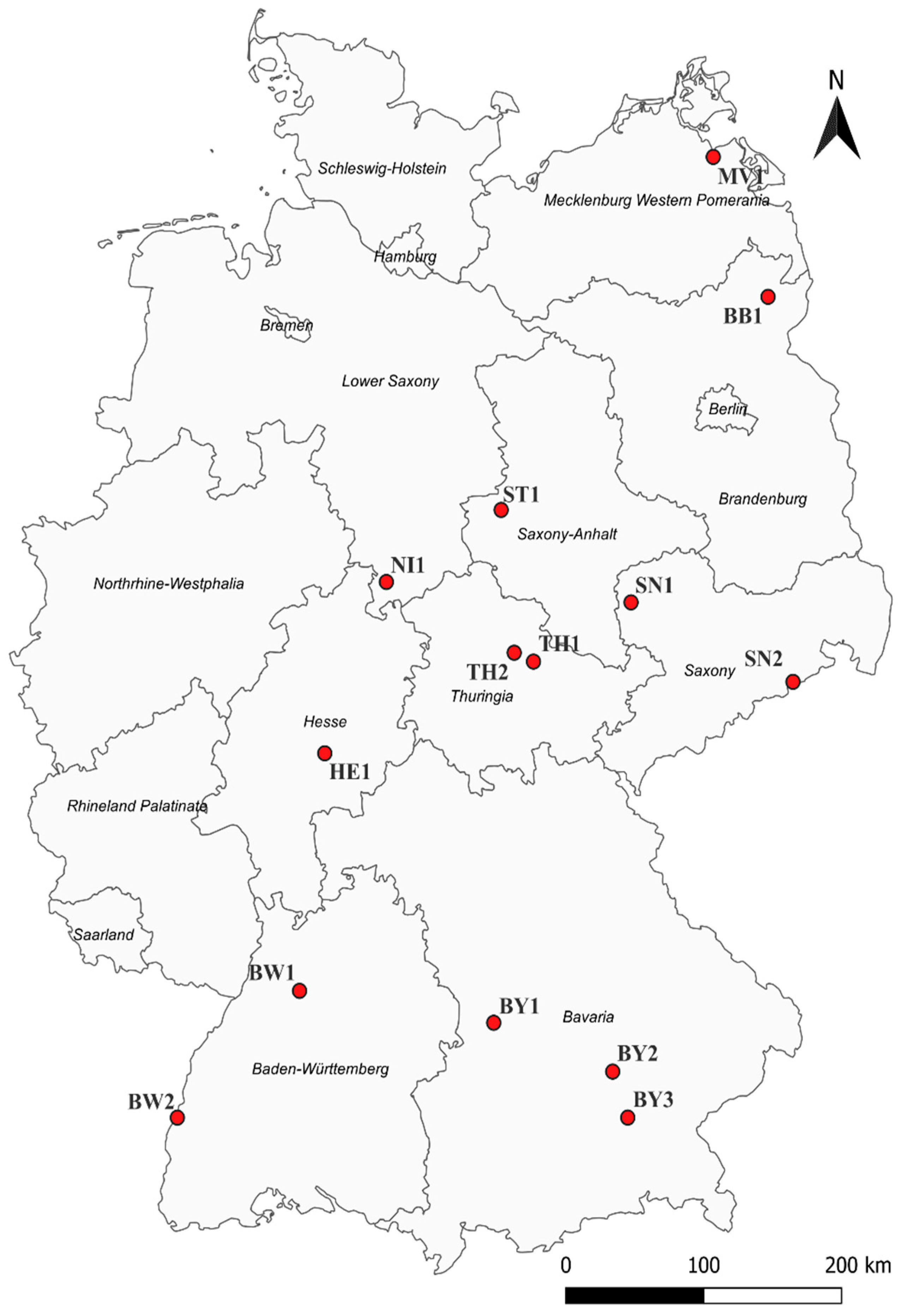
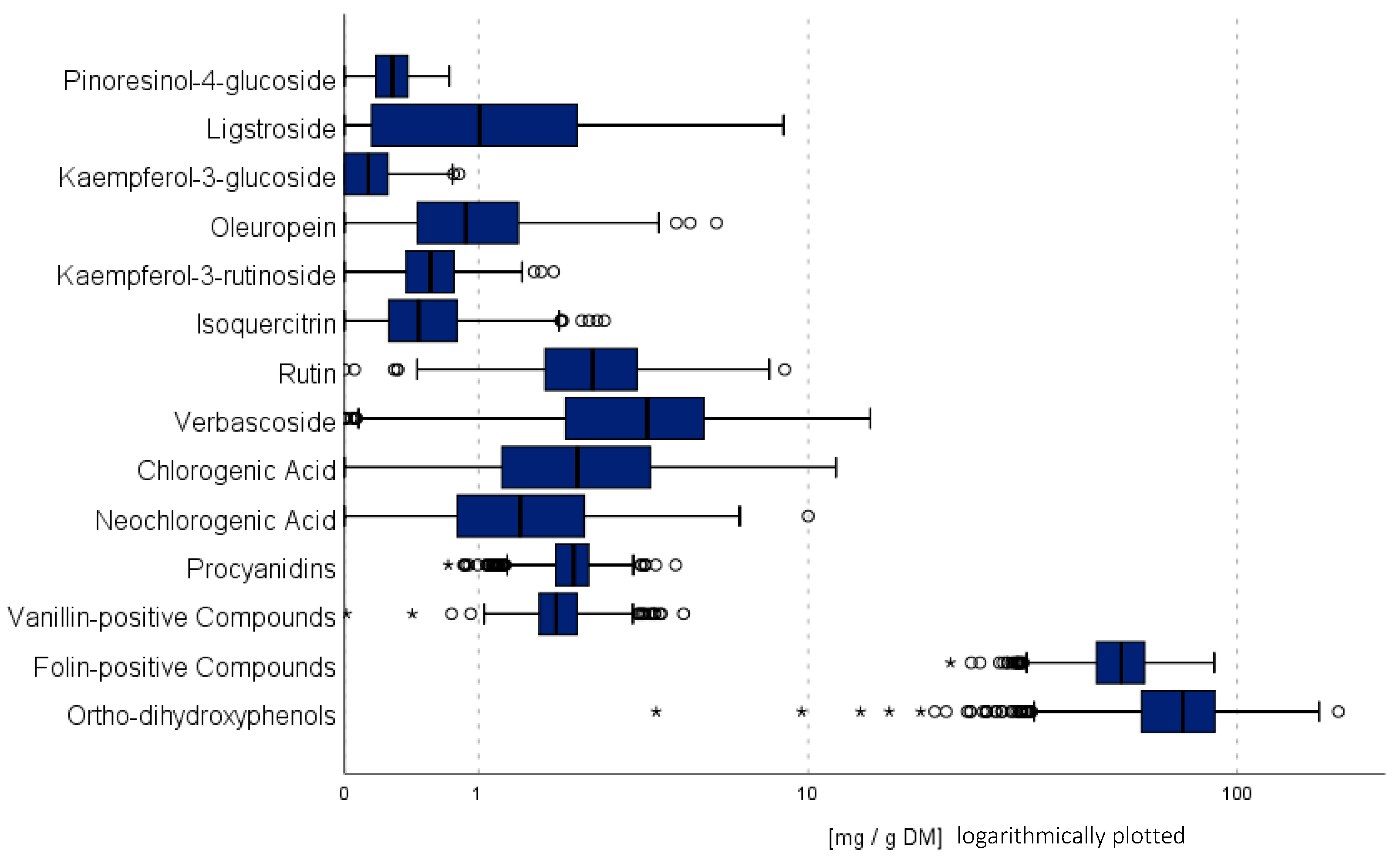
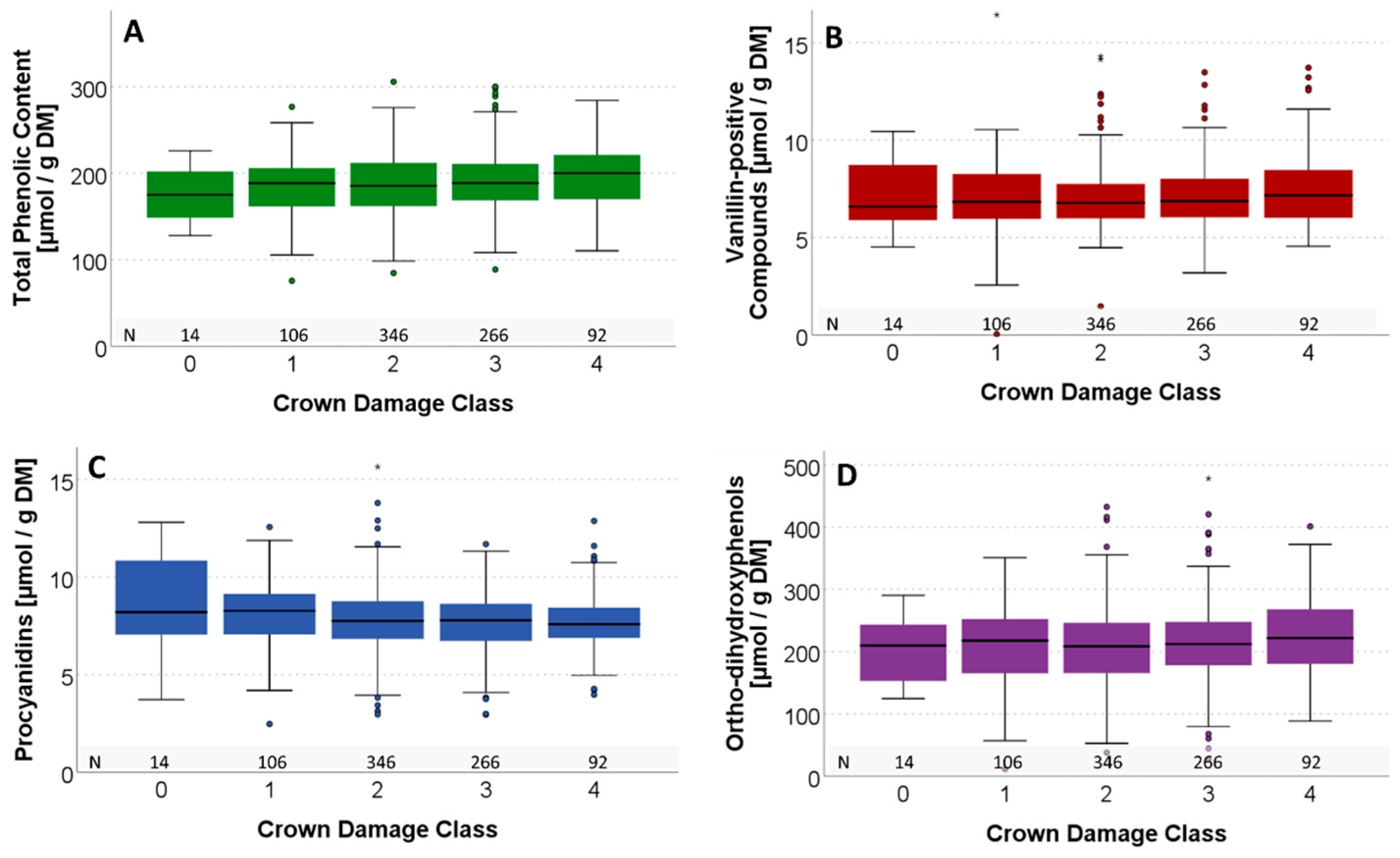
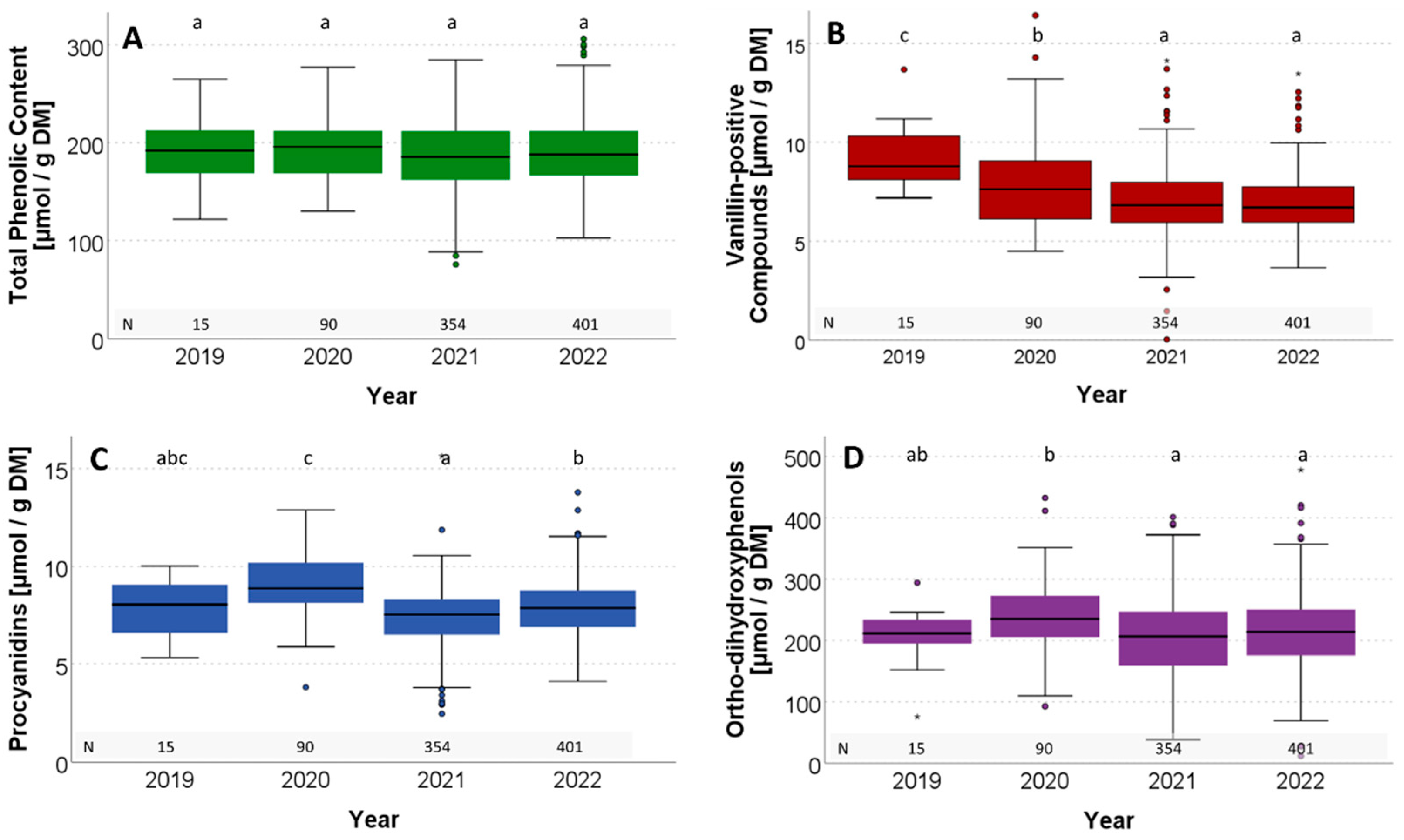
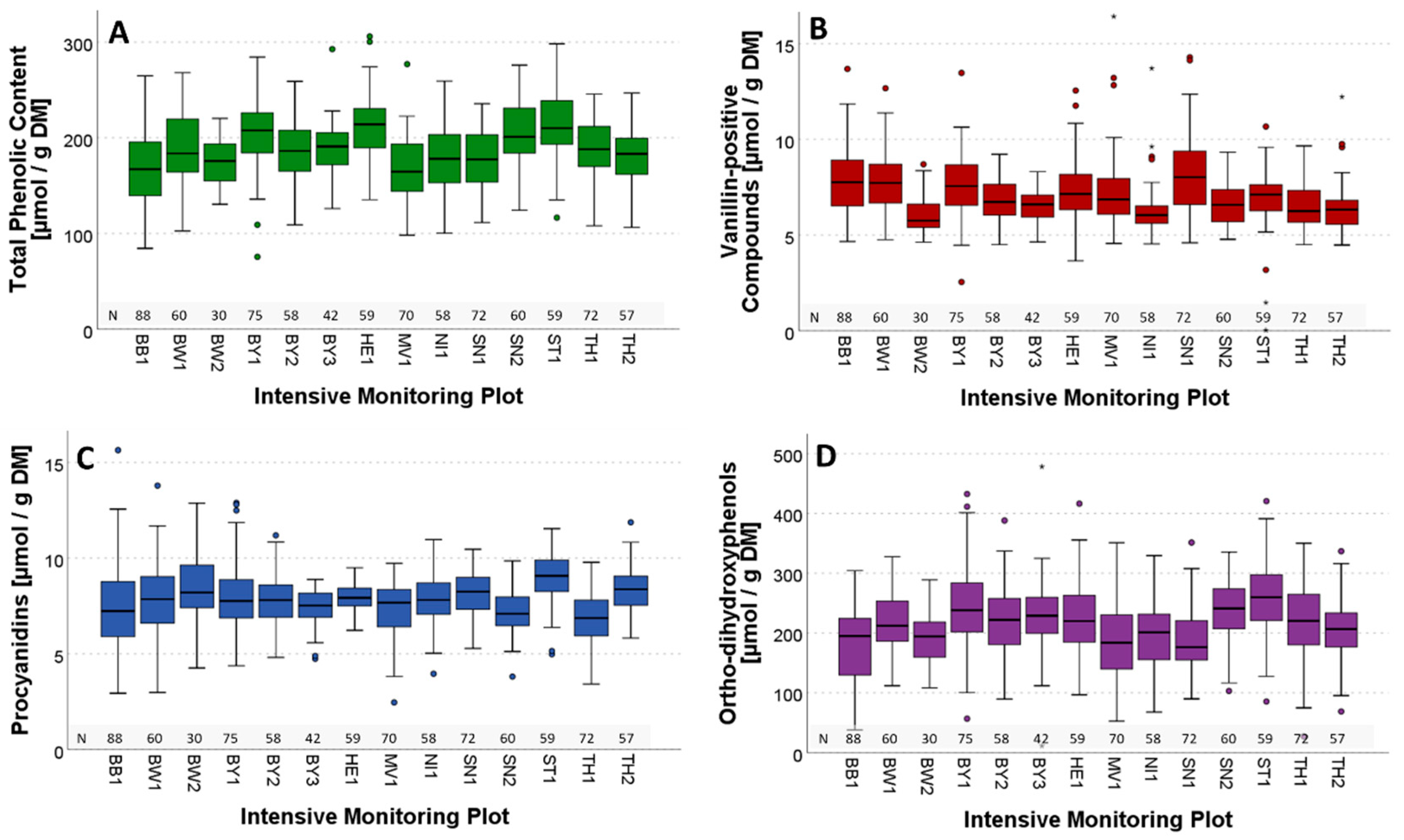
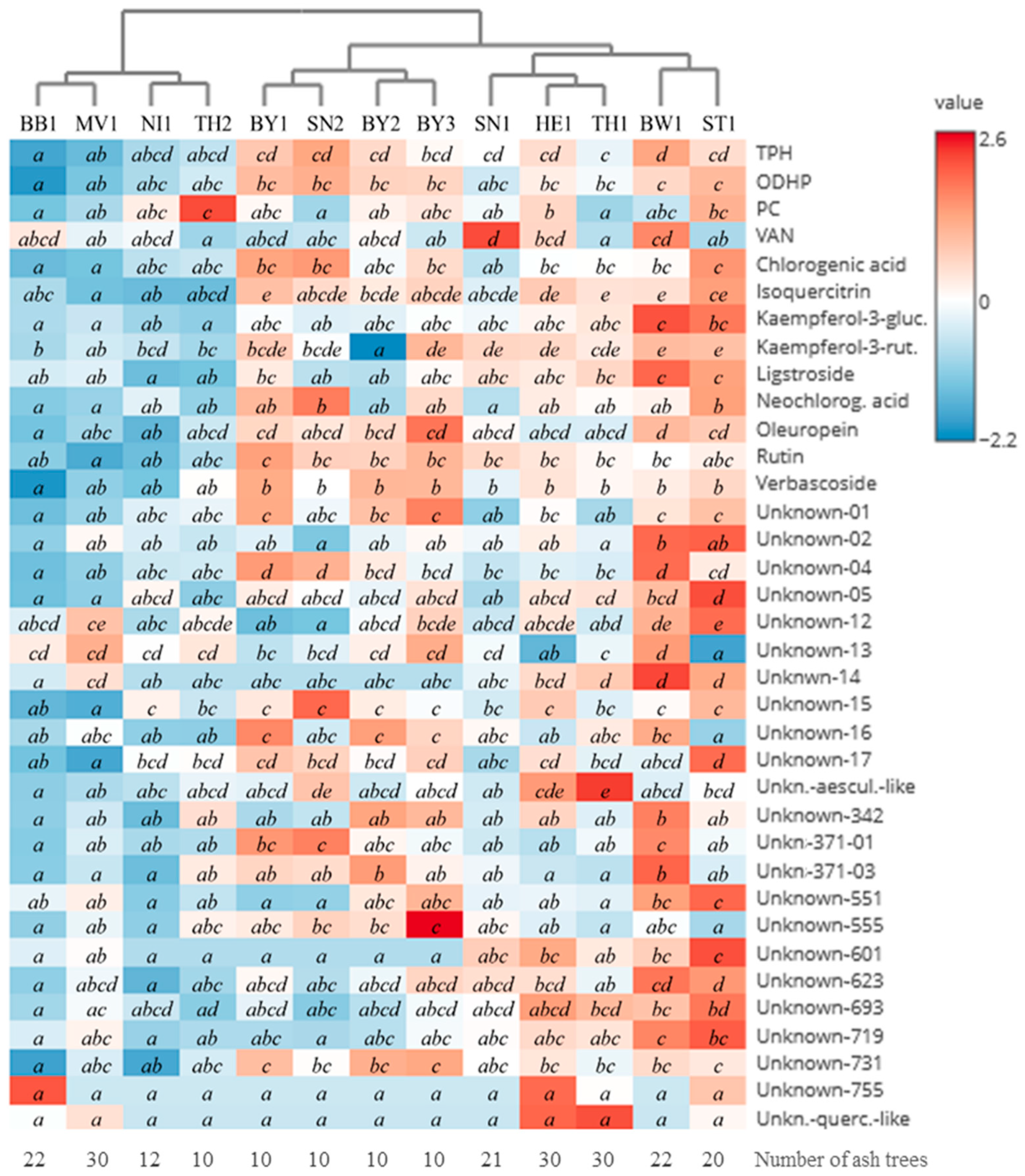
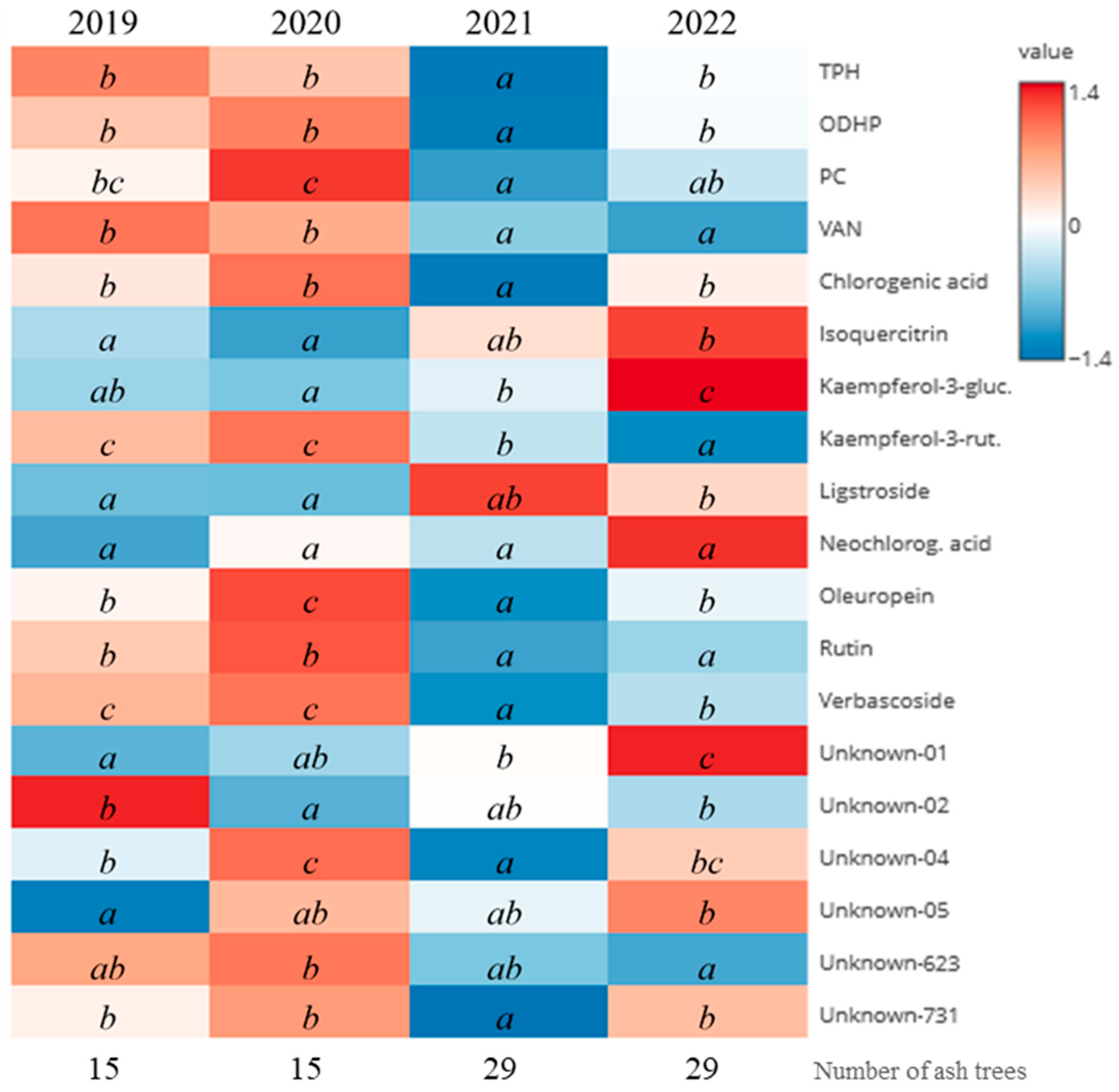
| Year | 2019 | 2020 | 2021 | 2022 | ||||
|---|---|---|---|---|---|---|---|---|
| IBF | Groups | Single | Groups | Single | Groups | Single | Groups | Single |
| BB1 | 15 | 15 | 15 | 15 | 29 | 29 | 29 | 29 |
| BW1 | - | - | - | - | 30 | 22 | 30 | 21 |
| BW2 | - | - | - | - | - | - | 30 | - |
| BY1 | - | - | 15 | 15 | 30 | 10 | 30 | 8 |
| BY2 | - | - | 15 | 15 | 13 | 10 | 30 | 10 |
| BY3 | - | - | - | - | 12 | 10 | 30 | 10 |
| HE1 | - | - | - | - | 30 | 30 | 29 | 14 |
| MV1 | - | - | 15 | 15 | 30 | 30 | 25 | 25 |
