Habitat Associations of Overwintering Bats in Managed Pine Forest Landscapes
Abstract
1. Introduction
2. Methods
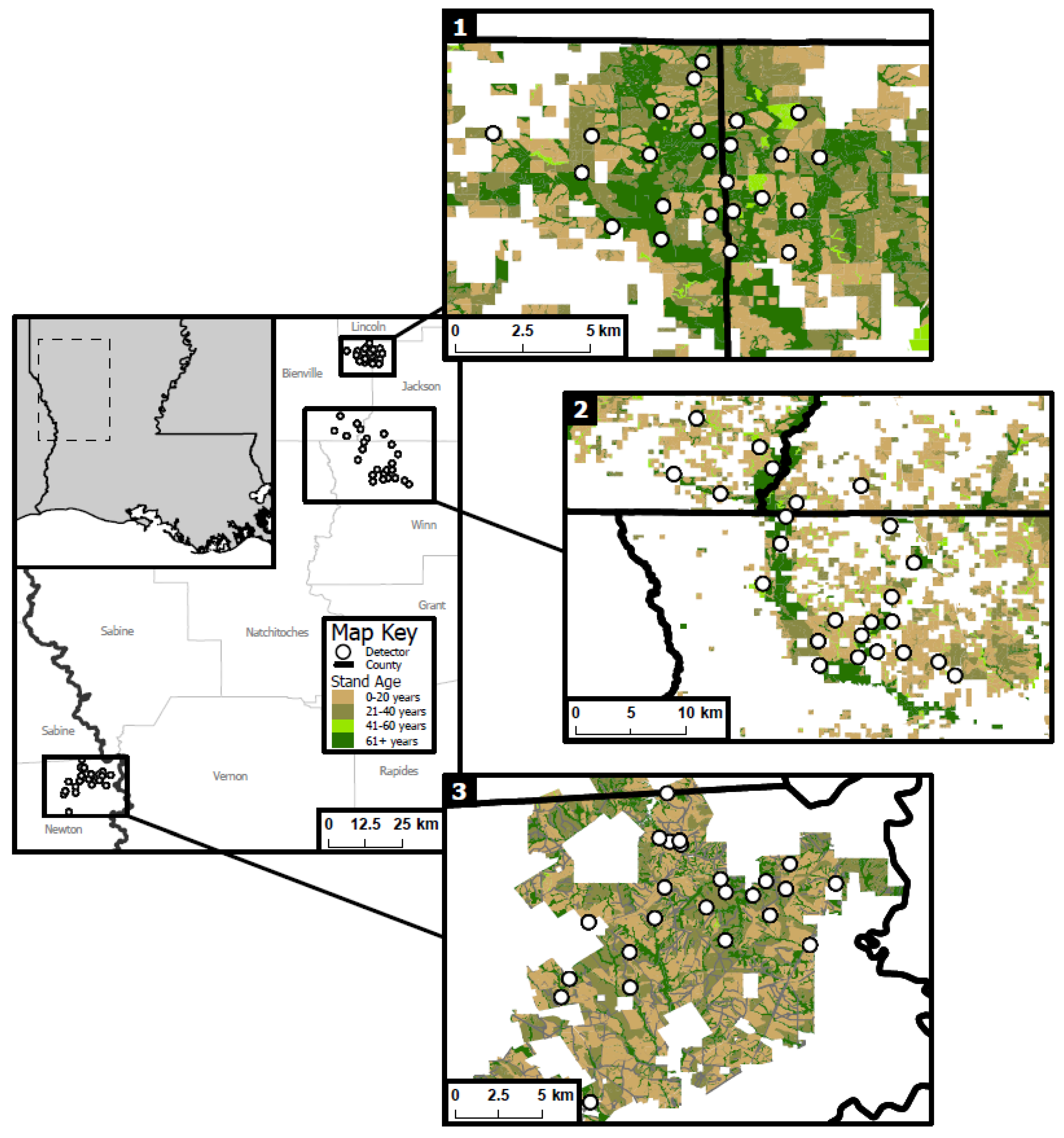
2.1. Study Area and Site Selection
2.2. Acoustic Detector Deployment and Monitoring
2.3. Acoustic Analysis
2.4. Microhabitat Assessment
2.5. Landscape Assessment
2.6. Statistical Analysis
3. Results
3.1. Principal Components Analysis
3.2. Habitat Association
4. Discussion
4.1. Species-Specific Habitat Use
4.1.1. Big Brown Bat (Eptesicus fuscus)
4.1.2. Eastern Red/Seminole Bat (Lasiurus borealis/seminolus)
4.1.3. Hoary Bat (Lasiurus cinereus)
4.1.4. Southeastern Myotis (Myotis austroriparius)
4.1.5. Evening Bat (Nycticeius humeralis)
4.1.6. Tricolored Bat (Perimyotis subflavus)
4.1.7. Brazilian Free-Tailed Bat (Tadarida brasiliensis)
4.2. Expanations for a Lack of Associations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Hastings, A. Disturbance, Coexistence, History, and Competition for Space. Theor. Popul. Biol. 1980, 18, 363–373. [Google Scholar] [CrossRef]
- Humphries, M.M.; Studd, E.K.; Menzies, A.K.; Boutin, S. To Everything There Is a Season: Summer-to-Winter Food Webs and the Functional Traits of Keystone Species. Integr. Comp. Biol. 2017, 57, 961–976. [Google Scholar] [CrossRef] [PubMed]
- Stevens, R.D.; Amarilla-Stevens, H.N. Dietary Patterns of Phyllostomid Bats in Interior Atlantic Forest of Eastern Paraguay. J. Mammal. 2021, 102, 685–694. [Google Scholar] [CrossRef]
- Marchand, P.J. Life in the Cold: An Introduction to Winter Ecology; University Press of New England: Lebanon, NH, USA, 2014. [Google Scholar]
- Dubowy, P.J. Waterfowl Communities and Seasonal Environments: Temporal Variability in Interspecific Competition. Ecology 1988, 69, 1439–1453. [Google Scholar] [CrossRef]
- Van Valen, L. Morphological Variation and Width of Ecological Niche. Am. Nat. 1965, 99, 377–390. [Google Scholar] [CrossRef]
- Speakman, J.R.; Thomas, D.W. Physiological Ecology and Energetics of Bats. In Bat Ecology; Kunz, T.H., Fenton, M.B., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 430–490. [Google Scholar]
- Czenze, Z.J.; Jonasson, K.A.; Willis, C.K.R. Thrifty Females, Frisky Males: Winter Energetics of Hibernating Bats from a Cold Climate. Physiol. Biochem. Zool. 2017, 90, 502–511. [Google Scholar] [CrossRef]
- Twente, J.W.; Twente, J.; Brack, V. The Duration of the Period of Hibernation of Three Species of Vespertilionid Bats. II. Laboratory Studies. Can. J. Zool. 1985, 63, 2955–2961. [Google Scholar] [CrossRef]
- Cryan, P.M. Seasonal Distribution of Migratory Tree Bats (Lasiurus and Lasionycteris) in North America. J. Mammal. 2003, 84, 579–593. [Google Scholar] [CrossRef]
- Webber, Q.M.R.; McGuire, L.P. Heterothermy, Body Size, and Locomotion as Ecological Predictors of Migration in Mammals. Mamm. Rev. 2022, 52, 82–95. [Google Scholar] [CrossRef]
- Boyles, J.G.; Dunbar, M.B.; Whitaker, J.J.O. Activity following Arousal in Winter in North American Vespertilionid Bats. Mamm. Rev. 2006, 36, 267–280. [Google Scholar] [CrossRef]
- Lausen, C.L.; Barclay, R.M.R. Winter Bat Activity in the Canadian Prairies. Can. J. Zool. 2006, 84, 1079–1086. [Google Scholar] [CrossRef]
- Falxa, G. Winter Foraging of Silver-Haired and California Myotis Bats in Western Washington. Northwest. Nat. 2007, 88, 98–100. [Google Scholar] [CrossRef]
- Schwab, N.A.; Mabee, T.J. Winter Acoustic Activity of Bats in Montana. Northwest. Nat. 2014, 95, 13–27. [Google Scholar] [CrossRef]
- White, J.A.; Andersen, B.R.; Otto, H.W.; Lemen, C.A.; Freeman, P.W. Winter Activity of Bats in Southeastern Nebraska: An Acoustic Study. Trans. Nebraska Acad. Sci. 2014, 34, 80–83. [Google Scholar]
- Grider, J.F.; Larsen, A.L.; Homyack, J.A.; Kalcounis-Ruepell, M.C. Winter Activity of Coastal Plain Populations of Bat Species Affected by White-Nose Syndrome and Wind Energy Facilities. PLoS ONE 2016, 11, e0166512. [Google Scholar] [CrossRef]
- Klug-Baerwald, B.J.; Gower, L.E.; Lausen, C.L.; Brigham, R.M. Environmental Correlates and Energetics of Winter Flight by Bats in Southern Alberta, Canada. Can. J. Zool. 2016, 94, 829–839. [Google Scholar] [CrossRef]
- Lemen, C.A.; Freeman, P.W.; White, J.A. Winter Activity of Myotis septentrionalis: Role of Temperature in Controlling Emergence from a Hibernaculum. Trans. Nebraska Acad. Sci. 2016, 36, 6–8. [Google Scholar]
- Bernard, R.F.; McCracken, G.F. Winter Behavior of Bats and the Progression of White-Nose Syndrome in the Southeastern United States. Ecol. Evol. 2017, 7, 1487–1496. [Google Scholar] [CrossRef]
- Johnson, J.S.; Treanor, J.J.; Lacki, M.J.; Baker, M.D.; Falxa, G.A.; Dodd, L.E.; Waag, A.G.; Lee, E.H. Migratory and Winter Activity of Bats in Yellowstone National Park. J. Mammal. 2017, 98, 211–221. [Google Scholar] [CrossRef]
