Comparative Transcriptome Analysis of Two Populations of Dastarcus helophoroides (Fairmaire) under High Temperature Stress
Abstract
:1. Introduction
2. Materials and Methods
2.1. Insects and Samples
2.2. RNA Extraction and Illumina Sequencing
2.3. Sequence Assembly and Gene Annotation
2.4. Gene Expression Quantification
3. Results
3.1. RNA Sequencing and Assembly
3.2. Gene Functional Annotation
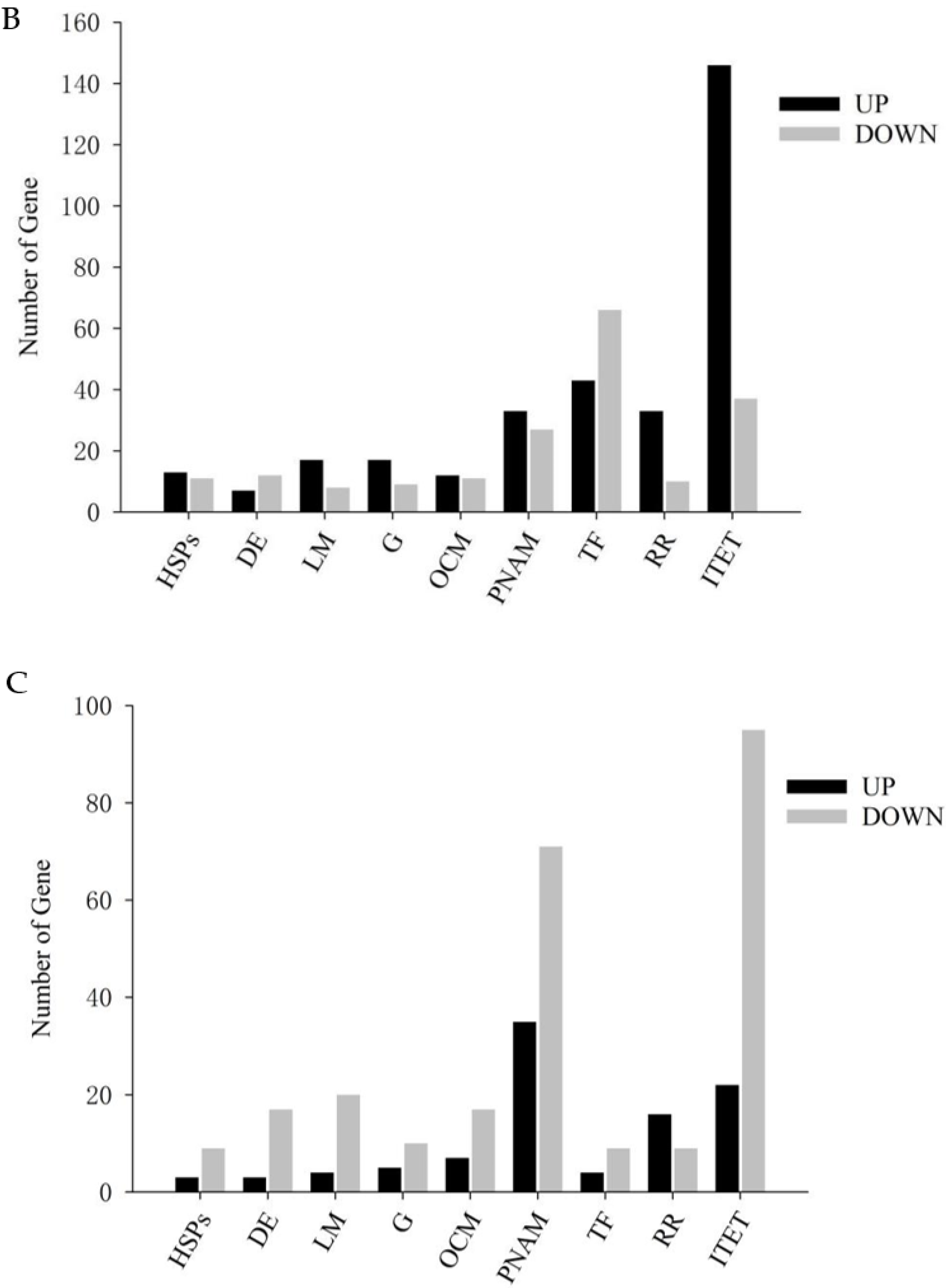
3.3. Quantitative Analysis of Differentially Expressed Genes
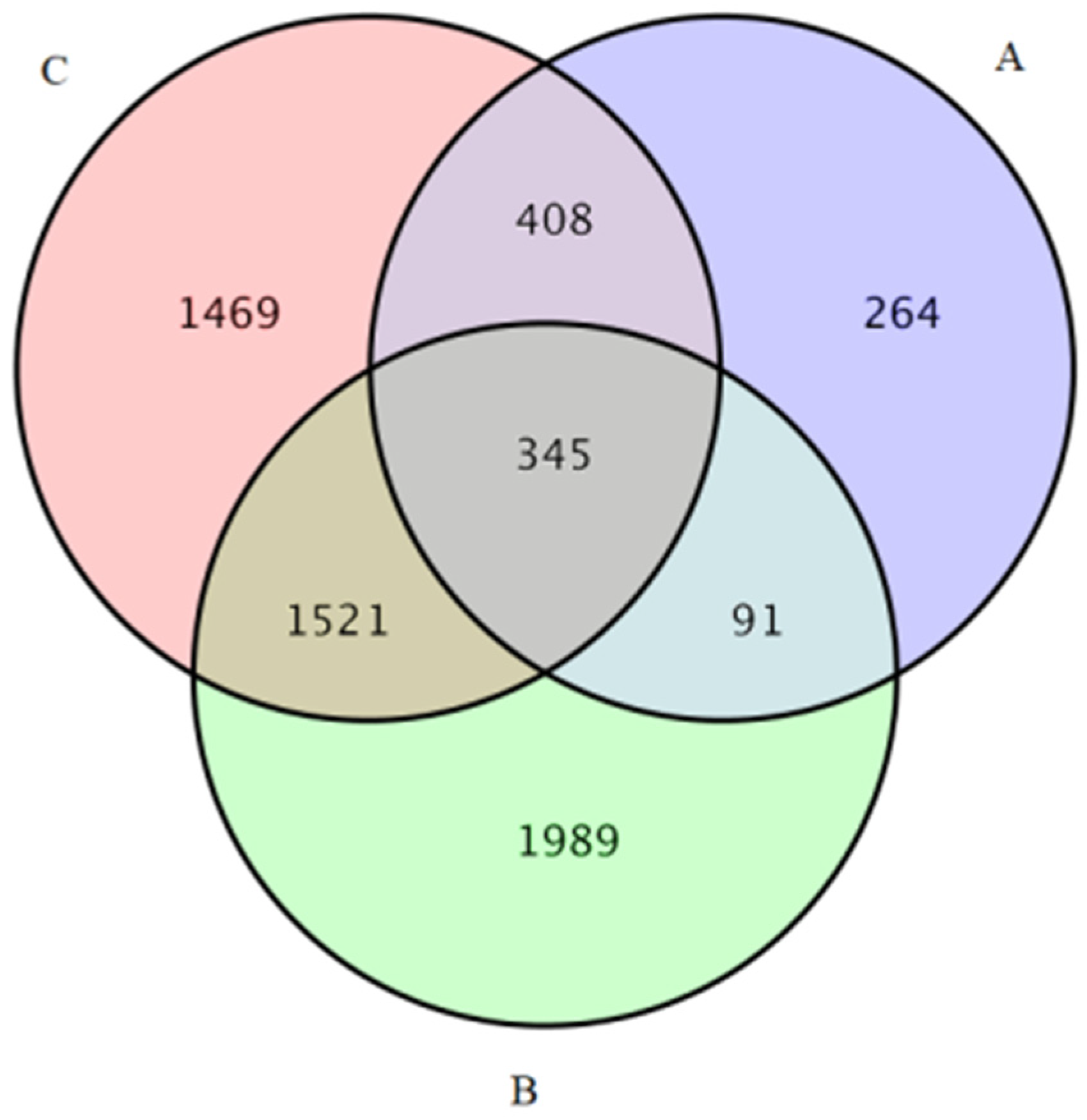
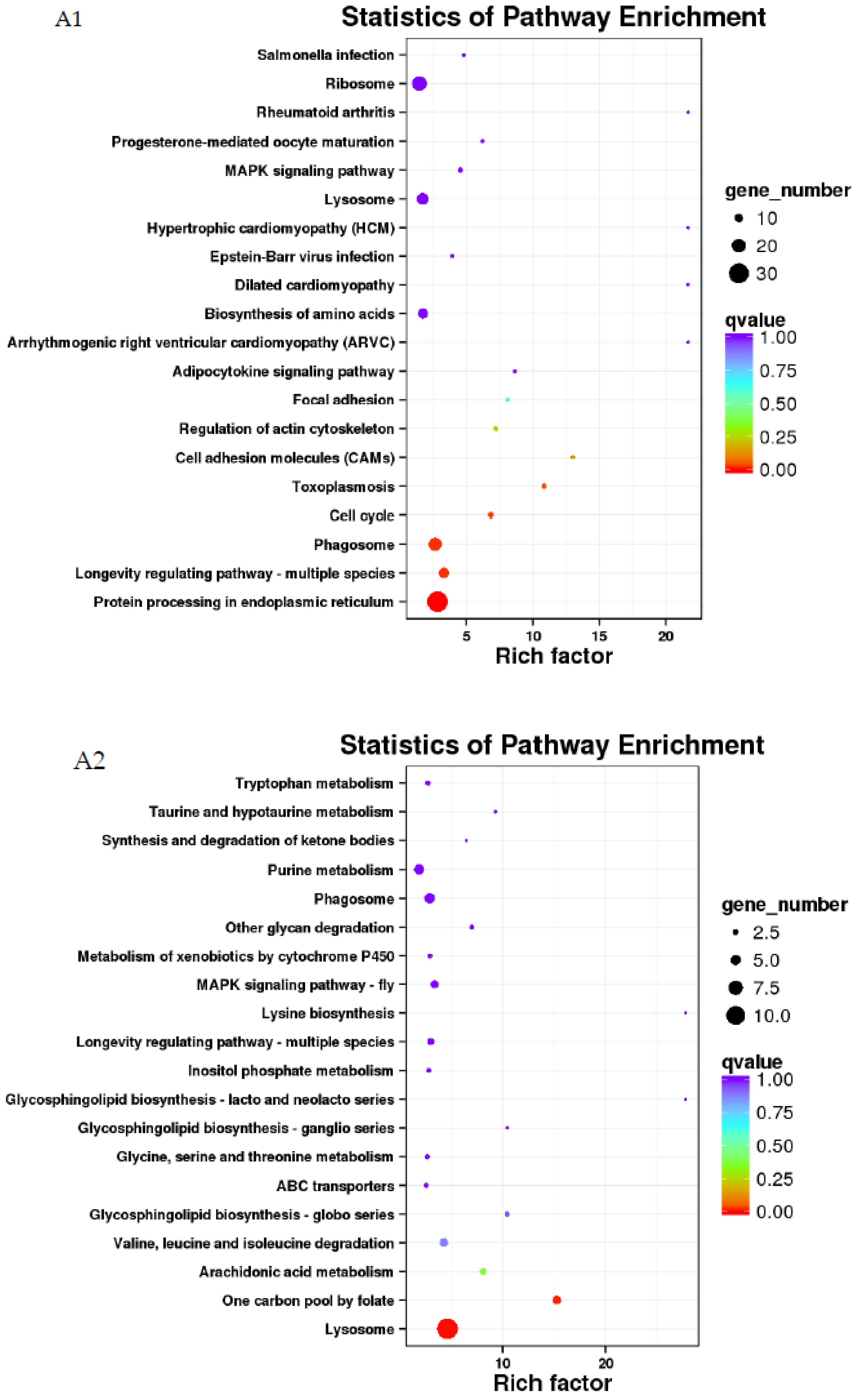
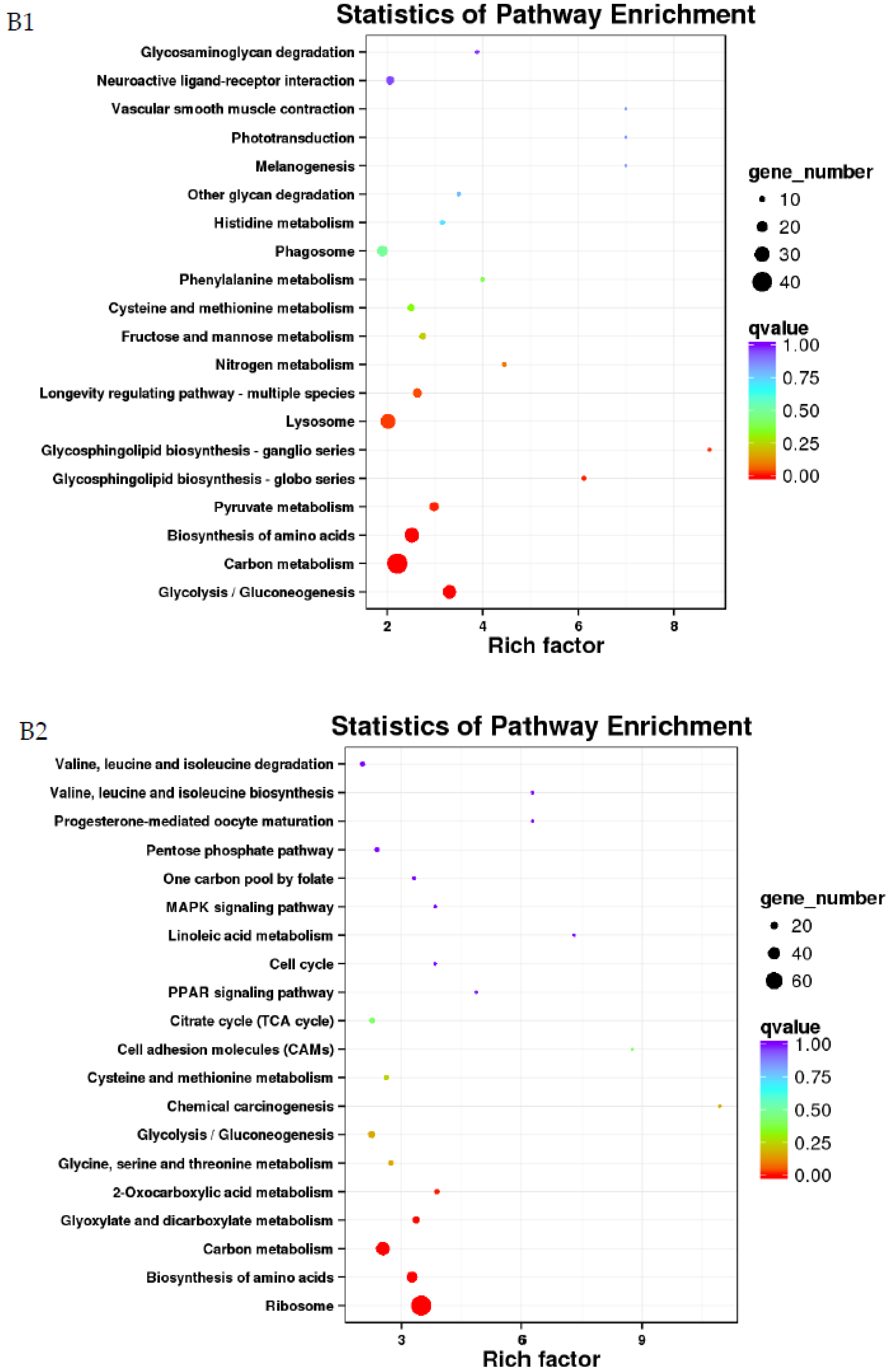
3.4. GO and KEGG Analysis of Differentially Expressed Genes
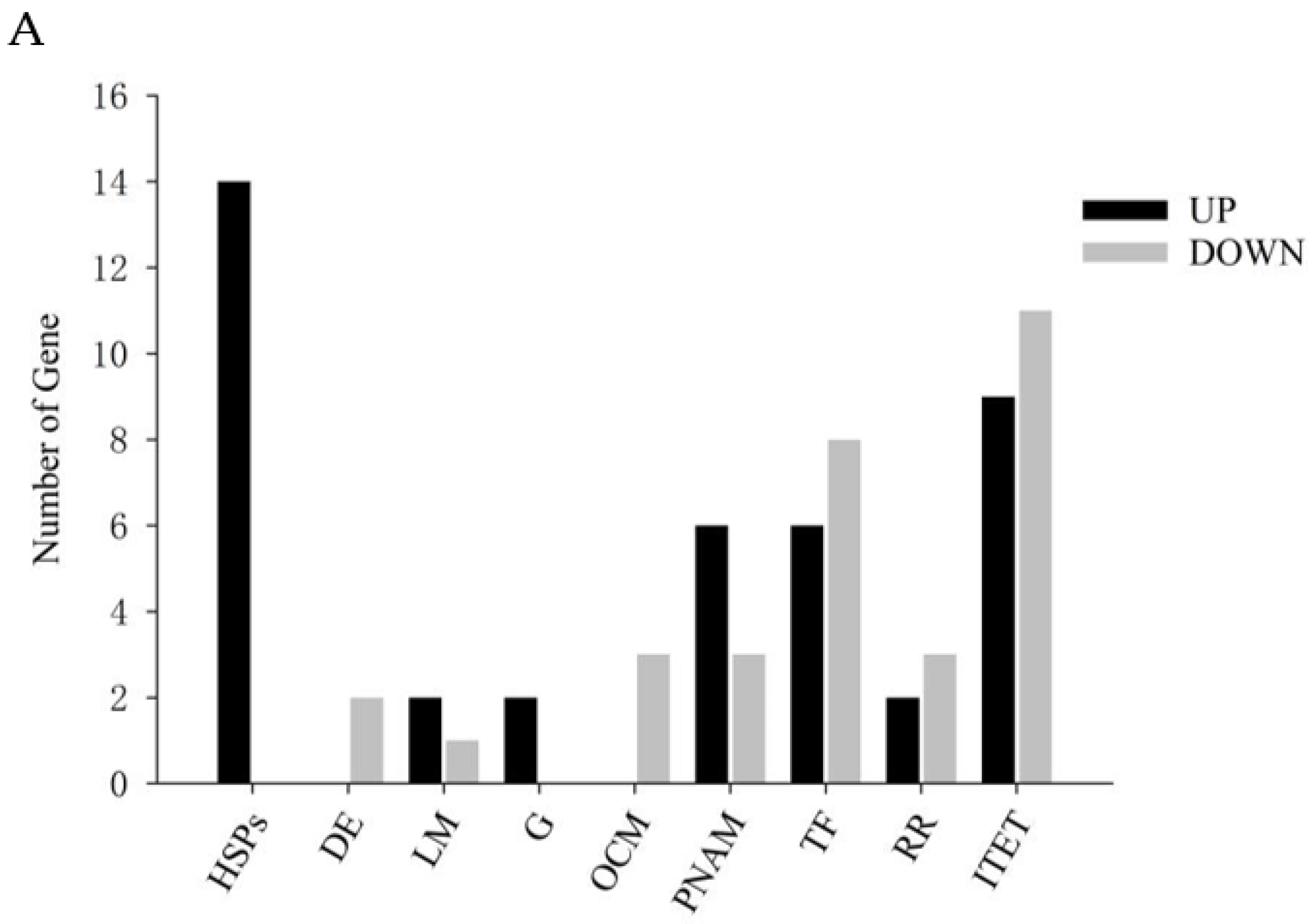
3.5. Analysis of Differentially Expressed Genes
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Gene ID | Gene Name | Readcount-TC | Readcount-TH | FDR | Regulated |
|---|---|---|---|---|---|
| c82189.graph_c0 | sHSPs | 4877 | 50087 | 0 | Up |
| c86948.graph_c1 | HSP70 | 1444 | 106,489 | 0 | Up |
| c79212.graph_c0 | sHSPs | 945 | 61,275 | 0 | Up |
| c86227.graph_c0 | HSP70 | 306 | 13,733 | 0 | Up |
| c91040.graph_c0 | HSP70 | 339 | 52,034 | 0 | Up |
| c92401.graph_c0 | HSP70 | 1503 | 92,464 | 0 | Up |
| c80481.graph_c0 | HSP90 | 22354 | 119,681 | 0 | Up |
| c79843.graph_c0 | sHSPs | 965 | 23,371 | 0 | Up |
| c82009.graph_c1 | HSP90 | 154 | 469 | 3.35 × 10−8 | Up |
| c77102.graph_c0 | HSP70 | 46 | 134 | 0.001081 | Up |
| c81481.graph_c0 | HSP40 | 4393 | 13020 | 3.80 × 10−11 | Up |
| c75847.graph_c0 | HSP40 | 24 | 105 | 7.14 × 10−6 | Up |
| c76869.graph_c0 | HSP40 | 9 | 46 | 0.004722 | Up |
| c84292.graph_c0 | HSP40 | 3153 | 27349 | 0 | Up |
| Gene ID | Gene Name | Readcount-TC | Readcount-TH | FDR | Regulated |
|---|---|---|---|---|---|
| c86948.graph_c1 | HSP70 | 2425 | 46786 | 0 | Up |
| c79212.graph_c0 | sHSPs | 1580 | 11134 | 4.86 × 10−13 | Up |
| c83631.graph_c0 | HSP40 | 2 | 347 | 0 | Up |
| c86227.graph_c0 | HSP70 | 361 | 7620 | 0 | Up |
| c91040.graph_c0 | HSP70 | 178 | 4234 | 0 | Up |
| c92401.graph_c0 | HSP70 | 2294 | 46106 | 0 | Up |