| NI1 | - | - | - | - | 30 | 12 | 28 | 11 |
| SN1 | - | - | 15 | 15 | 30 | 21 | 27 | 15 |
| SN2 | - | - | - | - | 30 | 10 | 30 | 10 |
| ST1 | - | - | - | - | 30 | 20 | 29 | 29 |
| TH1 | - | - | 15 | 15 | 30 | 30 | 27 | 15 |
| TH2 | - | - | - | - | 30 | 10 | 27 | 9 |
| TPH | ODHP | PC | VAN | ||||
|---|---|---|---|---|---|---|---|
| ODHP | 0.877 | TPH | 0.877 | Verbascoside | 0.442 | Rutin | 0.347 |
| Chlorogenic acid | 0.676 | Verbascoside | 0.774 | Unknown 623 | 0.372 | TPH | 0.344 |
| Rutin | 0.634 | Chlorogenic acid | 0.689 | Unknown 731 | 0.360 | Kaempferol-3-rutinoside | 0.282 |
| Isoquercitrin | 0.608 | Unknown 623 | 0.681 | Rutin | 0.318 | Pinoresinol-4-glu | 0.215 |
| Unknown 623 | 0.601 | Unknown 731 | 0.638 | 8-hydroxy-pinores-glu | 0.312 | Unknown 623 | 0.208 |
