Abstract
This study investigates the host diversity and characteristics of the rare parasitic plant Silverberry-like taxillus (Taxillus yadoriki). Seogwipo, Jeju Island, where T. yadoriki grows naturally, was examined to clarify the diversity of host species, their location, and the part of the tree where T. yadoriki is attached. A total of 687 host trees were found. Taxillus yadoriki was intensively distributed in the lowlands below 200 m a.s.l. The host trees belonged to 40 species, four varieties, three cultivars, and two hybrids from 32 genera and 21 families, of which 23 taxa were identified as host plants for the first time in this study. Incredibly, this plant was found to be able to parasitize a variety of plants from 12 orders. Taxillus yadoriki was found to be parasitic in three orders of Gymnosperms, Ginkgoales, Cupressales, and Pinales, as well as nine orders of Angiosperms, including Magnoliales, Rosales, and Ericales. Rosaceae was the most diverse at 18.0%, and most species were broad-leaved and arboreal. Among them, Cryptomeria japonica and Litsea japonica had the highest frequency of parasitization and T. yadoriki was mainly attached to the upper part of the main tree and the main branch; as a result, T. yadoriki was distributed and showed specificity in the position of disturbance on the host. T. yadoriki’s specific distribution model was built as a Maxent program. The area under the curve of the receiver operation characteristic was 0.948 ± 0.026 (mean ± standard deviation), and the performance of the constructed model was excellent. Of the total eight environmental factors, density of artistic forest and meant temperature of coldest quarter accounted for 75.3% of the total importance, which was the main distribution determinant of species. In the average distribution probability map of T. yadoriki, the critical probability for determining distribution was calculated to be 0.2898 ± 0.1018. Accordingly, the distribution of species is predicted to expand from Jeju Island to the entire southern and southeastern coasts.
1. Introduction
More than 4750 species of parasitic plants have been identified to date, comprising approximately 20 families, representing approximately 1% of all flowering plants [1,2,3]. These species have a wide global distribution and are found in all biomes. According to the type, parasitic plants can be categorized as obligate, facultative, hemi-, holo-, stem-, or root- [4,5,6]. More than 40 species in seven families grow naturally in Korea, and various types grow naturally in all climate zones, from the subtropical to the subarctic zones. Korean parasitic plants include Orobanche coerulescens, Aeginetia indica, and Cuscuta japonica, which are fully parasitic plants (Obligate), Monotropastrum humile and Monotropa hypopithys, which are saprophytes, and mistletoe, which is hemiparasitic [7,8]. Among them, hemiparasitic plants are both parasitic and photosynthetic and survive by attaching parasitic roots to the host plant to obtain moisture and nutrients [2,9]. Mistletoes that grow naturally in Korea include Viscum and Korthalsella in Santalaceae and Loranthus and Taxillus in Loranthaceae (Table 1). The genus Viscum includes Viscum album var. lutescens and Viscum album for. rubroauranticum, which grow naturally in cool and temperate deciduous broad-leaved tree forests and are known to be parasitic on Fagaceae, Betulaceae, and Aceraceae. Korthalsella japonica is distributed in temperate and subtropical regions and is parasitic on Theaceae, Oleaceae, and Lauraceae. Loranthus tanakae is mainly distributed in deciduous broad-leaved tree forests in central and northern temperate regions and is parasitic on Fagaceae and Betulaceae. Finally, T. yadoriki is mainly distributed in evergreen broad-leaved tree forests in temperate and subtropical regions and is parasitic on Lauraceae, Fagaceae, and Theaceae [10,11,12,13,14]. However, little is still known about the host plant diversity and ecological characteristics of the parasitic species.

Table 1.
Diversity of some parasitic plants in Korea.
Taxillus yadoriki, the target species of this study, is a mistletoe with a restricted distribution in Japan and Korea in Northeast Asia, and in Korea it grows naturally only on Jeju Island [11,13,15]. Due to the species’ narrow distribution range and small population size, it is designated as a critically endangered species by the Korea Forest Service and as Near Threatened according to the Ministry of Environment’s Red List Categories [16,17]. Due to the parasitic nature of the plant, which inhibits the growth of the host plant, it is recognized as an invasive plant to be removed from orchards and artificial forests that are managed for special purposes [18,19].
Like many other mistletoes, T. yadoriki has a long history of use in folk botany as a medicinal plant [14]. Recent studies have confirmed that it contains anti-cancer and anti-inflammatory substances. Notably, the diversity and content of medicinal substances in parasitic plants have been found to vary greatly depending on the type of host plant [20,21,22]. These results suggest that the potential of T. yadoriki as a medicinal plant is expected to vary depending on the host plant diversity. However, data on the host plant diversity of T. yadoriki are limited.