| c80481.graph_c0 | HSP90 | 27241 | 72567 | 0.000985 | Up |
| c90689.graph_c0 | HSP70 | 106 | 3226 | 0 | Up |
| c79843.graph_c0 | sHSPs | 2212 | 8574 | 3.64 × 10−7 | Up |
| c70517.graph_c0 | HSP70 | 2 | 19 | 0.002453 | Up |
| c84424.graph_c0 | HSP40 | 6 | 771 | 0 | Up |
| c86230.graph_c0 | HSP40 | 6 | 179 | 0 | Up |
| c84292.graph_c0 | HSP40 | 4523 | 9985 | 0.008762 | Up |
| c72158.graph_c0 | sHSPs | 646 | 2 | 0 | Down |
| c93293.graph_c0 | HSP70 | 33 | 0 | 7.85 × 10−5 | Down |
| c41847.graph_c0 | HSP60 | 65 | 0 | 3.59 × 10−8 | Down |
| c35384.graph_c0 | HSP70 | 24 | 0 | 0.001249 | Down |
| c86915.graph_c0 | HSP70 | 2679 | 408 | 9.32 × 10−5 | Down |
| c34577.graph_c0 | HSP40 | 75 | 0 | 5.20 × 10−9 | Down |
| c93606.graph_c0 | HSP70 | 26 | 0 | 0.0006555 | Down |
| c82009.graph_c1 | HSP90 | 74 | 0 | 6.26 × 10−9 | Down |
| c77102.graph_c0 | HSP70 | 22 | 0 | 0.00243 | Down |
| c75011.graph_c0 | HSP90 | 277 | 1 | 0 | Down |
| c75847.graph_c0 | HSP40 | 25 | 0 | 0.000902 | Down |
| c78753.graph_c0 | [Mn] SOD | 0 | 556 | 0 | Up |
| c88361.graph_c0 | Extracellular SOD | 265 | 967 | 1.13 × 10−6 | Up |
| c76588.graph_c0 | Prx5 | 6 | 631 | 0 | Up |
| c92939.graph_c0 | Prx2F | 3 | 46 | 1.90 × 10−7 | Up |
References
- Ma, C.S.; Ma, G.; Pincebourde, S. Survive a warming climate: Insect responses to extreme high temperatures. Annu. Rev. Entomol. 2021, 66, 163–184. [Google Scholar] [CrossRef]
- Yang, L.; Huang, L.F.; Wang, W.L.; Chen, E.H.; Chen, H.S.; Jiang, J.J. Effects of temperature on growth and development of the brown planthopper, Nilaparvata lugens (Homoptera: Delphacidae). Environ. Entomol. 2020, 50, 1–11. [Google Scholar] [CrossRef]
- Wolf, U.B.; Roland, G.; Marcel, N.; Nalini, P.; Martin, A.S. Stage- and sex-specific heat tolerance in the yellow dung fly Scathophaga stercoraria. J. Therm. Biol. 2014, 46, 1–9. [Google Scholar] [CrossRef]
- Thomson, L.J.; Robinson, M.; Hoffmann, A.A. Field and laboratory evidence for acclimation without costs in an egg parasitoid. Funct. Ecol. 2001, 15, 217–221. [Google Scholar] [CrossRef]
