Genetic Diversity and Population Structure of Capirona (Calycophyllum spruceanum Benth.) from the Peruvian Amazon Revealed by RAPD Markers
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. DNA Amplification
2.3. Data Analysis
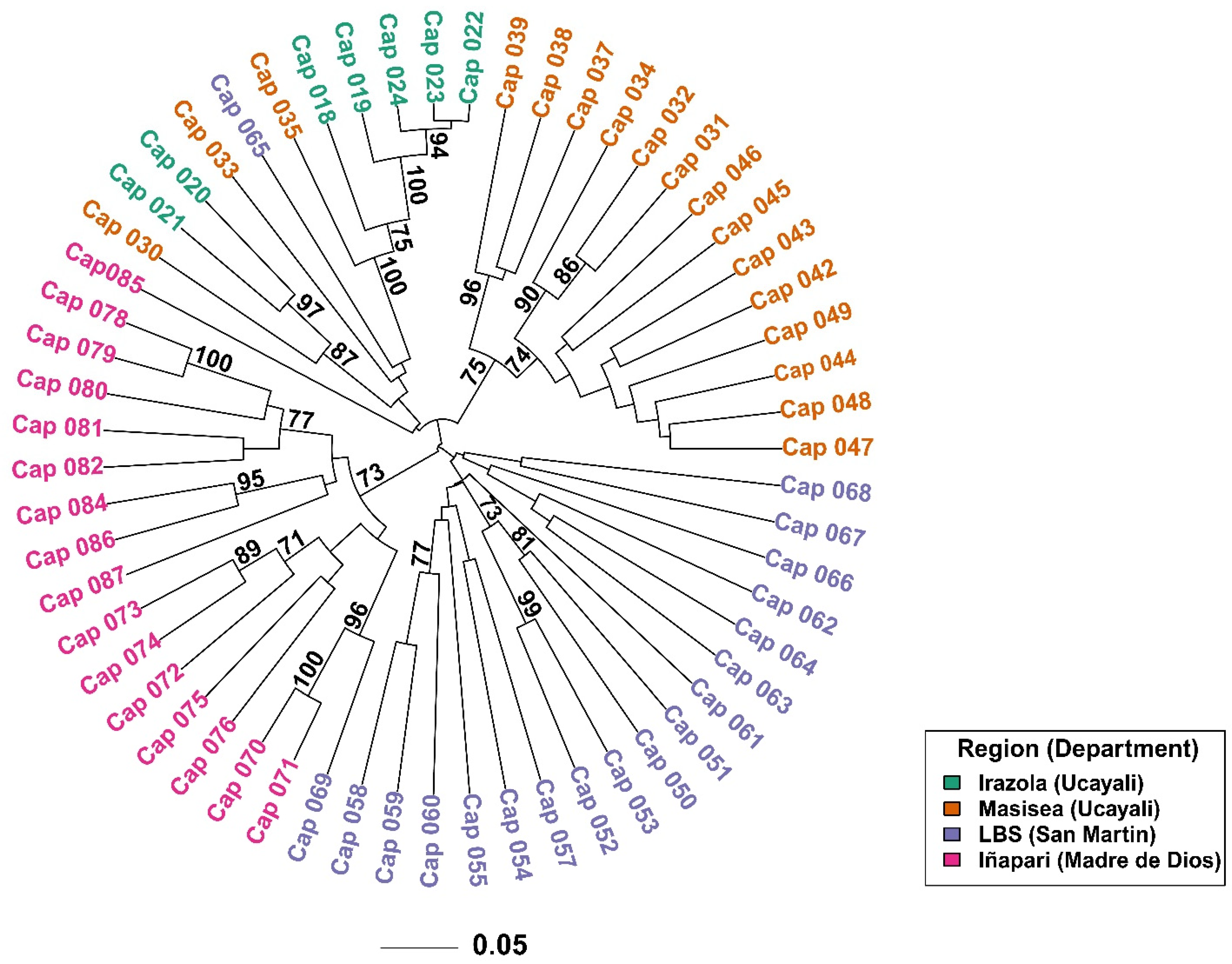
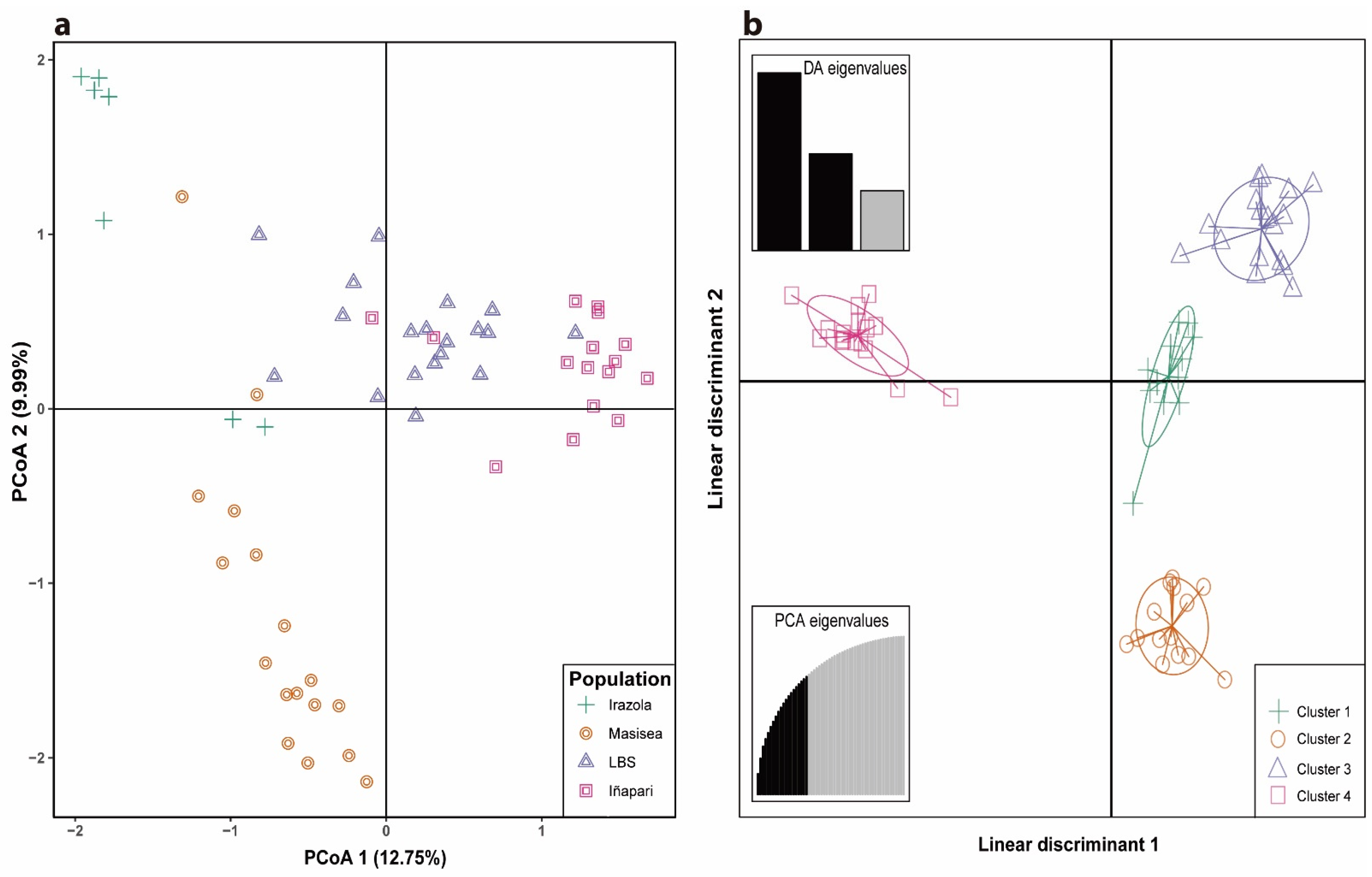
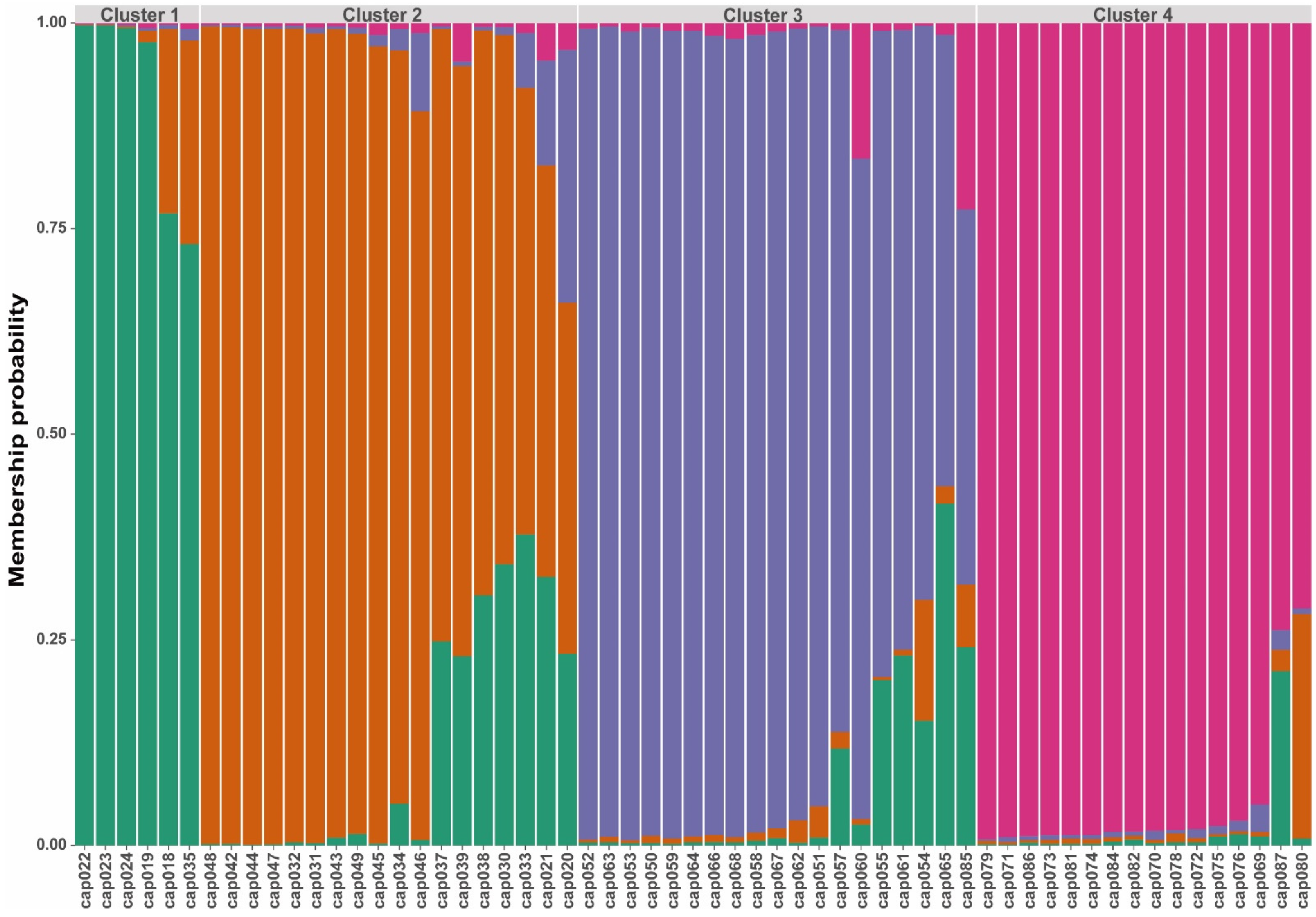
3. Results
3.1. RAPD Analysis
3.2. Genetic Diversity and Population Structure
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Russell, J.R.; Weber, J.C.; Booth, A.; Powell, W.; Dawson, C.S. Genetic variation of Calycophyllum spruceanum in the Peruvian Amazon Basin, revealed by amplified fragment length polymorphism (AFLP) analysis. Mol. Ecol. 1999, 8, 199–204. [Google Scholar] [CrossRef]
- Sears, R. New Forestry on the Floodplain: The ecology and Management of Calycophyllum spruceanum (Rubiaceae) on the Amazon Landscape. Ph.D. Thesis, Columbia University, New York, NY, USA, 2003. [Google Scholar]
- Tauchen, J.; Lojka, B.; Hlasna-Cepkova, P.; Svobodova, E.; Dvorakova, Z.; Rollo, A. Morphological and genetic diversity of Calycophyllum spruceanum (Benth) k. Schum (Rubiaceae) in Peruvian amazon. Agric. Trop. Subtrop. 2011, 44, 212–218. [Google Scholar]
- Weber, J.; Sotelo-Montes, C.; Labarta-Chávarri, R. Tree domestication in the Peruvian Amazon Basin—working with farmers for community development. Agrofor. Today 1997, 9, 4–8. [Google Scholar]
- Busch, J.; Amarjargal, O. Authority of Second-Tier governments to reduce deforestation in 30 tropical countries. Front. For. Glob. Chang. 2020, 3, 1. [Google Scholar] [CrossRef]
- Estoque, R.C.; Ooba, M.; Avitabile, V.; Hijioka, Y.; Dasgupta, R.; Togawa, T.; Muruyama, Y. The future of Southeast Asia’s forests. Nat. Commun 2019, 10, 1–12. [Google Scholar] [CrossRef]
- Rasolofoson, R.A.; Ferraro, P.J.; Jenkins, C.N.; Jones, J.P. effectiveness of community forest management at reducing deforestation in Madagascar. Biol. Conserv. 2015, 184, 271–277. [Google Scholar] [CrossRef]
- Singh, P.; Singh, S.P.; Tiwari, A.K.; Sharma, B.L. Genetic diversity of sugarcane hybrid cultivars by RAPDs markers. Biotech 2017, 7, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Bucci, G.; Vendramin, G.G.; Lelli, L.; Vicario, L. Assessing the genetic divergence of Pinus leucodermis Ant. endangered populations: Use of molecular markers for conservation purposes. Theor. Appl. Genet. 1997, 95, 1138–1146. [Google Scholar] [CrossRef]
