Transcriptome Analysis Provides Insights into Lignin Biosynthesis in Styrax tonkinensis Branches
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Location and Materials
2.2. Determination of Lignin Content
2.3. RNA Extraction, cDNA Library Construction and Sequencing
2.4. Quality Control and De Novo Assembly
2.5. Kyoto Encyclopedia of Genes and Genomes (KEGG) Enrichment
2.6. Quantitative Real-Time PCR (qRT-PCR) Analysis
2.7. Statistics Analysis
3. Results
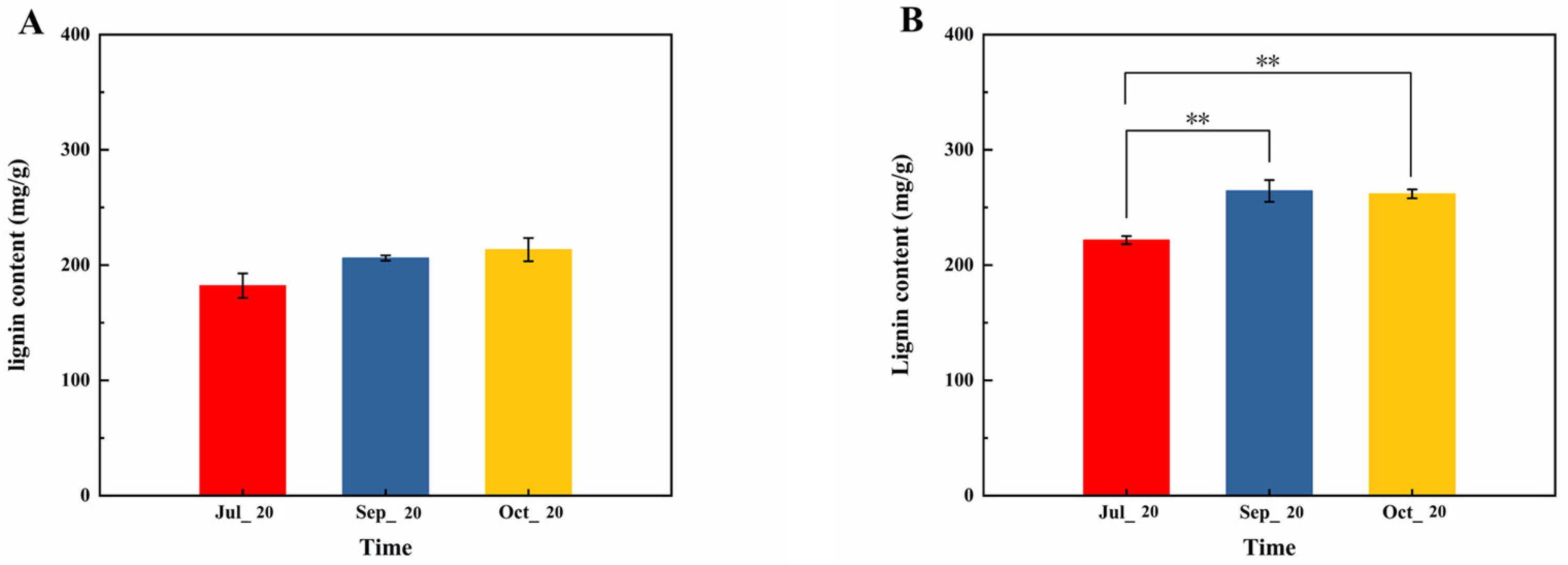
3.1. Changes in Lignin Content in Current-Year and Biennial Branches at Different Periods
3.2. De Novo Assembly and Quality Control
3.3. PCA Analysis
3.4. Analysis of DEGs
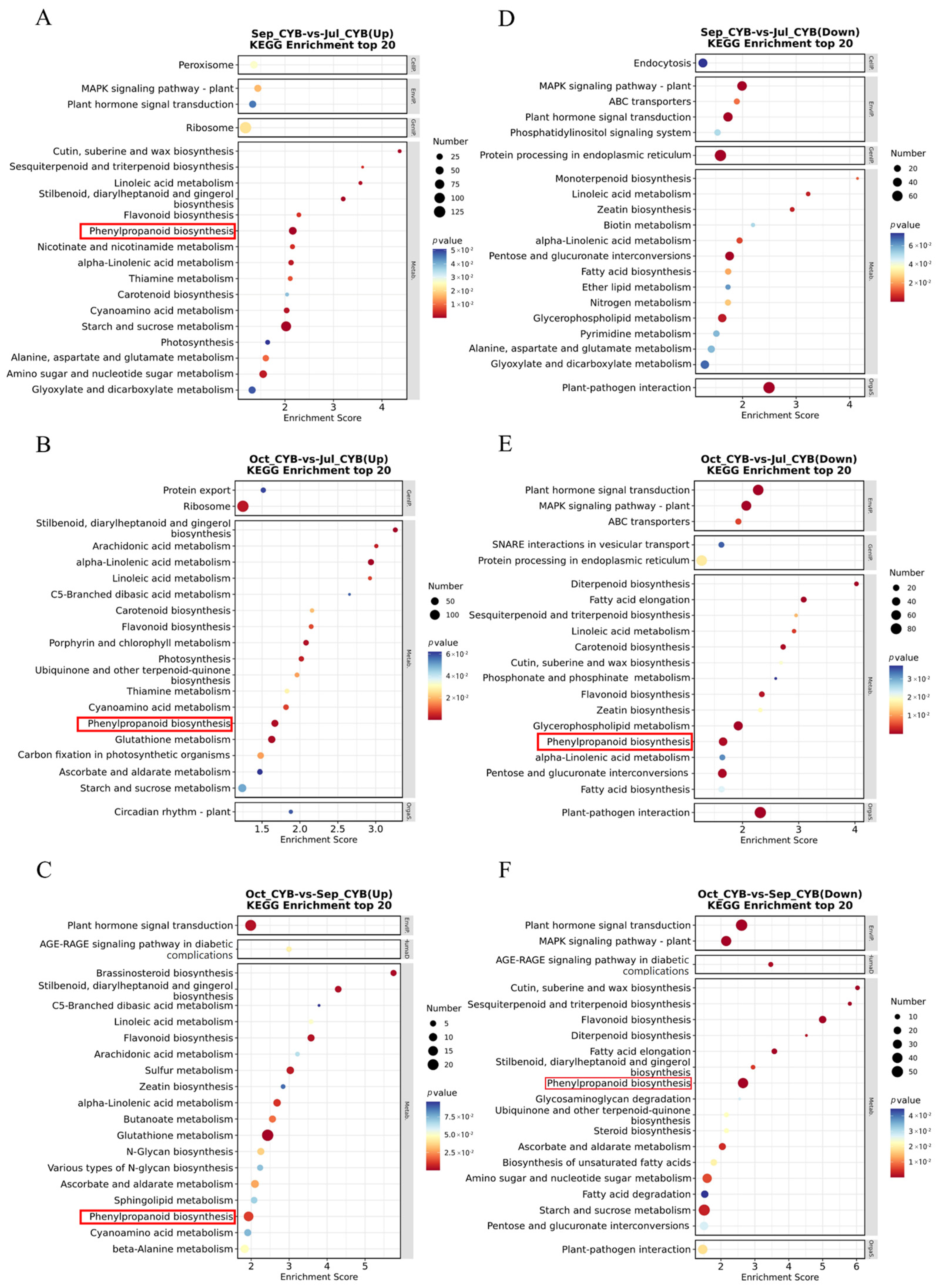
3.5. KEGG Pathway Enrichment Analysis of DEGs
3.6. Identification of DEGs Associated with Lignin Biosynthesis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sulis, D.B.; Jiang, X.; Yang, C.; Marques, B.M.; Matthews, M.L.; Miller, Z.; Lan, K.; Cofre-Vega, C.; Liu, B.; Sun, R.; et al. Multiplex CRISPR Editing of Wood for Sustainable Fiber Production. Science 2023, 381, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xie, S.; Yang, C.; Cao, D.; Fan, S.; Zhang, X. Comparative Transcriptome Analysis Reveals Candidate Genes and Pathways for Potential Branch Growth in Elm (Ulmus pumila) Cultivars. Biology 2022, 11, 711. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Chen, B.; Zhang, D. Transcript Profiling of Populus Tomentosa Genes in Normal, Tension, and Opposite Wood by RNA-Seq. BMC Genom. 2015, 16, 164. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.Y.; Yang, C.; Wu, Z.C.; Zhang, X.J.; Fan, S.J. Comprehensive Time-Course Transcriptome Reveals the Crucial Biological Pathways Involved in the Seasonal Branch Growth in Siberian Elm (Ulmus pumila). Int. J. Mol. Sci. 2023, 24, 14976. [Google Scholar] [CrossRef] [PubMed]
- Han, R.; Gu, C.; Li, R.; Xu, W.; Wang, S.; Liu, C.; Qu, C.; Chen, S.; Liu, G.; Yu, Q.; et al. Characterization and T-DNA Insertion Sites Identification of a Multiple-Branches Mutant Br in Betula platyphylla × Betula pendula. BMC Plant Biol. 2019, 19, 491. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Li, Y.; Liu, Y.; Guo, H.; Guo, J.; Du, Y.; Chen, Y.; Yu, C.; Zhong, F.; Lian, B.; et al. Genome-Wide Identification and Analysis of Monolignol Biosynthesis Genes in Salix matsudana Koidz and Their Relationship to Accelerated Growth. For. Res. 2021, 1, 8. [Google Scholar] [CrossRef]
