Seasonal Changes and the Interaction between the Horse Chestnut Leaf Miner Cameraria ohridella and Horse Chestnut Leaf Blotch Disease Caused by Guignardia aesculi
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Sampling of Horse Chestnut Leaves
2.3. Measurement of Proportion of Leaf Area Damaged by Leaf Miner and Fungus
2.4. Data Presentation and Statistical Analysis
2.5. Modeling Dynamics of Leaf Damage Caused by Cameraria ohridella and Guignardia aesculi
2.5.1. The Model
2.5.2. Estimation of Model Parameter Values
2.5.3. Evaluating the Model as a Predictive Tool
3. Results
3.1. Damage to A. hippocastanum Leaves during the Vegetation Period
3.2. Modeling Leaf Damage Caused by Cameraria ohridella and Guignardia aesculi
3.3. The Model as a Predictive Tool
4. Discussion
4.1. Damage to the Leaf Area of A. hippocastanum during the Vegetation Period
4.2. Interaction between C. ohridella and G. aesculi on the Leaves of Horse Chestnut Trees
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thomas, P.A.; Alhamd, O.; Iszkuło, G.; Dering, M.; Mukassabi, T.A. Biological Flora of the British Isles: Aesculus Hippocastanum. J. Ecol. 2019, 107, 992–1030. [Google Scholar] [CrossRef]
- Somme, L.; Moquet, L.; Quinet, M.; Vanderplanck, M.; Michez, D.; Lognay, G.; Jacquemart, A.-L. Food in a Row: Urban Trees Offer Valuable Floral Resources to Pollinating Insects. Urban Ecosyst. 2016, 19, 1149–1161. [Google Scholar] [CrossRef]
- Tuovinen, T. Influence of Surrounding Trees and Bushes on the Phytoseiid Mite Fauna on Apple Orchard Trees in Finland. Agric. Ecosyst. Environ. 1994, 50, 39–47. [Google Scholar] [CrossRef]
- Kabíček, J.; Řeháková, M. Phytoseiid Mite Community on Aesculus Hippocastanum in the Parks. Acta Fytotech. Zootech. 2004, 7, 114–115. [Google Scholar]
- Kopačka, M.; Stathakis, T.I.; Broufas, G.; Papadoulis, G.T.; Zemek, R. Diversity and Abundance of Phytoseiidae (Acari: Mesostigmata) on Horse Chestnut (Aesculus Hippocastanum L.) in an Urban Environment: A Comparison between Greece and the Czech Republic. Acarologia 2018, 58, 8390. [Google Scholar] [CrossRef]
- Deschka, G.; Dimić, N. Cameraria Ohridella Sp. n. (Lep., Lithocolletidae) from Macedonia, Yugoslavia. Acta Entomol. Jugosl. 1986, 22, 11–23. [Google Scholar]
- Grabenweger, G.; Grill, R. On the Place of Origin of Cameraria Ohridella Deschka & Dimic (Lepidoptera: Gracillariidae). Beitr. Z. Entomofaunist. 2000, 1, 9–17. [Google Scholar]