- Reynolds, D.S.; Shoemaker, K.; Von Oettingen, S.; Najjar, S. High Rates of Winter Activity and Arousals in Two New England Bat Species: Implications for a Reduced White-Nose Syndrome Impact? Northeast. Nat. 2017, 24, B188–B208. [Google Scholar] [CrossRef]
- Arndt, R.J.; Lima, S.L. Landscape-Wide Flight Activity by Wintering Bats Predictably Follows Pulses of Warmth in the Midwestern United States. J. Mammal. 2020, 101, 1489–1501. [Google Scholar] [CrossRef]
- Parker, K.A.; Li, H.; Kalcounis-Rueppell, M.C. Species-Specific Environmental Conditions for Winter Bat Acoustic Activity in North Carolina, United States. J. Mammal. 2020, 101, 1502–1512. [Google Scholar] [CrossRef]
- Jorge, M.H.; Sweeten, S.E.; True, M.C.; Freeze, S.R.; Cherry, M.J.; Garrison, E.P.; Taylor, H.; Gorman, K.M.; Ford, W.M. Fire, Land Cover, and Temperature Drivers of Bat Activity in Winter. Fire Ecol. 2021, 17, 19. [Google Scholar] [CrossRef]
- Shute, K.E.; Loeb, S.C.; Jachowski, D.S. Seasonal Shifts in Nocturnal Habitat Use by Coastal Bat Species. J. Wildl. Manag. 2021, 85, 964–978. [Google Scholar] [CrossRef]
- Stevens, R.D.; Garcia, C.J.; Madden, M.A.; Gregory, B.B.; Perry, R.W. Seasonal Changes in the Active Bat Community of the Kisatchie National Forest, Louisiana. Southeast. Nat. 2020, 19, 524–536. [Google Scholar] [CrossRef]
- Dunbar, M.B.; Tomasi, T.E. Arousal Patterns, Metabolic Rate, and an Energy Budget of Eastern Red Bats (Lasiurus Borealis) in Winter. J. Mammal. 2006, 87, 1096–1102. [Google Scholar] [CrossRef]
- Mormann, B.M.; Robbins, L.W. Winter Roosting Ecology of Eastern Red Bats in Southwest Missouri. J. Wildl. Manag. 2007, 71, 213–217. [Google Scholar] [CrossRef]
- Weller, T.J.; Castle, K.T.; Liechti, F.; Hei, C.D.; Schirmacher, M.R.; Cryan, P.M. First Direct Evidence of Long-Distance Seasonal Movements and Hibernation in a Migratory Bat. Sci. Rep. 2016, 6, 34585. [Google Scholar] [CrossRef]
- Perry, R.W.; Saugey, D.A.; Crump, B.G. Winter Roosting Ecology of Silver-Haired Bats in an Arkansas Forest. Southeast. Nat. 2010, 9, 563–572. [Google Scholar] [CrossRef]
- Geluso, K. Winter Activity of Brazilian Free-Tailed Bats (Tadarida Brasiliensis) at Carlsbad Cavern, New Mexico. Southwest. Nat. 2008, 53, 243–247. [Google Scholar] [CrossRef]
- Perry, R.W. A Review of Factors Affecting Cave Climates for Hibernating Bats in Temperate North America. Environ. Rev. 2013, 21, 28–39. [Google Scholar] [CrossRef]
- Brigham, R.M. Bats in Forests: What We Know and What We Need to Know. In Bats in Forests: Conservation and Management; Lacki, M.J., Hayes, J.P., Kurta, A., Eds.; The Johns Hopkins University Press: Baltimore, MD, USA, 2007; pp. 1–16. [Google Scholar]
- Humphrey, S.R. Nursery Roosts and Community Diversity of Nearctic Bats. J. Mammal. 1975, 56, 321–346. [Google Scholar] [CrossRef]
- Gehrt, S.D.; Chelsvig, J.E. Bat Activity in an Urban Landscape: Patterns at the Landscape and Microhabitat Scale. Ecol. Appl. 2003, 13, 939–950. [Google Scholar] [CrossRef]
- Duchamp, J.E.; Swihart, R.K. Shifts in Bat Community Structure Related to Evolved Traits and Features of Human-Altered Landscapes. Landsc. Ecol. 2008, 23, 849–860. [Google Scholar] [CrossRef]
- Hein, C.D.; Castleberry, S.B.; Miller, K.V. Site-Occupancy of Bats in Relation to Forested Corridors. For. Ecol. Manag. 2009, 257, 1200–1207. [Google Scholar] [CrossRef]
- Morris, A.D.; Miller, D.A.; Kalcounis-Rueppell, M.C. Use of Forest Edges by Bats in a Managed Pine Forest Landscape. J. Wildl. Manage. 2010, 74, 26–34. [Google Scholar] [CrossRef]
- Ethier, K.; Fahrig, L. Positive Effects of Forest Fragmentation, Independent of Forest Amount, on Bat Abundance in Eastern Ontario, Canada. Landsc. Ecol. 2011, 26, 865–876. [Google Scholar] [CrossRef]
- Jantzen, M.K.; Fenton, M.B. The Depth of Edge Influence among Insectivorous Bats at Forest-Field Interfaces. Can. J. Zool. 2013, 91, 287–292. [Google Scholar] [CrossRef]
- Kirkpatrick, L.; Maher, S.J.; Lopez, Z.; Lintott, P.R.; Bailey, S.A.; Dent, D.; Park, K.J. Bat Use of Commercial Coniferous Plantations at Multiple Spatial Scales: Management and Conservation Implications. Biol. Conserv. 2017, 206, 1–10. [Google Scholar] [CrossRef]
- Krusic, R.A.; Yamasaki, M.; Neefus, C.D.; Perkins, P.J. Bat Habitat Use in White Mountain National Forest. J. Wildl. Manag. 1996, 60, 625–631. [Google Scholar] [CrossRef]
- Grindal, S.D.; Brigham, R.M. Short-Term Effects of Small-Scale Habitat Disturbance on Activity by Activity by Insectivorous Bats. J. Wildl. Manag. 1998, 62, 996–1003. [Google Scholar] [CrossRef]
- Menzel, M.A.; Carter, T.C.; Menzel, J.M.; Ford, W.M.; Chapman, B.R. Effects of Group Selection Silviculture in Bottomland Hardwoods on the Spatial Activity Patterns of Bats. For. Ecol. Manag. 2002, 162, 209–218. [Google Scholar] [CrossRef]
