Genetic Diversity and Population Structure Analysis in the Chinese Endemic Species Michelia crassipes Based on SSR Markers
Abstract
1. Introduction
2. Materials and Methods
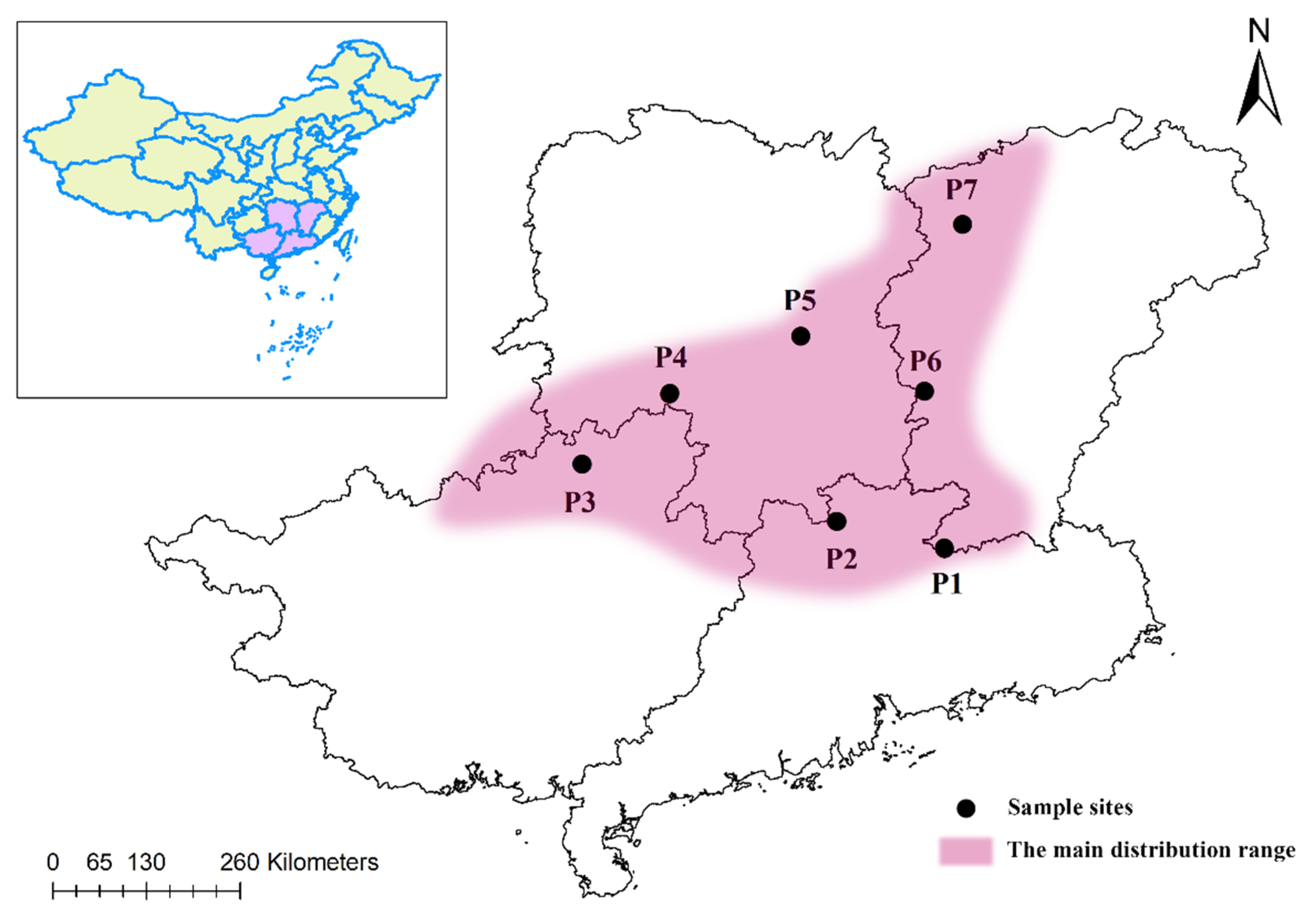
2.1. Plant Material
2.2. DNA Extraction
2.3. Primer Screening and PCR Amplification
2.4. Analysis of Genetic Diversity and Population Genetic Structure
3. Results and Analysis
3.1. Genetic Diversity of M. crassipes Populations
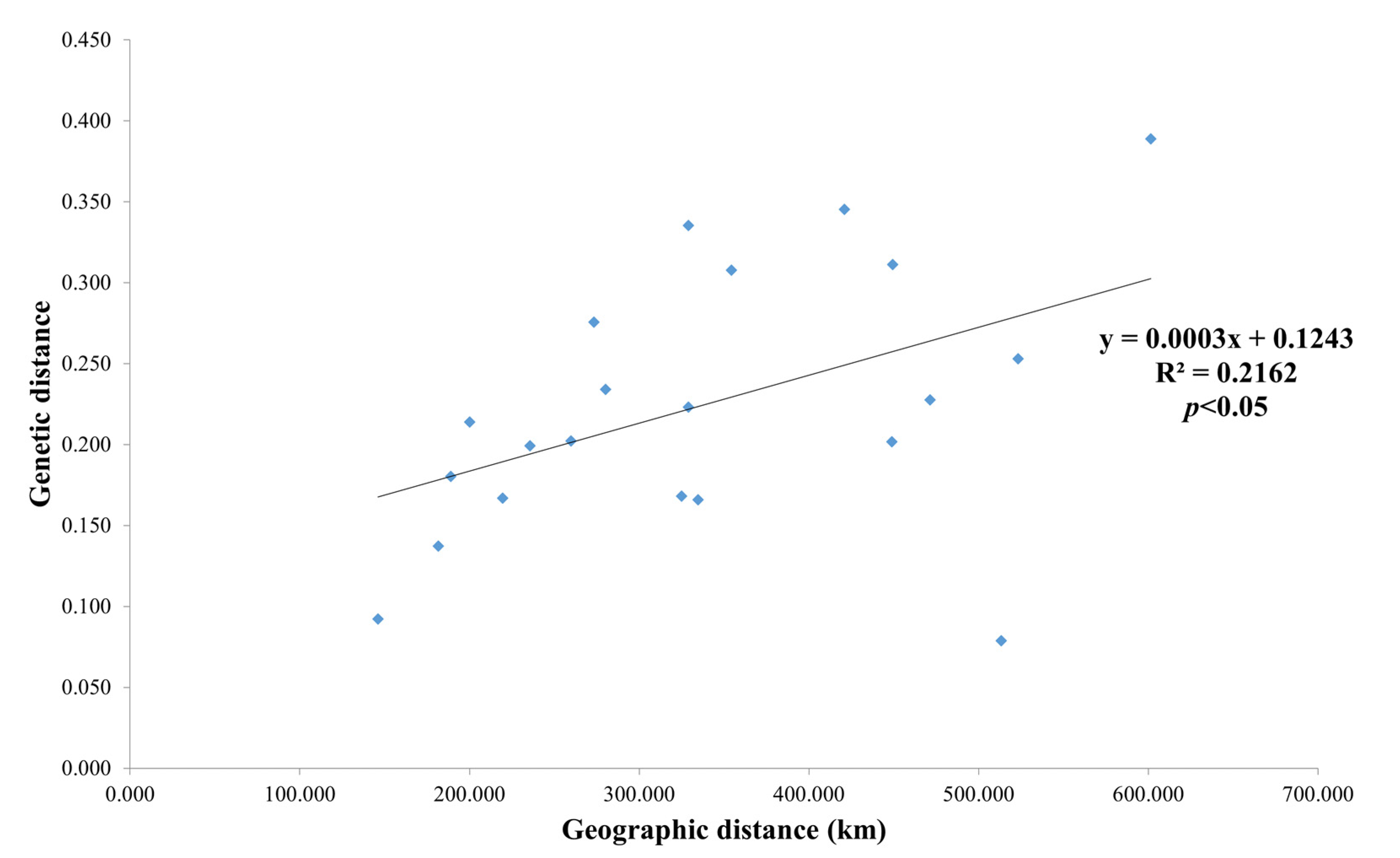
3.2. Genetic Differentiation and Gene Flow
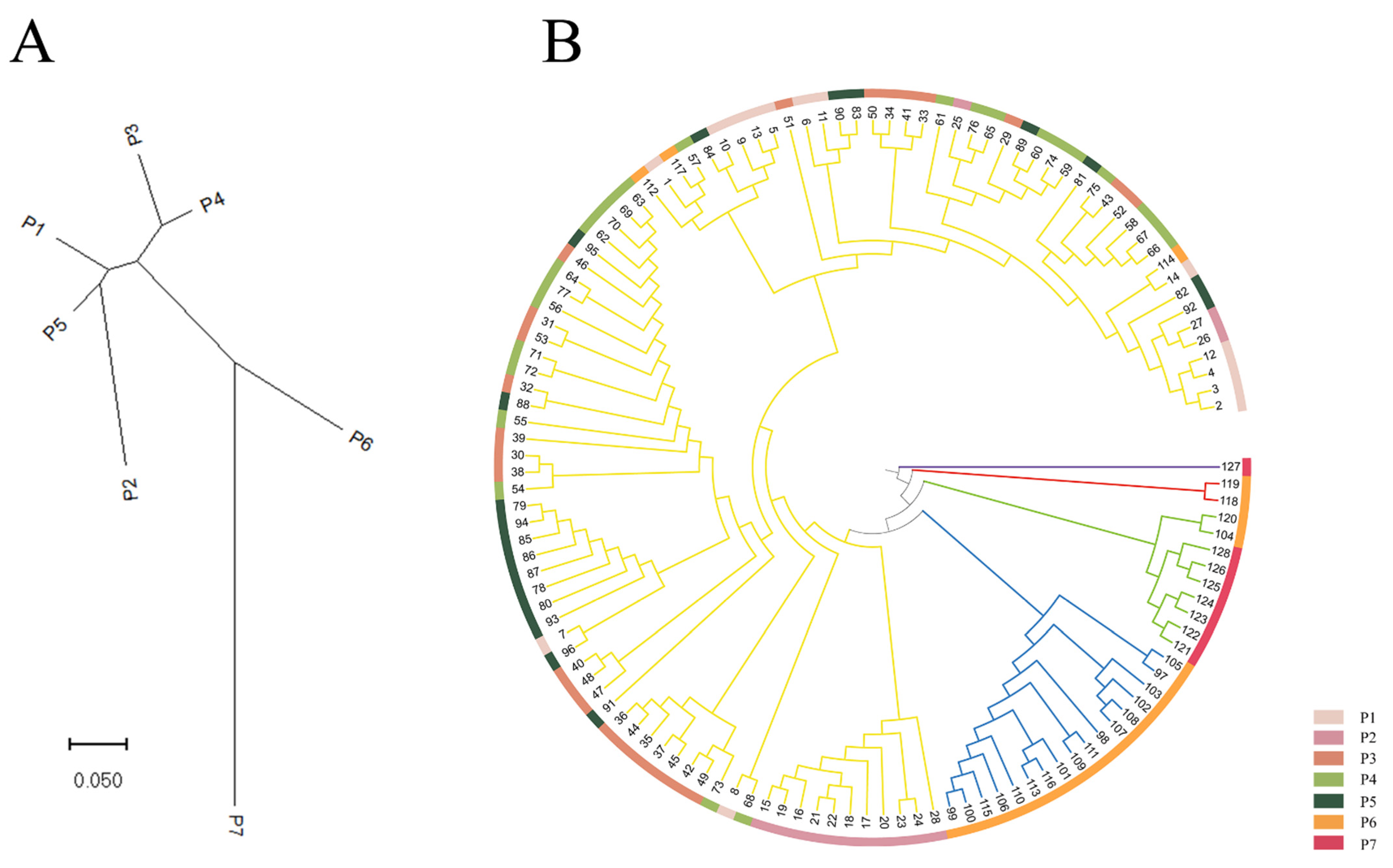
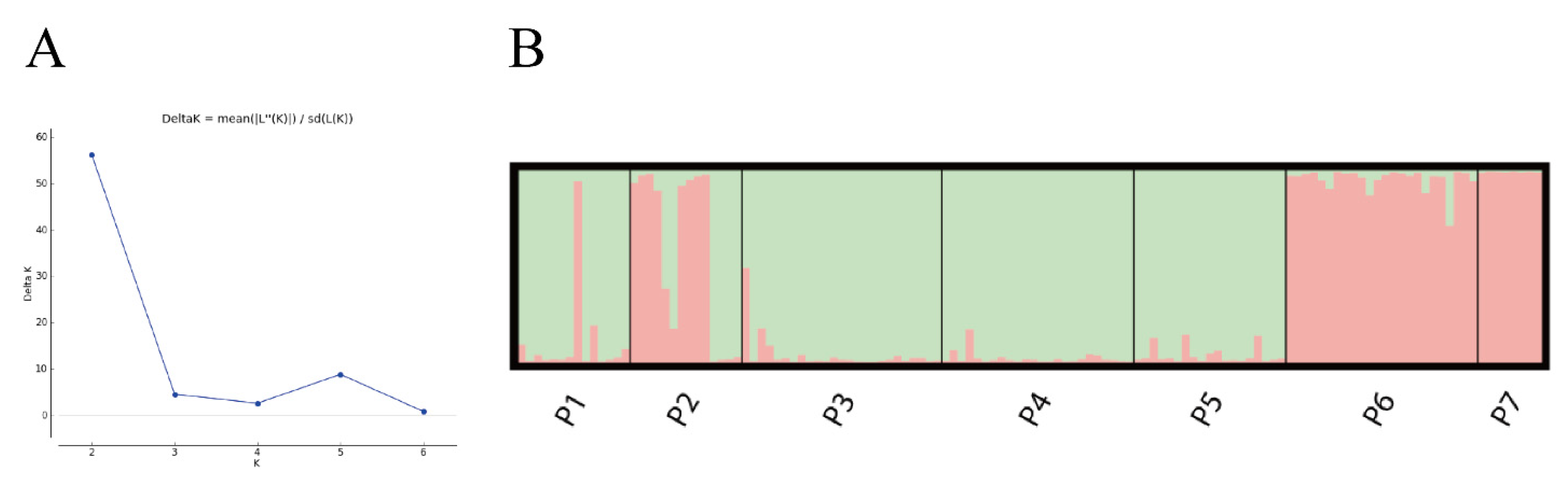
3.3. Population Clustering and Genetic Structure
4. Discussion
4.1. Genetic Diversity of Michelia crassipes
4.2. Genetic Differentiation and Genetic Structure
4.3. Conservation Proposal
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Flora of China Editorial Committee. Flora of China; Science Press: Beijing, China, 1996. [Google Scholar]
- Liu, D.; Ouyang, S.; Zeng, G. Rare ornamental flowering trees—M. crassipes. Plants 2002, 2, 18–19. [Google Scholar]
- Chai, Y.; Cai, M.; Jin, X.; Zhang, D. Pollination biology of Michelia crassipes. Guihaia 2017, 37, 1322–1329. [Google Scholar]
- Liu, Y. Magnolias of China; Beijing Science and Technology Press: Beijing, China, 2004. [Google Scholar]
- Shao, W.; Jiang, J.; Dong, R. A New Michelia Cultivar ‘Mengxing’. Acta Hortic. Sin. 2016, 43, 1219–1220. [Google Scholar] [CrossRef]
- Shao, W.; Jiang, J.; Dong, R.; Luan, Q. A New Variety, Michelia ‘Mengyuan’. Sci. Silvae Sin. 2015, 51, 155. [Google Scholar]
- Shao, W.; Jiang, J.; Dong, R. A New Michelia Cultivar ‘Mengzi’. Acta Hortic. Sin. 2015, 42, 1863–1864. [Google Scholar] [CrossRef]
- Bo, B. Kunming Institute of Botany successfully produces five new cultivars of the genus Michelia. China Flowers Hortic. 2011, 11, 40. [Google Scholar]
- Liao, J. Selective Breeding of New Cultivars for the Michelia crassipes Law. J. Northwest For. Univ. 2007, 22, 76–78. [Google Scholar]
- Yu, Q.; Liu, C.; Jin, X.; Tan, Y. Changes of Physiolocical indexes and Pigment during the Flowering Process in the Perianth of Michelia crassipes. J. Northeast. For. Univ. 2021, 49, 39–43. [Google Scholar] [CrossRef]
- Liu, C.; Yu, Q.; Li, Z.; Jin, X.; Xing, W. Metabolic and transcriptomic analysis related to flavonoid biosynthesis during the color formation of Michelia crassipes tepal. Plant Physiol. Biochem. 2020, 155, 938–951. [Google Scholar] [CrossRef] [PubMed]
- Gong, W.; Xiao, J.; Gan, Q.; Huang, F.; Ouyang, S.; He, L. The Effects of the Substrate and Rooting Reagent on the Rooting of Michelia Crassipes by Cutting Propagation. Fujian For. 2018, 6, 37–40. [Google Scholar]