- Gillies, A.C.M.; Cornelius, J.P.; Newton, A.C.; Navarro, C.; Hernández, M.; Wilson, J. Genetic variation in Costa Rican populations of the tropical timber species Cedrela odorata L., assessed using RAPDs. Mol. Ecol. 1997, 6, 1133–1145. [Google Scholar] [CrossRef]
- Nesbitt, K.A.; Potts, B.M.; Vaillancourt, R.E.; Reid, J.B. Fingerprinting and pedigree analysis in Eucalyptus globulus using RAPDs. Silvae Genet. 1997, 46, 6–11. [Google Scholar]
- Gillies, A.C.M.; Navarro, C.; Lowe, A.J.; Newtons, A.C.; Hernández, M.; Wilson, J. Genetic diversity in Mesoamerican populations of Mahogany (Swietenia macrophylla), assessed using RAPDs. Heredity 1999, 83, 722–732. [Google Scholar] [CrossRef]
- Baldoni, A.B.; Teodoro, L.P.R.; Teodoro, P.E.; Tonini, H.; Tardin, F.D.; Botin, A.A.; Hoogerheide, E.S.; Botelho, S.; Lulu, J.; Neto, A.; et al. Genetic diversity of Brazil nut tree (Bertholletia excelsa Bonpl.) in southern Brazilian Amazon. For. Ecol. Man. 2020, 458, 117795. [Google Scholar] [CrossRef]
- Chen, C.; Chu, Y.; Ding, C.; Su, X.; Huang, Q. Genetic diversity and population structure of black cottonwood (Populus deltoides) revealed using simple sequence repeat markers. BMC Genet. 2020, 21, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ministerio de Economía y Finanzas. Available online: https://www.mef.gob.pe/contenidos/doc_siga/catalogo/ctlogo_familias_madera_capirona.pdf (accessed on 20 August 2021).
- Cruz, W.; Saldaña, C.; Ramos, H.; Baselly, R.; Loli, J.C.; Cuellar, E. Estructura y diversidad genética de poblaciones naturales de Cedrelinga Cateniformis “tornillo” en la región oriental del Perú. Sci. Agropecu. 2020, 11, 521–528. [Google Scholar] [CrossRef]
- Tijerino, A.; Callejas, L.; Cerda-Granados, D.A. Assessment of genetic diversity in five Nicaraguan populations of Cedrela odorata L. (Meliaceae) using RAPD markers. Encuentro 2016, 103, 28–39. [Google Scholar] [CrossRef]
- Renau-Morata, B.; Nebauer, G.; Sales, E.; Allainguillaume, J.; Caligari, P.; Segura, J. Genetic diversity and structure of natural and managed populations of Cedrus atlantica (Pinaceae) assessed using random amplified polymorphic DNA. Am. J. Bot. 2005, 92, 875–884. [Google Scholar] [CrossRef] [PubMed]
- Ye, N.; Weeden, F.; Reisch, I. Genetic mapping and fingerprinting of grape vines using molecular markers. Am. J. Enol. Vitic. 1993, 44, 468. [Google Scholar]