- Liška, J. Verbreitung der Rosskastanienminiermotte in Tschechien. Forstsch. Aktuell 1997, 21, 1. [Google Scholar]
- Wittenberger, G. Die Roßkastanien-Miniermotte in Nordböhmen nebst einigen Hinweisen zum Vorkommen in Schlesien (Polen). Nassau. Ver. Für Nat. 1998, 98, 75–78. [Google Scholar]
- Šefrová, H.; Laštůvka, Z. Dispersal of the Horse-Chestnut Leafminer, Cameraria Ohridella Deschka & Dimic, 1986, in Europe: Its Course, Ways and Causes (Lepidoptera: Gracillariidae). Entomol. Z. 2001, 111, 194–198. [Google Scholar]
- Cebeci, H.; Acer, S. The Occurrence of Some Lepidopterous Species on the Horse Chestnut (Aesculus Hippocastanum L.) at Istanbul-Belgrad Forest in Turkey. Acta Agric. Slov. 2007, 89. [Google Scholar] [CrossRef]
- Péré, C.; Augustin, S.; Turlings, T.C.J.; Kenis, M. The Invasive Alien Leaf Miner Cameraria Ohridella and the Native Tree Acer Pseudoplatanus: A Fatal Attraction? Agric. For. Entomol. 2010, 12, 151–159. [Google Scholar] [CrossRef]
- Skuhravý, V. Zusammenfassende Betrachtung der Kenntnisse über die Roßkastanien-miniermotte, Cameraria ohridella Desch. & Dem. (Lep., Gracillariidae). J. Pest Sci. 1999, 72, 95–99. [Google Scholar] [CrossRef]
- Samek, T. Diapause of Cameraria Ohridella Deschka et Dimic and Its Impact on the Species Population Dynamics. J. For. Sci. 2012, 49, 252–258. [Google Scholar] [CrossRef]
- Weryszko-Chmielewska, E.; Haratym, W. Changes in Leaf Tissues of Common Horse Chestnut (Aesculus Hippocastanum L.) Colonised by the Horse-Chestnut Leaf Miner (Cameraria Ochridella Deschka and Dimić). Acta Agrobot. 2011, 64, 11–22. [Google Scholar] [CrossRef]
- Konarska, A.; Grochowska, M.; Haratym, W.; Tietze, M.; Weryszko-Chmielewska, E.; Lechowski, L. Changes in Aesculus Hippocastanum Leaves during Development of Cameraria Ohridella. Urban For. Urban Green. 2020, 56, 126793. [Google Scholar] [CrossRef]
- Thalmann, C.; Freise, J.; Heitland, W.; Bacher, S. Effects of Defoliation by Horse Chestnut Leafminer (Cameraria Ohridella) on Reproduction in Aesculus Hippocastanum. Trees Struct. Funct. 2003, 17, 383–388. [Google Scholar] [CrossRef]
- Nardini, A.; Raimondo, F.; Scimone, M.; Salleo, S. Impact of the Leaf Miner Cameraria Ohridella on Whole-Plant Photosynthetic Productivity of Aesculus Hippocastanum: Insights from a Model. Trees 2004, 18, 714–721. [Google Scholar] [CrossRef]
