Re-Assembly of the Longleaf Pine Ecosystem: Effects of Groundcover Seeding on Understory Community, Fire Behavior and Soil Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site and Experimental Design
2.2. Data Collection
2.2.1. Stand Structure
2.2.2. Groundcover Sampling
2.2.3. Fire Behavior
2.2.4. Soil Nutrients and Isotopic Ratios
2.3. Statistical Analysis
2.3.1. Understory Characteristics
2.3.2. Fire Behavior
2.3.3. Soil Nutrients and Isotopic Ratios
3. Results
3.1. Stand Structure
3.2. Understory Characteristics
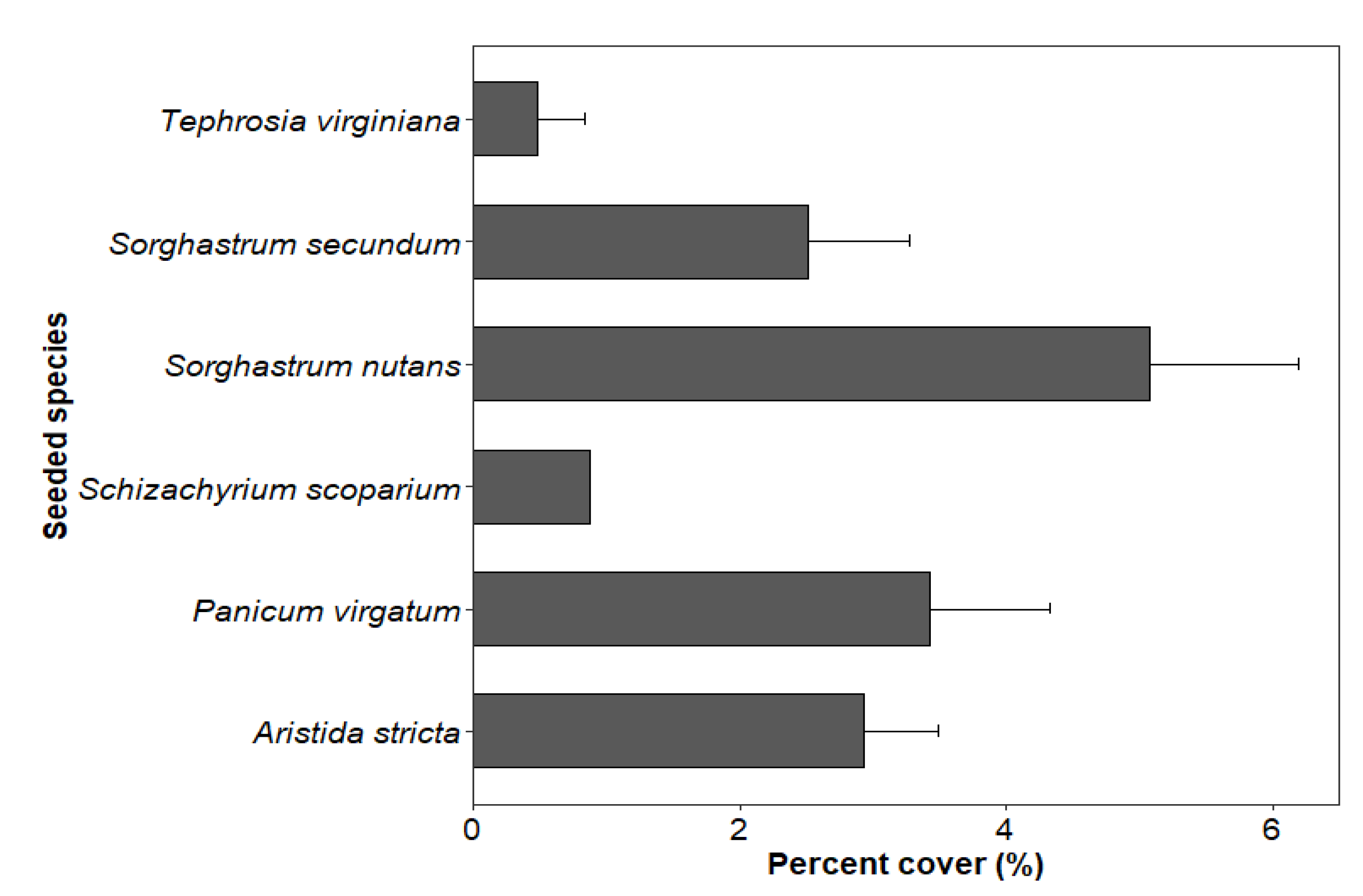
3.2.1. Status of Seeded Species
3.2.2. Plant Species Richness and Diversity
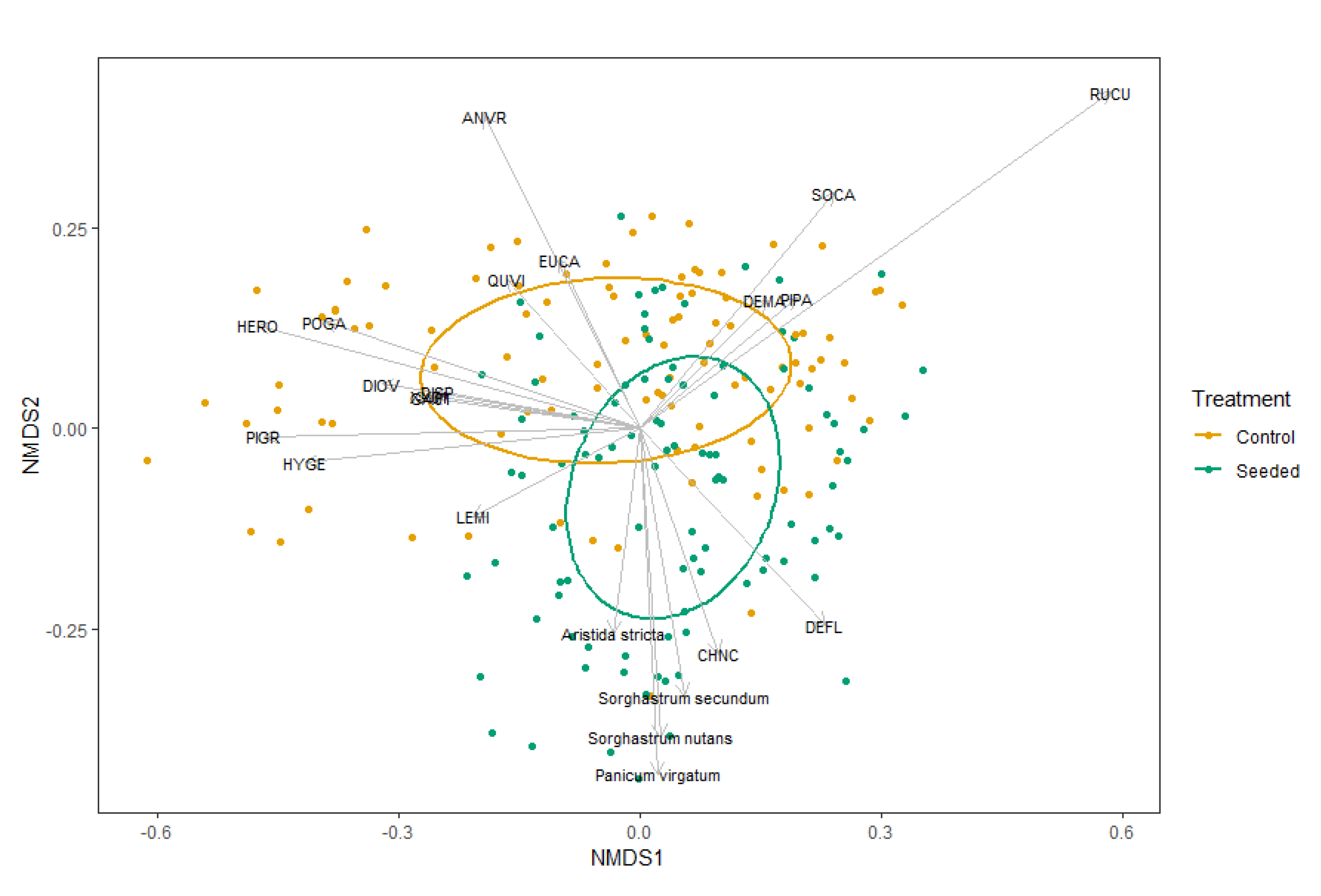
3.2.3. Community Species Composition
3.3. Fire Behavior
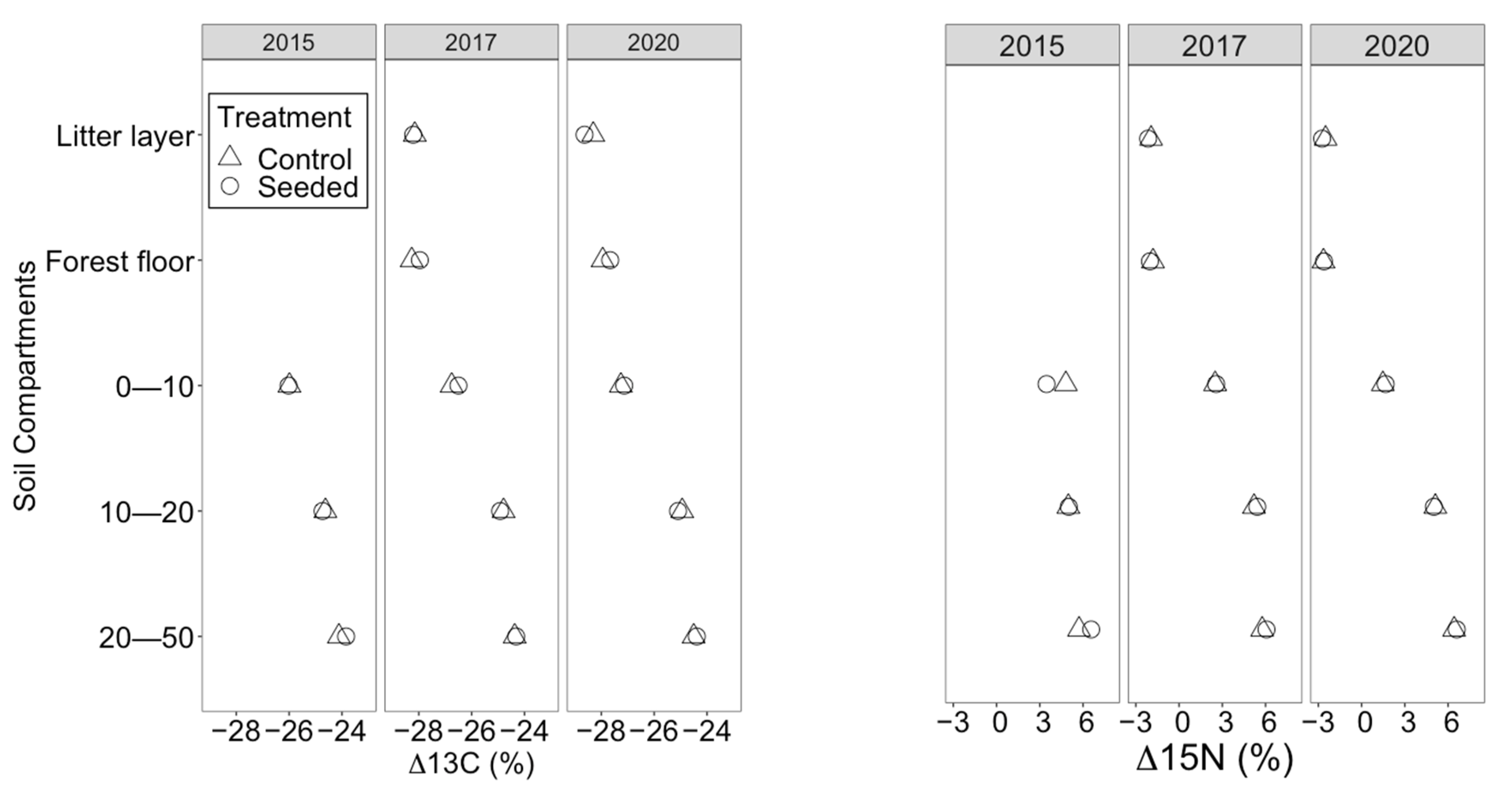
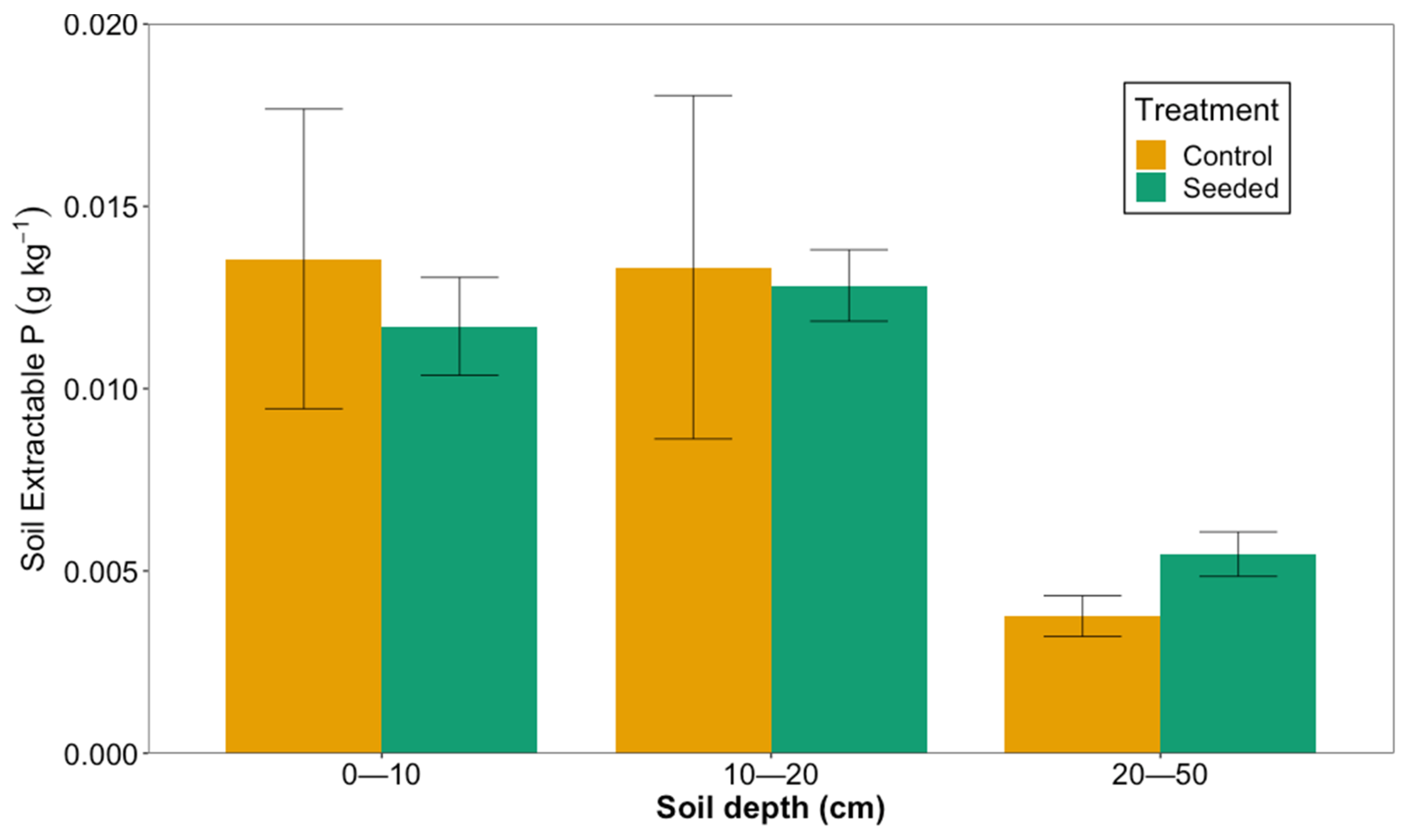
3.4. Nutrients and Isotopic Signatures Analysis
Phosphorus Analysis
4. Discussion
4.1. Understory Characteristics
4.2. Fire Behavior
4.3. Soil Nutrients and Isotopic Ratios
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Frost, C.C. Four centuries of changing landscape patterns in the longleaf pine ecosystem. In Proceedings of the Tall Timbers Fire Ecology Conference; Tall Timbers Research Station: Tallahassee, FL, USA, 1993; Volume 18, pp. 17–43. [Google Scholar]
- Noss, R.F. Longleaf pine and wiregrass: Keystone components of an endangered ecosystem. Nat. Areas J. 1989, 9, 211–213. [Google Scholar]
- Peet, R.K.; Allard, D.J. Longleaf pine vegetation of the Southern Atlantic and Eastern Gulf Coast Regions: A preliminary classification. In Proceedings of the Tall Timbers Fire Ecology Conference; Tall Timbers Research Station: Tallahassee, FL, USA, 1993; Volume 18, pp. 45–81. [Google Scholar]
- Walker, J.L.; Silletti, A.M. Restoring the ground layer of longleaf pine ecosystems. In The Longleaf Pine Ecosystem; Springer: Berlin/Heidelberg, Germany, 2007; pp. 297–333. [Google Scholar]
- Sharma, A.; Bohn, K.K.; Jose, S.; Miller, D.L. Seed Bank—Vegetation dynamics along a restoration management gradient in pine flatwoods ecosystems of the Florida Gulf Coast. Nat. Areas J. 2018, 38, 26–43. [Google Scholar] [CrossRef]