- Xu, X.N.; Wang, E.D. Statue and analysis of overseas natural enemies merchandise. Chin. J. Biol. Control. 2007, 23, 373–382. [Google Scholar] [CrossRef]
- Wang, Z.Z.; Liu, Y.Q.; Shi, M.; Huang, J.H.; Chen, X.X. Parasitoid wasps as effective biological control agents. J. Integr. Agric. 2019, 18, 705–715. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.S.; Chen, H.Y.; Li, B.P. Mass-Rearing and Utilization of Insect Natural Enemies; China Agriculture Science and Technology Press: Beijing, China, 2014; pp. 14–28. [Google Scholar]
- Debora, L.B.; Wagner, M.J.S.; Maria, G.N.P.; Lessando, G.M.; Narciso, G.R.C.; Guedes, G.M.C. Bioinsecticide-predator interactions: Azadirachtin behavioral and reproductive impairment of the coconut mite predator Neoseiulus baraki. PLoS ONE 2015, 10, e0118343. [Google Scholar] [CrossRef] [Green Version]
- Hoffmann, A.A.; Sørensen, J.G.; Loeschcke, V. Adaptation of Drosophila to temperature extremes: Bringing together quantitative and molecular approaches. J. Therm. Biol. 2003, 28, 175–216. [Google Scholar] [CrossRef]
- Dang, Y.Q.; Wang, X.X.; Yang, Z.Z. Advances in biological control of forest insect pests by using natural enemies in China. J. Environ. Entomol. 2018, 40, 242–255. [Google Scholar]
- Tang, Y.L.; Yang, Z.Q.; Wang, X.Y.; Tang, H.; Jiang, J.; Wei, K.; Lü, J. Biocontrol of oak longhorn beetle, Massicus raddei by releasing parasitoid Dastarcus helophoroides (Coleoptera: Bothrideridae) adults and eggs. Sci. Silvae Sin. 2012, 48, 186–191. [Google Scholar]
- Han, X.; Duan, Y.L.; Li, X.; Han, X.; Duan, Y.L.; Li, X.F.; Wang, Z.Y.; Zhang, Y.N.; Qiao, L.Q. Sensilla ultrastructure of antennae and mouthparts of the first instar Larvae of Dastarcus helophoroides (Coleoptera: Bothrideridae). For. Res. 2021, 34, 180–184. [Google Scholar] [CrossRef]
- Wei, J.R.; Yang, Z.Q.; Liu, Y.L.; Zhao, H.B.; Tang, H. Distribution and ecological biology of Dastarcus helophoroides. For. Pest Dis. 2009, 28, 16–18. [Google Scholar]
- Xie, J.L. Dastarcus helophoroides against Monochamus alternatus in pine forest. Biol. Disaster Sci. 2019, 42, 66–69. [Google Scholar]
- Zhang, Z.M. Studies on control effect of Dastarcus helophoroides on Monochamus alternates. For. Prospect. Des. 2019, 39, 42–45. [Google Scholar]
