Cold Acclimation Increases Freeze Tolerance in Acacia koa, a Tropical Tree Species Occurring over a Wide Elevational Gradient
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Acclimation Treatments
2.3. Freezing Tests and Conductivity Measurements
2.4. Statistical Analyses
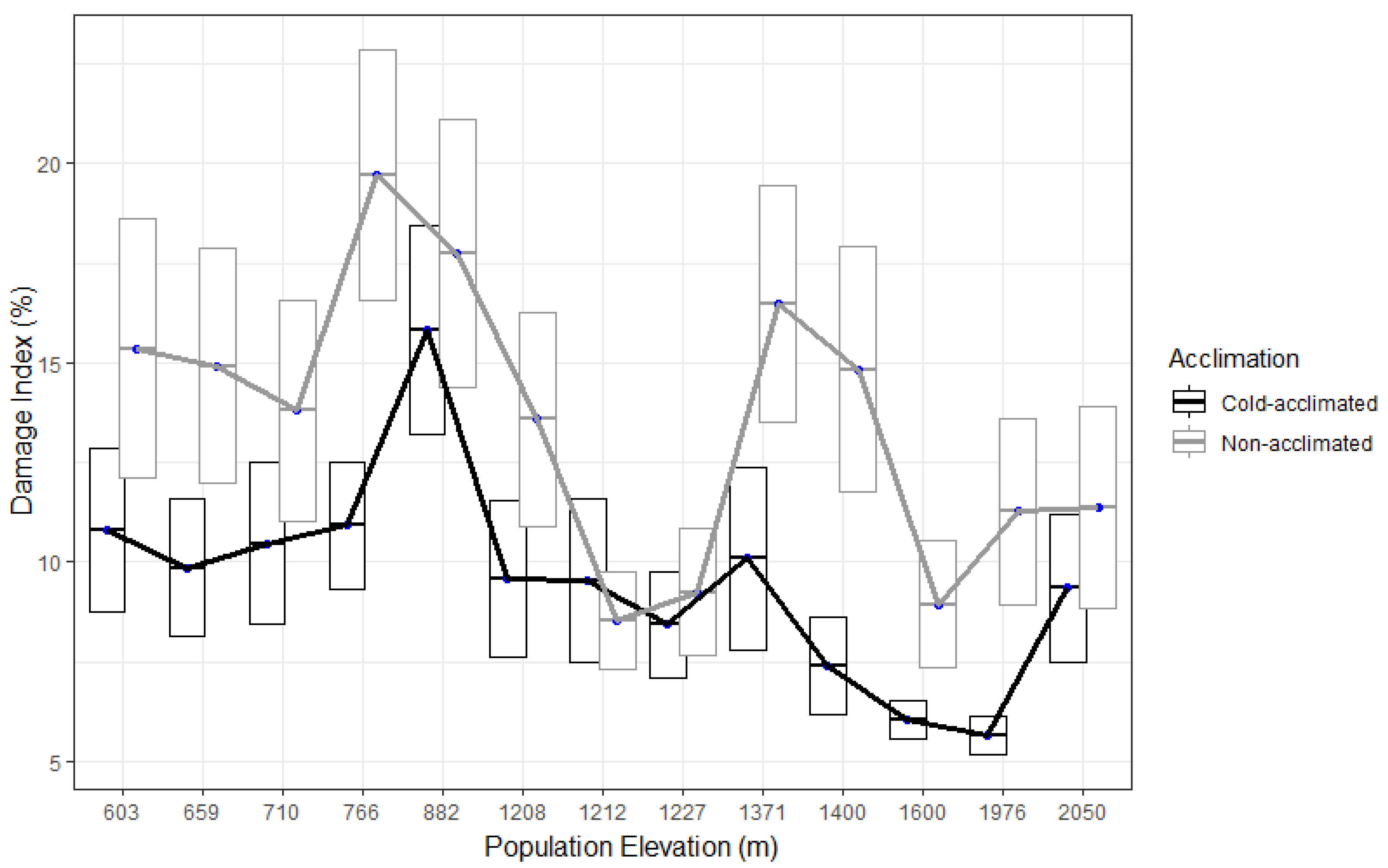
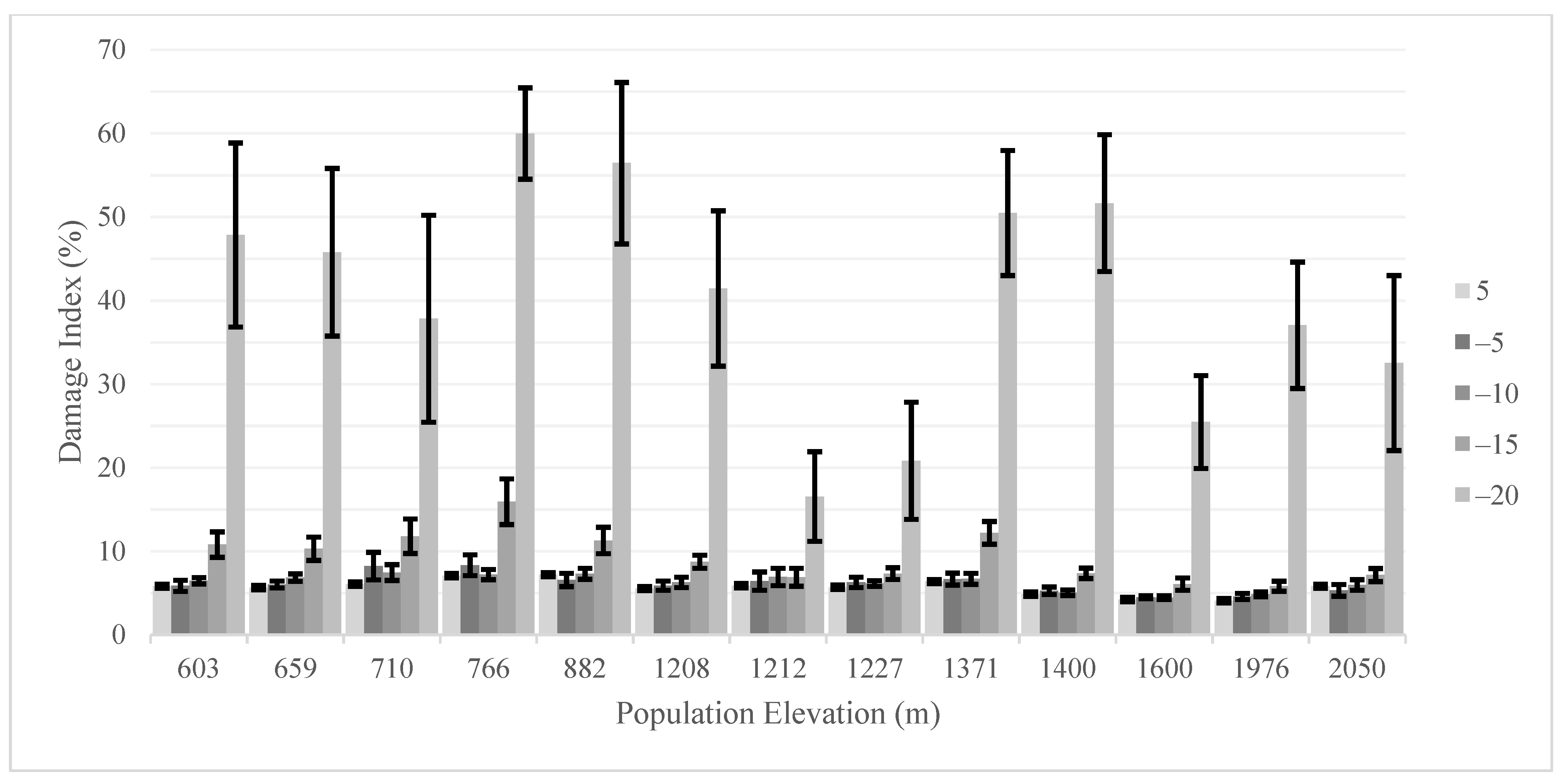
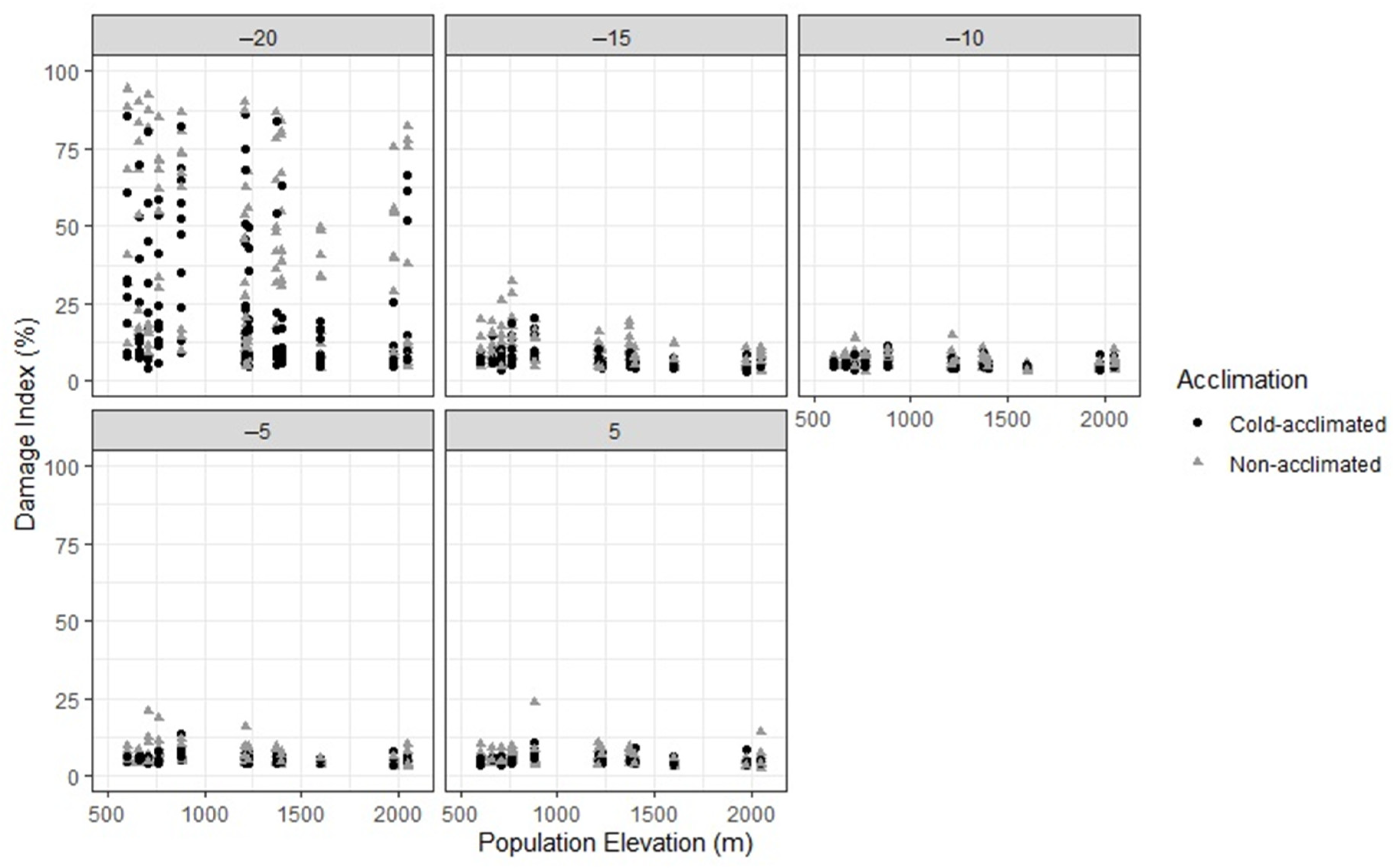
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rose, R.; Carlson, W.C.; Morgan, P. The Target Seedling Concept. In Proceedings of the Target Seedling Symposium, Combined Meeting of the Western Forest Nursery Assoc. Gen. Tech. Rep. RM-200, Roseburg, OR, USA, 13–17 August 1990; Rose, R., Campbell, S.J., Landis, T.D., Eds.; US Dept Agric Forest Serv Rocky Mtn Forest Range Exp Sta: Ft Collins, CO, USA, 1990. 286p. [Google Scholar]
- Landis, T.D.; Dumroese, R.K. Applying the target plant concept to nursery stock quality. In Plant Quality: A Key to Success in Forest Establishment; MacLennan, L., Fennessy, J., Eds.; National Council Forest Res Develop: Dublin, Ireland, 2006; pp. 1–10. [Google Scholar]
- Dumroese, R.K.; Landis, T.D.; Pinto, J.R.; Haase, D.L.; Wilkinson, K.W.; Davis, A.S. Meeting forest restoration challenges: Using the target plant concept. Reforesta 2016, 1, 37–52. [Google Scholar] [CrossRef] [Green Version]