- Means, D.B. Vertebrate faunal diversity of longleaf pine ecosystems. In The Longleaf Pine Ecosystem; Springer: Berlin/Heidelberg, Germany, 2007; pp. 157–213. [Google Scholar]
- Mitchell, R.J.; Hiers, J.K.; O’Brien, J.; Starr, G. Ecological forestry in the Southeast: Understanding the ecology of fuels. J. For. 2009, 107, 391–397. [Google Scholar]
- Ellair, D.P.; Platt, W.J. Fuel composition influences fire characteristics and understorey hardwoods in pine savanna. J. Ecol. 2013, 101, 192–201. [Google Scholar] [CrossRef]
- Dell, J.E.; Richards, L.A.; O’Brien, J.J.; Loudermilk, E.L.; Hudak, A.T.; Pokswinski, S.M.; Bright, B.C.; Hiers, J.K.; Williams, B.W.; Dyer, L.A. Overstory-derived surface fuels mediate plant species diversity in frequently burned longleaf pine forests. Ecosphere 2017, 8, e01964. [Google Scholar] [CrossRef]
- Kirkman, L.K.; Giencke, L.M. Restoring and managing a diverse ground cover. In Ecological Restoration and Management of Longleaf Pine Forests; CRC Press: Boca Raton, FL, USA, 2017; pp. 207–232. [Google Scholar]
- Wragg, P.D.; Mielke, T.; Tilman, D. Forbs, grasses, and grassland fire behaviour. J. Ecol. 2018, 106, 1983–2001. [Google Scholar] [CrossRef]
- Jose, S.; Jokela, E.J.; Miller, D.L. The Longleaf pine ecosystem: Ecology, silviculture, and restoration. Choice Rev. Online 2006, 44, 1519–1544. [Google Scholar] [CrossRef]
- Kirkman, L.K.; Jack, S.B. Ecological Restoration and Management of Longleaf Pine Forests; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Sharma, A.; Cory, B.; McKeithen, J.; Frazier, J. Structural diversity of the longleaf pine ecosystem. For. Ecol. Manag. 2020, 462, 117987. [Google Scholar] [CrossRef]
- Brudvig, L.A.; Grman, E.; Habeck, C.W.; Orrock, J.L.; Ledvina, J.A. Strong legacy of agricultural land use on soils and understory plant communities in longleaf pine woodlands. For. Ecol. Manag. 2013, 310, 944–955. [Google Scholar] [CrossRef]
- Ramsey, C.L.; Jose, S.; Brecke, B.J.; Merritt, S. Growth response of longleaf pine (Pinus palustris Mill.) seedlings to fertilization and herbaceous weed control in an old field in southern USA. For. Ecol. Manag. 2003, 172, 281–289. [Google Scholar] [CrossRef]