- Wei, J.R.; Yang, Z.Q.; Tang, H.; Ma, J.H.; Du, J.W. Behavior of a cerambycid parasitoid beetle (Dastarcus helophoroides). Sci. Silvae Sin. 2008, 44, 50–55. [Google Scholar]
- Gao, S.K.; Zahng, Y.L.; Tang, Y.L.; Yang, Z.Q.; Wang, X.Y.; Lu, J.F.; Wang, J.; Situ, C.N.; Fu, F.Y. Overwintering characteristics and cold-hardiness of biotype of Dastarcus helophoroides (Coleoptera: Bothrideridae) on Monochamus alternatus (Coleoptera: Cerambycidae). Sci. Silvae Sin. 2016, 52, 68–74. [Google Scholar]
- Huang, H.H.; Xu, Z.F.; Yang, Z.Q.; Wang, X.D.; Fan, J.X.; Qian, M.H.; Te, L.K.; Chen, J.J. Important natural enemy of the Monochamus alternatus Hope—Dastarcus helophoroides. Guangdong For. Sci. Technol. 2003, 19, 76–77, 82. [Google Scholar]
- Zheng, Y.N.; Jang, S.W.; Huang, F.Y.; Yang, Z.Q.; Gao, G.P.; Liu, P.X.; Shi, Y.; Fan, L.C.; Zhang, Y.L. Dastarcus helophoroides, Natural Enemy of Monochamus saltuarius was found in pine tree infected by Bursaphelenchus xylophilus in Liaoning, China. Chin. J. Biol. Control. 2020, 36, 315–318. [Google Scholar] [CrossRef]
- Li, G.H.; Li, B.P.; Xu, F.Y.; Zheng, H.Y.; Xie, C.X.; Meng, L. Parasitization preference and developmental performances in the ectoparasitoid Dastarcus helophoroides (Coleoptera: Bothrideridae). Acta Ecol. Sin. 2016, 36, 3796–3801. [Google Scholar]
- Hao, C.F.; Wang, H.P.; Zahng, Z.Q.; Chang, Y.; Li, M.L. Characterization and expression analysis of HSP70 gene in Dastarcus helophoroides. J. Northeast. For. Univ. 2016, 44, 108–115, 124. [Google Scholar] [CrossRef]
- Zhou, Y.J.; Lu, C.D.; Shen, H.Y.; Liang, Y.; Chen, Z.H.; Zhong, J.H.; Tan, J.J.; Wang, X.X.; Liang, G.H. Adaptation and physiological response of Dastarcus helophoroides (Fairmaire) to high temperature stress. Chin. J. Biol. Control. 2021, 37, 1179–1188. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.D.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.Y.; Kang, X.L.; Wu, H.H.; Kristop, S.; Zhang, J.Z.; Ma, E.; Zhu, K.Y. Transcriptome-wide survey, gene expression profiling and exogenous chemical-induced transcriptional responses of cytochrome P450 superfamily genes in migratory locust (Locusta migratoria). Insect Biochem. Mol. Biol. 2018, 100, 66–77. [Google Scholar] [CrossRef]