- Brooks, R.T. Habitat-Associated and Temporal Patterns of Bat Activity in a Diverse Forest Landscape of Southern New England, USA. Biodivers. Conserv. 2009, 18, 529–545. [Google Scholar] [CrossRef]
- Węgiel, A.; Grzywiński, W.; Ciechanowski, M.; Jaros, R.; Kalcounis-Rüppell, M.; Kmiecik, A.; Kmiecik, P.; Węgiel, J. The Foraging Activity of Bats in Managed Pine Forests of Different Ages. Eur. J. For. Res. 2019, 138, 383–396. [Google Scholar] [CrossRef]
- Miller, D.A.; Wigley, T.B.; Miller, K.V. Managed Forests and Conservation of Terrestrial Biodiversity in the Southern United States. J. For. 2009, 107, 197–203. [Google Scholar]
- Ober, H.K.; Jones, G.M.; Gottlieb, I.G.W.; Johnson, S.A.; Smith, L.; Brosi, B.J.; Fletcher, R.J. Bat Community Response to Intensification of Biomass Production for Bioenergy across the Southeastern United States. Ecol. Appl. 2020, 30, e02155. [Google Scholar] [CrossRef]
- Miller, D.A.; Arnett, E.B.; Lacki, M.J. Habitat Management for Forest-Roosting Bats of North America: A Critical Review of Habitat Studies. Wildl. Soc. Bull. 2003, 31, 30–44. [Google Scholar]
- Loeb, S.C.; Waldrop, T.A. Bat Activity in Relation to Fire and Fire Surrogate Treatments in Southern Pine Stands. For. Ecol. Manag. 2008, 255, 3185–3192. [Google Scholar] [CrossRef]
- Bender, M.J.; Castleberry, S.B.; Miller, D.A.; Bently Wigley, T. Site Occupancy of Foraging Bats on Landscapes of Managed Pine Forest. For. Ecol. Manag. 2015, 336, 1–10. [Google Scholar] [CrossRef]
- Vindigni, M.A.; Morris, A.D.; Miller, D.A.; Kalcounis-Rueppell, M.C. Use of Modified Water Sources by Bats in a Managed Pine Landscape. For. Ecol. Manag. 2009, 258, 2056–2061. [Google Scholar] [CrossRef]
- Kunz, T.H. Resource Utilization: Temporal and Spatial Components of Bat Activity in Central Iowa. J. Mammal. 1973, 54, 14–32. [Google Scholar] [CrossRef]
- Anthony, E.L.P.; Stack, M.H.; Kunz, T.H. Night Roosting and the Nocturnal Time Budget of the Little Brown Bat, Myotis Lucifugus: Effects of Reproductive Status, Prey Density, and Environmental Conditions. Oecologia 1981, 51, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Barclay, R.M.R. The Effect of Reproductive Condition on the Foraging Behavior of Female Hoary Bats, Lasiurus cinereus. Behav. Ecol. Sociobiol. 1989, 24, 31–37. [Google Scholar] [CrossRef]
- Frost, C. History and Future of the Longleaf Pine Ecosystem. In The Longleaf Pine Ecosystem; Jose, S., Jokela, E.J., Miller, D.I., Eds.; Springer: New York, NY, USA, 2006; pp. 9–48. [Google Scholar]
- Landers, J.L.; Van Lear, D.H.; Boyer, W.D. The Longleaf Pine Forests of the Southeast: Requiem or Renaissance? J. For. 1995, 93, 38–44. [Google Scholar]
- Holland, A.M.; Rutledge, B.T.; Jack, S.B.; Stober, J.M. The Longleaf Pine Forest: Long-Term Monitoring and Restoration of a Management Dependent Ecosystem. J. Nat. Conserv. 2019, 47, 38–50. [Google Scholar] [CrossRef]
- Wear, D.N.; Greis, J.G. The Southern Forest Resource Assessment: Summary Report; Southern Research Station: Asheville, NC, USA, 2002. [Google Scholar]
- Martinuzzi, S.; Withey, J.C.; Pidgeon, A.M.; Plantinga, A.J.; Mckerrow, A.J.; Williams, S.G.; Helmers, D.P.; Radeloff, V.C. Future Land-Use Scenarios and the Loss of Wildlife Habitats in the Southeastern United States. Ecol. Appl. 2015, 25, 160–171. [Google Scholar] [CrossRef]
- Bailey, R.G. Description of the Ecoregions of the United States; US Department of Agriculture, Forest Service: Fort Collins, CO, USA, 1995. [Google Scholar]
- Weller, T.J.; Zabel, C.J. Variation in Bat Detections in Forest to Detector Orientation. Wildl. Soc. Bull. 2011, 30, 922–930. [Google Scholar]
- Britzke, E.R.; Gillam, E.H.; Murray, K.L. Current State of Understanding of Ultrasonic Detectors for the Study of Bat Ecology. Acta Theriol. 2013, 58, 109–117. [Google Scholar] [CrossRef]
- Lowery, G.H. Mammals of Louisiana and Adjacent Waters; Louisiana State University Press: Baton Rouge, LA, USA, 1974. [Google Scholar]
- Lance, R.F.; Garrett, R.W. Bat Fauna of Central Louisiana Forests. Texas J. Sci. 1997, 49, 181–189. [Google Scholar]
- Stevens, R.D.; Garcia, C.J.; Bohlender, E.E.; Gregory, B.B. Distributional Updates and Conservation Status of Bats from Louisiana. Occas. Pap. Museum Texas Tech Univ. 2017, 348, 1–12. [Google Scholar]
- Foster, R.W.; Kurta, A. Roosting Ecology of the Northern Bat (Myotis septentrionalis) and Comparisons with the Endangered Indiana Bat (Myotis sodalis). J. Mammal. 1999, 80, 659–672. [Google Scholar] [CrossRef]
- Weller, T.J.; Zabel, C.J. Characteristics of Fringed Myotis Day Roosts in Northern California. J. Wildl. Manag. 2001, 65, 489–497. [Google Scholar] [CrossRef]