- Brockway, D.G. Restoration of Longleaf Pine Ecosystems; USDA Forest Service, Southern Research Station: Asheville, NC, USA, 2005; Volume 83. [Google Scholar]
- Sharma, A. Role of Uneven-Aged Silviculture and the Soil Seed Bank in Restoration of Longleaf Pine-Slash Pine (Pinus Palustris-Pinus Elliottii) Ecosystems. Ph.D. Thesis, University of Florida, Gainesville, FL, USA, 2012. [Google Scholar]
- Sharma, A.; Bohn, K.K.; McKeithen, J.; Singh, A. Effects of conversion harvests on light regimes in a southern pine ecosystem in transition from intensively managed plantations to uneven-aged stands. For. Ecol. Manag. 2019, 432, 140–149. [Google Scholar] [CrossRef]
- Kilgo, J.; Blake, J. Ecology and Management of a Forested Landscape: Fifty Years on the Savannah River Site; USDA Forest Service: Savannah River, New Ellenton, SC, USA, 2005. [Google Scholar]
- Cox, A.C.; Gordon, D.R.; Slapcinsky, J.L.; Seamon, G.S. Understory restoration in longleaf pine sandhills. Nat. Areas J. 2004, 24, 4–14. [Google Scholar]
- Vaughan, E. The Apalachicola Bluffs and Ravines Preserve in Northern Florida: A Longleaf Pine and Wiregrass Restoration Project; University of Minnesota: Minneapolis, MN, USA, 2001. [Google Scholar]
- Hattenbach, M.; Gordon, D.; Seamon, G.; Studenmund, R. Development of directseeding techniques to restore native groundcover in a sandhill ecosystem. In Proceedings of the Longleaf Pine Ecosystem Restoration Symposium: Ecological Restoration and Regional Conservation Strategies, Fort Lauderdale, FL, USA, 12–15 November 1998; pp. 64–70, Longleaf Alliance Report. [Google Scholar]
- U.S. Department of Agriculture Natural Resources Conservation Service [USDA NRCS]. 2017; Longleaf Pine Initiative: Conservation beyond Boundaries. Available online: https://www.nrcs.usda.gov/wps/portal/nrcs/detailfull/national/home/?cid=nrcsdev11_023913 (accessed on 13 April 2017).
- Guo, N.; Degen, A.A.; Deng, B.; Shi, F.; Bai, Y.; Zhang, T.; Long, R.; Shang, Z. Changes in vegetation parameters and soil nutrients along degradation and recovery successions on alpine grasslands of the Tibetan plateau. Agric. Ecosyst. Environ. 2019, 284, 106593. [Google Scholar] [CrossRef]