| Verbascoside | 0.577 | Rutin | 0.586 | ODHP | 0.299 | ODHP | 0.201 |
| Kaempferol-3-rutinoside | 0.514 | Unknown 371_01 | 0.563 | Chlorogenic acid | 0.263 | Unknown 719 | 0.189 |
| Unknown 17 | 0.512 | Unknown 02 | 0.521 | Unknown 17 | 0.258 | Unknown 601 | 0.180 |
| Unknown 01 | 0.507 | Unknown 04 | 0.513 | Kaempferol-3-rutinoside | 0.246 | PC | 0.173 |
| Unknown 731 | 0.502 | Unknown 17 | 0.507 | TPH | 0.235 | Unknown 551 | 0.159 |
| Phenolic Parameter | L | Y | L × Y | CDC | L × CDC | Y × CDC | L × Y × CDC | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TPH c | 0.072 | a | 0.000 | 0.072 | a | 0.002 | 0.018 | b | 0.005 | 0.020 | a | |||
| ODHP c | 0.048 | a | 0.005 | b | 0.038 | a | 0.002 | 0.035 | a | 0.006 | b | 0.013 | ||
| PC c | 0.052 | a | 0.028 | a | 0.060 | a | 0.002 | 0.000 | 0.001 | −0.001 | ||||
