Morphometric Analyses of Leaf Shapes in Four Sympatric Mediterranean Oaks and Hybrids in the Algerian Kabylie Forest
Abstract
1. Introduction
2. Materials and Methods
2.1. Morphometric Analysis
2.2. Data Analysis
3. Results
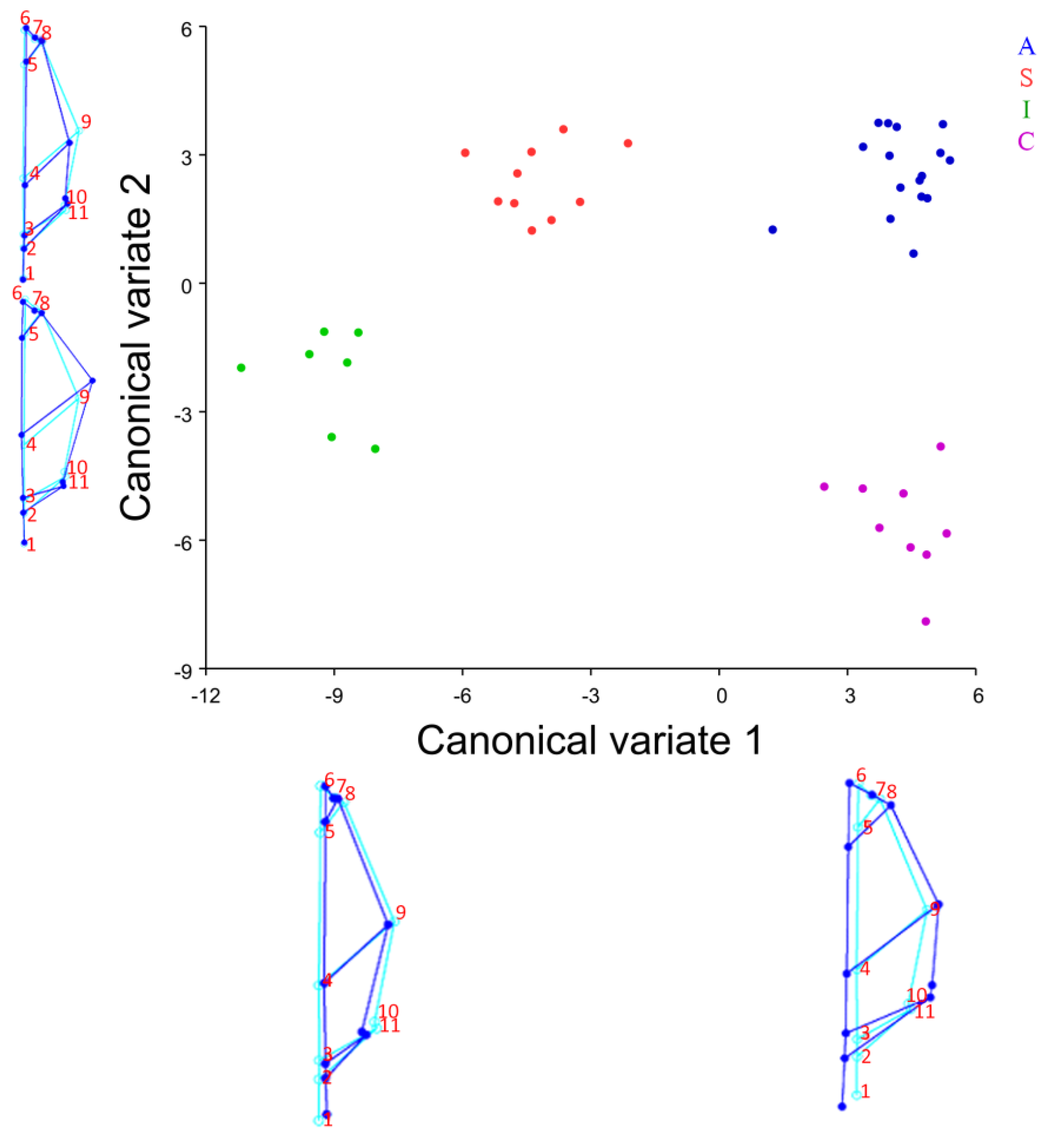
3.1. Morphological Variation in Oaks Species in the Akfadou Forest
3.2. Morphometric Leaf Variation between Species
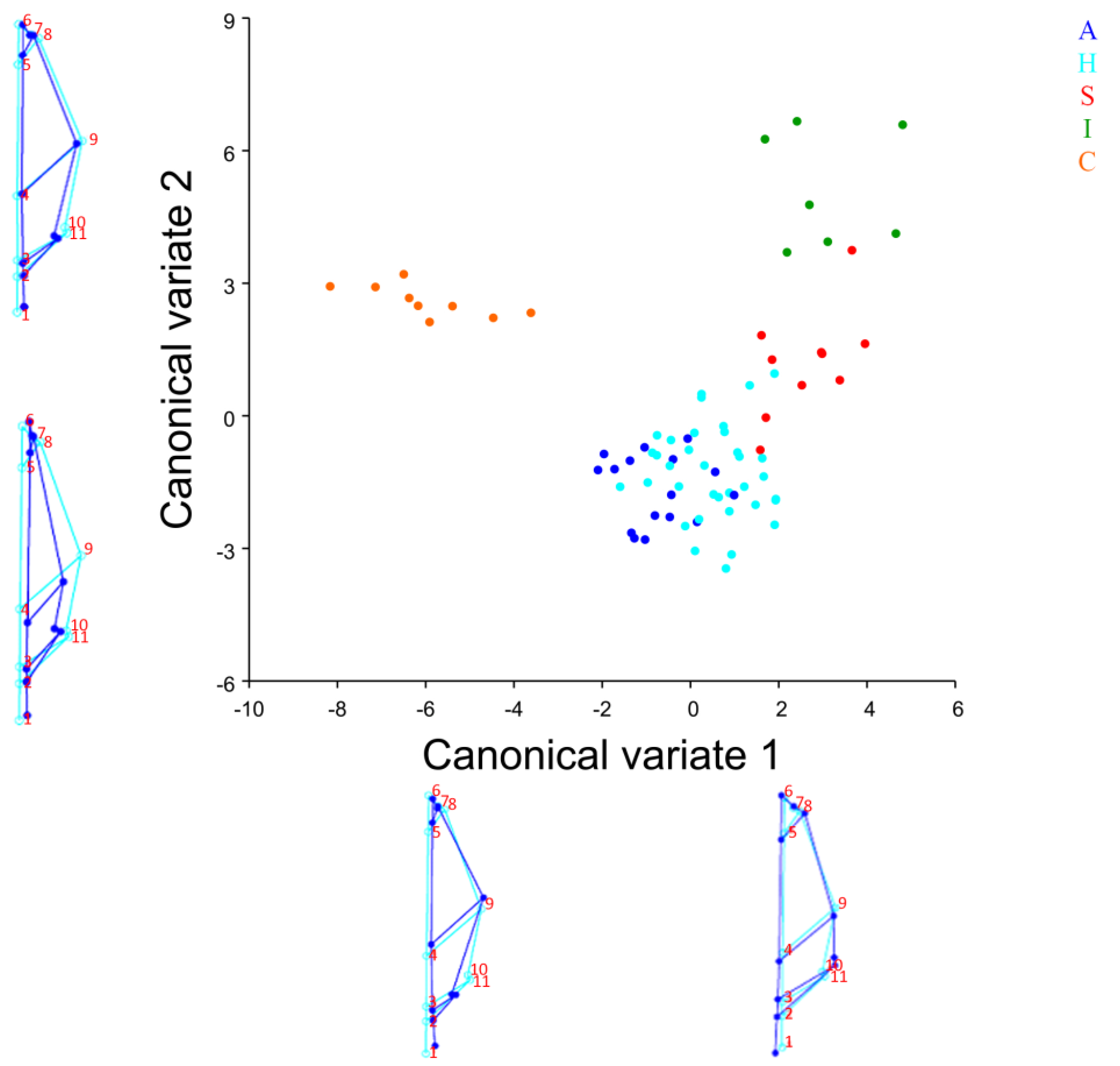
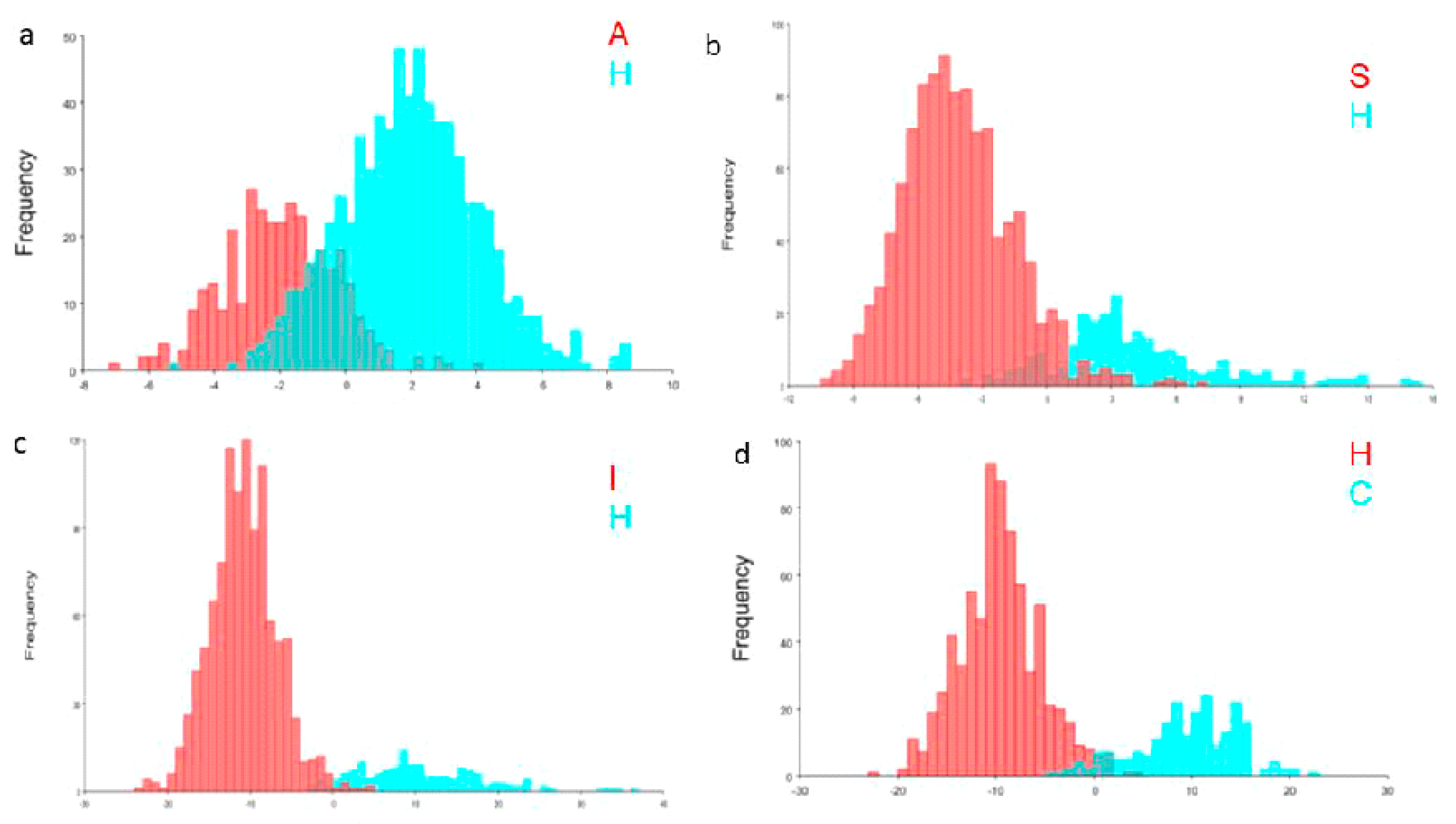
3.3. Morphometric Variations of Hybrids
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abbott, R.J.; Barton, N.H.; Good, J.M. Genomics of hybridization and its evolutionary consequences. Mol. Ecol. 2016, 25, 2325–2332. [Google Scholar] [PubMed]
- Seehausen, O. Hybridization and adaptive radiation. Trends Ecol. Evol. 2004, 19, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Rieseberg, L.H.; Raymond, O.; Rosenthal, D.M.; Lai, Z.; Livingstone, K.; Nakazato, T.; Durphy, J.L.; Schwarzbach, A.E.; Donovan, L.A.; Lexer, C. Major ecological transitions in wild sunflowers facilitated by hybridization. Science 2003, 301, 1211–1216. [Google Scholar] [CrossRef] [PubMed]
- Potts, B.M.; Reid, J.B. Hybridization as a dispersal mechanism. Evolution 1988, 42, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Petit, R.J.; Bodénès, C.; Ducousso, A.; Roussel, G.; Kremer, A. Hybridization as a mechanism of invasion in oaks. New Phytol. 2003, 161, 151–164. [Google Scholar] [CrossRef]
- Potts, B.M.; Dungey, H.S. Interspecific hybridization of Eucalyptus: Key issues for breeders and geneticists. New For. 2004, 27, 115–138. [Google Scholar] [CrossRef]
- Chan, W.Y.; Hoffmann, A.A.; van Oppen, M.J.H. Hybridization as a conservation management tool. Conserv. Lett. 2019, 12, e12652. [Google Scholar]
- Janes, J.K.; Hamilton, J.A. Mixing it up: The role of hybridization in forest management and conservation under climate change. Forests 2017, 8, 237. [Google Scholar] [CrossRef]
- Howard, D.J.; Preszler, R.W.; Williams, J.; Fenchel, S.; Boecklen, W.J. How discrete are oak species? Insights from a hybrid zone between Quercus grisea and Quercus gambelii. Evolution 1997, 51, 747–755. [Google Scholar] [CrossRef]