- Salleo, S.; Nardini, A.; Raimondo, F.; Lo Gullo, M.A.; Pace, F.; Giacomich, P. Effects of Defoliation Caused by the Leaf Miner Cameraria Ohridella on Wood Production and Efficiency in Aesculus Hippocastanum Growing in North-Eastern Italy. Trees 2003, 17, 367–375. [Google Scholar] [CrossRef]
- Pastirčáková, K.; Pastirčák, M.; Celar, F.; Shin, H.-D. Guignardia Aesculi on Species of Aesculus: New Records from Europe and Asia. Mycotaxon 2009, 108, 287–296. [Google Scholar] [CrossRef]
- Ianovici, N.; Ciocan, G.V.; Matica, A.; Scurtu, M.; Sesan, T.E. Study on the infestation by Cameraria ohridella on Aesculus hippocastanum foliage from timisoara, Romania. Ann. West Univ. Timiş. 2012, 15, 67–80. [Google Scholar]
- Scaramuzzi, G. Sul Seccume Delle Foglie d’ippocastano. Ann. Sper. Agric. 1954, 8, 1256–1281. [Google Scholar]
- Zimmermannová-Pastirčáková, K.; Janitor, A. Podiel fytopatogénnych húb pri usýchaní a odumieraní listov pagaštana konského (Aesculus hippocastanum L.). In Sborník XV. Českej a Slovenskej Konferencie o Ochrane Rastlín; Mendelova zemědelská a lesnická univerzita: Brno, Czech Republic, 2000; pp. 445–446. [Google Scholar]
- Zimmermannová, K. Houbové ochorenie listov pagaštana konského (Aesculus hippocastanum L.) a jeho výskyt na Slovensku. Folia Oecologica 2001, 28, 153–165. [Google Scholar]
- Bissett, J.; Darbyshire, S.J. Phyllosticta Sphaeropsoidea. Fungi Canadenses 1984, 280, 1–2. [Google Scholar]
- Chen, P.C.; Li, Y.Z.; Xu, Y.; Chi, X.Z.; Yan, W.; Ju, R.T. Main Pests in Imported Colored Arbors and the Occurrence. For. Pest Dis. 2007, 26, 31–34. [Google Scholar]
- Pastirčáková, K. Guignardia aesculi (Peck) Stewart—Fungal pathogen on Aesculus leaves in Slovakia. Acta Fytotech. Zootech. 2004, 7, 234–236. [Google Scholar]
- Kopačka, M.; Zemek, R. The Effect of Microclimatic Conditions on Guignardia Aesculi Infecting Horse Chestnut (Aesculus Hippocastanum) Trees. In Proceedings of the Dendrological Days in Mlyňany Arboretum SAS, Vieska nad Žitavou, Slovakia, 18–19 September 2012; pp. 18–19. [Google Scholar]
- Kobza, M.; Juhásová, G.; Adamčíková, K.; Onrušková, E. Tree Injection in the Management of Horse-Chestnut Leaf Miner, Cameraria Ohridella (Lepidoptera: Gracillariidae). Gesunde Pflanz. 2011, 62, 139–143. [Google Scholar] [CrossRef]
- Berger, C.; Laurent, F. Trunk Injection of Plant Protection Products to Protect Trees from Pests and Diseases. Crop Prot. 2019, 124, 104831. [Google Scholar] [CrossRef]