Therefore, this study determines the distribution status of T. yadoriki in Korea, as well as the diversity and host characteristics of its host plants. The host plant diversity identified for the first time in this study was described through comparison with the literature. Furthermore, by confirming which part is preferred when attaching to the host plant, we intend to not only reveal the attachment characteristics but also use it as basic information for management. In addition, we plant to use existing distribution information to derive environmental factors that determine the distribution of species and use these to analyze potential distribution sites for species. Our results will help to enhance the utilization potential of T. yadoriki, whose medicinal components vary depending on the host plant, and serve as a basis for the stable conservation and management of rare plants.
2. Materials and Methods
2.1. Field Research
Jeju Island (Jeju Special Self-Governing Province) is the southernmost part of the Korean Peninsula and has an area of approximately 1850 km2. It is and oval-shaped island that is wide form east to west. The climate is characterized by high temperature and precipitation in summer and low temperature and dry climate in winter (Figure 1).
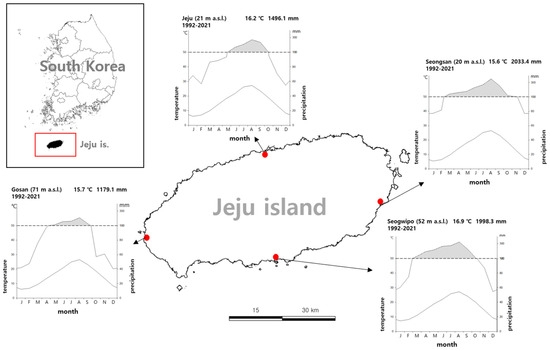
Figure 1.
Geography and bioclimate of the study area (1992–2021).
A total of eight field surveys were conducted from January to May 2021 on Jeju Island: 4 times in January, 3 times in February, and 1 time in May. The entire island was surveyed to determine the distribution range of the species, and GPS values were calibrated and recorded for the points where the species distribution was confirmed. Field identifications were made to determine host plant diversity, and photographic data, including geographic information, were stored. When precise identification through specimens was required, species were collected, evidence specimens were prepared, and the specimens were deposited in the Warm Temperate and Subtropical Forest Research Center herbarium after identification. The scientific names of the species were based on the Korean Standardized Inventory of Plants [17], and the original color Korean Flora [23] and original color Korean Standardized Flora [24] were considered. To identify the characteristics of the species’ attachment to the host plant, we carefully recorded the attached position and ecological characteristics of the host plant. The positions of attachment were categorized as trunk, branch, and twig according to the characteristics of the branch and recorded as the upper part, middle part, and lower part according to the position in the tree. The host plant was categorized according to taxonomic position, growth form [25], and disseminule forms [26].
2.2. Distribution Modeling
2.2.1. Data Collection
A species distribution model was created to identify potential distribution sites using the current distribution location of T. yadoriki. The scope of application of the species distribution model was limited to Jeju Island, and the coordinates collected through field surveys were used for analysis of species distribution information. The environmental data consisted of major climate data considering the characteristics of the native habitat of T. yadoriki and land cover data reflecting the habitat and host plant characteristics. For climate data, WorldClim ver. 2.1 bioclimatic variables (30″ × 30″) were used [27]. WorldClim consists of 19 variables that affect biological distribution.
For land cover data, the Ministry of Environment’s land cover map was used [28]. The land cover map was reorganized into six legends, considering the diversity and density of plants that T. yadoriki can host for each legend. Then, after creating a grid with the same pixel size as the climate data, the area of the land cover legend reconstructed from each grid was calculated, and then the density (0–1) was calculated as a percentage (Figure S1). The types and descriptions of the reconstructed land cover are as follows: (1) Barren land, being an area where vegetation is not covered; among the legends of the land cover map, ‘used area’ and ‘barren’ are included. (2) Artificial grassland, being an artificial grassland; this area occasionally includes artificially planted trees. (3) Natural grassland, being a natural grassland; this area occasionally includes artificially planted trees as well as rare, native trees, and includes natural grasslands and wetlands among the land cover map legends. (4) Artificial forest, being an artificially created forest composed mainly of an orchard cultivated on Jeju Island and a windproof planting forest surrounding it; forest density is relatively high, but diversity is low. (5) Natural forest, being Jeju Island’s native vegetation area; this area includes evergreen forests and coniferous forests and consists of high-density trees. (6) Aquatic area, being an area composed of water including rivers and lakes.