- Curtu, A.L.; Gailing, O.; Finkeldey, R. Evidence for hybridization and introgression within a species-rich oak (Quercus spp.) community. BMC Evol. Biol. 2007, 7, 218. [Google Scholar]
- Curtu, A.L.; Gailing, O.; Leinemann, L.; Finkeldey, R. Genetic variation and differentiation within a natural community of five oak species (Quercus spp.). Plant Biol. 2007, 9, 116–126. [Google Scholar] [PubMed]
- Antonecchia, G.; Fortini, P.; Lepais, O.; Gerber, S.; Léger, P.; Scippa, G.S.; Viscosi, V. Genetic structure of a natural oak community in central Italy: Evidence of gene flow between three sympatric white oak species (Quercus, Fagaceae). Ann. For. Res. 2015, 58, 205–216. [Google Scholar]
- Petit, R.J.; Pineau, E.; Demesure, B.; Bacilieri, R.; Ducousso, A.; Kremer, A. Chloroplast DNA footprints of postglacial recolonization by oaks. Proc. Natl. Acad. Sci. USA 1997, 94, 9996–10001. [Google Scholar] [PubMed]
- Whittemore, A.T.; Schaal, B.A. Interspecific gene flow in sympatric oaks. Proc. Natl. Acad. Sci. USA 1991, 88, 2540–2544. [Google Scholar] [CrossRef]
- Dumolin-Lapégue, S.; Kremer, A.; Petit, R.J. Are chloroplast and mitochondrial DNA variation species independent in oaks? Evolution 1999, 53, 1406–1413. [Google Scholar] [CrossRef]
- Sofletea, N.; Enescu, C.M.; Curtu, A.L. Small-Scale Morphologial Desriptors Analysis in four Romaninan Oak Stands reported to Series Lanuginosae Simk. Bull. Transilv. Univeristy Brasov. 2011, 4, 77–84. [Google Scholar]
- Burgarella, C.; Lorenzo, Z.; Jabbour-Zahab, R.; Lumaret, R.; Guichoux, E.; Petit, R.J.; Soto, A.; Gil, L. Detection of hybrids in nature: Application to oaks (Quercus suber and Q. ilex). Heredity 2009, 102, 442–452. [Google Scholar]
- Lepais, O.; Petit, R.J.; Guichoux, E.; Lavabre, J.E.; Alberto, F.; Kremer, A.; Gerber, S. Species relative abundance and direction of introgression in oaks. Mol. Ecol. 2009, 18, 2228–2242. [Google Scholar]