- Warrington, I.J.; Rook, D.A. Evaluation of techniques used in determining frost tolerance of forest planting stock: A review. N. Z. J. For. Sci. 1980, 10, 116–132. [Google Scholar]
- Krasowski, M.J.; Simpson, D.G. Frost-Related Problems in the Establishment of Coniferous Forests. In Conifer Cold Hardiness; Bigras, F.J., Colombo, S.J., Eds.; Springer: Dordrecht, The Netherlands, 2001; pp. 253–285. [Google Scholar] [CrossRef]
- Wallin, E.; Gräns, D.; Jacobs, D.F.; Lindström, A.; Verhoef, N. Short-day photoperiods affect expression of genes related to dormancy and freezing tolerance in Norway spruce seedlings. Annals For. Sci. 2017, 74, 59. [Google Scholar] [CrossRef] [Green Version]
- Malmqvist, C.; Wallertz, K.; Johansson, U. Survival, early growth and impact of damage by late-spring frost and winter desiccation on Douglas-fir seedlings in southern Sweden. New For. 2018, 49, 723–736. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilkinson, K.M.; Elevitch, C.R. Growing Koa, a Hawaiian Legacy Tree, 1st ed.; Permanent Agriculture Resources (PAR): Holualoa, HI, USA, 2003. [Google Scholar]
- Baker, P.J.; Scowcroft, P.G.; Ewel, J.J. Koa (Acacia koa) Ecology and Silviculture, General Technical Report; United States Department of Agriculture Forest: Washington, DC, USA, 2009.