- Lodhi, A.; Daly, J.; Ye, N.; Weeden, F.; Reisch, I. A molecular marker based linkage map of Vitis. Genome 1995, 38, 786–794. [Google Scholar] [CrossRef]
- Weeden, F.; Hemmat, M.; Lawson, M.; Lohdi, M.; Bell, L.; Manganaris, G.; Reisch, I.; Brown, K.; Ye, N. Development and application of molecular marker linkage maps in woody fruit crops. Euphytica 1994, 77, 71–75. [Google Scholar] [CrossRef]
- Büscher, N.; Zyprian, E.; Blaich, R. Identification of grapevine cultivars by DNA analyses: Pitfalls of random amplified polymorphic DNA techniques using 10mer primers. Vitis 1993, 32, 187–188. [Google Scholar]
- Xu, H.; Wilson, J.; Arulsekar, S.; Bakalinsky, A.T. Sequence-specific polymerase chain reaction markers derived from randomly amplified polymorphic DNA markers for fingerprinting grape (Vitis) rootstocks. J. Amer. Soc. Hort. Sci. 1995, 120, 714–720. [Google Scholar] [CrossRef]
- Gogorcena, Y.; Arulsekar, S.; Dandekar, M.; Parfitt, E. Molecular markers for grape characterization. Vitis 1993, 32, 183–185. [Google Scholar]
- Qu, X.; Lu, J.; Lamikanra, O. Genetic diversity in muscadine and American bunch grapes based on randomly amplified polymorphic DNA (RAPD) analysis. J. Amer. Soc. Hort. Sci. 1996, 121, 1020–1023. [Google Scholar] [CrossRef]
- Mendonça, G.; de Souza, M.; Vieira, A.; Estopa, A.; Reis, F.; Carvalho, D. Using random amplified polymorphic DNA to assess genetic diversity and structure of natural Calophyllum brasiliense (Clusiaceae) populations in riparian forests. Int. J. For. Res. 2014, 2014, 1–9. [Google Scholar]
- Basyuni, M.; Prayogi, H.; Putri, P.; Syahputra, I.; Siregar, S.; Risnasari, I.; Wati, R.; Arifiyanto, D. RAPDs markers on genetic diversity in three populations of pisifera type of oil palm (Elaeis guineensis). In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2018; Volume 130, p. 012050.guineensis. [Google Scholar]
- Kim, H.; Lei, P.; Wang, A.; Liu, S.; Zhao, Y.; Huang, F.; Yu, Z.; Zhu, G.; He, Z.; Tan, D.; et al. Genetic diversity of castor bean (Ricinus communis L.) revealed by ISSR and RAPDs markers. Agronomy 2021, 11, 457. [Google Scholar] [CrossRef]
- Vidal, J.R.; Moreno, S.; Masa, A.; Ortiz, J.M. Study of the genetic homogeneity of albariño (Vitis vinifera L.) growing in Galicia (Spain) using isozyme and RAPDs markers. Vitis 1998, 37, 145–146. [Google Scholar]
- Doyle, J.J.; Doyle, J.L. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem. Bulletin 1987, 19, 11–15. [Google Scholar]