- Lepais, O.; Gerber, S. Reproductive patterns shape introgression dynamics and species succession within the European white oak species complex. Evolution 2011, 65, 156–170. [Google Scholar]
- Gugger, P.F.; Fitz-Gibbon, S.T.; Albarrán-Lara, A.; Wright, J.W.; Sork, V.L. Landscape genomics of Quercus lobata reveals genes involved in local climate adaptation at multiple spatial scales. Mol. Ecol. 2021, 30, 406–423. [Google Scholar]
- Curtu, A.L.; Alizoti, P.; Ballian, D. Marginal/peripheral populations of forest tree species and their conservation status: Report for southeastern Europe. Ann. Silvic. Res. 2017, 41, 41–47. [Google Scholar]
- Kremer, A.; Zanetto, A.; Ducousso, A. Multilocus and Multitrait Measures of Differentiation for Gene Markers and Phenotypic Traits. Genetics 1997, 145, 1229–1241. [Google Scholar] [CrossRef] [PubMed]
- Kleinsmit, J.; Bacilieri, R.; Kremer, A.; Roloff, A. Genetic, morphological, ecological and phenological differentiation between Quercus petraea (Matt.) Liebl. and Quercus robur L. in a mixed stand of Northwest of France. Silvae Genet. 1995, 44, 1–10. [Google Scholar]
- Jensen, J.S. Provenance Variation in Phenotypic Traits in Quercus robur and Quercus petraea in Danish Provenance Trials. Scand. J. For. Res. 2000, 15, 297–308. [Google Scholar] [CrossRef]
- Ducousso, A.; Guyon, J.P.; Kremer, A. Latitudinal and altitudinal variation of bud burst in western populations of sessile oak (Quercus petraea (Matt) Liebl). Ann. Sci. For. 1996, 53, 775–782. [Google Scholar] [CrossRef]
- Ramírez-Valiente, J.A.; Santos del Blanco, L.; Alía, R.; Robledo-Arnuncio, J.J.; Climent, J. Adaptation of Mediterranean forest species to climate: Lessons from common garden experiments. J. Ecol. 2021, 1–21. [Google Scholar] [CrossRef]
- Jiménez, P.; de Heredia, U.L.; Collada, C.; Lorenzo, Z.; Gil, L. High variability of chloroplast DNA in three Mediterranean evergreen oaks indicates complex evolutionary history. Heredity 2004, 93, 510–515. [Google Scholar] [CrossRef][Green Version]