- McGroddy, M.E.; Daufresne, T.; Hedin, L.O. Scaling of C: N: P stoichiometry in forests worldwide: Implications of terrestrial redfield-type ratios. Ecology 2004, 85, 2390–2401. [Google Scholar] [CrossRef]
- Still, C.J.; Berry, J.A.; Collatz, G.J.; DeFries, R.S. Global distribution of C3 and C4 vegetation: Carbon cycle implications. Glob. Biogeochem. Cycles 2003, 17, 6-1–6-14. [Google Scholar] [CrossRef]
- Basu, S.; Agrawal, S.; Sanyal, P.; Mahato, P.; Kumar, S.; Sarkar, A. Carbon isotopic ratios of modern C3–C4 plants from the Gangetic Plain, India and its implications to paleovegetational reconstruction. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2015, 440, 22–32. [Google Scholar] [CrossRef]
- Boutton, T.W. Stable carbon isotope ratios of organic matter and their use as indicators of vegetation and climate changes. In Mass Spectrometry of Soils; Marcel Dekker: New York, NY, USA, 1996; pp. 47–82. [Google Scholar]
- Balesdent, J.; Girardin, C.; Mariotti, A. Site-Related^(13) C of Tree Leaves and Soil Organic Matter in a Temperate Forest. Ecology 1993, 74, 1713–1721. [Google Scholar] [CrossRef]
- Wedin, D.A. C4 grasses: Resource use, ecology, and global change. Warm-Seas. (C4) Grasses 2004, 45, 15–50. [Google Scholar]
- Wedin, D.A. Species, nitrogen, and grassland dynamics: The constraints of stuff. In Linking Species & Ecosystems; Springer: Berlin/Heidelberg, Germany, 1995; pp. 253–262. [Google Scholar]
- Seastedt, T. Mass, nitrogen, and phosphorus dynamics in foliage and root detritus of tallgrass prairie. Ecology 1988, 69, 59–65. [Google Scholar] [CrossRef]
- Loudermilk, E.L.; Achtemeier, G.L.; O’Brien, J.J.; Hiers, J.K.; Hornsby, B.S. High-resolution observations of combustion in heterogeneous surface fuels. Int. J. Wildland Fire 2014, 23, 1016–1026. [Google Scholar] [CrossRef]
- Fornara, D.A.; Tilman, D. Plant functional composition influences rates of soil carbon and nitrogen accumulation. J. Ecol. 2008, 96, 314–322. [Google Scholar] [CrossRef]
- Pau, S.; Edwards, E.J.; Still, C.J. Improving our understanding of environmental controls on the distribution of C3 and C4 grasses. Glob. Chang. Biol. 2013, 19, 184–196. [Google Scholar] [CrossRef] [PubMed]