2.2.2. Data Processing and Selection
The collected environmental data were cropped to Jeju Island, the scope of the model, with QGIS ver. 3.28.12 and converted to rasters with a resolution of 30″ × 30″ [QGIS.org, Zürich, Swiss] [29], and correlation analysis was performed with the cor.test function in R ver. 4.3.2 to consider collinearity among variables [R Core Team, Vienna, Austria] [30,31,32]. The correlation of climate variables in Jeju Island was high among annual and quarterly temperature and precipitation data (B01, B05, B06, B08, B09, B10, B11, B12, B13, B14, B16, B15, B17, B18, B19) and temperature deviation data (B02, B03, B04, B07) (Table S1). In our variable selection, we wanted to reflect the characteristics of a temperate plant. Therefore, we selected ‘Mean temperature of coldest quarter’ for annual and quarterly temperature and precipitation data, and ‘Temperature annual range’ for temperature anomaly data. Meanwhile, the land cover variables were independent, so all variables were selected. In the end, eight variables were used in the modeling (Figure S1).
2.2.3. Modeling
We believe that our island-wide field surveys have identified the majority of T. yadoriki distributions on Jeju Island, but there may still be residual potential T. yadoriki distributions. Therefore, we used the maximum entry model algorithm, which allows effective modeling even with occurrence data [33,34].
The maximum entry model algorithm was performed with Maxent software ver. 3.4.4 (New York, NY, USA) [35]. During modeling, parameters were adjusted for model validation and ecological interpretation. ‘Replicated run type’ was set to ‘Crossvalidate’ and ‘Replicates’ was set to ‘10’ for 10-fold cross validation. ‘Create response curves’ and ‘Do jacknife to measure variable importance’ were enabled to explore the relationship between T. yadoriki and its environment. ‘Output format’ was set to ‘Logistic’. The performance of the constructed model was evaluated by the area under the curve of the receiver operation characteristic. It has a value of 0.5–1, and the closer it is to 1, the better the model [36,37,38]. The model variables were evaluated using importance. In the calculated average distribution probability map, if the probability was higher than the maximum test sensitivity plus specificity logistic threshold, it was judged as a potential distribution possible area [39].
3. Results and Discussion
3.1. Distribution Range of Native Species
The native distribution of T. yadoriki was limited to the Seogwipo area on Jeju Island, and the distribution was confirmed at 687 points. T. yadoriki was found at altitudes ranging from 7 to 194 m above sea level, with an average altitude of 67.4 m, and was concentrated in the lowlands. The horizontal distribution range was confirmed from Andeok-myeon Andeok valley area (33°15′26″ N 126°21′23″ E), the westernmost part, to Namwon-eup (33°15′50″ N 126°37′9″ E), the easternmost part, and from Yeongcheon-dong area (33°17′55″ N 126°35′32″ E), the northernmost part to Songsan-dong area (33°14′20″ N 126°36′5″ E), the southernmost part (Figure 2). The average annual temperature (1992–2021) at the Seogwipo ASOS site (52 m above sea level) adjacent to the distribution site was 16.9 °C with an annual precipitation of 1989.3 mm (Figure 1 and Figure 2, [40]), and the area is considered to have a subtropical climate with a temperature index of 199.6. The region has the warmest climate in Korea, being the only region where the average daily temperature in winter is not below zero [40].
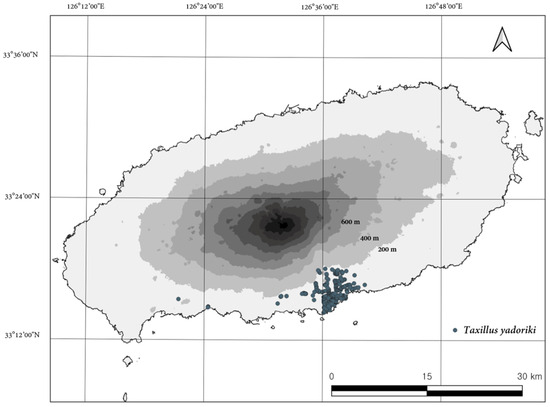
Figure 2.
Distribution of T. yadoriki in Jeju, Republic of Korea.
Parasitized trees and locations were diverse, including street trees, windbreaks, landscape trees, fruit trees, and natural forests, but high frequencies were found in croplands, roadsides, and garden green areas where anthropogenic activities are intensive. Most of these species occur as stand-alone trees or exhibit a linear distribution, and in many cases, are vegetated landscapes that play various roles, such as local bird migration corridors, refuges, and sanctuaries. Due to these native distribution characteristics, T. yadoriki has one of the narrowest distribution ranges among mistletoe species in Korea, which contrasts with other mistletoe species that are mainly distributed in natural forests composed of long- to old-growth trees [11,41,42,43].