- Belahbib, N.; Pemonge, M.-H.H.; Ouassou, A.; Sbay, H.; Kremer, A.; Petit, R.J. Frequent cytoplasmic exchanges between oak species that are not closely related: Quercus suber and Q. ilex in Morocco. Mol. Ecol. 2001, 10, 2003–2012. [Google Scholar] [CrossRef]
- Fortini, P.; Viscosi, V.; Maiuro, L.; Fineschi, S.; Vendramin, G.G. Comparative leaf surface morphology and molecular data of five oaks of Comparative leaf surface morphology and molecular data of five oaks of the subgenus Quercus Oerst (Fagaceae). Plant Biosyst. 2009, 143, 543–554. [Google Scholar] [CrossRef]
- Fortini, P.; Di Marzio, P.; Di Pietro, R. Differentiation and hybridization of Quercus frainetto, Q. petraea, and Q. pubescens (Fagaceae): Insights from macro-morphological leaf traits and molecular data. Plant. Syst. Evol. 2015, 301, 375–385. [Google Scholar] [CrossRef]
- Di Pietro, R.; Conte, A.L.; Di Marzio, P.; Gianguzzi, L.; Spampinato, G.; Caldarella, O.; Fortini, P. A multivariate morphometric analysis of diagnostic traits in southern Italy and Sicily pubescent oaks. Folia Geobot. 2020, 55, 163–183. [Google Scholar] [CrossRef]
- Di Pietro, R.; Marzio, P.D.; Medagli, P.; Misano, G.; Silletti, G.N.; Wagensommer, R.P.; Fortini, P. Evidence from multivariate morphometric study of the Quercus pubescens complex in southeast Italy. Bot. Serbica 2016, 40, 83–100. [Google Scholar]
- Proietti, E.; Filesi, L.; Di Marzio, P.; Di Pietro, R.; Masin, R.; Conte, A.L.; Fortini, P. Morphology, geometric morphometrics, and taxonomy in relict deciduous oaks woods in northern Italy. Rend. Lincei. Sci. Fis. Nat. 2021, 32, 549–564. [Google Scholar] [CrossRef]
- Vazquez, F.M.; Coombes, A.J.; Garcia-Alonso, D.; Marquez, F.; Rodriguez-Meireles, C.; Guerra Barrena, M.J.; Vila-Viçosa, C. Anotaciones a la nomenclatura del género Quercus L., (FAGACEAE) en la Península Ibérica y NW de África. Folia Bot. Extrem. 2018, 12, 5–79. [Google Scholar]
- Kremer, A.; Dupouey, J.L.; Deans, J.D.; Cottrell, J.; Csaikl, U.; Finkeldey, R.; Espinel, S.; Jensen, J.; Kleinschmit, J.; Van Dam, B.; et al. Leaf morphological differentiation between Quercus robur and Quercus petraea is stable across western European mixed oak stands. Ann. For. Sci. 2002, 59, 777–787. [Google Scholar] [CrossRef]