- Percival, G.C.; Holmes, S.P. The Influence of Systemic Inducing Agents on Horse Chestnut Leaf Miner (Cameraria Ohridella) Severity in White Flowering Horse Chestnut (Aesculus Hoppicastanum L.). Urban For. Urban Green. 2016, 20, 97–102. [Google Scholar] [CrossRef]
- Percival, G.C.; Banks, J.; Keary, I. Evaluation of Organic, Synthetic and Physical Insecticides for the Control of Horse Chestnut Leaf Miner (Cameraria Ohridella). Urban For. Urban Green. 2012, 11, 426–431. [Google Scholar] [CrossRef]
- Mösch, S.; Eilers, E.J.; Hommes, M. Biocontrol of Cameraria Ohridella by Insectivorous Birds in Different Landscape Contexts. BioControl 2018, 63, 215–225. [Google Scholar] [CrossRef]
- Klug, T.; Meyhöfer, R.; Kreye, M.; Hommes, M. Native Parasitoids and Their Potential to Control the Invasive Leafminer, Cameraria Ohridella DESCH. & DIM. (Lep.: Gracillariidae). Bull. Entomol. Res. 2008, 98, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Kehrli, P.; Lehmann, M.; Bacher, S. Mass-Emergence Devices: A Biocontrol Technique for Conservation and Augmentation of Parasitoids. Biol. Control 2005, 32, 191–199. [Google Scholar] [CrossRef]
- Zemek, R.; Prenerová, E.; Volter, L.; Weyda, F.; Skuhravý, V. Perspectives for the Biological Control of Cameraria Ohridella. Commun. Agric. Appl. Biol. Sci. 2007, 72, 521–526. [Google Scholar]
- Prenerová, E.; Zemek, R.; Volter, L.; Weyda, F. Strain of Entomopathogenic Fungus Isaria Fumosorosea CCM 8367 (CCEFO.011.PFR) and the Method for Controlling Insect and Mite Pests. EPO Patent EP2,313,488, 29 April 2015. [Google Scholar]
- Barta, M. In Planta Bioassay on the Effects of Endophytic Beauveria Strains against Larvae of Horse-Chestnut Leaf Miner (Cameraria Ohridella). Biol. Control 2018, 121, 88–98. [Google Scholar] [CrossRef]
- Richter, D.; Sermann, H.; Jackel, B.; Buttner, C.B. Pathogenicity of Entomopathogenic Fungi on Hibernating Pupae of Cameraria Ohridella Deschka & Dimic 1986 (Lepidoptera, Gracillariidae). Part 1: Pathogenicity against the Naked Pupa. Commun. Agric. Appl. Biol. Sci. 2007, 72, 399–410. [Google Scholar] [PubMed]
- Richter, D.; Sermann, H.; Jackel, B.; Buttner, C. Pathogenicity of Entomopathogenic Fungi on Hibernating Pupae of Cameraria Ohridella Deschka & Dimic 1986 (Lepidoptera, Gracillariidae). Part 2: Efficacy of Entomopathogenic Fungi against Pupa in Her Pupal Cell. Commun. Agric. Appl. Biol. Sci. 2007, 72, 411–421. [Google Scholar]