- Chen, C.; Chen, H.; Han, C.; Liu, Z.; Ni, M.; Wu, Q.; Yu, F. Transcriptomic and Metabolomic Analysis Unravels the Molecular Regulatory Mechanism of Fatty Acid Biosynthesis in Styrax tonkinensis Seeds under Methyl Jasmonate Treatment. Int. J. Mol. Sci. 2022, 23, 6190. [Google Scholar] [CrossRef]
- Flora of China Editorial Committee. Flora of China; Science Press: Beijing, China, 2018; Volume 60, p. 84. [Google Scholar]
- Pinyopusarerk, K. Styrax Tonkinensis: Taxonomy, Ecology, Silviculture and Uses; Australian Centre for International Agricultural Research: Canberra, Australia, 1994. [Google Scholar]
- Matsuo, M.U.; Sujan, K.C.; Hirota, I.; Kojima, M.; Yoshida, M.; Yamamoto, H. Utilisation of Styrax tonkinensis Wood in Laos and Its Physical Properties. J. Trop. For. Sci. 2016, 28, 298–307. [Google Scholar]
- Luo, Y.C.; Yang, Y.; Zeng, Z.G.; Jie, J.L.; Huang, W.C.; Zhan, Y.S.; Zhou, X.P. Analysis of Wood Properties of Styrax tonkinensis and Its Utilization. Acta Agric. Univ. Jiangxiensis 2007, 29, 77–80. (In Chinese) [Google Scholar]
- Jędrzejczak, P.; Collins, M.N.; Jesionowski, T.; Klapiszewski, Ł. The Role of Lignin and Lignin-Based Materials in Sustainable Construction—A Comprehensive Review. Int. J. Biol. Macromol. 2021, 187, 624–650. [Google Scholar] [CrossRef]
- Riki, J.T.B.; Sotannde, O.A.; Oluwadare, A.O. Anatomical and Chemical Properties of Wood and Their Practical Implications in Pulp and Paper Production: A Review. J. Res. For. Wildl. Environ. 2019, 11, 358–368. [Google Scholar]
- Funda, T.; Fundová, I.; Fries, A.; Wu, H.X. Genetic Improvement of the Chemical Composition of Scots Pine (Pinus sylvestris L.) Juvenile Wood for Bioenergy Production. GCB Bioenergy 2020, 12, 848–863. [Google Scholar] [CrossRef]
- Wang, J.P.; Matthews, M.L.; Williams, C.M.; Shi, R.; Yang, C.; Tunlaya-Anukit, S.; Chen, H.C.; Li, Q.; Liu, J.; Lin, C.Y.; et al. Improving Wood Properties for Wood Utilization through Multi-Omics Integration in Lignin Biosynthesis. Nat. Commun. 2018, 9, 1579. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Dai, X.; Pang, H.; Cheng, Y.; Huang, X.; Li, H.; Yan, X.; Lu, F.; Wei, H.; Sederoff, R.R.; et al. BEL1-like Homeodomain Protein BLH6a Is a Negative Regulator of CAld5H2 in Sinapyl Alcohol Monolignol Biosynthesis in Poplar. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef]