- Gugerli, F.; Walser, J.C.; Dounavi, K.; Holderegger, R.; Finkeldey, R. Coincidence of small-scale spatial discontinuities in leaf morphology and nuclear microsatellite variation of Quercus petraea and Q. robur in a mixed forest. Ann. Bot. 2007, 99, 713–722. [Google Scholar] [CrossRef]
- Ramírez-Valiente, J.A.; Etterson, J.R.; Deacon, N.J.; Cavender-Bares, J. Evolutionary potential varies across populations and traits in the neotropical oak Quercus oleoides. Tree Physiol. 2019, 39, 427–439. [Google Scholar] [CrossRef]
- Enescu, C.M.; Curtu, A.L.; Sofletea, N. Is Quercus virgiliana a distinct morphological and genetic entity among European white oaks? Turk. J. Agric. 2013, 37, 632–641. [Google Scholar] [CrossRef]
- Di Pietro, R.; Conte, A.L.; Di Marzio, P.; Fortini, P.; Farris, E.; Gianguzzi, L.; Müller, M.; Rosati, L.; Spampinato, G.; Gailing, O. Does the genetic diversity among pubescent white oaks in southern Italy, Sicily and Sardinia islands support the current taxonomic classification? Eur. J. For. Res. 2021, 140, 355–371. [Google Scholar] [CrossRef]
- Midolo, G.; Wellstein, C.; Faurby, S. Individual fitness is decoupled from coarse-scale probability of occurrence in North American trees. Ecography 2021, 44, 789–801. [Google Scholar] [CrossRef]
- Wellstein, C.; Spada, F. The status of Quercus pubescens willd. in Europe. In Warm-Temperate Deciduous Forests around the Northern Hemisphere, Geobotany Studies; Box, E., Fujiiwara, K., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 153–163. [Google Scholar]
- Mir, C.; Toumi, L.; Jarne, P.; Sarda, V.; Di Giusto, F.; Lumaret, R. Endemic North African Quercus afares Pomel originates from hybridisation between two genetically very distant oak species (Q. suber L. and Q. canariensis Willd.): Evidence from nuclear and cytoplasmic markers. Heredity 2006, 96, 175–184. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Messaoudène, M.; Laribi, M.; Derridj, A. Étude de la diversité floristique de la forêt de l’Akfadou (Algérie). Bois For. Trop. 2007, 291, 75–81. [Google Scholar]