- Scott, J.M.; Mountainspring, S.; Ramsey, F.L.; Kepler, C.B. Forest bird communities of the Hawaiian Islands: Their dynamics, ecology, and conservation. Stud. Avian Biol. 1986, 9, 1–431. [Google Scholar]
- Little, E.L.; Skolmen, R.G. Common Forest Trees of Hawaii (Native and Introduced). In USDA Forest Service Agriculture Handbook No; U.S. Department of Agriculture: Washington, DC, USA, 1989; Volume 679. [Google Scholar]
- Yanagida, J.F.; Friday, J.B.; Illukpitiya, P.; Mamiit, R.J.; Edwards, Q. Economic value of Hawai’i’s forest industry in 2001. Econ. Issues 2004, 7, 1–3. [Google Scholar]
- Scowcroft, P.G.; Jeffrey, J. Potential significance of frost, topographic relief, and Acacia koa stands to restoration of mesic Hawaiian forests on abandoned rangeland. For. Ecol. Manag. 1999, 114, 447–458. [Google Scholar] [CrossRef]
- Friday, J.B.; Cordell, S.; Giardina, C.P.; Inman-Narahari, F.; Koch, N.; Leary, J.J.K.; Litton, C.M.; Trauernicht, C. Future directions for forest restoration in Hawaii. New For. 2015, 46, 733–746. [Google Scholar] [CrossRef]
- Pejchar, L.; Press, D.M. Achieving conservation objectives through production forestry: The case of Acacia koa on Hawaii Island. Environ. Sci. Policy 2006, 9, 439–447. [Google Scholar] [CrossRef]