- Chai, Y.; Hu, X.; Zhang, D.; Liu, X.; Liu, C.; Jin, X. Studies on Compatibility of lnterspecific Hybridization Between Michelia crassipes and M. Figo, M. maudiae, M. platypetala. Acta Hortic. Sin. 2018, 45, 1970–1978. [Google Scholar] [CrossRef]
- Liu, X.; Tang, S.; Sun, Q.; Yao, G. Geographic Distribution of Magnoliaceae Plants in East China Plant Zone. For. Inventory Plan. 2008, 10, 40–43. [Google Scholar]
- Yang, C.; Chen, J.; Fang, X.; Yang, l. Excellent garden tree species Michelia crassipes. Guizhou For. Sci. Technol. 2003, 6, 16–18. [Google Scholar]
- Xu, F.; Zhu, B.; Pan, W.; Zhu, Z.; Zhong, N.; Li, W.; Zhao, Q. Study on the Genetic Relationship of 11 Michelia Species Based on SSR Markers. Guangdong Agric. Sci. 2021, 48, 87–93. [Google Scholar] [CrossRef]
- Biodiversity Committee; Chinese Academy of Sciences. Principles and Methods of Biodiversity Research; Science and Technology of China Press: Beijing, China, 1994. [Google Scholar]
- Booy, G.; Hendriks, R.; Smulders, M.; Van Groenendael, J.; Vosman, B. Genetic diversity and the survival of populations. Plant Biol. 2000, 2, 379–395. [Google Scholar] [CrossRef]
- Xu, Y.; Zang, R. Theoretical and practical research on conservation of Wild Plants with Extremely Small Populations in China. Biodivers. Sci. 2022, 30, 84–105. [Google Scholar] [CrossRef]
- Liu, R.; Li, J.; Yu, J.; Yu, W.; Huang, X.; Han, S.; Lu, M. SSR Marker-based Analysis on Genetic Relationship of 16 Torreya grandis Germplasms. Mol. Plant Breed. 2022, 20, 4144–4152. [Google Scholar] [CrossRef]
- Khodadadi, M.; Fotokian, M.H.; Miransari, M. Genetic Diversity of Wheat (Triticum aestivum L.) Genotypes Based on Cluster and Principal Component Analyses for Breeding Strategies. Aust. J. Crop Sci. 2011, 5, 17–24. [Google Scholar]
- Wu, Q.; Zang, F.; Ma, Y.; Zheng, Y.; Zang, D. Analysis of genetic diversity and population structure in endangered Populus wulianensis based on 18 newly developed EST-SSR markers. Glob. Ecol. Conserv. 2020, 24, e01329. [Google Scholar] [CrossRef]
- Li, S.; Liu, S.-L.; Pei, S.-Y.; Ning, M.-M.; Tang, S.-Q. Genetic diversity and population structure of Camellia huana (Theaceae), a limestone species with narrow geographic range, based on chloroplast DNA sequence and microsatellite markers. Plant Divers. 2020, 42, 343–350. [Google Scholar] [CrossRef]
- Kumar, M.; Chaudhary, V.; Sirohi, U.; Singh, M.K.; Malik, S.; Naresh, R. Biochemical and molecular markers for characterization of chrysanthemum germplasm: A review. J. Pharmacogn. Phytochem. 2018, 7, 2641–2652. [Google Scholar]
- Wang, X.; Gao, C.; Li, K. Construction strategy and verification of timber-used germplasm conservation bank of Pinus vunnanensis. J. Plant Resour. Environ. 2019, 28, 105–114. [Google Scholar]
- Chen, X.; Shen, X. Forest Tree Breeding; Higher Education Press: Beijing, China, 2005. [Google Scholar]