- Kalmus, M.; Sermann, H.; Buettner, C. Efficacy of Entomopathogenic Fungi against Eggs and Larvae of the Horse Chestnut Leafminer Cameraria Ohridella. J. Plant Dis. Prot. 2007, 114, 92. [Google Scholar]
- Nedveckytė, I.; Pečiulytė, D.; Būda, V. Fungi Associated with Horse-Chestnut Leaf Miner Moth Cameraria Ohridella Mortality. Forests 2021, 12, 58. [Google Scholar] [CrossRef]
- Robert, S.; Petra, C.; Juraj, M.; Marek, B. Natural Prevalence of Entomopathogenic Fungi in Hibernating Pupae of Cameraria Ohridella (Lepidoptera: Gracillariidae) and Virulence of Selected Isolates. Plant Prot. Sci. 2016, 52, 199–208. [Google Scholar] [CrossRef]
- Matuska-Lyzwa, J.; Kaca, W.; Zarnowiec, P. Biological Activity of Wild Isolates of Entomopathogenic Nematodes to Horse-Chestnut Leaf Miner (Cameraria Ohridella). Pol. J. Environ. Stud. 2015, 24, 1181–1184. [Google Scholar] [CrossRef]
- Pastirčáková, K. In Vitro Interactions between Trichoderma Harzianum and Pathogenic Fungi Damaging Horse-Chestnut (Aesculus Hippocastanum) Leaves and Fruits. Biol. Lett. 2019, 54, 21–35. [Google Scholar] [CrossRef][Green Version]
- Baraniak, E.; Walczak, U.; Tryjanowski, P.; Zduniak, P. Effect of Distance between Host Trees and Leaf Litter Removal on Population Density of Cameraria Ohridella Deschka & Dimic, 1986 (Lepidoptera, Gracillariidae)—Pest of Chestnut (Aesculus Sp.) Trees. Pol. J. Ecol. 2004, 52, 569–574. [Google Scholar]
- Kehrli, P.; Bacher, S. Date of Leaf Litter Removal to Prevent Emergence of Cameraria Ohridella in the Following Spring. Entomol. Exp. Appl. 2003, 107, 159–162. [Google Scholar] [CrossRef]
- Flückiger, W.; Braun, S. Stress Factors of Urban Trees and Their Relevance for Vigour and Predisposition for Parasite Attacks. Acta Hortic. 1999, 325–334. [Google Scholar] [CrossRef]
- Gilbert, M.; Svatoš, A.; Lehmann, M.; Bacher, S. Spatial Patterns and Infestation Processes in the Horse Chestnut Leafminer Cameraria Ohridella: A Tale of Two Cities. Entomol. Exp. Appl. 2003, 107, 25–37. [Google Scholar] [CrossRef]
- Bhatti, I.; Ozanne, C.; Shaw, P. Parasitoids and Parasitism Rates of the Horse Chestnut Leaf Miner Cameraria Ohridella Deschka and Dimić [Lepidoptera: Gracillariidae] across Four Sites in South-West London. Arboric. J. 2013, 35, 147–159. [Google Scholar] [CrossRef]