- Ament, M.R.; Tierney, J.A.; Hedin, L.O.; Hobbie, E.A.; Wurzburger, N. Phosphorus and species regulate N2 fixation by herbaceous legumes in longleaf pine savannas. Oecologia 2018, 187, 281–290. [Google Scholar] [CrossRef]
- Markewitz, D.; Sartori, F.; Craft, C. Soil change and carbon storage in longleaf pine stands planted on marginal agricultural lands. Ecol. Appl. 2002, 12, 1276–1285. [Google Scholar] [CrossRef]
- Butnor, J.R.; Johnsen, K.H.; Maier, C.A.; Nelson, C.D. Intra-annual variation in soil C, N and nutrients pools after prescribed fire in a mississippi longleaf pine (Pinus palustris Mill.) plantation. Forests 2020, 11, 181. [Google Scholar] [CrossRef]
- Ficken, C.D.; Wright, J.P. Effects of fire frequency on litter decomposition as mediated by changes to litter chemistry and soil environmental conditions. PLoS ONE 2017, 12, e0186292. [Google Scholar] [CrossRef] [PubMed]
- Carter, M.C.; Foster, C.D. Prescribed burning and productivity in southern pine forests: A review. For. Ecol. Manag. 2004, 191, 93–109. [Google Scholar] [CrossRef]
- Hendricks, J.J.; Wilson, C.A.; Boring, L.R. Foliar litter position and decomposition in a fire-maintained longleaf pine-wiregrass ecosystem. Can. J. For. Res. 2002, 32, 928–941. [Google Scholar] [CrossRef]
- Pritchett, W.; Smith, W. Forest fertilization in the US southeast. For. Soils For. Land Manag. 1975, 467–476. [Google Scholar]
- Ledvina, J.; McShea, W.J.; Bourg, N.A.; Herrmann, V.; Akre, T.; Johnson, A.E. Management Regime and Field Age Affect Species Richness and Cover of Native Forbs and Exotic Species in Virginia Grasslands. Ecol. Restor. 2020, 38, 83–93. [Google Scholar] [CrossRef]
- Chapin, F.S., III. The mineral nutrition of wild plants. Annu. Rev. Ecol. Syst. 1980, 11, 233–260. [Google Scholar] [CrossRef]
- Goebel, P.C.; Palik, B.J.; Kirkman, L.K.; Drew, M.B.; West, L.; Pederson, D.C. Forest ecosystems of a Lower Gulf Coastal Plain landscape: Multifactor classification and analysis. J. Torrey Bot. Soc. 2001, 128, 47–75. [Google Scholar] [CrossRef]