- Wagner, W.L.; Herbst, D.R.; Sohmer, H.S. Manual of the Flowering Plants of Hawaii; Bernice, P., Ed.; University of Hawaii Press: Bishop Museum Press: Honolulu, HI, USA, 1990. [Google Scholar]
- Mueller-Dombois, D. Forest dynamics in Hawai’i. Trends Ecol. Evol. 1987, 2, 216–220. [Google Scholar] [CrossRef]
- Elevitch, C.R.; Wilkinson, K.M.; Friday, J.B. Acacia koa (koa) and Acacia koaia (koai‘a). In Species Profiles for Pacific Island Agroforestry; Elevitch, C.R., Ed.; Permanent Agriculture Resources (PAR): Holualoa, HI, USA, 2006. [Google Scholar]
- Dudley, N.S.; Jones, T.C.; James, R.L.; Sniezko, R.A.; Cannon, P.; Borthakur, D. Applied disease screening and selection program for resistance to vascular wilt in Hawaiian Acacia koa. South. For. 2015, 77, 65–73. [Google Scholar] [CrossRef]
- Scowcroft, P.G.; Meinzer, F.C.; Goldstein, G.; Melcher, P.J.; Jeffrey, J. Moderating night radiative cooling reduces frost damage to Metrosideros polymorpha seedlings used for forest restoration in Hawaii. Rest. Ecol. 2000, 8, 161–169. [Google Scholar] [CrossRef]
- Rose, K.M.E.; Friday, J.B.; Oliet, J.A.; Jacobs, D.F. Canopy openness affects microclimate and performance of underplanted trees in restoration of high-elevation tropical pasturelands. Agric. For. Meterol. 2020, 292–293, 108105. [Google Scholar] [CrossRef]