- United States Geological Survey USGS National Transportation Dataset (NTD) for Louisiana. Available online: https://www.sciencebase.gov/catalog/item/5a5f36c4e4b06e28e9bfc1ca (accessed on 30 April 2019).
- Texas Department of Transportation TxDOT Roadways. Available online: https://gis-txdot.opendata.arcgis.com/datasets/txdot-roadways/explore?location=31.121687%2C-100.055172%2C6.76 (accessed on 30 April 2019).
- United States Geological Survey National Hydrography Dataset. Available online: https://www.usgs.gov/national-hydrography/national-hydrography-dataset (accessed on 6 April 2019).
- United States Geological Survey. The National Map. Available online: https://www.usgs.gov/programs/national-geospatial-program/national-map (accessed on 6 April 2019).
- Multi-Resolution Land Characteristics Consortium NLCD 2016 Land Cover (CONUS). Available online: https://www.mrlc.gov/data/nlcd-2016-land-cover-conus (accessed on 6 April 2019).
- Ford, W.M.; Menzel, J.M.; Menzel, M.A.; Edwards, J.W.; Kilgo, J.C. Presence and Absence of Bats across Habitat Scales in the Upper Coastal Plain of South Carolina. J. Wildl. Manag. 2006, 70, 1200–1209. [Google Scholar] [CrossRef]
- McGarigal, K.; Cushman, S.A.; Ene, E. FRAGSTATS v4: Spatial Pattern Analysis Program for Categorical and Continuous Maps. Available online: http://www.umass.edu/landeco/research/fragstats/fragstats.html (accessed on 21 December 2021).
- Team, R.C. R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org (accessed on 21 December 2021).
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; R Core Team. Nlme: Linear and Nonlinear Mixed Effects Models. Available online: https://cran.r-project.org/package=nlme (accessed on 21 December 2021).
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous Inference in General Parametric Models. Biometr. J. 2008, 50, 346–363. [Google Scholar] [CrossRef] [PubMed]
- Wold, S.; Esbensen, K.; Geladi, P. Principal Component Analysis. Chemom. Intell. Lab. Syst. 1987, 2, 37–52. [Google Scholar] [CrossRef]
- Peres-Neto, P.R.; Jackson, D.A.; Somers, K.M. How Many Principal Components? Stopping Rules for Determining the Number of Non-Trivial Axes Revisited. Comput. Stat. Data Anal. 2005, 49, 974–997. [Google Scholar] [CrossRef]
- Hair, J.F.; Black, W.C.; Babin, B.J.; Anderson, R.E.; Tatham, R.L. Multivariate Data Analysis, 6th ed.; Pearson Prentice Hall: Upper Saddle River, NJ, USA, 2006. [Google Scholar]
- Andersen, B.R. Influences of Habitat and Temperature on Winter Bat Assemblages; Texas Tech University: Lubbock, TX, USA, 2020. [Google Scholar]
- Norberg, U.M.; Rayner, J.M.V. Ecological Morphology and Flight in Bats (Mammalia: Chiroptera): Wing Adaptations, Flight Performance, Foraging Strategy and Echolocation. Philos. Trans. R. Soc. London B Biol. Sci. 1987, 316, 335–427. [Google Scholar]
- Norberg, U.M. Wing Design, Flight Performance, and Habitat Use in Bats. In Ecological Morphology: Integrative Organismal Biology; Wainwright, P.C., Reilly, S.M., Eds.; University of Chicago Press: Chicago, IL, USA, 1994; pp. 205–239. [Google Scholar]
- Marinello, M.M.; Bernard, E. Wing Morphology of Neotropical Bats: A Quantitative and Qualitative Analysis with Implications for Habitat Use. Can. J. Zool. 2014, 92, 141–147. [Google Scholar] [CrossRef]
- Mellanby, K. Low Temperature and Insect Activity. Proc. R. Soc. B Biol. Sci. 1939, 127, 473–487. [Google Scholar]
- Heinrich, B. Flight of Winter Moths near 0 °C. Science 1985, 228, 177–179. [Google Scholar] [CrossRef]
- Holyoak, M.; Jarosik, V.; Novák, I. Weather-Induced Changes in Moth Activity Bias Measurement of Long-Term Population Dynamics from Light Trap Samples. Entomol. Exp. Appl. 1997, 83, 329–335. [Google Scholar] [CrossRef]
- Hein, C.D.; Castleberry, S.B.; Miller, K.V. Male Seminole Bat Winter Roost-Site Selection in a Managed Forest. J. Wildl. Manage. 2008, 72, 1756–1764. [Google Scholar] [CrossRef]
- Neece, B.D.; Loeb, S.C.; Jachowski, D.S. Variation in Regional and Landscape Effects on Occupancy of Temperate Bats in the Southeastern U.S. PLoS ONE 2018, 13, e0206857. [Google Scholar] [CrossRef] [PubMed]
- Menzel, M.A.; Menzel, J.M.; Ford, W.M.; Edwards, J.W.; Carter, T.C.; Churchill, J.B.; Kilgo, J.C. Home Range and Habitat Use of Male Rafinesque’s Big-Eared Bats (Corynorhinus rafinesquii). Am. Midl. Nat. 2001, 145, 402–408. [Google Scholar] [CrossRef]
- Faure, P.A.; Fullard, J.H.; Dawson, J.W. The Gleaning Attacks of the Northern Long-Eared Bat, Myotis septentrionalis, Are Relatively Inaudible to Moths. J. Exp. Biol. 1993, 178, 173–189. [Google Scholar] [CrossRef] [PubMed]
- Murray, K.L.; Britzke, E.R.; Hadley, B.M.; Robbins, L.W. Surveying Bat Communities: A Comparison between Mist Nets and the Anabat II Bat Detector System. Acta Chiropterolog. 1999, 1, 105–112. [Google Scholar]