- Li, B.; Gu, W.; Lu, B. A study on phenotypic diversity of seeds and cones characteristics in Pinus bungeana. Biodivers. Sci. 2002, 10, 181–188. [Google Scholar]
- Thiel, T.; Michalek, W.; Varshney, R.; Graner, A. Exploiting EST databases for the development and characterization of gene-derived SSR-markers in barley (Hordeum vulgare L.). Theor. Appl. Genet. 2003, 106, 411–422. [Google Scholar] [CrossRef]
- Koressaar, T.; Remm, M. Enhancements and modifications of primer design program Primer3. Bioinformatics 2007, 23, 1289–1291. [Google Scholar] [CrossRef]
- Peakall, R.; Smouse, P.E. GenAlEx 6.5: Genetic analysis in Excel. Population genetic software for teaching and research—An update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef]
- Kalinowski, S.T.; Taper, M.L.; Marshall, T.C. Revising how the computer program CERVUS accommodates genotyping error increases success in paternity assignment. Mol. Ecol. 2007, 16, 1099–1106. [Google Scholar] [CrossRef]
- Yeh, F.C.; Yang, R.C.; Boyle, T.B.; Ye, Z.; Mao, J.X. POPGENE, the user-friendly shareware for population genetic analysis. Mol. Biol. Biotechnol. Cent. Univ. Alta. Can. 1997, 10, 295–301. [Google Scholar]
- Pritchard, J.K.; Stephens, M.; Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 2000, 155, 945–959. [Google Scholar] [CrossRef] [PubMed]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef]
- Earl, D.A.; VonHoldt, B.M. STRUCTURE HARVESTER: A website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv. Genet. Resour. 2012, 4, 359–361. [Google Scholar] [CrossRef]
- Jakobsson, M.; Rosenberg, N.A. CLUMPP: A cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics 2007, 23, 1801–1806. [Google Scholar] [CrossRef]
- Rosenberg, N.A. DISTRUCT: A program for the graphical display of population structure. Mol. Ecol. Notes 2004, 4, 137–138. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular evolutionary genetics analysis version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef]
- Botstein, D.; White, R.L.; Skolnick, M.; Davis, R.W. Construction of a genetic linkage map in man using restriction fragment length polymorphisms. Am. J. Hum. Genet. 1980, 32, 314. [Google Scholar] [PubMed]
- Wright, S. The genetical structure of populations. Ann. Eugen. 1949, 15, 323–354. [Google Scholar] [CrossRef] [PubMed]
- Belaj, A.; Muñoz-Diez, C.; Baldoni, L.; Porceddu, A.; Barranco, D.; Satovic, Z. Genetic diversity and population structure of wild olives from the north-western Mediterranean assessed by SSR markers. Ann. Bot. 2007, 100, 449–458. [Google Scholar] [CrossRef]
- Nybom, H.; Bartish, I.V. Effects of life history traits and sampling strategies on genetic diversity estimates obtained with RAPD markers in plants. Perspect. Plant Ecol. Evol. Syst. 2000, 3, 93–114. [Google Scholar] [CrossRef]