| VAN c | 0.080 | a | 0.007 | a | 0.054 | a | 0.001 | 0.008 | 0.003 | −0.015 | ||||
| Chlorogenic acid d | 0.056 | a | 0.009 | b | 0.028 | a | 0.002 | −0.001 | 0.004 | −0.004 | ||||
| Isoquercitrin d | 0.052 | a | 0.046 | a | 0.050 | a | −0.002 | 0.014 | −0.004 | −0.003 | ||||
| Kaempferol-3-glucoside d | 0.034 | a | 0.058 | a | 0.043 | a | 0.000 | −0.003 | −0.005 | −0.008 | ||||
| Kaempferol-3-rutinoside d | 0.089 | a | 0.023 | a | 0.040 | a | 0.004 | 0.005 | −0.006 | 0.010 | ||||
| Ligstroside d | 0.048 | a | 0.068 | a | 0.019 | b | −0.002 | −0.010 | −0.006 | −0.011 | ||||
| Neochlorogenic acid d | 0.071 | a | 0.002 | 0.023 | b | 0.006 | 0.006 | 0.003 | −0.007 | |||||
| Oleuropein d | 0.043 | a | 0.026 | a | 0.047 | a | 0.004 | 0.011 | −0.007 | −0.008 | ||||
| Rutin d | 0.017 | b | 0.013 | a | 0.098 | a | 0.005 | 0.019 | −0.006 | −0.001 | ||||
| Verbascoside d | 0.055 | a | 0.039 | a | 0.042 | a | −0.002 | 0.019 | −0.002 | 0.003 | ||||
| Unknown-01 d | 0.071 | a | 0.051 | a | 0.054 | a | −0.001 | 0.038 | a | −0.002 | 0.016 | |||
| Unknown-02 d | 0.121 | a | −0.002 | 0.047 | a | 0.002 | 0.002 | −0.004 | −0.001 | |||||
| Unknown-04 d | 0.126 | a | 0.012 | a | 0.080 | a | −0.001 | 0.021 | b | 0.000 | −0.010 | |||
| Unknown-05 d | 0.115 | a | −0.001 | 0.075 | a | 0.005 | 0.019 | 0.007 | −0.003 | |||||
| Unknown-12 e | 0.075 | a | 0.009 | b | 0.137 | a | 0.003 | −0.003 | 0.003 | −0.007 | ||||
| Unknown-13 e | 0.046 | b | 0.062 | a | 0.106 | a | −0.003 | 0.002 | 0.004 | −0.009 | ||||
| Unknown-14 f | 0.252 | a | n.a. | n.a. | 0.004 | 0.015 | n.a. | n.a. | ||||||