- Broders, H.G.; Findlay, C.S.; Zheng, L. Effects of Clutter on Echolocation Call Structure of Myotis septentrionalis and M. lucifugus. J. Mammal. 2004, 85, 273–281. [Google Scholar] [CrossRef]
- Meierhofer, M.B.; Johnson, J.S.; Leivers, S.J.; Pierce, B.L.; Evans, J.E.; Morrison, M.L. Winter Habitats of Bats in Texas. PLoS ONE 2019, 14, e0220839. [Google Scholar] [CrossRef]
- Trousdale, A.W.; Beckett, D.C. Characteristics of Tree Roosts of Rafinesque’s Big-Eared Bat (Corynorhinus rafinesquii) in Southeastern Mississippi. Am. Midl. Nat. 2005, 154, 442–449. [Google Scholar] [CrossRef]
- Whitaker, J.O.; Rissler, L.J. Seasonal Activity of Bats at Copperhead Cave. Proc. Indiana Acad. Sci. 1992, 101, 127–135. [Google Scholar]
- Whitaker, J.O.; Rissler, L.J. Winter Activity of Bats at a Mine Entrance in Vermillion County, Indiana. Am. Midl. Nat. 1992, 127, 52–59. [Google Scholar] [CrossRef]
- Schmidly, D.J.; Bradley, R.D. The Mammals of Texas, 7th ed.; University of Texas Press: Austin, TX, USA, 2016. [Google Scholar]
- Crnkovic, A.C. Discovery of Northern Long-Eared Myotis, Myotis septentrionalis (Chiroptera: Vespertilionidae), in Louisiana. Southwest. Nat. 2003, 48, 715–717. [Google Scholar] [CrossRef]
- Kurta, A.; Baker, R.H. Eptesicus fuscus. Mamm. Species 1990, 356, 1–10. [Google Scholar] [CrossRef]
- Ellis, A.M.; Patton, L.L.; Castleberry, S.B. Bat Activity in Upland and Riparian Habitats in the Georgia Piedmont. Proc. Annu. Conf. Southeast. Assoc. Fish Wildl. Agencies 2002, 56, 210–218. [Google Scholar]
- Menzel, J.; Menzel, M.; Kilgo, J.; Ford, W.M.; Edwards, J.W.; McCracken, G.F. Effect of Habitat and Foraging Height on Bat Activity in the Coastal Plain of South Carolina. J. Wildl. Manag. 2005, 69, 235–245. [Google Scholar] [CrossRef]
- Cryan, P.M.; Bogan, M.A.; Yanega, G.M. Roosting Habits of Four Bat Species in the Black Hills of South Dakota. Acta Chiropterolog. 2001, 3, 43–52. [Google Scholar]
- Rancourt, S.J.; Rule, M.I.; O’Connell, M.A. Maternity Roost Site Selection of Big Brown Bats in Ponderosa Pine Forests of the Channeled Scablands of North Eastern Washington State, USA. For. Ecol. Manag. 2007, 248, 183–192. [Google Scholar] [CrossRef]
- Perry, R.W.; Thill, R.E. Roost Selection by Big Brown Bats in Forests of Arkansas: Importance of Pine Snags and Open Forest Habitats to Males. Southeast. Nat. 2008, 7, 607–618. [Google Scholar] [CrossRef]
- Barclay, R.M.R.; Brigham, R.M. Prey Detection, Dietary Niche Breadth, and Body Size in Bats: Why Are Aerial Insectivorous Bats so Small? Am. Nat. 1991, 137, 693–703. [Google Scholar]
- Loeb, S.C.; O’Keefe, J.M. Habitat Use by Forest Bats in South Carolina in Relation to Local, Stand, and Landscape Characteristics. J. Wildl. Manag. 2006, 70, 1210–1218. [Google Scholar] [CrossRef]
- Whitaker, J.O.; Gummer, S.L. Hibernation of the Big Brown Bat Eptesicus Fuscus in Buildings. J. Mammal. 1992, 73, 312–316. [Google Scholar] [CrossRef]
- Whitaker, J.O.; Gummer, S.L. Population Structure and Dynamics of Big Brown Bats (Eptesicus fuscus) Hibernating in Buildings in Indiana. Am. Midl. Nat. 2000, 143, 389–396. [Google Scholar] [CrossRef]
- Halsall, A.L.; Boyles, J.G.; Whitaker, J.O. Body Temperature Patterns of Big Brown Bats during Winter in a Building Hibernaculum. J. Mammal. 2012, 93, 497–503. [Google Scholar] [CrossRef]
- Furlonger, C.L.; Dewar, H.J.; Fenton, M.B. Habitat Use by Foraging Insectivorous Bats. Can. J. Zool. 1987, 65, 284–288. [Google Scholar] [CrossRef]
- Jung, T.S.; Thompson, I.D.; Titman, R.D.; Applejohn, A.P. Habitat Selection by Forest Bats in Relation to Mixed-Wood Stand Types and Structure in Central Ontario. J. Wildl. Manag. 1999, 63, 1306–1319. [Google Scholar] [CrossRef]
- Hutchinson, J.T.; Lacki, M.J. Foraging Behavior and Habitat Use of Red Bats in Mixed Mesophytic Forests of the Cumberland Plateau, Kentucky. In 12th Central Hardwood Forest Conference, U.S. Forest Service; Stringer, J.W., Loftis, D.L., Eds.; Southern Experiment Station: Asheville, NC, USA, 1999; pp. 171–177. [Google Scholar]
- Elmore, L.W.; Miller, D.A.; Vilella, F.J. Foraging Area Size and Habitat Use by Red Bats (Lasiurus borealis) in an Intensively Managed Pine Landscape in Mississippi. Am. Midl. Nat. 2005, 153, 405–417. [Google Scholar] [CrossRef]
- Jorge, M.H.; Ford, W.M.; Sweeten, S.E.; Freeze, S.R.; True, M.C.; Germain, M.J.S.; Taylor, H.; Gorman, K.M.; Garrison, E.P.; Cherry, M.J. Winter Roost Selection of Lasiurine Tree Bats in a Pyric Landscape. PLoS ONE 2021, 16, e0245695. [Google Scholar] [CrossRef]
- Menzel, M.A.; Carter, T.C.; Chapman, B.R.; Laerm, J. Quantitative Comparison of Tree Roosts Used by Red Bats (Lasiurus borealis) and Seminole Bats (L. seminolus). Can. J. Zool. 1998, 76, 630–634. [Google Scholar] [CrossRef][Green Version]