3.2. Host Plant Diversity and Species Characteristics
T. yadoriki was found to be able to parasitize a variety of plants from 12 orders. The identified host plants belonged to 49 taxa (40 species, four varieties, three cultivars, and two hybrids) in 32 genera and 21 families. Phylogenetically, T. yadoriki was found to be parasitic in three orders of Gymnosperms, Ginkgoales, Cupressales, and Pinales, as well as nine orders of Angiosperms, including Magnoliales, Rosales, and Ericales. Although these host plants show great differences in the structural aspect of sap movement, the fact that these species can parasitize is a very specific phenomenon. In detail, three taxa, three genera, and three families (Ginkgo biloba, Cryptomeria japonica, and Torreya nucifera) in Gymnosperma and 46 taxa, 28 genera, and 17 families of Lauraceae, Fagaceae, and Rosaceae in Dicotyledon among Angiosperma were identified as host plants. This was 3.3 times higher than the host plant diversity of 15 taxa, 14 genera, and eight families recorded in previous studies in Korea for T. yadoriki. We identified 23 taxa of new host plants [17,20,44,45]. This value was also the highest host plant diversity compared to other mistletoes in Korea, confirming that T. yadoriki utilizes a wide range of host plants in Korea (Table 2). Internationally, T. yadoriki host plant diversity is known for 57 taxa, 53 species, three variants, and one hybrid in 38 genera and 26 families (Table 3). Of these, 24 families have species in common with Korea. However, seven families, including Illiciaceae, Moraceae, and Myrtaceae; 12 genera, including Ficus, Ulmus, and Litsea; and 25 taxa, including Torreya nucifera, Eriobotrya japonica, Cornus kousa, and Nerium oleander, were identified as host plants only in Korea, and our report first records their host potential. Among the foreign host plants, Quercus gilva, Q. acuta, Carpinus tschonoskii, Toxicodendron succedaneum, Symplocos prunifolia, and Pittosporum tobira are species that grow naturally in Korea and are likely to contribute as host plants in Korea, although parasitism was not confirmed in this study [10,12,46,47].

Table 2.
Host plant diversity of T. yadoriki in Korea (○: first record).

Table 3.
Comparison of host plant diversity between Korea and other countries (+: present).
The newly identified host plants in this study include 17 genera, including Torreya, Cornus, Elaeagnus, and Magnolia, and 25 taxa, including Ginkgo biloba, Prunus spachiana f. ascendens, Ulmus parvifolia, Elaeagnus umbellata, and Illicium anisatum. The diversity of host plants varies by family, with nine taxa (18.0%) in Rosaceae, six taxa (12.0%) in Lauraceae, and five taxa (10.0%) in Theaceae. However, there are 13 taxa in one genus and one family, accounting for 26.0% of the total, suggesting that the phylogenetic dependence on host plants is not high.
The growth form of the host plant was dominated by arboreal and sub-arboreal trees with 40 taxa (80.0%) and was parasitic on shrubby trees (10 taxa, 20.0%), including Elaeagnus umbellata, Litsea japonica, and Ficus erecta Thunb. (Table 4). The average height of the host tree was 6.2 m, with a range of sizes from 1.5 to 15 m.

Table 4.
Ecological characteristics of host plants and parasitic character of T. yadoriki.
Among the host plants, native, cultivated, and planted species accounted for 66.0%, 18.0%, and 16.0%, respectively. The highest dispersal type of host plant was animal dispersal, with 60.0% (30 taxa). Coniferous and evergreen broad-leaved trees and deciduous broad-leaved trees accounted for 6.0%, 44.0%, and 50.0%, respectively. Species with a relatively high frequency of parasitization by T. yadoriki were Cryptomeria japonica (189 sites), Litsea japonica (109 sites), Prunus × yedoensis (88 sites), Machilus thunbergii (66 sites), Neolitsea sericea (61 sites), and Camellia japonica (23 sites). Meanwhile, there were some species with high occurrence in the study area but that were not identified as parasitic, such as Ilex rotunda, Pinus thunbergii, Melia azedarach, Elaeocarpus sylvestris var. ellipticus, and Cinnamomum camphora (one individual). Mistletoe has host specificity and preference through the evolutionary process of speciation [5,48,49,50], and some species are thought to have host-plant exclusivity of T. yadoriki.
3.3. Attachment Characteristics within the Host Plant
Attachment of T. yadoriki was identified on host plants of 48 taxa, 39 species, four varieties, three cultivars, and two hybrids from 31 genera and 21 families.
The distribution of the attachment location of T. yadoriki based on the host plant’s tree type was 62.7% (431 individuals) in the middle part, 31.7% (218 individuals) in the upper part, and 5.5% (38 individuals) in the lower part. The distribution of branch types with parasitic attachment was highest at 58.1% (399) on the branch, followed by 20.7% (142) on the twig and 21.3% (146) on the trunk (Table 5). Different tree species showed differences in attachment characteristics. For Cryptomeria japonica, a candelabra-type tree with a relatively upright trunk and a long main stem, T. yadoriki was mainly distributed in the trunk and middle part. Zelkova-type trees with widely spreading branches, Quercus acutissima, Quercus glauca, Quercus serrata, Magnolia kobus, Neolitsea sericea, showed attachment of T. yadoriki on these branches, while other shrubby species with relatively small tree heights showed a predominant attachment of T. yadoriki to the upper part and main branches.