- Jagiełło, R.; Baraniak, E.; Karolewski, P.; Łakomy, P.; Behnke-Borowczyk, J.; Walczak, U.; Giertych, M.J. Ecophysiological Aspects of the Interaction between Cameraria Ohridella and Guignardia Aesculi on Aesculus Hippocastanum. Dendrobiology 2017, 78, 146–156. [Google Scholar] [CrossRef]
- Jagiełło, R.; Łakomy, P.; Łukowski, A.; Giertych, M.J. Spreading-the-Risk Hypothesis May Explain Cameraria Ohridella Oviposition in Relation to Leaf Blotch Disease. Arthropod-Plant Interact. 2019, 13, 787–795. [Google Scholar] [CrossRef]
- Johne, A.B.; Weissbecker, B.; Schütz, S. Approaching Risk Assessment of Complex Disease Development in Horse Chestnut Trees: A Chemical Ecologist’s Perspective. J. Appl. Entomol. 2008, 132, 349–359. [Google Scholar] [CrossRef]
- Kopacka, M.; Zemek, R. Spatial Variability in the Level of Infestation of the Leaves of Horse Chestnut by the Horse Chestnut Leaf Miner, Cameraria Ohridella (Lepidoptera: Gracillariidae) and in the Number of Adult Moths and Parasitoids Emerging from Leaf Litter in an Urban Environment. Eur. J. Entomol. 2017, 114, 42–52. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 Years of Image Analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Škaloudová, B.; Křivan, V.; Zemek, R. Computer-Assisted Estimation of Leaf Damage Caused by Spider Mites. Comput. Electron. Agric. 2006, 53, 81–91. [Google Scholar] [CrossRef]
- The Java Language Specification, 3rd ed.; Java Series; Gosling, J., Ed.; Addison-Wesley: Upper Saddle River, NJ, USA, 2005; ISBN 978-0-321-24678-3. [Google Scholar]
- Sokal, R.R.; Rohlf, F.J. Biometry: The Principles and Practice of Statistics in Biological Research; Freeman: San Francisco, CA, USA, 1969; ISBN 978-0-7167-0663-2. [Google Scholar]
- Zar, J.H. Biostatistical Analysis, 5th ed.; Prentice-Hall/Pearson: Upper Saddle River, NJ, USA, 2010; ISBN 978-0-13-100846-5. [Google Scholar]
- Kukula-Mlynarczyk, A.; Hurej, M. Incidence, Harmfulness and Some Elements of the Horse Chestnut Leafminer (Cameraria Ohridella Deschka & Dimic) Control on White Horse Chestnut (Aesculus Hippocastanum L. J. Plant Prot. Res. 2007, 47, 53–64. [Google Scholar]
- Hatcher, P.E. Three-Way Interactions between Plant Pathogenic Fungi, Herbivorous Insects and Their Host Plants. Biol. Rev. 1995, 70, 639–694. [Google Scholar] [CrossRef]