- Cruz, W.; Ramos, H.; y Cuellar, J. Manual de Protocolos para el Estudio de Diversidad Genética en Especies Forestales Nativas: Tornillo (Cedrelinga cateniformis Ducke), Capirona (Calycophyllum spruceanum Benth.), Shihuahuaco (Dipteryx sp.), Ishpingo (Amburana sp.) y Castaña (Bertholletia Excelsa); Instituto Nacional de Innovación Agraria: Lima, Perú, 2019. [Google Scholar]
- Goyal, P.; Jain, R.; Kachhwaha, S.; Kothari, S.L. Assessment of genetic diversity in Pithecellobium dulce (Roxb.) Benth. germplasm using RAPDs and ISSR markers. Trees 2014, 29, 637–653. [Google Scholar] [CrossRef]
- Chia-Wong, J.A.; Garcia, C.L.; Suni, N.M.; Eskes, B. Characterization of Theobroma cacao L. collection at Tingo Maria using ISSR molecular markers. Rev. Aporte St. 2011, 4, 195–202. [Google Scholar]
- Chesnokov, Y.V.; Artemyeva, A.M. Evaluation of the measure of polymorphism information of genetic diversity. Agric. Biol. 2015, 5. [Google Scholar]
- Prevosti, A.; Ocana, J.; Alonso, G. Distances between populations of Drosophila subobscura, based on chromosome arrangement frequencies. Theor. Appl. Gene. 1975, 45, 231–241. [Google Scholar] [CrossRef]
- Kamvar, Z.N.; Tabima, J.F.; Grünwald, N.J. Poppr: An R package for genetic analysis of populations with clonal, partially clonal, and/or sexual reproduction. PeerJ 2014, 2, e281. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, J.K.; Stephens, M.; Rosenberg, N.A.; Donnelly, P. Association mapping in structured populations. Am. J. Hum. Genet. 2000, 67, 170–181. [Google Scholar] [CrossRef] [PubMed]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef] [PubMed]
- Francis, R.M. pophelper: An R package and web app to analyse and visualize population structure. Mol. Ecol. Resour. 2017, 17, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Caye, K.; Deist, T.M.; Martins, H.; Michel, O.; François, O. TESS3: Fast inference of spatial population structure and genome scans for selection. Mol. Ecol. Resour. 2016, 16, 540–548. [Google Scholar] [CrossRef]
- Waples, R.S.; Gaggiotti, O. What is a population? an empirical evaluation of some genetic methods for identifying the number of gene pools and their degree of connectivity. Mol. Ecol. 2006, 15, 1419–1439. [Google Scholar] [CrossRef] [PubMed]