Table 5.
Diversity, frequency and ecological characters of host plant of T. yadoriki in Korea (* new record).
These attachment traits are thought to be highly correlated with the characteristic that T. yadoriki dispersal is driven by avian feeding. The tendency to attach is considered to occur at a high frequency in areas with high branch density and suitable conditions for birds. It is necessary to conduct further studies on the bird diversity involved in the dispersal of T. yadoriki, the effects of bird feeding and digestion on germination, the external morphological characteristics of seed attachment sites on seed germination and settlement, and pharmacological activity, depending on the host plant.
3.4. Species Distribution Model
T. yadoriki’s specific distribution model was built as a maximum entry algorithm. The area under the curve of the receiver operation characteristic was 0.948 ± 0.026 (mean ± standard deviation), and the performance of the constructed model was excellent. Of the total eight environmental factors, ‘density of artistic forest’ and ‘meant temperature of coldest quarter’ accounted for 75.3% of the total importance, which was the main distribution determinant of T. yadoriki (Figure 3). The most important distribution determinant was ‘density of artistic forest’ (52.3% ± 2.0%). And mean temperature of coldest quarter was the second most important determinant of distribution (23.0% ± 4.3%). The distribution probability of species tended to be proportional to the ‘density of artificial forest’. The distribution probability of T. yadoriki then increased rapidly when the ‘mean temperature of coldest quarter’ was above 5–6 °C.
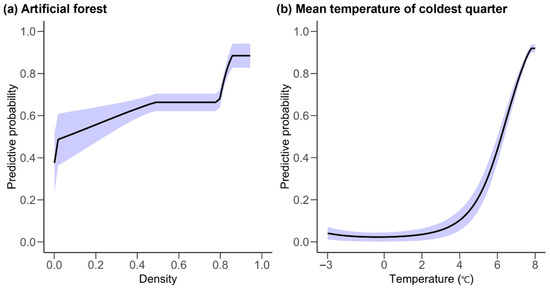
Figure 3.
The response curve showing the density of artificial forests (a) and mean temperature of coldest quarter (b), which are the main distribution factors of Taxillus yadoriki. The line represents the mean, and the blue ribbon represents the standard deviation.
In the average distribution probability map of species, the critical probability for determining distribution was calculated to be 0.2898 ± 0.1018. Therefore, T. yadoriki is predicted to be distributed throughout the south and southeast coasts of Jeju Island (Figure 4).
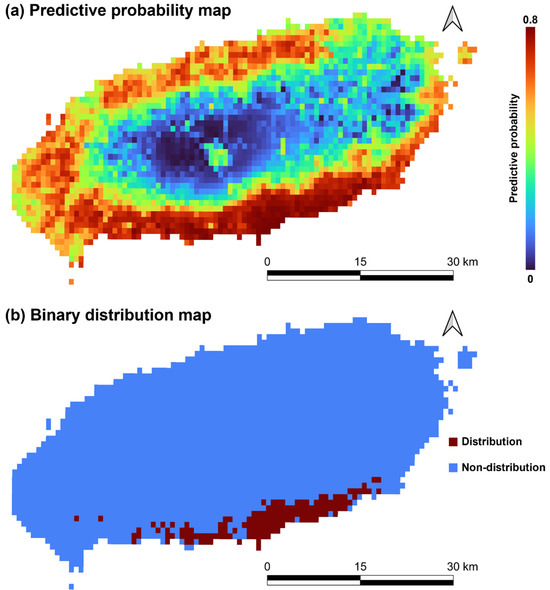
Figure 4.
Probability map (a) and binarized distribution map (b) predicted by Maxent of Taxillus yadoriki in Jeju Island. Binarization was done with maximum test sensitivity plus specificity logistic threshold.
The constructed species distribution model was complementary to the field research results. In the modeling, T. yadoriki preferred artificial forests. This reflects the field surveys’ preference for small, human-managed forests and populations over natural forests, and is consistent with the characteristics associated with dispersal by small birds. For these small birds, artificial forests, such as orchards, provide protection from their natural enemies, large animals, while also providing an abundance of food [51,52]. The proportional relationship between the probability of distribution of T. yadoriki and the mean temperature of the coldest quarter also reflects the fact that the areas where T. yadoriki occurred in field surveys do not drop below freezing in winter. The model refines this further and suggests a quantitative distribution limit temperature of 5–6 °C. Finally, the species distribution model predicts that the distribution of T. yadoriki extends beyond the southern and southwestern distributions of Jeju Island, as well as the southern and southeastern coasts, which were the areas investigated by field researchers. This is because the low-elevation coastal plain of Jeju Island, with the exception of the northeast, is covered by operational orchards, providing physical habitat for T. yadoriki, but its distribution is limited by the lowest temperatures of the year.