- Messaoudène, M.; Tessier, L. Relations cerne-climat dans des peuplements de Quercus afares willd et Quercus canariensis Pomel en Algerie. Ann. Sci. For. 1997, 54, 347–358. [Google Scholar] [CrossRef]
- Viscosi, V.; Cardini, A. Leaf morphology, taxonomy and geometric morphometrics: A simplified protocol for beginners. PLoS ONE 2011, 6, e25630. [Google Scholar] [CrossRef]
- Viscosi, V.; Loy, A.; Fortini, P. Geometric Morphometric Analysis as a Tool to Explore Covariation between Shape and Other Quantitative Leaf Traits in European White Oaks. Tools for Identifying Biodiversity: Progress and Problems. In Proceedings of the International Congress, Paris, France, 20–22 September 2010; Nimis, P.M., Vignes Lebbe, R., Eds.; EUT Edizioni Università di Trieste: Trieste, Italy, 2010; pp. 257–261. [Google Scholar]
- Quézel, P.; Santa, S. Nouvelle Flore de l’Algérie et des Régions Désertiques Méridionales, Volume 1; CNRS ed.: Paris, France, 1962; pp. 1–565. [Google Scholar]
- Quézel, P.; Santa, S. Nouvelle Flore de l’Algérie et des Régions Désertiques Méridionales, Volume 2; CNRS ed.: Paris, France, 1963; pp. 566–1170. [Google Scholar]
- Mhamdi, S.; Brendel, O.; Montpied, P.; Ghouil-Amimi, H.; Hasnaoui, I.; Dreyer, E. Leaf morphology displays no detectable spatial organisation in the relict Quercus afares Pomel compared to the co-occurring parental species Q. canariensis Willd. and Q. suber L. Ann. For. Sci. 2013, 70, 675–684. [Google Scholar] [CrossRef][Green Version]
- Liu, Y.; Li, Y.; Song, J.; Zhang, R.; Yan, Y.; Wang, Y.; Du, F.K. Geometric morphometric analyses of leaf shapes in two sympatric Chinese oaks: Quercus dentata Thunberg and Quercus aliena Blume (Fagaceae). Ann. For. Sci. 2018, 75, 90. [Google Scholar] [CrossRef]
- Viscosi, V. Geometric morphometrics and leaf phenotypic plasticity: Assessing fluctuating asymmetry and allometry in European white oaks (Quercus). Bot. J. Linn. Soc. 2015, 179, 335–348. [Google Scholar] [CrossRef]