- Frantz, A.C.; Cellina, S.; Krier, A.; Schley, L.; Burke, T. Using spatial Bayesian methods to determine the genetic structure of a continuously distributed population: Clusters or isolation by distance? J. Appl. Ecol. 2009, 46, 493–505. [Google Scholar] [CrossRef]
- Janes, J.K.; Miller, J.M.; Dupuis, J.R.; Malenfant, R.M.; Gorrell, J.C.; Cullingham, C.I.; Andrew, R.L. The K= 2 conundrum. Mol. Ecol. 2017, 26, 3594–3602. [Google Scholar] [CrossRef]
- Iorizzo, M.; Senalik, D.A.; Ellison, S.L.; Grzebelus, D.; Cavagnaro, P.F.; Allender, C.; Brunet, J.; Spooner, D.M.; Van Deynze, A.; Simon, P.W. Genetic structure and domestication of carrot (Daucus carota subsp. sativus) (Apiaceae). Am. J. Bot. 2013, 100, 930–938. [Google Scholar] [CrossRef]
- Arbizu, C.I.; Ellison, S.L.; Senalik, D.; Simon, P.W.; Spooner, D.M. Genotyping-by-sequencing provides the discriminating power to investigate the subspecies of Daucus carota (Apiaceae). BMC Evol. Biol. 2016, 16, 1–16. [Google Scholar] [CrossRef]
- Nei, M. Genetic distance between populations. Am. Nat. 1972, 106, 283–292. [Google Scholar] [CrossRef]
- Botstein, D.; White, R.L.; Skolnick, M.; Davis, R.W. Construction of a genetic-linkage map in man using restriction fragment length polymorphisms. Am. J. Hum. Genet. 1980, 32, 314–331. [Google Scholar]
- Dávila-Lara, A.; Affenzeller, M.; Tribsch, A.; Díaz and Comes, H.P. AFLP diversity and spatial structure of Calycophyllum candidissimum (Rubiaceae), a dominant tree species of Nicaragua’s critically endangered seasonally dry forest. Heredity 2017, 4, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Montes, C.S.; Vidaurre, H.; Weber, J. Variation in stem-growth and branch-wood traits among provenances of Calycophyllum spruceanum Benth. from the Peruvian Amazon. New For. 2003, 26, 1–16. [Google Scholar] [CrossRef]
- Tuisima-Coral, L.L.; Hlásná Čepková, P.; Weber, J.C.; Lojka, B. Preliminary evidence for domestication effects on the genetic diversity of Guazuma crinita in the Peruvian Amazon. Forests 2020, 11, 795. [Google Scholar] [CrossRef]
- Eltaher, S.; Sallam, A.; Belamkar, V.; Emara, H.A.; Nower, A.A.; Salem, K.F.M.; Poland, J.; Baenziger, P.S. Genetic diversity and population structure of F3:6 nebraska winter wheat genotypes using genotyping-by-sequencing. Front. Genet. 2018, 9, 76. [Google Scholar] [CrossRef] [PubMed]
- Hennink, S.; Zeven, A.C. The interpretation of Nei and Shannon-Weaver within population variation indices. Euphytica 1990, 51, 235–240. [Google Scholar] [CrossRef]