- National Climatic Data Center, n.d. Hawaii Annual Temperatures and Extremes. Available online: http://coolweather.net/statetemperature/hawaii_temperature.htm (accessed on 4 May 2019).
- Western Regional Climate Center, n.d. Climate of Hawaii. Available online: https://wrcc.dri.edu/narratives/HAWAII.htm (accessed on 4 May 2019).
- Allen, J.A. Acacia koa A. Gray. In Tropical Tree Seed Manual: Part II, Species Descriptions; U.S. Department of Agriculture: Washington, DC, USA, 2002; pp. 253–255. [Google Scholar]
- Lipp, C.C.; Goldstein, G.; Meinzer, F.C.; Niemczura, W. Freezing tolerance and avoidance in high-elevation Hawaiian plants. Plant Cell Environ. 1994, 17, 1035–1044. [Google Scholar] [CrossRef]
- University of Hawai’i at Manoa. Mean Annual Air Temperature-State of Hawaii. 2014. Available online: http://evapotranspiration.geography.hawaii.edu/assets/files/MapImages/AirTemp_MeanAnn.jpg (accessed on 23 October 2018).
- Rueda Krauss, O. Parent Tree Selection and Evaluation of Frost Resistance, Wood Quality, and Seed Relatedness of Acacia koa. Open Access Theses 245. 2014. Available online: http://docs.lib.purdue.edu/open_access_theses/245 (accessed on 19 June 2018).