- Keys, J.E.; Carpenter, C.A.; Hooks, S.L.; Koenig, F.; McNab, W.H.; Russell, W.; Smith, M.-L. Ecological Units of the Eastern United States: First Approximation; US Department of Agriculture, Forest Service Atlanta: Atlanta, GA, USA, 1995.
- Johnson, R.; Gjerstad, D. Restoring the overstory of longleaf pine ecosystems. In The Longleaf Pine Ecosystem; Springer: Berlin/Heidelberg, Germany, 2007; pp. 271–295. [Google Scholar]
- Hainds, M.J. Establishing longleaf seedlingson agricultural fields and pastures. In Proceedings of the 4th Biennial Regional Longleaf Alliance Conference, Southern Pines, NC, USA, 17–20 November 2002; pp. 69–73. [Google Scholar]
- Battaglia, M.A.; Mou, P.; Palik, B.; Mitchell, R.J. The effect of spatially variable overstory on the understory light environment of an open-canopied longleaf pine forest. Can. J. For. Res. 2002, 32, 1984–1991. [Google Scholar] [CrossRef]
- Frazer, G.W.; Canham, C.D.; Lertzman, K.P. Gap Light Analyzer (GLA), Version 2.0: Imaging Software to Extract Canopy Structure and Gap Light Transmission Indices from True-Colour Fisheye Photographs, Users Manual and Program Documentation; Simon Fraser University: Burnaby, BC, Canada; The Institute of Ecosystem Studies: Millbrook, NY, USA, 1999; p. 36. [Google Scholar]
- Braun-Blanquet, J. PflanzensoziologiePlant Sociology. The Study of Plant Communities; McGraw-Hill Book Company, Inc.: New York, NY, USA; London, UK, 1932. [Google Scholar]
- Bigelow, S.W.; Whelan, A.W. Longleaf pine proximity effects on air temperatures and hardwood top-kill from prescribed fire. Fire Ecol. 2019, 15, 15. [Google Scholar] [CrossRef]
- Butler, B.; Teske, C.; Jimenez, D.; O’Brien, J.; Sopko, P.; Wold, C.; Vosburgh, M.; Hornsby, B.; Loudermilk, E. Observations of energy transport and rate of spreads from low-intensity fires in longleaf pine habitat–RxCADRE 2012. Int. J. Wildland Fire 2015, 25, 76–89. [Google Scholar] [CrossRef]
- Simard, A.J.; Eenigenburg, J.E.; Adams, K.B.; Nissen Jr, R.L.; Deacon, A.G. A general procedure for sampling and analyzing wildland fire spread. For. Sci. 1984, 30, 51–64. [Google Scholar]
- Wade, D.D. A Guide for Prescribed Fire in Southern Forests; US Department of Agriculture, Forest Service, Southern Region: Atlanta, GA, USA, 1989.