- Rosyara, U.R.; De Jong, W.S.; Douches, D.S.; Endelman, J.B. Software for genome-wide association studies in autopolyploids and its application to potato. Plant Gen. 2016, 9. [Google Scholar] [CrossRef] [PubMed]
- Campoy, J.A.; Lerigoleur-Balsemin, E.; Christmann, H.; Beauvieux, R.; Girollet, N.; Quero-García, J.; Dirlewanger, E.; Ba reneche, T. Genetic diversity, linkage disequilibrium, population structure and construction of a core collection of Prunus avium L. landraces and bred cultivars. BMC Plant Biol. 2016, 16, 1–15. [Google Scholar] [CrossRef]
- Lee, K.J.; Lee, J.-R.; Sebastin, R.; Shin, M.-J.; Kim, S.-H.; Cho, G.-T.; Hyun, D.Y. Assessment of genetic diversity of tea germplasm for its management and sustainable use in Korea genebank. Forests 2019, 10, 780. [Google Scholar] [CrossRef]
- González, N.A. Identificación y Validación de Single Nucleotide Polymorphism (SNPs) Distribuidos en el Genoma de Eucalyptus globulus. Master’s Thesis, Universidad de Concepción, Concepción, Chile, 2015. [Google Scholar]
- Durán Reyes, R.F. Modelos de Predicción Genómicos para la Selección de Genotipos de Eucalyptus globulus en Base a Densidad de la Madera y Volumen. Ph.D. Thesis, Universidad de Concepción, Concepción, Chile, 2017. [Google Scholar]
- Liu, C.; Dou, Y.; Guan, X.; Fu, Q.; Zhang, Z.; Hu, Z.; Zheng, J.; Lu, Y.; Li, W. De novo transcriptomic analysis and development of EST-SSRs for Sorbus pohuashanensis (Hance) Hedl. PLoS ONE 2017, 12, e0179219. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Zheng, J.; Jiao, Z.; Zhou, J.; He, K.; Wang, B.; He, X. Transcriptome sequencing and EST-SSR marker development in Salix babylonica and S. suchowensis. Tree Genet. Genomes 2019, 15, 9. [Google Scholar] [CrossRef]
- Wu, Q.; Zang, F.; Xie, X.; Ma, Y.; Zheng, Y.; Zang, D. Full-length transcriptome sequencing analysis and development of EST-SSR markers for the endangered species Populus wulianensis. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
| Code | Region | Department | Latitude | Longitude | Code | Region | Department | Latitude | Longitude |
|---|---|---|---|---|---|---|---|---|---|