- Menzel, M.A.; Carter, T.C.; Ford, W.M.; Chapman, B.R.; Ozier, J. Summer Roost Tree Selection by Eastern Red (Lasiurus borealis), Seminole (L. seminolus), and Evening (Nycticeius humeralis) Bats in the Upper Coastal Plain of South Carolina. Proc. Annu. Conf. / Southeast. Assoc. Fish Wildl. Agencies 2000, 54, 304–313. [Google Scholar]
- Hutchinson, J.T.; Lacki, M.J. Selection of Day Roosts by Red Bats in Mixed Mesophytic Forests. J. Wildl. Manag. 2000, 64, 87–94. [Google Scholar] [CrossRef]
- Mager, K.J.; Nelson, T.A. Roost-Site Selection by Eastern Red Bats (Lasiurus borealis). Am. Midl. Nat. 2001, 145, 120–126. [Google Scholar] [CrossRef]
- Flinn, J.R.; Perry, R.W.; Robbins, L.W. Winter Roosting by Eastern Red Bats in Ozark Mountain Forests of Missouri. Forests 2021, 12, 1769. [Google Scholar] [CrossRef]
- Perry, R.W.; Thill, R.E. Summer Roosting by Adult Male Seminole Bats in the Ouachita Mountains, Arkansas. Am. Midl. Nat. 2007, 158, 361–368. [Google Scholar] [CrossRef]
- Hein, C.D.; Castleberry, S.B.; Miller, K.V. Winter Roost-Site Selection by Seminole Bats in the Lower Coastal Plain of South Carolina. Southeast. Nat. 2005, 4, 473–478. [Google Scholar] [CrossRef]
- Hein, C.D.; Castleberry, S.B.; Miller, K.V. Sex-Specific Summer Roost-Site Selection by Seminole Bats in Response to Landscape-Level Forest Management. J. Mammal. 2008, 89, 964–972. [Google Scholar] [CrossRef]
- Perry, R.W. Migration and Recent Range Expansion of Seminole Bats (Lasiurus Seminolus) in the United States. J. Mammal. 2018, 99, 1478–1485. [Google Scholar] [CrossRef]
- Menzel, J.M.; Menzel, M.A.; Kilgo, J.C.; Ford, W.M.; Carter, T.C.; Edwards, J.W. Bats of the Savannah River Site and Vicinity; US Department of Agriculture, Forest Service, Southern Research Station: Asheville, NC, USA, 2003.
- Willis, C.K.R.; Brigham, R.M. Physiological and Ecological Aspects of Roost Selection by Reproductive Female Hoary Bats (Lasiurus cinereus). J. Mammal. 2005, 86, 85–94. [Google Scholar] [CrossRef]
- Perry, R.W.; Thill, R.E. Roost Characteristics of Hoary Bats in Arkansas. Am. Midl. Nat. 2007, 158, 132–138. [Google Scholar] [CrossRef]
- Veilleux, J.P.; Moosman, P.R.; Reynolds, D.S.; Lagory, K.E.; Walston, L.J. Observations of Summer Roosting and Foraging Behavior of a Hoary Bat (Lasiurus cinereus) in Southern New Hampshire. Northeast. Nat. 2009, 16, 148–153. [Google Scholar] [CrossRef]
- Klug, B.J.; Goldsmith, D.A.; Barclay, R.M.R. Roost Selection by the Solitary, Foliage-Roosting Hoary Bat (Lasiurus cinereus) during Lactation. Can. J. Zool. 2012, 90, 329–336. [Google Scholar] [CrossRef]
- Marín, G.; Ramos-H, D.; Cafaggi, D.; Sierra-Durán, C.; Gallegos, A.; Romero-Ruiz, A.; Medellín, R. Challenging Hibernation Limits of Hoary Bats: The Southernmost Record of Lasiurus cinereus Hibernating in North America. Mamm. Biol. 2021, 101, 287–291. [Google Scholar] [CrossRef]
- Saugey, D.A.; Vaughn, R.L.; Crump, B.G.; Heidt, G.A. Notes on the Natural History of Lasiurus borealis in Arkansas. J. Arkanas Acad. Sci. 1998, 52, 92–98. [Google Scholar]
- Boyles, J.G.; Timpone, J.C.; Robbins, L.W. Late-Winter Observations of Red Bats, Lasiurus borealis, and Evening Bats, Nycticeius humeralis, in Missouri. Bat Res. News 2003, 44, 59–61. [Google Scholar]
- Bender, M.J.; Hartman, G.D. Bat Activity Increases with Barometric Pressure and Temperature during Autumn in Central Georgia. Southeast. Nat. 2015, 14, 231–242. [Google Scholar] [CrossRef]
- Rice, D.W. Life History and Ecology of Myotis austroriparius in Florida. J. Mammal. 1957, 38, 15–32. [Google Scholar] [CrossRef]
- Fleming, H.L.; Jones, J.C.; Belant, J.L.; Richardson, D.M. Multi-Scale Roost Site Selection by Rafinesque’s Big-Eared Bat (Corynorhinus rafinesquii) and Southeastern Myotis (Myotis austroriparius) in Mississippi. Am. Midl. Nat. 2013, 169, 43–55. [Google Scholar] [CrossRef]
- Meierhofer, M.B.; Leivers, S.J.; Fern, R.R.; Wolf, L.K.; Young, J.H.; Pierce, B.L.; Evans, J.W.; Morrison, M.L. Structural and Environmental Predictors of Presence and Abundance of Tri-Colored Bats in Texas Culverts. J. Mammal. 2019, 100, 1274–1281. [Google Scholar] [CrossRef]
- Boyles, J.G.; Robbins, L.W. Characteristics of Summer and Winter Roost Trees Used by Evening Bats (Nycticeius humeralis) in Southwestern Missouri. Am. Midl. Nat. 2006, 155, 210–220. [Google Scholar] [CrossRef]
- Kaarakka, H.M.; White, J.P.; Redell, J.A.; Luukkonen, K.L. Notes on Capture and Roost Characteristics of Three Female Evening Bats (Nycticeius humeralis) in Southern Wisconsin. Am. Midl. Nat. 2018, 180, 168–172. [Google Scholar] [CrossRef]
- Humphrey, S.R.; Cope, J.B. Records of Migration of the Evening Bat, Nycticeius humeralis. J. Mammal. 1968, 49, 329. [Google Scholar] [CrossRef]