- Hamrick, J.L.; Godt, M.J.W.; Sherman-Broyles, S.L. Factors influencing levels of genetic diversity in woody plant species. In Population Genetics of Forest Trees; Springer: Berlin/Heidelberg, Germany, 1992; pp. 95–124. [Google Scholar]
- Qin, A.; Ma, F.; Xu, G.; Shi, Z.; Chen, Q. Population structure and dynamic characteristics of a rare and endangered tree species Michelia wilsonii Finet et Gagn. Acta Ecol. Sin. 2020, 40, 4445–4454. [Google Scholar]
- Wei, Y.; Hong, F.; Yuan, L.; Kong, Y.; Shi, Y. Population Distribution and Age Structure Characteristics of Michelia shiluensis, an Endangered and Endemic Species in Hainan lslano. Chin. J. Trop. Crops 2017, 38, 2280–2284. [Google Scholar]
- Li, H.; Xiong, Z.; Chen, Z.; Yang, C. A Preliminary Investigation of Rare and Endangered Michelia angustioblonga. J. Sichuan For. Sci. Technol. 2016, 37, 96–98. [Google Scholar] [CrossRef]
- Guan, Z.; Zhang, L. The Study on Conservation and Utilization of Endangered Plant Michelia hedyosperma. Chin. Wild Plant Resour. 2004, 23, 11–14. [Google Scholar]
- Zhao, N.; Park, S.; Zhang, Y.-Q.; Nie, Z.-L.; Ge, X.-J.; Kim, S.; Yan, H.-F. Fingerprints of climatic changes through the late Cenozoic in southern Asian flora: Magnolia section Michelia (Magnoliaceae). Ann. Bot. 2022, 130, 41–52. [Google Scholar] [CrossRef]
- Reed, D.H.; Frankham, R. Correlation between fitness and genetic diversity. Conserv. Biol. 2003, 17, 230–237. [Google Scholar] [CrossRef]
- Niu, Y.; Bhatt, A.; Peng, Y.; Chen, W.; Gao, Y.; Zhan, X.; Zhang, Z.; Hu, W.; Song, M.; Yu, Z. Genetic diversity and population structure analysis of Emmenopterys henryi Oliv., an endangered relic species endemic to China. Genet. Resour. Crop Evol. 2021, 68, 1135–1148. [Google Scholar] [CrossRef]
- Ye, X.; Wen, G.; Zhang, M.; Liu, Y.; Fan, H.; Zhang, G.; Chen, S.; Liu, B. Genetic diversity and genetic structure of a rare and endangered species Semiliquidambar cathayensis Hung T.Chano. Plant Sci. J. 2021, 39, 415–423. [Google Scholar]
- Wang, S.-Q. Genetic diversity and population structure of the endangered species Paeonia decomposita endemic to China and implications for its conservation. BMC Plant Biol. 2020, 20, 510. [Google Scholar] [CrossRef]
- Kikuchi, S.; Osone, Y. Subspecies divergence and pronounced phylogenetic incongruence in the East-Asia-endemic shrub Magnolia sieboldii. Ann. Bot. 2021, 127, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chen, W.; Luo, J.; Yao, Z.; Yu, Q.; Wang, Y.; Zhang, S.; Liu, Z.; Zhang, M.; Shen, Y. Development of EST-SSR markers and their application in an analysis of the genetic diversity of the endangered species Magnolia sinostellata. Mol. Genet. Genom. 2019, 294, 135–147. [Google Scholar] [CrossRef]