| cap018 | Irazola | Ucayali | 8°51′ S | 75°7′ W | cap057 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap019 | Irazola | Ucayali | 8°51′ S | 75°7′ W | cap058 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap020 | Irazola | Ucayali | 8°51′ S | 75°7′ W | cap059 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap021 | Irazola | Ucayali | 8°51′ S | 75°7′ W | cap060 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap022 | Irazola | Ucayali | 8°51′ S | 75°7′ W | cap061 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap023 | Irazola | Ucayali | 8°51′ S | 75°7′ W | cap062 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap024 | Irazola | Ucayali | 8°51′ S | 75°7′ W | cap063 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap030 | Masisea | Ucayali | 8°41′ S | 73°37′ W | cap064 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap031 | Masisea | Ucayali | 8°42′ S | 73°40′ W | cap065 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap032 | Masisea | Ucayali | 8°42′ S | 73°40′ W | cap066 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap033 | Masisea | Ucayali | 8°43′ S | 73°40′ W | cap067 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap034 | Masisea | Ucayali | 8°43′ S | 73°40′ W | cap068 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap035 | Masisea | Ucayali | 8°43′ S | 73°40′ W | cap069 | LBS | San Martín | 6°28′ S | 76°20′ W |
| cap037 | Masisea | Ucayali | 8°49′ S | 73°44′ W | cap070 | Iñapari | Madre de Dios | 10°60′ S | 69°53′ W |
| cap038 | Masisea | Ucayali | 8°50′ S | 73°45′ W | cap071 | Iñapari | Madre de Dios | 10°60′ S | 69°53′ W |
| cap039 | Masisea | Ucayali | 8°50′ S | 73°47′ W | cap072 | Iñapari | Madre de Dios | 10°57′S | 69°54′ W |
| cap042 | Masisea | Ucayali | 8°50′ S | 73°47′ W | cap073 | Iñapari | Madre de Dios | 10°59′ S | 69°53′ W |
| cap043 | Masisea | Ucayali | 8°50′ S | 73°47′ W | cap074 | Iñapari | Madre de Dios | 10°59′ S | 69°54′ W |
| cap044 | Masisea | Ucayali | 8°50′ S | 73°47′ W | cap075 | Iñapari | Madre de Dios | 10°59′ S | 69°53′ W |
| cap045 | Masisea | Ucayali | 8°53′ S | 73°52′ W | cap076 | Iñapari | Madre de Dios | 10°58′ S | 69°53′ W |
| cap046 | Masisea | Ucayali | 8°53′ S | 73°52′ W | cap078 | Iñapari | Madre de Dios | 10°58′ S | 69°54′ W |
| cap047 | Masisea | Ucayali | 8°40′ S | 74°11′ W | cap079 | Iñapari | Madre de Dios | 10°58′ S | 69°54′ W |
| cap048 | Masisea | Ucayali | 8°41′ S | 74°12′ W | cap080 | Iñapari | Madre de Dios | 10°58′ S | 69°54′ W |
| cap049 | Masisea | Ucayali | 8°41′ S | 74°12′ W | cap081 | Iñapari | Madre de Dios | 10°58′ S | 69°54′ W |
| cap050 | LBS | San Martín | 6°28′ S | 76°20′ W | cap082 | Iñapari | Madre de Dios | 10°58′ S | 69°54′ W |