- Watkins, L.C. Observations on the Distribution and Natural History of the Evening Bat (Nycticeius humeralis) in Northwestern Missouri and Adjacent Iowa. Trans. Kans. Acad. Sci. 1969, 72, 330–336. [Google Scholar] [CrossRef]
- Perry, R.W.; Thill, R.E. Diurnal Roosts of Male Evening Bats (Nycticeius humeralis) in Diversely Managed Pine- Hardwood Forests. Am. Midl. Nat. 2008, 160, 374–385. [Google Scholar] [CrossRef]
- Sandel, J.K.; Benatar, G.R.; Burke, K.M.; Walker, C.W.; Lacher, J.; Honeycutt, R.L. Use and Selection of Winter Hibernacula by the Eastern Pipistrelle (Pipistrellus subflavus) in Texas. J. Mammal. 2001, 82, 173–178. [Google Scholar] [CrossRef]
- Ferrara, F.J.; Leberg, P.L. Characteristics of Positions Selected by Day-Roosting Bats under Bridges in Louisiana. J. Mammal. 2005, 86, 729–735. [Google Scholar] [CrossRef]
- Leivers, S.J.; Meierhofer, M.B.; Pierce, B.L.; Evans, J.W.; Morrison, M.L. External Temperature and Distance from Nearest Entrance Influence Microclimates of Cave and Culvert-Roosting Tri-Colored Bats (Perimyotis subflavus). Ecol. Evol. 2019, 9, 14042–14052. [Google Scholar] [CrossRef]
- Newman, B.A.; Loeb, S.C.; Jachowski, D.S. Winter Roosting Ecology of Tricolored Bats (Perimyotis subflavus) in Trees and Bridges. J. Mammal. 2021, 102, 1331–1341. [Google Scholar] [CrossRef]
- LaVaL, R.K. Observations on the Biology of Tadarida brasiliensis cynocephala in Southeastern Louisiana. Am. Midl. Nat. 1973, 89, 112–120. [Google Scholar] [CrossRef]
- Constantine, D.G. Ecological Observations on Lasiurine Bats in Georgia. J. Mammal. 1958, 39, 64–70. [Google Scholar] [CrossRef]
- Erickson, J.L.; West, S.D. Associations of Bats with Local Structure and Landscape Features of Forested Stands in Western Oregon and Washington. Biol. Conserv. 2003, 109, 95–102. [Google Scholar] [CrossRef]
- Miles, A.C.; Castleberry, S.B.; Miller, D.A.; Conner, L.M. Multi-Scale Roost-Site Selection by Evening Bats on Pine-Dominated Landscapes in Southwest Georgia. J. Wildl. Manag. 2006, 70, 1191–1199. [Google Scholar] [CrossRef]
- Florence, R.G. Ecology and Silviculture of Eucalypt Forests; CSIRO Publishing: Collingwood, Australia, 1996. [Google Scholar]
- Chen, G.; Pan, S.; Hayes, D.J.; Tian, H. Spatial and Temporal Patterns of Plantation Forests in the United States since the 1930s: An Annual and Gridded Data Set for Regional Earth System Modeling. Earth Syst. Sci. Data 2017, 9, 545–556. [Google Scholar] [CrossRef]
- Thomas, D.W. The Distribution of Bats in Different Ages of Douglas-Fir Forests. J. Wildl. Manag. 1988, 52, 619–626. [Google Scholar] [CrossRef]
- Crampton, L.H.; Barclay, R.M.R. Selection of Roosting and Foraging Habitat by Bats in Different-Aged Aspen Mixedwood Stands. Conserv. Biol. 1998, 12, 1347–1358. [Google Scholar] [CrossRef]
- Menzel, M.A.; Carter, T.C.; Ford, W.M.; Chapman, B.R. Tree-Roost Characteristics of Subadult and Female Adult Evening (Nycticeius humeralis) in the Upper Coastal Plain of South Carolina. Am. Midl. Nat. 2001, 145, 112–119. [Google Scholar] [CrossRef]
- Manly, B.F.; McDonald, L.L.; Thomas, D.L.; Mcdonald, T.L.; Erickson, W.P. Resource Selection by Animals: Statistical Design and Analysis for Field Studies; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002. [Google Scholar]
- Barclay, R.M.R.; Fullard, J.H.; Jacobs, D.S. Variation in the Echolocation Calls of the Hoary Bat (Lasiurus cinereus): Influence of Body Size, Habitat Structure, and Geographic Location. Can. J. Zool. 1999, 77, 530–534. [Google Scholar] [CrossRef]
- Lemen, C.; Freeman, P.W.; White, J.A.; Andersen, B.R. The Problem of Low Agreement among Automated Identification Programs for Acoustical Surveys of Bats. West. North Am. Nat. 2015, 75, 218–225. [Google Scholar] [CrossRef]
- Russo, D.; Voigt, C.C. The Use of Automated Identification of Bat Echolocation Calls in Acoustic Monitoring: A Cautionary Note for a Sound Analysis. Ecol. Indic. 2016, 66, 598–602. [Google Scholar] [CrossRef]
- Cryan, P.M.; Veilleux, J.P. Migration and Use of Autumn, Winter and Spring Roosts by Tree Bats. In Bats in Forests: Conservation and Management; Hayes, J.P., Lacki, M.J., Kurta, A., Eds.; The Johns Hopkins University Press: Baltimore, MD, USA, 2007; pp. 153–175. [Google Scholar]
- Hayes, J.P.; Loeb, S.C. The Influences of Forest Management on Bats in North America. In Bats in Forests: Conservation and Management; Lacki, M.J., Hayes, J.P., Kurta, A., Eds.; The Johns Hopkins University Press: Baltimore, MD, USA, 2007; pp. 207–236. [Google Scholar]
| Microhabitat | H-PC1 | H-PC2 | H-PC3 | Landscape | L450-PC1 | L450-PC2 | L1000-PC1 | L1000-PC2 |
|---|---|---|---|---|---|---|---|---|
| Stand Age (Years) | 0.36 | −0.22 | <0.01 | Landcover Diversity | −0.40 | −0.29 | −0.34 | −0.30 |