- Kuo, S.; Sparks, D.; Page, A.; Helmke, P.; Loeppert, R. Phosphorus. Methods of Soil Analysis. Part 3. Chemical Methods. Madison; Soil Science Society of America, Inc.: Madison, WI, USA; American Society of Agronomy, Inc.: Madison, WI, USA, 1996. [Google Scholar]
- Magurran, A.E. Ecological Diversity and Its Measurement; Princeton University Press: Princeton, NJ, USA, 1988. [Google Scholar]
- Bates, D.M.; Maechler, M.; Bolker, B. Package ‘lme4′ (Version 0.999999-0): Linear Mixed-Effects Models Using S4 Classes. Available online: cran.r-project.org/web/packages/lme4/lme4.pdf (accessed on 10 March 2022).
- Montgomery, D. Design and Analysis of Experiments, 2nd ed.; John Wiley and Sons: New York, NY, USA, 1984. [Google Scholar]
- Cáceres, M.D.; Legendre, P. Associations between species and groups of sites: Indices and statistical inference. Ecology 2009, 90, 3566–3574. [Google Scholar] [CrossRef]
- Phillips, D.L.; Gregg, J.W. Uncertainty in source partitioning using stable isotopes. Oecologia 2001, 127, 171–179. [Google Scholar] [CrossRef]
- Fox, J.; Weisberg, S. An R Companion to Applied Regression; Sage publications: Los Angeles, CA, USA, 2018. [Google Scholar]
- Van Lear, D.H.; Carroll, W.D.; Kapeluck, P.R.; Johnson, R. History and restoration of the longleaf pine-grassland ecosystem: Implications for species at risk. For. Ecol. Manag. 2005, 211, 150–165. [Google Scholar] [CrossRef]
- Brudvig, L.A.; Damschen, E.I. Land-use history, historical connectivity, and land management interact to determine longleaf pine woodland understory richness and composition. Ecography 2011, 34, 257–266. [Google Scholar] [CrossRef]
- Peterson, D.W.; Reich, P.B.; Wrage, K.J. Plant functional group responses to fire frequency and tree canopy cover gradients in oak savannas and woodlands. J. Veg. Sci. 2007, 18, 3–12. [Google Scholar] [CrossRef]
- Farrar, R.M. Thinning longleaf pine on average sites. J. For. 1968, 66, 906–909. [Google Scholar]
- Kirkman, L.K.; Coffey, K.L.; Mitchell, R.J.; Moser, E.B. Ground cover recovery patterns and life-history traits: Implications for restoration obstacles and opportunities in a species-rich savanna. J. Ecol. 2004, 92, 409–421. [Google Scholar] [CrossRef]
- Brockway, D.G.; Lewis, C.E. Long-term effects of dormant-season prescribed fire on plant community diversity, structure and productivity in a longleaf pine wiregrass ecosystem. Forest Ecology and Management. For. Ecol. Manag. 1997, 96, 167–183. [Google Scholar] [CrossRef]
- Hedman, C.W.; Grace, S.L.; King, S.E. Vegetation composition and structure of southern coastal plain pine forests: An ecological comparison. For. Ecol. Manag. 2000, 134, 233–247. [Google Scholar] [CrossRef]
- Aschenbach, T.A.; Foster, B.L.; Imm, D.W. The Initial Phase of a Longleaf Pine-Wiregrass Savanna Restoration: Species Establishment and Community Responses. Restor. Ecol. 2010, 18, 762–771. [Google Scholar] [CrossRef]
- Reed, H.E.; Seastedt, T.R.; Blair, J.M. Ecological consequences of c 4 grass invasion of a c 4 grassland: A dilemma for management. Ecol. Appl. 2005, 15, 1560–1569. [Google Scholar] [CrossRef]
- Gagnon, P.; Harms, K.; Platt, W.; Passmore, H.; Myers, J. Small-Scale Variation in Fuel Loads Differentially Affects Two Co-Dominant Bunchgrasses. PLoS ONE 2012, 7, e29674. [Google Scholar] [CrossRef]
- Simpson, K.J.; Ripley, B.S.; Christin, P.A.; Belcher, C.M.; Lehmann, C.E.; Thomas, G.H.; Osborne, C.P. Determinants of flammability in savanna grass species. J. Ecol. 2016, 104, 138–148. [Google Scholar] [CrossRef]
- Ripley, B.; Visser, V.; Christin, P.-A.; Archibald, S.; Martin, T.; Osborne, C. Fire ecology of C3 and C4 grasses depends on evolutionary history and frequency of burning but not photosynthetic type. Ecology 2015, 96, 2679–2691. [Google Scholar] [CrossRef] [PubMed]
- Tilman, D.; Wedin, D.; Knops, J. Productivity and sustainability influenced by biodiversity in grassland ecosystems. Nature 1996, 379, 718–720. [Google Scholar] [CrossRef]
- Viera-Vargas, M.S.; Souto, C.M.; Urquiaga, S.; Boddey, R.M. Quantification of the contribution of N2 fixation to tropical forage legumes and transfer to associated grass. Soil Biol. Biochem. 1995, 27, 1193–1200. [Google Scholar] [CrossRef]