4. Conclusions
The host plants of Taxillus yadoriki were diverse, including 49 taxa, 32 genera, and 21 families. Twenty-three host plants were identified for the first time. In Korea, T. yadoriki was limited to the Seogwipo area and was mainly parasitic on street trees and landscape trees subjected to anthropogenic disturbance. While the host plant species diversity was high, the frequency of occurrence was particularly high for certain species, including Cryptomeria japonica and Litsea japonica. T. yadoriki was commonly attached to both woody and broadleaf host plants, with the most frequent attachment location being the middle part of the host plant and the most frequent attachment site being the branch. In addition, as there were tree species not specifically parasitic in the area where they grow naturally, they were determined to have host specificity and exclusivity. The field survey results were supported using a species distribution model, and potential distribution areas were predicted. T. yadoriki requires the presence of woody plants on which it can parasitize, as well as temperatures that do not fall below freezing throughout the year in habitats subject to artificial disturbance. Accordingly, it was predicted that the distribution would expand to the south and southeast coasts of Jeju Island, which is suitable for the distribution determinants. Further studies are needed to analyze the potential distribution and cultivation sites of T. yadoriki, which is highly valued as a medicinal plant, as well as the variation of pharmacological activity according to the host plant, alongside cultivation technology and management techniques.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/f15050799/s1, Table S1: Summary of Correlations between 25 environmental variables. Figure S1: Diversity in environmental factors used in species distribution model for Taxillus yadoriki in Jeju Island.
Author Contributions
Conceptualization, B.C.; methodology, C.L. and B.C.; Software, C.L. and B.C.; formal analysis and investigation, B.C. and E.P.; resources, B.C. and E.P.; data curation, B.C. and C.L.; writing—original draft preparation, B.C. and C.L.; writing—review and editing, B.C., C.L., E.P. and E.Y.; visualization C.L.; project administration, B.C. and E.Y.; funding acquisition, B.C. and E.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This study was conducted with the support of the “Research on the establishment of foundations for conservation and utilization of useful forest life resources in the warm-temperate and subtropical regions (’23), National Institute of Forest Science (FE0200-2021-01-2023)”. Provided by the Korea Forest Service (Korea Forestry Promotion Institute).
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available since these are research data conducted as a project for obtaining specific research results and intellectual property rights at the Nation Institute of Forest Science. When the project is completed, it is planned to be publicly provided through the institution’s original and independent system.
Acknowledgments
We would like to thank J.E. Yang for assisting with the field survey and data organization regarding the distribution and characteristics of the species.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Musselman, L.J.; Press, M.C. Introduction to parasitic plants. In Parasitic Plants; Pres, M.C., Graves, J.D., Eds.; Springer Science & Business Media: Berlin/Heidelberg, Germany, 1995; pp. 1–13. [Google Scholar]
- Nickrent, D.L. Parasitic plants of the world. In Parasitic Plants of the Iberian Peninsula and Balearic Islands; López-Sáez, J.A., Catalán, P., Sáez, L., Eds.; Mundi-Prensa Libros: Madrid, Spain, 2002; pp. 7–27. [Google Scholar]
- Nickrent, D.L. Parasitic angiosperms: How often and how many? Taxon 2020, 69, 5–27. [Google Scholar] [CrossRef]
- Douglas, A.E. Symbiotic Interactions; Oxford University Press: Oxford, UK, 1994. [Google Scholar]
- Thompson, R.L.; Poindexterss, D.B. Host specificity of American mistletoe (Phoradendron leucarpum, Viscaceae) in Garrard County, Kentucky. J. Kentucky Acad. Sci. 2005, 66, 40–43. [Google Scholar] [CrossRef]
- Twyford, A.D. Parasitic plants. Curr. Biol. 2018, 28, 857–859. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.D. Santalaceae R.Br. In The Genera of vascular Plants of Korea; Park, C.W., Ed.; Academy Publishing, Co.: Seoul, Republic of Korea, 2007; pp. 653–655. [Google Scholar]
- Kim, Y.D. Loranthaceae Juss. In The Genera of vascular Plants of Korea; Park, C.W., Ed.; Academy Publishing Co.: Seoul, Republic of Korea, 2007; pp. 656–657. [Google Scholar]
- Těšitel, J. Functional biology of parasitic plants: A review. Plant Ecol. Evol. 2016, 149, 5–20. [Google Scholar] [CrossRef]
- Fukunaga, T.; Nishiya, K.; Kajikawa, I.; Takeya, K.; Itokawa, H. Studies on the constituents of Japanese mistletoes from different host trees, and their antimicrobial and hypotensive properties. Chem. Pharm. Bull. 1989, 37, 1543–1546. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.S.; Kim, S.Y.; Sun, B.Y.; Yi, J.S. A review of the taxonomic and ecological characteristics of Korean mistletoe types (Viscum, Korthalsella, Loranthus and Taxillus). Korean J. Plant Tax. 2013, 43, 81–89. [Google Scholar] [CrossRef]
- Ishida, H.; Kuroda, A.; Iwakiri, K. Overgrowth of the hemiparasitic plant species Taxillus yadoriki (Loranthaceae) and the debilitation of its host trees in a park in Miyazaki City, Miyazaki Prefecture. Veg. Sci. 2016, 33, 15–32. [Google Scholar]
- GBIF (Global Biodiversity Information Facility). Biodiversity Research. 2022. Available online: https://www.gbif.org (accessed on 12 December 2022).