- Chen, F.; Tobimatsu, Y.; Havkin-Frenkel, D.; Dixon, R.A.; Ralph, J. A Polymer of Caffeyl Alcohol in Plant Seeds. Proc. Natl. Acad. Sci. USA 2012, 109, 1772–1777. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Tobimatsu, Y.; Jackson, L.; Nakashima, J.; Ralph, J.; Dixon, R.A. Novel Seed Coat Lignins in the Cactaceae: Structure, Distribution and Implications for the Evolution of Lignin Diversity. Plant J. 2013, 73, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Chanoca, A.; de Vries, L.; Boerjan, W. Lignin Engineering in Forest Trees. Front. Plant Sci. 2019, 10, 912. [Google Scholar] [CrossRef]
- Bedon, F.; Legay, S. Lignin Synthesis, Transcriptional Regulation and Potential for Useful Modification in Plants. CABI Rev. 2011, 1–28. [Google Scholar] [CrossRef]
- Zhang, J.; Tuskan, G.A.; Tschaplinski, T.J.; Muchero, W.; Chen, J.G. Transcriptional and Post-Transcriptional Regulation of Lignin Biosynthesis Pathway Genes in Populus. Front. Plant Sci. 2020, 11, 534575. [Google Scholar] [CrossRef]
- Han, C.; Chen, J.; Liu, Z.; Chen, H.; Yu, F.; Yu, W. Morphological and Physiological Responses of Melia azedarach Seedlings of Different Provenances to Drought Stress. Agronomy 2022, 12, 1461. [Google Scholar] [CrossRef]
- Wan, Z.B.; Zhan, M.L.; Fang, L.J.; Pan, J. Study of Growth Rhythm of Reserved Seedlings of Styrax tonkinensis. For. By-Prod. Spec. China 2010, 1, 19–21. (In Chinese) [Google Scholar] [CrossRef]
- Guo, X.L.; Yu, B.Y.; Zhang, S.K.; Li, J.Y.; Wang, J.; Huang, J.G. Research Progresses on Xylem Formation Dynamics and Its Regulation Mechanism. J. Trop. Subtrop. Bot. 2019, 27, 541–547. (In Chinese) [Google Scholar]
- van der Weijde, T.; Torres, A.F.; Dolstra, O.; Dechesne, A.; Visser, R.G.F.; Trindade, L.M. Impact of Different Lignin Fractions on Saccharification Efficiency in Diverse Species of the Bioenergy Crop Miscanthus. Bioenerg. Res. 2016, 9, 146–156. [Google Scholar] [CrossRef]
- Chen, H.; Wu, Q.; Ni, M.; Chen, C.; Han, C.; Yu, F. Transcriptome Analysis of Endogenous Hormone Response Mechanism in Roots of Styrax Tonkinensis Under Waterlogging. Front. Plant Sci. 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A Flexible Trimmer for Illumina Sequence Data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-Length Transcriptome Assembly from RNA-Seq Data without a Reference Genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; He, Y.; Yu, Y.; Zhou, M.; Zhao, L.; Xia, X.; Wang, C. Transcriptomic Analysis of Seasonal Gene Expression and Regulation during Xylem Development in “Shanxin” Hybrid Poplar (Populus davidiana × Populus bolleana). Forests 2021, 12, 451. [Google Scholar] [CrossRef]