- Costa e Silva, F.; Shvaleva, A.; Broetto, F.; Ortuño, M.F.; Rodrigues, M.L.; Almeida, M.H.; Chaves, M.M. and Pereira, J.S. Acclimation to short-term low temperatures in two Eucalyptus globulus clones with contrasting drought resistance. Tree Physiol. 2009, 29, 77–86. [Google Scholar] [CrossRef]
- Zuther, E.; Schulz, E.; Childs, L.H.; Hincha, D.K. Clinal variation in the non-acclimated and cold-acclimated freezing tolerance of Arabidopsis thaliana accessions. Plant Cell Environ. 2012, 35, 1860–1878. [Google Scholar] [CrossRef]
- Ritchie, G.A. Measuring Cold Hardiness. In Techniques and Approaches in Forest Tree Ecophysiology; Lassoie, J.P., Hinckley, T.M., Eds.; CRC Press: Boca Raton, FL, USA, 1991; pp. 557–582. [Google Scholar]
- Haase, D.L. Seedling phenology and cold hardiness: Moving targets. In National Proceedings: Forest and Conservation Nursery Associations-2010; Riley, L.E., Haase, D.L., Pinto, J.R., Eds.; Proc. RMRS-P-65; USDA Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA, 2011; pp. 121–127. [Google Scholar]
- Jacobs, D.F.; Wilson, B.C.; Ross-Davis, A.L.; Davis, A.S. Cold hardiness and transplant response of Juglans nigra seedlings subjected to alternative storage regimes. Ann. For. Sci. 2008, 65, 606. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.M.; Walker, S.C. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67. [Google Scholar] [CrossRef]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82. [Google Scholar] [CrossRef] [Green Version]
- Loik, M.E.; Redar, S.P. Microclimate, freezing tolerance, and cold acclimation along an elevation gradient for seedlings of the Great Basin Desert shrub, Artemisia tridentata. J. Arid Environ. 2003, 54, 769–782. [Google Scholar] [CrossRef] [Green Version]
- Melcher, P.J.; Cordell, S.; Jones, T.J.; Scowcroft, P.G.; Niemczura, W.; Giambelluca, T.W.; Goldstein, G. Supercooling capacity increases from sea level to tree line in the Hawaiian tree species Metrosideros polymorpha. Int. J. Plant Sci. 2000, 161, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Hadley, J.L.; Amundson, R.G. Effects of radiational heating at low air temperature on water balance, cold tolerance, and visible injury of red spruce foliage. Tree Physiol. 1992, 11, 1–17. [Google Scholar] [CrossRef]
- Sutinen, M.-L.; Palta, J.P.; Reich, P.B. Seasonal differences in freezing stress resistance of needles of Pinus nigra and Pinus resinosa: Evaluation of the electrolyte leakage method. Tree Physiol. 1992, 11, 241–254. [Google Scholar] [CrossRef] [Green Version]
- Islam, M.A.; Apostol, K.G.; Jacobs, D.F.; Dumroese, R.K. Fall fertilization of Pinus resinosa seedlings: Nutrient uptake, cold hardiness, and morphological development. Ann. For. Sci. 2009, 66, 7. [Google Scholar] [CrossRef] [Green Version]
- Palta, J.P.; Jensen, K.G.; Li, P.H. Cell membrane alterations following a slow freeze-thaw cycle: Ion leakage, injury and recovery. In Plant Cold Hardiness and Freezing Stress. Mechanisms and Crop Implications; Li, P.H., Sakai, A., Eds.; Academic Press: New York, NY, USA, 1982; Volume 2, pp. 221–242. [Google Scholar]
- Peixoto, M.M.; Friesen, P.C.; Sage, R.F. Winter cold-tolerance thresholds in field-grown Miscanthus hybrid rhizomes. J. Exp. Bot. 2015, 66, 4415–4425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peixoto, M.D.M.; Sage, R.F. Improved experimental protocols to evaluate cold tolerance thresholds in Miscanthus and switchgrass rhizomes. GCB Bioenergy 2016, 8, 257–268. [Google Scholar] [CrossRef] [Green Version]