- Klingenberg, C.P. MorphoJ: An integrated software package for geometric morphometrics. Mol. Ecol. Resour. 2011, 11, 353–357. [Google Scholar] [CrossRef]
- Viscosi, V.; Lepais, O.; Gerber, S.; Fortini, P. Leaf morphological analyses in four European oak species (Quercus) and their hybrids: A comparison of traditional and geometric morphometric methods. Plant Biosyst. 2009, 143, 564–574. [Google Scholar] [CrossRef]
- Alberto, F.J.; Aitken, S.N.; Alía, R.; González-Martínez, S.C.; Hänninen, H.; Kremer, A.; Lefèvre, F.; Lenormand, T.; Yeaman, S.; Whetten, R.; et al. Potential for evolutionary responses to climate change—evidence from tree populations. Glob. Chang. Biol. 2013, 19, 1645–1661. [Google Scholar] [CrossRef]
- Valladares, F.; Matesanz, S.; Guilhaumon, F.; Araújo, M.B.; Balaguer, L.; Benito-Garzón, M.; Cornwell, W.; Gianoli, E.; van Kleunen, M.; Naya, D.E.; et al. The effects of phenotypic plasticity and local adaptation on forecasts of species range shifts under climate change. Ecol. Lett. 2014, 17, 1351–1364. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Valiente, J.A.; Valladares, F.; Delgado, A.; Nicotra, A.B.; Aranda, I. Understanding the importance of intrapopulation functional variability and phenotypic plasticity in Quercus suber. Tree Genet. Genomes 2015, 11, 35. [Google Scholar] [CrossRef]
- Viscosi, V.; Antonecchia, G.; Lepais, O.; Fortini, P.; Gerber, S.; Loy, A. Leaf Shape and Size Differentiation in White Oaks: Assessment of Allometric Relationshops among Three Sympatric Species and Their Hybrids. Int. J. Plant Sci. 2012, 173, 874–875. [Google Scholar] [CrossRef]
- Franjić, J.; Liber, Z.; Škvorc, Ž.; Idžojtić, M.; Šoštarić, R.; Stančić, Z. Morphological and molecular differentiation of the Croatian populations of Quercus pubescens Willd. (Fagaceae). Acta Soc. Bot. Pol. 2006, 75, 123–130. [Google Scholar] [CrossRef]
- Fortini, P.; Antonecchia, G.; Di Marzio, P.; Maiuro, L.; Viscosi, V. Role of micromorphological leaf traits and molecular data in taxonomy of three sympatric white oak species and their hybrids (Quercus L.). Plant Biosyst. 2013, 149, 546–558. [Google Scholar] [CrossRef]