- Gao, Y.; Chen, X.B.; Zhang, Z.Y. Advances in Research on Lignin Biosynthesis and Its Molecular Regulation. Biotechnol. Bull. 2007, 2, 47–51. (In Chinese) [Google Scholar]
- Dixon, R.A.; Barros, J. Lignin Biosynthesis: Old Roads Revisited and New Roads Explored. Open Biol. 2019, 9, 190215. [Google Scholar] [CrossRef]
- Li, Y.; Wu, Q.; Men, X.; Wu, F.; Zhang, Q.; Li, W.; Sun, L.; Xing, S. Transcriptome and Metabolome Analyses of Lignin Biosynthesis Mechanism of Platycladus Orientalis. PeerJ 2022, 10, e14172. [Google Scholar] [CrossRef]
- Tong, N.N.; Peng, L.P.; Liu, Z.A.; Li, Y.; Zhou, X.Y.; Wang, X.R.; Shu, Q.Y. Comparative Transcriptomic Analysis of Genes Involved in Stem Lignin Biosynthesis in Woody and Herbaceous Paeonia Species. Physiol. Plant. 2021, 173, 961–977. [Google Scholar] [CrossRef] [PubMed]
- Rakoczy, M.; Femiak, I.; Alejska, M.; Figlerowicz, M.; Podkowinski, J. Sorghum CCoAOMT and CCoAOMT-like Gene Evolution, Structure, Expression and the Role of Conserved Amino Acids in Protein Activity. Mol. Genet. Genom. 2018, 293, 1077–1089. [Google Scholar] [CrossRef]
- Zhong, R.; Morrison, W.H.; Himmelsbach, D.S.; Poole, F.L.; Ye, Z.H. Essential Role of Caffeoyl Coenzyme A O -Methyltransferase in Lignin Biosynthesis in Woody Poplar Plants. Plant Physiol. 2000, 124, 563–578. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Liu, F.; Xing, H.; Mao, K.; Chen, G.; Guo, Q.; Chen, J. Correlation Analysis of Lignin Accumulation and Expression of Key Genes Involved in Lignin Biosynthesis of Ramie (Boehmeria nivea). Genes 2019, 10, 389. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ji, X.; Mao, R.; Cao, F.; Li, M. Increasing Lignin Accumulation in Arabidopsis and Poplar by Overexpressing a CCoAOMT Gene from the Dove Tree (Davidia involucrata Baill.). J. Plant Growth Regul. 2023, 42, 4095–4105. [Google Scholar] [CrossRef]
- Day, A.; Neutelings, G.; Nolin, F.; Grec, S.; Habrant, A.; Crônier, D.; Maher, B.; Rolando, C.; David, H.; Chabbert, B.; et al. Caffeoyl Coenzyme A O-Methyltransferase down-Regulation Is Associated with Modifications in Lignin and Cell-Wall Architecture in Flax Secondary Xylem. Plant Physiol. Biochem. 2009, 47, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Zhang, Y.; Xu, J.; Niu, X.; Qi, J.; Tao, A.; Zhang, L.; Fang, P.; Lin, L.; Su, J. The CCoAOMT1 Gene from Jute (Corchorus capsularis L.) Is Involved in Lignin Biosynthesis in Arabidopsis Thaliana. Gene 2014, 546, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.J.; Yang, Y.Z.; Teng, R.M.; Liu, H.; Li, H.; Zhuang, J. Identification and Expression Analysis of Caffeoyl-Coenzyme A O-Methyltransferase Family Genes Related to Lignin Biosynthesis in Tea Plant (Camellia sinensis). Protoplasma 2021, 258, 115–127. [Google Scholar] [CrossRef]