- NIBR (National Institute of Biological Resources). Species Information Search. 2022. Available online: https://www.nibr.go.kr (accessed on 12 December 2022).
- Satake, Y.; Hara, H.; Watari, S.; Tominari, T. Wild Flowers of Japan (Woody Plants); Heibonsha: Tokyo, Japan, 1989; pp. 100–102. [Google Scholar]
- KNA (Korea National Arboretum). Rare Plants Data Book in Korea; Korea National Arboretum: Seoul, Republic of Korea, 2008. [Google Scholar]
- KBIS (Korea Biodiversity Information System). Species Information Search. 2022. Available online: http://www.nature.go.kr (accessed on 12 December 2022).
- Griebel, A.; Watson, D.; Pendall, E. Mistletoe, friend and foe: Synthesizing ecosystem implications of mistletoe infection. Environ. Res. Lett. 2017, 12, 115012. [Google Scholar] [CrossRef]
- Bell, D.M.; Pabst, R.J.; Shaw, D.C. Tree growth declines and mortality were associated with a parasitic plant during warm and dry climatic conditions in a temperate coniferous forest ecosystem. Glob. Chang. Biol. 2020, 26, 1714–1724. [Google Scholar] [CrossRef]
- Park, S.B.; Park, G.H.; Kim, H.N.; Son, H.J.; Song, H.M.; Kim, H.S.; Jeong, J.B. Anti-inflammatory effect of the extracts from the branch of Taxillus yadoriki being parasitic in Neolitsea sericea in LPS-stimulated RAW264. 7 cells. Biomed. Pharmacother. 2018, 104, 1–7. [Google Scholar] [CrossRef]
- Park, S.B.; Park, G.H.; Kim, H.N.; Song, H.M.; Song, H.J.; Park, J.A.; Kim, H.S.; Jeong, J.B. Ethanol extracts from the branch of Taxillus yadoriki parasitic to Neolitsea sericea induces cyclin D1 proteasomal degradation through cyclin D1 nuclear export. BMC Complement. Altern Med. 2018, 18, 189. [Google Scholar] [CrossRef] [PubMed]
- Park, S.B.; Kim, H.N.; Kim, J.D.; Park, G.H.; Eo, H.J.; An, M.H.; Jeong, J.B. Inhibitory effect of the branch extracts from Taxillus yadoriki parasitic to Neolitsea sericea against the cell proliferation in human lung cancer cells, A549. Korean J. Plant Res. 2019, 32, 109–115. [Google Scholar]
- Lee, T.B. Illustrated Flora of Korea; Hyangmunsa: Seoul, Republic of Korea, 2004. [Google Scholar]
- Lee, W.T. Lineamenta Flora Korea; Academy Press: Seongnam, Republic of Korea, 1996. [Google Scholar]
- Mucina, L.; Rutherford, M.C.; Powrie, L.W. The logic of the map: Approaches and procedures. In The Vegetation of South Africa, Lesotho and Swaziland; Mucina, L., Rutherford, M.C., Eds.; South African National Biodiversity Institute: Pretoria, South Africa, 2006; pp. 13–30. [Google Scholar]
- Numata, M. Ecological judgement of grassland condition and trend: I. Judgement by biological spectra. J. Jpn. Soc. Grassl. Sci. 1965, 11, 20–33. [Google Scholar]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- MoE (Ministry of Environment) Land Cover Map. 2022. Available online: https://egis.me.go.kr (accessed on 12 December 2022).
- QGIS.org. QGIS Geographic Information System. Available online: http://www.qgis.org (accessed on 1 March 2024).
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org (accessed on 1 March 2024).