- Dodd, R.S.; Afzal-Rafii, Z. Selection and dispersal in a multispecies oak hybrid zone. Evolution 2004, 58, 261–269. [Google Scholar] [CrossRef]
- Radford, E.; Catullo, G.; de Montmollin, B. Important Plant Areas of the South and East Mediterranean Region: Priority Sites for Conservation; IUCN: Gland, Switzerland; Malaga, Spain, 2011. [Google Scholar]


| Size | Shape | |||||||
|---|---|---|---|---|---|---|---|---|
| Effect | DF | Sum of Squares | F | Percentage of Variation (%) | DF | Sum of Squares | F | Percentage of Variation (%) |
| Plot | 15 | 27,722,600.99 | 19.35 *** | 21.53 | 270 | 6.71 *** | 9.66 | |
| Species | 35 | 60,215,876.60 | 18.02 *** | 46.77 | 630 | 8.13 | 5.02 *** | 22.11 |
| Trees | 81 | 12,391,290.78 | 1.6 ** | 9.63 | 1458 | 7.41 | 1.98 *** | 20.14 |
| Leaves | 293 | 27,979,062.07 | 92.49 *** | 21.73 | 5274 | 13.56 | 19.85 *** | 36.87 |
| Error | 415 | 428,489.35 | 0.33 | 7470 | 0.97 | 2.63 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akli, A.; Lorenzo, Z.; Alía, R.; Rabhi, K.; Torres, E. Morphometric Analyses of Leaf Shapes in Four Sympatric Mediterranean Oaks and Hybrids in the Algerian Kabylie Forest. Forests 2022, 13, 508. https://doi.org/10.3390/f13040508
Akli A, Lorenzo Z, Alía R, Rabhi K, Torres E. Morphometric Analyses of Leaf Shapes in Four Sympatric Mediterranean Oaks and Hybrids in the Algerian Kabylie Forest. Forests. 2022; 13(4):508. https://doi.org/10.3390/f13040508
Chicago/Turabian StyleAkli, Amel, Zaida Lorenzo, Ricardo Alía, Khellaf Rabhi, and Enrique Torres. 2022. "Morphometric Analyses of Leaf Shapes in Four Sympatric Mediterranean Oaks and Hybrids in the Algerian Kabylie Forest" Forests 13, no. 4: 508. https://doi.org/10.3390/f13040508
APA StyleAkli, A., Lorenzo, Z., Alía, R., Rabhi, K., & Torres, E. (2022). Morphometric Analyses of Leaf Shapes in Four Sympatric Mediterranean Oaks and Hybrids in the Algerian Kabylie Forest. Forests, 13(4), 508. https://doi.org/10.3390/f13040508