- Zhao, D.; Luan, Y.; Shi, W.; Zhang, X.; Meng, J.; Tao, J. A Paeonia Ostii Caffeoyl-CoA O-Methyltransferase Confers Drought Stress Tolerance by Promoting Lignin Synthesis and ROS Scavenging. Plant Sci. 2021, 303, 110765. [Google Scholar] [CrossRef]
- Ye, Z.H.; Zhong, R.; Morrison Iii, W.H.; Himmelsbach, D.S. Caffeoyl Coenzyme A O-Methyltransferase and Lignin Biosynthesis. Phytochemistry 2001, 57, 1177–1185. [Google Scholar] [CrossRef]
- Jouanin, L.; Goujon, T.; de Nadaï, V.; Martin, M.-T.; Mila, I.; Vallet, C.; Pollet, B.; Yoshinaga, A.; Chabbert, B.; Petit-Conil, M.; et al. Lignification in Transgenic Poplars with Extremely Reduced Caffeic Acid O-Methyltransferase Activity1. Plant Physiol. 2000, 123, 1363–1374. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H. Lignin Biosynthesis by Suppression of Two O-Methyltrans-Ferases. Chin. Sci. Bull. 2002, 47, 1092. [Google Scholar] [CrossRef]
- Zhu, Q.; Zhang, K.; Chen, W.; Li, X.; Zhu, X. Transcriptomic and Metabolomic Analyses Reveal Key Factors Regulating Chilling Stress-Induced Softening Disorder in Papaya Fruit. Postharvest Biol. Technol. 2023, 205, 112534. [Google Scholar] [CrossRef]
- Whetten, R.W.; MacKay, J.J.; Sederoff, R.R. Recent Advances In Understanding Lignin Biosynthesis. Annu. Rev. Plant. Physiol. Plant. Mol. Biol. 1998, 49, 585–609. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; Xu, C.; Li, X.; Ferguson, I.; Chen, K. Accumulation of Lignin in Relation to Change in Activities of Lignification Enzymes in Loquat Fruit Flesh after Harvest. Postharvest Biol. Technol. 2006, 40, 163–169. [Google Scholar] [CrossRef]
- Blee, K.A.; Choi, J.W.; O’Connell, A.P.; Schuch, W.; Lewis, N.G.; Bolwell, G.P. A Lignin-Specific Peroxidase in Tobacco Whose Antisense Suppression Leads to Vascular Tissue Modification. Phytochemistry 2003, 64, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Jaberian Hamedan, H.; Sohani, M.M.; Aalami, A.; Nazarideljou, M.J. Genetic Engineering of Lignin Biosynthesis Pathway Improved Stem Bending Disorder in Cut Gerbera (Gerbera jamesonii) Flowers. Sci. Hortic. 2019, 245, 274–279. [Google Scholar] [CrossRef]