- Padalia, H.; Srivastava, V.; Kushwaha, S.P.S. Modeling potential invasion range of alien invasive species, Hyptis suaveolens (L.) Poit. in India: Comparison of MaxEnt and GARP. Ecol. Inform. 2014, 22, 36–43. [Google Scholar] [CrossRef]
- Negrini, M.; Fidelis, E.G.; Picanço, M.C.; Ramos, R.S. Mapping of the Steneotarsonemus spinki invasion risk in suitable areas for rice (Oryza sativa) cultivation using MaxEnt. Exp. Appl. Acarol. 2020, 80, 445–461. [Google Scholar] [CrossRef] [PubMed]
- Elith, J.H.; Graham, C.P.; Anderson, R.; Dudík, M.; Ferrier, S.; Guisan, A.E.; Zimmermann, N. Novel methods improve prediction of species’ distributions from occurrence data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Ngarega, B.K.; Chaibva, P.; Masocha, V.F.; Saina, J.K.; Khine, P.K.; Schneider, H. Application of MaxEnt modeling to evaluate the climate change effects on the geographic distribution of Lippia javanica (Burm. f.) Spreng in Africa. Environ. Monit. Assess. 2024, 196, 62. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.J. A Brief Tutorial on Maxent. Network of Conservation Educators and Practitioners, Center for Biodiversity and Conservation, American Museum of Natural History. Lessons Conserv. 2009, 3, 108–135. [Google Scholar]
- Swets, J.A. Measuring the accuracy of diagnostic systems. Science 1988, 240, 1285–1293. [Google Scholar] [CrossRef]
- Elith, J.; Phillips, S.J.; Hastie, T.; Dudík, M.; Chee, Y.E.; Yates, C.J. A statistical explanation of MaxEnt for ecologists. Divers. Distrib. 2011, 17, 43–57. [Google Scholar] [CrossRef]
- Shi, J.; Xia, M.; He, G.; Gonzalez, N.C.; Zhou, S.; Lan, K.; Ouyang, L.; Shen, X.; Jiang, X.; Cao, F.; et al. Predicting Quercus gilva distribution dynamics and its response to climate change induced by GHGs emission through MaxEnt modeling. J. Environ. Manag. 2024, 357, 120841. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.R.; Hernández, M.J.P.; Báez, W.L.; Ramos, J.H.; Flores, H.J.M.; Uicab, J.V.C.; Santos, M.D.R. The ecological niche as a tool for predicting potential areas of two pine species. Mex. J. For. Sci. 2018, 9, 47–68. [Google Scholar]
- KMA (Korea Meteorological Administration). Climate Data. 2022. Available online: https://www.weather.go.kr (accessed on 12 December 2022).
- Lee, S.G. Distribution, habitat characteristics and assessment of the conservation status of a rare mistletoe species, Loranthus tanakae (Loranthaceae) in Korea. J. Korean Forest Soc. 2013, 102, 428–436. [Google Scholar]
- Lee, B.D. Ecological environment of native habitats and host plant in mistletoe (Viscum album var. coloratum). Korean J. Plant Res. 2009, 22, 389–393. [Google Scholar]
- Venturelli, M. Estudos sobre Struthanthus vulgaris Mart.: Anatomia do fruto e semente e aspectos de germinac¸ a. o, crescimento e desenvolvimento. Rev. Bras. Bot. 1981, 4, 131–147. [Google Scholar]
- Kim, T.Y.; Kim, J.S. Woody Plants of the Korean Peninsula; Dolbegae: Paju, Republic of Korea, 2018. [Google Scholar]
- Choi, K.; Park, K.W.; Kim, H.J.; Lee, J.D.; Koo, J.C.; Whang, S.S. Anatomy of the Korean mistletoe and their haustorial features in host plants. Korean J. Plant Taxon. 2009, 39, 4–11. [Google Scholar] [CrossRef]
- Yoshikawa, E.; Yoneda, M.; Miyamoto, K.; Iwanaga, F.; Yamamoto, F. Leaf water relations and assimilation in Scurruta yadoriki and its host species. Tree For. Hearth 2009, 13, 58–66. [Google Scholar]
- Makino, T. Makino’s Illustrated Pocket Book of Japanese in Colour; Hokuryukan Co.: Tokyo, Japan, 1985. [Google Scholar]
- Guerra, T.J.; Pizo, M.A.; Silva, W.R. Host specificity and aggregation for a widespread mistletoe in Campo Rupestre vegetation. Flora 2018, 238, 148–154. [Google Scholar] [CrossRef]
- Okubamichael, D.Y.; Griffiths, M.E.; Ward, D. Host specificity in parasitic plants—Perspectives from mistletoes. AoB Plants 2016, 8, 69. [Google Scholar] [CrossRef]
- Norton, D.A.; Carpenter, M.A. Mistletoes as parasites: Host specificity and speciation. Trends Ecol. Evol. 1998, 13, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Tworek, S. Different bird strategies and their responses to habitat changes in an agricultural landscape. Ecol. Res. 2002, 17, 339–359. [Google Scholar] [CrossRef]
- Brambilla, M.; Assandri, G.; Martino, G.; Bogliani, G.; Pedrini, P. The importance of residual habitats and crop management for the conservation of birds breeding in intensive orchards. Ecol. Res. 2015, 30, 597–604. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).