- Weyda, F.; Pflegerová, J.; Stašková, T.; Tomčala, A.; Prenerová, E.; Zemek, R.; Volter, L.; Kodrík, D. Ultrastructural and Biochemical Comparison of Summer Active and Summer Diapausing Pupae of the Horse Chestnut Leaf Miner, Cameraria Ohridella (Lepidoptera: Gracillariidae). Eur. J. Entomol. 2015, 112, 197–203. [Google Scholar] [CrossRef]
- Menkis, A.; Povilaitienė, A.; Marčiulynas, A.; Lynikienė, J.; Gedminas, A.; Marčiulynienė, D. Occurrence of Common Phyllosphere Fungi of Horse-Chestnut (Aesculus Hippocastanum) Is Unrelated to Degree of Damage by Leafminer (Cameraria Ohridella). Scand. J. For. Res. 2019, 34, 26–32. [Google Scholar] [CrossRef]
- Straw, N.A.; Williams, D.T. Impact of the Leaf Miner Cameraria Ohridella (Lepidoptera: Gracillariidae) and Bleeding Canker Disease on Horse-Chestnut: Direct Effects and Interaction. Agric. For. Entomol. 2013, 15, 321–333. [Google Scholar] [CrossRef]
- Percival, G.C.; Banks, J.M. Studies of the Interaction between Horse Chestnut Leaf Miner (Cameraria Ohridella) and Bacterial Bleeding Canker (Pseudomonas Syringae Pv. Aesculi). Urban For. Urban Green. 2014, 13, 403–409. [Google Scholar] [CrossRef]
- Koskella, B.; Meaden, S.; Crowther, W.J.; Leimu, R.; Metcalf, C.J.E. A Signature of Tree Health? Shifts in the Microbiome and the Ecological Drivers of Horse Chestnut Bleeding Canker Disease. New Phytol. 2017, 215, 737–746. [Google Scholar] [CrossRef]
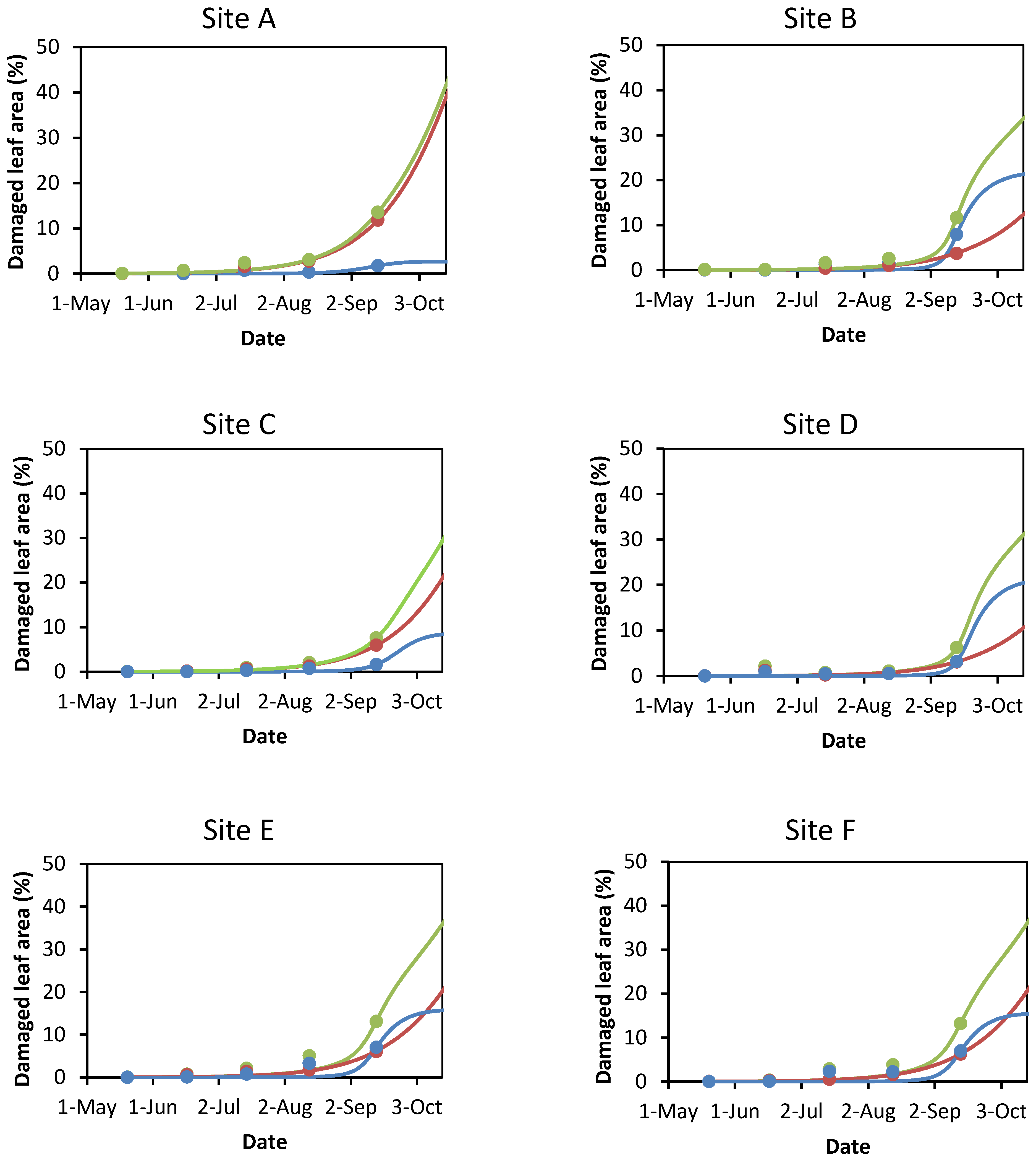
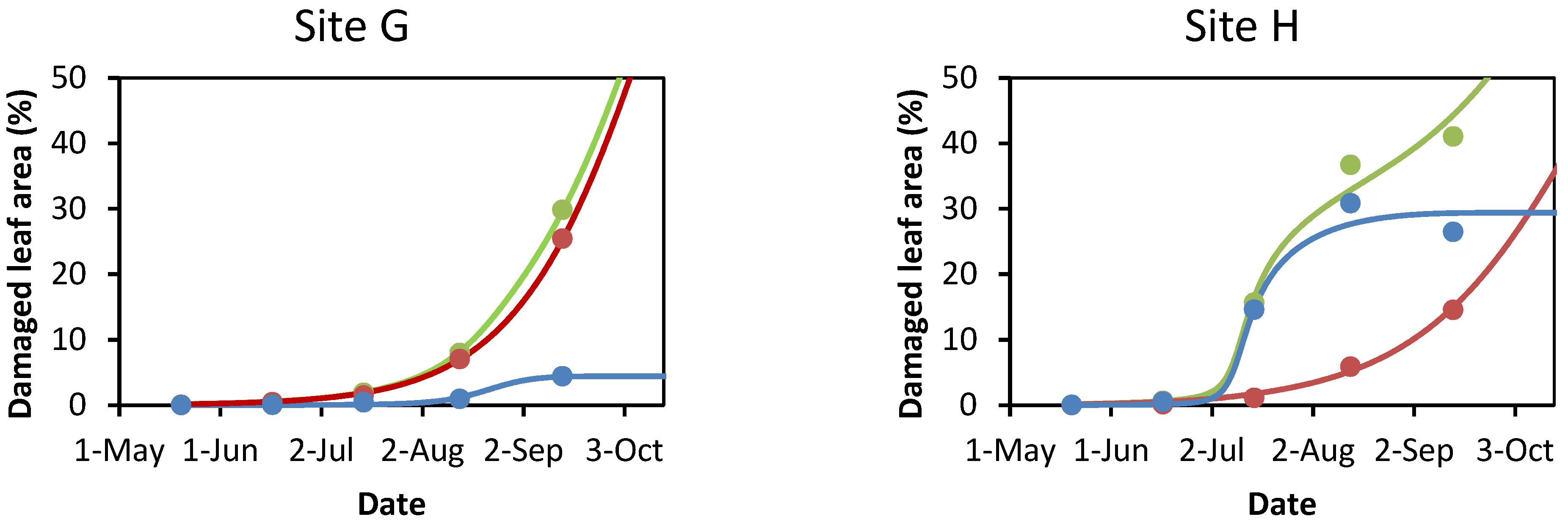
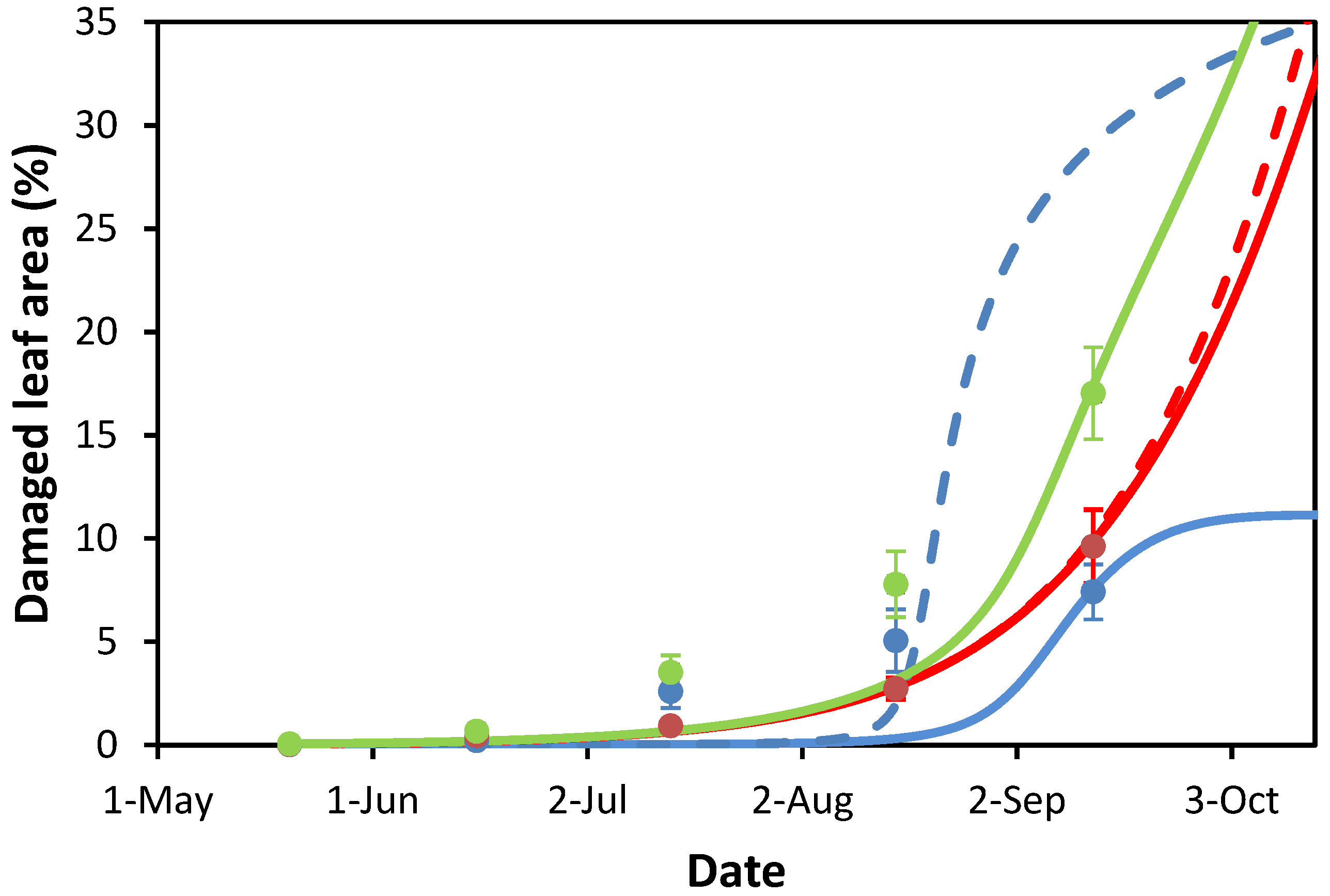
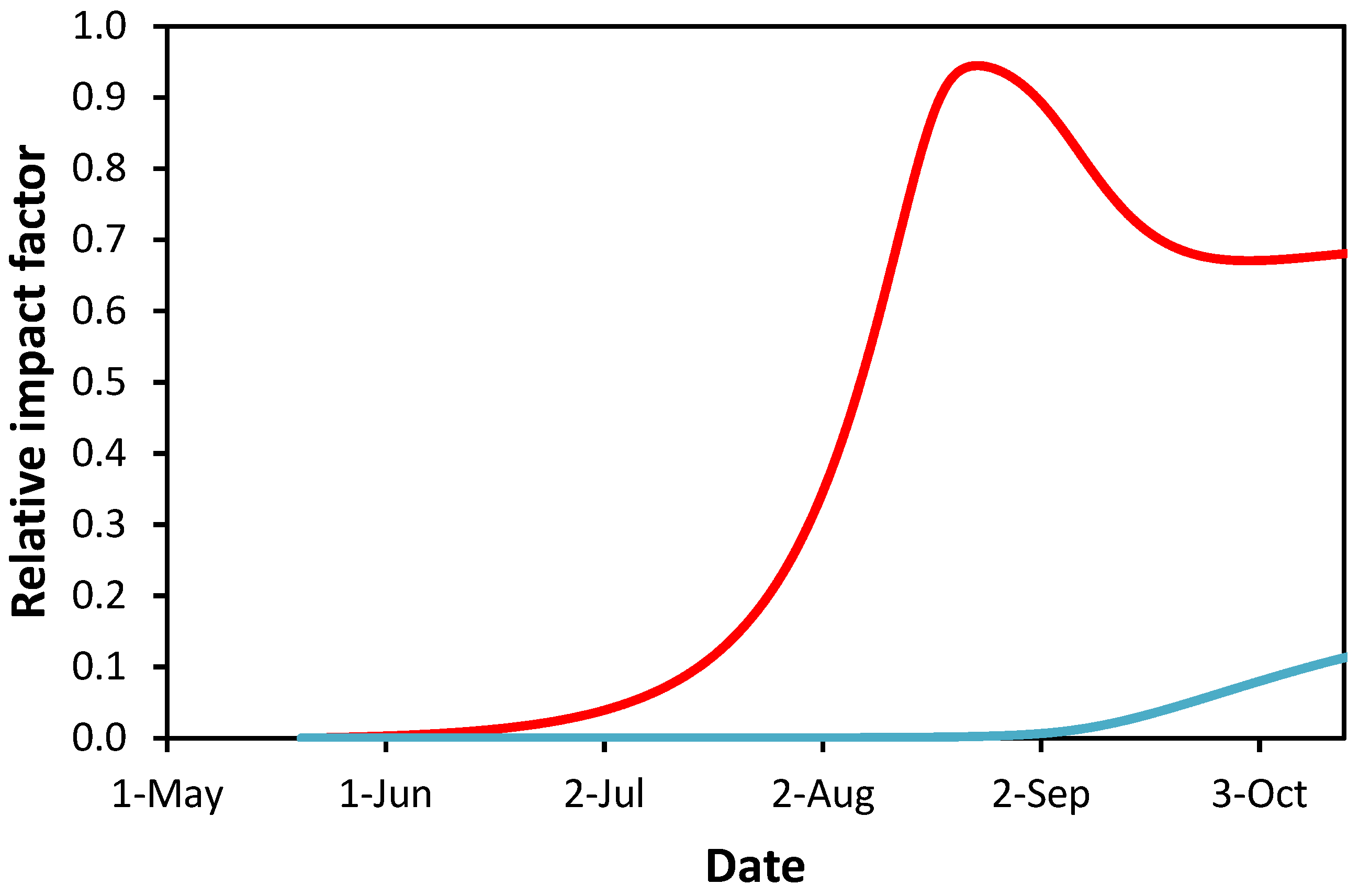
| Study Site | Cadastral Area | Green Public Open Space | Horse Chestnut Trees | Avg. Age ± SEM & of Horse Chestnut Tree | |
|---|---|---|---|---|---|
| Label | Name | (Hectares) | (Hectares) | (Number) | (Year) |
| A | City centre | 133 | 12 | 186 | 74.99 ± 1.86 |
| B | Šumava and Máj estate | 183 | 58 | 40 | 39.02 ± 3.19 |
| C | Vltava estate | 92 | 34 | 40 | 41.79 ± 3.49 |
| D | Třebotovice and Kaliště village | 30 | 2 | 5 | 17. 82 ± 6.86 |
| E | Rožnov estate | 294 | 29 | 57 | 70.49 ± 4.43 |
| F | Pražské předměstí estate | 124 | 37 | 49 | 49.74 ± 2.75 |
| G | Stromovka Park | 1 | 1 | 6 | 43.53 ± 4.13 |
| H | Nádražní Street | 1 | 1 | 18 | 77.74 ± 3.02 |
| Sampling Date | Leaf Area Damaged by Cameraria ohridella | Leaf Area Damaged by Guinardia aesculi | ||
|---|---|---|---|---|
| Avg. ± SEM | Range | Avg. ± SEM | Range | |
| from 16 to 24 May | 0.018 ± 0.002 | 0.000–0.329 | 0.031 ± 0.005 | 0.000–0.591 |