- Tamaki, I.; Kawashima, N.; Setsuko, S.; Itaya, A.; Tomaru, N. Morphological and genetic divergence between two lineages of Magnolia salicifolia (Magnoliaceae) in Japan. Biol. J. Linn. Soc. 2018, 125, 475–490. [Google Scholar] [CrossRef]
- Xiong, M.; Tian, S.; Zhang, Z.; Fan, D.; Zhang, Z. Population genetic structure and conservation units of Sinomanglietia glauca (Magnoliaceae). Biodivers. Sci. 2014, 22, 476–484. [Google Scholar]
- Yu, H.-H.; Yang, Z.-L.; Sun, B.; Liu, R.-n. Genetic diversity and relationship of endangered plant Magnolia officinalis (Magnoliaceae) assessed with ISSR polymorphisms. Biochem. Syst. Ecol. 2011, 39, 71–78. [Google Scholar] [CrossRef]
- Xiang, C.; Zhu, X.; Zhang, H.; Qiu, D. Genetic Diversity of Endangered Plant Michelia wilsonii. J. Northwest For. Univ. 2009, 24, 66–69. [Google Scholar]
- Zhao, X.; Ma, Y.; Sun, W.; Wen, X.; Milne, R. High genetic diversity and low differentiation of Michelia coriacea (Magnoliaceae), a critically endangered endemic in southeast Yunnan, China. Int. J. Mol. Sci. 2012, 13, 4396–4411. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Liu, T.; Xie, Y.; Wei, Y.; Xie, Z.; Shi, Y.; Deng, X. High Genetic Diversity and Low Differentiation in Michelia shiluensis, an Endangered Magnolia Species in South China. Forests 2020, 11, 469. [Google Scholar] [CrossRef]
- Lin, Y. The Population Genetic Structure and Gene Flow of Kmeria septentrionalis Assessed by Microsatellite Markers. Master’s Thesis, Guangxi Normal University, Guilin, China, 2012. [Google Scholar]
- Li, K.; Chen, L.; Feng, Y.; Yao, J.; Li, B.; Xu, M.; Li, H. High genetic diversity but limited gene flow among remnant and fragmented natural populations of Liriodendron chinense Sarg. Biochem. Syst. Ecol. 2014, 54, 230–236. [Google Scholar] [CrossRef]
- Bernard, A.; Barreneche, T.; Lheureux, F.; Dirlewanger, E. Analysis of genetic diversity and structure in a worldwide walnut (Juglans regia L.) germplasm using SSR markers. PLoS ONE 2018, 13, e0208021. [Google Scholar] [CrossRef]
- Ebadi, A.; Ghaderi, N.; Vafaee, Y. Genetic diversity of Iranian and some European grapes as revealed by nuclear and chloroplast microsatellite and SNP molecular markers. J. Hortic. Sci. Biotechnol. 2019, 94, 599–610. [Google Scholar] [CrossRef]
- Xiao, Z.; Zhai, M.; Wang, Z.; Xu, J.; Li, L.; Yang, H. Application of microsatellite DNA on analyzing genetic diversity of Juglans regia. J. Cent. South Univ. For. Technol. 2014, 34, 55–61. [Google Scholar] [CrossRef]
- Zhao, P.; Li, D.; Ma, J.; Liang, W.; Pang, X.; Long, C.; Ma, J.; Guo, H. SSR marker development, genetic diversity and population structure analysis in oil tree species Vernicia montana. J. Beijing For. Univ. 2021, 43, 50–61. [Google Scholar]
- Paetkau, D. The molecular basis and evolutionary history of a microsatellite null allele in bears. Mol. Ecol. 1995, 4, 519–520. [Google Scholar] [CrossRef] [PubMed]