- Liu, D.; Yuan, H.; Yao, Y.; Cheng, L.; Tang, L.; Kang, Q.; Song, X.; Chen, S.; Wu, G. Genome-Wide Identification and Expression Pattern Analysis of the F5H Gene Family in Flax (Linum usitatissimum L.). Agronomy 2023, 13, 1108. [Google Scholar] [CrossRef]
- Fan, D.; Li, C.; Fan, C.; Hu, J.; Li, J.; Yao, S.; Lu, W.; Yan, Y.; Luo, K. MicroRNA6443-Mediated Regulation of FERULATE 5-HYDROXYLASE Gene Alters Lignin Composition and Enhances Saccharification in Populus tomentosa. New Phytol. 2020, 226, 410–425. [Google Scholar] [CrossRef]
- Öhman, D.; Demedts, B.; Kumar, M.; Gerber, L.; Gorzsás, A.; Goeminne, G.; Hedenström, M.; Ellis, B.; Boerjan, W.; Sundberg, B. MYB103 Is Required for FERULATE-5-HYDROXYLASE Expression and Syringyl Lignin Biosynthesis in Arabidopsis Stems. Plant J. 2013, 73, 63–76. [Google Scholar] [CrossRef]
- Ruegger, M.; Meyer, K.; Cusumano, J.C.; Chapple, C. Regulation of Ferulate-5-Hydroxylase Expression in Arabidopsis in the Context of Sinapate Ester Biosynthesis1. Plant Physiol. 1999, 119, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Yan, X.; Ran, S.; Ralph, J.; Smith, R.A.; Chen, X.; Qu, C.; Li, J.; Liu, L. Knockout of the Lignin Pathway Gene BnF5H Decreases the S/G Lignin Compositional Ratio and Improves Sclerotinia Sclerotiorum Resistance in Brassica Napus. Plant Cell Environ. 2022, 45, 248–261. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Yue, J.; Yang, H.; Zhu, C.; Zhu, F.; Li, J.; Xu, R.; Gao, J.; Zhou, D.; Deng, X.; et al. Integration of Metabolome, Histochemistry and Transcriptome Analysis Provides Insights into Lignin Accumulation in Oleocellosis-Damaged Flavedo of Citrus Fruit. Postharvest Biol. Technol. 2021, 172, 111362. [Google Scholar] [CrossRef]
- Gui, J.; Luo, L.; Zhong, Y.; Sun, J.; Umezawa, T.; Li, L. Phosphorylation of LTF1, an MYB Transcription Factor in Populus, Acts as a Sensory Switch Regulating Lignin Biosynthesis in Wood Cells. Mol. Plant 2019, 12, 1325–1337. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.Q.; Huang, J.Q.; Reyt, G.; Song, T.; Love, A.; Tiemessen, D.; Xue, P.Y.; Wu, W.K.; George, M.W.; Chen, X.Y.; et al. A Dirigent Protein Complex Directs Lignin Polymerization and Assembly of the Root Diffusion Barrier. Science 2023, 382, 464–471. [Google Scholar] [CrossRef]
- Khan, P.; Tong, L.; Khan, S.U.; Zhang, C.; Wang, W. Lignin: A Defensive Shield Halting the Environmental Stresses—A Review. Appl. Ecol. Env. Res. 2022, 20, 1991–2015. [Google Scholar] [CrossRef]