| from 13 to 21 June | 0.214 ± 0.117 | 0.000–28.071 | 0.448 ± 0.126 | 0.000–27.612 |
| from 11 to 19 July | 2.594 ± 0.402 | 0.003–37.582 | 0.924 ± 0.095 | 0.000–14.882 |
| from 11 to 22 August | 5.058 ± 0.756 | 0.000–78.790 | 2.726 ± 0.255 | 0.022–22.636 |
| from 9 to 19 September | 7.416 ± 0.666 | 0.006–60.686 | 9.612 ± 0.891 | 0.000–78.864 |
| Species | |||
|---|---|---|---|
| C. ohridella | G. aesculi | ||
| Parameters | α | 3.0066 | 0.0478 |
| β | 1.5023 | 1.0096 | |
| γ | 14.611 | 0.6732 | |
| Initial damage | Site A | 5.42 × 10−5 | 6.54 × 10−4 |
| (p(0)) | Site B | 4.35 × 10−5 | 2.11 × 10−4 |
| Site C | 4.30 × 10−5 | 3.25 × 10−4 | |
| Site D | 3.96 × 10−5 | 1.73 × 10−4 | |
| Site E | 4.82 × 10−5 | 3.44 × 10−4 | |
| Site F | 4.84 × 10−5 | 3.51 × 10−4 | |
| Site G | 8.96 × 10−5 | 1.538 × 10−3 | |
| Site H | 2.29 × 10−4 | 1.453 × 10−3 | |
| Periods | C. ohridella | G. aesculi | ||||
|---|---|---|---|---|---|---|
| r | F1,6 | p | r | F1,6 | p | |
| Period 1 vs. Period 5 | 0.3975 | 1.126 | 0.3295 | −0.1823 | 0.206 | 0.6657 |
| Period 2 vs. Period 5 | −0.2950 | 0.572 | 0.4781 | −0.1429 | 0.125 | 0.7358 |
| Period 3 vs. Period 5 | 0.9698 | 95.02 | <0.0001 | 0.6848 | 5.299 | 0.0609 |
| Period 4 vs. Period 5 | 0.9715 | 100.7 | <0.0001 | 0.9555 | 62.96 | 0.0002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kopačka, M.; Nachman, G.; Zemek, R. Seasonal Changes and the Interaction between the Horse Chestnut Leaf Miner Cameraria ohridella and Horse Chestnut Leaf Blotch Disease Caused by Guignardia aesculi. Forests 2021, 12, 952. https://doi.org/10.3390/f12070952
Kopačka M, Nachman G, Zemek R. Seasonal Changes and the Interaction between the Horse Chestnut Leaf Miner Cameraria ohridella and Horse Chestnut Leaf Blotch Disease Caused by Guignardia aesculi. Forests. 2021; 12(7):952. https://doi.org/10.3390/f12070952
Chicago/Turabian StyleKopačka, Michal, Gösta Nachman, and Rostislav Zemek. 2021. "Seasonal Changes and the Interaction between the Horse Chestnut Leaf Miner Cameraria ohridella and Horse Chestnut Leaf Blotch Disease Caused by Guignardia aesculi" Forests 12, no. 7: 952. https://doi.org/10.3390/f12070952
APA StyleKopačka, M., Nachman, G., & Zemek, R. (2021). Seasonal Changes and the Interaction between the Horse Chestnut Leaf Miner Cameraria ohridella and Horse Chestnut Leaf Blotch Disease Caused by Guignardia aesculi. Forests, 12(7), 952. https://doi.org/10.3390/f12070952