| Unknown-15 e | 0.090 | a | 0.104 | a | 0.040 | a | 0.018 | a | 0.002 | −0.003 | 0.001 | |||
| Unknown-16 e | 0.043 | a | 0.028 | a | 0.126 | a | 0.009 | b | 0.004 | 0.002 | 0.002 | |||
| Unknown-17 e | 0.101 | a | 0.002 | 0.037 | a | −0.002 | 0.010 | −0.004 | −0.016 | |||||
| Unknown-aesculin-like e | 0.033 | a | 0.004 | 0.079 | a | 0.006 | 0.030 | b | −0.003 | −0.002 | ||||
| Unknown-342 e | 0.043 | a | 0.025 | a | 0.019 | b | 0.035 | a | 0.010 | −0.004 | −0.003 | |||
| Unknown-371-01 e | 0.100 | a | 0.058 | a | 0.015 | b | 0.008 | b | 0.010 | −0.003 | −0.014 | |||
| Unknown-371-03 e | 0.051 | a | 0.085 | a | 0.018 | b | 0.014 | a | 0.023 | b | 0.000 | −0.002 | ||
| Unknown-551 e | 0.097 | a | 0.020 | a | 0.048 | a | 0.001 | 0.016 | 0.008 | b | −0.005 | |||
| Unknown-555 e | 0.054 | a | 0.018 | a | 0.017 | b | 0.000 | 0.009 | −0.004 | −0.006 | ||||
| Unknown-601 e | 0.120 | a | 0.055 | a | 0.078 | a | −0.005 | 0.010 | −0.004 | −0.019 | ||||
| Unknown-623 d | 0.099 | a | 0.032 | a | 0.019 | b | 0.003 | −0.004 | 0.000 | −0.011 | ||||
| Unknown-693 e | 0.050 | a | 0.013 | a | 0.011 | 0.006 | −0.002 | −0.004 | −0.005 | |||||
| Unknown-719 g | 0.065 | b | 0.000 | 0.004 | 0.001 | −0.014 | 0.000 | 0.026 | b | |||||
| Unknown-731 d | 0.051 | a | 0.032 | a | 0.055 | a | 0.008 | b | 0.005 | −0.006 | −0.006 | |||
| Unknown-755 e | 0.000 | 0.001 | −0.006 | −0.006 | −0.043 | −0.003 | −0.030 | |||||||
| Unknown-quercetin-like f | 0.037 | b | n.a. | n.a. | −0.007 | 0.050 | n.a. | n.a. | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Häuser, H.; Pilger, A.; Ulrichs, C.; Kätzel, R. Phenolic Leaf Compounds in Ash Trees (Fraxinus excelsior L.) in the Context of Ash Dieback. Forests 2025, 16, 1387. https://doi.org/10.3390/f16091387
Häuser H, Pilger A, Ulrichs C, Kätzel R. Phenolic Leaf Compounds in Ash Trees (Fraxinus excelsior L.) in the Context of Ash Dieback. Forests. 2025; 16(9):1387. https://doi.org/10.3390/f16091387
Chicago/Turabian StyleHäuser, Henriette, Angela Pilger, Christian Ulrichs, and Ralf Kätzel. 2025. "Phenolic Leaf Compounds in Ash Trees (Fraxinus excelsior L.) in the Context of Ash Dieback" Forests 16, no. 9: 1387. https://doi.org/10.3390/f16091387
APA StyleHäuser, H., Pilger, A., Ulrichs, C., & Kätzel, R. (2025). Phenolic Leaf Compounds in Ash Trees (Fraxinus excelsior L.) in the Context of Ash Dieback. Forests, 16(9), 1387. https://doi.org/10.3390/f16091387