- Chen, Y.; Yi, N.; Yao, S.B.; Zhuang, J.; Fu, Z.; Ma, J.; Yin, S.; Jiang, X.; Liu, Y.; Gao, L.; et al. CsHCT-Mediated Lignin Synthesis Pathway Involved in the Response of Tea Plants to Biotic and Abiotic Stresses. J. Agric. Food Chem. 2021, 69, 10069–10081. [Google Scholar] [CrossRef]
- Li, D.; Yang, J.; Pak, S.; Zeng, M.; Sun, J.; Yu, S.; He, Y.; Li, C. PuC3H35 Confers Drought Tolerance by Enhancing Lignin and Proanthocyanidin Biosynthesis in the Roots of Populus Ussuriensis. New Phytol. 2022, 233, 390–408. [Google Scholar] [CrossRef]
- Rogers, L.A.; Campbell, M.M. The Genetic Control of Lignin Deposition during Plant Growth and Development. New Phytol. 2004, 164, 17–30. [Google Scholar] [CrossRef]
- Xie, M.; Zhang, J.; Tschaplinski, T.J.; Tuskan, G.A.; Chen, J.G.; Muchero, W. Regulation of Lignin Biosynthesis and Its Role in Growth-Defense Tradeoffs. Front. Plant Sci. 2018, 9, 1427. [Google Scholar] [CrossRef]
- Xiao, R.; Zhang, C.; Guo, X.; Li, H.; Lu, H. MYB Transcription Factors and Its Regulation in Secondary Cell Wall Formation and Lignin Biosynthesis during Xylem Development. Int. J. Mol. Sci. 2021, 22, 3560. [Google Scholar] [CrossRef]
- Sun, Y.; Jiang, C.; Jiang, R.; Wang, F.; Zhang, Z.; Zeng, J. A Novel NAC Transcription Factor From Eucalyptus, EgNAC141, Positively Regulates Lignin Biosynthesis and Increases Lignin Deposition. Front. Plant Sci. 2021, 12, 642090. [Google Scholar] [CrossRef]
- Quan, M.; Du, Q.; Xiao, L.; Lu, W.; Wang, L.; Xie, J.; Song, Y.; Xu, B.; Zhang, D. Genetic Architecture Underlying the Lignin Biosynthesis Pathway Involves Noncoding RNAs and Transcription Factors for Growth and Wood Properties in Populus. Plant Biotechnol. J. 2019, 17, 302–315. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, C.; Xu, Q.; Chen, H.; Peng, H.; Yu, F. Transcriptome Analysis Provides Insights into Lignin Biosynthesis in Styrax tonkinensis Branches. Forests 2024, 15, 601. https://doi.org/10.3390/f15040601
Han C, Xu Q, Chen H, Peng H, Yu F. Transcriptome Analysis Provides Insights into Lignin Biosynthesis in Styrax tonkinensis Branches. Forests. 2024; 15(4):601. https://doi.org/10.3390/f15040601
Chicago/Turabian StyleHan, Chao, Qiunuan Xu, Hong Chen, Huiwu Peng, and Fangyuan Yu. 2024. "Transcriptome Analysis Provides Insights into Lignin Biosynthesis in Styrax tonkinensis Branches" Forests 15, no. 4: 601. https://doi.org/10.3390/f15040601
APA StyleHan, C., Xu, Q., Chen, H., Peng, H., & Yu, F. (2024). Transcriptome Analysis Provides Insights into Lignin Biosynthesis in Styrax tonkinensis Branches. Forests, 15(4), 601. https://doi.org/10.3390/f15040601