- Barros, J.; Winkler, F.M.; Velasco, L.A. Assessing the genetic diversity in Argopecten nucleus (Bivalvia: Pectinidae), a functional hermaphrodite species with extremely low population density and self-fertilization: Effect of null alleles. Ecol. Evol. 2020, 10, 3919–3931. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Cole, J.W.; Grond-Ginsbach, C. Departure from Hardy Weinberg equilibrium and genotyping error. Front. Genet. 2017, 8, 167. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zheng, Y.; Li, C.; Lin, F.; Huang, P. Genomic Characteristics and Population Genetic Variation of Dalbergia cultrata Graham ex Benth in China. For. Res. 2022, 35, 44–53. [Google Scholar] [CrossRef]
- Nybom, H. Comparison of different nuclear DNA markers for estimating intraspecific genetic diversity in plants. Mol. Ecol. 2004, 13, 1143–1155. [Google Scholar] [CrossRef]
- Morjan, C.L.; Rieseberg, L.H. How species evolve collectively: Implications of gene flow and selection for the spread of advantageous alleles. Mol. Ecol. 2004, 13, 1341–1356. [Google Scholar] [CrossRef]
- Govindaraju, D.R. Relationship between dispersal ability and levels of gene flow in plants. Oikos 1988, 52, 31–35. [Google Scholar] [CrossRef]
- Wright, S. Evolution and the Genetics of Populations, Volume 4: Variability within and among Natural Populations; University of Chicago press: Chicago, IL, USA, 1984; Volume 4. [Google Scholar]
- Liu, H.; Gao, J.; Song, C.; Yu, S. Conservation status and human disturbance of the habitats of Michelia crassipes Law in China. China Environ. Sci. 2019, 39, 3976–3981. [Google Scholar] [CrossRef]
- Collevatti, R.G.; Telles, M.P.; Lima, J.S.; Gouveia, F.O.; Soares, T.N. Contrasting spatial genetic structure in Annona crassiflora populations from fragmented and pristine savannas. Plant Syst. Evol. 2014, 300, 1719–1727. [Google Scholar] [CrossRef]
- Guo, Z.; Cui, G. Geomorphologic regionalization of China aimed at construction of nature reserve system. Acta Ecol. Sin. 2013, 33, 6264–6276. [Google Scholar]
- Lu, J.; Zhang, Y.; Diao, X.; Yu, K.; Dai, X.; Qu, P.; Crabbe, M.J.C.; Zhang, T.; Qiao, Q. Evaluation of genetic diversity and population structure of Fragaria nilgerrensis using EST-SSR markers. Gene 2021, 796–797, 145791. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Jiao, Z.; Zheng, J.; Dou, Q.; Huang, L.; Wang, B.; Huang, R. Construction of Fingerprint of Michelia Germplasm by Fluorescent SSR Markers. Mol. Plant Breed. 2018, 16, 4705–4714. [Google Scholar]


| Primer Pair | Primer Sequence (5′-3′) | Repeat Motif | Annealing Temperature (°C) | Product Size/bp |
|---|---|---|---|---|
| M17 | F:GCTGCAGAGGCATTATGGTT | (ATC)6 | 60 | 239 |
| R:ATGTCTGGTGGGCAGATGAT | ||||