| % Canopy Clutter | 0.27 | 0.11 | −0.18 | Total Edge (m) | −0.38 | −0.04 | −0.35 | −0.02 |
| % Understory Clutter | −0.04 | 0.05 | 0.43 | Total Forest Edge (m) | −0.38 | 0.21 | −0.40 | −0.04 |
| # Trees | 0.29 | 0.42 | −0.12 | Total Core Area (m2) | 0.42 | −0.01 | 0.38 | 0.10 |
| Mean DBH (cm) | −0.04 | −0.45 | −0.33 | Total Contagion | 0.28 | 0.09 | 0.41 | 0.08 |
| Mean Tree Height (m) | 0.11 | −0.38 | −0.33 | % Open Water | 0.09 | −0.12 | 0.02 | −0.17 |
| # Conifers | −0.22 | 0.39 | −0.30 | % Developed, Open Space | −0.11 | 0.27 | −0.12 | 0.08 |
| # Hardwoods | 0.42 | 0.01 | 0.15 | % Developed, Low Intensity | −0.23 | 0.22 | −0.18 | 0.07 |
| # Overstory Trees | 0.01 | 0.47 | −0.35 | % Developed, Moderate Intensity | −0.08 | 0.26 | −0.18 | 0.31 |
| # Understory Trees | 0.39 | 0.05 | 0.22 | % Developed, High Intensity | −0.16 | 0.06 | −0.11 | 0.19 |
| # Snags | 0.34 | 0.02 | −0.05 | % Barren Land | −0.05 | 0.16 | −0.10 | 0.24 |
| % Snags | 0.31 | −0.02 | −0.05 | % Deciduous Forest | −0.13 | −0.16 | −0.02 | −0.17 |
| Distance to Water (m) | −0.28 | −0.10 | −0.08 | % Coniferous Forest | 0.09 | 0.55 | 0.02 | 0.52 |
| Distance to Road (m) | −0.11 | 0.01 | 0.43 | % Mixed Forest | −0.07 | −0.02 | −0.11 | 0.17 |
| Elevation (m) | −0.12 | 0.13 | 0.26 | % Shrub/Scrub | −0.27 | −0.04 | −0.25 | −0.26 |
| % Hay/Pasture | NA | NA | 0.02 | −0.03 | ||||
| % Herbaceous | −0.25 | −0.03 | −0.29 | 0.13 | ||||
| % Woody Wetlands | 0.12 | −0.50 | 0.17 | −0.44 | ||||
| % Emergent Wetlands | −0.13 | −0.22 | −0.05 | −0.25 | ||||
| Proportion of Variance | 0.304 | 0.178 | 0.152 | 0.223 | 0.139 | 0.245 | 0.141 |
| Species | # of Detector Nights Present | Temperature Cutoff (°C) | # of Detector Nights Present Above Threshold | % Detected Nights Discarded |
|---|---|---|---|---|
| All Species | 937 | 11.5 | 657 | 29.9% |
| Big brown bat | 182 | 15.5 | 145 | 20.3% |
| Eastern red/Seminole bat | 609 | 12 | 517 | 15.1% |
| Hoary bat | 134 | 10 | 128 | 5.2% |
| Southeastern myotis | 389 | 7 | 364 | 6.4% |
| Evening bat | 102 | 9.5 | 96 | 5.9% |
| Tricolored bat | 256 | 14.0 | 221 | 13.7% |
| Brazilian free-tailed bat | 158 | 8.5 | 153 | 3.2% |
| Species | Number of Passes | Number of Sites Detected | Percentage of Sites Detected | Mean Number of Bat Passes per Site per Night (±SE) |
|---|---|---|---|---|
| All Bats | 36,724 | 72 | 100% | 24.0 ± 3.6 |
| Rafinesque’s big-eared bat * | 12 | 8 | 11.1% | 0.008 ± 0.003 |
| Big brown bat | 2711 | 50 | 69.4% | 1.8 ± 0.4 |
| Eastern red/Seminole bat | 6129 | 72 | 100% | 3.85 ± 0.4 |
| Hoary bat | 608 | 34 | 47.2% | 0.4 ± 0.1 |
| Northern yellow bat * | 119 | 6 | 8.3% | 0.03 ± 0.02 |
| Silver-haired bat * | 52 | 15 | 20.8% | 0.07 ± 0.03 |
| Southeastern myotis | 1900 | 64 | 88.9% | 1.1 ± 0.1 |
| Northern long-eared bat * | 101 | 34 | 47.2% | 0.07 ± 0.02 |
| Evening bat | 344 | 37 | 51.3% | 0.22 ± 0.06 |
| Tricolored bat | 1725 | 56 | 77.8% | 1.1 ± 0.2 |
| Brazilian free-tailed bat | 340 | 50 | 69.4% | 0.2 ± 0.04 |
| Species | Model Sig. | Sunset Temp | H-PC1 | H-PC2 | H-PC3 | L1000-PC1 | L1000-PC2 |
|---|---|---|---|---|---|---|---|
| All Species | p < 0.0001 | 0.25 (p < 0.0001) | n.s. | n.s. | n.s. | n.s. | 8.60 (p = 0.04) |
| Big brown bat | p < 0.0001 | 0.06 (p = 0.002) | n.s. | n.s. | −1.45 (p = 0.003) | n.s. | −2.06 (p = 0.004) |
| Eastern red/Seminole bats | p < 0.0001 | 0.02 (p = 0.008) | n.s. | n.s. | 1.25 (p = 0.001) | n.s. | n.s. |
| Hoary bat | p = 0.04 | 0.003 (p = 0.001) | n.s. | n.s. | n.s. | n.s. | n.s. |
| Southeastern myotis | p = 0.18 | ||||||
| Evening bat | p < 0.001 | 0.003 (p = 0.0002) | n.s. | n.s. | n.s. | −0.13 (p = 0.03) | 0.18 (p = 0.01) |
| Tricolored bat | p < 0.0001 | 0.03 (p < 0.0001) | n.s. | n.s. | n.s. | n.s. | n.s. |
| Brazilian free-tailed bat | p < 0.0001 | 0.004 (p < 0.0001) | n.s. | n.s. | n.s. | n.s. | 0.10 (p = 0.01) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andersen, B.R.; McGuire, L.P.; Wigley, T.B.; Miller, D.A.; Stevens, R.D. Habitat Associations of Overwintering Bats in Managed Pine Forest Landscapes. Forests 2022, 13, 803. https://doi.org/10.3390/f13050803
Andersen BR, McGuire LP, Wigley TB, Miller DA, Stevens RD. Habitat Associations of Overwintering Bats in Managed Pine Forest Landscapes. Forests. 2022; 13(5):803. https://doi.org/10.3390/f13050803
Chicago/Turabian StyleAndersen, Brett R., Liam P. McGuire, Thomas Bently Wigley, Darren A. Miller, and Richard D. Stevens. 2022. "Habitat Associations of Overwintering Bats in Managed Pine Forest Landscapes" Forests 13, no. 5: 803. https://doi.org/10.3390/f13050803
APA StyleAndersen, B. R., McGuire, L. P., Wigley, T. B., Miller, D. A., & Stevens, R. D. (2022). Habitat Associations of Overwintering Bats in Managed Pine Forest Landscapes. Forests, 13(5), 803. https://doi.org/10.3390/f13050803