- Kuerban, T.S.; Lu, X.Y.; Liu, X.N.; Ma, J. Transcriptomic analysis of the desert beetle Microdera punctipennis (Coleoptera: Tenebrionidae) in response to short-term cold stress. Acta Entomol. Sin. 2016, 59, 581–591. [Google Scholar] [CrossRef]
- Wang, L.K.; Feng, Z.X.; Wang, X.; Wang, X.W.; Zhang, X.G. DEGseq: An R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 2010, 26, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.M.; Wu, Z.X.; Wang, F.H. Response of insects to extreme high and low temperature stresses. J. Environ. Entomol. 2010, 32, 250–255. [Google Scholar]
- Zhang, W.; Song, W.; Zhang, Z.Q.; Wang, H.D.; Yang, M.M.; Guo, R.J.; Li, M.L. Transcriptome analysis of Dastarcus helophoroides (Coleoptera: Bothrideridae) using Illumina HiSeq sequencing. PLoS ONE. 2014, 9, e100673. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.H.; Ding, S.B.; Zhang, Y.M.; Wang, Y. Effects of artificial diet on the growth & development and fecundity in Helicoverpa armigera. J. Henan Agric. Univ. 2010, 44, 78–82. [Google Scholar] [CrossRef]
- Miao, S.M.; Liao, P.; Ye, M.; Mao, J.J.; Wang, M.Q.; Zhang, L.S.; Chen, H.Y.; Liu, C.X. Inbreeding depression in captive-raised Arma chinensis. Chin. J. Biol. Control. 2019, 35, 191–196. [Google Scholar] [CrossRef]
- Pu, Y.C.; Huang, B.; Hou, Y.M. Inbreeding depression in captive-raised diamondback moths. Chin. J. Appl. Entomol. 2015, 52, 1244–1250. [Google Scholar]
- Shan, Y.M.; Zhang, Y.; Huo, Z.J.; Pang, B.P.; Sun, X.T. Molecular cloning of a serine protease gene DdSP and its response to temperature stress in Galeruca daurica (Coleoptera: Chrysomelidae). Acta Entomol. Sin. 2018, 61, 761–770. [Google Scholar] [CrossRef]
- Wang, C.H.; Sun, S.Q.; Xu, J.L.; Zhao, X.L.; Xue, C.B. Differential expressed genes and their pathways of the resistance to flubendiamide in Plutella xylostella. Sci. Agric. Sin. 2018, 51, 2106–2137. [Google Scholar]