| M19 | F:AGCTTCCGGGACTTCAAACT | (CAG)5 | 60 | 237 |
| R:CGTAGGCAAGAACGGATGAT | ||||
| M20 | F:CCCTACTTCGACCATTGCAT | (TTG)6 | 60 | 275 |
| R:CGAACCAAGAGTCCGTCAAT | ||||
| M25 | F:TGCTCCATAAGGTAGGGCAT | (TC)6 | 60 | 251 |
| R:TCACTCCCTCTGAAGCCATT | ||||
| M27 | F:GAAGACGCTTTCCTGCAGAT | (AT)6 | 60 | 218 |
| R:CACATCAAGCTTGCACGAGT | ||||
| M28 | F:GCCTAAGGCGCTTCTTTTCT | (GT)8 | 60 | 237 |
| R:ATGGCTAAACGAAGGGAGGT | ||||
| M29 | F:AGGGGCAGAAGATGTCCTTT | (GT)6 | 60 | 267 |
| R:TGCCTTGGCTTAGAAGCTGT | ||||
| M30 | F:GTGGTATTGTTGGGTTCGCT | (CA)6 | 60 | 276 |
| R:CCTCCAAAACCCTCCTCTTT | ||||
| M31 | F:CCATGGGCAAGAGAATCAGT | (CA)6 | 60 | 246 |
| R:GAAGTTCAGCCAACCACCAT | ||||
| M32 | F:TCGTCCCTGGAATGAGAAGT | (CA)6 | 60 | 240 |
| R:CTTAACCGATGGTGGCTTGT | ||||
| N2 | F:GCGTAATTTCGTCACCCACT | (AT)6 | 60 | 231 |
| R:CCTGACCGTTGGTTTGAGTT | ||||
| N8 | F:CCCACCCTCAAACATCACTT | (GC)6 | 60 | 230 |
| R:AATACCTTGGTTGAGGGGGT | ||||
| N10 | F:TGCAAGAGTCATCCATTTCAG | (AAG)5 | 60 | 260 |
| R:GCTGGGGACGTAATGTTGTT | ||||
| N13 | F:CCTCACACTCACAGCAGCAT | (CAG)6 | 62 | 218 |
| R:GTGGAGCTGTTACCGTTGGT |
| Locus | Na | Ne | PPB (%) | I | Ho | He | PIC | Fis | Fit | Fst | Nm | Fn | HWE |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| M17 | 14 | 3.594 | 85.71% | 1.457 | 0.821 | 0.697 | 0.755 | −0.178 | −0.036 | 0.121 | 1.818 | −0.037 | *** |
| M19 | 7 | 1.457 | 85.71% | 0.526 | 0.291 | 0.273 | 0.458 | −0.065 | 0.340 | 0.380 | 0.408 | 0.221 | * |
| M20 | 19 | 2.722 | 88.89% | 1.289 | 0.459 | 0.600 | 0.708 | 0.234 | 0.354 | 0.156 | 1.349 | 0.144 | NS |
| M25 | 22 | 6.601 | 90.48% | 2.016 | 0.791 | 0.831 | 0.879 | 0.048 | 0.112 | 0.067 | 3.457 | 0.014 | NS |
| M27 | 31 | 4.428 | 93.33% | 1.828 | 0.260 | 0.769 | 0.806 | 0.662 | 0.675 | 0.039 | 6.115 | 0.577 | *** |
| M28 | 20 | 3.956 | 94.74% | 1.628 | 0.747 | 0.739 | 0.765 | −0.012 | 0.047 | 0.058 | 4.064 | −0.011 | NS |
| M29 | 18 | 2.669 | 94.44% | 1.094 | 0.217 | 0.580 | 0.610 | 0.625 | 0.677 | 0.137 | 1.573 | 0.567 | *** |
| M30 | 12 | 1.804 | 100.00% | 0.783 | 0.226 | 0.407 | 0.570 | 0.444 | 0.632 | 0.339 | 0.487 | 0.510 | *** |
| M31 | 8 | 1.703 | 87.50% | 0.617 | 0.420 | 0.351 | 0.426 | −0.199 | 0.101 | 0.250 | 0.752 | 0.012 | NS |
| M32 | 14 | 2.469 | 100.00% | 1.006 | 0.292 | 0.511 | 0.639 | 0.430 | 0.591 | 0.282 | 0.635 | 0.334 | *** |
| N2 | 14 | 1.815 | 92.31% | 0.785 | 0.199 | 0.395 | 0.498 | 0.496 | 0.638 | 0.281 | 0.640 | 0.540 | *** |
| N8 | 15 | 2.238 | 100.00% | 0.907 | 0.476 | 0.451 | 0.519 | −0.056 | 0.227 | 0.268 | 0.684 | 0.144 | * |
| N10 | 14 | 1.471 | 92.86% | 0.519 | 0.247 | 0.236 | 0.242 | −0.046 | 0.080 | 0.121 | 1.823 | 0.093 | ND |
| N13 | 10 | 3.183 | 70.00% | 1.240 | 0.777 | 0.663 | 0.667 | −0.173 | −0.075 | 0.083 | 2.750 | −0.042 | *** |
| Mean | 15.571 | 2.865 | 91.14% | 1.121 | 0.445 | 0.536 | 0.610 | 0.158 | 0.312 | 0.185 | 1.897 | 0.219 |