| Population | Elevation (m) | Annual Rainfall (mm) | Coordinates | Land Cover | |
|---|---|---|---|---|---|
| Latitude | Longitude | ||||
| BK-Saddle 1 | 603 | 5707 | 19.67932° N, | 155.182° W | Lowland rainforest |
| BK-Saddle 2 | 659 | 5733 | 19.67932° N, | 155.1899° W | Lowland rainforest |
| BK-Saddle 3 | 710 | 5779 | 19.69229° N, | 155.2015° W | Lowland rainforest |
| BK-Saddle 4 | 766 | 5903 | 19.69476° N, | 155.2144° W | Lowland rainforest |
| BK-Saddle 5 | 882 | 6227 | 19.70094° N, | 155.2353° W | Lowland rainforest |
| BK-MLSR 3 | 1208 | 1871 | 19.43908° N, | 155.3005° W | Montane-subalpine mesic forest |
| BK-MLSR 2 | 1212 | 1846 | 19.43338° N, | 155.2957° W | Montane-subalpine dry shrubland |
| BK-MLSR 1 | 1227 | 2036 | 19.43358° N, | 155.2811° W | Montane-subalpine mesic forest |
| Gaspar’s Dairy | 1371 | 892 | 19.48653° N, | 155.8261° W | Montane-subalpine mesic forest |
| Kona Hema | 1400 | 809 | 19.19974° N, | 155.795° W | Montane-subalpine mesic forest |
| Umikoa | 1600 | 1992 | 19.92584° N, | 155.3476° W | Introduced perennial grassland |
| BIR | 1976 | 1593 | 19.8569° N, | 155.3401° W | Perennial grassland |
| BK-MLSR 4 | 2050 | 1631 | 19.49243° N, | 155.3865° W | Montane-Subalpine Dry Shrubland |
| Conditions | Photoperiod (Hours) | Temp. Day (°C) | Temp. Night (°C) | RH (%) | Watering Interval (Days) | Light Intensity (μmol/m2/s) |
|---|---|---|---|---|---|---|
| Greenhouse | 14 | ~25.5 | ~22.2 | <~80 | 1–2 | ~500 |
| Week 1 growth chamber | 11 | 15 | 10 | ~80–85 | 4 | 200 |
| Week 2 growth chamber | 11 | 12 | 6 | ~80–85 | 10 | 200 |
| Week 3 growth chamber | 11 | 8 | 4 | ~80–85 | 10 | 200 |
| Week 4 growth chamber | 11 | 8 | 4 | ~80–85 | 10 | 200 |
| Week 5 growth chamber | 11 | 8 | 4 | ~80–85 | 10 | 200 |
| Week 6 growth chamber | 11 | 8 | 4 | ~80–85 | 10 | 200 |
| Fixed Effects | Estimate | Std. Error | df | t Value | Pr(>|t|) |
|---|---|---|---|---|---|
| (Intercept) | 7.961 × 10−1 | 3.048 × 10−2 | 3.183 × 10 | 26.123 | <0.001 |
| Population elevation | −7.400 × 10−5 | 1.889 × 10−5 | 1.266 × 103 | −3.917 | <0.001 |
| Test temperature | −3.106 × 10−2 | 2.086 × 10−3 | 1.266 × 103 | −14.888 | <0.001 |
| Acclimation | −9.003 × 10−3 | 2.838 × 10−2 | 6.115 × 10 | −0.317 | 0.762 |
| (Test temperature)2 | 4.658 × 10 | 2.149 × 10−1 | 1.266 × 103 | 21.672 | <0.001 |
| Pop. elev. × Test temperature | 5.472 × 10−6 | 1.519 × 10−6 | 1.266 × 103 | 3.603 | <0.001 |
| Test temperature × Acclimation | 8.225 × 10−3 | 1.398 × 10−3 | 1.266 × 103 | 5.885 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ayala-Jacobo, L.M.; Woeste, K.E.; Jacobs, D.F. Cold Acclimation Increases Freeze Tolerance in Acacia koa, a Tropical Tree Species Occurring over a Wide Elevational Gradient. Forests 2021, 12, 1089. https://doi.org/10.3390/f12081089
Ayala-Jacobo LM, Woeste KE, Jacobs DF. Cold Acclimation Increases Freeze Tolerance in Acacia koa, a Tropical Tree Species Occurring over a Wide Elevational Gradient. Forests. 2021; 12(8):1089. https://doi.org/10.3390/f12081089
Chicago/Turabian StyleAyala-Jacobo, Lilian M., Keith E. Woeste, and Douglass F. Jacobs. 2021. "Cold Acclimation Increases Freeze Tolerance in Acacia koa, a Tropical Tree Species Occurring over a Wide Elevational Gradient" Forests 12, no. 8: 1089. https://doi.org/10.3390/f12081089
APA StyleAyala-Jacobo, L. M., Woeste, K. E., & Jacobs, D. F. (2021). Cold Acclimation Increases Freeze Tolerance in Acacia koa, a Tropical Tree Species Occurring over a Wide Elevational Gradient. Forests, 12(8), 1089. https://doi.org/10.3390/f12081089