- Zhao, X.; Chen, M.J.; Zhao, Y.; Zha, L.; Yang, H.L.; Wu, Y.J. GC–MS-based nontargeted and targeted metabolic profiling identifies changes in the Lentinula edodes mycelial metabolome under high-temperature stress. Int. J. Mol. Sci. 2019, 20, 2330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, C.C.; Wang, W.L.; Huhe, B.T. Sequencing and analysis of the transcriptome of Parabronema skrjabini in camle. Chin. J. Vet. Sci. 2017, 37, 671–675. [Google Scholar] [CrossRef]
- Fan, L.F.; Zhong, Y.S.; Lin, J.R. Comparison of related enzyme activities between diapause and nondiapause eggs of the silkworm, Bombyx mori. Acta Entomol. Sin. 2011, 54, 1258–1263. [Google Scholar] [CrossRef]
- Zwingmann, C.; Richter-Landsberg, C.; Leibfritz, D. 13C isotopomer analysis of glucose and alanine metabolism reveals cytosolic pyruvate compartmentation as part of energy metabolism in astrocytes. Glia 2001, 34, 200–212. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Wang, M.; Wang, S.L.; Wu, Q.J.; Xu, B.Y.; Zhang, Y.J. Advance on glycosphingolipids as receptor for Bacillus thuringiensis toxins. Chin. J. Pestic. Sci. 2010, 12, 13–21. [Google Scholar]
- John, T. Heat shock proteins and Drosophila aging. Exp. Gerontol. 2011, 46, 355–362. [Google Scholar] [CrossRef] [Green Version]
- Lü, J.H.; Huo, M.F. Transcriptome analysis reveals heat tolerance of Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae) adults. J. Stored Prod. Res. 2018, 78, 59–66. [Google Scholar] [CrossRef]
- Ilyasov, R.A.; Lee, M.L.; Yunusbaev, U.; Nikolenko, A.; Kwon, H.-W. Estimation of C-derived introgression into A. m. mellifera colonies in the Russian Urals using microsatellite genotyping. Genes Genom. 2020, 42, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Li, K.; Zhu, J.Y.; Fang, Q.; Ye, G.Y.; Wang, H.; Li, K.; Zhu, J.Y. Cloning and expression pattern of heat shock protein genes from the endoparasitoid wasp, Pteromalus puparum in response to environmental stresses. Arch. Insect Biochem. Physiol. 2012, 79, 247–263. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Wang, Y.N.; Zhu, N.; Chen, X.X. Four heat shock protein genes of the endoparasitoid wasp, Cotesia vestalis, and their transcriptional profiles in relation to developmental stages and temperature. PLoS ONE. 2013, 8, e59721. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, R.; Qi, L.D.; Du, Y.Z.; Li, Y.X. Thermotolerance and heat-shock protein gene expression patterns in Bemisia tabaci (Hemiptera: Aleyrodidae) mediterranean in relation to developmental stage. J. Econ. Entomol. 2017, 110, 2190–2198. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.O.; Lei, Z.R. Current developments of heat shock proteins in insect. Sci. Agric. Sin. 2005, 38, 2023–2034. [Google Scholar]
- Zhang, T.H.; Zhang, X.H. Research progress of heat shock protein in insect. J. Shanxi Agric. Univ. 2019, 39, 35–40. [Google Scholar] [CrossRef]
- Gu, L.L.; Li, M.Z.; Wang, G.R.; Liu, X.D. Multigenerational heat acclimation increases thermal tolerance and expression levels of Hsp70 and Hsp90 in the rice leaf folder larvae. J. Therm. Biol. 2019, 81, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Kodrík, D.; Bednářová, A.; Zemanová, M.; Krishnan, N. Hormonal regulation of response to oxidative stress in insects—An update. Int. J. Mol. Sciences 2015, 16, 25788–25816. [Google Scholar] [CrossRef] [Green Version]
- Joanisse, D.; Storey, K. Oxidative stress and antioxidants in overwintering larvae of cold-hardy goldenrod gall insects. J. Exp. Biol. 1996, 199, 1483–1491. [Google Scholar] [CrossRef]
- Danilenko, V.N.; Devyatkin, A.V.; Marsova, M.V.; Shibilova, M.U.; Ilyasov, R.A.; Shmyrev, V.I. Common inflammatory mechanisms in COVID-19 and Parkinson’s diseases: The role of microbiome, pharmabiotics and postbiotics in their prevention. J. Inflamm. Res. 2021, 14, 6349–6381. [Google Scholar] [CrossRef]
- Katsura, T.; Nobuyuki, T.; Kenji, K.; Mitsuaki, M.; Yoichi, N. Extracellular superoxide dismutase induced by dopamine in cultured astrocytes Neurochem. Res. 2013, 38, 32–41. [Google Scholar] [CrossRef]
- Radyuk, S.N.; Klichko, V.I.; Spinola, B.; Sohal, R.S.; Orr, W.C. The peroxiredoxin gene family in drosophila melanogaster. Free Radic. Biol. Med. 2001, 31, 1090–1100. [Google Scholar] [CrossRef]