- McLauchlan, K. The nature and longevity of agricultural impacts on soil carbon and nutrients: A review. Ecosystems 2006, 9, 1364–1382. [Google Scholar] [CrossRef]
- Knoepp, J.D.; Taylor, S.R.; Boring, L.R.; Miniat, C.F. Influence of forest disturbance on stable nitrogen isotope ratios in soil and vegetation profiles. Soil Sci. Soc. Am. J. 2015, 2015, 1470–1481. [Google Scholar] [CrossRef]
- Kitayama, K.; Iwamoto, K. Patterns of natural 15 N abundance in the leaf-to-soil continuum of tropical rain forests differing in N availability on Mount Kinabalu, Borneo. Plant Soil 2001, 229, 203–212. [Google Scholar] [CrossRef]
- Smith2, B.N.; Epsten, S. Two Categories of 1CC/12C Ratios for Higher Plantsl. Plant Physiol. 1971, 47, 380–384. [Google Scholar] [CrossRef]
- Lefroy, R.D.; Blair, G.J.; Strong, W.M. Changes in soil organic matter with cropping as measured by organic carbon fractions and 13C natural isotope abundance. Plant Soil 1993, 155, 399–402. [Google Scholar] [CrossRef]
- Prober, S.M.; Thiele, K.R.; Lunt, I.D. Identifying ecological barriers to restoration in temperate grassy woodlands: Soil changes associated with different degradation states. Aust. J. Bot. 2002, 50, 699–712. [Google Scholar] [CrossRef]
- Prober, S.M.; Lunt, I.D.; Thiele, K.R. Determining reference conditions for management and restoration of temperate grassy woodlands: Relationships among trees, topsoils and understorey flora in little-grazed remnants. Aust. J. Bot. 2002, 50, 687–697. [Google Scholar] [CrossRef]

| Response Variables | Seeded | Control | p-Value |
|---|---|---|---|
| Species Richness (species m−2) | 11.28 (0.29) | 10.34 (0.29) | 0.21 |
| Shannon Diversity Index | 1.74 (0.35) | 1.69 (0.04) | 0.40 |
| Soil C and N | Year | Seeded | Control |
|---|---|---|---|
| Mineral C (g kg−1) | 2015 | 5.35 ± 1.28 | 4.78 ± 0.86 |
| 2017 | 5.62 ± 1.74 | 5.88 ± 1.86 | |
| 2020 | 8.40 ± 3.28 | 6.87 ± 2.47 | |
| Mineral N (g kg−1) | 2015 | 0.24 ± 0.05 | 0.22 ± 0.04 |
| 2017 | 0.25 ± 0.07 | 0.23 ± 0.06 | |
| 2020 | 0.34 ± 0.12 | 0.28 ± 0.10 | |
| Organic C (g kg−1) | 2017 | 458.40 ± 18.04 | 453.28 ± 21.74 |
| 2020 | 474.50 ± 7.86 | 466.16 ± 6.95 | |
| Organic N (g kg−1) | 2017 | 4.09 ± 0.45 | 4.71 ± 1.59 |
| 2020 | 4.51 ± 1.03 | 4.45 ± 1.07 |
| Fixed Effect | δ13C | δ15N | C | N | ||||
|---|---|---|---|---|---|---|---|---|
| df | χ2 | df | χ2 | df | χ2 | df | χ2 | |
| Treatment | 1 | 0 | 1 | 0.07 | 1 | 1.74 | 1 | 3.55 |
| Depth | 2 | 283 *** | 2 | 474 *** | 2 | 154 *** | 2 | 178 *** |
| Year | 2 | 33 *** | 1 | 13.2 *** | 1 | 28.1 *** | 2 | 22.5 *** |
| Fixed Effects | δ13C | δ15N | C | N |
|---|---|---|---|---|
| χ2 | χ2 | χ2 | χ2 | |
| Treatment | 0.001 | 0.11 | 2.69 | 0.02 |
| Layer | 23.03 *** | 0.19 | 36.47 *** | 117.93 *** |
| Year | 0.83 | 77.555 *** | 1503 *** | 59.66 *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baniya, B.; Bigelow, S.W.; Sharma, A.; Taylor, S.; Vogel, J.G.; Brantley, S.T. Re-Assembly of the Longleaf Pine Ecosystem: Effects of Groundcover Seeding on Understory Community, Fire Behavior and Soil Properties. Forests 2022, 13, 519. https://doi.org/10.3390/f13040519
Baniya B, Bigelow SW, Sharma A, Taylor S, Vogel JG, Brantley ST. Re-Assembly of the Longleaf Pine Ecosystem: Effects of Groundcover Seeding on Understory Community, Fire Behavior and Soil Properties. Forests. 2022; 13(4):519. https://doi.org/10.3390/f13040519
Chicago/Turabian StyleBaniya, Benju, Seth W. Bigelow, Ajay Sharma, Scott Taylor, Jason G. Vogel, and Steven T. Brantley. 2022. "Re-Assembly of the Longleaf Pine Ecosystem: Effects of Groundcover Seeding on Understory Community, Fire Behavior and Soil Properties" Forests 13, no. 4: 519. https://doi.org/10.3390/f13040519
APA StyleBaniya, B., Bigelow, S. W., Sharma, A., Taylor, S., Vogel, J. G., & Brantley, S. T. (2022). Re-Assembly of the Longleaf Pine Ecosystem: Effects of Groundcover Seeding on Understory Community, Fire Behavior and Soil Properties. Forests, 13(4), 519. https://doi.org/10.3390/f13040519