| Populations | Na | Ne | I | Ho | He | HWE |
|---|---|---|---|---|---|---|
| P1 | 4.929 | 2.590 | 1.063 | 0.505 | 0.528 | M17 **, M20 *, M27 ***, M29 *, N2 *** |
| P2 | 4.571 | 2.624 | 0.920 | 0.348 | 0.439 | M17 *, M20 *, M27 ***, M28 ***, M29 **, M30 **, M32 ***, N13 *** |
| P3 | 6.214 | 3.225 | 1.213 | 0.540 | 0.583 | M19 ***, M25 *, M27 ***, M29 ***, M30 ***, N2 *** |
| P4 | 5.571 | 2.395 | 0.990 | 0.398 | 0.478 | M17 ***, M20 *, M25 *, M27 ***, M29 ***, M30 ***, M32 ***, N2 ***, N10 ***, N13 ** |
| P5 | 5.429 | 2.673 | 1.046 | 0.433 | 0.501 | M17 ***, M25 *, M27 ***, M28 *, M29 ***, M30 **, M32 ***, N13 ** |
| P6 | 8.500 | 3.723 | 1.494 | 0.488 | 0.666 | M17 ***, M19 **, M20 **, M27 ***, M28 **, M29 ***, M30 ***, M32 ***, N2 ***, N13 ** |
| P7 | 4.643 | 2.825 | 1.120 | 0.401 | 0.557 | M17 *, M20 **, M25 ***, M29 **, M32 **, N8 *, N13 ** |
| Mean | 5.694 | 2.865 | 1.121 | 0.445 | 0.536 |
| Source of Variation | d.f. | SSD | MSD | Variance Component | Variance Ratio | p Value |
|---|---|---|---|---|---|---|
| Among populations | 6 | 333.849 | 55.642 | 2.564 | 21.09% | 0.001 |
| Within populations | 121 | 1161.221 | 9.597 | 9.597 | 78.91% | 0.001 |
| Populations | P1 | P2 | P3 | P4 | P5 | P6 | P7 |
|---|---|---|---|---|---|---|---|
| P1 | 2.220 | 2.708 | 3.099 | 4.877 | 2.680 | 1.459 | |
| P2 | 0.101 | 2.182 | 1.854 | 2.919 | 1.573 | 0.883 | |
| P3 | 0.085 | 0.103 | 5.934 | 3.062 | 2.357 | 1.478 | |
| P4 | 0.075 | 0.119 | 0.040 | 3.611 | 2.093 | 1.415 | |
| P5 | 0.049 | 0.079 | 0.075 | 0.065 | 2.136 | 1.172 | |
| P6 | 0.085 | 0.137 | 0.096 | 0.107 | 0.105 | 1.827 | |
| P7 | 0.146 | 0.221 | 0.145 | 0.150 | 0.176 | 0.120 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiao, Y.; Jiang, X.; Lu, C.; Liu, J.; Diao, S.; Jiang, J. Genetic Diversity and Population Structure Analysis in the Chinese Endemic Species Michelia crassipes Based on SSR Markers. Forests 2023, 14, 508. https://doi.org/10.3390/f14030508
Xiao Y, Jiang X, Lu C, Liu J, Diao S, Jiang J. Genetic Diversity and Population Structure Analysis in the Chinese Endemic Species Michelia crassipes Based on SSR Markers. Forests. 2023; 14(3):508. https://doi.org/10.3390/f14030508
Chicago/Turabian StyleXiao, Yuguang, Xiaolong Jiang, Chengcheng Lu, Jun Liu, Shu Diao, and Jingmin Jiang. 2023. "Genetic Diversity and Population Structure Analysis in the Chinese Endemic Species Michelia crassipes Based on SSR Markers" Forests 14, no. 3: 508. https://doi.org/10.3390/f14030508
APA StyleXiao, Y., Jiang, X., Lu, C., Liu, J., Diao, S., & Jiang, J. (2023). Genetic Diversity and Population Structure Analysis in the Chinese Endemic Species Michelia crassipes Based on SSR Markers. Forests, 14(3), 508. https://doi.org/10.3390/f14030508