- Kang, S.W.; Rhee, S.G.; Chang, T.S.; Chang, T.S.; Jeong, W.; Choi, M.H. 2-Cys peroxiredoxin function in intracellular signal transduction: Therapeutic implications. Trends Mol. Med. 2005, 11, 571–578. [Google Scholar] [CrossRef] [PubMed]
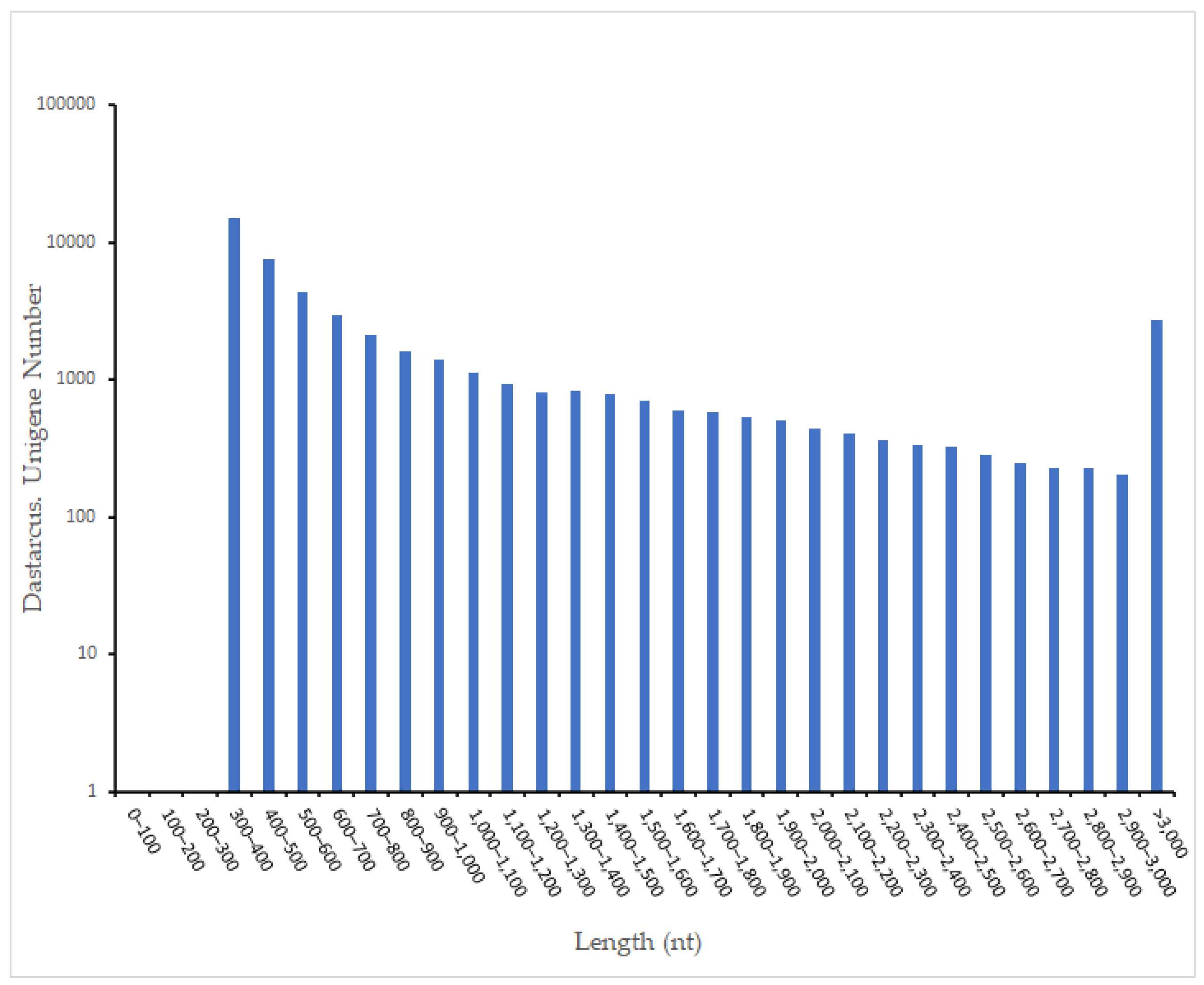
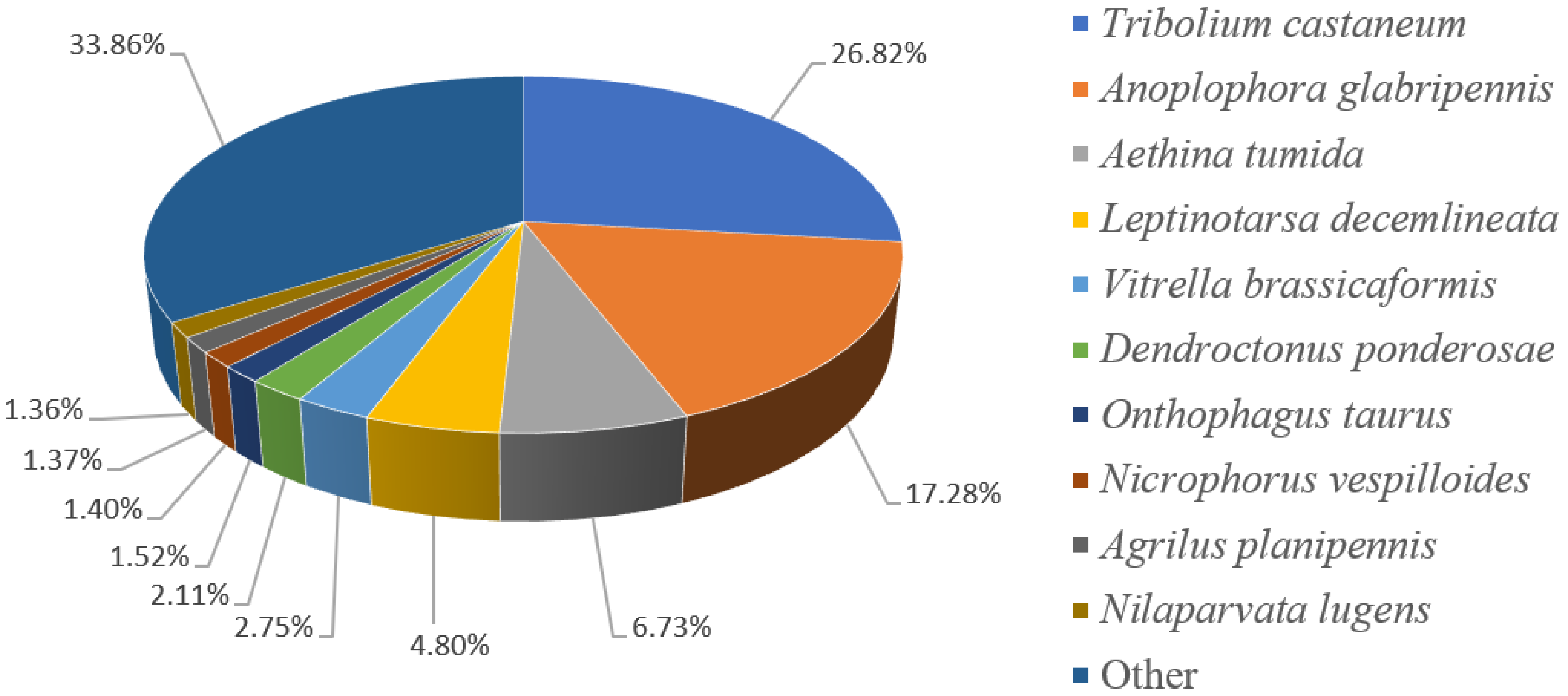
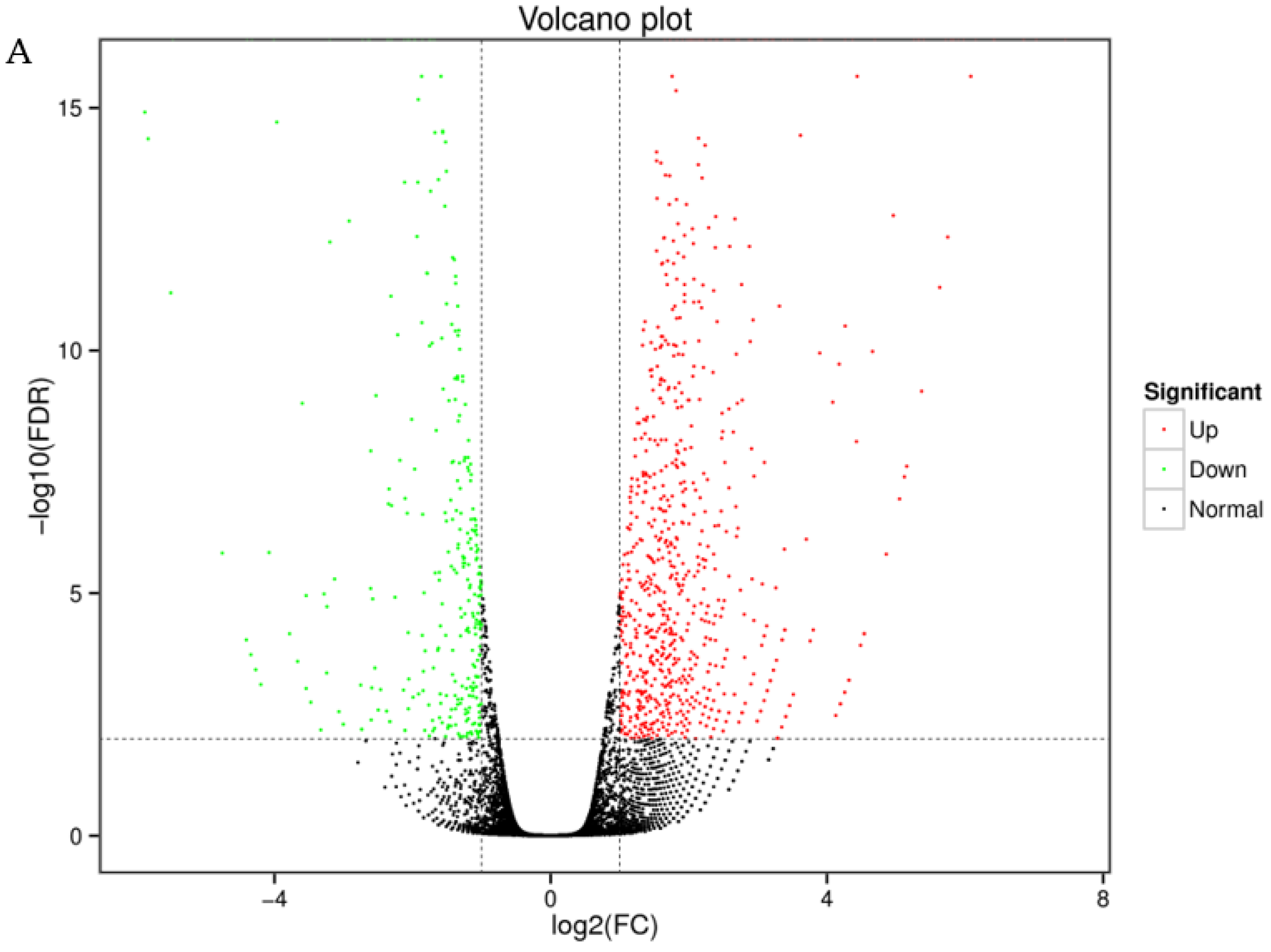
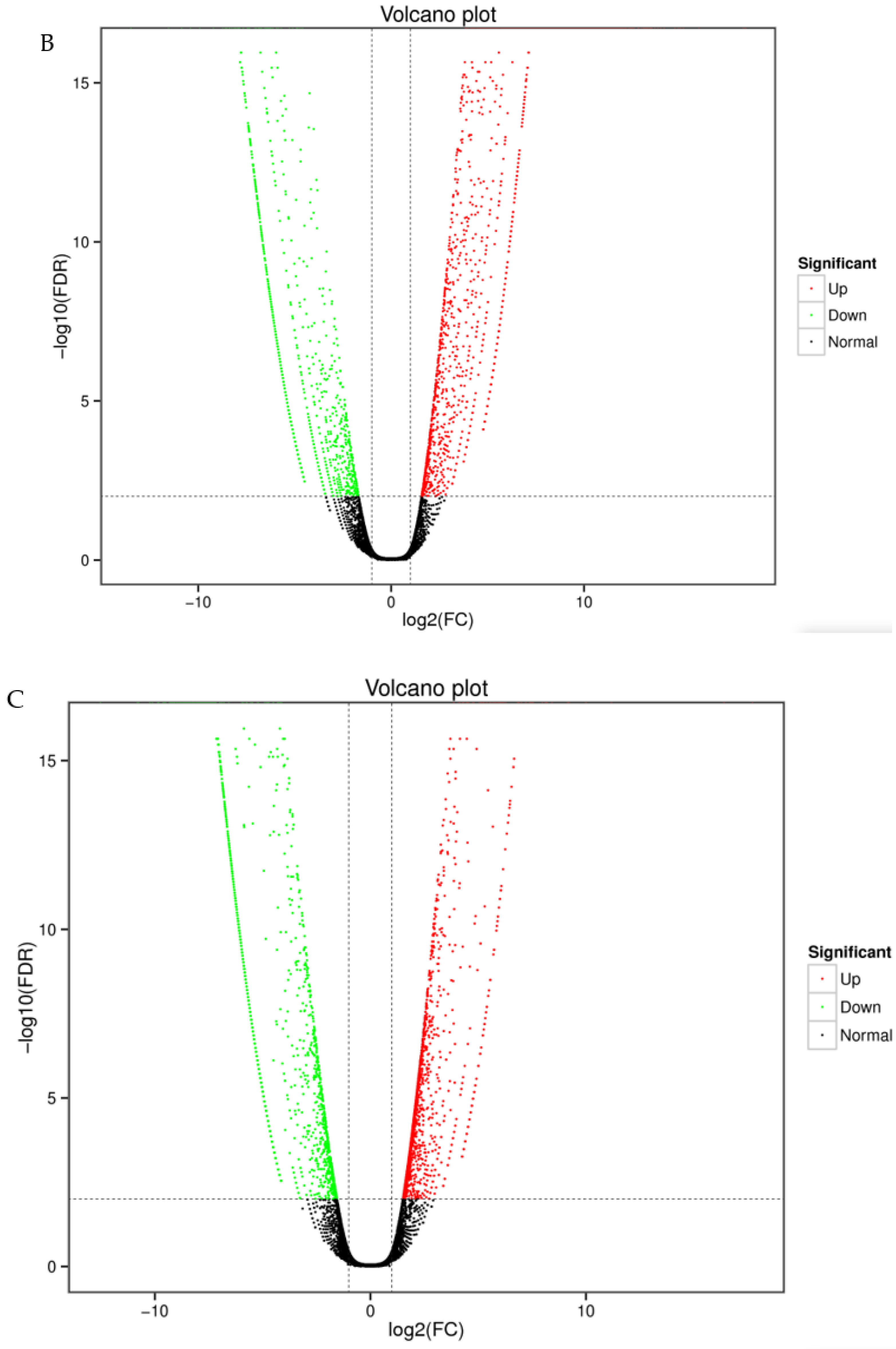


| Sample | Total Number | Total Length (bp) | Mean Length (bp) | N50 | GC (%) |
|---|---|---|---|---|---|
| All unigenes | 47,763 | 47,252,603 | 989.31 | 1607 | 42.95–44.14 |
| Annotation Database | Annotated Number | Length ≥ 1000 bp | Proportion (%) |
|---|---|---|---|
| COG | 6555 | 4187 | 32.70 |
| GO | 9404 | 5754 | 46.91 |
| KEGG | 9206 | 5799 | 45.92 |
| KOG | 12,290 | 7822 | 61.31 |
| Pfam | 14,516 | 9105 | 72.41 |
| Swissprot | 10,888 | 7099 | 54.32 |
| eggNOG | 17,623 | 10,088 | 87.91 |
| Nr | 18,803 | 10,666 | 93.80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, H.; He, H.; Lu, C.; Liang, Y.; Wu, H.; Zheng, L.; Wang, X.; Liang, G. Comparative Transcriptome Analysis of Two Populations of Dastarcus helophoroides (Fairmaire) under High Temperature Stress. Forests 2022, 13, 13. https://doi.org/10.3390/f13010013
Shen H, He H, Lu C, Liang Y, Wu H, Zheng L, Wang X, Liang G. Comparative Transcriptome Analysis of Two Populations of Dastarcus helophoroides (Fairmaire) under High Temperature Stress. Forests. 2022; 13(1):13. https://doi.org/10.3390/f13010013
Chicago/Turabian StyleShen, Haiyan, Huan He, Ciding Lu, Yun Liang, Hongmin Wu, Lingzhi Zheng, Xiaoyi Wang, and Guanghong Liang. 2022. "Comparative Transcriptome Analysis of Two Populations of Dastarcus helophoroides (Fairmaire) under High Temperature Stress" Forests 13, no. 1: 13. https://doi.org/10.3390/f13010013
APA StyleShen, H., He, H., Lu, C., Liang, Y., Wu, H., Zheng, L., Wang, X., & Liang, G. (2022). Comparative Transcriptome Analysis of Two Populations of Dastarcus helophoroides (Fairmaire) under High Temperature Stress. Forests, 13(1), 13. https://doi.org/10.3390/f13010013