| cap051 | LBS | San Martín | 6°28′ S | 76°20′ W | cap084 | Iñapari | Madre de Dios | 10°58′ S | 69°54′ W |
| cap052 | LBS | San Martín | 6°28′ S | 76°20′ W | cap085 | Iñapari | Madre de Dios | 10°60′ S | 69°53′ W |
| cap053 | LBS | San Martín | 6°28′ S | 76°20′ W | cap086 | Iñapari | Madre de Dios | 11°0′ S | 69°53′ W |
| cap054 | LBS | San Martín | 6°28′ S | 76°20′ W | cap087 | Iñapari | Madre de Dios | 11°0′ S | 69°53′ W |
| cap055 | LBS | San Martín | 6°28′ S | 76°20′ W |
| Cluster | N | H | Lambda | He | PIC | PPL |
|---|---|---|---|---|---|---|
| 1 | 6 | 1.79 | 0.83 | 0.12 | 0.20 | 51.08% |
| 2 | 18 | 2.89 | 0.94 | 0.3 | 0.29 | 83.33% |
| 3 | 19 | 2.94 | 0.95 | 0.41 | 0.38 | 96.24% |
| 4 | 16 | 2.77 | 0.94 | 0.29 | 0.20 | 83.33% |
| Total | 59 | 4.08 | 0.98 | 0.41 |
| Cluster | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| 1 | ||||
| 2 | 0.182 | |||
| 3 | 0.198 | 0.125 | ||
| 4 | 0.269 | 0.167 | 0.123 |
| Source | df | SS | MS | Est.Var. | % |
|---|---|---|---|---|---|
| Between clusters | 3 | 584.14 | 194.71 | 11.69 | 28.44% |
| Within clusters | 55 | 1617.01 | 29.40 | 29.4 | 71.56% |
| Total | 58 | 2201.15 | 37.95 | 41.09 | 100.00% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saldaña, C.L.; Cancan, J.D.; Cruz, W.; Correa, M.Y.; Ramos, M.; Cuellar, E.; Arbizu, C.I. Genetic Diversity and Population Structure of Capirona (Calycophyllum spruceanum Benth.) from the Peruvian Amazon Revealed by RAPD Markers. Forests 2021, 12, 1125. https://doi.org/10.3390/f12081125
Saldaña CL, Cancan JD, Cruz W, Correa MY, Ramos M, Cuellar E, Arbizu CI. Genetic Diversity and Population Structure of Capirona (Calycophyllum spruceanum Benth.) from the Peruvian Amazon Revealed by RAPD Markers. Forests. 2021; 12(8):1125. https://doi.org/10.3390/f12081125
Chicago/Turabian StyleSaldaña, Carla L., Johan D. Cancan, Wilbert Cruz, Mirian Y. Correa, Miriam Ramos, Eloy Cuellar, and Carlos I. Arbizu. 2021. "Genetic Diversity and Population Structure of Capirona (Calycophyllum spruceanum Benth.) from the Peruvian Amazon Revealed by RAPD Markers" Forests 12, no. 8: 1125. https://doi.org/10.3390/f12081125
APA StyleSaldaña, C. L., Cancan, J. D., Cruz, W., Correa, M. Y., Ramos, M., Cuellar, E., & Arbizu, C. I. (2021). Genetic Diversity and Population Structure of Capirona (Calycophyllum spruceanum Benth.) from the Peruvian Amazon Revealed by RAPD Markers. Forests, 12(8), 1125. https://doi.org/10.3390/f12081125







